Submitted:
15 July 2025
Posted:
15 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
3. Oxidative Stress and Neurodegeneration in Alzheimer’s Disease
| Component | Effects of Free Radicals |
| Proteins | - Changes in amino acids - Peptide chain breakage - Protein denaturation - Loss of activity |
| Lipids | - Lipid peroxidation - Alteration of membranes |
| Sugars | - Glucose autoxidation |
| DNA | - DNA breakage - Base mutations - Alteration of gene expression |
4. Oxidative Stress and Neurodegeneration in Alzheimer’s Disease
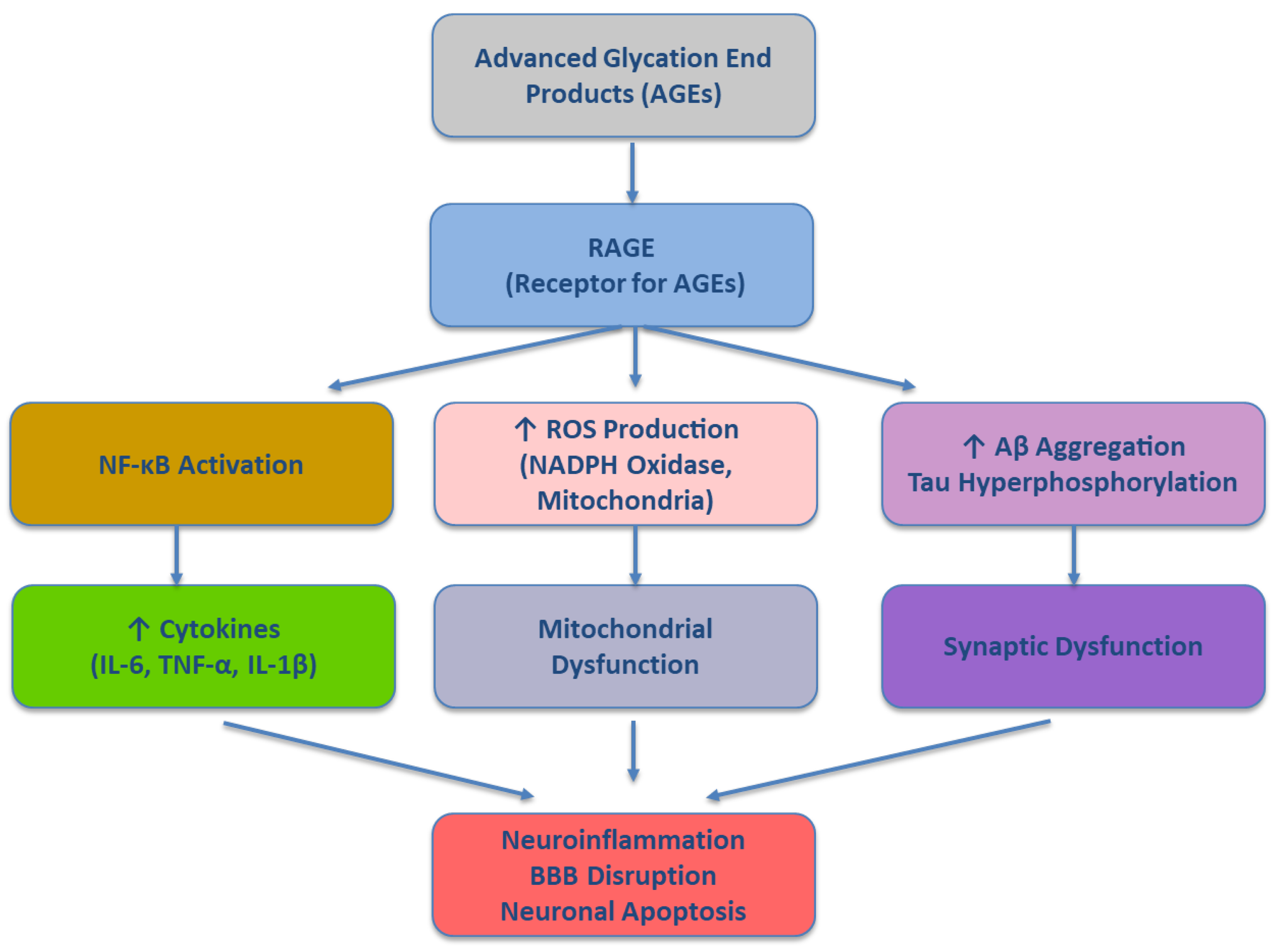
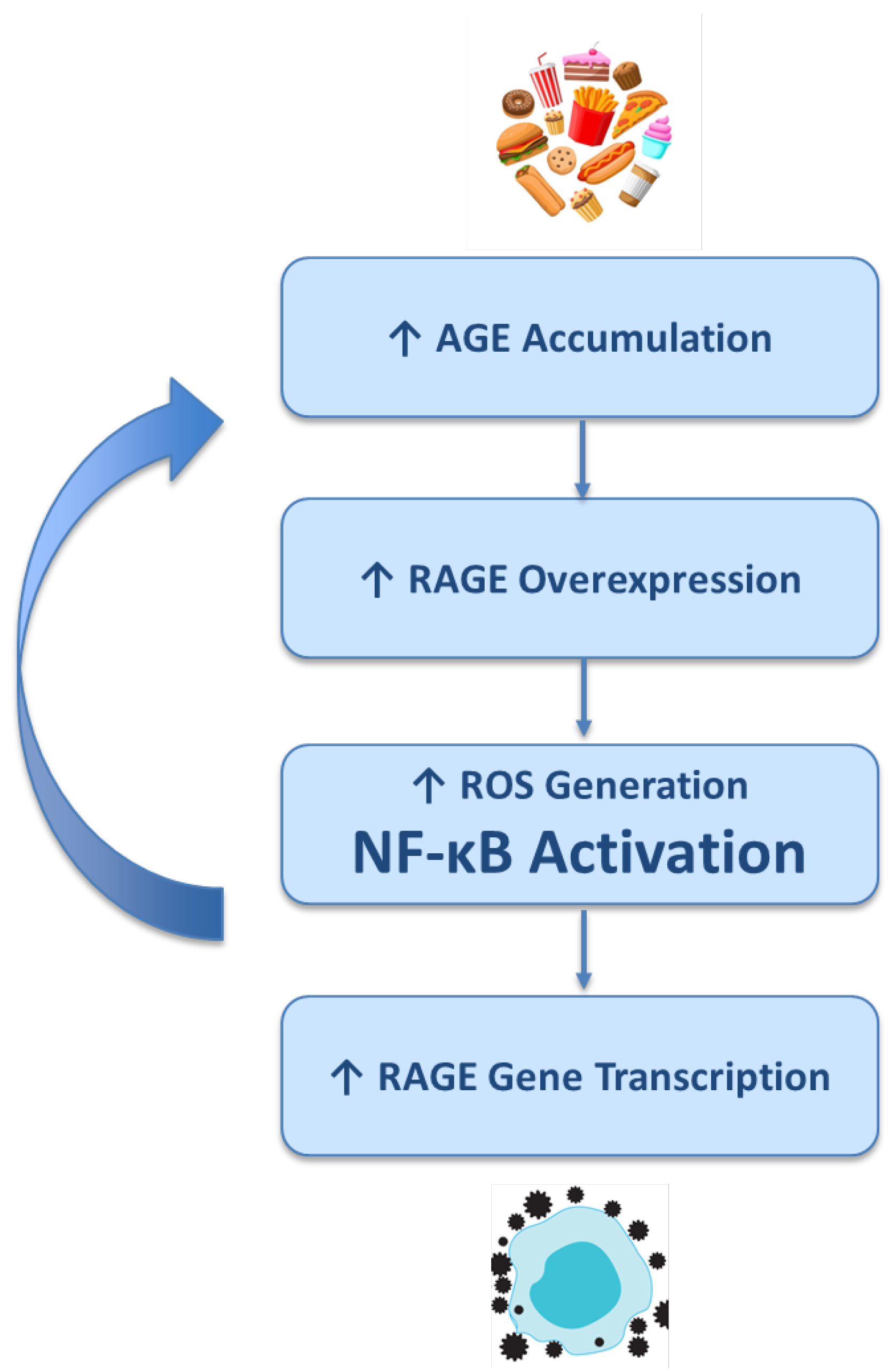
5. The AGE–RAGE Axis as a Central Node in Aging-Related Metabolic and Inflammatory Dysfunction
6. Therapeutic Strategies Targeting AGE Formation, Detoxification, and RAGE Signaling in AD
6.1. Inhibitors of AGE Formation
6.2. Enhancing AGE Detoxification and Clearance
6.3. RAGE Antagonists and Receptor-Targeted Therapies
6.4. Nutritional and Lifestyle Interventions
7. Conclusions
7.1. Open Research Questions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
References
- Wang, S.; Jiang, Y.; Yang, A.; Meng, F.; Zhang, J. The Expanding Burden of Neurodegenerative Diseases: An Unmet Medical and Social Need. Aging Dis 2024, 0-. [CrossRef]
- Kamatham, P.T.; Shukla, R.; Khatri, D.K.; Vora, L.K. Pathogenesis, Diagnostics, and Therapeutics for Alzheimer’s Disease: Breaking the Memory Barrier. Ageing Res Rev 2024, 101, 102481. [CrossRef]
- Mendez, M.F. Early-Onset Alzheimer’s Disease: Nonamnestic Subtypes and Type 2 AD. Arch Med Res 2012, 43, 677–685. [CrossRef]
- Valdez-Gaxiola, C.A.; Rosales-Leycegui, F.; Gaxiola-Rubio, A.; Moreno-Ortiz, J.M.; Figuera, L.E.; Valdez-Gaxiola, C.A.; Rosales-Leycegui, F.; Gaxiola-Rubio, A.; Moreno-Ortiz, J.M.; Figuera, L.E. Early- and Late-Onset Alzheimer’s Disease: Two Sides of the Same Coin? Diseases 2024, Vol. 12, Page 110 2024, 12, 110. [CrossRef]
- Mecocci, P.; Baroni, M.; Senin, U.; Boccardi, V. Brain Aging and Late-Onset Alzheimer’s Disease: A Matter of Increased Amyloid or Reduced Energy? Journal of Alzheimer’s Disease 2018, 64, S397–S404. [CrossRef]
- Dominguez-Gortaire, J.; Ruiz, A.; Porto-Pazos, A.B.; Rodriguez-Yanez, S.; Cedron, F. Alzheimer’s Disease: Exploring Pathophysiological Hypotheses and the Role of Machine Learning in Drug Discovery. International Journal of Molecular Sciences 2025, Vol. 26, Page 1004 2025, 26, 1004. [CrossRef]
- Uddin, M.S.; Kabir, M.T.; Rahman, M.S.; Behl, T.; Jeandet, P.; Ashraf, G.M.; Najda, A.; Bin-Jumah, M.N.; El-Seedi, H.R.; Abdel-Daim, M.M. Revisiting the Amyloid Cascade Hypothesis: From Anti-Aβ Therapeutics to Auspicious New Ways for Alzheimer’s Disease. Int J Mol Sci 2020, 21, 5858. [CrossRef]
- Kazemeini, S.; Nadeem-Tariq, A.; Shih, R.; Rafanan, J.; Ghani, N.; Vida, T.A. From Plaques to Pathways in Alzheimer’s Disease: The Mitochondrial-Neurovascular-Metabolic Hypothesis. Int J Mol Sci 2024, 25, 11720. [CrossRef]
- Botella Lucena, P.; Heneka, M.T. Inflammatory Aspects of Alzheimer’s Disease. Acta Neuropathol 2024, 148. [CrossRef]
- Kothandan, D.; Singh, D.S.; Yerrakula, G.; D, B.; N, P.; B, V.S.S.; A, R.; VG, S.R.; S, K.; M, J. Advanced Glycation End Products-Induced Alzheimer’s Disease and Its Novel Therapeutic Approaches: A Comprehensive Review. Cureus 2024, 16, e61373. [CrossRef]
- Fornai, F.; Li, W.; Chen, Q.; Peng, C.; Yang, D.; Liu, S.; Lv, Y.; Jiang, L.; Xu, S.; Huang, L. Roles of the Receptor for Advanced Glycation End Products and Its Ligands in the Pathogenesis of Alzheimer’s Disease. 2025. [CrossRef]
- Vitorakis, N.; Piperi, C. Pivotal Role of AGE-RAGE Axis in Brain Aging with Current Interventions. Ageing Res Rev 2024, 100, 102429. [CrossRef]
- Chen, J.; Mooldijk, S.S.; Licher, S.; Waqaz, K.; Ikram, M.K.; Uitterlinden, A.G.; Zillikens, M.C. Advanced Glycation End Products, Their Receptor and the Risk of Dementia in the General Population: A Prospective Cohort Study. Alzheimer’s & Dementia 2020, 16, e043005. [CrossRef]
- Chen, J.; Mooldijk, S.S.; Licher, S.; Waqas, K.; Ikram, M.K.; Uitterlinden, A.G.; Zillikens, M.C.; Ikram, M.A. Assessment of Advanced Glycation End Products and Receptors and the Risk of Dementia. JAMA Netw Open 2021, 4. [CrossRef]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr Biol 2014, 24, R453. [CrossRef]
- Jomova, K.; Raptova, R.; Alomar, S.Y.; Alwasel, S.H.; Nepovimova, E.; Kuca, K.; Valko, M. Reactive Oxygen Species, Toxicity, Oxidative Stress, and Antioxidants: Chronic Diseases and Aging. Arch Toxicol 2023, 97, 2499. [CrossRef]
- Martemucci, G.; Costagliola, C.; Mariano, M.; D’andrea, L.; Napolitano, P.; D’Alessandro, A.G. Free Radical Properties, Source and Targets, Antioxidant Consumption and Health. Oxygen 2022, Vol. 2, Pages 48-78 2022, 2, 48–78. [CrossRef]
- Timalsina, D.R.; Abichandani, L.; Ambad, R. A Review Article on Oxidative Stress Markers F2-Isoprostanes and Presenilin-1 in Alzheimer’s Disease. J Pharm Bioallied Sci 2025, 17, S109–S112. [CrossRef]
- Kim, S.; Jung, U.J.; Kim, S.R. Role of Oxidative Stress in Blood–Brain Barrier Disruption and Neurodegenerative Diseases. Antioxidants 2024, Vol. 13, Page 1462 2024, 13, 1462. [CrossRef]
- Olufunmilayo, E.O.; Gerke-Duncan, M.B.; Holsinger, R.M.D. Oxidative Stress and Antioxidants in Neurodegenerative Disorders. Antioxidants 2023, Vol. 12, Page 517 2023, 12, 517. [CrossRef]
- Li, J.; Wuliji, O.; Li, W.; Jiang, Z.G.; Ghanbari, H.A. Oxidative Stress and Neurodegenerative Disorders. Int J Mol Sci 2013, 14, 24438–24475. [CrossRef]
- Yoritaka, A.; Hattori, N.; Uchida, K.; Tanaka, M.; Stadtman, E.R.; Mizuno, Y. Immunohistochemical Detection of 4-Hydroxynonenal Protein Adducts in Parkinson Disease. Proc Natl Acad Sci U S A 1996, 93, 2696–2701. [CrossRef]
- Gabbita, S.P.; Lovell, M.A.; Markesbery, W.R. Increased Nuclear DNA Oxidation in the Brain in Alzheimer’ s Disease. J Neurochem 1998, 71, 2034–2040. [CrossRef]
- Alam, Z.I.; Jenner, A.; Daniel, S.E.; Lees, A.J.; Cairns, N.; Marsden, C.D.; Jenner, P.; Halliwell, B. Oxidative DNA Damage in the Parkinsonian Brain: An Apparent Selective Increase in 8-Hydroxyguanine Levels in Substantia Nigra. J Neurochem 1997, 69, 1196–1203. [CrossRef]
- Rajmohan, R.; Reddy, P.H. Amyloid Beta and Phosphorylated Tau Accumulations Cause Abnormalities at Synapses of Alzheimer’s Disease Neurons. J Alzheimers Dis 2017, 57, 975. [CrossRef]
- Roy, R.G.; Mandal, P.K.; Maroon, J.C. Oxidative Stress Occurs Prior to Amyloid Aβ Plaque Formation and Tau Phosphorylation in Alzheimer’s Disease: Role of Glutathione and Metal Ions. ACS Chem Neurosci 2023, 14, 2944–2954. [CrossRef]
- Twarda-clapa, A.; Olczak, A.; Białkowska, A.M.; Koziołkiewicz, M. Advanced Glycation End-Products (AGEs): Formation, Chemistry, Classification, Receptors, and Diseases Related to AGEs. Cells 2022, 11, 1312. [CrossRef]
- Moldogazieva, N.T.; Mokhosoev, I.M.; Mel’Nikova, T.I.; Porozov, Y.B.; Terentiev, A.A. Oxidative Stress and Advanced Lipoxidation and Glycation End Products (ALEs and AGEs) in Aging and Age-Related Diseases. Oxid Med Cell Longev 2019, 2019. [CrossRef]
- Prasad, K. AGE–RAGE Stress: A Changing Landscape in Pathology and Treatment of Alzheimer’s Disease. Mol Cell Biochem 2019, 459, 95–112. [CrossRef]
- Ballard, C.; Gauthier, S.; Corbett, A.; Brayne, C.; Aarsland, D.; Jones, E. Alzheimer’s Disease. The Lancet 2011, 377, 1019–1031. [CrossRef]
- Cai, Z.; Liu, N.; Wang, C.; Qin, B.; Zhou, Y.; Xiao, M.; Chang, L.; Yan, L.J.; Zhao, B. Role of RAGE in Alzheimer’s Disease. Cell Mol Neurobiol 2016, 36, 483–495. [CrossRef]
- Baek, S.H.; Hong, S.; Kim, E.; Park, S.; Lee, M.; Park, J.; Cho, Y.; Yoon, H.; Kim, D.; Yun, Y.; et al. A Novel RAGE Modulator Induces Soluble RAGE to Reduce BACE1 Expression in Alzheimer’s Disease. Advanced Science 2025, 12, 2407812. [CrossRef]
- Fang, F.; Lue, L.-F.; Yan, S.; Xu, H.; Luddy, J.S.; Chen, D.; Walker, D.G.; Stern, D.M.; Yan, S.; Schmidt, A.M.; et al. RAGE-Dependent Signaling in Microglia Contributes to Neuroinflammation, Aβ Accumulation, and Impaired Learning/Memory in a Mouse Model of Alzheimer’s Disease. The FASEB Journal 2010, 24, 1043. [CrossRef]
- DaRocha-Souto, B.; Coma, M.; Pérez-Nievas, B.G.; Scotton, T.C.; Siao, M.; Sánchez-Ferrer, P.; Hashimoto, T.; Fan, Z.; Hudry, E.; Barroeta, I.; et al. Activation of Glycogen Synthase Kinase-3 Beta Mediates β-Amyloid Induced Neuritic Damage in Alzheimer’s Disease. Neurobiol Dis 2011, 45, 425. [CrossRef]
- D’Alessandro, M.C.B.; Kanaan, S.; Geller, M.; Praticò, D.; Daher, J.P.L. Mitochondrial Dysfunction in Alzheimer’s Disease. Ageing Res Rev 2025, 107, 102713. [CrossRef]
- Kierdorf, K.; Fritz, G. RAGE Regulation and Signaling in Inflammation and Beyond. J Leukoc Biol 2013, 94, 55–68. [CrossRef]
- Takata, F.; Nakagawa, S.; Matsumoto, J.; Dohgu, S. Blood-Brain Barrier Dysfunction Amplifies the Development of Neuroinflammation: Understanding of Cellular Events in Brain Microvascular Endothelial Cells for Prevention and Treatment of BBB Dysfunction. Front Cell Neurosci 2021, 15, 661838. [CrossRef]
- Koerich, S.; Parreira, G.M.; Almeida, D.L. de; Vieira, R.P.; Oliveira, A.C.P. de Receptors for Advanced Glycation End Products (RAGE): Promising Targets Aiming at the Treatment of Neurodegenerative Conditions. Curr Neuropharmacol 2023, 21, 219. [CrossRef]
- Fritz, G. RAGE: A Single Receptor Fits Multiple Ligands. Trends Biochem Sci 2011, 36, 625–632. [CrossRef]
- Dong, H.; Zhang, Y.; Huang, Y.; Deng, H. Pathophysiology of RAGE in Inflammatory Diseases. Front Immunol 2022, 13, 931473. [CrossRef]
- Zhou, M.; Zhang, Y.; Shi, L.; Li, L.; Zhang, D.; Gong, Z.; Wu, Q. Activation and Modulation of the AGEs-RAGE Axis: Implications for Inflammatory Pathologies and Therapeutic Interventions – A Review. Pharmacol Res 2024, 206, 107282. [CrossRef]
- Affuso, F.; Micillo, F.; Fazio, S. Insulin Resistance, a Risk Factor for Alzheimer’s Disease: Pathological Mechanisms and a New Proposal for a Preventive Therapeutic Approach. Biomedicines 2024, Vol. 12, Page 1888 2024, 12, 1888. [CrossRef]
- Lemche, E.; Killick, R.; Mitchell, J.; Caton, P.W.; Choudhary, P.; Howard, J.K. Molecular Mechanisms Linking Type 2 Diabetes Mellitus and Late-Onset Alzheimer’s Disease: A Systematic Review and Qualitative Meta-Analysis. Neurobiol Dis 2024, 196, 106485. [CrossRef]
- Fang, F.; Lue, L.-F.; Yan, S.; Xu, H.; Luddy, J.S.; Chen, D.; Walker, D.G.; Stern, D.M.; Yan, S.; Schmidt, A.M.; et al. RAGE-Dependent Signaling in Microglia Contributes to Neuroinflammation, Aβ Accumulation, and Impaired Learning/Memory in a Mouse Model of Alzheimer’s Disease. The FASEB Journal 2010, 24, 1043. [CrossRef]
- Valiukas, Z.; Tangalakis, K.; Apostolopoulos, V.; Feehan, J. Microglial Activation States and Their Implications for Alzheimer’s Disease. J Prev Alzheimers Dis 2025, 12, 100013. [CrossRef]
- Wendimu, M.Y.; Hooks, S.B. Microglia Phenotypes in Aging and Neurodegenerative Diseases. Cells 2022, 11, 2091. [CrossRef]
- Wan, W.; Chen, H.; Li, Y. The Potential Mechanisms of Aβ-Receptor for Advanced Glycation End-Products Interaction Disrupting Tight Junctions of the Blood-Brain Barrier in Alzheimer’s Disease. International Journal of Neuroscience 2014, 124, 75–81. [CrossRef]
- Takata, F.; Nakagawa, S.; Matsumoto, J.; Dohgu, S. Blood-Brain Barrier Dysfunction Amplifies the Development of Neuroinflammation: Understanding of Cellular Events in Brain Microvascular Endothelial Cells for Prevention and Treatment of BBB Dysfunction. Front Cell Neurosci 2021, 15, 661838. [CrossRef]
- Thornalley, P.J. Use of Aminoguanidine (Pimagedine) to Prevent the Formation of Advanced Glycation Endproducts. Arch Biochem Biophys 2003, 419, 31–40. [CrossRef]
- Ooi, H.; Nasu, R.; Furukawa, A.; Takeuchi, M.; Koriyama, Y. Pyridoxamine and Aminoguanidine Attenuate the Abnormal Aggregation of β-Tubulin and Suppression of Neurite Outgrowth by Glyceraldehyde-Derived Toxic Advanced Glycation End-Products. Front Pharmacol 2022, 13, 921611. [CrossRef]
- Berends, E.; Pencheva, M.G.; van de Waarenburg, M.P.H.; Scheijen, J.L.J.M.; Hermes, D.J.H.P.; Wouters, K.; van Oostenbrugge, R.J.; Foulquier, S.; Schalkwijk, C.G. Glyoxalase 1 Overexpression Improves Neurovascular Coupling and Limits Development of Mild Cognitive Impairment in a Mouse Model of Type 1 Diabetes. Journal of Physiology 2024, 602. [CrossRef]
- Webster, J.; Urban, C.; Berbaum, K.; Loske, C.; Alpar, A.; Gärtner, U.; De Arriba, S.G.; Arendt, T.; Münch, G. The Carbonyl Scavengers Aminoguanidine and Tenilsetam Protect against the Neurotoxic Effects of Methylglyoxal. Neurotox Res 2005, 7, 95–101. [CrossRef]
- Burstein, A.H.; Sabbagh, M.; Andrews, R.; Valcarce, C.; Dunn, I.; Altstiel, L. Development of Azeliragon, an Oral Small Molecule Antagonist of the Receptor for Advanced Glycation Endproducts, for the Potential Slowing of Loss of Cognition in Mild Alzheimer’s Disease. J Prev Alzheimers Dis 2018, 5, 1–6. [CrossRef]
- Deane, R.; Singh, I.; Sagare, A.P.; Bell, R.D.; Ross, N.T.; LaRue, B.; Love, R.; Perry, S.; Paquette, N.; Deane, R.J.; et al. A Multimodal RAGE-Specific Inhibitor Reduces Amyloid β-Mediated Brain Disorder in a Mouse Model of Alzheimer Disease. Journal of Clinical Investigation 2012, 122, 1377–1392. [CrossRef]
- Crunkhorn, S. Neurodegenerative Disease: Taming the RAGE of Alzheimer’s Disease. Nat Rev Drug Discov 2012, 11, 351. [CrossRef]
- Erusalimsky, J.D. The Use of the Soluble Receptor for Advanced Glycation-End Products (SRAGE) as a Potential Biomarker of Disease Risk and Adverse Outcomes. Redox Biol 2021, 42, 101958. [CrossRef]
- Uribarri, J.; Woodruff, S.; Goodman, S.; Cai, W.; Chen, X.; Pyzik, R.; Yong, A.; Striker, G.E.; Vlassara, H. Advanced Glycation End Products in Foods and a Practical Guide to Their Reduction in the Diet. J Am Diet Assoc 2010, 110, 911. [CrossRef]
- Garay-Sevilla, M.E.; Rojas, A.; Portero-Otin, M.; Uribarri, J. Dietary AGEs as Exogenous Boosters of Inflammation. Nutrients 2021, Vol. 13, Page 2802 2021, 13, 2802. [CrossRef]
- J, W.; E, V.; A, D.; S, H.; J, V.; J, V.; R, V.; M, D.; B, V.; M, F.; et al. Cooking Methods Affect Advanced Glycation End Products and Lipid Profiles: A Randomized Cross-over Study in Healthy Subjects. Cell Rep Med 2025, 6. [CrossRef]
- González, I.; Morales, M.A.; Rojas, A. Polyphenols and AGEs/RAGE Axis. Trends and Challenges. Food Research International 2020, 129. [CrossRef]
- Aatif, M. Current Understanding of Polyphenols to Enhance Bioavailability for Better Therapies. Biomedicines 2023, 11, 2078. [CrossRef]
- Brimson, J.M.; Prasanth, M.I.; Malar, D.S.; Thitilertdecha, P.; Kabra, A.; Tencomnao, T.; Prasansuklab, A. Plant Polyphenols for Aging Health: Implication from Their Autophagy Modulating Properties in Age-Associated Diseases. Pharmaceuticals 2021, Vol. 14, Page 982 2021, 14, 982. [CrossRef]
- Małkowska, P. Positive Effects of Physical Activity on Insulin Signaling. Current Issues in Molecular Biology 2024, Vol. 46, Pages 5467-5487 2024, 46, 5467–5487. [CrossRef]
- Clemente-Suárez, V.J.; Rubio-Zarapuz, A.; Belinchón-deMiguel, P.; Beltrán-Velasco, A.I.; Martín-Rodríguez, A.; Tornero-Aguilera, J.F. Impact of Physical Activity on Cellular Metabolism Across Both Neurodegenerative and General Neurological Conditions: A Narrative Review. Cells 2024, 13, 1940. [CrossRef]
| Feature | Early-Onset AD (EOAD) | Late-Onset AD (LOAD) |
| Age of onset | <65 years | ≥65 years |
| Genetic associations | APP, PSEN1, PSEN2 mutations | APOE ε4 allele, polygenic risk loci |
| Pathogenesis | Dominant amyloidogenic pathway | Multifactorial (metabolic, vascular, oxidative) |
| Oxidative stress involvement | Less prominent | Highly implicated |
| Frequency | ~5–10% of AD cases | ~90–95% of AD cases |
| Strategy | Mechanism | Examples | Status |
| Inhibition of AGE formation | Carbonyl scavengers, Maillard blockers | Aminoguanidine, Pyridoxamine | Preclinical |
| AGE cross-link breakers | Disruption of protein–AGE bonds | ALT-711 (Alagebrium) | Experimental |
| Enhancement of detoxification | Upregulation of GLO1, antioxidant boosting | Sulforaphane, NAC, GSH | Investigational |
| RAGE antagonism | Blocking AGE–RAGE interaction | Azeliragon, FPS-ZM1, sRAGE | Phase II/III |
| Lifestyle modification | Reduction of exogenous AGEs | Diet, polyphenols, exercise | Observational |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




