Submitted:
15 July 2025
Posted:
16 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Antibodies
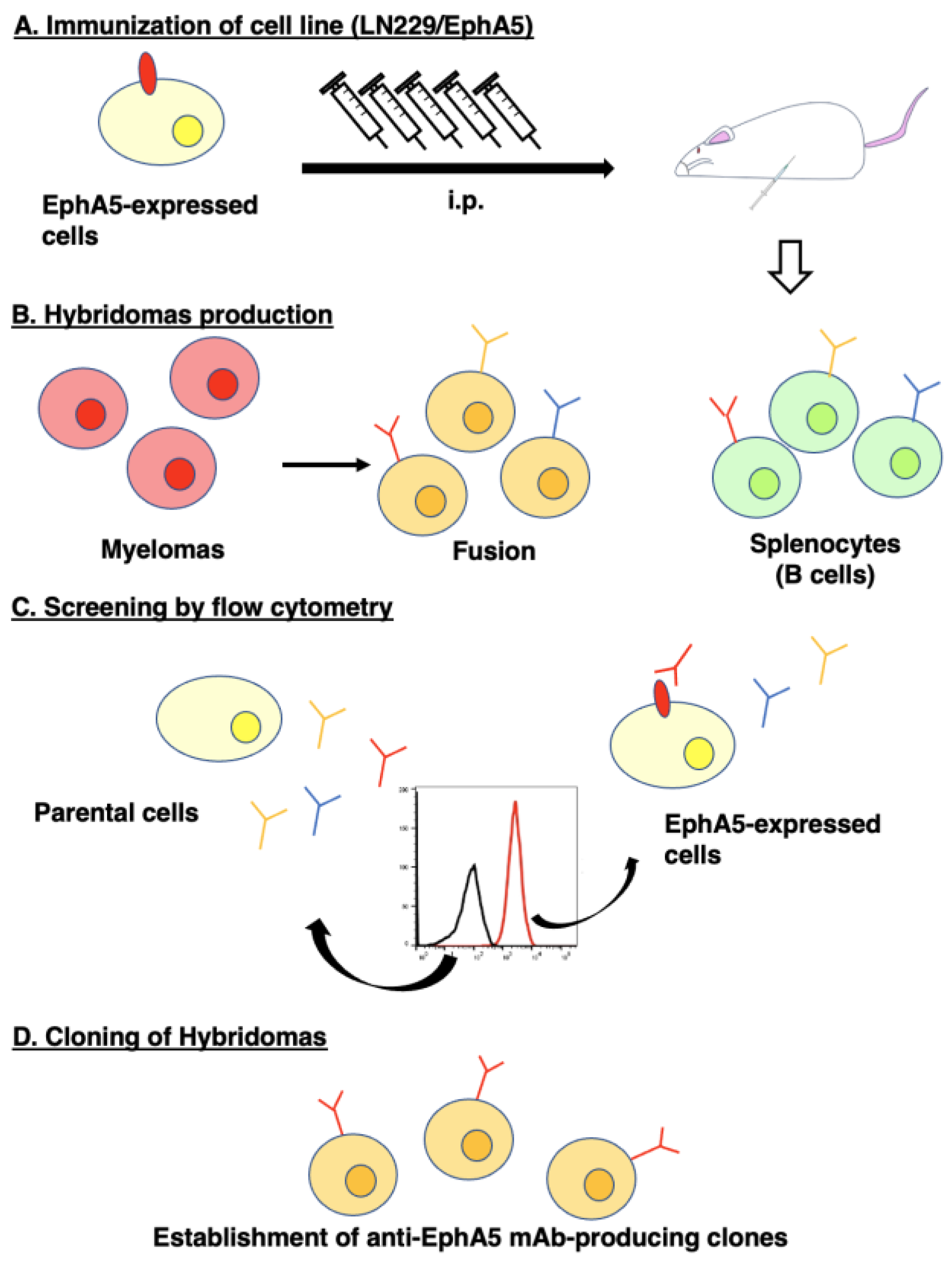
2.3. Hybridoma Production
2.4. Flow Cytometry
2.5. Determination of the Binding Affinity by Flow Cytometry
2.6. Immunohistochemistry
3. Results
3.1. Development of an Anti-EphA5 mAb, Ea5Mab-7 Using the CBIS Method
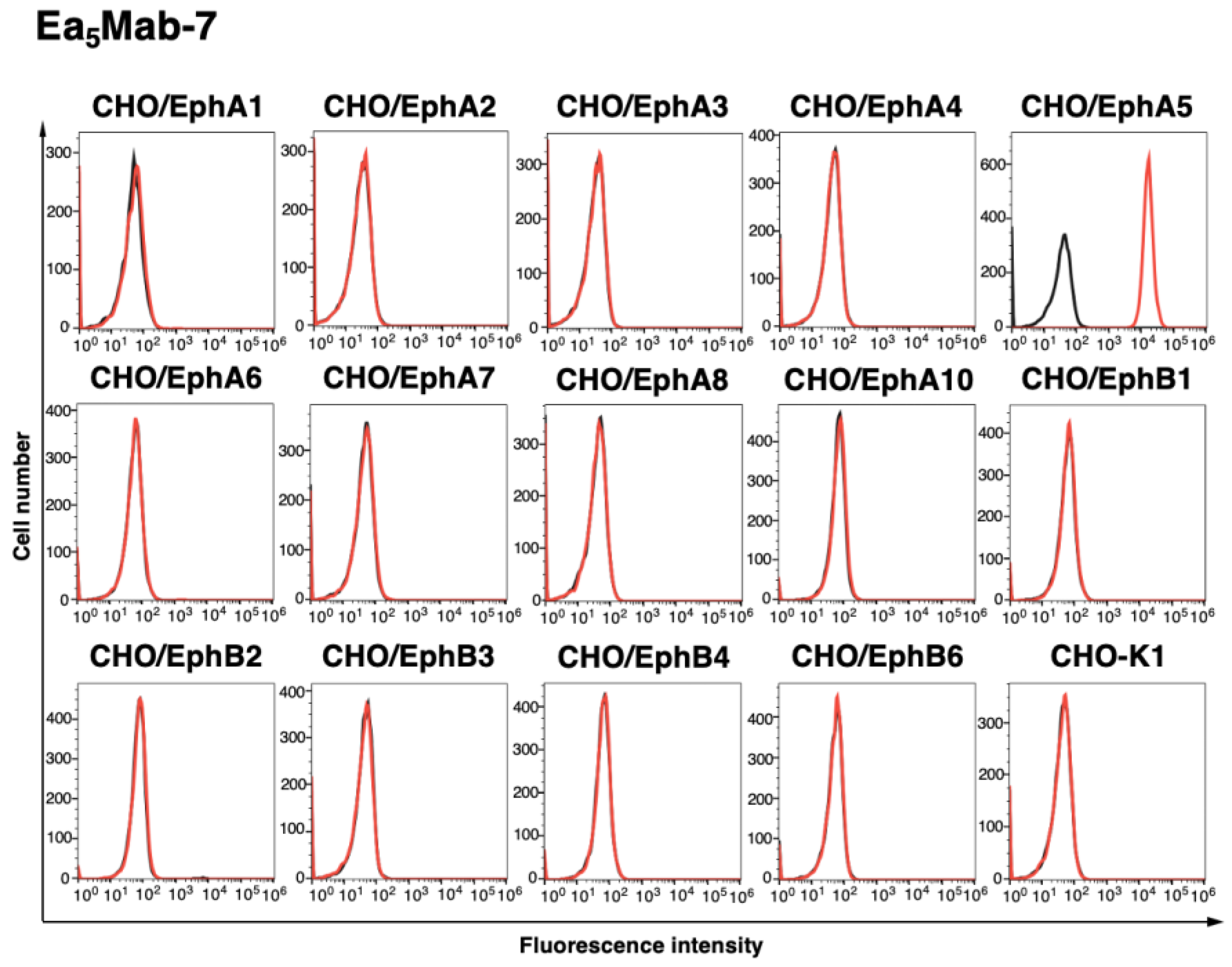
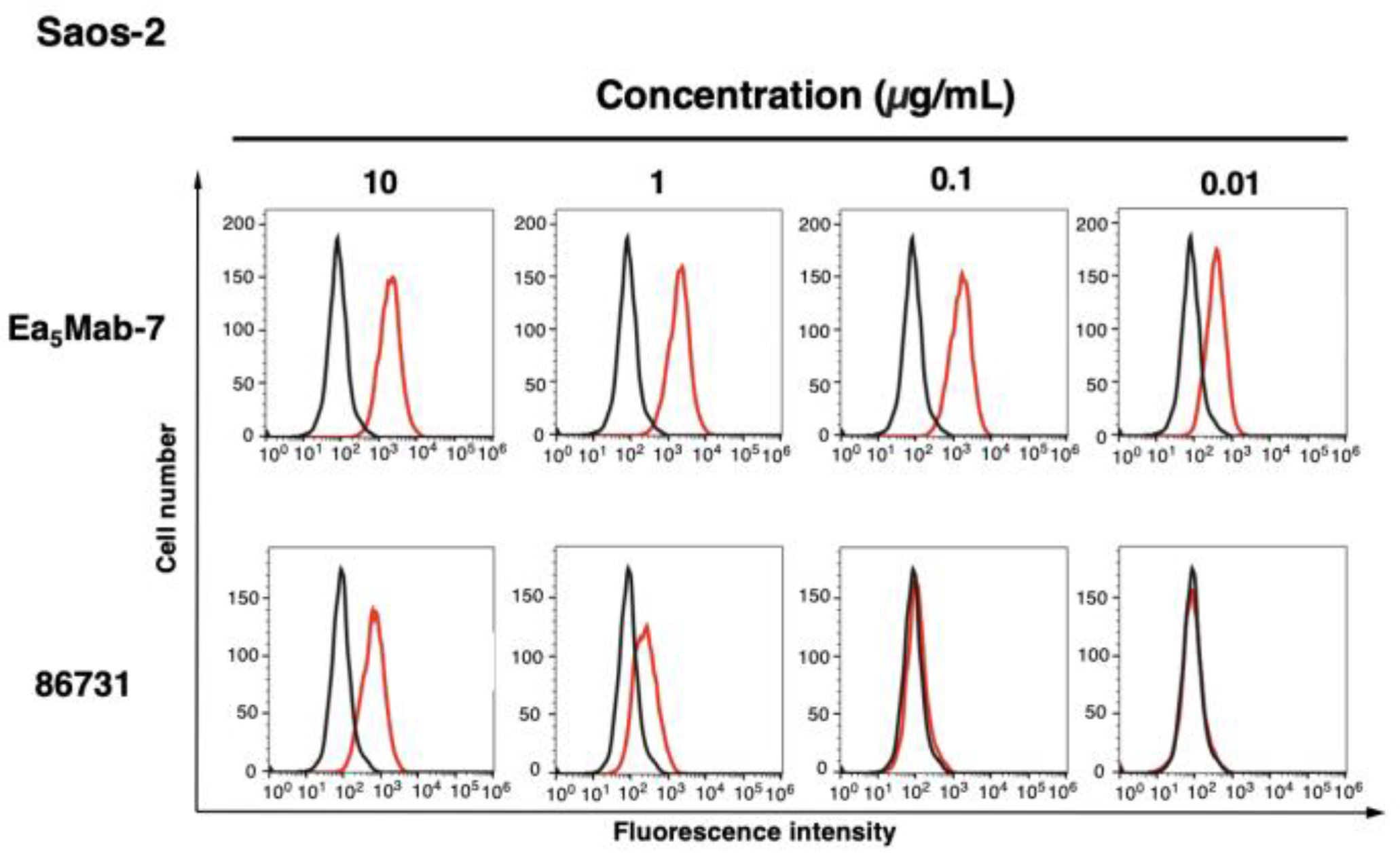
3.2. Investigation of the Reactivity of Anti-EphA5 mAbs Using Flow Cytometry
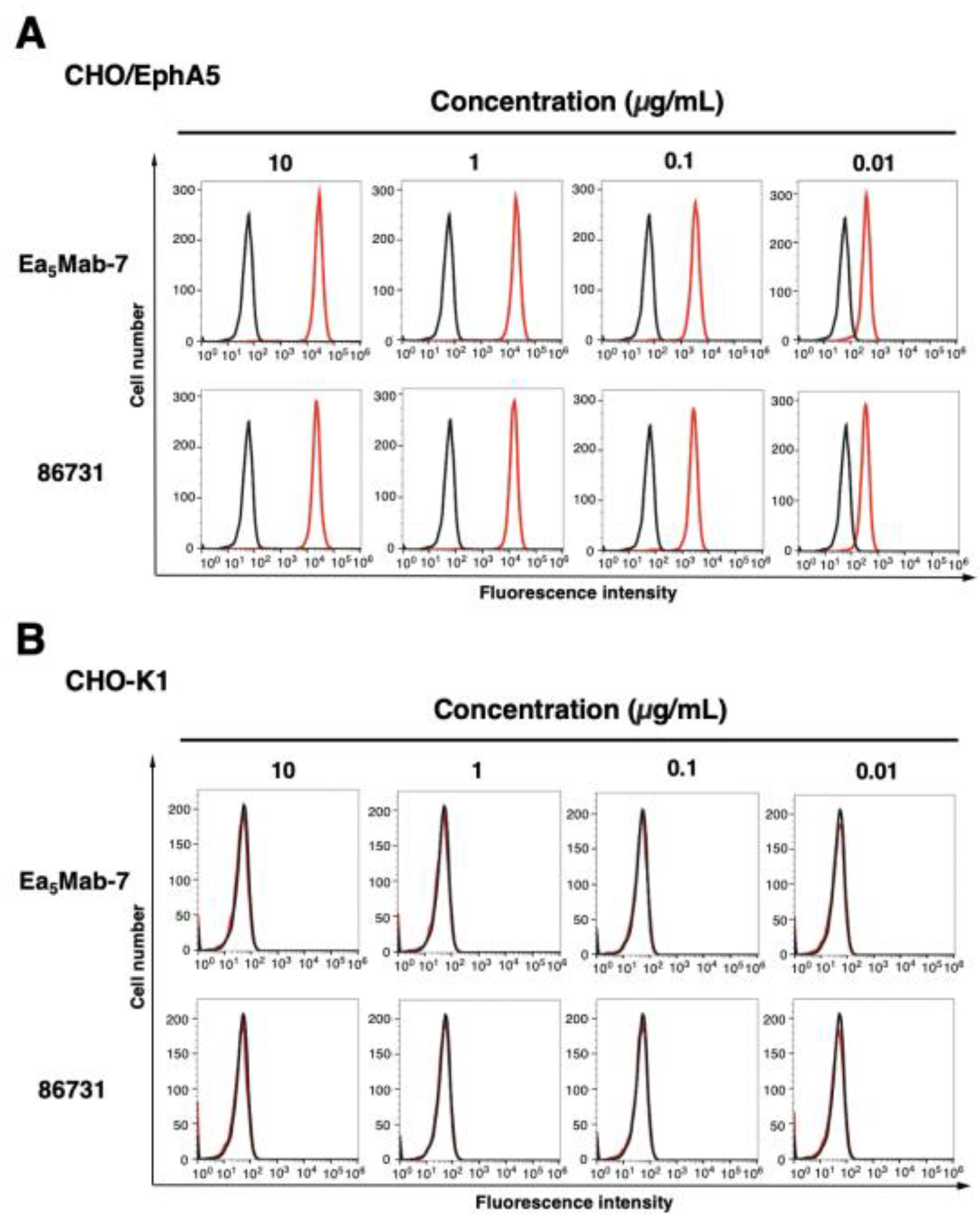
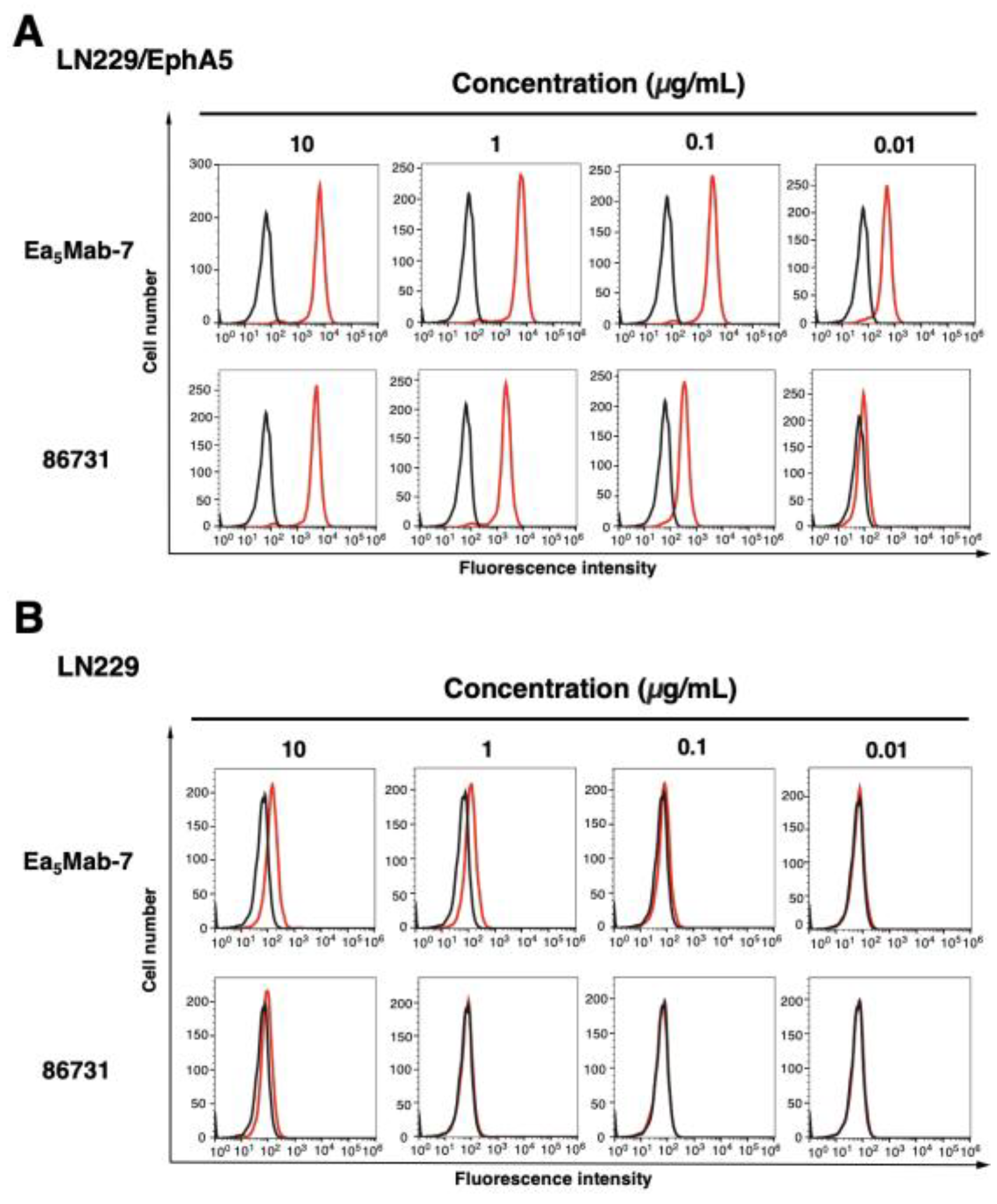
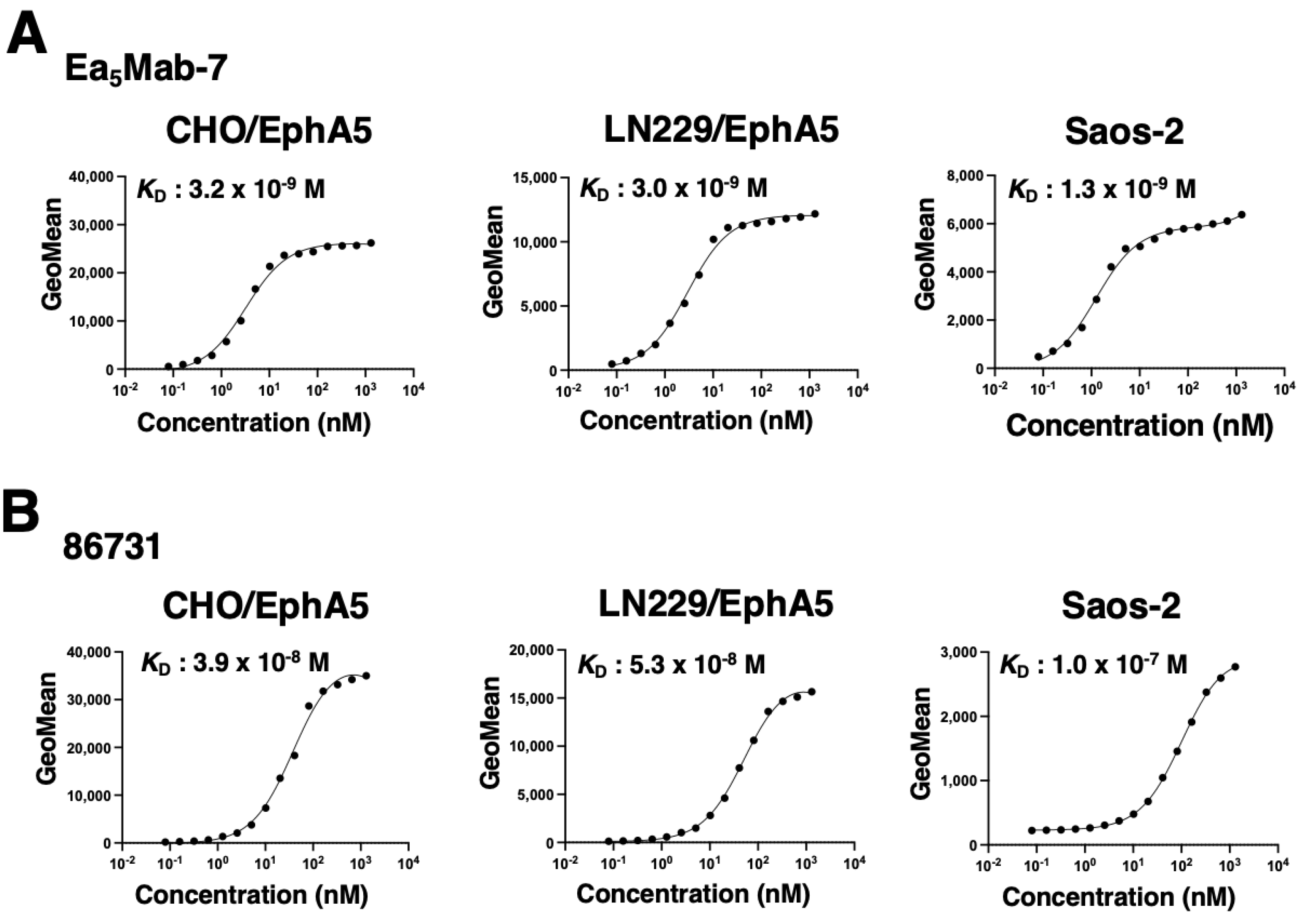
3.3. Determination of the Binding Affinity of Anti-EphA5 mAbs Using Flow Cytometry
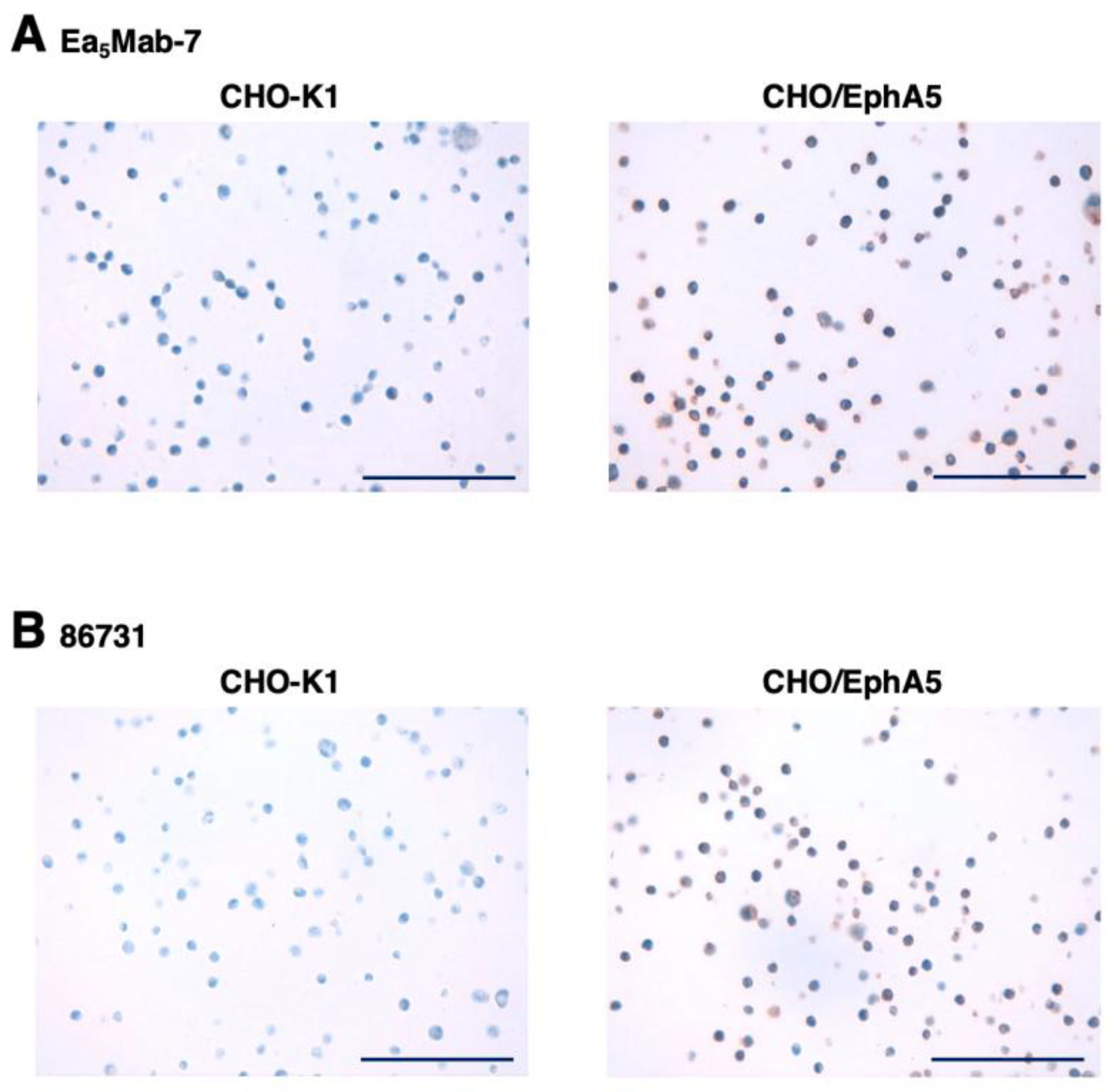
3.4. IHC Using Anti-EphA5 mAbs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pasquale, E.B. Eph receptors and ephrins in cancer progression. Nat Rev Cancer 2024, 24, 5–27. [Google Scholar] [CrossRef] [PubMed]
- Pasquale, E.B. Eph receptor signaling complexes in the plasma membrane. Trends Biochem Sci 2024, 49, 1079–1096. [Google Scholar] [CrossRef] [PubMed]
- Lisabeth, E.M.; Falivelli, G.; Pasquale, E.B. Eph receptor signaling and ephrins. Cold Spring Harb Perspect Biol 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Alford, S.C.; Bazowski, J.; Lorimer, H.; Elowe, S.; Howard, P.L. Tissue transglutaminase clusters soluble A-type ephrins into functionally active high molecular weight oligomers. Exp Cell Res 2007, 313, 4170–4179. [Google Scholar] [CrossRef]
- Wykosky, J.; Palma, E.; Gibo, D.M.; et al. Soluble monomeric EphrinA1 is released from tumor cells and is a functional ligand for the EphA2 receptor. Oncogene 2008, 27, 7260–7273. [Google Scholar] [CrossRef]
- Alford, S.; Watson-Hurthig, A.; Scott, N.; et al. Soluble ephrin a1 is necessary for the growth of HeLa and SK-BR3 cells. Cancer Cell Int 2010, 10, 41. [Google Scholar] [CrossRef]
- Song, Y.; Zhao, X.P.; Song, K.; Shang, Z.J. Ephrin-A1 is up-regulated by hypoxia in cancer cells and promotes angiogenesis of HUVECs through a coordinated cross-talk with eNOS. PLoS One 2013, 8, e74464. [Google Scholar] [CrossRef]
- Arora, S.; Scott, A.M.; Janes, P.W. Eph Receptors in Cancer. Biomedicines 2023, 11. [Google Scholar] [CrossRef]
- Huang, W.; Lin, A.; Luo, P.; et al. EPHA5 mutation predicts the durable clinical benefit of immune checkpoint inhibitors in patients with lung adenocarcinoma. Cancer Gene Ther 2021, 28, 864–874. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, J.; Zhang, W.; et al. EphA5 knockdown enhances the invasion and migration ability of esophageal squamous cell carcinoma via epithelial-mesenchymal transition through activating Wnt/β-catenin pathway. Cancer Cell Int 2020, 20, 20. [Google Scholar] [CrossRef]
- Oh, S.Y.; Jang, G.; Kim, J.; et al. Identification of New Pathogenic Variants of Hereditary Diffuse Gastric Cancer. Cancer Res Treat 2024, 56, 1126–1135. [Google Scholar] [CrossRef]
- Wang, G.; Xu, G.; Fan, Y.; et al. EPHA5 promotes cell proliferation and inhibits apoptosis in Follicular Thyroid Cancer via the STAT3 signaling pathway. Oncogenesis 2025, 14, 12. [Google Scholar] [CrossRef]
- Janes, P.W.; Vail, M.E.; Ernst, M.; Scott, A.M. Eph Receptors in the Immunosuppressive Tumor Microenvironment. Cancer Res 2021, 81, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Shiuan, E.; Chen, J. Eph Receptor Tyrosine Kinases in Tumor Immunity. Cancer Res 2016, 76, 6452–6457. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, H.X.; Lin, W.H.; et al. EPHA7 mutation as a predictive biomarker for immune checkpoint inhibitors in multiple cancers. BMC Med 2021, 19, 26. [Google Scholar] [CrossRef] [PubMed]
- Harly, C.; Joyce, S.P.; Domblides, C.; et al. Human γδ T cell sensing of AMPK-dependent metabolic tumor reprogramming through TCR recognition of EphA2. Sci Immunol 2021, 6. [Google Scholar] [CrossRef]
- Funk, S.D.; Yurdagul, A., Jr.; Albert, P.; et al. EphA2 activation promotes the endothelial cell inflammatory response: a potential role in atherosclerosis. Arterioscler Thromb Vasc Biol 2012, 32, 686–695. [Google Scholar] [CrossRef]
- Zhang, A.; Xing, J.; Xia, T.; et al. EphA2 phosphorylates NLRP3 and inhibits inflammasomes in airway epithelial cells. EMBO Rep 2020, 21, e49666. [Google Scholar] [CrossRef]
- Itai, S.; Yamada, S.; Kaneko, M.K.; et al. Establishment of EMab-134, a Sensitive and Specific Anti-Epidermal Growth Factor Receptor Monoclonal Antibody for Detecting Squamous Cell Carcinoma Cells of the Oral Cavity. Monoclon Antib Immunodiagn Immunother 2017, 36, 272–281. [Google Scholar] [CrossRef]
- Itai, S.; Fujii, Y.; Kaneko, M.K.; et al. H(2)Mab-77 is a Sensitive and Specific Anti-HER2 Monoclonal Antibody Against Breast Cancer. Monoclon Antib Immunodiagn Immunother 2017, 36, 143–148. [Google Scholar] [CrossRef]
- Ubukata, R.; Suzuki, H.; Hirose, M.; et al. Establishment of a highly sensitive and specific anti-EphB2 monoclonal antibody (Eb2Mab-12) for flow cytometry. MI 2025. [Google Scholar] [CrossRef]
- Satofuka, H.; Suzuki, H.; Tanaka, T.; et al. Development of an anti-human EphA2 monoclonal antibody Ea2Mab-7 for multiple applications. Biochemistry and Biophysics Reports 2025, 42, 101998. [Google Scholar] [CrossRef] [PubMed]
- Satofuka, H.; Suzuki, H.; Tanaka, T.; et al. A novel anti-mouse CCR7 monoclonal antibody, C(7)Mab-7, demonstrates high sensitivity in flow cytometry, western blot, and immunohistochemistry. Biochem Biophys Rep 2025, 41, 101948. [Google Scholar]
- Suzuki, H.; Tanaka, T.; Li, G.; et al. Development of a Sensitive Anti-Mouse CCR5 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024, 43, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Ubukata, R.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Development of an anti-CDH15/M-cadherin monoclonal antibody Ca15Mab-1 for flow cytometry, immunoblotting, and immunohistochemistry. Biochemistry and Biophysics Reports 2025, 43, 102138. [Google Scholar] [CrossRef]
- Fujii, Y.; Kaneko, M.K.; Kato, Y. MAP Tag: A Novel Tagging System for Protein Purification and Detection. Monoclon Antib Immunodiagn Immunother 2016, 35, 293–299. [Google Scholar] [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kuno, A.; et al. Inhibition of tumor cell-induced platelet aggregation using a novel anti-podoplanin antibody reacting with its platelet-aggregation-stimulating domain. Biochem Biophys Res Commun 2006, 349, 1301–1307. [Google Scholar] [CrossRef]
- Shoaib, Z.; Fan, T.M.; Irudayaraj, J.M.K. Osteosarcoma mechanobiology and therapeutic targets. Br J Pharmacol 2022, 179, 201–217. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, K.; Hu, J.; Zhang, C. Advances and challenges in the treatment of osteosarcoma. Prog Biophys Mol Biol 2025, 197, 60–74. [Google Scholar] [CrossRef]
- Harper, K.; Sathiadoss, P.; Saifuddin, A.; Sheikh, A. A review of imaging of surface sarcomas of bone. Skeletal Radiol 2021, 50, 9–28. [Google Scholar] [CrossRef]
- Brar, G.S.; Schmidt, A.A.; Willams, L.R.; Wakefield, M.R.; Fang, Y. Osteosarcoma: current insights and advances. Explor Target Antitumor Ther 2025, 6, 1002324. [Google Scholar] [CrossRef]
- Hasegawa, J.; Sue, M.; Yamato, M.; et al. Novel anti-EPHA2 antibody, DS-8895a for cancer treatment. Cancer Biol Ther 2016, 17, 1158–1167. [Google Scholar] [CrossRef]
- Gan, H.K.; Parakh, S.; Lee, F.T.; et al. A phase 1 safety and bioimaging trial of antibody DS-8895a against EphA2 in patients with advanced or metastatic EphA2 positive cancers. Invest New Drugs 2022, 40, 747–755. [Google Scholar] [CrossRef]
- Offenhäuser, C.; Al-Ejeh, F.; Puttick, S.; et al. EphA3 Pay-Loaded Antibody Therapeutics for the Treatment of Glioblastoma. Cancers (Basel) 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Day, B.W.; Stringer, B.W.; Al-Ejeh, F.; et al. EphA3 maintains tumorigenicity and is a therapeutic target in glioblastoma multiforme. Cancer Cell 2013, 23, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Ohishi, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Anti-HER2 Cancer-Specific mAb, H(2)Mab-250-hG(1), Possesses Higher Complement-Dependent Cytotoxicity than Trastuzumab. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. A Cancer-Specific Monoclonal Antibody against HER2 Exerts Antitumor Activities in Human Breast Cancer Xenograft Models. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Guo, W.; Wang, X.; et al. Circulating Exosomal PD-L1 at Initial Diagnosis Predicts Outcome and Survival of Patients with Osteosarcoma. Clin Cancer Res 2023, 29, 659–666. [Google Scholar] [CrossRef]
- Cai, R.; Zhu, H.; Liu, Y.; et al. To be, or not to be: the dilemma of immunotherapy for non-small cell lung cancer harboring various driver mutations. J Cancer Res Clin Oncol 2023, 149, 10027–10040. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



