Submitted:
02 July 2025
Posted:
03 July 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sample Preparation of Pre-Filled Syringes
2.2. Chemical Stability
2.3. Chromatographic Method
2.4. Physical Stability
3. Results
3.1. Validation of the Analytical Method
3.2. Stability Study
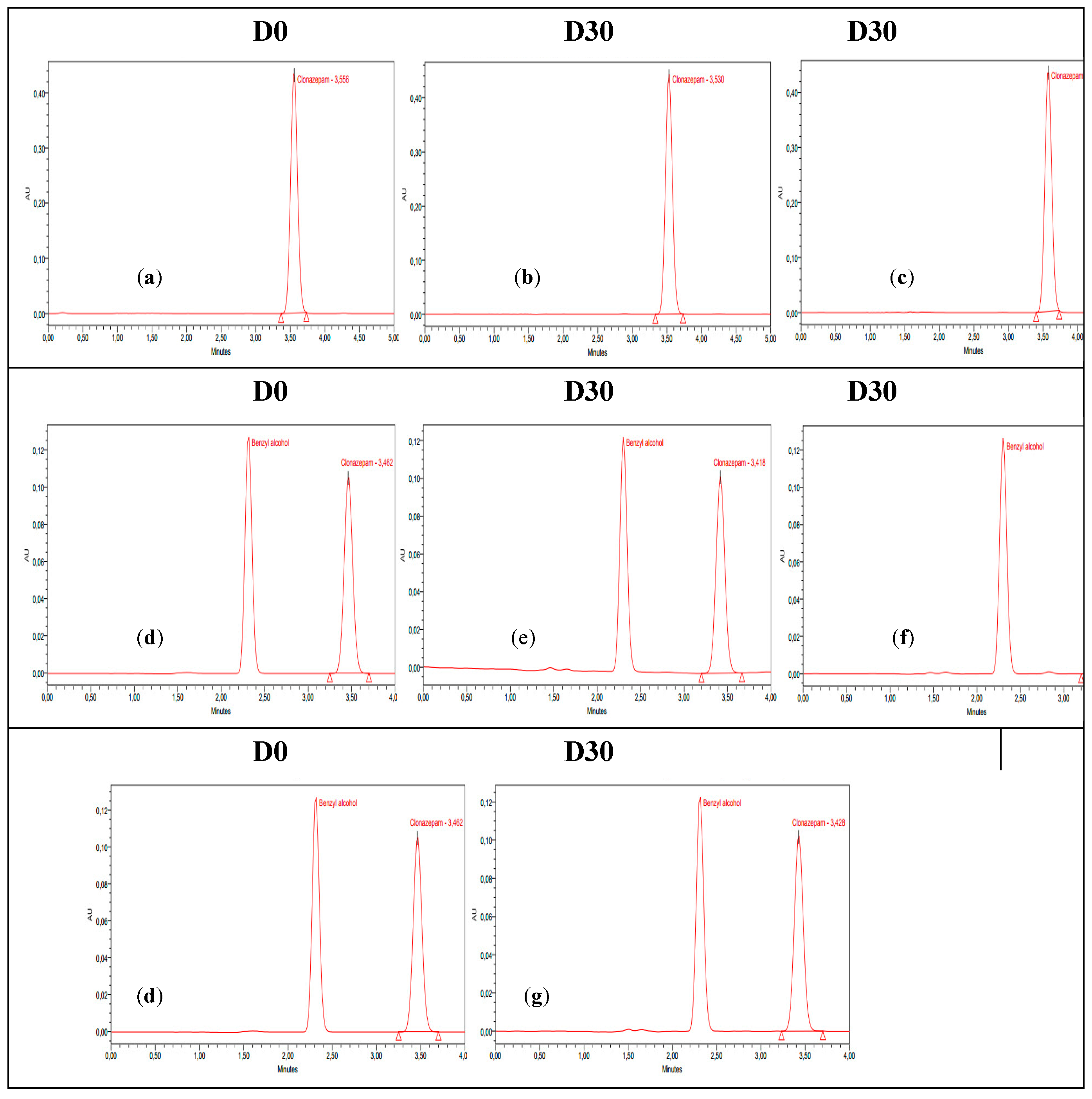
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| GABA | Gamma-aminobutyric acid |
| HPLC | High-performance liquid chromatography |
| ICH | International Conference on Harmonisation |
| INSST | Spanish National Institute for Safety and Health at Work |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| NIOSH | Health of the United States |
| R2 | Correlation coefficient |
| RSD% | Relative standard deviation |
| SD | Standard deviation |
| SEFH | Spanish Society of Hospital Pharmacy |
References
- Spanish Medicines and Medical Devices Agency. Datasheet of Rivotril 1 mg/mL concentrate and solvent for injectable solution. [Internet]. Datasheet of Rivotril 1 mg/mL concentrate and solvent for injectable solution [cited 2024 Aug 12];Available from: https://cima.aemps.es/cima/dochtml/ft/52332/FT_52332.
- Spanish Medicines and Medical Devices Agency. Datasheet of Rivotril 2,5 mg/mlL oral drops solution [Internet]. Datasheet of Rivotril 2,5 mg/mlL oral drops solution [cited 2024 Aug 12];Available from: https://cima.aemps.es/cima/dochtml/ft/52333/FT_52333.
- National Institute for Occupational Safety and Health. Hazardous drugs. Preventive measures for their preparation and administration [Internet]. Medicam. Peligr. Medidas Prev. Para Su Prep. Adm.2016 [cited 2024 Aug 12];Available from: https://www.insst.es/documentacion/catalogo-de-publicaciones/medicamentos-peligrosos.
- Connor TH, MacKenzie, BA, DeBord DG, Trout DB, O’Callaghan JP, Ovesen JL, Whittaker C. Cincinnati, OH, National Institute for Occupational Safety and Health. NIOSH [2020]. NIOSH list of hazardous drugs in healthcare settings 2020 [Internet]. [cited 2024 Aug 5];Available from: https://www.cdc.gov/niosh/docket/review/docket233c/pdfs/DRAFT-NIOSH-Hazardous-Drugs-List-2020.
- Lal, R. Drugs in Pregnancy and Lactation: Improved Benefit-Risk Information [Internet]. 2015 [cited 2024 Aug 27];Available from: https://www.fda.gov/files/drugs/published/%22Drugs-in-Pregnancy-and-Lactation--Improved-Benefit-Risk-Information%22-January-22--2015-Issue.
- Asociación Española de Farmacéuticos de la Industria (AEFI). Validación de métodos analíticos. Monografía. Comisión de normas de buena fabricación y control de calidad. Barcelona: Edicion Hewlett Packard; 2001.
- European Directorate for the Quality of Medicines & HealthCare. European Pharmacopoeia. 11th ed. Estrasburgo: European Directorate for the Quality of Medicines & HealthCare; 2022.
- United States Pharmacopeial Convention. United States Pharmacopeia and National Formulary (USP-NF). 2022nd ed. Rockville: US Pharmacopeia Convention, Inc; 2022.
- Allen LV, Erickson MA. Stability of acetazolamide, allopurinol, azathioprine, clonazepam, and flucytosine in extemporaneously compounded oral liquids. Am J Health-Syst Pharm AJHP Off J Am Soc Health-Syst Pharm 1996, 53, 1944–1949. [Google Scholar]
- Polonini HC, Loures S, Lima LC, Ferreira AO, Brandão MAF. Stability of Atenolol, Clonazepam, Dexamethasone, Diclofenac Sodium, Diltiazem, Enalapril Maleate, Ketoprofen, Lamotrigine, Penicillamine-D, and Thiamine in SyrSpend SF PH4 Oral Suspensions. Int J Pharm Compd 2016, 20, 167–174. [Google Scholar]
- Abraham, J. International Conference On Harmonisation Of Technical Requirements For Registration Of Pharmaceuticals For Human Use [Internet]. In: Brouder A, Tietje C, editors. Handbook of Transnational Economic Governance Regimes. Brill; 2009 [cited 2018 Jun 6]. page 1041–54. Available from: http://booksandjournals.brillonline.com/content/books/10.1163/ej.9789004163300.i-1081.
- Sagar Baliram, PM. A Validated Stability–Indicating HPLC Method estimation of Clonazepam In the bulk drug and Pharmaceutical Dosage Form. Pharm Anal Acta [Internet] 2015 [cited 2018 ];06(02). Available from: https://www.omicsonline.org/open-access/a-validated-stabilityindicating-hplc-method-estimation-of-clonazepam-in-the-bulk-drug-and-pharmaceutical-dosage-form-2153-2435.1000332.php? 20 May 4032. [Google Scholar]
- Statistical validation: Quantitative determination (General Explanations). Basle: Hoffman F. La Roche, 1987:1-9.
- N. Miller James, C. N. Miller James, C. Miller Jane. Statistics and Chemometrics for Analytical Chemistry. 5a. Harlow: Pearson Education Limited; 2005.
- Agencia Española de Medicamentos y Productos Sanitarios. Contaminación por partículas: particulas visibles. Real Farmacopea Española [Internet]. [cited 2024 Sep 10];Available from: https://extranet.boe.es/farmacopea/doc.php? 2092.
- European Agency for Safety and Health at Work. Guidance for the safe management of hazardous medicinal products at work | Safety and health at work EU-OSHA [Internet]. 2023 [cited 2024 Aug 28];Available from: https://osha.europa.

|
Theoretical concentration (µg/mL) |
20 (80%) | 25 (100%) | 30 (120%) |
| Mean (µg/mL) | 20,04 | 24,99 | 30,06 |
| SD | 0,03 | 0,02 | 0,21 |
| RSD% | 0,15 | 0,09 | 0,68 |
| Accuracy% | 100,20 | 99,98 | 100,12 |
|
Theoretical concentration (µg/mL) |
20 (80%) | 25 (100%) | 30 (120%) |
| Mean (µg/mL) | 20,04 | 25,04 | 30,10 |
| SD | 0,07 | 0,06 | 0,21 |
| RSD% | 0,33 | 0,23 | 0,71 |
| Accuracy% | 100,21 | 100,15 | 100,32 |
| Room temperature |
Refrigeration conditions Protected from light |
||||
|---|---|---|---|---|---|
| D0 | D30 | D0 | D30 | ||
| Average Recovery % of concentration | 100 | 100,33±0,01 | 100 | 97,82±0,02 | |
| pH | 4,63±0,02 | 4,65±0,06 | 4,65±0,03 | 4,65±0,05 | |
| Colour | Blue | Blue | Blue | Blue | |
| Crystals≥10 µm/mL | 0 | 0 | 0 | 0 | |
| Room temperature | Room temperature protected from light |
Refrigeration conditions protected from light |
||
|---|---|---|---|---|
| D0 | D30 | D30 | D30 | |
| Average Recovery % of concentration | 100 | 100,87±0,01 | 98,14±0,02 | 98,02±0,02 |
| pH | 4,15±0,08 | 4,27±0,15 | 4,30±0,07 | 4,17±0,04 |
| Colour | Transparent | Transparent | Transparent | Transparent |
| Crystals ≥10 µm/mL | 0 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




