Submitted:
15 July 2025
Posted:
17 July 2025
Read the latest preprint version here
Abstract

Keywords:
1. Introduction
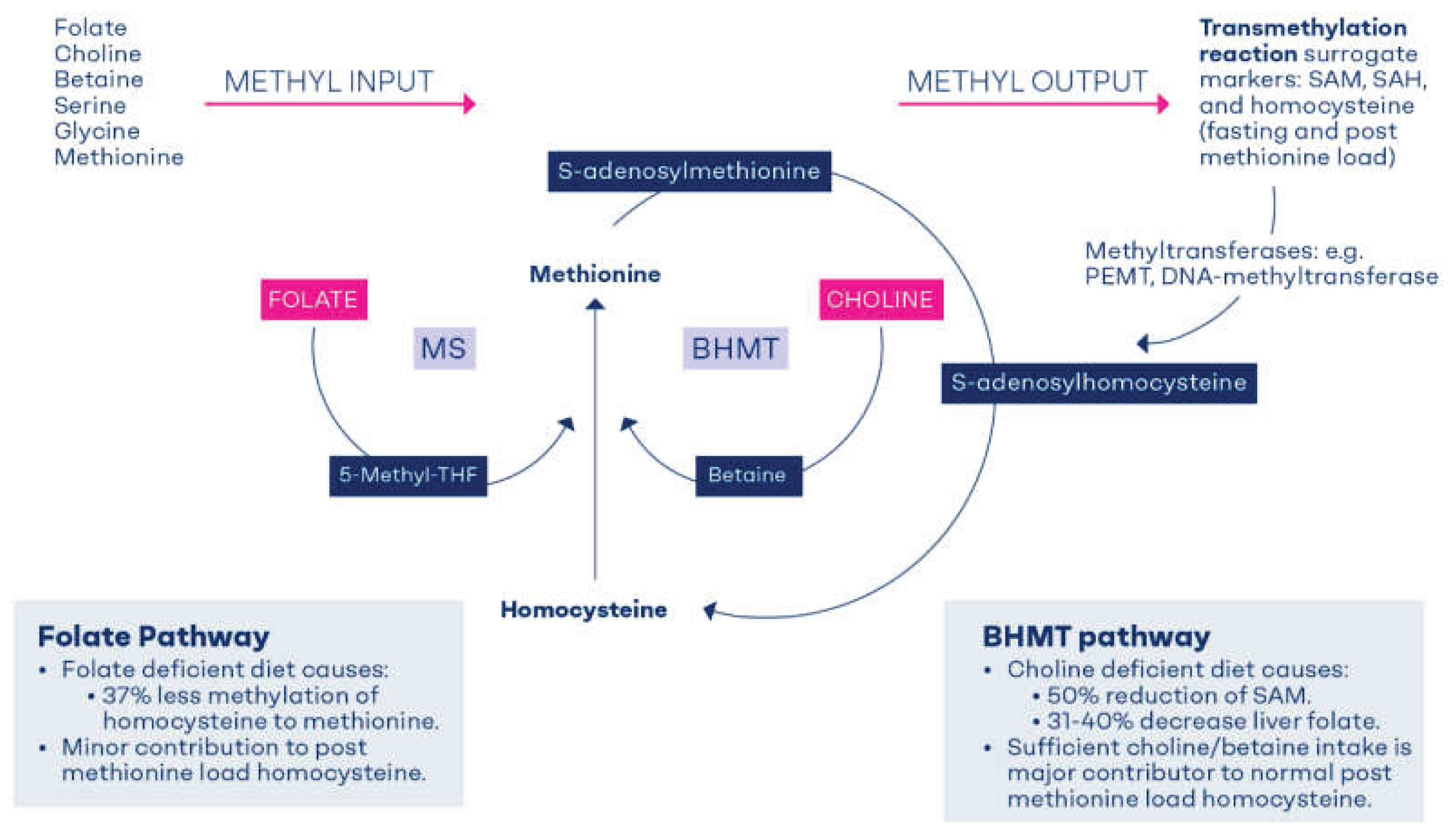
2. Formation of Methyl Groups in One Carbon Metabolism
3. Functional Test to Differentiate Between Contributions of Folate and Choline to C1-Metabolism
4. Safeguarding the Methyl Balance Through the Diet or Methylneogenesis
5. Factors Affecting Methylneogenesis
| Factor | Explanation | Reference |
|---|---|---|
| Dietary deficiency of selected nutrients | Sufficient intakes of folate, choline, betaine, methionine and serine can compensate for temporary lack of other nutrients with methyl donor function. | |
| Sex | Expression and activity of several genes in C1-metabolism differ by sex. | [41] |
| Age | Estrogen-dependent regulation of PEMT gene provides additional source of choline in liver of premenopausal and pregnant women. | [48,49] |
| Polymorphisms in genes involved in the folate pathway | Individuals with MTHFR677TT genotype may be prone to low folate and rely more on generating SAM from choline, betaine and other nutrients. | [55] |
| polymorphisms in genes that rely on SAM | PEMT gene polymorphisms could reduce endogenous synthesis of phosphatidylcholine and thus the net flux of methyl groups. | [50,51] |
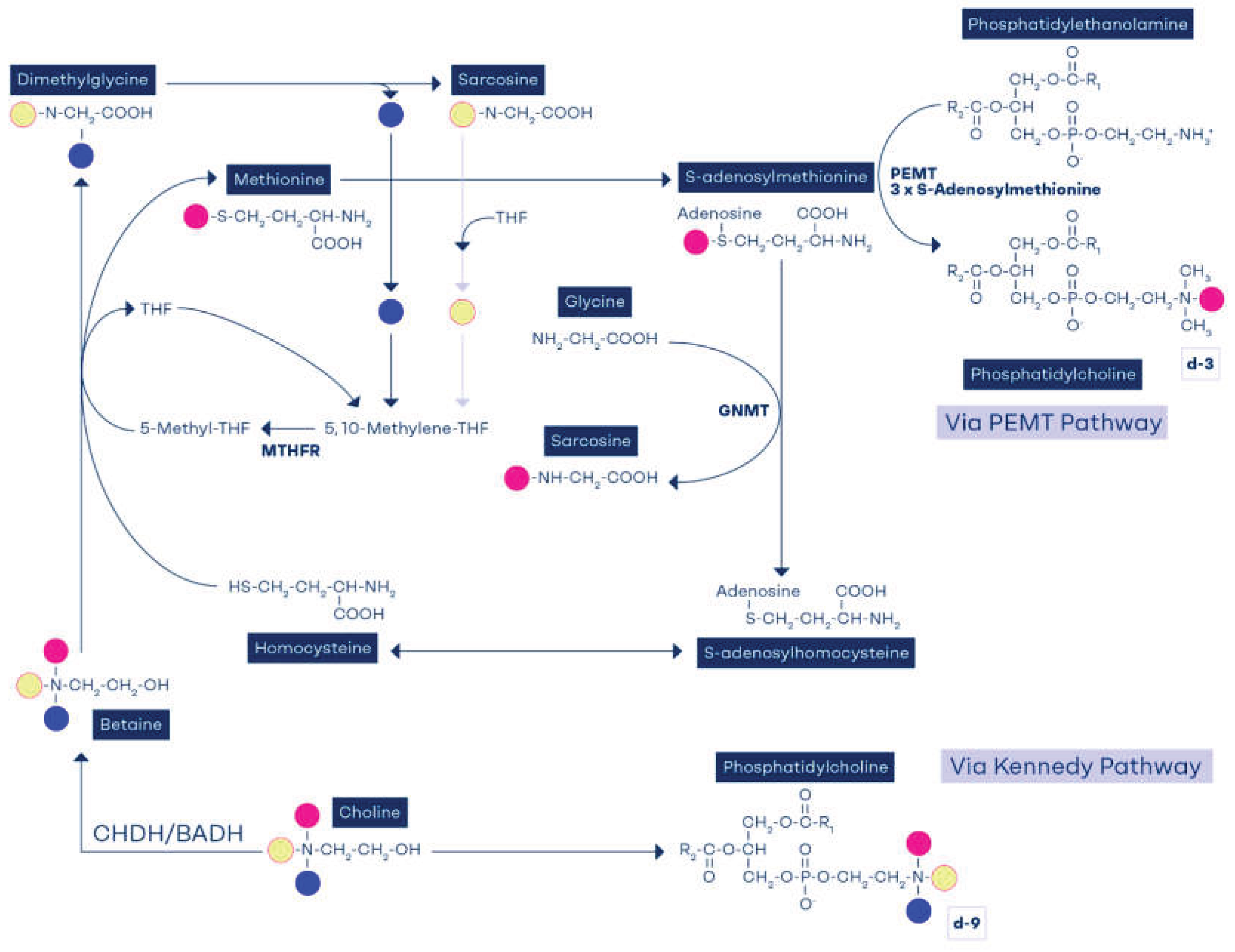
6. Phosphatidylethanolamine methyltransferase Role in Methylneogenesis
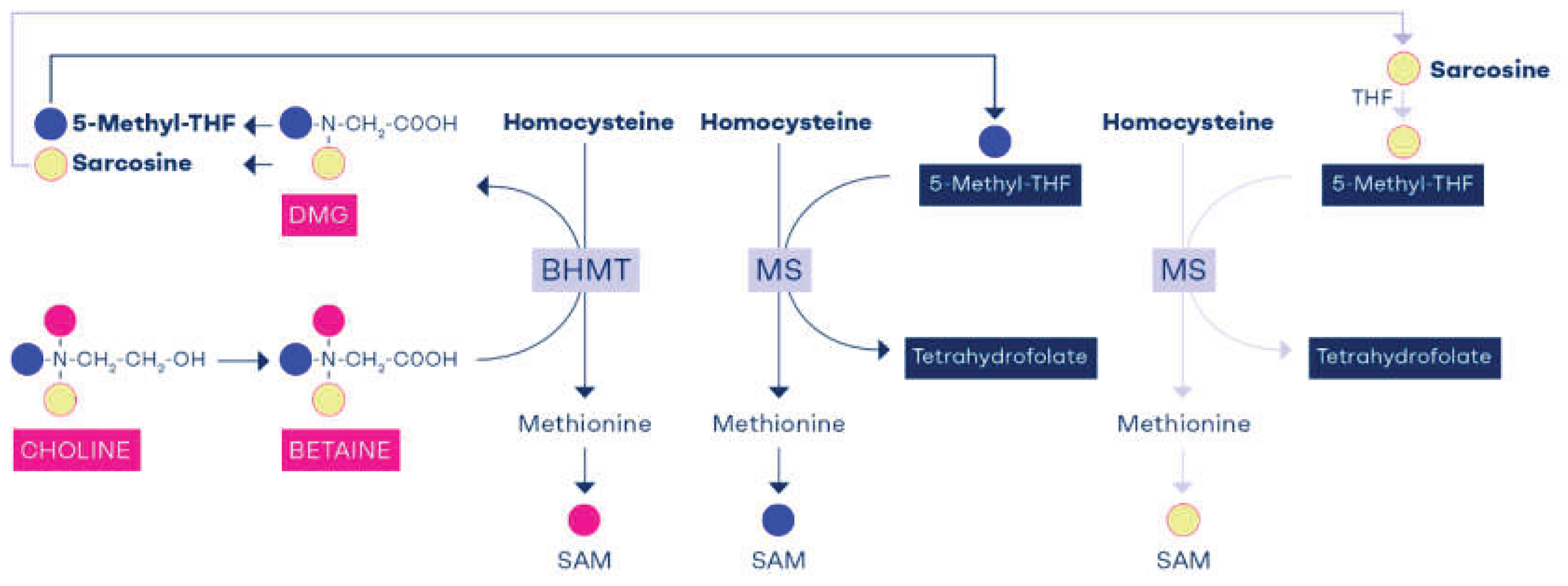
7. Tracking the Methyl Groups of Betaine and Choline
8. Interdependency of Folate and Choline
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Finkelstein J.D., Kyle W.E., Harris B.J. Methionine metabolism in mammals: regulatory effects of S-adenosylhomocysteine. Arch. Biochem. Biophys. 1974, 165, 774-779. [CrossRef]
- Kim Y.I., Miller J.W., da Costa K.A., Nadeau M., Smith D., Selhub J., Zeisel S.H., Mason J.B. Severe folate deficiency causes secondary depletion of choline and phosphocholine in rat liver. J Nutr. 1994, 124, 2197-2203. [CrossRef]
- Johnson B.C., James M.F. Choline deficiency in the baby pig. J. Nutr. 1948, 36, 339-349. [CrossRef]
- Selhub J., Seyoum E., Pomfret E.A., Zeisel S.H. Effects of choline deficiency and methotrexate treatment upon liver folate content and distribution. Cancer Res 1991, 51, 16-21.
- Horne D.W., Cook R.J., Wagner C. Effect of dietary methyl group deficiency on folate metabolism in rats. J Nutr. 1989, 119, 618-621. [CrossRef]
- Varela-Moreiras G., Ragel C., Perez de M.J. Choline deficiency and methotrexate treatment induces marked but reversible changes in hepatic folate concentrations, serum homocysteine and DNA methylation rates in rats. J Am. Coll. Nutr. 1995, 14, 480-485. [CrossRef]
- Teng Y.W., Mehedint M.G., Garrow T.A., Zeisel S.H. Deletion of betaine-homocysteine S-methyltransferase in mice perturbs choline and 1-carbon metabolism, resulting in fatty liver and hepatocellular carcinomas. J Biol. Chem 2011, 286, 36258-36267. [CrossRef]
- Steenge G.R., Verhoef P., Katan M.B. Betaine supplementation lowers plasma homocysteine in healthy men and women. J Nutr. 2003, 133, 1291-1295. [CrossRef]
- Atkinson W., Elmslie J., Lever M., Chambers S.T., George P.M. Dietary and supplementary betaine: acute effects on plasma betaine and homocysteine concentrations under standard and postmethionine load conditions in healthy male subjects. Am. J Clin Nutr. 2008, 87, 577-585. [CrossRef]
- Cuskelly G.J., Stacpoole P.W., Williamson J., Baumgartner T.G., Gregory J.F., III. Deficiencies of folate and vitamin B(6) exert distinct effects on homocysteine, serine, and methionine kinetics. Am J Physiol Endocrinol. Metab 2001, 281, E1182-E1190. [CrossRef]
- Mudd S.H., Poole J.R. Labile methyl balances for normal humans on various dietary regimens. Metabolism 1975, 24, 721-735. [CrossRef]
- Chen L.H., Liu M.L., Hwang H.Y., Chen L.S., Korenberg J., Shane B. Human methionine synthase. cDNA cloning, gene localization, and expression. J Biol. Chem 1997, 272, 3628-3634. [CrossRef]
- Finkelstein J.D. Pathways and regulation of homocysteine metabolism in mammals. Semin. Thromb. Hemost. 2000, 26, 219-225. [CrossRef]
- Galletti P., De R.M., Cotticelli M.G., Morana A., Vaccaro R., Zappia V. Biochemical rationale for the use of CDPcholine in traumatic brain injury: pharmacokinetics of the orally administered drug. J Neurol. Sci. 1991, 103 Suppl, S19-S25. [CrossRef]
- Stipanuk M.H. Metabolism of sulfur-containing amino acids. Annu. Rev. Nutr 1986, 6, 179-209. [CrossRef]
- Schafer C., Hoffmann L., Heldt K., Lornejad-Schafer M.R., Brauers G., Gehrmann T., Garrow T.A., Haussinger D., Mayatepek E., Schwahn B.C. et al. Osmotic regulation of betaine homocysteine-S-methyltransferase expression in H4IIE rat hepatoma cells. Am. J Physiol Gastrointest. Liver Physiol 2007, 292, G1089-G1098. [CrossRef]
- Millian N.S., Garrow T.A. Human betaine-homocysteine methyltransferase is a zinc metalloenzyme. Arch Biochem. Biophys. 1998, 356, 93-98. [CrossRef]
- McKeever M.P., Weir D.G., Molloy A., Scott J.M. Betaine-homocysteine methyltransferase: organ distribution in man, pig and rat and subcellular distribution in the rat. Clin Sci (Lond) 1991, 81, 551-556. [CrossRef]
- Wilcken D.E., Wilcken B., Dudman N.P., Tyrrell P.A. Homocystinuria--the effects of betaine in the treatment of patients not responsive to pyridoxine. N. Engl. J Med 1983, 309, 448-453. [CrossRef]
- Wilcken D.E., Dudman N.P., Tyrrell P.A. Homocystinuria due to cystathionine beta-synthase deficiency--the effects of betaine treatment in pyridoxine-responsive patients. Metabolism 1985, 34, 1115-1121. [CrossRef]
- Buchman A.L., Dubin M., Jenden D., Moukarzel A., Roch M.H., Rice K., Gornbein J., Ament M.E., Eckhert C.D. Lecithin increases plasma free choline and decreases hepatic steatosis in long-term total parenteral nutrition patients. Gastroenterology 1992, 102, 1363-1370. [CrossRef]
- Selhub J., Jacques P.F., Wilson P.W., Rush D., Rosenberg I.H. Vitamin status and intake as primary determinants of homocysteinemia in an elderly population. JAMA 1993, 270, 2693-2698. [CrossRef]
- Chiuve S.E., Giovannucci E.L., Hankinson S.E., Zeisel S.H., Dougherty L.W., Willett W.C., Rimm E.B. The association between betaine and choline intakes and the plasma concentrations of homocysteine in women. Am. J Clin Nutr. 2007, 86, 1073-1081. [CrossRef]
- Olthof M.R., Brink E.J., Katan M.B., Verhoef P. Choline supplemented as phosphatidylcholine decreases fasting and postmethionine-loading plasma homocysteine concentrations in healthy men. Am. J Clin Nutr. 2005, 82, 111-117. [CrossRef]
- Olthof M.R., van V.T., Boelsma E., Verhoef P. Low dose betaine supplementation leads to immediate and long term lowering of plasma homocysteine in healthy men and women. J Nutr. 2003, 133, 4135-4138. [CrossRef]
- Ubbink J.B., Becker P.J., Delport R., Bester M., Riezler R., Vermaak W.J. Variability of post-methionine load plasma homocysteine assays. Clin Chim. Acta 2003, 330, 111-119. [CrossRef]
- van der Griend R., Haas F.J., Duran M., Biesma D.H., Meuwissen O.J., Banga J.D. Methionine loading test is necessary for detection of hyperhomocysteinemia. J. Lab Clin Med 1998, 132, 67-72. [CrossRef]
- van der Griend R., Biesma D.H., Banga J.D. Postmethionine-load homocysteine determination for the diagnosis hyperhomocysteinaemia and efficacy of homocysteine lowering treatment regimens. Vasc. Med 2002, 7, 29-33. [CrossRef]
- Bostom A.G., Jacques P.F., Nadeau M.R., Williams R.R., Ellison R.C., Selhub J. Post-methionine load hyperhomocysteinemia in persons with normal fasting total plasma homocysteine: initial results from the NHLBI Family Heart Study. Atherosclerosis 1995, 116, 147-151. [CrossRef]
- Lever M., Slow S., McGregor D.O., Dellow W.J., George P.M., Chambers S.T. Variability of plasma and urine betaine in diabetes mellitus and its relationship to methionine load test responses: an observational study. Cardiovasc. Diabetol. 2012, 11, 34. [CrossRef]
- Holm P.I., Bleie O., Ueland P.M., Lien E.A., Refsum H., Nordrehaug J.E., Nygard O. Betaine as a determinant of postmethionine load total plasma homocysteine before and after B-vitamin supplementation. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 301-307. [CrossRef]
- Holm P.I., Ueland P.M., Vollset S.E., Midttun O., Blom H.J., Keijzer M.B., den Heijer M. Betaine and folate status as cooperative determinants of plasma homocysteine in humans. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 379-385. [CrossRef]
- Lee J.E., Jacques P.F., Dougherty L., Selhub J., Giovannucci E., Zeisel S.H., Cho E. Are dietary choline and betaine intakes determinants of total homocysteine concentration? Am. J Clin Nutr. 2010, 91, 1303-1310. [CrossRef]
- da Costa K.A., Gaffney C.E., Fischer L.M., Zeisel S.H. Choline deficiency in mice and humans is associated with increased plasma homocysteine concentration after a methionine load. Am. J Clin Nutr. 2005, 81, 440-444. [CrossRef]
- Verhoef P., Steenge G.R., Boelsma E., van V.T., Olthof M.R., Katan M.B. Dietary serine and cystine attenuate the homocysteine-raising effect of dietary methionine: a randomized crossover trial in humans. Am J Clin Nutr 2004, 80, 674-679. [CrossRef]
- Figueroa-Torres A.G., Matias-Aguilar L.O., Coria-Ramirez E., Bonilla-Gonzalez E., Gonzalez-Marquez H., Ibarra-Gonzalez I., Hernandez-Lopez J.R., Hernandez-Juarez J., Dominguez-Reyes V.M., Isordia-Salas I. et al. Cystathionine beta-synthase and methylenetetrahydrofolate reductase mutations in Mexican individuals with hyperhomocysteinemia. SAGE Open. Med 2020, 8, 2050312120974193. [CrossRef]
- Lievers K.J., Kluijtmans L.A., Heil S.G., Boers G.H., Verhoef P., den H.M., Trijbels F.J., Blom H.J. Cystathionine beta-synthase polymorphisms and hyperhomocysteinaemia: an association study. Eur. J Hum. Genet. 2003, 11, 23-29. [CrossRef]
- Bhat D.S., Gruca L.L., Bennett C.D., Katre P., Kurpad A.V., Yajnik C.S., Kalhan S.C. Evaluation of tracer labelled methionine load test in vitamin B-12 deficient adolescent women. PLoS. ONE. 2018, 13, e0196970. [CrossRef]
- Chiang E.P., Selhub J., Bagley P.J., Dallal G., Roubenoff R. Pyridoxine supplementation corrects vitamin B6 deficiency but does not improve inflammation in patients with rheumatoid arthritis. Arthritis Res Ther. 2005, 7, R1404-R1411. [CrossRef]
- de J.R., Griffioen P.H., van Z.B., Brouns R.M., Visser W., Lindemans J. Evaluation of a shorter methionine loading test. Clin Chem Lab Med 2004, 42, 1027-1031. [CrossRef]
- Sadre-Marandi F., Dahdoul T., Reed M.C., Nijhout H.F. Sex differences in hepatic one-carbon metabolism. BMC. Syst. Biol. 2018, 12, 89. [CrossRef]
- Nelen W.L., Blom H.J., Thomas C.M., Steegers E.A., Boers G.H., Eskes T.K. Methylenetetrahydrofolate reductase polymorphism affects the change in homocysteine and folate concentrations resulting from low dose folic acid supplementation in women with unexplained recurrent miscarriages. J Nutr 1998, 128, 1336-1341. [CrossRef]
- Mudd S.H., Brosnan J.T., Brosnan M.E., Jacobs R.L., Stabler S.P., Allen R.H., Vance D.E., Wagner C. Methyl balance and transmethylation fluxes in humans. Am. J Clin Nutr. 2007, 85, 19-25. [CrossRef]
- Mudd S.H., Ebert M.H., Scriver C.R. Labile methyl group balances in the human: the role of sarcosine. Metabolism 1980, 29, 707-720. [CrossRef]
- Storch K.J., Wagner D.A., Burke J.F., Young V.R. Quantitative study in vivo of methionine cycle in humans using [methyl-2H3]- and [1-13C]methionine. Am J Physiol 1988, 255, E322-E331. [CrossRef]
- Lamers Y., Williamson J., Gilbert L.R., Stacpoole P.W., Gregory J.F., III. Glycine turnover and decarboxylation rate quantified in healthy men and women using primed, constant infusions of [1,2-(13)C2]glycine and [(2)H3]leucine. J Nutr 2007, 137, 2647-2652. [CrossRef]
- Im Y.S., Chiang P.K., Cantoni G.L. Guanidoacetate methyltransferase. Purification and molecular properties. J Biol Chem 1979, 254, 11047-11050. [CrossRef]
- Resseguie M., Song J., Niculescu M.D., da Costa K.A., Randall T.A., Zeisel S.H. Phosphatidylethanolamine N-methyltransferase (PEMT) gene expression is induced by estrogen in human and mouse primary hepatocytes. FASEB J 2007, 21, 2622-2632. [CrossRef]
- Resseguie M.E., da Costa K.A., Galanko J.A., Patel M., Davis I.J., Zeisel S.H. Aberrant estrogen regulation of PEMT results in choline deficiency-associated liver dysfunction. J Biol. Chem 2011, 286, 1649-1658. [CrossRef]
- da Costa K.A., Kozyreva O.G., Song J., Galanko J.A., Fischer L.M., Zeisel S.H. Common genetic polymorphisms affect the human requirement for the nutrient choline. FASEB J 2006, 20, 1336-1344. [CrossRef]
- Fischer L.M., daCosta K.A., Kwock L., Stewart P.W., Lu T.S., Stabler S.P., Allen R.H., Zeisel S.H. Sex and menopausal status influence human dietary requirements for the nutrient choline. Am. J Clin Nutr. 2007, 85, 1275-1285. [CrossRef]
- Schwahn B.C., Laryea M.D., Chen Z., Melnyk S., Pogribny I., Garrow T., James S.J., Rozen R. Betaine rescue of an animal model with methylenetetrahydrofolate reductase deficiency. Biochem. J 2004, 382, 831-840. [CrossRef]
- Chen Z., Karaplis A.C., Ackerman S.L., Pogribny I.P., Melnyk S., Lussier-Cacan S., Chen M.F., Pai A., John S.W., Smith R.S. et al. Mice deficient in methylenetetrahydrofolate reductase exhibit hyperhomocysteinemia and decreased methylation capacity, with neuropathology and aortic lipid deposition. Hum. Mol. Genet. 2001, 10, 433-443. [CrossRef]
- Colson N.J., Naug H.L., Nikbakht E., Zhang P., McCormack J. The impact of MTHFR 677 C/T genotypes on folate status markers: a meta-analysis of folic acid intervention studies. Eur. J Nutr 2017, 56, 247-260. [CrossRef]
- Yan J., Wang W., Gregory J.F., III, Malysheva O., Brenna J.T., Stabler S.P., Allen R.H., Caudill M.A. MTHFR C677T genotype influences the isotopic enrichment of one-carbon metabolites in folate-compromised men consuming d9-choline. Am. J Clin Nutr. 2011, 93, 348-355. [CrossRef]
- Vance D.E., Ridgway N.D. The methylation of phosphatidylethanolamine. Prog. Lipid Res. 1988, 27, 61-79. [CrossRef]
- DeLong C.J., Hicks A.M., Cui Z. Disruption of choline methyl group donation for phosphatidylethanolamine methylation in hepatocarcinoma cells. J Biol. Chem 2002, 277, 17217-17225. [CrossRef]
- Vance D.E., Walkey C.J., Cui Z. Phosphatidylethanolamine N-methyltransferase from liver. Biochim. Biophys. Acta 1997, 1348, 142-150. [CrossRef]
- Stead L.M., Brosnan J.T., Brosnan M.E., Vance D.E., Jacobs R.L. Is it time to reevaluate methyl balance in humans? Am. J Clin Nutr. 2006, 83, 5-10. [CrossRef]
- Shields D.J., Lingrell S., Agellon L.B., Brosnan J.T., Vance D.E. Localization-independent regulation of homocysteine secretion by phosphatidylethanolamine N-methyltransferase. J Biol. Chem 2005, 280, 27339-27344. [CrossRef]
- Jacobs R.L., Stead L.M., Devlin C., Tabas I., Brosnan M.E., Brosnan J.T., Vance D.E. Physiological regulation of phospholipid methylation alters plasma homocysteine in mice. J Biol. Chem 2005, 280, 28299-28305. [CrossRef]
- Jacobs R.L., Devlin C., Tabas I., Vance D.E. Targeted deletion of hepatic CTP:phosphocholine cytidylyltransferase alpha in mice decreases plasma high density and very low density lipoproteins. J Biol Chem 2004, 279, 47402-47410. [CrossRef]
- Schwahn B.C., Wendel U., Lussier-Cacan S., Mar M.H., Zeisel S.H., Leclerc D., Castro C., Garrow T.A., Rozen R. Effects of betaine in a murine model of mild cystathionine-beta-synthase deficiency. Metabolism 2004, 53, 594-599. [CrossRef]
- Schwahn B.C., Chen Z., Laryea M.D., Wendel U., Lussier-Cacan S., Genest J., Jr., Mar M.H., Zeisel S.H., Castro C., Garrow T. et al. Homocysteine-betaine interactions in a murine model of 5,10-methylenetetrahydrofolate reductase deficiency. FASEB J 2003, 17, 512-514. [CrossRef]
- Watkins S.M., Zhu X., Zeisel S.H. Phosphatidylethanolamine-N-methyltransferase activity and dietary choline regulate liver-plasma lipid flux and essential fatty acid metabolism in mice. J Nutr 2003, 133, 3386-3391. [CrossRef]
- DeLong C.J., Shen Y.J., Thomas M.J., Cui Z. Molecular distinction of phosphatidylcholine synthesis between the CDP-choline pathway and phosphatidylethanolamine methylation pathway. J Biol. Chem 1999, 274, 29683-29688. [CrossRef]
- Augustin P., Hromic A., Pavkov-Keller T., Gruber K., Macheroux P. Structure and biochemical properties of recombinant human dimethylglycine dehydrogenase and comparison to the disease-related H109R variant. FEBS J 2016, 283, 3587-3603. [CrossRef]
- Robinson J.L., McBreairty L.E., Randell E.W., Harding S.V., Bartlett R.K., Brunton J.A., Bertolo R.F. Betaine or folate can equally furnish remethylation to methionine and increase transmethylation in methionine-restricted neonates. J Nutr Biochem. 2018, 59, 129-135. [CrossRef]
- Barak A.J., Kemmy R.J. Methotrexate effects on hepatic betaine levels in choline-supplemented and choline-deficient rats. Drug Nutr. Interact. 1982, 1, 275-278.
- Barak A.J., Tuma D.J., Beckenhauer H.C. Methotrexate hepatotoxicity. J Am Coll. Nutr 1984, 3, 93-96. [CrossRef]
- Freeman-Narrod M., Narrod S.A., Custer R.P. Chronic toxicity of methotrexate in rats: partial to complete projection of the liver by choline: Brief communication. J Natl. Cancer Inst. 1977, 59, 1013-1017. [CrossRef]
- Pomfret E.A., daCosta K.A., Zeisel S.H. Effects of choline deficiency and methotrexate treatment upon rat liver. J Nutr Biochem. 1990, 1, 533-541. [CrossRef]
- Svardal A.M., Ueland P.M., Berge R.K., Aarsland A., Aarsaether N., Lonning P.E., Refsum H. Effect of methotrexate on homocysteine and other sulfur compounds in tissues of rats fed a normal or a defined, choline-deficient diet. Cancer Chemother. Pharmacol. 1988, 21, 313-318. [CrossRef]
- Poirier L.A., Grantham P.H., Rogers A.E. The effects of a marginally lipotrope-deficient diet on the hepatic levels of S-adenosylmethionine and on the urinary metabolites of 2-acetylaminofluorene in rats. Cancer Res. 1977, 37, 744-748.
- Barak A.J., Kemmy R.J., Tuma D.J. The effect of methotrexate on homocysteine methylating agents in rat liver. Drug Nutr Interact. 1982, 1, 303-306.
- Shivapurkar N., Poirier L.A. Tissue levels of S-adenosylmethionine and S-adenosylhomocysteine in rats fed methyl-deficient, amino acid-defined diets for one to five weeks. Carcinogenesis 1983, 4, 1051-1057. [CrossRef]
- Zeisel S.H., Zola T., daCosta K.A., Pomfret E.A. Effect of choline deficiency on S-adenosylmethionine and methionine concentrations in rat liver. Biochem. J 1989, 259, 725-729.
| Factor | PML-homocysteine | Reference |
|---|---|---|
| Elevated fasting plasma homocysteine | ↑↑ | [30,31] |
| Higher plasma betaine | ↓ ↓ ↓ | [30,31,32] |
| Higher betaine intake (dietary and supplemental) | ↓ ↓ ↓ | [8,9,33] |
| Higher choline intake (dietary and supplemental) | ↓ ↓ ↓ | [9,33,34] |
| Acute intake of choline/betaine (single dose studies or after a meal) | ↓ ↓ ↓ | [24,25] |
| Higher serine and cysteine intake | ↓ ↓ | [35] |
| Higher plasma folate | ↓ | [32] |
| Higher folate intake (dietary and supplemental) | (↓↑) | [8] |
| Common polymorphisms affecting transsulfuration pathway such as in cystathionine β-synthase | (↓↑) | [36,37] |
| Low vitamin B12 status | (↑) | [38] |
| Pyridoxine (vitamin B6) supplementation | (↓) | [31,39] |
| ↓ lower, (↑) slightly higher, (↓) slightly lower, (↓↑) no clear effect. | ||
| PML-homocysteine, µmol/L | F-homocysteine, µmol/L | Delta (PML- minus F-homocysteine), µmol/L | Percent change (PML vs. baseline homocysteine), % | Relationship between PML-homocysteine and F-homocysteine | |
| 1- Native situation - no supplement (Olthof et al., [24] and Steenge et al., [8]) | |||||
| 32.6 | 15.6 | 17.0 | +109.0 | PML-homocysteine = F-homocysteine * 2.09. | |
| 34.8 | 12.2 | 22.6 | +185.2 | PML-homocysteine = F-homocysteine * 2.85. | |
| 31.6 | 13.0 | 18.6 | +143.1 | PML-homocysteine = F-homocysteine * 2.43. | |
| Mean = 33.0 | 13.6 | 19.4 | +145.8 | PML-homocysteine = F-homocysteine * 2.46. | |
| 2- Supplemented with 2.6 g/d choline for 2 weeks (Olthof et al., [24]) | |||||
| 22.3 | 13.6 | 8.7 | +64.0 | PML-homocysteine = F-homocysteine * 1.64. Methionine load test in choline intake-optimized persons led to roughly 55% lower PML-homocysteine compared to non-supplemented people (8.7 vs. 19.4 µmol/L) |
|
| 3- Supplemented with 3*2 g/d betaine for 6 weeks (Steenge et al., [8]) | |||||
| 17.6 | 10.9 | 6.7 | +61.5 | PML-homocysteine = F-homocysteine * 1.62. Methionine load test in betaine intake optimized persons led to roughly 65% lower PML-homocysteine compared to non-supplemented people (6.7 vs. 19.4 µmol/L) |
|
| 4- Supplemented with 400 µg* 2/d folic acid for 6 weeks (Steenge et al., [8]) | |||||
| 33 | 10.7 | 22.3 | +208.4 | PML-homocysteine = F-homocysteine * 3.1. Optimization of folate status has no lowering effect on PML-homocysteine compared to non-supplemented people (22.3 vs. 19.4 µmol/L) |
|
|
| Question | Elaboration |
|---|---|
| Sex differences: Is the methylation flux higher in men than in women? | The expressions of several enzymes in C1-metabolism show sex-differences [41]. For example, men have higher plasma homocysteine and betaine than women and PEMT gene is upregulated by estrogen. |
| Is there a dose response relationship between choline intake and PML-homocysteine? | A dose response relationship between betaine intake and PML-homocysteine has been demonstrated [25]. Does the same apply for choline intake and what is the intake level to achieve a maximal reduction of PML-homocysteine? |
| May high dose betaine or choline compensate for folate deficiency in term of lowering PML-homocysteine? | Addressing metabolic capacity to upregulate methyl group flow via betaine/choline in people with folate deficiency or MTHFRC677T TT genotype. |
| Can PML-homocysteine be used to define the optimal intake of choline or betaine in pregnant and lactating women? | Homocysteine concentrations after a methionine load test can be tested before and after loading the gap of choline or betaine intakes. |
| Is | |
| Can PML-homocysteine test be used to identify women at high risk of neural tube defects or other pregnancy complications such as recurrent pregnancy loos, gestational diabetes or preeclampsia? | In one study among women with a history of recurrent pregnancy loss, folic acid supplementation (0.5 mg/d for 2 months) did not lower PML-homocysteine in 53% of the women [42]. In theory, the PML-homocysteine test may identify women who could benefit from choline/betaine supplements through increasing methyl group flux via the BHMT pathway and normalizing PML-homocysteine. This may influence disease risk. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




