Submitted:
16 July 2025
Posted:
17 July 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sample Preparation of Pre-Filled Syringes


2.2. Chemical Stability
2.3. Chromatographic Method
2.4. Physical Stability
3. Results
3.1. Validation of the Analytical Method
| Theoretical Concentration (µg/mL) |
20 (80%) |
25 (100%) | 30 (120%) |
|---|---|---|---|
| Mean (µg/mL) | 20,04 | 24,99 | 30,06 |
| SD | 0,03 | 0,02 | 0,21 |
| RSD% | 0,15 | 0,09 | 0,68 |
| Accuracy% | 100,20 | 99,98 | 100,12 |
| Theoretical Concentration (µg/mL) |
20 (80%) |
25 (100%) | 30 (120%) |
|---|---|---|---|
| Mean (µg/mL) | 20,04 | 25,04 | 30,10 |
| SD | 0,07 | 0,06 | 0,21 |
| RSD% | 0,33 | 0,23 | 0,71 |
| Accuracy% | 100,21 | 100,15 | 100,32 |
3.2. Stability Study
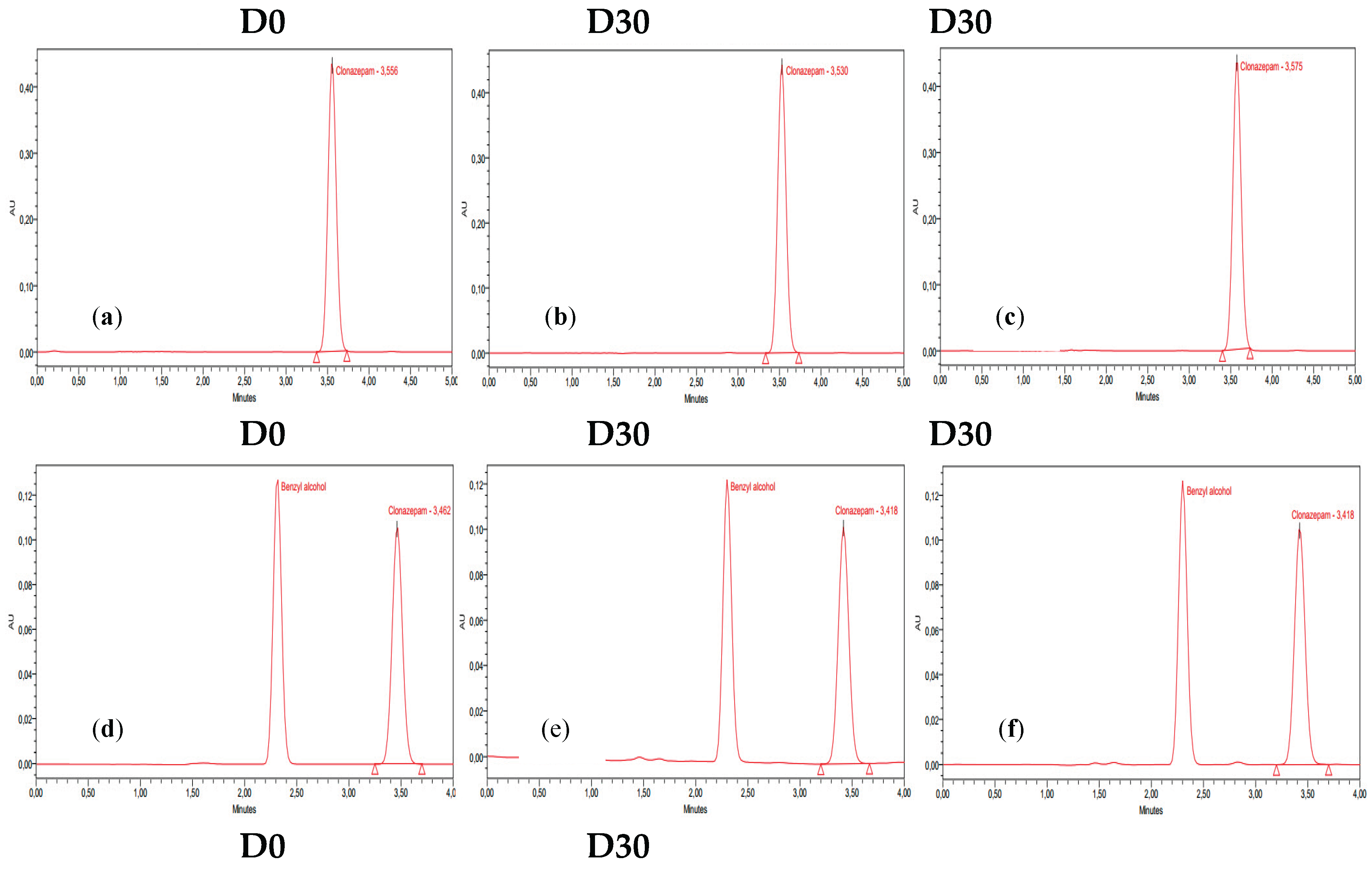
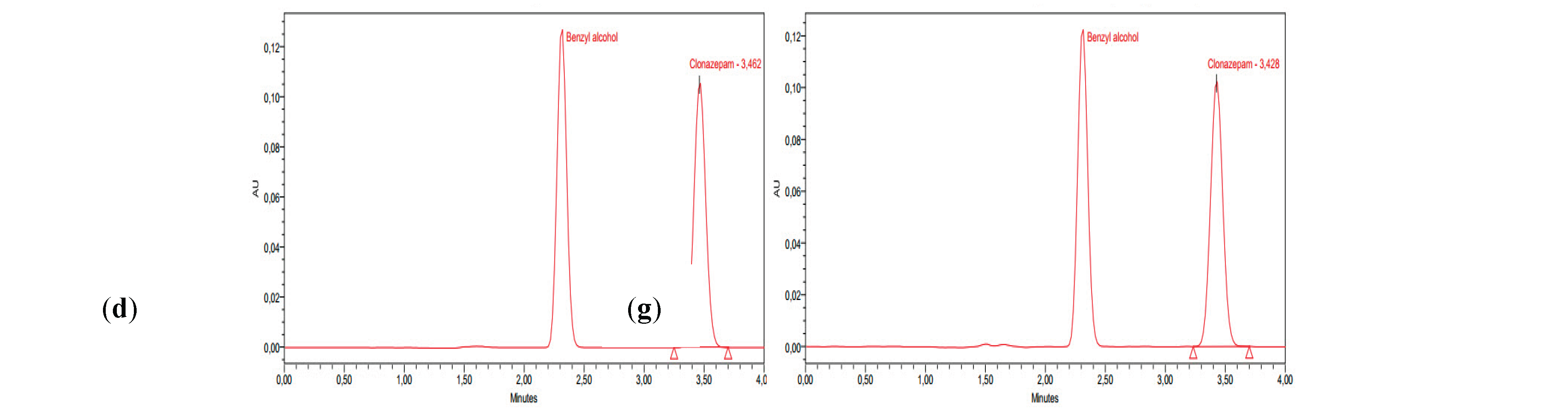
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| GABA | Gamma-aminobutyric acid |
| HPLC | High-performance liquid chromatography |
| ICH | International Conference on Harmonisation |
| INSST | Spanish National Institute for Safety and Health at Work |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| NIOSH | Health of the United States |
| R2 | Correlation coefficient |
| RSD% | Relative standard deviation |
| SD | Standard deviation |
| SEFH | Spanish Society of Hospital Pharmacy |
References
- Spanish Medicines and Medical Devices Agency. Datasheet of Rivotril 1 mg/mL concentrate and solvent for injectable solution. [Internet]. Datasheet of Rivotril 1 mg/mL concentrate and solvent for injectable solution [cited 2024 Aug 12];Available from: https://cima.aemps.es/cima/dochtml/ft/52332/FT_52332.html.
- Spanish Medicines and Medical Devices Agency. Datasheet of Rivotril 2,5 mg/mlL oral drops solution [Internet]. Datasheet of Rivotril 2,5 mg/mlL oral drops solution [cited 2024 Aug 12];Available from: https://cima.aemps.es/cima/dochtml/ft/52333/FT_52333.html#5-propiedades-farmacol-gicas.
- National Institute for Occupational Safety and Health. Hazardous drugs. Preventive measures for their preparation and administration [Internet]. Medicam. Peligr. Medidas Prev. Para Su Prep. Adm.2016 [cited 2024 Aug 12];Available from: https://www.insst.es/documentacion/catalogo-de-publicaciones/medicamentos-peligrosos.-medidas-de-prevencion-para-su-preparacion-y-administracion.
- Connor TH, MacKenzie, BA, DeBord DG, Trout DB, O’Callaghan JP, Ovesen JL, Whittaker C. Cincinnati, OH, National Institute for Occupational Safety and Health. NIOSH [2020]. NIOSH list of hazardous drugs in healthcare settings 2020 [Internet]. [cited 2024 Aug 5];Available from: https://www.cdc.gov/niosh/docket/review/docket233c/pdfs/DRAFT-NIOSH-Hazardous-Drugs-List-2020.pdf.
- Lal, R. Drugs in Pregnancy and Lactation: Improved Benefit-Risk Information [Internet]. 2015 [cited 2024 Aug 27];Available from: https://www.fda.gov/files/drugs/published/%22Drugs-in-Pregnancy-and-Lactation--Improved-Benefit-Risk-Information%22-January-22--2015-Issue.pdf.
- Asociación Española de Farmacéuticos de la Industria (AEFI). Validación de métodos analíticos. Monografía. Comisión de normas de buena fabricación y control de calidad. Barcelona: Edicion Hewlett Packard; 2001.
- European Directorate for the Quality of Medicines & HealthCare. European Pharmacopoeia. 11th ed. Estrasburgo: European Directorate for the Quality of Medicines & HealthCare; 2022.
- United States Pharmacopeial Convention. United States Pharmacopeia and National Formulary (USP-NF). 2022nd ed. Rockville: US Pharmacopeia Convention, Inc; 2022.
- Allen LV, Erickson MA. Stability of acetazolamide, allopurinol, azathioprine, clonazepam, and flucytosine in extemporaneously compounded oral liquids. Am J Health-Syst Pharm AJHP Off J Am Soc Health-Syst Pharm 1996;53(16):1944–9. [CrossRef]
- Polonini HC, Loures S, Lima LC, Ferreira AO, Brandão MAF. Stability of Atenolol, Clonazepam, Dexamethasone, Diclofenac Sodium, Diltiazem, Enalapril Maleate, Ketoprofen, Lamotrigine, Penicillamine-D, and Thiamine in SyrSpend SF PH4 Oral Suspensions. Int J Pharm Compd 2016;20(2):167–74.
- Abraham, J. International Conference On Harmonisation Of Technical Requirements For Registration Of Pharmaceuticals For Human Use [Internet]. In: Brouder A, Tietje C, editors. Handbook of Transnational Economic Governance Regimes. Brill; 2009 [cited 2018 Jun 6]. page 1041–54.Available from: http://booksandjournals.brillonline.com/content/books/10.1163/ej.9789004163300.i-1081.897. [CrossRef]
- Sagar Baliram PM. A Validated Stability–Indicating HPLC Method estimation of Clonazepam In the bulk drug and Pharmaceutical Dosage Form. Pharm Anal Acta [Internet] 2015 [cited 2018 May 20];06(02). Available from: https://www.omicsonline.org/open-access/a-validated-stabilityindicating-hplc-method-estimation-of-clonazepam-in-the-bulk-drug-and-pharmaceutical-dosage-form-2153-2435.1000332.php?aid=40328. [CrossRef]
- Statistical validation: Quantitative determination (General Explanations). Basle: Hoffman F. La Roche, 1987:1-9.
- N. Miller James, C. Miller Jane. Statistics and Chemometrics for Analytical Chemistry. 5a. Harlow: Pearson Education Limited; 2005. [CrossRef]
- Agencia Española de Medicamentos y Productos Sanitarios. Contaminación por partículas: particulas visibles. Real Farmacopea Española [Internet]. [cited 2024 Sep 10];Available from: https://extranet.boe.es/farmacopea/doc.php?id=20920.
- European Agency for Safety and Health at Work. Guidance for the safe management of hazardous medicinal products at work | Safety and health at work EU-OSHA [Internet]. 2023 [cited 2024 Aug 28];Available from: https://osha.europa.eu/en/publications/guidance-safe-management-hazardous-medicinal-products-work.
| Room temperature | Refrigeration conditions Protected from light |
|||||
|---|---|---|---|---|---|---|
| D0 | D30 | D0 | D30 | |||
| Average Recovery % of concentration | 100 | 100,33±0,01 | 100 | 97,82±0,02 | ||
| pH | 4,63±0,02 | 4,65±0,06 | 4,65±0,03 | 4,65±0,05 | ||
| Colour | Blue | Blue | Blue | Blue | ||
| Crystals≥10 µm/mL | 0 | 0 | 0 | 0 | ||
| Room temperature | Room temperature protected from light |
Refrigeration conditions protected from light |
||
|---|---|---|---|---|
| D0 | D30 | D30 | D30 | |
| Average Recovery % of concentration | 100 | 100,87±0,01 | 98,14±0,02 | 98,02±0,02 |
| pH | 4,15±0,08 | 4,27±0,15 | 4,30±0,07 | 4,17±0,04 |
| Colour | Transparent | Transparent | Transparent | Transparent |
| Crystals ≥10 µm/mL | 0 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




