1. Introduction
Of its many functions, the lung is essential to oxygenate blood and assist in exchange of carbon dioxide with the environment through a closely integrated system of blood vessels and conducting airways [
1]. The right and left lungs are divided into three and two lobes, respectively, aerated by the conducting system which divides from the trachea into bronchi and further branches until microscopic terminal alveoli are formed and lined by alveolar capillaries [
2]. Of note, the lung receives a dual blood supply by the pulmonary arteries, which carry deoxygenated blood to the alveoli, as well as the bronchial arteries, which carry oxygenated blood to the parenchyma. This blood mixes before returning to the heart through the pulmonary veins [
3]. The arterial system derived from the pulmonary arteries, tracks closely with the airways, while the bronchial arteries supply lymph nodes, visceral pleura, nerves, and pulmonary vasa vasorum in addition to the lung parenchyma [
3] tracking more loosely along these structures after associating with the main pulmonary bronchi [
4]. The bronchial circulation is usually derived from branches of the thoracic aorta and occasionally the pulmonary circulation.
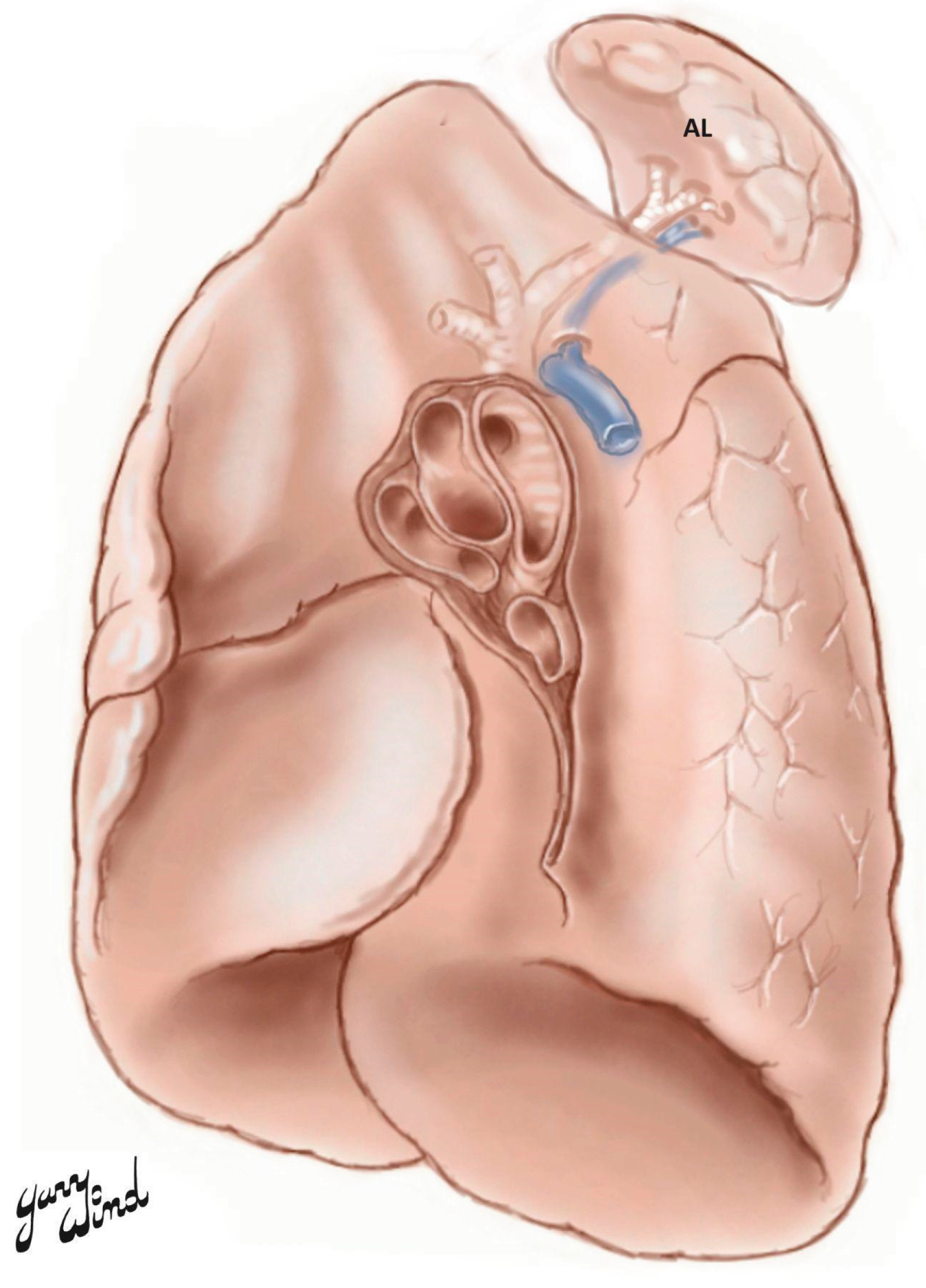
The azygos lobe (AL) was first described by Dr. Heinrich August Wrisberg in 1777 regarding a three-year-old boy in which there were apparent configurations of vascular bilaterally, resulting in bilateral clefting [
5]. An AL occurs when the posterior cardinal vein fails to migrate past the apex of a lung and instead, penetrates the nearby lobe [
6]. Left-sided ALs are rare, where the majority are on the right side [
7]. Plural layers that follow the vein entrap a portion of the right upper lobe [
6,
8]. The resulting structure resembles mesentery due to two folds of parietal and two folds of visceral plural [
6]. Additionally, the bronchial and arterial connections arise from the apical or posterior segments of the right upper lobe. The azygos vein (AV) terminates in the superior vena cava (SVC), and occasionally the right brachiocephalic vein (BCV) [
6]. The classification system set out in 1919 by Stibbe continues to be used to describe the position of the azygos fissure (AF) as shown in
Table 1, below.
A fine convex line represents the AF between the right upper lobe and the AL on radiography [
6]. This corresponds to a small triangular-shaped extra-plural tissue between the prior layers of the plural, also known as the trigone parietale [
6]. As a consequence of development, the AL may penetrate deeply behind the inferior vena cava, which can be seen on lateral chest radiographs and cross-sectional imaging [
6]. The right BCV–SVC complex may be visible on a chest radiograph due to an intrapulmonary course of these veins; however, this is rare [
6]. The right BCV-SVC complex course is variable and has not been well described in patients with an AL. Of note, anatomic differences, such as scars, bullae, displaced fissures, and supernumerary fissures, may mimic an AF; however, a false AL is identified when the AV is not present [
6].
2. Case Description
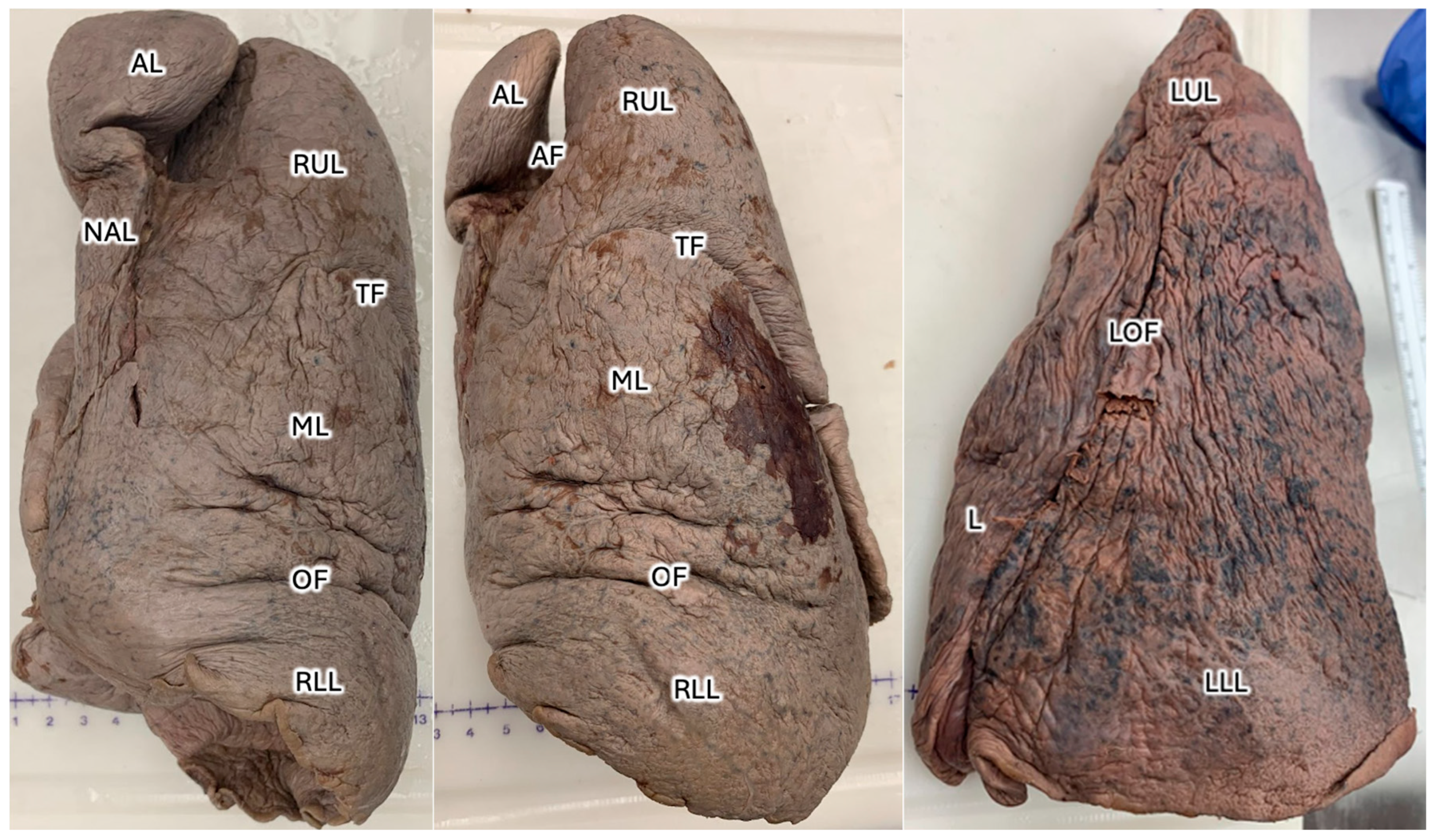
Sixty total donors were dissected at the Uniformed Services University, including 48 donors by first-year medical students, six dissected by the students within the graduate school of nursing, and six donors prosected by anatomy faculty at the Uniformed Services University of the Health Sciences, revealed an AL located on the posterior surface of the right apical lung in a 38-year-old male donor with cause of death listed as prostate cancer, hypertension, atrial fibrillation, and Type II diabetes mellitus. The patient’s medical history was notable for Ogilvie’s syndrome, multiple traumatic brain injuries (TBIs), and chronic respiratory failure. The AL, located on the posterior apical surface of the right lung, measured 5 cm in width and 8 cm in length at the time of dissection (
Figure 1). The left lung showed signs of atrophy and discoloration, suggestive of pollution or smoking earlier in life.
Measurements were obtained using ImageJ software, a free and open-source image analysis platform [
9], to extract specific dimensions from photographic images of the donor lungs (
Figure 2a,
Figure 3,
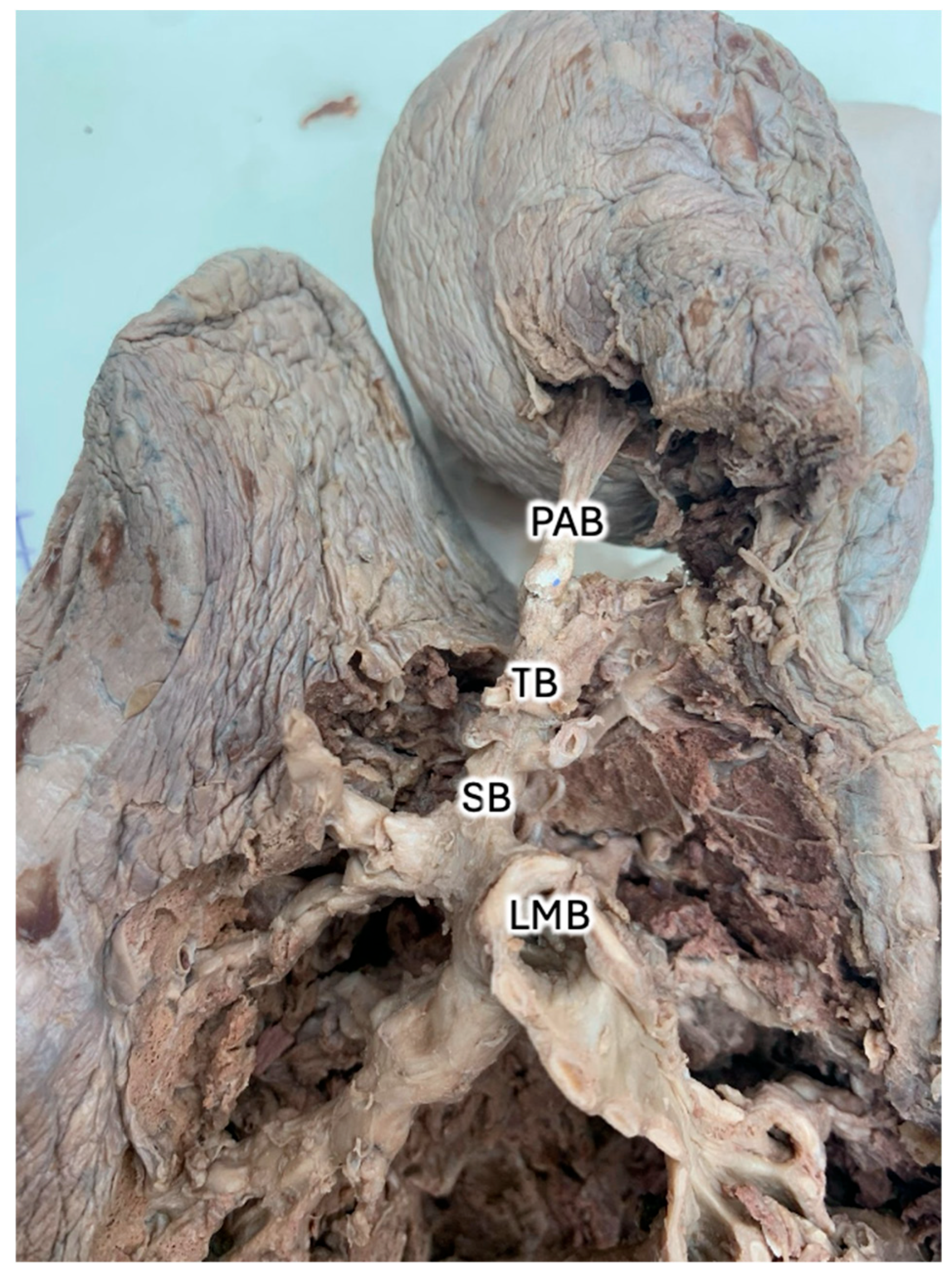
Figure 4). At its greatest lateral dimension, the AL measured 5.50cm. The widths of the right and left lungs were 11.86cm and 9.82cm, respectively. At its greatest vertical dimension, the AL measured 12.20cm. The lengths of the right and left lungs were 23.27cm and 15.81cm, respectively. Of note, the lobe was shaped such that there was a rounder terminus near the apex and an eccentric neck proximal to the hilum, and the lobe was oriented posteromedial to the right superior lobe. The terminal region reflects the greatest diameter of the AL; whereas the neck measured 2.56cm at its greatest width and 8.74cm at its greatest length. The angle of the AF is best observed from the anterior aspect and measured 17.01 degrees from the midline. Of note, the transverse fissure and right oblique fissure measure 49.64 degrees and 85.93 degrees from the midline, respectively. When observing the primary bronchiole leading to the AL, the greatest diameter was measured as 0.52 cm, and branches at 26.21 degrees from the proximal bronchiole.
3. Discussion
In addition to knowledge of typical accessory fissures [
10], azygos variants are important for radiologic evaluation. Pathologic states that affect the volume within the AL or others may affect the distribution of the horizontal and oblique fissures and accessory fissures. Subtle changes in fissure distribution are helpful in the assessment of volume loss associated with chronic lung disease [
11] Additionally, AL variants contribute to complications in surgeries to include sympathectomy and lobectomy [
12]. In the case of this patient, who experienced acute colonic pseudo-obstruction (Ogilvie’s Syndrome), colonic distention from lack of motility [
13] likely prevented full expansion of the lungs through diaphragmatic pressure. This may have contributed to azygos airway or vascular obstruction. Although Ogilvie’s syndrome is not listed as a cause of death for this patient, there have been examples of Ogilvie’s syndrome leading to respiratory compromise and death [
14]. In addition to other comorbidities, a pathologic state imposed on an azygos variant may have contributed to the patient’s chronic respiratory failure through poorly ventilated regions or contributing to dead space. Although hypoxic pulmonary vasoconstriction is a compensatory mechanism for ventilation/perfusion mismatch and can normally compensate for mild ventilatory defects, multiple pathologic states superimposed may overwhelm compensation [
15]. Additionally, the gravitational distribution of blood flow typically does not significantly utilize apical vasculature; however, this region may be under greater use while a patient is in a recumbent position [
2]. Given that this patient’s AL is posterolateral, it is likely that their AL received some gravity-dependent pulmonary blood flow.
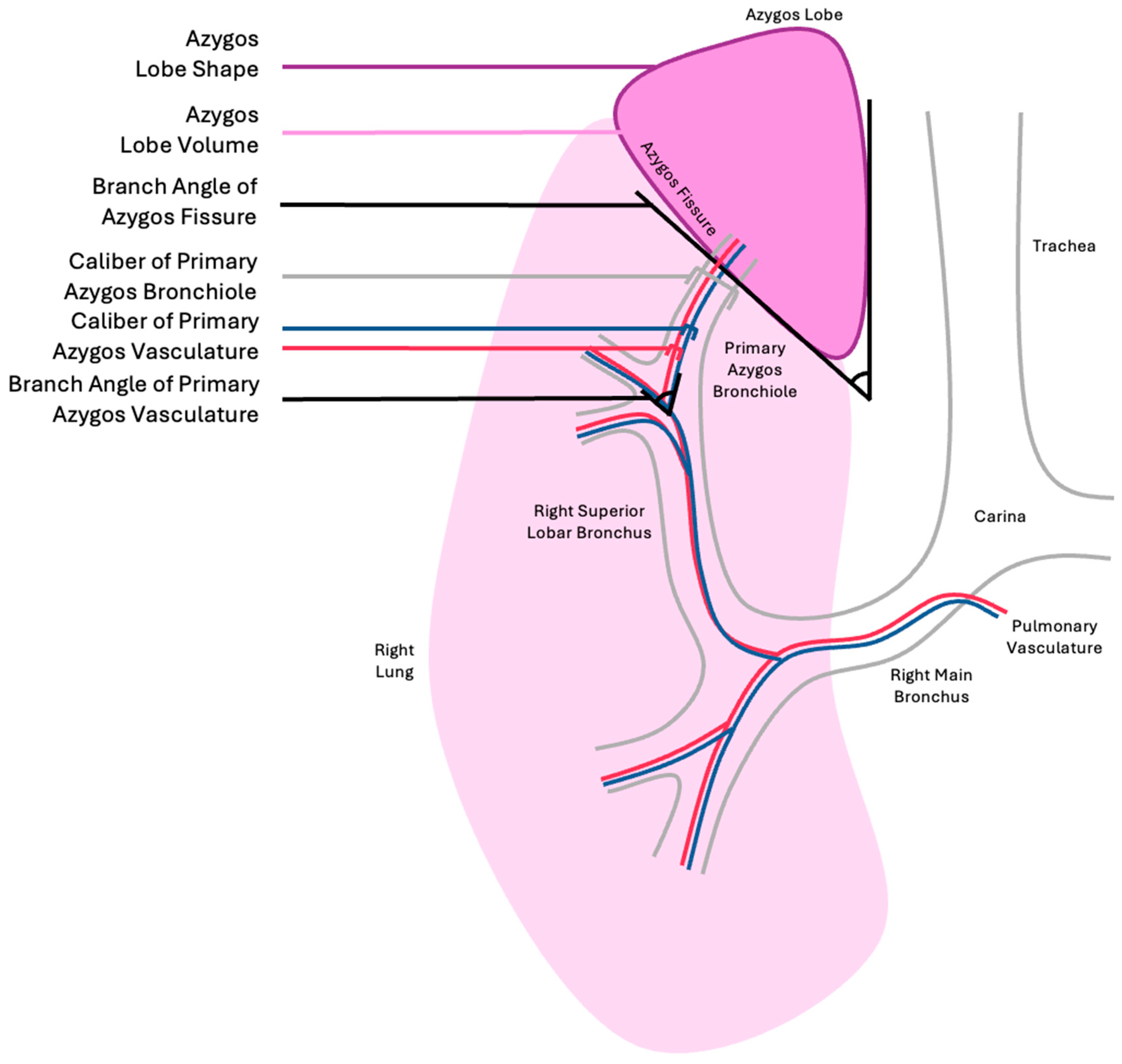
The historical classification of ALs is subjective and fails to capture important information. In this donor, the AL appeared to have a narrower neck than the apical portion. The AF angle may provide insight into the orientation of vascular and airway supplies; however, it may have limited utility in predicting the caliber of the airway or large pulmonary arteries/veins, and is only a proxy for the branching angle.
Figure 4 reflects that the direction of the primary azygos bronchiole after its bifurcation appears orthogonal to the azygos fissure; however, it is unclear if this relationship is consistent among ALs of varying dimensions. The arterial vessels associated with the donor AL were not adequately captured to comment on their branching pattern; however, pulmonary and bronchial vasculature tend to follow the conducting air structures and are assumed to be subject to similar geometric confinement as the primary azygos bronchiole and its derivatives. A more practical classification of ALs could take into account factors that would impact function, such as volume, shape, angle of AF branching, angle of primary azygos pulmonary vasculature branching, caliber of pulmonary vasculature, angle of primary azygos bronchiole branching, and caliber of primary azygos bronchiole, as reflected in
Table 2.
Of note, basic chest imaging, such as a chest x-ray, can only, on occasion, reveal the orientation of the AF without reflecting the lobe volume or the branching angle for the bronchopulmonary vasculature and airways. As evidenced by this donor, azygos bronchial branching may not closely mirror the angle of the AF. Computed tomography and magnetic resonance imaging may yield more useful data that could inform the classification of the AL for patient care. Further research is needed to create a generalizable AL classification scheme. It is unknown whether some AL variants definitively increase the risk of pulmonary dysfunction. Further research that accounts for vascular, airway, and AL morphometry through careful donor dissections or advanced radiography may provide insight into the risk stratification of patients with ALs to create a robust classification scheme.
4. Conclusions
An AL is an uncommon variant that is often unrecognized by the patient or their care team; however, its presence may have implications for the patient’s radiologic evaluation and surgical planning as well as their clinical course with regard to conditions that affect the lungs. An AL can be differentiated from a bulla or other pathological conditions by the presence of an AV on advanced imaging and occasionally on plain film chest X-rays. Further, the presence of additional tissue and vascular structures may serve as a nidus for complications in surgeries involving the posterior apical thorax. The donor with an AL variant described in this case had their clinical course affected by the presence of Olgolvie’s syndrome, which is known to negatively impact pulmonary function. Historical classifications of AL variants are not easily interpreted or applied widely to justify their sustainment. Enhanced classification of AL variants through more meaningful morphometric data is necessary to stratify patient risk for pulmonary compromise among other life-threatening conditions.
Author Contributions
Investigation: David Johnson, Guinevere Granite; Writing - original draft preparation, David Johnson, Guinevere Granite; Conceptualization: David Johnson, Guinevere Granite; Writing – Reviewing and editing: David Johnson, Guinevere Granite, Elizabeth Maynes, Jordan Dimitrakoff, Kerrie Lashley, Juan Jose Valenzuela-Fuenzalida, Maria Ximena Leighton, Yolanda Roth, Joanne Lenert; Validation: David Johnson, Guinevere Granite, Elizabeth Maynes, Jordan Dimitrakoff, Kerrie Lashley, Juan Jose Valenzuela-Fuenzalida, Juan Jose Valenzuela-Fuenzalida, Maria Ximena Leighton, Yolanda Roth, Joanne Lenert; Supervision: Guinevere Granite. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding
Informed Consent Statement
The donor included in this case report was a USU Willed Body donor that provided their remains to the Uniformed Services University of the Health Sciences for educational and research purposes. Informed consent was waived at the time of donation.
Acknowledgments
We would like to thank the family of our donor for their beneficent contribution. Without their generosity, this article would not have been possible. We would also like to thank Dr. Gary Wind for his artistic contribution of the article’s schematic. More of his artwork can be found on his educational website at http://www.vesalius.com. We would also like to thank 2LT Ava Dimmick, 2LT Jeffrey Kim, ENS Jamie Woodhead, and ENS Tyler Sturgill for their careful dissection of this donor.
Conflicts of Interest
The authors declare no conflict of interest.
Disclaimer
The contents of this presentation are the sole responsibility of the author(s) and do not necessarily reflect the views, opinions or policies of Uniformed Services University of the Health Sciences (USUHS), The Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc., the Department of Defense (DoD) or the Departments of the Army, Navy, or Air Force. Mention of trade names, commercial products, or organizations does not imply endorsement by the U.S. Government.
References
- Levitzky, M.G. Pulmonary Physiology. Tenth edition. McGraw Hill Medical; 2022.
- West, J.B.; Luks, A. West’s Respiratory Physiology : The Essentials. Eleventh edition. Wolters Kluwer; 2021. [Google Scholar]
- Suresh, K.; Shimoda, L.A. Lung Circulation. Compr Physiol. 2016, 6, 897–943. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cudkowicz, L.; Armstrong, J.B. Observations on the normal anatomy of the bronchial arteries. Thorax. 1951, 6, 343–358. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Boyden, E.A. The distribution of bronchi in gross anomalies of the right upper lobe particularly lobes subdivided by the azygos vein and those containing pre-eparterial bronchi. Radiology. 1952, 58, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Mata, J.; Cáceres, J.; Alegret, X.; Coscojuela, P.; De Marcos, J.A. Imaging of the azygos lobe: normal anatomy and variations. AJR Am J Roentgenol. 1991, 156, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Takasugi, J.E.; Godwin, J.D. Left azygos lobe. Radiology. 1989, 171, 133–134. [Google Scholar] [CrossRef] [PubMed]
- Stibbe, E.P. The Accessory Pulmonary Lobe of the Vena Azygos. J Anat. 1919, 53 (Pt 4), 305–314. [Google Scholar] [PubMed] [PubMed Central]
- Rasband, W.S.; ImageJ; US National Institutes of Health, Bethesda, Maryland, USA. https://imagej.net/ij/, 1997-2018.
- Godwin, J.D.; Tarver, R.D. Accessory fissures of the lung. AJR Am J Roentgenol. 1985, 144, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Kalchiem-Dekel, O.; Galvin, J.R.; Burke, A.P.; Atamas, S.P.; Todd, N.W. Interstitial Lung Disease and Pulmonary Fibrosis: A Practical Approach for General Medicine Physicians with Focus on the Medical History. J Clin Med. 2018, 7, 476. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tran, C.T.; Miao, K.H.; Lui, F. Anatomy, Thorax, Lung Azygos Lobe. [Updated 2023 Jul 24]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available online: https://www.ncbi.nlm.nih.gov/books/NBK518977/.
- Maloney, N.; Vargas, H.D. Acute intestinal pseudo-obstruction (Ogilvie's syndrome). Clin Colon Rectal Surg. 2005, 18, 96–101. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dewey, J.; Prahlow, J.A. Acute colonic pseudo-obstruction (Ogilvie syndrome) leading to respiratory compromise and death. J Forensic Sci. 2021, 66, 2515–2520. [Google Scholar] [CrossRef] [PubMed]
- Tarry, D.; Powell, M. Hypoxic Pulmonary Vasoconstriction. BJA Education. 2017, 17, 208–213. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).