Submitted:
04 July 2025
Posted:
07 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mitochondrial Mechanisms in Muscle Aging and Disease
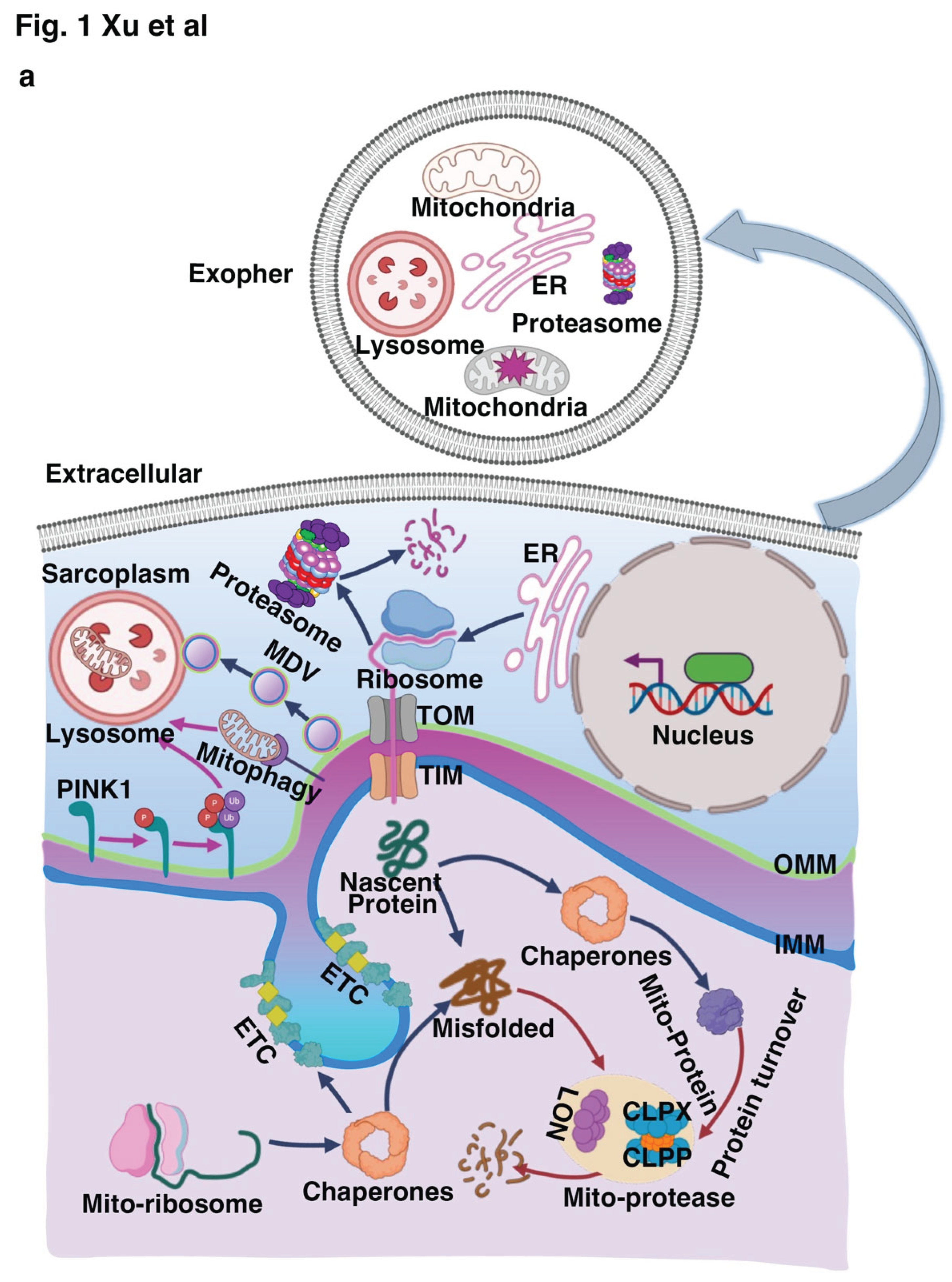
2.1. Mitochondrial Quality Control (MQC) System
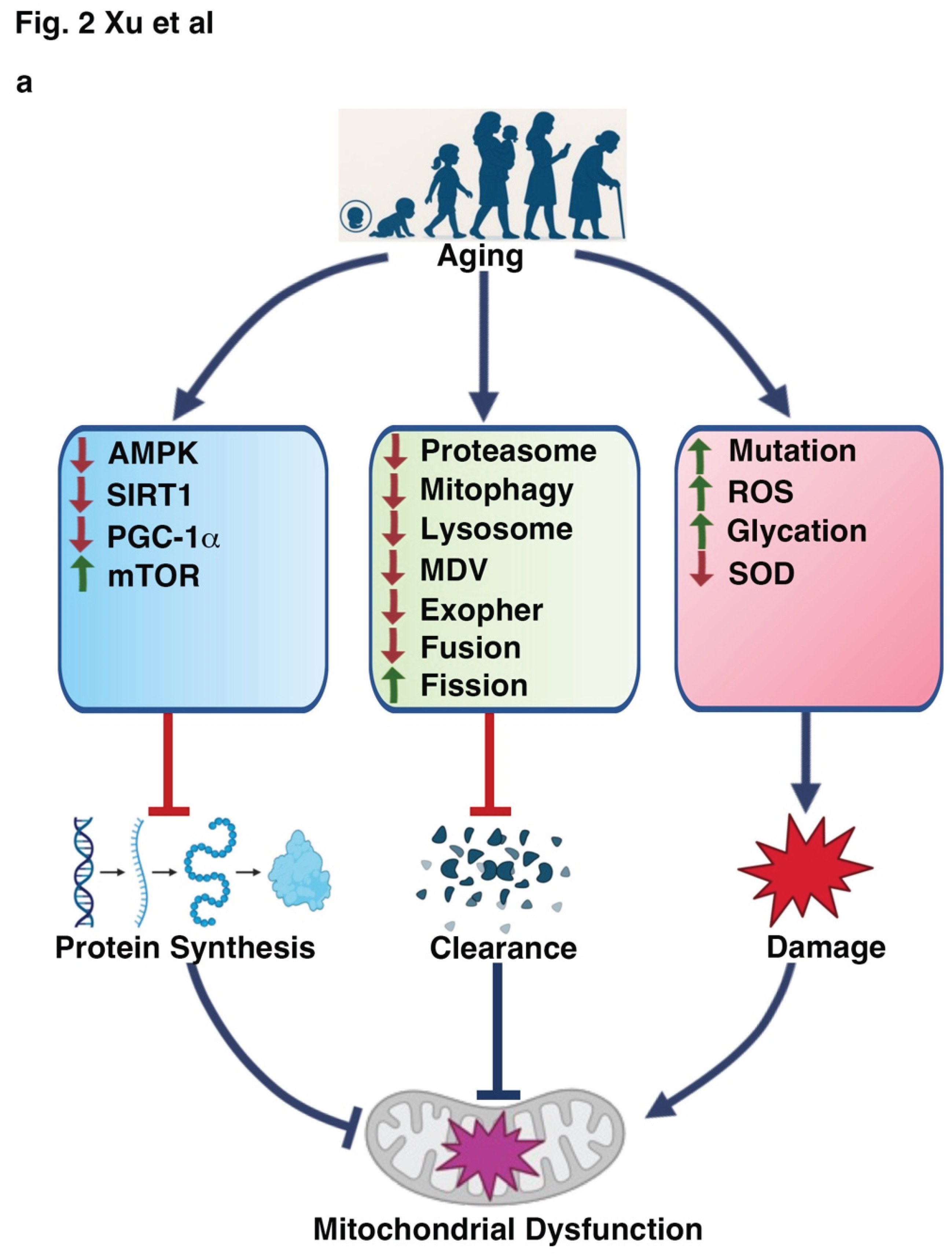
2.2. Mitochondrial Alterations Induced by Aging
2.2.1. Reduced Mitochondrial Biogenesis and Content
2.2.2. Disrupted Mitochondrial Dynamics:
2.2.3. Increased Oxidative Stress
2.2.4. Altered Mitochondrial Unfolded Protein Response (UPRmt)
3. Non-Mitochondrial Mechanisms in Muscle Aging and Disease
3.1. Muscle Stem Cell Dysfunction
3.2. Neuromuscular Junction Dysfunction
3.3. Inflammation and Immune Cell Infiltration
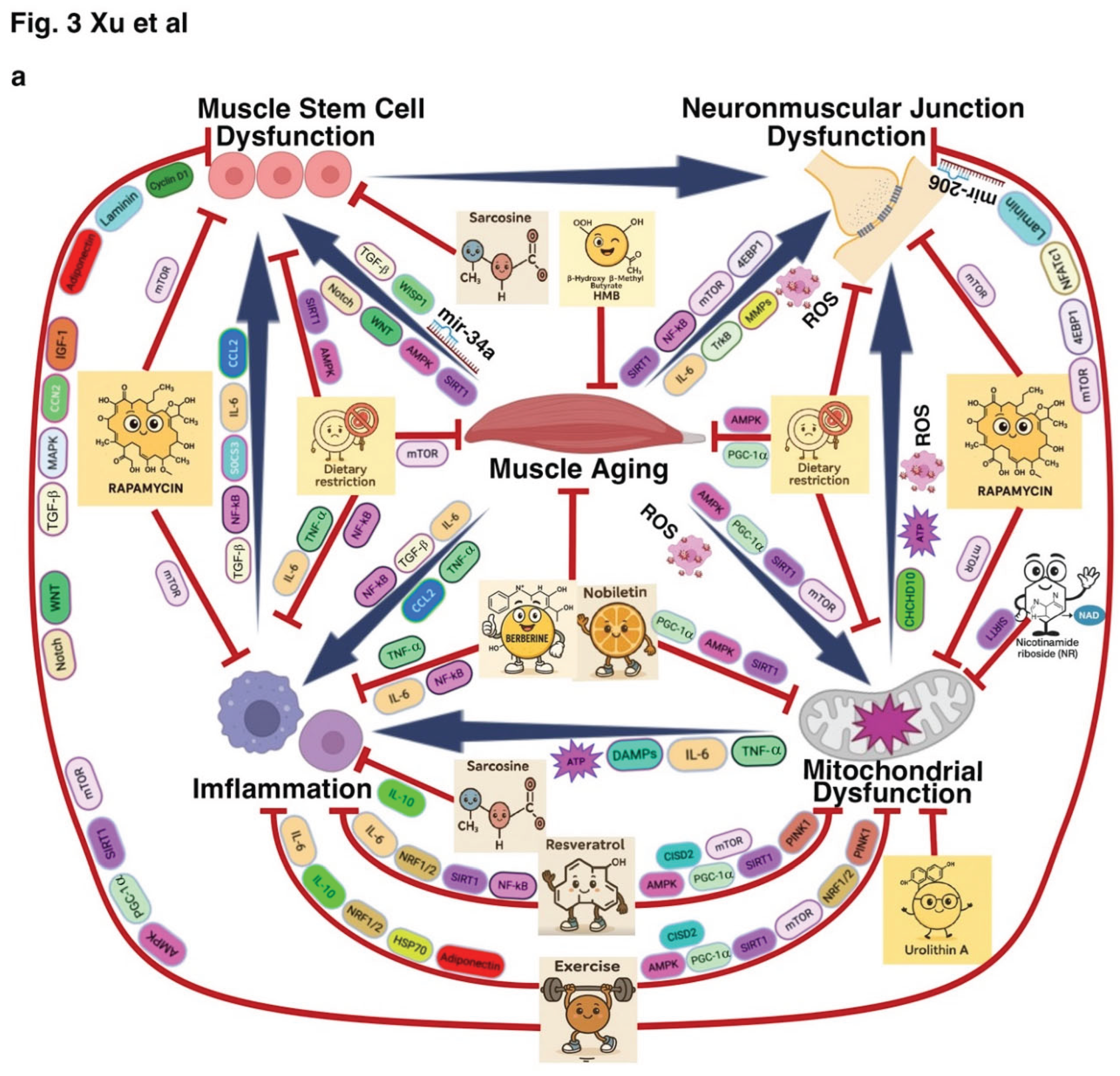
4. Therapeutic Strategies Targeting Muscle Aging and Muscle-Related Diseases
4.1. Lifestyle Interventions: Exercise and Dietary Restriction
4.2. Pharmacological Strategies Targeting Metab[120,189–228olic and Proteostatic Pathways
4.3. Biological Interventions Targeting Stem Cell Function and Neuromuscular Integrity
Conclusion and Future Directions
Author Contributions
References
- Mukund K, Subramaniam S. Skeletal muscle: A review of molecular structure and function, in health and disease. Wiley Interdiscip Rev Syst Biol Med. 2020;12(1):e1462. [CrossRef]
- Tieland M, Trouwborst I, Clark BC. Skeletal muscle performance and ageing. J Cachexia Sarcopenia Muscle. 2018;9(1):3-19.
- Frontera WR, Ochala J. Skeletal muscle: a brief review of structure and function. Calcif Tissue Int. 2015;96(3):183-95. [CrossRef]
- Biolo G, Cederholm T, Muscaritoli M. Muscle contractile and metabolic dysfunction is a common feature of sarcopenia of aging and chronic diseases: From sarcopenic obesity to cachexia. Clin Nutr. 2014;33(5):737-48. [CrossRef]
- Kedlian VR, Wang Y, Liu T, Chen X, Bolt L, Tudor C, et al. Human skeletal muscle aging atlas. Nat Aging. 2024;4(5):727-44. [CrossRef]
- Dao T, Green AE, Kim YA, Bae SJ, Ha KT, Gariani K, et al. Sarcopenia and Muscle Aging: A Brief Overview. Endocrinol Metab (Seoul). 2020;35(4):716-32. [CrossRef]
- Picca A, Calvani R, Bossola M, Allocca E, Menghi A, Pesce V, et al. Update on mitochondria and muscle aging: all wrong roads lead to sarcopenia. Biological Chemistry. 2018;399(5):421-36. [CrossRef]
- Demontis F, Piccirillo R, Goldberg AL, Perrimon N. Mechanisms of skeletal muscle aging: insights from Drosophila and mammalian models. Dis Model Mech. 2013;6(6):1339-52. [CrossRef]
- Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186(2):243-78. [CrossRef]
- Leduc-Gaudet JP, Hussain SNA, Barreiro E, Gouspillou G. Mitochondrial Dynamics and Mitophagy in Skeletal Muscle Health and Aging. International Journal of Molecular Sciences. 2021;22(15). [CrossRef]
- Hood DA, Memme JM, Oliveira AN, Triolo M. Maintenance of Skeletal Muscle Mitochondria in Health, Exercise, and Aging. Annu Rev Physiol. 2019;81:19-41. [CrossRef]
- Coen PM, Musci RV, Hinkley JM, Miller BF. Mitochondria as a Target for Mitigating Sarcopenia. Front Physiol. 2019;9. [CrossRef]
- Alway SE, Mohamed JS, Myers MJ. Mitochondria Initiate and Regulate Sarcopenia. Exerc Sport Sci Rev. 2017;45(2):58-69. [CrossRef]
- Johnson ML, Robinson MM, Nair KS. Skeletal muscle aging and the mitochondrion. Trends Endocrinol Metab. 2013;24(5):247-56.
- Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, et al. Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci U S A. 2005;102(15):5618-23. [CrossRef]
- Peake J, Della Gatta P, Cameron-Smith D. Aging and its effects on inflammation in skeletal muscle at rest and following exercise-induced muscle injury. Am J Physiol-Reg I. 2010;298(6):R1485-R95. [CrossRef]
- Jang YC, Van Remmen H. Age-associated alterations of the neuromuscular junction. Experimental Gerontology. 2011;46(2-3):193-8.
- Cui H, Kong Y, Zhang H. Oxidative stress, mitochondrial dysfunction, and aging. J Signal Transduct. 2012;2012:646354.
- Gonzalez-Freire M, de Cabo R, Studenski SA, Ferrucci L. The neuromuscular junction: aging at the crossroad between nerves and muscle. Front Aging Neurosci. 2014;6. [CrossRef]
- Quiros PM, Langer T, Lopez-Otin C. New roles for mitochondrial proteases in health, ageing and disease. Nat Rev Mol Cell Biol. 2015;16(6):345-59. [CrossRef]
- Sousa-Victor P, Garcia-Prat L, Serrano AL, Perdiguero E, Munoz-Canoves P. Muscle stem cell aging: regulation and rejuvenation. Trends Endocrinol Metab. 2015;26(6):287-96. [CrossRef]
- Campanari ML, García-Ayllón MS, Ciura S, Sáez-Valero J, Kabashi E. Neuromuscular Junction Impairment in Amyotrophic Lateral Sclerosis: Reassessing the Role of Acetylcholinesterase. Front Mol Neurosci. 2016;9. [CrossRef]
- Iyer SR, Shah SB, Lovering RM. The Neuromuscular Junction: Roles in Aging and Neuromuscular Disease. Int J Mol Sci. 2021;22(15).
- Pratt J, De Vito G, Narici M, Boreham C. Neuromuscular Junction Aging: A Role for Biomarkers and Exercise. J Gerontol a-Biol. 2021;76(4):576-85. [CrossRef]
- Antuna E, Cachan-Vega C, Bermejo-Millo JC, Potes Y, Caballero B, Vega-Naredo I, et al. Inflammaging: Implications in Sarcopenia. Int J Mol Sci. 2022;23(23). [CrossRef]
- Chen M, Wang Y, Deng S, Lian Z, Yu K. Skeletal muscle oxidative stress and inflammation in aging: Focus on antioxidant and anti-inflammatory therapy. Front Cell Dev Biol. 2022;10:964130. [CrossRef]
- Miwa S, Kashyap S, Chini E, von Zglinicki T. Mitochondrial dysfunction in cell senescence and aging. J Clin Invest. 2022;132(13). [CrossRef]
- Zhang X, Gao Y, Zhang S, Wang Y, Pei X, Chen Y, et al. Mitochondrial dysfunction in the regulation of aging and aging-related diseases. Cell Commun Signal. 2025;23(1):290. [CrossRef]
- Cartee GD, Hepple RT, Bamman MM, Zierath JR. Exercise Promotes Healthy Aging of Skeletal Muscle. Cell Metab. 2016;23(6):1034-47. [CrossRef]
- Blau HM, Cosgrove BD, Ho AT. The central role of muscle stem cells in regenerative failure with aging. Nat Med. 2015;21(8):854-62. [CrossRef]
- Chatre L, Verdonk F, Rocheteau P, Crochemore C, Chretien F, Ricchetti M. A novel paradigm links mitochondrial dysfunction with muscle stem cell impairment in sepsis. Biochim Biophys Acta Mol Basis Dis. 2017;1863(10 Pt B):2546-53. [CrossRef]
- Hong X, Isern J, Campanario S, Perdiguero E, Ramirez-Pardo I, Segales J, et al. Mitochondrial dynamics maintain muscle stem cell regenerative competence throughout adult life by regulating metabolism and mitophagy. Cell Stem Cell. 2022;29(10):1506-8.
- Bhattacharya D, Shah V, Oresajo O, Scime A. p107 mediated mitochondrial function controls muscle stem cell proliferative fates. Nat Commun. 2021;12(1):5977. [CrossRef]
- Bhattacharya D, Scime A. Mitochondrial Function in Muscle Stem Cell Fates. Front Cell Dev Biol. 2020;8:480. [CrossRef]
- Miao YM, Xie LY, Song JM, Cai X, Yang JH, Ma XL, et al. Unraveling the causes of sarcopenia: Roles of neuromuscular junction impairment and mitochondrial dysfunction. Physiol Rep. 2024;12(1). [CrossRef]
- Xu HY, Ranjit R, Richardson A, Van Remmen H. Muscle mitochondrial catalase expression prevents neuromuscular junction disruption, atrophy, and weakness in a mouse model of accelerated sarcopenia. J Cachexia Sarcopeni. 2021;12(6):1582-96. [CrossRef]
- Xiao YT, Zhang JM, Shu XQ, Bai L, Xu WT, Wang AL, et al. Loss of mitochondrial protein CHCHD10 in skeletal muscle causes neuromuscular junction impairment. Human Molecular Genetics. 2020;29(11):1784-96. [CrossRef]
- Anagnostou ME, Hepple RT. Mitochondrial Mechanisms of Neuromuscular Junction Degeneration with Aging. Cells. 2020;9(1). [CrossRef]
- Liu WX, Klose A, Forman S, Paris ND, Pierre LWL, Cortés-Lopéz M, et al. Loss of adult skeletal muscle stem cells drives age-related neuromuscular junction degeneration. Elife. 2017;6. [CrossRef]
- Verschuuren JJ, Palace J, Murai H, Tannemaat MR, Kaminski HJ, Bril V. Advances and ongoing research in the treatment of autoimmune neuromuscular junction disorders. Lancet Neurol. 2022;21(2):189-202. [CrossRef]
- Fu X, Xiao J, Wei Y, Li S, Liu Y, Yin J, et al. Combination of inflammation-related cytokines promotes long-term muscle stem cell expansion. Cell Res. 2015;25(9):1082-3.
- Ennis WJ, Sui A, Bartholomew A. Stem Cells and Healing: Impact on Inflammation. Adv Wound Care (New Rochelle). 2013;2(7):369-78. [CrossRef]
- Palacios D, Mozzetta C, Consalvi S, Caretti G, Saccone V, Proserpio V, et al. TNF/p38alpha/polycomb signaling to Pax7 locus in satellite cells links inflammation to the epigenetic control of muscle regeneration. Cell Stem Cell. 2010;7(4):455-69.
- Van Dyke JM, Smit-Oistad IM, Macrander C, Krakora D, Meyer MG, Suzuki M. Macrophage-mediated inflammation and glial response in the skeletal muscle of a rat model of familial amyotrophic lateral sclerosis (ALS). Exp Neurol. 2016;277:275-82. [CrossRef]
- Wang J, Leung KS, Chow SK, Cheung WH. Inflammation and age-associated skeletal muscle deterioration (sarcopaenia). J Orthop Translat. 2017;10:94-101. [CrossRef]
- Westbrook R, Chung T, Lovett J, Ward C, Joca H, Yang H, et al. Kynurenines link chronic inflammation to functional decline and physical frailty. JCI Insight. 2020;5(16). [CrossRef]
- Bartman S, Coppotelli G, Ross JM. Mitochondrial Dysfunction: A Key Player in Brain Aging and Diseases. Curr Issues Mol Biol. 2024;46(3):1987-2026. [CrossRef]
- Son JM, Lee C. Aging: All roads lead to mitochondria. Semin Cell Dev Biol. 2021;116:160-8. [CrossRef]
- Jadiya P, Tomar D. Mitochondrial Protein Quality Control Mechanisms. Genes (Basel). 2020;11(5). [CrossRef]
- Russell AP, Foletta VC, Snow RJ, Wadley GD. Skeletal muscle mitochondria: a major player in exercise, health and disease. Biochim Biophys Acta. 2014;1840(4):1276-84. [CrossRef]
- Hepple RT. Mitochondrial involvement and impact in aging skeletal muscle. Front Aging Neurosci. 2014;6:211. [CrossRef]
- Ni HM, Williams JA, Ding WX. Mitochondrial dynamics and mitochondrial quality control. Redox Biol. 2015;4:6-13. [CrossRef]
- Baker MJ, Palmer CS, Stojanovski D. Mitochondrial protein quality control in health and disease. Br J Pharmacol. 2014;171(8):1870-89. [CrossRef]
- Dillon LM, Rebelo AP, Moraes CT. The role of PGC-1 coactivators in aging skeletal muscle and heart. IUBMB Life. 2012;64(3):231-41. [CrossRef]
- Halling JF, Jessen H, Nohr-Meldgaard J, Buch BT, Christensen NM, Gudiksen A, et al. PGC-1alpha regulates mitochondrial properties beyond biogenesis with aging and exercise training. Am J Physiol Endocrinol Metab. 2019;317(3):E513-E25. [CrossRef]
- Baraldo M, Geremia A, Pirazzini M, Nogara L, Solagna F, Türk C, et al. Skeletal muscle mTORC1 regulates neuromuscular junction stability. J Cachexia Sarcopeni. 2020;11(1):208-25. [CrossRef]
- Ogasawara R, Fujita S, Hornberger TA, Kitaoka Y, Makanae Y, Nakazato K, et al. The role of mTOR signalling in the regulation of skeletal muscle mass in a rodent model of resistance exercise. Sci Rep. 2016;6:31142. [CrossRef]
- Liu X, Yuan H, Niu Y, Niu W, Fu L. The role of AMPK/mTOR/S6K1 signaling axis in mediating the physiological process of exercise-induced insulin sensitization in skeletal muscle of C57BL/6 mice. Biochim Biophys Acta. 2012;1822(11):1716-26. [CrossRef]
- Parkington JD, LeBrasseur NK, Siebert AP, Fielding RA. Contraction-mediated mTOR, p70S6k, and ERK1/2 phosphorylation in aged skeletal muscle. J Appl Physiol (1985). 2004;97(1):243-8.
- Chen ZP, Stephens TJ, Murthy S, Canny BJ, Hargreaves M, Witters LA, et al. Effect of exercise intensity on skeletal muscle AMPK signaling in humans. Diabetes. 2003;52(9):2205-12. [CrossRef]
- Salminen A, Kaarniranta K, Kauppinen A. Age-related changes in AMPK activation: Role for AMPK phosphatases and inhibitory phosphorylation by upstream signaling pathways. Ageing Res Rev. 2016;28:15-26. [CrossRef]
- Yuan Y, Cruzat VF, Newsholme P, Cheng J, Chen Y, Lu Y. Regulation of SIRT1 in aging: Roles in mitochondrial function and biogenesis. Mech Ageing Dev. 2016;155:10-21. [CrossRef]
- Myers MJ, Shepherd DL, Durr AJ, Stanton DS, Mohamed JS, Hollander JM, et al. The role of SIRT1 in skeletal muscle function and repair of older mice. J Cachexia Sarcopeni. 2019;10(4):929-49. [CrossRef]
- Price NL, Gomes AP, Ling AJ, Duarte FV, Martin-Montalvo A, North BJ, et al. SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell Metab. 2012;15(5):675-90. [CrossRef]
- Zhao T, Fan J, Abu-Zaid A, Burley SK, Zheng XFS. Nuclear mTOR Signaling Orchestrates Transcriptional Programs Underlying Cellular Growth and Metabolism. Cells. 2024;13(9). [CrossRef]
- Fan J, Yuan Z, Burley SK, Libutti SK, Zheng XFS. Amino acids control blood glucose levels through mTOR signaling. Eur J Cell Biol. 2022;101(3):151240. [CrossRef]
- Yapa NMB, Lisnyak V, Reljic B, Ryan MT. Mitochondrial dynamics in health and disease. FEBS Lett. 2021;595(8):1184-204. [CrossRef]
- Raven KD, Kapetanovic R. Mitochondrial dynamics: Regulating cell metabolism, homoeostasis, health and disease. Semin Cell Dev Biol. 2024;161-162:20-1. [CrossRef]
- Tilokani L, Nagashima S, Paupe V, Prudent J. Mitochondrial dynamics: overview of molecular mechanisms. Essays Biochem. 2018;62(3):341-60. [CrossRef]
- Marinkovic M, Novak I. A brief overview of BNIP3L/NIX receptor-mediated mitophagy. FEBS Open Bio. 2021;11(12):3230-6. [CrossRef]
- Liu K, Zhao Q, Sun H, Liu L, Wang C, Li Z, et al. BNIP3 (BCL2 interacting protein 3) regulates pluripotency by modulating mitochondrial homeostasis via mitophagy. Cell Death Dis. 2022;13(4):334. [CrossRef]
- Fu T, Xu Z, Liu L, Guo Q, Wu H, Liang X, et al. Mitophagy Directs Muscle-Adipose Crosstalk to Alleviate Dietary Obesity. Cell Rep. 2018;23(5):1357-72. [CrossRef]
- Carter HN, Kim Y, Erlich AT, Zarrin-Khat D, Hood DA. Autophagy and mitophagy flux in young and aged skeletal muscle following chronic contractile activity. J Physiol. 2018;596(16):3567-84. [CrossRef]
- Ashrafi G, Schwarz TL. The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ. 2013;20(1):31-42. [CrossRef]
- Nouri K, Feng Y, Schimmer AD. Mitochondrial ClpP serine protease-biological function and emerging target for cancer therapy. Cell Death Dis. 2020;11(10):841. [CrossRef]
- Pharaoh G, Sataranatarajan K, Street K, Hill S, Gregston J, Ahn B, et al. Metabolic and Stress Response Changes Precede Disease Onset in the Spinal Cord of Mutant SOD1 ALS Mice. Front Neurosci. 2019;13:487. [CrossRef]
- Bota DA, Van Remmen H, Davies KJ. Modulation of Lon protease activity and aconitase turnover during aging and oxidative stress. FEBS Lett. 2002;532(1-2):103-6. [CrossRef]
- Bota DA, Davies KJ. Lon protease preferentially degrades oxidized mitochondrial aconitase by an ATP-stimulated mechanism. Nat Cell Biol. 2002;4(9):674-80. [CrossRef]
- Guo C, Xiao Y, Gu J, Zhao P, Hu Z, Zheng J, et al. ClpP/ClpX deficiency impairs mitochondrial functions and mTORC1 signaling during spermatogenesis. Commun Biol. 2023;6(1):1012. [CrossRef]
- Picca A, Guerra F, Calvani R, Romano R, Coelho-Junior HJ, Bucci C, et al. Mitochondrial-derived vesicles in skeletal muscle remodeling and adaptation. Semin Cell Dev Biol. 2023;143:37-45. [CrossRef]
- Konig T, McBride HM. Mitochondrial-derived vesicles in metabolism, disease, and aging. Cell Metab. 2024;36(1):21-35. [CrossRef]
- Sugiura A, McLelland GL, Fon EA, McBride HM. A new pathway for mitochondrial quality control: mitochondrial-derived vesicles. EMBO J. 2014;33(19):2142-56. [CrossRef]
- Melentijevic I, Toth ML, Arnold ML, Guasp RJ, Harinath G, Nguyen KC, et al. C. elegans neurons jettison protein aggregates and mitochondria under neurotoxic stress. Nature. 2017;542(7641):367-71. [CrossRef]
- Roberts RF, Tang MY, Fon EA, Durcan TM. Defending the mitochondria: The pathways of mitophagy and mitochondrial-derived vesicles. Int J Biochem Cell Biol. 2016;79:427-36. [CrossRef]
- Konig T, Nolte H, Aaltonen MJ, Tatsuta T, Krols M, Stroh T, et al. MIROs and DRP1 drive mitochondrial-derived vesicle biogenesis and promote quality control. Nat Cell Biol. 2021;23(12):1271-86. [CrossRef]
- Liang W, Sagar S, Ravindran R, Najor RH, Quiles JM, Chi L, et al. Mitochondria are secreted in extracellular vesicles when lysosomal function is impaired. Nat Commun. 2023;14(1):5031. [CrossRef]
- Turek M, Banasiak K, Piechota M, Shanmugam N, Macias M, Sliwinska MA, et al. Muscle-derived exophers promote reproductive fitness. EMBO Rep. 2021;22(8):e52071. [CrossRef]
- Nicolas-Avila JA, Lechuga-Vieco AV, Esteban-Martinez L, Sanchez-Diaz M, Diaz-Garcia E, Santiago DJ, et al. A Network of Macrophages Supports Mitochondrial Homeostasis in the Heart. Cell. 2020;183(1):94-109 e23. [CrossRef]
- Reiter RJ, Tan DX, Rosales-Corral S, Galano A, Zhou XJ, Xu B. Mitochondria: Central Organelles for Melatonin's Antioxidant and Anti-Aging Actions. Molecules. 2018;23(2). [CrossRef]
- Sladowska M, Turek M, Kim MJ, Drabikowski K, Mussulini BHM, Mohanraj K, et al. Proteasome activity contributes to pro-survival response upon mild mitochondrial stress in Caenorhabditis elegans. PLoS Biol. 2021;19(7):e3001302. [CrossRef]
- Ruan L, Wang Y, Zhang X, Tomaszewski A, McNamara JT, Li R. Mitochondria-Associated Proteostasis. Annu Rev Biophys. 2020;49:41-67. [CrossRef]
- Wong YC, Ysselstein D, Krainc D. Mitochondria-lysosome contacts regulate mitochondrial fission via RAB7 GTP hydrolysis. Nature. 2018;554(7692):382-6. [CrossRef]
- Triolo M, Hood DA. Manifestations of Age on Autophagy, Mitophagy and Lysosomes in Skeletal Muscle. Cells. 2021;10(5). [CrossRef]
- Xiao Y, Karam C, Yi J, Zhang L, Li X, Yoon D, et al. ROS-related mitochondrial dysfunction in skeletal muscle of an ALS mouse model during the disease progression. Pharmacol Res. 2018;138:25-36. [CrossRef]
- Wang F, Wang X, Liu Y, Zhang Z. Effects of Exercise-Induced ROS on the Pathophysiological Functions of Skeletal Muscle. Oxid Med Cell Longev. 2021;2021:3846122. [CrossRef]
- Lian D, Chen MM, Wu H, Deng S, Hu X. The Role of Oxidative Stress in Skeletal Muscle Myogenesis and Muscle Disease. Antioxidants (Basel). 2022;11(4). [CrossRef]
- Hollander J, Fiebig R, Gore M, Bejma J, Ookawara T, Ohno H, et al. Superoxide dismutase gene expression in skeletal muscle: fiber-specific adaptation to endurance training. Am J Physiol. 1999;277(3):R856-62. [CrossRef]
- Hollander J, Bejma J, Ookawara T, Ohno H, Ji LL. Superoxide dismutase gene expression in skeletal muscle: fiber-specific effect of age. Mech Ageing Dev. 2000;116(1):33-45. [CrossRef]
- Hood DA, Tryon LD, Carter HN, Kim Y, Chen CC. Unravelling the mechanisms regulating muscle mitochondrial biogenesis. Biochem J. 2016;473(15):2295-314. [CrossRef]
- Mancinelli R, Checcaglini F, Coscia F, Gigliotti P, Fulle S, Fano-Illic G. Biological Aspects of Selected Myokines in Skeletal Muscle: Focus on Aging. Int J Mol Sci. 2021;22(16). [CrossRef]
- Yeo D, Kang C, Gomez-Cabrera MC, Vina J, Ji LL. Intensified mitophagy in skeletal muscle with aging is downregulated by PGC-1alpha overexpression in vivo. Free Radical Bio Med. 2019;130:361-8. [CrossRef]
- Garcia S, Nissanka N, Mareco EA, Rossi S, Peralta S, Diaz F, et al. Overexpression of PGC-1alpha in aging muscle enhances a subset of young-like molecular patterns. Aging Cell. 2018;17(2).
- Bujak AL, Crane JD, Lally JS, Ford RJ, Kang SJ, Rebalka IA, et al. AMPK activation of muscle autophagy prevents fasting-induced hypoglycemia and myopathy during aging. Cell Metab. 2015;21(6):883-90. [CrossRef]
- Thompson AM, Wagner R, Rzucidlo EM. Age-related loss of SirT1 expression results in dysregulated human vascular smooth muscle cell function. Am J Physiol-Heart C. 2014;307(4):H533-H41. [CrossRef]
- Koltai E, Szabo Z, Atalay M, Boldogh I, Naito H, Goto S, et al. Exercise alters SIRT1, SIRT6, NAD and NAMPT levels in skeletal muscle of aged rats. Mech Ageing Dev. 2010;131(1):21-8. [CrossRef]
- Tang H, Inoki K, Brooks SV, Okazawa H, Lee M, Wang J, et al. mTORC1 underlies age-related muscle fiber damage and loss by inducing oxidative stress and catabolism. Aging Cell. 2019;18(3):e12943. [CrossRef]
- Vernier M, Giguere V. Aging, senescence and mitochondria: the PGC-1/ERR axis. J Mol Endocrinol. 2021;66(1):R1-R14.
- Lin Y, Zhan M, Chen X, Xiao X. Biological function of EPHB4 in the aging process of vascular endothelial cells: mtDNA molecular mechanism and MAPK/PGC-1/TFAM signaling pathway. Int J Biol Macromol. 2025;293:138536.
- Gonzalez AA, Kumar R, Mulligan JD, Davis AJ, Saupe KW. Effects of aging on cardiac and skeletal muscle AMPK activity: basal activity, allosteric activation, and response to in vivo hypoxemia in mice. Am J Physiol Regul Integr Comp Physiol. 2004;287(5):R1270-5. [CrossRef]
- Jiang D, Liu C, Chen Y, Xing X, Zheng D. Whole body vibration activates AMPK/CPT1 signaling pathway of skeletal muscle in young and aging mice based on metabolomics study. Endocr J. 2022;69(5):585-96. [CrossRef]
- Seo AY, Joseph AM, Dutta D, Hwang JCY, Aris JP, Leeuwenburgh C. New insights into the role of mitochondria in aging: mitochondrial dynamics and more. Journal of Cell Science. 2010;123(15):2533-42. [CrossRef]
- Chan DC. Fusion and fission: interlinked processes critical for mitochondrial health. Annu Rev Genet. 2012;46:265-87. [CrossRef]
- Romanello V, Sandri M. Implications of mitochondrial fusion and fission in skeletal muscle mass and health. Semin Cell Dev Biol. 2023;143:46-53. [CrossRef]
- Iqbal S, Ostojic O, Singh K, Joseph AM, Hood DA. Expression of mitochondrial fission and fusion regulatory proteins in skeletal muscle during chronic use and disuse. Muscle Nerve. 2013;48(6):963-70. [CrossRef]
- Liguori I, Russo G, Curcio F, Bulli G, Aran L, Della-Morte D, et al. Oxidative stress, aging, and diseases. Clin Interv Aging. 2018;13:757-72.
- Shaw PX, Werstuck G, Chen Y. Oxidative stress and aging diseases. Oxid Med Cell Longev. 2014;2014:569146.
- El Assar M, Alvarez-Bustos A, Sosa P, Angulo J, Rodriguez-Manas L. Effect of Physical Activity/Exercise on Oxidative Stress and Inflammation in Muscle and Vascular Aging. Int J Mol Sci. 2022;23(15). [CrossRef]
- Puengpan S, Phetrungnapha A, Sattayakawee S, Tunsophon S. Phycocyanin attenuates skeletal muscle damage and fatigue via modulation of Nrf2 and IRS-1/AKT/mTOR pathway in exercise-induced oxidative stress in rats. PLoS One. 2024;19(9):e0310138. [CrossRef]
- Yang J, Luo J, Tian X, Zhao Y, Li Y, Wu X. Progress in Understanding Oxidative Stress, Aging, and Aging-Related Diseases. Antioxidants (Basel). 2024;13(4). [CrossRef]
- Qiu X, Brown K, Hirschey MD, Verdin E, Chen D. Calorie restriction reduces oxidative stress by SIRT3-mediated SOD2 activation. Cell Metab. 2010;12(6):662-7. [CrossRef]
- Dikalova AE, Itani HA, Nazarewicz RR, McMaster WG, Flynn CR, Uzhachenko R, et al. Sirt3 Impairment and SOD2 Hyperacetylation in Vascular Oxidative Stress and Hypertension. Circ Res. 2017;121(5):564-74. [CrossRef]
- Marchi S, Giorgi C, Suski JM, Agnoletto C, Bononi A, Bonora M, et al. Mitochondria-ros crosstalk in the control of cell death and aging. J Signal Transduct. 2012;2012:329635. [CrossRef]
- Dai DF, Chiao YA, Marcinek DJ, Szeto HH, Rabinovitch PS. Mitochondrial oxidative stress in aging and healthspan. Longev Healthspan. 2014;3:6. [CrossRef]
- Pellegrino MW, Nargund AM, Haynes CM. Signaling the mitochondrial unfolded protein response. Biochim Biophys Acta. 2013;1833(2):410-6. [CrossRef]
- Jovaisaite V, Mouchiroud L, Auwerx J. The mitochondrial unfolded protein response, a conserved stress response pathway with implications in health and disease. J Exp Biol. 2014;217(Pt 1):137-43. [CrossRef]
- Mesbah Moosavi ZS, Hood DA. The unfolded protein response in relation to mitochondrial biogenesis in skeletal muscle cells. Am J Physiol Cell Physiol. 2017;312(5):C583-C94.
- Naresh NU, Haynes CM. Signaling and Regulation of the Mitochondrial Unfolded Protein Response. Cold Spring Harb Perspect Biol. 2019;11(6). [CrossRef]
- Shin CS, Meng S, Garbis SD, Moradian A, Taylor RW, Sweredoski MJ, et al. LONP1 and mtHSP70 cooperate to promote mitochondrial protein folding. Nat Commun. 2021;12(1):265. [CrossRef]
- Seli E, Wang T, Horvath TL. Mitochondrial unfolded protein response: a stress response with implications for fertility and reproductive aging. Fertil Steril. 2019;111(2):197-204. [CrossRef]
- Munoz-Carvajal F, Sanhueza M. The Mitochondrial Unfolded Protein Response: A Hinge Between Healthy and Pathological Aging. Front Aging Neurosci. 2020;12:581849. [CrossRef]
- Wang Y, Li J, Zhang Z, Wang R, Bo H, Zhang Y. Exercise Improves the Coordination of the Mitochondrial Unfolded Protein Response and Mitophagy in Aging Skeletal Muscle. Life (Basel). 2023;13(4). [CrossRef]
- Gopinath SD, Rando TA. Stem cell review series: aging of the skeletal muscle stem cell niche. Aging Cell. 2008;7(4):590-8. [CrossRef]
- Lukjanenko L, Karaz S, Stuelsatz P, Gurriaran-Rodriguez U, Michaud J, Dammone G, et al. Aging Disrupts Muscle Stem Cell Function by Impairing Matricellular WISP1 Secretion from Fibro-Adipogenic Progenitors. Cell Stem Cell. 2019;24(3):433-46 e7. [CrossRef]
- Sousa-Victor P, Neves J, Munoz-Canoves P. Muscle stem cell aging: identifying ways to induce tissue rejuvenation. Mech Ageing Dev. 2020;188:111246. [CrossRef]
- Memczak S, Belmonte JC. Overcoming muscle stem cell aging. Curr Opin Genet Dev. 2023;83:102127. [CrossRef]
- Fulzele S, Mendhe B, Khayrullin A, Johnson M, Kaiser H, Liu Y, et al. Muscle-derived miR-34a increases with age in circulating extracellular vesicles and induces senescence of bone marrow stem cells. Aging (Albany NY). 2019;11(6):1791-803. [CrossRef]
- Xu Y, He Z, Song M, Zhou Y, Shen Y. A microRNA switch controls dietary restriction-induced longevity through Wnt signaling. EMBO Rep. 2019;20(5). [CrossRef]
- Hu Q, Xu Y, Song M, Dai Y, Antebi A, Shen Y. BLMP-1 is a critical temporal regulator of dietary-restriction-induced response in Caenorhabditis elegans. Cell Rep. 2024;43(3):113959. [CrossRef]
- Pan C, Xiong Y, Lv X, Xia Y, Zhang S, Chen H, et al. UbcD1 regulates Hedgehog signaling by directly modulating Ci ubiquitination and processing. EMBO Rep. 2017;18(11):1922-34. [CrossRef]
- Fan J, Gao Y, Lu Y, Wu W, Yuan S, Wu H, et al. PKAc-directed interaction and phosphorylation of Ptc is required for Hh signaling inhibition in Drosophila. Cell Discov. 2019;5:44. [CrossRef]
- Bloch-Gallego E. Mechanisms controlling neuromuscular junction stability. Cellular and Molecular Life Sciences. 2015;72(6):1029-43. [CrossRef]
- Liu W, Wei-LaPierre L, Klose A, Dirksen RT, Chakkalakal JV. Inducible depletion of adult skeletal muscle stem cells impairs the regeneration of neuromuscular junctions. Elife. 2015;4. [CrossRef]
- Sousa-Victor P, Garcia-Prat L, Munoz-Canoves P. New mechanisms driving muscle stem cell regenerative decline with aging. Int J Dev Biol. 2018;62(6-7-8):583-90. [CrossRef]
- Londhe P, Guttridge DC. Inflammation induced loss of skeletal muscle. Bone. 2015;80:131-42. [CrossRef]
- Lepore E, Casola I, Dobrowolny G, Musarò A. Neuromuscular Junction as an Entity of Nerve-Muscle Communication. Cells. 2019;8(8). [CrossRef]
- Personius KE, Parker SD. TrkB expression at the neuromuscular junction is reduced during aging. Muscle Nerve. 2013;47(4):532-8. [CrossRef]
- Mantilla CB, Stowe JM, Sieck DC, Ermilov LG, Greising SM, Zhang C, et al. TrkB kinase activity maintains synaptic function and structural integrity at adult neuromuscular junctions. J Appl Physiol. 2014;117(8):910-20. [CrossRef]
- Greising SM, Stowe JM, Sieck GC, Mantilla CB. Role of TrkB kinase activity in aging diaphragm neuromuscular junctions. Experimental Gerontology. 2015;72:184-91. [CrossRef]
- Simó A, Just-Borràs L, Cilleros-Mañé V, Hurtado E, Nadal L, Tomàs M, et al. BDNF-TrkB Signaling Coupled to nPKCε and cPKCβI Modulate the Phosphorylation of the Exocytotic Protein Munc18-1 During Synaptic Activity at the Neuromuscular Junction. Front Mol Neurosci. 2018;11. [CrossRef]
- Snyder-Warwick AK, Satoh A, Santosa KB, Imai S, Jablonka-Shariff A. Hypothalamic Sirt1 protects terminal Schwann cells and neuromuscular junctions from age-related morphological changes. Aging Cell. 2018;17(4). [CrossRef]
- Ang STJ, Crombie EM, Dong H, Tan KT, Hernando A, Yu DJ, et al. Muscle 4EBP1 activation modifies the structure and function of the neuromuscular junction in mice. Nature Communications. 2022;13(1). [CrossRef]
- Genin EC, Madji Hounoum B, Bannwarth S, Fragaki K, Lacas-Gervais S, Mauri-Crouzet A, et al. Mitochondrial defect in muscle precedes neuromuscular junction degeneration and motor neuron death in CHCHD10(S59L/+) mouse. Acta Neuropathol. 2019;138(1):123-45. [CrossRef]
- Dagdeviren S, Jung DY, Friedline RH, Noh HL, Kim JH, Patel PR, et al. IL-10 prevents aging-associated inflammation and insulin resistance in skeletal muscle. FASEB J. 2017;31(2):701-10. [CrossRef]
- Qi G, Mi Y, Fan R, Li R, Liu Z, Liu X. Nobiletin Protects against Systemic Inflammation-Stimulated Memory Impairment via MAPK and NF-kappaB Signaling Pathways. J Agric Food Chem. 2019;67(18):5122-34. [CrossRef]
- Belizario JE, Fontes-Oliveira CC, Borges JP, Kashiabara JA, Vannier E. Skeletal muscle wasting and renewal: a pivotal role of myokine IL-6. Springerplus. 2016;5:619. [CrossRef]
- Ma JF, Sanchez BJ, Hall DT, Tremblay AK, Di Marco S, Gallouzi IE. STAT3 promotes IFNgamma/TNFalpha-induced muscle wasting in an NF-kappaB-dependent and IL-6-independent manner. Embo Mol Med. 2017;9(5):622-37.
- Chen F, Fu J, Feng H. IL-6 Promotes Muscle Atrophy by Increasing Ubiquitin-Proteasome Degradation of Muscle Regeneration Factors After Cerebral Infarction in Rats. Neuromolecular Med. 2025;27(1):3. [CrossRef]
- Thoma A, Lightfoot AP. NF-kB and Inflammatory Cytokine Signalling: Role in Skeletal Muscle Atrophy. Adv Exp Med Biol. 2018;1088:267-79.
- Mourkioti F, Rosenthal N. NF-kappaB signaling in skeletal muscle: prospects for intervention in muscle diseases. J Mol Med (Berl). 2008;86(7):747-59. [CrossRef]
- Choudhary S, Sinha S, Zhao Y, Banerjee S, Sathyanarayana P, Shahani S, et al. NF-kappaB-inducing kinase (NIK) mediates skeletal muscle insulin resistance: blockade by adiponectin. Endocrinology. 2011;152(10):3622-7. [CrossRef]
- Remels AH, Gosker HR, Verhees KJ, Langen RC, Schols AM. TNF-alpha-induced NF-kappaB activation stimulates skeletal muscle glycolytic metabolism through activation of HIF-1alpha. Endocrinology. 2015;156(5):1770-81.
- Biggar Y, Kamath AA, Breedon SA, Storey KB. NF-kappaB signaling and its anti-apoptotic effects in liver & skeletal muscle of dehydrated Xenopus laevis. Exp Cell Res. 2025;449(1):114579. [CrossRef]
- Ismaeel A, Kim JS, Kirk JS, Smith RS, Bohannon WT, Koutakis P. Role of Transforming Growth Factor-beta in Skeletal Muscle Fibrosis: A Review. Int J Mol Sci. 2019;20(10). [CrossRef]
- Song Y, Shen H, Schenten D, Shan P, Lee PJ, Goldstein DR. Aging enhances the basal production of IL-6 and CCL2 in vascular smooth muscle cells. Arterioscler Thromb Vasc Biol. 2012;32(1):103-9. [CrossRef]
- Picca A, Lezza AMS, Leeuwenburgh C, Pesce V, Calvani R, Bossola M, et al. Circulating Mitochondrial DNA at the Crossroads of Mitochondrial Dysfunction and Inflammation During Aging and Muscle Wasting Disorders. Rejuv Res. 2018;21(4):350-9. [CrossRef]
- Marchi S, Guilbaud E, Tait SWG, Yamazaki T, Galluzzi L. Mitochondrial control of inflammation. Nat Rev Immunol. 2023;23(3):159-73.
- Brack AS, Conboy IM, Conboy MJ, Shen J, Rando TA. A temporal switch from notch to Wnt signaling in muscle stem cells is necessary for normal adult myogenesis. Cell Stem Cell. 2008;2(1):50-9. [CrossRef]
- Luo D, Renault VM, Rando TA. The regulation of Notch signaling in muscle stem cell activation and postnatal myogenesis. Semin Cell Dev Biol. 2005;16(4-5):612-22. [CrossRef]
- Wang GQ, Wang Y, Xiong Y, Chen XC, Ma ML, Cai R, et al. Sirt1 AS lncRNA interacts with its mRNA to inhibit muscle formation by attenuating function of miR-34a. Sci Rep. 2016;6:21865. [CrossRef]
- Zhang C, Zhang Y, Zhang W, Tong H, Li S, Yan Y. WISP1 promotes bovine MDSC differentiation via recruitment of ANXA1 for the regulation of the TGF-beta signalling pathway. Mol Cell Biochem. 2020;470(1-2):215-27. [CrossRef]
- Josephson AM, Bradaschia-Correa V, Lee S, Leclerc K, Patel KS, Muinos Lopez E, et al. Age-related inflammation triggers skeletal stem/progenitor cell dysfunction. Proc Natl Acad Sci U S A. 2019;116(14):6995-7004. [CrossRef]
- Irfan M, Marzban H, Chung S. C5L2 CRISPR KO enhances dental pulp stem cell-mediated dentinogenesis via TrkB under TNFalpha-induced inflammation. Front Cell Dev Biol. 2024;12:1338419. [CrossRef]
- Zuo B, Fan X, Xu D, Zhao L, Zhang B, Li X. Deciphering the mitochondria-inflammation axis: Insights and therapeutic strategies for heart failure. Int Immunopharmacol. 2024;139:112697. [CrossRef]
- Nunes JPS, Roda VMP, Andrieux P, Kalil J, Chevillard C, Cunha-Neto E. Inflammation and mitochondria in the pathogenesis of chronic Chagas disease cardiomyopathy. Exp Biol Med (Maywood). 2023;248(22):2062-71. [CrossRef]
- Malnoe D, Bories M, Pierre-Jean M, Marchand T, Le Corre P. Inflammation Decreases Ciclosporin Metabolism in Allogeneic Hematopoietic Stem Cell Transplantation Recipients. J Clin Pharmacol. 2025;65(3):328-39. [CrossRef]
- Voloboueva LA, Giffard RG. Inflammation, mitochondria, and the inhibition of adult neurogenesis. J Neurosci Res. 2011;89(12):1989-96.
- Liu J, Han Z, Han Z, He Z. Mesenchymal stem cell-conditioned media suppresses inflammation-associated overproliferation of pulmonary artery smooth muscle cells in a rat model of pulmonary hypertension. Exp Ther Med. 2016;11(2):467-75.
- Medeiros C, Frederico MJ, da Luz G, Pauli JR, Silva AS, Pinho RA, et al. Exercise training reduces insulin resistance and upregulates the mTOR/p70S6k pathway in cardiac muscle of diet-induced obesity rats. J Cell Physiol. 2011;226(3):666-74.
- Axelrod CL, Fealy CE, Mulya A, Kirwan JP. Exercise training remodels human skeletal muscle mitochondrial fission and fusion machinery towards a pro-elongation phenotype. Acta Physiol (Oxf). 2019;225(4):e13216. [CrossRef]
- Kim Y, Triolo M, Hood DA. Impact of Aging and Exercise on Mitochondrial Quality Control in Skeletal Muscle. Oxid Med Cell Longev. 2017;2017. [CrossRef]
- Zou K, De Lisio M, Huntsman HD, Pincu Y, Mahmassani Z, Miller M, et al. Laminin-111 improves skeletal muscle stem cell quantity and function following eccentric exercise. Stem Cells Transl Med. 2014;3(9):1013-22. [CrossRef]
- Peake JM, Neubauer O, Della Gatta PA, Nosaka K. Muscle damage and inflammation during recovery from exercise. J Appl Physiol (1985). 2017;122(3):559-70. [CrossRef]
- Lee TXY, Wu J, Jean WH, Condello G, Alkhatib A, Hsieh CC, et al. Reduced stem cell aging in exercised human skeletal muscle is enhanced by ginsenoside Rg1. Aging (Albany NY). 2021;13(12):16567-76. [CrossRef]
- Moradi N, Sanfrancesco VC, Champsi S, Hood DA. Regulation of lysosomes in skeletal muscle during exercise, disuse and aging. Free Radic Biol Med. 2024;225:323-32. [CrossRef]
- Moore DR, Atherton PJ, Rennie MJ, Tarnopolsky MA, Phillips SM. Resistance exercise enhances mTOR and MAPK signalling in human muscle over that seen at rest after bolus protein ingestion. Acta Physiol (Oxf). 2011;201(3):365-72. [CrossRef]
- Nishimune H, Stanford JA, Mori Y. Role of Exercise in Maintaining the Integrity of the Neuromuscular Junction. Muscle Nerve. 2014;49(3):315-24. [CrossRef]
- Hargreaves M, Spriet LL. Skeletal muscle energy metabolism during exercise. Nat Metab. 2020;2(9):817-28.
- Lambert CP, Wright NR, Finck BN, Villareal DT. Exercise but not diet-induced weight loss decreases skeletal muscle inflammatory gene expression in frail obese elderly persons. J Appl Physiol (1985). 2008;105(2):473-8.
- Sharples AP, Hughes DC, Deane CS, Saini A, Selman C, Stewart CE. Longevity and skeletal muscle mass: the role of IGF signalling, the sirtuins, dietary restriction and protein intake. Aging Cell. 2015;14(4):511-23. [CrossRef]
- Keith N, Bronson RT, Lipman RD, Ding W, Lamont L, Cosmas AC, et al. Diet restriction and age alters skeletal muscle capillarity in B6C3F1 mice. J Am Aging Assoc. 2000;23(3):141-5. [CrossRef]
- Nam SY, Kim KR, Cha BS, Song YD, Lim SK, Lee HC, et al. Low-dose growth hormone treatment combined with diet restriction decreases insulin resistance by reducing visceral fat and increasing muscle mass in obese type 2 diabetic patients. Int J Obes Relat Metab Disord. 2001;25(8):1101-7. [CrossRef]
- Magwere T, Goodall S, Skepper J, Mair W, Brand MD, Partridge L. The effect of dietary restriction on mitochondrial protein density and flight muscle mitochondrial morphology in Drosophila. J Gerontol A Biol Sci Med Sci. 2006;61(1):36-47. [CrossRef]
- Colom B, Oliver J, Roca P, Garcia-Palmer FJ. Caloric restriction and gender modulate cardiac muscle mitochondrial H2O2 production and oxidative damage. Cardiovasc Res. 2007;74(3):456-65. [CrossRef]
- Chen D, Bruno J, Easlon E, Lin SJ, Cheng HL, Alt FW, et al. Tissue-specific regulation of SIRT1 by calorie restriction. Genes Dev. 2008;22(13):1753-7. [CrossRef]
- Fulco M, Cen Y, Zhao P, Hoffman EP, McBurney MW, Sauve AA, et al. Glucose restriction inhibits skeletal myoblast differentiation by activating SIRT1 through AMPK-mediated regulation of Nampt. Dev Cell. 2008;14(5):661-73. [CrossRef]
- Hepple RT, Qin M, Nakamoto H, Goto S. Caloric restriction optimizes the proteasome pathway with aging in rat plantaris muscle: implications for sarcopenia. Am J Physiol Regul Integr Comp Physiol. 2008;295(4):R1231-7. [CrossRef]
- Canto C, Auwerx J. Caloric restriction, SIRT1 and longevity. Trends Endocrinol Metab. 2009;20(7):325-31.
- Schenk S, McCurdy CE, Philp A, Chen MZ, Holliday MJ, Bandyopadhyay GK, et al. Sirt1 enhances skeletal muscle insulin sensitivity in mice during caloric restriction. J Clin Invest. 2011;121(11):4281-8. [CrossRef]
- Tauriainen E, Storvik M, Finckenberg P, Merasto S, Martonen E, Pilvi TK, et al. Skeletal muscle gene expression profile is modified by dietary protein source and calcium during energy restriction. J Nutrigenet Nutrigenomics. 2011;4(1):49-62. [CrossRef]
- Cerletti M, Jang YC, Finley LW, Haigis MC, Wagers AJ. Short-term calorie restriction enhances skeletal muscle stem cell function. Cell Stem Cell. 2012;10(5):515-9. [CrossRef]
- Jang YC, Liu Y, Hayworth CR, Bhattacharya A, Lustgarten MS, Muller FL, et al. Dietary restriction attenuates age-associated muscle atrophy by lowering oxidative stress in mice even in complete absence of CuZnSOD. Aging Cell. 2012;11(5):770-82. [CrossRef]
- Mercken EM, Majounie E, Ding J, Guo R, Kim J, Bernier M, et al. Age-associated miRNA alterations in skeletal muscle from rhesus monkeys reversed by caloric restriction. Aging (Albany NY). 2013;5(9):692-703. [CrossRef]
- Wang CC, Adochio RL, Leitner JW, Abeyta IM, Draznin B, Cornier MA. Acute effects of different diet compositions on skeletal muscle insulin signalling in obese individuals during caloric restriction. Metabolism. 2013;62(4):595-603. [CrossRef]
- Suga T, Kinugawa S, Takada S, Kadoguchi T, Fukushima A, Homma T, et al. Combination of exercise training and diet restriction normalizes limited exercise capacity and impaired skeletal muscle function in diet-induced diabetic mice. Endocrinology. 2014;155(1):68-80. [CrossRef]
- Wong KE, Mikus CR, Slentz DH, Seiler SE, DeBalsi KL, Ilkayeva OR, et al. Muscle-Specific Overexpression of PGC-1alpha Does Not Augment Metabolic Improvements in Response to Exercise and Caloric Restriction. Diabetes. 2015;64(5):1532-43. [CrossRef]
- Keogh K, Kenny DA, Cormican P, McCabe MS, Kelly AK, Waters SM. Effect of Dietary Restriction and Subsequent Re-Alimentation on the Transcriptional Profile of Bovine Skeletal Muscle. PLoS One. 2016;11(2):e0149373. [CrossRef]
- Li Y, Li F, Wu L, Wei H, Liu Y, Li T, et al. Effects of dietary protein restriction on muscle fiber characteristics and mTORC1 pathway in the skeletal muscle of growing-finishing pigs. J Anim Sci Biotechnol. 2016;7(1):47. [CrossRef]
- Margolis LM, Rivas DA, Berrone M, Ezzyat Y, Young AJ, McClung JP, et al. Prolonged Calorie Restriction Downregulates Skeletal Muscle mTORC1 Signaling Independent of Dietary Protein Intake and Associated microRNA Expression. Front Physiol. 2016;7:445. [CrossRef]
- Oku Y, Tanabe R, Nakaoka K, Yamada A, Noda S, Hoshino A, et al. Influences of dietary vitamin D restriction on bone strength, body composition and muscle in rats fed a high-fat diet: involvement of mRNA expression of MyoD in skeletal muscle. J Nutr Biochem. 2016;32:85-90. [CrossRef]
- Elashry MI, Matsakas A, Wenisch S, Arnhold S, Patel K. The effect of caloric restriction on the forelimb skeletal muscle fibers of the hypertrophic myostatin null mice. Acta Histochem. 2017;119(5):582-91. [CrossRef]
- Stockinger J, Maxwell N, Shapiro D, deCabo R, Valdez G. Caloric Restriction Mimetics Slow Aging of Neuromuscular Synapses and Muscle Fibers. J Gerontol A Biol Sci Med Sci. 2017;73(1):21-8. [CrossRef]
- Vanacore D, Messina G, Lama S, Bitti G, Ambrosio P, Tenore G, et al. Effect of restriction vegan diet's on muscle mass, oxidative status, and myocytes differentiation: A pilot study. J Cell Physiol. 2018;233(12):9345-53. [CrossRef]
- Walters RO, Arias E, Diaz A, Burgos ES, Guan FX, Tiano S, et al. Sarcosine Is Uniquely Modulated by Aging and Dietary Restriction in Rodents and Humans. Cell Reports. 2018;25(3):663-+. [CrossRef]
- Yoshida S, Yamahara K, Kume S, Koya D, Yasuda-Yamahara M, Takeda N, et al. Role of dietary amino acid balance in diet restriction-mediated lifespan extension, renoprotection, and muscle weakness in aged mice. Aging Cell. 2018;17(4):e12796.
- Serna JDC, Caldeira da Silva CC, Kowaltowski AJ. Functional changes induced by caloric restriction in cardiac and skeletal muscle mitochondria. J Bioenerg Biomembr. 2020;52(4):269-77. [CrossRef]
- Bareja A, Draper JA, Katz LH, Lee DE, Grimsrud PA, White JP. Chronic caloric restriction maintains a youthful phosphoproteome in aged skeletal muscle. Mech Ageing Dev. 2021;195:111443. [CrossRef]
- Hernandez-Saavedra D, Moody L, Tang X, Goldberg ZJ, Wang AP, Chen H, et al. Caloric restriction following early-life high fat-diet feeding represses skeletal muscle TNF in male rats. J Nutr Biochem. 2021;91:108598. [CrossRef]
- Myers MJ, Shaik F, Shaik F, Alway SE, Mohamed JS. Skeletal Muscle Gene Expression Profile in Response to Caloric Restriction and Aging: A Role for SirT1. Genes (Basel). 2021;12(5). [CrossRef]
- Swaminathan A, Fokin A, Venckunas T, Degens H. Methionine restriction plus overload improves skeletal muscle and metabolic health in old mice on a high fat diet. Sci Rep. 2021;11(1):1260. [CrossRef]
- Ham DJ, Borsch A, Chojnowska K, Lin S, Leuchtmann AB, Ham AS, et al. Distinct and additive effects of calorie restriction and rapamycin in aging skeletal muscle. Nat Commun. 2022;13(1):2025. [CrossRef]
- Lv S, Shen Q, Li H, Chen Q, Xie W, Li Y, et al. Caloric restriction delays age-related muscle atrophy by inhibiting 11beta-HSD1 to promote the differentiation of muscle stem cells. Front Med (Lausanne). 2022;9:1027055. [CrossRef]
- Swaminathan A, Cesanelli L, Venckunas T, Degens H. Impact of methionine restriction on muscle aerobic metabolism and hypertrophy in young and old mice on an obesogenic diet. Growth Factors. 2022;40(3-4):108-18. [CrossRef]
- Ersoy U, Altinpinar AE, Kanakis I, Alameddine M, Gioran A, Chondrogianni N, et al. Lifelong dietary protein restriction induces denervation and skeletal muscle atrophy in mice. Free Radic Biol Med. 2024;224:457-69. [CrossRef]
- Heymsfield SB, Yang S, McCarthy C, Brown JB, Martin CK, Redman LM, et al. Proportion of caloric restriction-induced weight loss as skeletal muscle. Obesity (Silver Spring). 2024;32(1):32-40. [CrossRef]
- Li S, Zhong H, Wang Z, Chen J, Huang Z, Zou T, et al. Dietary protein restriction regulates skeletal muscle fiber metabolic characteristics associated with the FGF21-ERK1/2 pathway. iScience. 2024;27(3):109249.
- Vermeij WP, Alyodawi K, van Galen I, von der Heide JL, Birkisdottir MB, Van't Sant LJ, et al. Improved health by combining dietary restriction and promoting muscle growth in DNA repair-deficient progeroid mice. J Cachexia Sarcopenia Muscle. 2024;15(6):2361-74. [CrossRef]
- Zador E. Paradoxical Effect of Caloric Restriction on Overload-Induced Muscle Growth in Diastolic Heart Failure. JACC Basic Transl Sci. 2024;9(2):241-3. [CrossRef]
- Zhang TR, Chiang CH, Hsu TC, Wang CY, Chen CY. Age and dietary restriction modulate mitochondrial quality in quadriceps femoris muscle of male mice. Biogerontology. 2024;25(3):447-59. [CrossRef]
- Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell. 2006;127(6):1109-22. [CrossRef]
- Jiang QY, Cheng XF, Cui YY, Xia Q, Yan XY, Zhang MY, et al. Resveratrol regulates skeletal muscle fibers switching through the AdipoR1-AMPK-PGC-1α pathway. Food & Function. 2019;10(6):3334-43. [CrossRef]
- Ringholm S, Olesen J, Pedersen JT, Brandt CT, Halling JF, Hellsten Y, et al. Effect of lifelong resveratrol supplementation and exercise training on skeletal muscle oxidative capacity in aging mice; impact of PGC-1α. Experimental Gerontology. 2013;48(11):1311-8. [CrossRef]
- Higashida K, Kim SH, Jung SR, Asaka M, Holloszy JO, Han DH. Effects of resveratrol and SIRT1 on PGC-1alpha activity and mitochondrial biogenesis: a reevaluation. PLoS Biol. 2013;11(7):e1001603.
- Alway SE, McCrory JL, Kearcher K, Vickers A, Frear B, Gilleland DL, et al. Resveratrol Enhances Exercise-Induced Cellular and Functional Adaptations of Skeletal Muscle in Older Men and Women. J Gerontol A Biol Sci Med Sci. 2017;72(12):1595-606. [CrossRef]
- Sin TK, Yung BY, Siu PM. Modulation of SIRT1-Foxo1 signaling axis by resveratrol: implications in skeletal muscle aging and insulin resistance. Cell Physiol Biochem. 2015;35(2):541-52. [CrossRef]
- Wang DT, Yin Y, Yang YJ, Lv PJ, Shi Y, Lu L, et al. Resveratrol prevents TNF-alpha-induced muscle atrophy via regulation of Akt/mTOR/FoxO1 signaling in C2C12 myotubes. Int Immunopharmacol. 2014;19(2):206-13. [CrossRef]
- Dirks Naylor AJ. Cellular effects of resveratrol in skeletal muscle. Life Sci. 2009;84(19-20):637-40.
- Gordon BS, Delgado Diaz DC, Kostek MC. Resveratrol decreases inflammation and increases utrophin gene expression in the mdx mouse model of Duchenne muscular dystrophy. Clin Nutr. 2013;32(1):104-11. [CrossRef]
- Su LY, Huang WC, Kan NW, Tung TH, Huynh LBP, Huang SY. Effects of Resveratrol on Muscle Inflammation, Energy Utilisation, and Exercise Performance in an Eccentric Contraction Exercise Mouse Model. Nutrients. 2023;15(1). [CrossRef]
- Sadeghi A, Seyyed Ebrahimi SS, Golestani A, Meshkani R. Resveratrol Ameliorates Palmitate-Induced Inflammation in Skeletal Muscle Cells by Attenuating Oxidative Stress and JNK/NF-kappaB Pathway in a SIRT1-Independent Mechanism. J Cell Biochem. 2017;118(9):2654-63. [CrossRef]
- Tang H, Shrager JB, Goldman D. Rapamycin protects aging muscle. Aging (Albany NY). 2019;11(16):5868-70. [CrossRef]
- Ramos FJ, Chen SC, Garelick MG, Dai DF, Liao CY, Schreiber KH, et al. Rapamycin reverses elevated mTORC1 signaling in lamin A/C-deficient mice, rescues cardiac and skeletal muscle function, and extends survival. Sci Transl Med. 2012;4(144):144ra03.
- Dickinson JM, Fry CS, Drummond MJ, Gundermann DM, Walker DK, Glynn EL, et al. Mammalian Target of Rapamycin Complex 1 Activation Is Required for the Stimulation of Human Skeletal Muscle Protein Synthesis by Essential Amino Acids. J Nutr. 2011;141(5):856-62. [CrossRef]
- Bolster DR, Kubica N, Crozier SJ, Williamson DL, Farrell PA, Kimball SR, et al. Immediate response of mammalian target of rapamycin (mTOR)-mediated signalling following acute resistance exercise in rat skeletal muscle. J Physiol. 2003;553(Pt 1):213-20. [CrossRef]
- Ghaffari MH, Schuh K, Dusel G, Frieten D, Koch C, Prehn C, et al. Mammalian target of rapamycin signaling and ubiquitin-proteasome-related gene expression in skeletal muscle of dairy cows with high or normal body condition score around calving. J Dairy Sci. 2019;102(12):11544-60. [CrossRef]
- Kawakami Y, Hambright WS, Takayama K, Mu X, Lu A, Cummins JH, et al. Rapamycin Rescues Age-Related Changes in Muscle-Derived Stem/Progenitor Cells from Progeroid Mice. Mol Ther Methods Clin Dev. 2019;14:64-76. [CrossRef]
- Kido K, Sase K, Yokokawa T, Fujita S. Enhanced skeletal muscle insulin sensitivity after acute resistance-type exercise is upregulated by rapamycin-sensitive mTOR complex 1 inhibition. Sci Rep. 2020;10(1):8509. [CrossRef]
- Yang Y, Sadri H, Prehn C, Adamski J, Rehage J, Danicke S, et al. Proteasome activity and expression of mammalian target of rapamycin signaling factors in skeletal muscle of dairy cows supplemented with conjugated linoleic acids during early lactation. J Dairy Sci. 2020;103(3):2829-46. [CrossRef]
- Zhang Z, Xu HN, Li S, Jr AD, Chellappa K, Davis JG, et al. Rapamycin maintains NAD(+)/NADH redox homeostasis in muscle cells. Aging (Albany NY). 2020;12(18):17786-99. [CrossRef]
- Bhat OM, Yuan X, Kukreja RC, Li PL. Regulatory role of mammalian target of rapamycin signaling in exosome secretion and osteogenic changes in smooth muscle cells lacking acid ceramidase gene. FASEB J. 2021;35(7):e21732. [CrossRef]
- Elliehausen CJ, Minton DM, Nichol AD, Konopka AR. Skeletal muscle mitochondrial respiration in a model of age-related osteoarthritis is impaired after dietary rapamycin. Exp Gerontol. 2021;155:111579. [CrossRef]
- Orenduff MC, Coleman MF, Glenny EM, Huffman KM, Rezeli ET, Bareja A, et al. Differential effects of calorie restriction and rapamycin on age-related molecular and functional changes in skeletal muscle. Exp Gerontol. 2022;165:111841. [CrossRef]
- Sirago G, Picca A, Calvani R, Coelho-Junior HJ, Marzetti E. Mammalian Target of Rapamycin (mTOR) Signaling at the Crossroad of Muscle Fiber Fate in Sarcopenia. Int J Mol Sci. 2022;23(22). [CrossRef]
- Ding Y, Wang P, Li C, Zhang Y, Yang C, Zhou X, et al. Sodium Butyrate Induces Mitophagy and Apoptosis of Bovine Skeletal Muscle Satellite Cells through the Mammalian Target of Rapamycin Signaling Pathway. Int J Mol Sci. 2023;24(17). [CrossRef]
- Ato S, Oya C, Ogasawara R. Rapamycin administration causes a decrease in muscle contractile function and systemic glucose intolerance concomitant with reduced skeletal muscle Rictor, the mTORC2 component, expression independent of energy intake in young rats. PLoS One. 2024;19(12):e0312859. [CrossRef]
- Yu Y, Zhao Y, Teng F, Li J, Guan Y, Xu J, et al. Berberine Improves Cognitive Deficiency and Muscular Dysfunction via Activation of the AMPK/SIRT1/PGC-1a Pathway in Skeletal Muscle from Naturally Aging Rats. J Nutr Health Aging. 2018;22(6):710-7.
- Gomes AP, Duarte FV, Nunes P, Hubbard BP, Teodoro JS, Varela AT, et al. Berberine protects against high fat diet-induced dysfunction in muscle mitochondria by inducing SIRT1-dependent mitochondrial biogenesis. Bba-Mol Basis Dis. 2012;1822(2):185-95. [CrossRef]
- Jeong HW, Hsu KC, Lee JW, Ham M, Huh JY, Shin HJ, et al. Berberine suppresses proinflammatory responses through AMPK activation in macrophages. Am J Physiol-Endoc M. 2009;296(4):E955-E64. [CrossRef]
- Nohara K, Mallampalli V, Nemkov T, Wirianto M, Yang J, Ye Y, et al. Nobiletin fortifies mitochondrial respiration in skeletal muscle to promote healthy aging against metabolic challenge. Nat Commun. 2019;10(1):3923. [CrossRef]
- Potue P, Wunpathe C, Maneesai P, Kukongviriyapan U, Prachaney P, Pakdeechote P. Nobiletin alleviates vascular alterations through modulation of Nrf-2/HO-1 and MMP pathways in l-NAME induced hypertensive rats. Food Funct. 2019;10(4):1880-92.
- Amarsanaa K, Kim HJ, Ko EA, Jo J, Jung SC. Nobiletin Exhibits Neuroprotective Effects against Mitochondrial Complex I Inhibition via Regulating Apoptotic Signaling. Exp Neurobiol. 2021;30(1):73-86.
- Wang HH, Sun YN, Qu TQ, Sang XQ, Zhou LM, Li YX, et al. Nobiletin Prevents D-Galactose-Induced C2C12 Cell Aging by Improving Mitochondrial Function. Int J Mol Sci. 2022;23(19). [CrossRef]
- Lee SH, Li XH, Lu QY, Zhan CL, Kim JD, Lee GH, et al. Nobiletin enhances mitochondrial function by regulating SIRT1/PGC-1alpha signaling in porcine oocytes during in vitro maturation. Biochem Biophys Res Commun. 2024;706:149747.
- Yao S, Yuan Y, Zhang H, Meng X, Jin L, Yang J, et al. Berberine attenuates the abnormal ectopic lipid deposition in skeletal muscle. Free Radic Biol Med. 2020;159:66-75. [CrossRef]
- Li X, Xu S, Zhang Y, Li K, Gao XJ, Guo MY. Berberine Depresses Inflammation and Adjusts Smooth Muscle to Ameliorate Ulcerative Colitis of Cats by Regulating Gut Microbiota. Microbiol Spectr. 2022;10(6):e0320722. [CrossRef]
- Wu Y, Yang Y, Du C, Peng X, Fan W, Chang B, et al. Berberine attenuates obesity-induced skeletal muscle atrophy via regulation of FUNDC1 in skeletal muscle of mice. Sci Rep. 2025;15(1):4918. [CrossRef]
- Nguyen-Ngo C, Salomon C, Quak S, Lai A, Willcox JC, Lappas M. Nobiletin exerts anti-diabetic and anti-inflammatory effects in an in vitro human model and in vivo murine model of gestational diabetes. Clin Sci (Lond). 2020;134(6):571-92.
- Yang G, Lin CC, Yang Y, Yuan L, Wang P, Wen X, et al. Nobiletin Prevents Trimethylamine Oxide-Induced Vascular Inflammation via Inhibition of the NF-kappaB/MAPK Pathways. J Agric Food Chem. 2019;67(22):6169-76. [CrossRef]
- Li XX, Chen SG, Yue GG, Kwok HF, Lee JK, Zheng T, et al. Natural flavone tricin exerted anti-inflammatory activity in macrophage via NF-kappaB pathway and ameliorated acute colitis in mice. Phytomedicine. 2021;90:153625.
- Chang BY, Bae JH, Lim CY, Kim YH, Kim TY, Kim SY. Tricin-enriched Zizania latifolia ameliorates non-alcoholic fatty liver disease through AMPK-dependent pathways. Food Sci Biotechnol. 2023;32(14):2117-29. [CrossRef]
- Zhu YF, Zhou XY, Lan C, Wen YP, Fu HJ, Li ZC, et al. Tricin Delays Aging and Enhances Muscle Function via Activating AMPK-Mediated Autophagy in Diverse Model Organisms. J Agric Food Chem. 2025;73(17):10246-64. [CrossRef]
- Fletcher RS, Ratajczak J, Doig CL, Oakey LA, Callingham R, Da Silva Xavier G, et al. Nicotinamide riboside kinases display redundancy in mediating nicotinamide mononucleotide and nicotinamide riboside metabolism in skeletal muscle cells. Mol Metab. 2017;6(8):819-32. [CrossRef]
- Elhassan YS, Kluckova K, Fletcher RS, Schmidt MS, Garten A, Doig CL, et al. Nicotinamide Riboside Augments the Aged Human Skeletal Muscle NAD(+) Metabolome and Induces Transcriptomic and Anti-inflammatory Signatures. Cell Rep. 2019;28(7):1717-28 e6. [CrossRef]
- Khan NA, Auranen M, Paetau I, Pirinen E, Euro L, Forsstrom S, et al. Effective treatment of mitochondrial myopathy by nicotinamide riboside, a vitamin B3. Embo Mol Med. 2014;6(6):721-31. [CrossRef]
- Liu Y, Ge M, Xiao X, Lu Y, Zhao W, Zheng K, et al. Sarcosine decreases in sarcopenia and enhances muscle regeneration and adipose thermogenesis by activating anti-inflammatory macrophages. Nat Aging. 2025. [CrossRef]
- Cichello SA, Weisinger RS, Schuijers J, Jois M. 1-Sarcosine-angiotensin II infusion effects on food intake, weight loss, energy expenditure, and skeletal muscle UCP3 gene expression in a rat model. J Cachexia Sarcopenia Muscle. 2014;5(3):239-46.
- Ryu D, Mouchiroud L, Andreux PA, Katsyuba E, Moullan N, Nicolet-Dit-Felix AA, et al. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat Med. 2016;22(8):879-88. [CrossRef]
- Rodriguez J, Pierre N, Naslain D, Bontemps F, Ferreira D, Priem F, et al. Urolithin B, a newly identified regulator of skeletal muscle mass. J Cachexia Sarcopenia Muscle. 2017;8(4):583-97. [CrossRef]
- Luan P, D'Amico D, Andreux PA, Laurila PP, Wohlwend M, Li H, et al. Urolithin A improves muscle function by inducing mitophagy in muscular dystrophy. Sci Transl Med. 2021;13(588). [CrossRef]
- Singh A, D'Amico D, Andreux PA, Fouassier AM, Blanco-Bose W, Evans M, et al. Urolithin A improves muscle strength, exercise performance, and biomarkers of mitochondrial health in a randomized trial in middle-aged adults. Cell Rep Med. 2022;3(5):100633. [CrossRef]
- Ghosh N, Das A, Biswas N, Gnyawali S, Singh K, Gorain M, et al. Urolithin A augments angiogenic pathways in skeletal muscle by bolstering NAD(+) and SIRT1. Sci Rep. 2020;10(1):20184. [CrossRef]
- Faitg J, D'Amico D, Rinsch C, Singh A. Mitophagy Activation by Urolithin A to Target Muscle Aging. Calcif Tissue Int. 2024;114(1):53-9. [CrossRef]
- He X, Wu Z, Jiang J, Xu W, Yuan A, Liao F, et al. Urolithin A Protects against Hypoxia-Induced Pulmonary Hypertension by Inhibiting Pulmonary Arterial Smooth Muscle Cell Pyroptosis via AMPK/NF-kappaB/NLRP3 Signaling. Int J Mol Sci. 2024;25(15). [CrossRef]
- Moradi N, Champsi S, Hood DA. Sulforaphane, Urolithin A, and ZLN005 induce time-dependent alterations in antioxidant capacity, mitophagy, and mitochondrial biogenesis in muscle cells. Sports Med Health Sci. 2025;7(1):16-27. [CrossRef]
- Wilhelmsen A, Karagounis LG, Bennett AJ, D'Amico D, Fouassier AM, Jones SW, et al. The polyphenol metabolite urolithin A suppresses myostatin expression and augments glucose uptake in human skeletal muscle cells. Nutr Metab (Lond). 2025;22(1):12. [CrossRef]
- Walsh ME, Bhattacharya A, Sataranatarajan K, Qaisar R, Sloane L, Rahman MM, et al. The histone deacetylase inhibitor butyrate improves metabolism and reduces muscle atrophy during aging. Aging Cell. 2015;14(6):957-70. [CrossRef]
- Cruz-Jentoft AJ. Beta-Hydroxy-Beta-Methyl Butyrate (HMB): From Experimental Data to Clinical Evidence in Sarcopenia. Curr Protein Pept Sci. 2018;19(7):668-72. [CrossRef]
- Portal S, Eliakim A, Nemet D, Halevy O, Zadik Z. Effect of HMB supplementation on body composition, fitness, hormonal profile and muscle damage indices. J Pediatr Endocrinol Metab. 2010;23(7):641-50. [CrossRef]
- Slater GJ, Jenkins D. Beta-hydroxy-beta-methylbutyrate (HMB) supplementation and the promotion of muscle growth and strength. Sports Med. 2000;30(2):105-16.
- Alon T, Bagchi D, Preuss HG. Supplementing with beta-hydroxy-beta-methylbutyrate (HMB) to build and maintain muscle mass: a review. Res Commun Mol Pathol Pharmacol. 2002;111(1-4):139-51.
- Russ DW, Acksel C, Boyd IM, Maynard J, McCorkle KW, Edens NK, et al. Dietary HMB and beta-alanine co-supplementation does not improve in situ muscle function in sedentary, aged male rats. Appl Physiol Nutr Metab. 2015;40(12):1294-301.
- Shirvani H, Rahmati-Ahmadabad S, Kowsari E, Fry H, Kazemi M, Kaviani M. Effects of 2-week HMB-FA supplementation with or without eccentric resistance exercise on expression of some genes related to muscle protein turnover and serum irisin and IGF-1 concentrations. Gene. 2020;760:145018. [CrossRef]
- Cavallucci V, Pani G. The Leucine Catabolite and Dietary Supplement beta-Hydroxy-beta-Methyl Butyrate (HMB) as an Epigenetic Regulator in Muscle Progenitor Cells. Metabolites. 2021;11(8). [CrossRef]
- Tsuchiya Y, Ueda H, Sugita N, Ochi E. Low Dose of beta-Hydroxy-beta-Methylbutyrate (HMB) Alleviates Muscle Strength Loss and Limited Joint Flexibility following Eccentric Contractions. J Am Coll Nutr. 2021;40(3):211-8. [CrossRef]
- Viana MV, Becce F, Pantet O, Schmidt S, Bagnoud G, Thaden JJ, et al. Impact of beta-hydroxy-beta-methylbutyrate (HMB) on muscle loss and protein metabolism in critically ill patients: A RCT. Clin Nutr. 2021;40(8):4878-87.
- Prado CM, Orsso CE, Pereira SL, Atherton PJ, Deutz NEP. Effects of beta-hydroxy beta-methylbutyrate (HMB) supplementation on muscle mass, function, and other outcomes in patients with cancer: a systematic review. J Cachexia Sarcopenia Muscle. 2022;13(3):1623-41.
- Ferreira SC, Souza NP, Amado LRN, Melo JOF, Reis IA, Anastacio LR. The effect of beta-hydroxy beta-methylbutyrate (HMB) with nutritional counselling on anthropometric muscle mass markers, strength, functionality, and quality of life in patients on the waiting list for liver transplantation: A double-blind study. Nutrition. 2023;110:112021.
- Manzano M, Giron MD, Salto R, Burgio C, Reinoso A, Cabrera E, et al. Arginine and Lysine Supplementation Potentiates the Beneficial beta-Hydroxy ss-Methyl Butyrate (HMB) Effects on Skeletal Muscle in a Rat Model of Diabetes. Nutrients. 2023;15(22). [CrossRef]
- Cohen-Or M, Chapnik N, Froy O. beta-Hydroxy-beta-methylbutyrate (HMB) increases muscle mass and diminishes weight gain in high-fat-fed mice. J Nutr Biochem. 2025;142:109926. [CrossRef]
- Pham T, MacRae CL, Broome SC, D'Souza R F, Narang R, Wang HW, et al. MitoQ and CoQ10 supplementation mildly suppresses skeletal muscle mitochondrial hydrogen peroxide levels without impacting mitochondrial function in middle-aged men. Eur J Appl Physiol. 2020;120(7):1657-69. [CrossRef]
- Campbell MD, Duan J, Samuelson AT, Gaffrey MJ, Merrihew GE, Egertson JD, et al. Improving mitochondrial function with SS-31 reverses age-related redox stress and improves exercise tolerance in aged mice. Free Radic Biol Med. 2019;134:268-81. [CrossRef]
- Rudolph TE, Mayorga EJ, Roths M, Rhoads RP, Baumgard LH, Selsby JT. The effect of Mitoquinol (MitoQ) on heat stressed skeletal muscle from pigs, and a potential confounding effect of biological sex. J Therm Biol. 2021;97:102900. [CrossRef]
- Broome SC, Pham T, Braakhuis AJ, Narang R, Wang HW, Hickey AJR, et al. MitoQ supplementation augments acute exercise-induced increases in muscle PGC1alpha mRNA and improves training-induced increases in peak power independent of mitochondrial content and function in untrained middle-aged men. Redox Biol. 2022;53:102341. [CrossRef]
- Pin F, Huot JR, Bonetto A. The Mitochondria-Targeting Agent MitoQ Improves Muscle Atrophy, Weakness and Oxidative Metabolism in C26 Tumor-Bearing Mice. Front Cell Dev Biol. 2022;10:861622.
- Graham ZA, DeBerry JJ, Cardozo CP, Bamman MM. A 50 kdyne contusion spinal cord injury with or without the drug SS-31 was not associated with major changes in muscle mass or gene expression 14 d after injury in young male mice. Physiol Rep. 2021;9(4):e14751. [CrossRef]
- Graham ZA, DeBerry JJ, Cardozo CP, Bamman MM. SS-31 does not prevent or reduce muscle atrophy 7 days after a 65 kdyne contusion spinal cord injury in young male mice. Physiol Rep. 2022;10(10):e15266. [CrossRef]
- Loffredo FS, Steinhauser ML, Jay SM, Gannon J, Pancoast JR, Yalamanchi P, et al. Growth Differentiation Factor 11 Is a Circulating Factor that Reverses Age-Related Cardiac Hypertrophy. Cell. 2013;153(4):828-39. [CrossRef]
- Brack AS, Conboy MJ, Roy S, Lee M, Kuo CJ, Keller C, et al. Increased Wnt signaling during aging alters muscle stem cell fate and increases fibrosis. Science. 2007;317(5839):807-10. [CrossRef]
- Rebo J, Mehdipour M, Gathwala R, Causey K, Liu Y, Conboy MJ, et al. A single heterochronic blood exchange reveals rapid inhibition of multiple tissues by old blood. Nat Commun. 2016;7:13363. [CrossRef]
- Sahu A, Clemens ZJ, Shinde SN, Sivakumar S, Pius A, Bhatia A, et al. Regulation of aged skeletal muscle regeneration by circulating extracellular vesicles. Nat Aging. 2021;1(12):1148-61. [CrossRef]
- Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature. 2005;433(7027):760-4. [CrossRef]
- Conese M, Carbone A, Beccia E, Angiolillo A. The Fountain of Youth: A Tale of Parabiosis, Stem Cells, and Rejuvenation. Open Med (Wars). 2017;12:376-83. [CrossRef]
- Fish LA, Fallon JR. Multiple MuSK signaling pathways and the aging neuromuscular junction. Neurosci Lett. 2020;731:135014. [CrossRef]
- Zhou Z, Dun L, Wei B, Gan Y, Liao Z, Lin X, et al. Musk Ketone Induces Neural Stem Cell Proliferation and Differentiation in Cerebral Ischemia via Activation of the PI3K/Akt Signaling Pathway. Neuroscience. 2020;435:1-9. [CrossRef]
- Madison RD, Robinson GA. Muscle-Derived Extracellular Vesicles Influence Motor Neuron Regeneration Accuracy. Neuroscience. 2019;419:46-59. [CrossRef]
- Zhou J, Liu B, Liang C, Li Y, Song YH. Cytokine Signaling in Skeletal Muscle Wasting. Trends Endocrinol Metab. 2016;27(5):335-47. [CrossRef]
- Tang Y, Reay DP, Salay MN, Mi MY, Clemens PR, Guttridge DC, et al. Inhibition of the IKK/NF-kappaB pathway by AAV gene transfer improves muscle regeneration in older mdx mice. Gene Ther. 2010;17(12):1476-83. [CrossRef]
- Messina S, Bitto A, Aguennouz M, Minutoli L, Monici MC, Altavilla D, et al. Nuclear factor kappa-B blockade reduces skeletal muscle degeneration and enhances muscle function in Mdx mice. Exp Neurol. 2006;198(1):234-41. [CrossRef]
- Huang L, Li M, Deng C, Qiu J, Wang K, Chang M, et al. Potential Therapeutic Strategies for Skeletal Muscle Atrophy. Antioxidants (Basel). 2022;12(1). [CrossRef]
- Cohen TJ, Waddell DS, Barrientos T, Lu Z, Feng G, Cox GA, et al. The histone deacetylase HDAC4 connects neural activity to muscle transcriptional reprogramming. J Biol Chem. 2007;282(46):33752-9. [CrossRef]
- Miska EA, Langley E, Wolf D, Karlsson C, Pines J, Kouzarides T. Differential localization of HDAC4 orchestrates muscle differentiation. Nucleic Acids Res. 2001;29(16):3439-47.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



