1. Introduction
Since the 1990s, gastrointestinal (GI) imaging has undergone significant advances, becoming an integral part of routine clinical practice. Among second-level imaging modalities, Magnetic Resonance Imaging (MRI) stands out for its noninvasive, radiation-free evaluation of gastrointestinal disease, offering superior soft tissue contrast, multiplanar imaging, and functional assessment.
Once a supplementary technique, MRI is now a cornerstone in the diagnosis, staging, and monitoring of a wide range of gastrointestinal diseases, including inflammatory bowel disease (IBD), rectal cancer, and graft versus host disease (GVHD) [
1].
Recent years have also seen a growing interest among gastroenterologists in intestinal ultrasound (IUS), which has led to an increase in research and publications in this area.
MRI, however, remains the gold standard for most gastrointestinal diseases and, in particular, for imaging chronic inflammatory bowel disease.
Its panoramic visualization of the small and large intestine detailed rectal-anal assessment, high tissue contrast, and multiparametric capabilities-including T1-weighted (with/without gadolinium), T2-weighted, and diffusion-weighted imaging (DWI)-enable comprehensive assessment of structural and functional changes. In addition, MRI contrast agents have fewer contraindications and lower nephrotoxicity than CT iodinated agents.
Despite these strengths, MRI scanning has its limitations. Patient-related factors, such as claustrophobia, the need for collaboration during long scanning times and contraindications due to metal implants, may restrict its use, particularly among paediatric, geriatric or critically ill patients. Additionally, high costs and limited availability may reduce accessibility in some healthcare facilities, which could explain the growing interest in more affordable ultrasound scanning.
Italy has long been at the forefront of radiological innovation, with Italian radiologists making significant contributions to the advancement of MRI protocols, the standardization of gastrointestinal imaging and its integration into multidisciplinary care. This review provides a comprehensive overview of recent advances in gastrointestinal MRI, focusing on pioneering Italian research and clinical practice (
Figure 1).
Key areas explored include magnetic resonance enterography (MRE), high-resolution rectal imaging, functional pelvic floor studies, and MRI applications in neuroendocrine tumour evaluation. The review also explores emerging frontiers in gastrointestinal MRI, such as the assessment of intestinal motility, molecular imaging and artificial intelligence (AI). Although motility imaging is not yet standard in clinical protocols, it offers dynamic insights beyond static anatomy.
Meanwhile, AI and radiomics are set to transform gastrointestinal imaging by extracting quantitative biomarkers, improving diagnostic accuracy and supporting personalized treatment strategies.
Finally, this article aims to equip radiologists, gastroenterologists, and GI specialists with an up-to-date, evidence-based synthesis of the evolving role of MRI in gastrointestinal imaging, with a special emphasis on Italian contributions that continue to shape global practice.
2. Gastrointestinal Ultrasound (GIUS)
Gastrointestinal ultrasound (GIUS) is increasingly recognized as a pivotal, non-invasive imaging modality in the evaluation of both functional and inflammatory gastrointestinal disorders. The 2021 EFSUMB consensus statement titled “
Gastrointestinal Ultrasound in Functional Disorders of the Gastrointestinal Tract” highlights the expanding role of GIUS as a frontline diagnostic tool in patients with symptoms such as dysphagia, reflux, dyspepsia, bloating, abdominal pain, and altered bowel habits. Its real-time capabilities—including dynamic assessment of motility, peristalsis, luminal filling and emptying, perfusion, and blood flow—are achieved with high spatial and temporal resolution. Importantly, GIUS not only assists in excluding structural abnormalities but also offers valuable insight into functional pathophysiology, allowing for ongoing evaluation and monitoring of therapeutic response. The consensus provides robust clinical recommendations and emphasizes the integration of GIUS into routine workflows, thanks to its noninvasiveness, repeatability, and clinical versatility [
2]. Additionally, the 2018 EFSUMB guidelines, “
EFSUMB Recommendations and Clinical Guidelines for Intestinal Ultrasound (GIUS) in Inflammatory Bowel Diseases” reinforce the efficacy of GIUS in the management of chronic inflammatory conditions such as Crohn’s disease and ulcerative colitis. These guidelines, developed by a European expert task force involving both radiologists and gastroenterologists (including Italian ones) outlines standardized protocols for assessing bowel wall thickness, vascularity, strictures, fistulas, and abscesses—parameters essential for disease activity evaluation and therapeutic monitoring. With diagnostic performance often comparable to CT and MRI, GIUS offers a radiation-free alternative that enables frequent follow-up and timely clinical decisions [
3]. Together, these guidelines establish GIUS as a crucial imaging modality, enhancing diagnostic accuracy and patient care in both functional and inflammatory gastrointestinal disease contexts.
3. MR Enterography (MRE)
Magnetic Resonance Enterography (MRE) has become the imaging modality of choice for evaluating Crohn’s disease (CD) in both adults and children. It can effectively assess mural and transmural inflammation, as well as any complications, including strictures, fistulas and abscesses, at any intestinal location, without being invasive. Starting from the early papers published around 2000 [
4,
5,
6,
7,
8,
9,
10], it has been widely investigated and validated and has become the primary imaging modality for assessing Crohn's disease (CD) in adults and children [
4,
5,
6,
7,
8,
9,
10,
11,
12,
13].
3.1. Magnetic Resonance Enterography (MRE) Technique: Updated Recommendations
Standard Magnetic Resonance Enterography (MRE) protocols have become increasingly structured and refined, ensuring consistent, high-quality imaging across institutions, although new imaging sequences and technical improvements are continuously introduced and the technique is still evolving. The procedure requires bowel distension using an oral iso-osmotic contrast agent, such as polyethylene glycol (PEG)-based solutions. These contrast agents are well tolerated, promote uniform small bowel filling, and enable adequate visualization of the intestinal lumen and walls. Patients are instructed to ingest approximately 1.5 to 2 liters of the contrast solution over 30–45 minutes prior to the examination, often in divided doses to optimize tolerability and distension. To reduce bowel peristalsis and minimize motion artifacts during scanning, antiperistaltic agents—most commonly intramuscular glucagon or intravenous scopolamine butylbromide—are administered immediately before imaging. These agents improve image quality by suppressing motion and enhancing visualization of fine mural features such as ulceration or edema. The MRE technique has been recently reanalyzed and refined by Hameed et al. in practical recommendation papers offering stepwise protocols for optimal image acquisition in patients with Crohn’s disease [
13,
15]. The recommended imaging workflow begins with high-resolution T2-weighted sequences in axial and coronal planes to assess mural edema and stratification, followed by Gadolinium- enhanced T1-weighted imaging for hyperenhancement evaluation, and concludes with diffusion-weighted imaging (DWI) to detect active inflammation based on restricted diffusion. This comprehensive approach captures both anatomical and functional aspects of disease. The guide also emphasizes integrating validated scoring systems such as MaRIA and adopting structured reporting to ensure consistency and clarity in multidisciplinary discussions [
13,
15]. Cine-MRI, which captures real-time bowel motility, is increasingly recommended to aid in the evaluation of strictures and motility disorders that may be missed on static sequences [
4,
7].
Similarly, the ESGAR/ECCO Diagnostic consensus guidelines highlights MRE—alongside intestinal ultrasound (IUS) as a frontline imaging modality for luminal Crohn’s disease [
15,
16].
Mural thickening, gadolinium wall hyperenhancement, wall edema, ulceration, and motility changes are critical imaging markers of Crohn’s disease activity, and advocate for standardized reporting with treatment response categories such as transmural remission, partial response, or progression [
17,
18].
Based on these recommendations, a practical protocol should incorporate T2, post-contrast T1, DWI, and possibly cine-MRI sequences. Activity scores like simplified-MaRIA should be used routinely to assess inflammation. In stricturing disease, serial MRE—such as baseline and 12- month follow-up as shown in the STRIDENT trial—is essential for monitoring transmural healing [
19]. Structured reports should document activity, strictures, and motility, while correlating imaging findings with laboratory markers (e.g., CRP, fecal calprotectin) and complementary modalities like IUS for a fully integrated evaluation [
15,
16,
17,
18,
19,
20,
21].
A group of Italian radiologists coordinated by F. Maccioni, under the patronage of SIGENP (Italian Society of Pediatric Gastroenterology, Hepatology and Nutrition), is involved in developing new guidelines for pediatric MR enterography. Pediatric MRE in fact requires specific preparation and technical considerations, and the expertise of dedicated teams of radiologists and technicians.
3.2. MRE: Practical Recommendations3.3. MRI Assessment of Crohn’s Disease Activity
| Recommendation |
Details |
| Combine anatomical and functional sequences |
Use T2-weighted, post-contrast T1-weighted, diffusion-weighted imaging (DWI), and cine-MRI for comprehensive assessment of inflammation, complications, and motility. |
| Apply validated scoring methods |
Implement scoring systems such as MaRIA or Nancy consistently to quantify disease activity and monitor treatment response. |
| Intensify imaging frequency |
In patients with stricturing Crohn’s disease, perform MRE at baseline and again at 12 months to evaluate transmural healing (as recommended in the STRIDENT trial). |
| Adopt structured reporting |
Use ESR/ESGAR-endorsed templates to document stricture presence, bowel motility, and activity scores for multidisciplinary communication. |
| Integrate biomarkers |
Correlate MRE findings with laboratory markers (e.g., CRP, fecal calprotectin) and complementary imaging such as intestinal ultrasound. |
Magnetic Resonance Enterography (MRE) is now firmly established as the gold standard for assessing disease activity in Crohn’s disease (CD), particularly for small bowel involvement. It provides comprehensive evaluation of transmural inflammation and extraintestinal complications without ionizing radiation, making it well-suited for repeated assessments [
4,
5,
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17,
18,
19]. Hallmark imaging features of active CD include bowel wall thickening, mural hyperenhancement, edema on T2-weighted images, ulceration, restricted diffusion on DWI, and perienteric inflammatory changes such as mesenteric fat stranding and vascular engorgement [
4,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17,
18]. To quantify disease severity, several standardized scoring systems have been developed and validated, including the Magnetic Resonance Index of Activity (MaRIA), simplified MaRIA, Clermont score, Nancy score, and Crohn’s Disease MRI Index (CDMI) [
11,
17,
18]. These indices integrate morphological and contrast-enhanced features to provide reproducible segmental activity assessment. A 2025 systematic review by Dal Buono et al. confirmed that MRE demonstrates high diagnostic accuracy, with sensitivity and specificity exceeding 90% for active inflammation across multiple studies [
17].
Recent international guidance, such as the RSNA-endorsed “How I Do It” recommendations published in Radiology [
13], emphasizes a standardized MRE protocol combining T2-weighted, post-contrast T1-weighted, and diffusion-weighted imaging sequences, with optional cine-MRI for motility and stricture assessment [
15].
Table 2 illustrates the MRE protocols used in the Radiological Department of Umberto I, Sapienza University Hospital.
The STRIDENT trial (2025) further highlighted the clinical utility of MRE in monitoring transmural healing, reporting complete stricture resolution in 22% of patients treated with anti-TNF therapy [
19]. Structured reporting and scoring are increasingly integrated into multidisciplinary IBD management to enhance treatment planning and longitudinal monitoring.
The joint guidelines from the European Society of Gastrointestinal and Abdominal Radiology (ESGAR) and the European Crohn’s and Colitis Organisation (ECCO) provide comprehensive recommendations for the use of cross-sectional imaging in the diagnosis and management of Crohn’s disease. These state that Magnetic Resonance Enterography (MRE) and Intestinal Ultrasound (IUS) should be considered first-line imaging modalities for suspected luminal Crohn’s disease, while CT should be reserved for emergency cases or when MRI is not feasible [
15,
16]. Key imaging features of active disease include bowel wall thickening (>3 mm), T2 hyperintense mural edema, contrast enhancement, ulcerations, perienteric inflammation, and motility abnormalities. These findings should be documented using structured reports, along with disease phenotype (inflammatory, stricturing, penetrating) and treatment response categories (transmural remission, partial response, stable disease, progression) [
13,
14]. Italy has played a significant role in implementing and expanding these guidelines, especially through multicenter studies and collaborations between radiologists and gastroenterologists. A national survey conducted by an Italian group, leaded by Bonifacio C. et al, revealed high adherence to ESGAR guidelines in high-volume centers, although the consistent use of structured reporting and scoring systems is still evolving [
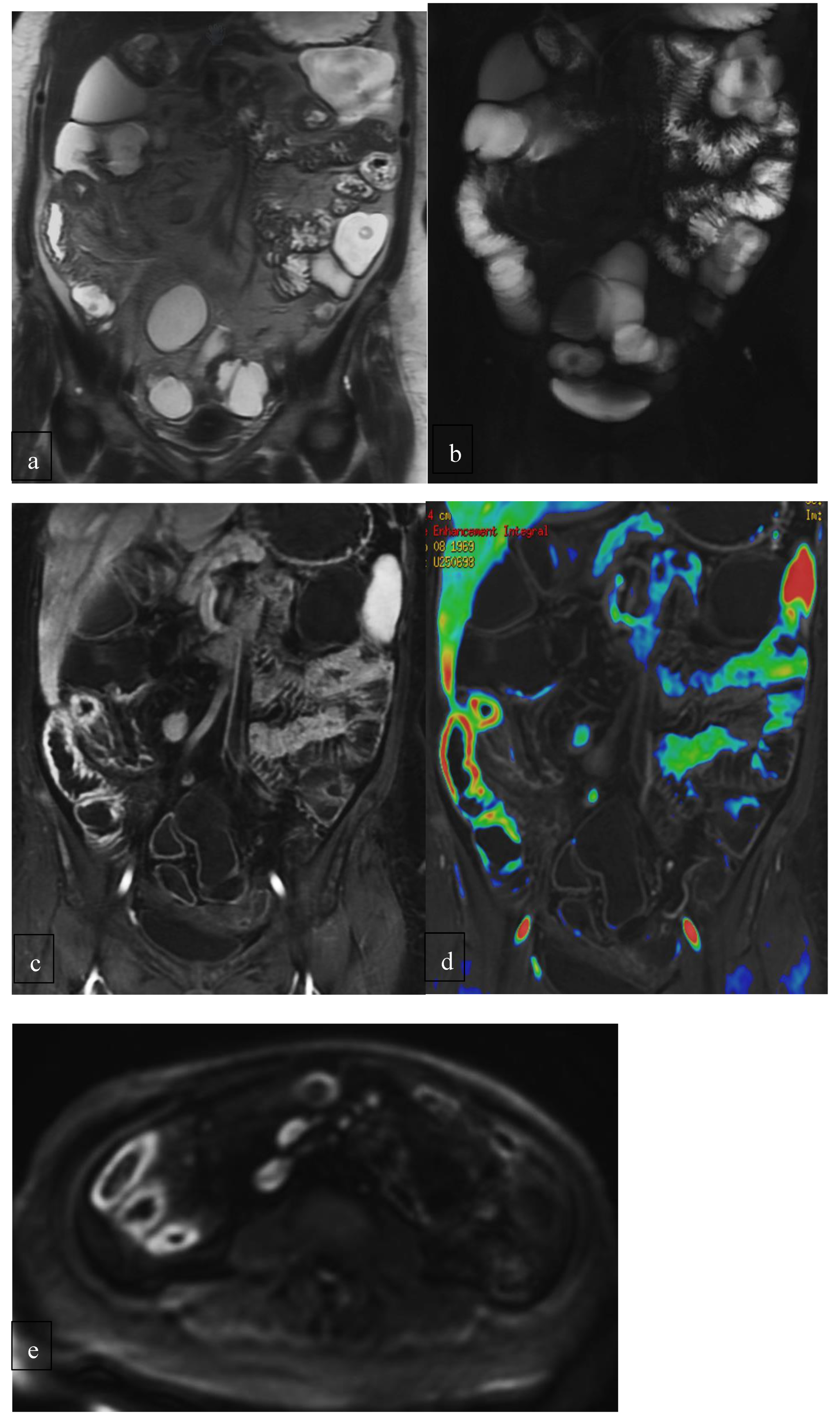
22]. The group led by Maccioni and colleagues at the Department of Radiology of Policlinico Umberto I, Sapienza University, employs innovative MRE (Magnetic Resonance Enterography) techniques. These include T2-weighted sequences, T2 with fat signal suppression, DWI, post-Gadolinium T1-weighted sequences, and fusion imaging. This advanced imaging approach enables not only accurate diagnosis but also effective follow-up of patients with Crohn's disease (
Figure 2).
In a multicenter study published in 2012, Maccioni’s group prospectively compared pediatric and adult patients with Crohn’s disease using MR enterography (MRE) across nine intestinal segments [
23]. Their findings revealed a distinct phenotype in children, who exhibited more extensive colonic and rectal involvement, whereas adults predominantly showed ileal disease. This work was instrumental in illustrating the diagnostic importance of segment-specific assessment in pediatric IBD. Further reinforcing the utility of MRE in children, a 2014 study led by Maccioni and published in
AJR demonstrated high diagnostic accuracy in a cohort of 50 pediatric patients, reporting an overall sensitivity of 94.5%, specificity of 97%, with interobserver agreement (κ = 0.93) [
24]. These results not only confirmed the robustness of MRE in clinical pediatric settings but also laid the groundwork for its broader adoption in centers across Italy and Europe. By 2019, the team had expanded their cohort to 350 patients, offering a more granular comparison of disease patterns in children versus adults [
25]. This larger study confirmed a significantly higher prevalence of rectal (29.7% vs 13.5%) and perianal disease (34.6% vs 16.1%) in the pediatric population, with corresponding odds ratios of 2.8 and 2.7, respectively—highlighting the clinical implications for early diagnosis and tailored treatment strategies [
25]. Complementary national efforts by Italian collaborative groups—such as the SIGENP Imaging Group—have focused on standardizing pediatric MRE protocols, reducing scan time, avoiding sedation when possible, and enhancing compliance through child-friendly techniques. Looking ahead, researchers are increasingly exploring the use of radiomics and artificial intelligence (AI) applied to pediatric MRE. Preliminary interesting studies on pediatric patients show promising results in predicting mucosal healing and treatment response based on quantitative image features and machine learning algorithms [
26,
27].
3.4. ESGAR Guidelines for MRI in Perianal Crohn’s Disease
The European Society of Gastrointestinal and Abdominal Radiology (ESGAR) has developed expert consensus guidelines on the optimal use of MRI in the evaluation of perianal Crohn’s disease (PFCD) and anal sepsis, published following a structured Delphi process involving 13 radiologists experienced in pelvic MRI, here including Italian radiologists [
28]. These guidelines emphasize that MRI is the gold standard imaging modality for PFCD, given its superior soft tissue contrast resolution and multiplanar capabilities. CT remains an alternative primarily for urgent septic presentations when MRI is unavailable. The recommended MRI acquisition protocol includes thin-slice (≤3 mm) high-resolution T2-weighted and T1-weighted sequences in at least two orthogonal planes (preferably axial and coronal to the anal canal), with optional fat suppression. Post-contrast T1-weighted sequences and diffusion-weighted imaging (DWI) are also encouraged to assess active inflammation, fistulas, and abscesses. The guidelines support classifying fistulas using the Parks classification and defining anatomical location of internal and external openings using clock-face notation or radial sectors. Notably, the size of any perianal abscess should be reported due to its prognostic and therapeutic implications—interobserver reliability for abscess size has shown excellent agreement (ICC ~0.81).
Structured reporting is a cornerstone of the ESGAR guidelines. Reports should include: the type of fistula, disease activity features (e.g., T2 hyperintensity, enhancement, diffusion restriction), presence and size of abscesses, and any secondary tracts. These reports align radiological interpretation with clinical decision-making, particularly surgical planning. ESGAR recommends delaying post-surgical MRI (e.g., after EUA) for at least 4 weeks to avoid misinterpretation due to post-procedural inflammation [
28]. In the pediatric setting, the same imaging protocols can generally be applied without major modifications.
Recently a new imaging-guided, anatomy-based classification of anorectal fistulas, the Garg classification has been proposed following a large multicentric retrospective Italian study [
29]. The new classification included 6 severity grades based on the characteristics of the primary tract: submucosal, inter-sphincteric, low trans-sphincteric, high trans-sphincteric, multiple, and supra-sphincteric/extra-sphincteric. Each grade was further subdivided into 3 subtypes (A, B, C) based on the extension of secondary tracts. Preliminary results demonstrated high accuracy of the new classification in defining disease severity [
29,
30].
4. High-Resolution Rectal MRI: Clinical Applications and the Italian Experience
High-resolution rectal MRI has become the gold standard for the local staging and restaging of rectal cancer, offering unmatched soft tissue contrast and spatial resolution. It allows precise evaluation of tumor invasion depth, involvement of the mesorectal fascia (circumferential resection margin, CRM), extramural vascular invasion (EMVI), and potential sphincter involvement—features that are crucial in selecting candidates for neoadjuvant chemoradiotherapy or sphincter-preserving surgery [
31,
32].
Post-treatment MRI also plays a central role in assessing therapeutic response through the magnetic resonance tumor regression grade (mrTRG), supporting “watch-and-wait” strategies in selected complete responders. Advances such as 3T MRI scanners and the comparison between diffusion-weighted imaging (DWI) and apparent diffusion coefficient (ADC) have improved diagnostic accuracy, allowing better visualization of residual tumor tissue and early identification of non-responders [
33,
34].
Italy has significantly contributed to protocol standardization and multidisciplinary integration in rectal cancer imaging. The COMRE group (REctal COMmittee) recently explored the joint role of MRI and endosonography in optimizing post-treatment pathways [
35]. At the Agostino Gemelli Hospital, Barbaro et al. investigated the role of PET-MRI in predicting metastatic risk based on morphologic tumor features in mucinous and non-mucinous cancers, influencing aggressive therapeutic planning [
36]. Furthermore, since 2020, several Italian teams have pioneered the integration of radiomics and AI into rectal MRI workflows. These approaches aim to extract high-dimensional imaging biomarkers for improved staging, therapy stratification, and prognosis prediction, supporting a move toward personalized oncology [
37,
38,
39,
40,
41]. In summary, high-resolution MRI of the rectum is a cornerstone of colorectal cancer management. The Italian experience has provided valuable advances in technique, structured interpretation, and radiomics application, helping to refine clinical decision-making and surgical planning.
5. Small Bowel Neoplasms
The diagnosis of intestinal tumors in adults continues to be a significant diagnostic challenge, primarily due to their nonspecific clinical presentation, anatomical location, and often subtle imaging findings. This is particularly true for tumors of the small bowel, which are rare, comprising less than 5% of all gastrointestinal neoplasms, but potentially aggressive if not identified early. As outlined in The Many Faces of Intestinal Tumors in Adults, Including the Primary Role of CT Imaging in Emergencies and the Important Role of Cross-Sectional Imaging: A Pictorial Review, accurate diagnosis frequently hinges on the effective use of advanced imaging modalities. The study by Brogna et al. [
42] further emphasizes the crucial role of integrated imaging strategies in tackling these diagnostically elusive tumors. Small bowel neoplasms are often discovered in acute clinical scenarios, such as intestinal obstruction or gastrointestinal hemorrhage, where symptoms prompt urgent imaging evaluation. In such cases, computed tomography (CT) is the primary modality due to its wide availability, speed, and ability to provide a comprehensive overview of both the intraluminal and extraluminal compartments. CT not only allows the identification of masses or wall thickening but also detects complications such as ischemia, perforation, or metastasis that may accompany malignant lesions.
However, as Brogna et al. and Masselli et al. point out, reliance solely on CT in elective or subacute settings may miss subtle or early-stage lesions, leading to delayed diagnosis. Masselli et al., in their article Magnetic Resonance of Small Bowel Tumors [
12], present MRI, particularly MR enterography and MR enteroclysis, as an invaluable adjunct or alternative to CT in the non-emergency evaluation of small bowel neoplasms. MRI offers superior soft-tissue contrast resolution and the ability to evaluate the bowel wall and surrounding structures in greater detail, without ionizing radiation, a key advantage in younger patients and those requiring serial imaging. Their work highlights how dynamic contrast-enhanced sequences, diffusion-weighted imaging, and functional assessment of bowel motility contribute to more accurate tumor characterization, differentiation between benign and malignant lesions, and assessment of disease extent. MRI is particularly useful for detecting submucosal tumors such as gastrointestinal stromal tumors (GISTs), carcinoids, and lymphoma, which may appear inconspicuous on CT.
Together, these findings support the increasing consensus that a multimodal, cross-sectional imaging approach, leveraging the strengths of both CT and MRI, is essential for optimizing diagnostic accuracy in small bowel tumors. Early and accurate imaging not only guides surgical or oncological management but also plays a critical role in avoiding unnecessary interventions and improving overall patient outcomes. Overall, both CT and MRI should be considered complementary rather than competing modalities, each with a distinct but overlapping role in the evolving imaging paradigm for small bowel neoplasms. The integration of these techniques into a tailored diagnostic workflow, depending on clinical presentation and patient factors, represents best practice in contemporary gastrointestinal radiology.
5.1. Pancreatic Neuroendocrine Neoplasms
Pancreatic neuroendocrine neoplasms (PanNENs), arising from diffuse neuroendocrine cells, represent the second most common group of pancreatic tumors and demonstrate significant biological heterogeneity [
43]. A multimodal imaging strategy—comprising ultrasound (US), contrast-enhanced CT (sensitivity 69–94%), MRI (sensitivity 79–100%), and functional imaging, is recommended for initial diagnosis, staging, surveillance, and treatment monitoring [
43,
44]. A pivotal Italian retrospective study by Farchione et al. (2016) compared morphological MRI sequences, diffusion-weighted imaging (DWI), and 68Ga-DOTANOC PET/CT, finding similar detection rates for primary and metastatic lesions around 90%, thus promoting MRI as a radiation-free surveillance alternative, especially in MEN-1 carriers [
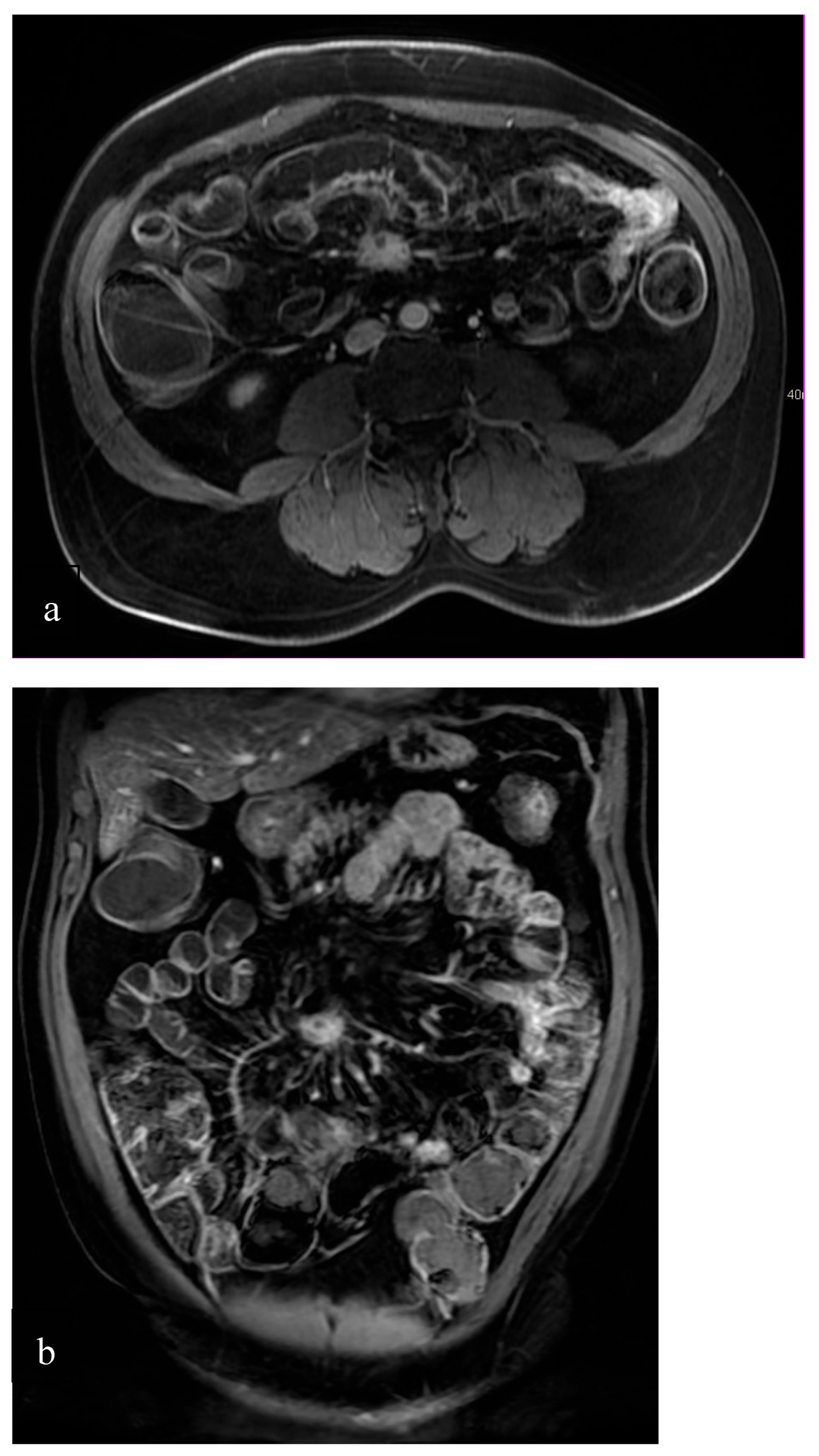
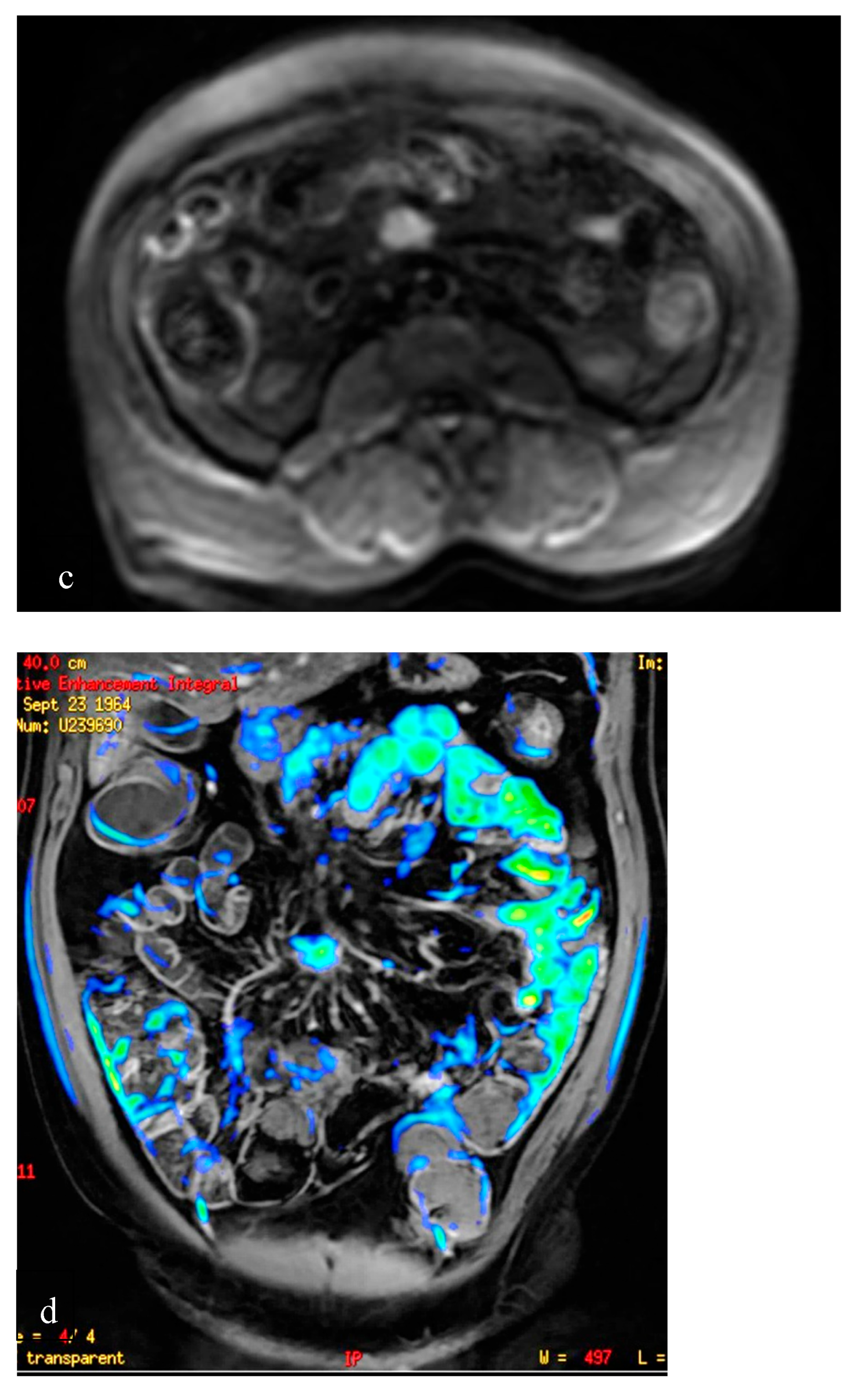
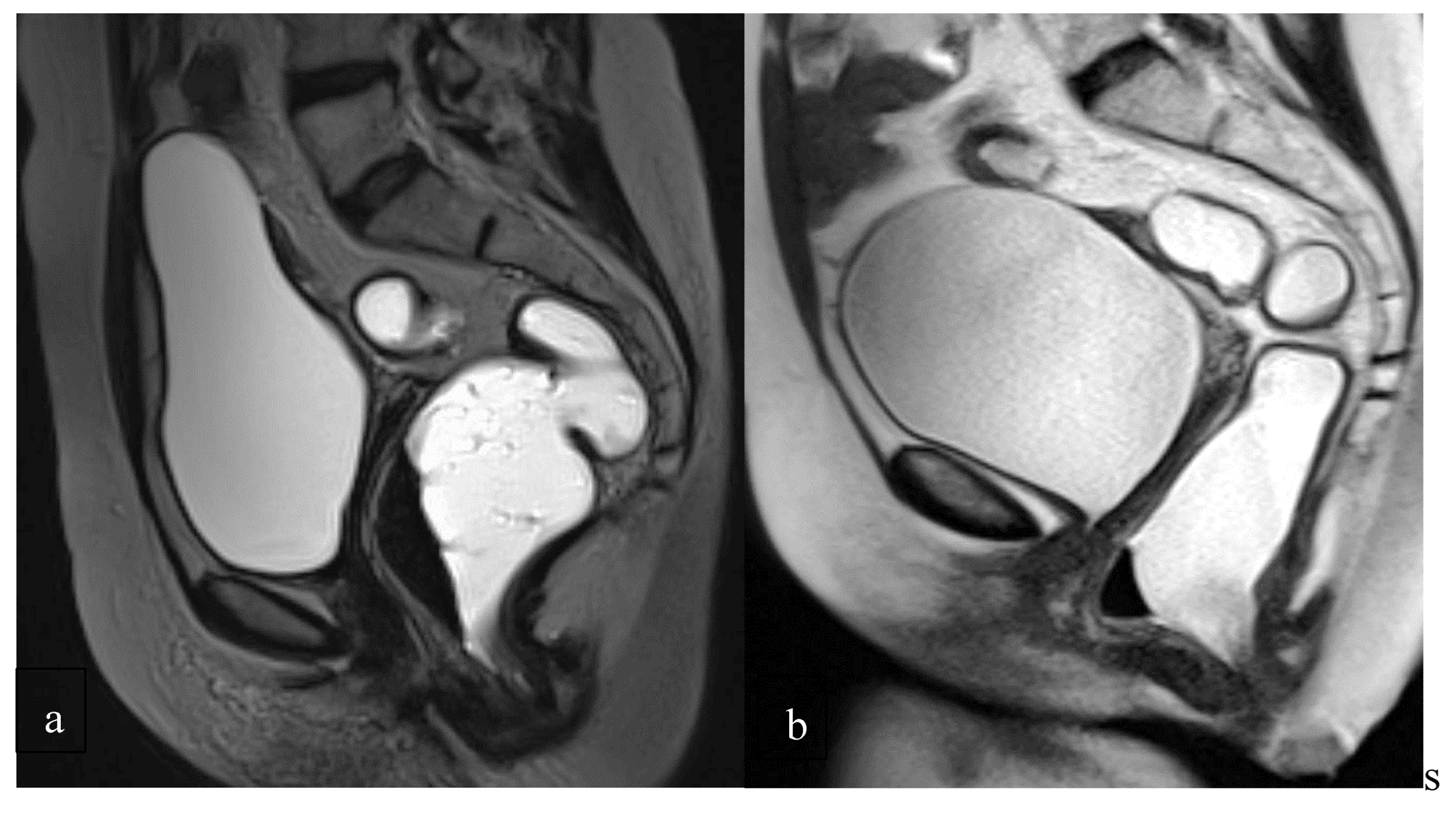
45]. A major contribution from Giannetta et al. focused on detecting and localizing insulinomas using a combined MRI and PET/CT multiparametric approach. Their work demonstrated that combining standard morphological MRI sequences (T2W-TSE ± fat suppression; T1 in- and out-of-phase; dynamic 3D T1 GRE with arterial, portal, and late phases) with DWI (b-values up to 1000 s/mm²) can identify sub-centimeter lesions even when PET is inconclusive (
Figure 3). These results support MRI’s role in both diagnosis and pre-surgical planning, particularly in small, functioning PanNENs. On MRI, PanNENs are typically hypointense on T1 and hyperintense on T2, with early arterial enhancement and slower washout—differentiating them from adenocarcinomas. DWI adds sensitivity for small and non-contrast-enhancing lesions. Hepatic metastases are well-demonstrated using hepatobiliary contrast agents (e.g., Gd-BOPTA), with DWI and high-resolution T1 detecting lesions ≥5 mm reliably [
43,
44,
45]. Functioning PanNENs exhibit characteristic MRI features: gastrinomas display T2 hyperintensity and ring-like enhancement in the arterial phase, whereas insulinomas show bright T2 signal due to their rich vascular content. Non-functioning tumors often have heterogeneous enhancement and restricted diffusion proportional to their Ki-67 proliferation index. Finally, recent Italian studies have advanced the application of radiomics and machine learning in PanNEN imaging [
45].
6. Peritoneal Carcinosis
Imaging of Peritoneal Carcinomatosis: Clinical Insights and Italian Contributions Peritoneal carcinomatosis (PC) represents a critical diagnostic and prognostic challenge in patients with advanced abdominal malignancies, including gastrointestinal and gynecological cancers. The group led by Reginelli A., Giacobbe G., and Del Canto MT in their review, “Peritoneal Carcinosis: What the Radiologist Needs to Know”, emphasized the central role of imaging—ultrasound (US), computed tomography (CT), and magnetic resonance imaging (MRI)—in the detection, staging, and multidisciplinary management of PC [
46]. Each modality offers unique advantages: US is often the first-line tool, especially for identifying ascites and peritoneal nodules as small as 2–3 mm, while CT is considered the current gold standard due to its wide availability, rapid acquisition, and high spatial resolution. It is particularly effective in mapping larger implants and assessing disease burden across the peritoneal cavity, especially when combined with delayed contrast-enhanced phases and proper bowel preparation. However, its sensitivity decreases in small bowel mesentery and subphrenic areas [
33].
MRI complements CT with superior soft-tissue contrast and sensitivity for smaller lesions, particularly when incorporating diffusion-weighted imaging (DWI) and delayed gadolinium-enhanced sequences. The work of Miceli et al. (2023) in Cancers further advances this perspective, presenting a detailed overview of the role of cross-sectional imaging and radiomics in predicting resectability in advanced ovarian cancer with peritoneal involvement. They underscore that MRI, especially when optimized with DWI and late-phase post-contrast imaging, can detect sub-centimeter implants that may be missed by CT, particularly in critical surgical regions such as the diaphragmatic peritoneum or small bowel serosa [
47,
48].
A key innovation highlighted by Miceli’s group is the integration of radiomics, the extraction of high-dimensional quantitative features from CT and MRI data, which provides a noninvasive method for tumor characterization and prediction of surgical outcomes. Their study illustrates how radiomic models, when combined with clinical and laboratory data (e.g., CA-125), can predict the likelihood of achieving complete cytoreduction—thereby helping avoid unnecessary laparotomy in non-resectable cases. These models represent a significant advance in personalized medicine and align with the evolving role of imaging as a decision-making tool rather than a mere diagnostic adjunct. The Italian experience has been pivotal in this field. These efforts not only improve lesion detectability but also facilitate multidisciplinary discussions with gynecologic, oncologists and surgeons [
49].
In conclusion, the combined use of US, CT, MRI, and radiomics offers a comprehensive strategy for managing peritoneal carcinomatosis. The integration of advanced imaging with predictive modeling, as seen in the work of Miceli et al., represents a paradigm shift in oncologic imaging—transforming radiology into a central pillar of precision oncology and surgical planning [
47,
48].
7. Imaging Gastrointestinal Graft-versus-Host Disease (GI-GVHD)
GI-GVHD, a common complication after allogeneic hematopoietic stem cell transplantation, typically presents nonspecific gastrointestinal symptoms. Although endoscopic biopsy remains the diagnostic gold standard, imaging is invaluable, particularly in unstable or thrombocytopenic patients.
Historically, contrast-enhanced CT (CECT) has been used to identify bowel wall thickening, mucosal hyperenhancement, layering, the “comb sign,” and ascites—features corroborated by early studies like those of Kalantari et al [
50]. Maccioni et al recently proposed a multiparametric MRI scoring system for acute intestinal GVHD, incorporating six significant imaging feature: bowel wall stratification (on both T2 and post-contrast T1 sequences), ascites, retroperitoneal edema, and declivous soft-tissue changes. This diagnostic score achieved 84.6% sensitivity and 100% specificity (AUC = 0.962), while a broader severity score of 15 signs predicted 1-month mortality with 100% sensitivity and 90% specificity. MRI findings consistently indicated the ileum as the most frequently involved segment (~85%), revealing that MRI detected a higher disease burden than clinical staging alone [
51]. Earlier MRI assessments reported wall thickening and stratification, but without scoring or prognostic insight, and displayed lower diagnostic accuracy (sensitivity 81%, specificity 36%) [
52,
53]. Brogna’s team recently refined the CT features of GI-GVHD [
54]. In summary, combined CT and multiparametric MRI offers a powerful, complementary diagnostic framework. The Italian-led MRI scoring system with its high accuracy and prognostic value, paves the way for less invasive, repeatable imaging-based tools in GI-GVHD management.
8. Functional Imaging of the Pelvic Floor with MR Defecography: Italian Contributions and ECCO–ESGAR Guidelines
Magnetic Resonance Defecography (MRD) is now widely regarded as the reference standard for the non-invasive, dynamic assessment of pelvic floor disorders. It offers superior soft tissue contrast and enables real-time visualization of all three pelvic compartments—anterior, middle, and posterior—during rest, squeeze, straining, and evacuation. These capabilities make MRD essential for diagnosing a wide range of conditions, including obstructed defecation syndrome, pelvic organ prolapse, rectocele, enterocele, intussusception, and functional disorders such as anismus. Italian researchers have been instrumental in the development and clinical standardization of MRD from 2013 on [
55].
The 2013 ECCO–ESGAR joint consensus guidelines formally endorse MRD in the evaluation of patients with Crohn’s disease presenting with obstructive or complex perianal symptoms, particularly when clinical examination or conventional imaging proves inconclusive [
56]. These guidelines highlight the value of MRD in identifying both structural and functional pelvic floor abnormalities, thus supporting its role in guiding surgical planning and conservative management strategies. Bitti et al. contributed significantly by publishing comprehensive imaging protocols incorporating high-resolution T2-weighted sequences in axial, sagittal, and coronal planes, followed by dynamic cine imaging during rectal evacuation using 120–250 mL of ultrasound gel. The introduction of anatomical reference lines—such as the pubococcygeal, H, and M lines—allows for reproducible quantification of pelvic descent and compartmental dysfunction [
57]. In 2016 technical issues of rectal distension have been extensively discussed by Maccioni et al., comparing two different MR defecography techniques for evaluating pelvic floor disorders: air-balloon versus gel for rectal filling [
58] In 2018 Maccioni and Alt’s publication laid the foundation for standardized pelvic floor MRI and MRD protocols, detailing optimal patient positioning, imaging sequences, and key diagnostic parameters. Maccioni’s group highlighted the critical elements to include in MR defecography reports, distinguishing between static anatomical data and dynamic functional findings. Key static features include sacrococcygeal morphology, the configuration of endopelvic and paravaginal fasciae, the levator ani muscle components, and relevant neural pathways. Dynamic parameters include rectal capacity, continence, vesicourethral angles, the anorectal angle, and detailed descriptions of pelvic compartment behavior during evacuation (
Figure 4). Her work set clinical and educational benchmarks that have shaped subsequent practice [
59]. Similarly, in 2024, Piloni, Manisco, and Fogante proposed a structured reporting template in the Journal of Imaging, aimed at standardizing MRD reporting for evacuation disorders. This approach seeks to minimize missing critical data, thus improving communication between radiologists and surgeons [
60]. They conducted one of the most extensive multicenter evaluations of MRD in patients with obstructed defecation syndrome, both before and after surgery, demonstrating the technique's accuracy in assessing anorectal angles, rectoceles, pelvic descent, and puborectalis muscle function. Recent publications have also emphasized the need for structured radiological reporting to maximize MRD’s clinical utility [
60]. Further advancements by Iacobellis et al. validated MRD’s diagnostic accuracy by correlating imaging results with clinical findings, reinforcing its importance in preoperative planning. Reginelli’s group explored innovative directions, including 3D pelvic modeling and the integration of artificial intelligence, aiming to enhance diagnostic reproducibility and surgical precision [
61].
Supporting these developments, a study by Colarieti et al. involving 218 patients demonstrated that MRD allowed radiologists to reach a diagnostic conclusion in 97% of cases, with a 92% concordance rate between radiologic findings and initial clinical suspicion. Surgeons rated the diagnostic quality of the radiology reports as excellent in 47%, good in 23%, sufficient in 16%, and insufficient in only 14% of cases. MRD findings were concordant and added clinically relevant information in 86.3% of cases, reinforcing its value in surgical decision-making. Ultimately, MR defecography not only confirms clinical suspicion but frequently provides additional insights that enhance treatment planning and patient outcomes [
62].
9. Motility Studies of the Small Bowel
Magnetic Resonance Imaging (MRI) of intestinal motility, also known as cine-MRI, is a non-invasive method that captures the dynamic peristaltic activity of the small bowel using time-resolved sequences like balanced steady-state free precession (bSSFP). Seminal work by de Jonge et al. showed that acquisition at ≥1 frame per second over at least a 15-second breath-hold provides stable and reproducible motility indices [
63]. Quantitative analysis using optical-flow or non-rigid registration algorithms allows computation of motility scores, which correlate negatively with inflammation in Crohn’s disease—evidenced in terminal ileal motility studies demonstrating lower motility indices in inflamed segments (e.g., 0.19 vs. 0.37 in healthy bowel; AUC ≈0.97) [
64]. Advanced 3D cine-MRI techniques (e.g. centerline-aware motion estimation) now enable volumetric differentiation between peristalsis and luminal motion, achieving AUC ≈0.97 for distinguishing motile from non-motile segments [
65]. Alex Menys and colleagues at UCL and Amsterdam Medical Center have been pioneers in this field, with publications on global small bowel motility, quantitative motility mapping in Crohn’s disease, and the relationship between motility variance and clinical symptoms [
66]. Together, these advances position cine-MRI as a precise, radiation-free modality for assessing small bowel function in disorders like Crohn’s disease, chronic pseudo-obstruction, and IBS.
10. Artificial Intelligence in Imaging of Intestinal Pathology and Tumors
Artificial Intelligence (AI) is a multidisciplinary field, whose goal is to provide an effective tool that is able to perform tasks, solve problems, and provide reliable results that facilitate the work in daily clinical practice, in which subsets of machine learning (ML), deep learning (DL), and radiomics, for example, are recognized; through these processes it is possible to perform tasks such as performing image classification, segmentation, and lesion detection on radiological images [
67]. The research group of Leonardo Da Rio, Marco Spadaccini, Tommaso Lorenzo Parigi et al. emphasized the relevant role that AI could have in the management of patients with IBD, which is complex and costly, optimizing clinical efficiency and healthcare resources [
68]. The potential applications of artificial intelligence range from diagnosis to treatment and follow-up. Given the essential role of imaging in gastroenterology, and particularly in IBD, AI-based image analysis can be used in numerous applications, such as evaluation of intestinal lesions, early detection of their evolution into cancer, and assessment of disease activity. This group emphasizes how AI can be useful in the interpretation of radiological images in patients with Crohn's disease for example, obtained by enterography-CT and in the characterization of intestinal fibrosis, integrating and correlating data even within large databases, assessing disease activity and reducing interobserver variability. The research group of Anna Lucia Cannarozzi, Luca Massimino, Anna Latiano et al. emphasizes how automated image analysis using AI has made it possible to detect malignant lesions earlier and to predict the evolution of some cancers. Moreover, thanks to international data-sharing collaborations, AI tools are expected to be trained on full images and big data in the coming years, so as to reduce bias in the field due to the heterogeneity of IBD patients [
67]. Fibrosthenic Crohn Disease is an area of unmet therapeutic need: the detection and characterization of stenosis is a well an area well suited for AI models. Good concordance has been demonstrated between (semi)automated measurements and an experienced radiologist's assessment of parameters such as bowel wall thickness, prestenotic dilatation, and minimal luminal diameter. In particular, AI was able to quantify the intestinal fibrosis by automated assessment, which was not inferior to expert radiological assessment and significantly faster. AI has not yet been evaluated for radiologic assessment radiologic evaluation of perianal fistulized CD, which promises to be quite challenging given the complex morphology and heterogeneity of this disease phenotype [
69]. So through the use of AI one could go so far as to identify pathologic thickening of the intestinal wall on, for example, images obtained from intestinal ultrasound, but this requires the use of cine images and not just still images, and concordance would have to be demonstrated on larger samples. The unpredictability of the course of IBD is one of the most challenging aspects of disease management, but using ML algorithms trained on large datasets such as medical records, laboratory results, imaging studies, and lifestyle factors could identify patterns and predictors of disease progression, thereby generating risk profiles for individual patients, estimating the likelihood of a flare-up, complication, or need for surgery, thereby also reducing health care expenditures.
Conclusions
Italian Radiologists have contributed significantly to the advancement and clinical integration of GI-MRI, for diagnostic, functional and prognostic applications, also offering a post-graduate Master Degree focused on Gastrointestinal Imaging (since 2021,
https://www.uniroma1.it/it/offerta-formativa/master/2025/imaging-gastrointestinale ). Italian research groups have played a key role in validating the use of MRE in Crohn’s Disease, also developing pediatric-specific protocols, in cooperation with SIGENP society (Società Italiana di Gastroenterologia Epatologia e Nutrizione Pediatrica). Furthermore, Italian centers made significant contributions to the staging and restaging of rectal cancer—particularly in assessing circumferential resection margins (CRM), extramural vascular invasion (EMVI), and sphincter involvement. Italian teams have also played a central role in standardizing post-treatment assessment protocols such as the magnetic resonance tumor regression grade (mrTRG) and collaborated closely with surgical groups to implement MRI in "watch-and-wait" strategies.
In high-resolution rectal MRI, Italian radiologists have likewise been a pioneers in functional pelvic floor imaging, especially through the development of dynamic MR defecography protocols and guidelines. Italian investigators have performed foundational work comparing supine and seated imaging using open MRI systems, correlating findings with manometry and clinical outcomes in obstructed defecation syndrome and pelvic organ prolapse. In the realm small intestine tumours and neuroendocrine tumors (NETs), Italian radiologists working within ENETS-accredited centers have helped integrate contrast-enhanced and DWI MRI into hybrid imaging algorithms, enhancing tumor detection, characterization, and staging. Lastly, Italy has emerged as a hub for innovation in artificial intelligence (AI) and radiomics applied to gastrointestinal MRI, with academic institutions in Milan, Rome, and Bologna leading the development of automated segmentation tools, radiomic biomarkers for treatment response, and AI-assisted interpretation in IBD and rectal cancer.
Despite the ongoing challenges of standardization and validation, these contributions have established Italian radiology as a leader in precision gastrointestinal imaging, with a strong interdisciplinary approach, combining radiology, gastroenterology and surgical oncology.
All these important clinical contributions of Italian radiology in the field of gastrointestinal imaging reveal the importance of close clinical-radiological collaboration, which in recent years has proven to be crucial in achieving significant advances in the study of the gastrointestinal tract.
References
- Maccioni, F.; Busato, L.; Valenti, A.; Cardaccio, S.; Longhi, A.; Catalano, C. Magnetic Resonance Imaging of the Gastrointestinal Tract: Current Role, Recent Advancements and Future Prospectives. Diagnostics 2023, 13, 2410. [Google Scholar] [CrossRef] [PubMed]
- Maconi, G.; Hausken, T.; Dietrich, C.F.; Pallotta, N.; Sporea, I.; Nurnberg, D.; Dirks, K.; Romanini, L.; Serra, C.; Braden, B.; Sparchez, Z.; Gilja, O.H. Gastrointestinal Ultrasound in Functional Disorders of the Gastrointestinal Tract - EFSUMB Consensus Statement. Ultrasound Int Open 2021, 7, E14–E24. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maconi, G.; Nylund, K.; Ripolles, T.; Calabrese, E.; Dirks, K.; Dietrich, C.F.; Hollerweger, A.; Sporea, I.; Saftoiu, A.; Maaser, C.; Hausken, T.; Higginson, A.P.; Nürnberg, D.; Pallotta, N.; Romanini, L.; Serra, C.; Gilja, O.H. EFSUMB Recommendations and Clinical Guidelines for Intestinal Ultrasound (GIUS) in Inflammatory Bowel Diseases. Ultraschall Med 2018, 39, 304–317. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F.; Viscido, A.; Broglia, L.; Marrollo, M.; Masciangelo, R.; Caprilli, R.; et al. Evaluation of Crohn disease activity with magnetic resonance imaging. Abdom Imaging 2000, 25, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Koh, D.M.; Miao, Y.; Chinn, R.J.; et al. MR imaging evaluation of the activity of Crohn’s disease. American Journal of Roentgenology 2001, 177, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Low, R.N.; Sebrechts, C.P.; Politoske, D.A.; et al. Crohn disease with endoscopic correlation: single-shot fast spin echo and gadolinium enhanced fat suppressed spoiled gradient echo MR imaging. Radiology 2002, 222, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Florie, J.; Waser, M.N.; Arts Cieslik, K. , et al. Dynamic contrast enhanced MRI of the bowel wall for assessment of disease activity in Crohn’s disease. AJR 2006, 186, 1384–1392. [Google Scholar] [CrossRef] [PubMed]
- Laghi, A.; Borrelli, O.; Paolantonio, P.; Dito, L.; Buena de Mesquita, M.; Falconieri, P.; Passariello, R.; Cucchiara, S. Contrast enhanced magnetic resonance imaging of the terminal ileum in children with Crohn’s disease. Gut 2003, 52, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Fidler, J.L.; Fletcher, J.G.; Bruining, D.H.; et al. Crohn’s disease: correlation of findings at MR enterography with clinical disease activity and endoscopic findings. Radiology 2006, 239, 367–375. [Google Scholar]
- Maccioni, F.; Bruni, A.; Viscido, A.; Colaiacomo, M.C.; Cocco, A.; Montesani, C.; et al. MR Imaging in Patients with Crohn Disease: Value of T2- versus T1-weighted Gadolinium-enhanced MR Sequences with Use of an Oral Superparamagnetic Contrast Agent. Radiology 2006, 238, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Rimola, J.; Rodriguez, S.; García-Bosch, O.; Ordás, I.; Ayala, E.; Aceituno, M.; et al. Magnetic resonance for assessment of disease activity and severity in ileocolonic Crohn’s disease. Gut 2009, 58, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Masselli, G.; Gualdi, G. MR imaging of the small bowel. Radiology 2012, 264, 333–348. [Google Scholar] [CrossRef] [PubMed]
- Hameed, M.; Kumar, S.; Taylor, S.A. How I Do It: Cross-sectional Imaging in Small-Bowel Crohn Disease and Ulcerative Colitis. Radiology 2025, 314, e241452. [Google Scholar] [CrossRef] [PubMed]
- Hameed, M.; De Kock, I.; Stoker, J.; Taylor, S.A. ESR Essentials: diagnosis and assessment of treatment response in patients with luminal Crohn’s disease-practice recommendations by the European Society of Gastrointestinal and Abdominal Radiology. Eur Radiol 2025. [Google Scholar] [CrossRef] [PubMed]
- Panes, J.; Bouhnik, Y.; Reinisch, W.; Stoker, J.; Taylor, S.A.; Baumgart, D.C.; et al. Imaging techniques for assessment of inflammatory bowel disease: joint ECCO and ESGAR evidence-based consensus guidelines. J Crohns Colitis 2013, 7, 556–585. [Google Scholar] [CrossRef] [PubMed]
- Maaser, C.; Sturm, A.; Vavricka, S.R.; Kucharzik, T.; Fiorino, G.; Annese, V.; Calabrese, E.; Baumgart, D.C.; Bettenworth, D.; Borralho Nunes, P.; Burisch, J.; Castiglione, F.; Eliakim, R.; Ellul, P.; González-Lama, Y.; Gordon, H.; Halligan, S.; Katsanos, K.; Kopylov, U.; Kotze, P.G.; Krustinš, E.; Laghi, A.; Limdi, J.K.; Rieder, F.; Rimola, J.; Taylor, S.A.; Tolan, D.; van Rheenen, P.; Verstockt, B.; Stoker, J.; European Crohn’s and Colitis Organisation [ECCO] and the European Society of Gastrointestinal and Abdominal Radiology [ESGAR]. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis 2019, 13, 144–164. [Google Scholar] [CrossRef] [PubMed]
- Dal Buono, A.; Faita, F.; Armuzzi, A.; Jairath, V.; Peyrin-Biroulet, L.; Danese, S.; et al. Assessment of activity and severity of inflammatory bowel disease in cross-sectional imaging techniques: a systematic review. J Crohns Colitis 2025, 19, jjaf023. [Google Scholar] [CrossRef] [PubMed]
- Seo, N. Comprehensive Review of Magnetic Resonance Enterography-Based Activity Scoring Systems for Crohn’s Disease. Investig Magn Reson Imaging 2025, 29, 1. [Google Scholar] [CrossRef]
- Lovett, G.C.; Schulberg, J.D.; Hamilton, A.L.; Wright, E.K.; Sutherland, T.R.; Ross, A.L.; et al. Crohn’s Disease Stricture Response to Treatment Assessed with Magnetic Resonance Imaging and Intestinal Ultrasound: STRIDENT Randomized Trial. Inflamm Bowel Dis 2025, izaf073. [Google Scholar] [CrossRef] [PubMed]
- Panés, J.; Bouzas, R.; Chaparro, M.; García-Sánchez, V.; Gisbert, J.P.; Martínez de Guereñu, B.; et al. Systematic review: the use of ultrasonography, computed tomography and magnetic resonance imaging for the diagnosis, assessment of activity and abdominal complications of Crohn’s disease. Aliment Pharmacol Ther 2011, 34, 125–145. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.A.; Mallett, S.; Bhatnagar, G.; Baldwin-Cleland, R.; Bloom, S.; Gupta, A.; et al. Diagnostic accuracy of magnetic resonance enterography and small bowel ultrasound for the extent and activity of newly diagnosed and relapsed Crohn’s disease (METRIC): a multicentre trial. Lancet Gastroenterol Hepatol 2018, 3, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Bonifacio, C.; Dal Buono, A.; Levi, R.; Gabbiadini, R.; Reca, C.; Bezzio, C.; et al. Reporting of Magnetic Resonance Enterography in Inflammatory Bowel Disease: Results of an Italian Survey. J. Clin. Med 2024. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F.; Viola, F.; Carrozzo, F.; Di Nardo, G.; Pino, A.R.; Staltari, I.; et al. Differences in the location and activity of intestinal Crohn’s disease lesions between adult and paediatric patients detected with MRI. Eur Radiol 2012, 22, 2465–2477. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F.; Al Ansari, N.; Mazzamurro, F.; Civitelli, F.; Viola, F.; Cucchiara, S.; et al. Detection of Crohn disease lesions of the small and large bowel in pediatric patients: diagnostic value of MR enterography versus reference examinations. AJR Am J Roentgenol 2014, 203, W533–W542. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F.; Bencardino, D.; Buonocore, V.; Mazzamurro, F.; Viola, F.; Oliva, S.; et al. MRI reveals different Crohn’s disease phenotypes in children and adults. Eur Radiol 2019, 29, 5082–5092. [Google Scholar] [CrossRef] [PubMed]
- Alyami, A.S. The Role of Radiomics in Fibrosis Crohn’s Disease: A Review. Diagnostics 2023, 13, 1623. [Google Scholar] [CrossRef] [PubMed]
- Debnath, P.; Dillman, J.R. Quantitative MRI in children with Crohn’s disease – where do we stand? Pediatric Radiology 2024, 54, 1785–1796. [Google Scholar] [CrossRef] [PubMed]
- Halligan, S.; Tolan, D.; Amitai, M.M.; Hoeffel, C.; Kim, S.H.; Maccioni, F.; et al. ESGAR consensus statement on the imaging of fistula-in-ano and other causes of anal sepsis. Eur Radiol 2020, 30, 4734–4740. [Google Scholar] [CrossRef] [PubMed]
- Brillantino, A.; Iacobellis, F.; Marano, L.; Renzi, A.; Talento, P.; Brusciano, L.; et al. Validation of a novel imaging-guided and anatomy-based classification system for anorectal fistulas: a retrospective clinical evaluation study. Ann Coloproctology 2025, 41, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Lo Re, G.; Tudisca, C.; Vernuccio, F.; Picone, D.; Cappello, M.; Agnello, F.; Galia, M.; Galfano, M.C.; Biscaldi, E.; Salerno, S.; Pinto, A.; Midiri, M.; Lagalla, R. Erratum to: MR imaging of perianal fistulas in Crohn's disease: sensitivity and specificity of STIR sequences. Radiol Med 2016, 121, 252. https://doi.org/10.1007/s11547-016-0625-6. Erratum for: Radiol Med 2016, 121, 243–251. [CrossRef] [PubMed]
- Caruso, D.; Polici, M.; Bellini, D.; Laghi, A. ESR Essentials: Imaging in colorectal cancer-practice recommendations by ESGAR. Eur Radiol 2024, 34, 5903–5910. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.; Zerunian, M.; De Santis, D.; Biondi, T.; Paolantonio, P.; Rengo, M.; et al. Magnetic Resonance of Rectal Cancer Response to Therapy: An Image Quality Comparison between 3.0 and 1.5 Tesla. BioMed Res Int 2020, 2020, 9842732. [Google Scholar] [CrossRef] [PubMed]
- Iafrate, F.; Ciccarelli, F.; Masci, G.M.; Grasso, D.; Marruzzo, F.; De Felice, F.; et al. Predictive role of diffusion-weighted MRI in the assessment of response to total neoadjuvant therapy in locally advanced rectal cancer. Eur Radiol 2023, 33, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Amodeo, S.; Rosman, A.S.; Desiato, V.; Hindman, N.M.; Newman, E.; Berman, R.; et al. MRI-Based Apparent Diffusion Coefficient for Predicting Pathologic Response of Rectal Cancer After Neoadjuvant Therapy: Systematic Review and Meta-Analysis. AJR Am J Roentgenol 2018, 211, W205–16. [Google Scholar] [CrossRef] [PubMed]
- Scabini, S.; Romana, C.; Sartini, M.; Attieh, A.; Marrone, C.; Cristina, M.L.; et al. The experience of the COMRE group (REctal COMmittee): can magnetic resonance imaging and endosonography really help the clinical pathway after NCRT in rectal cancer? Int J Surg Lond Engl 2023, 109, 2991–2995. [Google Scholar] [CrossRef] [PubMed]
- Barbaro, B.; Leccisotti, L.; Vecchio, F.M.; Di Matteo, M.; Serra, T.; Salsano, M.; et al. The potential predictive value of MRI and PET-CT in mucinous and nonmucinous rectal cancer to identify patients at high risk of metastatic disease. Br J Radiol. 90, 20150836. [CrossRef] [PubMed]
- Della Corte, A.; Mori, M.; Calabrese, F.; Palumbo, D.; Ratti, F.; Palazzo, G.; et al. Preoperative MRI radiomic analysis for predicting local tumor progression in colorectal liver metastases before microwave ablation. Int J Hyperth Off J Eur Soc Hyperthermic Oncol North Am Hyperth Group 2024, 41, 2349059. [Google Scholar] [CrossRef] [PubMed]
- Vicini, S.; Bortolotto, C.; Rengo, M.; Ballerini, D.; Bellini, D.; Carbone, I.; et al. A narrative review on current imaging applications of artificial intelligence and radiomics in oncology: focus on the three most common cancers. Radiol Med (Torino) 2022, 127, 819–836. [Google Scholar] [CrossRef] [PubMed]
- Granata, V.; Fusco, R.; De Muzio, F.; Brunese, M.C.; Setola, S.V.; Ottaiano, A.; et al. Radiomics and machine learning analysis by computed tomography and magnetic resonance imaging in colorectal liver metastases prognostic assessment. Radiol Med (Torino) 2023, 128, 1310–1332. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.; Polici, M.; Zerunian, M.; Pucciarelli, F.; Guido, G.; Polidori, T.; et al. Radiomics in Oncology, Part 1: Technical Principles and Gastrointestinal Application in CT and MRI. Cancers 2021, 13, 2522. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, R.; Trinci, M.; Casinelli, A.; Treballi, F.; Leone, E.; Caruso, D.; et al. Radiomics in radiology: What the radiologist needs to know about technical aspects and clinical impact. Radiol Med (Torino) 2024, 129, 1751–1765. [Google Scholar] [CrossRef] [PubMed]
- Brogna, B.; Maccioni, F.; Sgambato, D.; Capuano, F.; Iovine, L.; Guarino, S.; Di Libero, L.; Amendola, A.; Faggioni, L.; Cioni, D. The Many Faces of Intestinal Tumors in Adults, Including the Primary Role of CT Imaging in Emergencies and the Important Role of Cross-Sectional Imaging: A Pictorial Review. Healthcare (Basel) 2025, 13, 1071. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Prosperi, D.; Gentiloni Silveri, G.; Panzuto, F.; Faggiano, A.; Russo, V.M.; Caruso, D.; et al. Nuclear Medicine and Radiological Imaging of Pancreatic Neuroendocrine Neoplasms: A Multidisciplinary Update. J Clin Med 2022, 11, 6836. [Google Scholar] [CrossRef] [PubMed]
- De Robertis, R.; Cingarlini, S.; Tinazzi Martini, P.; Ortolani, S.; Butturini, G.; Landoni, L.; et al. Pancreatic neuroendocrine neoplasms: Magnetic resonance imaging features according to grade and stage. World J Gastroenterol 2017, 23, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Farchione, A.; Rufini, V.; Brizi, M.G.; Iacovazzo, D.; Larghi, A.; Massara, R.M.; et al. Evaluation of the Added Value of Diffusion-Weighted Imaging to Conventional Magnetic Resonance Imaging in Pancreatic Neuroendocrine Tumors and Comparison With 68Ga-DOTANOC Positron Emission Tomography/Computed Tomography. Pancreas 2016, 45, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Della Corte, A.; Mori, M.; Calabrese, F.; Palumbo, D.; Ratti, F.; Palazzo, G.; et al. Preoperative MRI radiomic analysis for predicting local tumor progression in colorectal liver metastases before microwave ablation. Int J Hyperth Off J Eur Soc Hyperthermic Oncol North Am Hyperth Group 2024, 41, 2349059. [Google Scholar] [CrossRef] [PubMed]
- Reginelli, A.; Giacobbe, G.; Del Canto, M.T.; Alessandrella, M.; Balestrucci, G.; Urraro, F.; et al. Peritoneal Carcinosis: What the Radiologist Needs to Know. Diagn Basel Switz 2023, 13, 1974. [Google Scholar] [CrossRef] [PubMed]
- Miceli, V.; Gennarini, M.; Tomao, F.; Cupertino, A.; Lombardo, D.; Palaia, I.; et al. Imaging of Peritoneal Carcinomatosis in Advanced Ovarian Cancer: CT, MRI, Radiomic Features and Resectability Criteria. Cancers 2023, 15, 5827. [Google Scholar] [CrossRef] [PubMed]
- Michielsen, K.; Vergote, I.; Op de Beeck, K.; Amant, F.; Leunen, K.; Moerman, P.; et al. Whole-body MRI with diffusion-weighted sequence for staging of patients with suspected ovarian cancer: a clinical feasibility study in comparison to CT and FDG-PET/CT. Eur Radiol 2014, 24, 889–901. [Google Scholar] [CrossRef] [PubMed]
- Brodoefel, H.; Bethge, W.; Vogel, M.; Fenchel, M.; Faul, C.; Wehrmann, M.; et al. Early and late-onset acute GvHD following hematopoietic cell transplantation: CT features of gastrointestinal involvement with clinical and pathological correlation. Eur J Radiol 2010, 73, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F.; La Rocca, U.; Milanese, A.; Busato, L.; Cleri, A.; Lopez, M.; et al. Multi-parametric MRI in the diagnosis and scoring of gastrointestinal acute graft-versus-host disease. Eur Radiol 2023, 33, 5911–5923. [Google Scholar] [CrossRef] [PubMed]
- Derlin, T.; Laqmani, A.; Veldhoen, S.; Apostolova, I.; Ayuk, F.; Adam, G.; et al. Magnetic resonance enterography for assessment of intestinal graft-versus-host disease after allogeneic stem cell transplantation. Eur Radiol 2015, 25, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Budjan, J.; Michaely, H.J.; Attenberger, U.; Haneder, S.; Heidenreich, D.; Kreil, S.; et al. Assessment of acute intestinal graft versus host disease by abdominal magnetic resonance imaging at 3 Tesla. Eur Radiol 2014, 24, 1835–1844. [Google Scholar] [CrossRef] [PubMed]
- Brogna, B.; Frieri, C.; Risitiano, A.M.; Urciuoli, L.; Storti, G.; Santoro, L.; Urciuoli, E.; De Chiara, G.; Cretella, P.; Sementa, C.; Musto, L.A.; Maccioni, F. Intestinal and Extraintestinal Findings of Graft-versus-Host Disease on CT: A Case Series with Radiological and Histopathological Correlations. Biomedicines 2024, 12, 1516. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F. Introduction to the feature section on functional imaging of the pelvic floor. Abdom Imaging 2013, 38, 881–3. [Google Scholar] [CrossRef] [PubMed]
- El Sayed, R.F.; Alt, C.D.; Maccioni, F.; Meissnitzer, M.; Masselli, G.; Manganaro, L.; Vinci, V.; Weishaupt, D.; ESUR and ESGAR Pelvic Floor Working Group. Magnetic resonance imaging of pelvic floor dysfunction - joint recommendations of the ESUR and ESGAR Pelvic Floor Working Group. Eur Radiol 2017, 27, 2067–2085. [Google Scholar] [CrossRef] [PubMed]
- Bitti, G.T.; Argiolas, G.M.; Ballicu, N.; Caddeo, E.; Cecconi, M.; Demurtas, G.; Matta, G.; Peltz, M.T.; Secci, S.; Siotto, P. Pelvic floor failure: MR imaging evaluation of anatomic and functional abnormalities. Radiographics 2014, 34, 429–48. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, F.; Al Ansari, N.; Buonocore, V.; Mazzamurro, F.; Indinnimeo, M.; Mongardini, M.; Catalano, C. Prospective Comparison between two different magnetic resonance defecography techniques for evaluating pelvic floor disorders: air-balloon versus gel for rectal filling. Eur Radiol 2016, 26, 1783–1791. https://doi.org/10.1007/s00330-015-4016-5. Epub 2015 Oct 6. Erratum in: Eur Radiol 2017, 27, 1333. [CrossRef] [PubMed]
- Maccioni F, Alt CD. MRI of the Pelvic Floor and MR Defecography. 2018 Mar 21. In: Hodler J,Kubik-Huch RA, von Schulthess GK, editors. Diseases of the Abdomen and Pelvis 2018-2021:Diagnostic Imaging - IDKD Book [Internet]. Cham (CH): Springer; 2018. Chapter 2. [PubMed]
- Piloni, V.; Manisco, T.; Fogante, M. Magnetic Resonance Imaging Template to Standardize Reporting of Evacuation Disorders. J Imaging 2024, 10, 302. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, F.; Reginelli, A.; Berritto, D.; Gagliardi, G.; Laporta, A.; Brillantino, A.; Renzi, A.; Scaglione, M.; Masselli, G.; Barile, A.; Romano, L.; Cappabianca, S.; Grassi, R. Pelvic floor dysfunctions: how to image patients? Jpn J Radiol 2020, 38, 47–63. [Google Scholar] [CrossRef] [PubMed]
- Colarieti, A.; Stuto, A.; Cellerino, P.; Sardanelli, F. Clinical value of MR defecography: What additional knowledge is provided by the radiologist to the surgeon? Eur J Radiol. 2024. [CrossRef] [PubMed]
- de Jonge, C.S.; Gollifer, R.M.; Nederveen, A.J.; Atkinson, D.; Taylor, S.A.; Stoker, J.; et al. Dynamic MRI for bowel motility imaging-how fast and how long? Br J Radiol 2018, 91, 20170845. [Google Scholar] [CrossRef] [PubMed]
- Menys, A.; Atkinson, D.; Odille, F.; Ahmed, A.; Novelli, M.; Rodriguez-Justo, M.; et al. Quantified terminal ileal motility during MR enterography as a potential biomarker of Crohn’s disease activity: a preliminary study. Eur Radiol 2012, 22, 2494–2501. [Google Scholar] [CrossRef] [PubMed]
- van Harten, L.D.; de Jonge, C.S.; Struik, F.; Stoker, J.; Išgum, I. Quantitative Analysis of Small Intestinal Motility in 3D Cine-MRI Using Centerline-Aware Motion Estimation. J Magn Reson Imaging JMRI 2025, 61, 1956–1966. [Google Scholar] [CrossRef] [PubMed]
- Menys, A.; Taylor, S.A.; Emmanuel, A.; Ahmed, A.; Plumb, A.A.; Odille, F.; et al. Global small bowel motility: assessment with dynamic MR imaging. Radiology 2013, 269, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Cannarozzi, A.L.; Massimino, L.; Latiano, A.; Parigi, T.L.; Giuliani, F.; Bossa, F.; et al. Artificial intelligence: A new tool in the pathologist’s armamentarium for the diagnosis of IBD. Comput Struct Biotechnol J 2024, 23, 3407–3417. [Google Scholar] [CrossRef] [PubMed]
- Da Rio, L.; Spadaccini, M.; Parigi, T.L.; Gabbiadini, R.; Dal Buono, A.; Busacca, A.; et al. Artificial intelligence and inflammatory bowel disease: Where are we going? World J Gastroenterol 2023, 29, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Sedano, R.; Solitano, V.; Vuyyuru, S.K.; Yuan, Y.; Hanžel, J.; Ma, C.; et al. Artificial intelligence to revolutionize IBD clinical trials: a comprehensive review. Ther Adv Gastroenterol 2025, 18, 17562848251321915. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).