Submitted:
01 July 2025
Posted:
03 July 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Huh7 Cell Culture, Infection, and Co-Culture Experiments
THP-1 Cell Culture, Infection, and Flow Cytometry Analysis
Exosome Isolation and Characterization
Plaque Assay
Exosome Protein Extraction and Cargo Profiling
Western Blotting
Overexpression Plasmid Transfection
RNA Immunoprecipitation (RIP) and Co-Immunoprecipitation Analysis
Quantitative Real-Time PCR Assay for DENV Genomic RNA and Host Gene Transcripts
Single-Cell Transcriptomic Analysis
Data Availability
Ethics Statement
Results
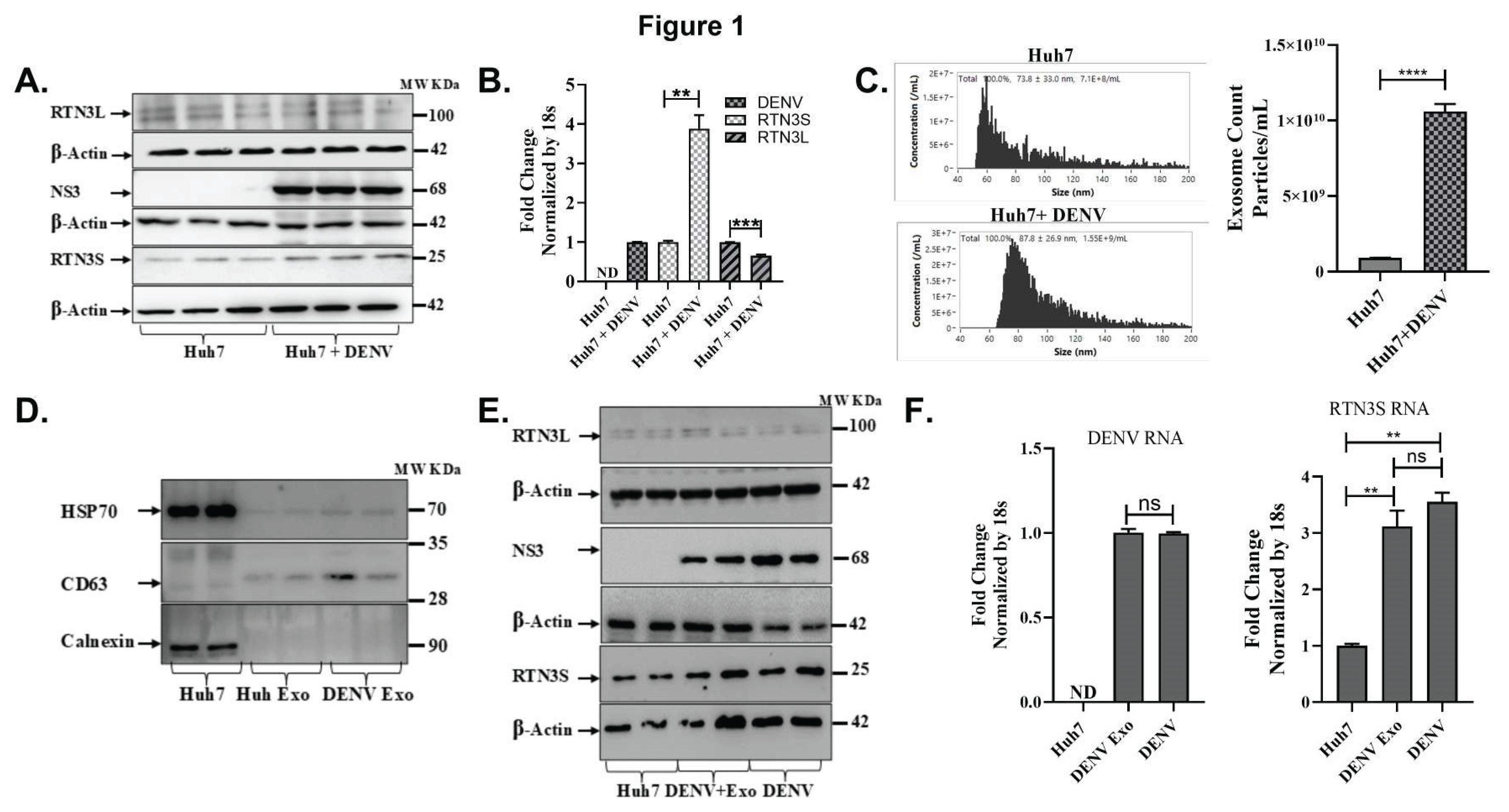
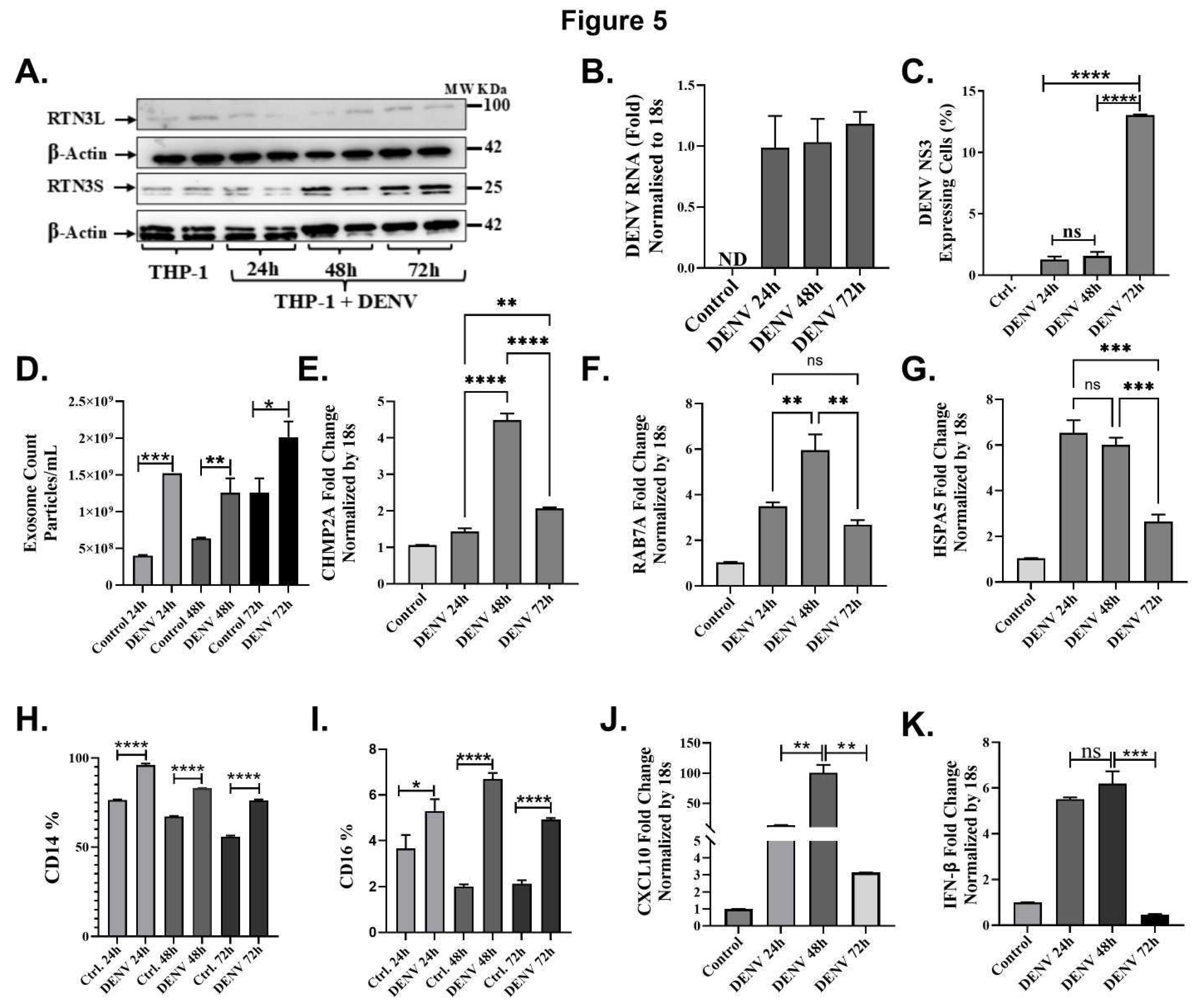
DENV Infection Upregulates RTN3 Isoforms and Exosome Secretion
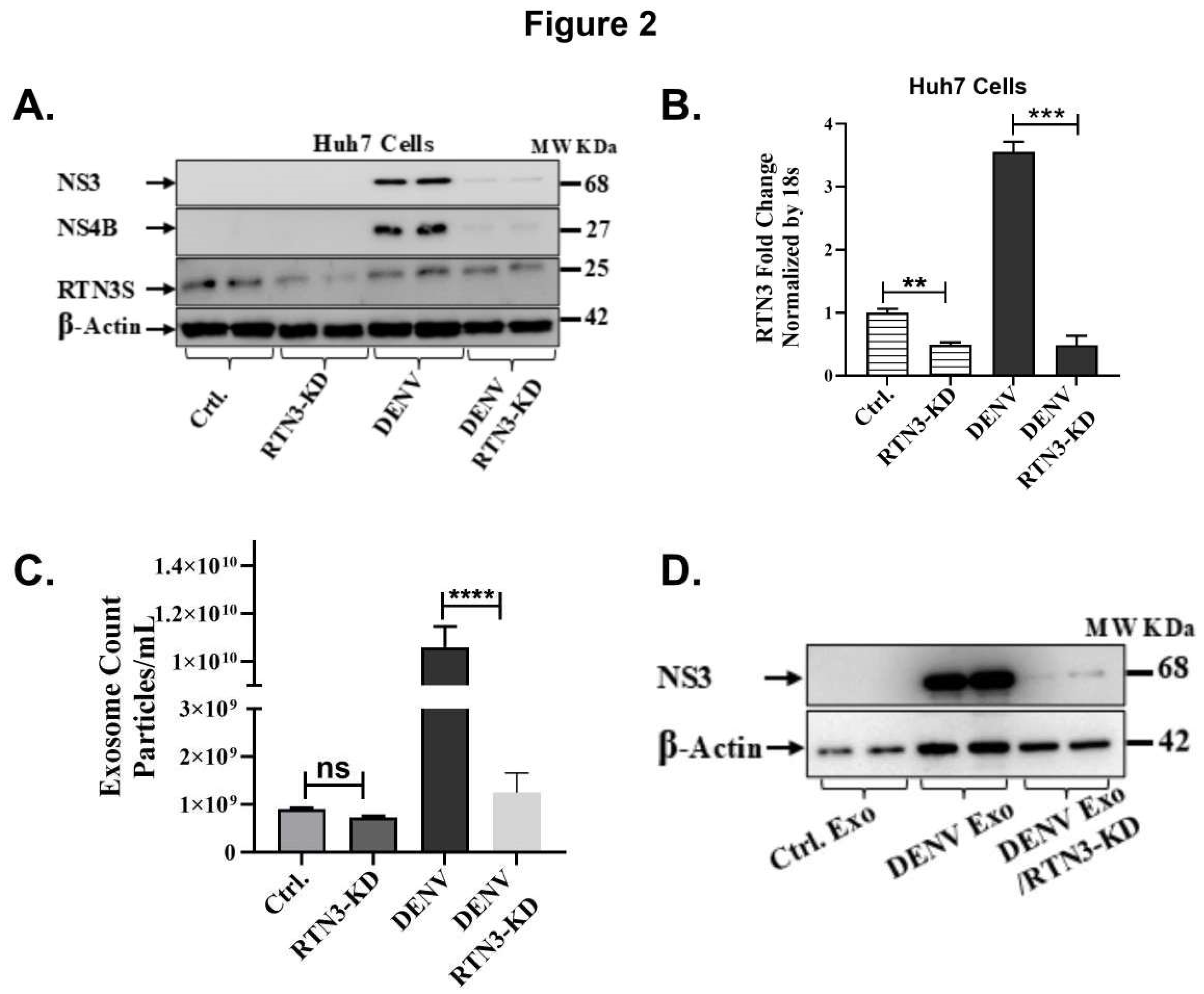
RTN3 Is Required for Virus-Induced Exosome Release and Viral Cargo Loading
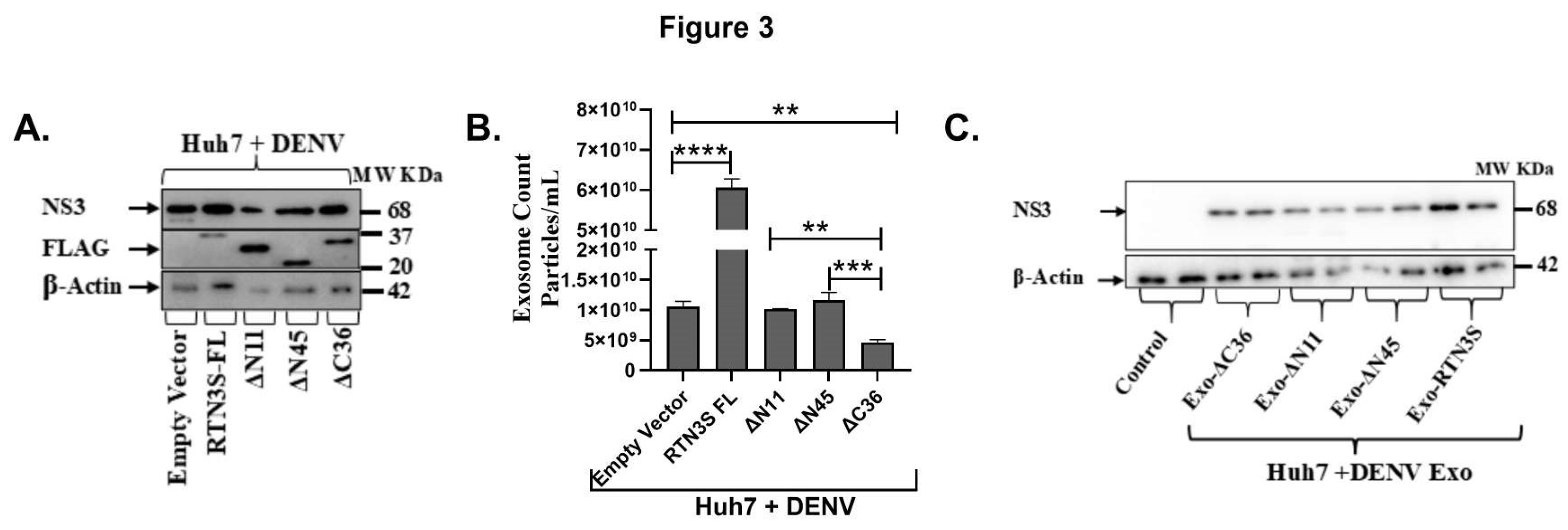
RTN3S Overexpression and Truncated Mutants Differentially Affect Exosomal Viral Packaging and Infectivity
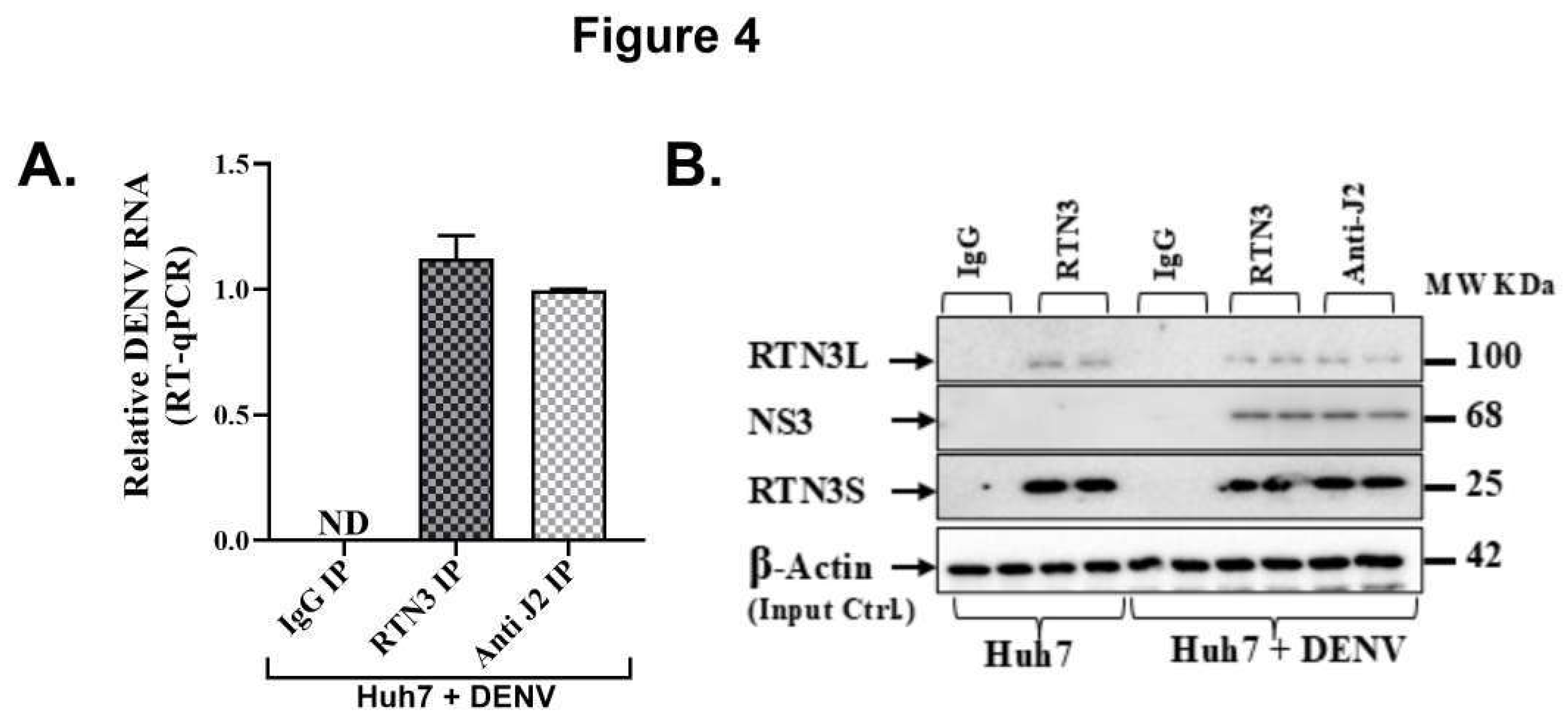
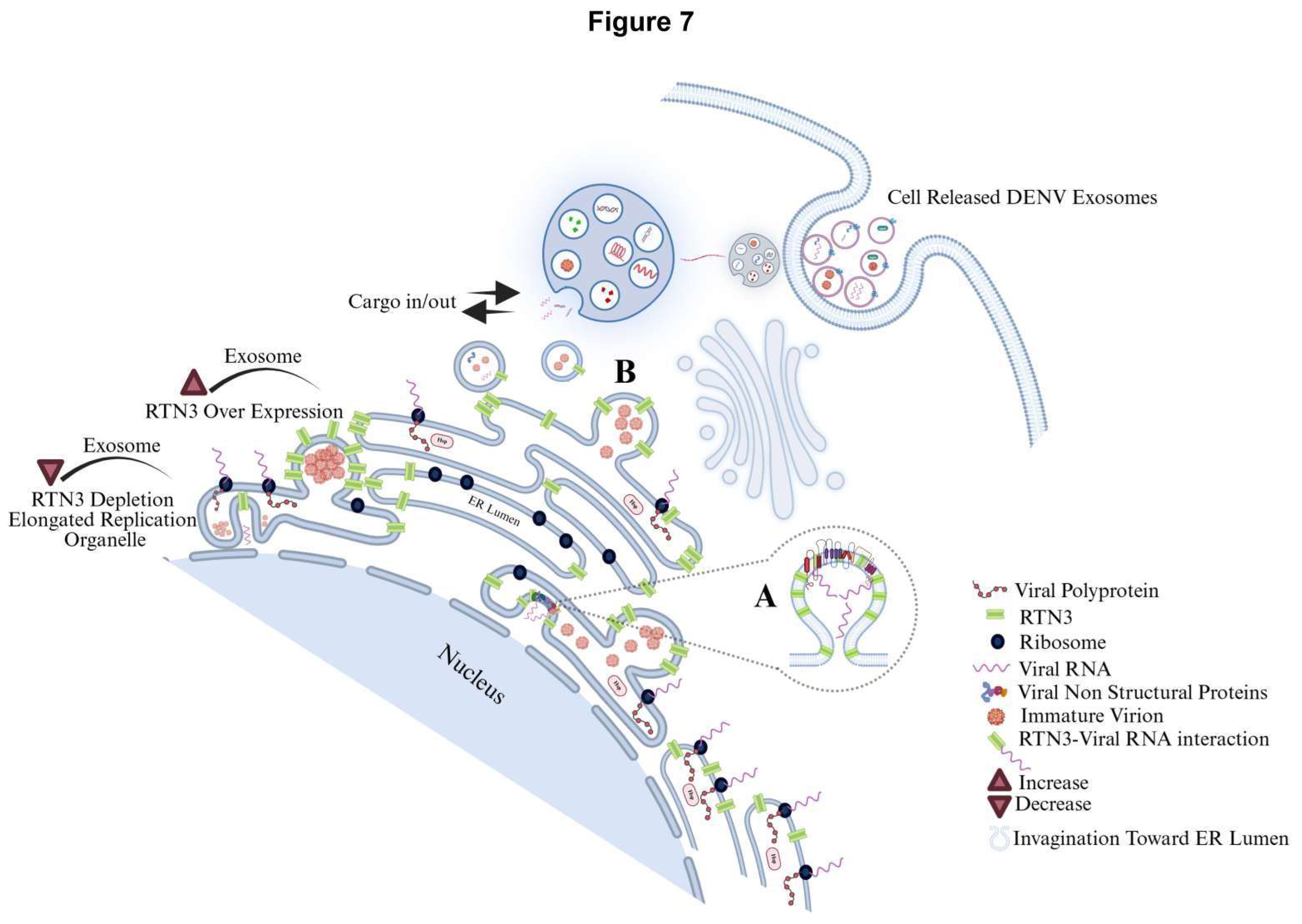
RTN3 Associates with DENV Replication Complexes and Viral RNA
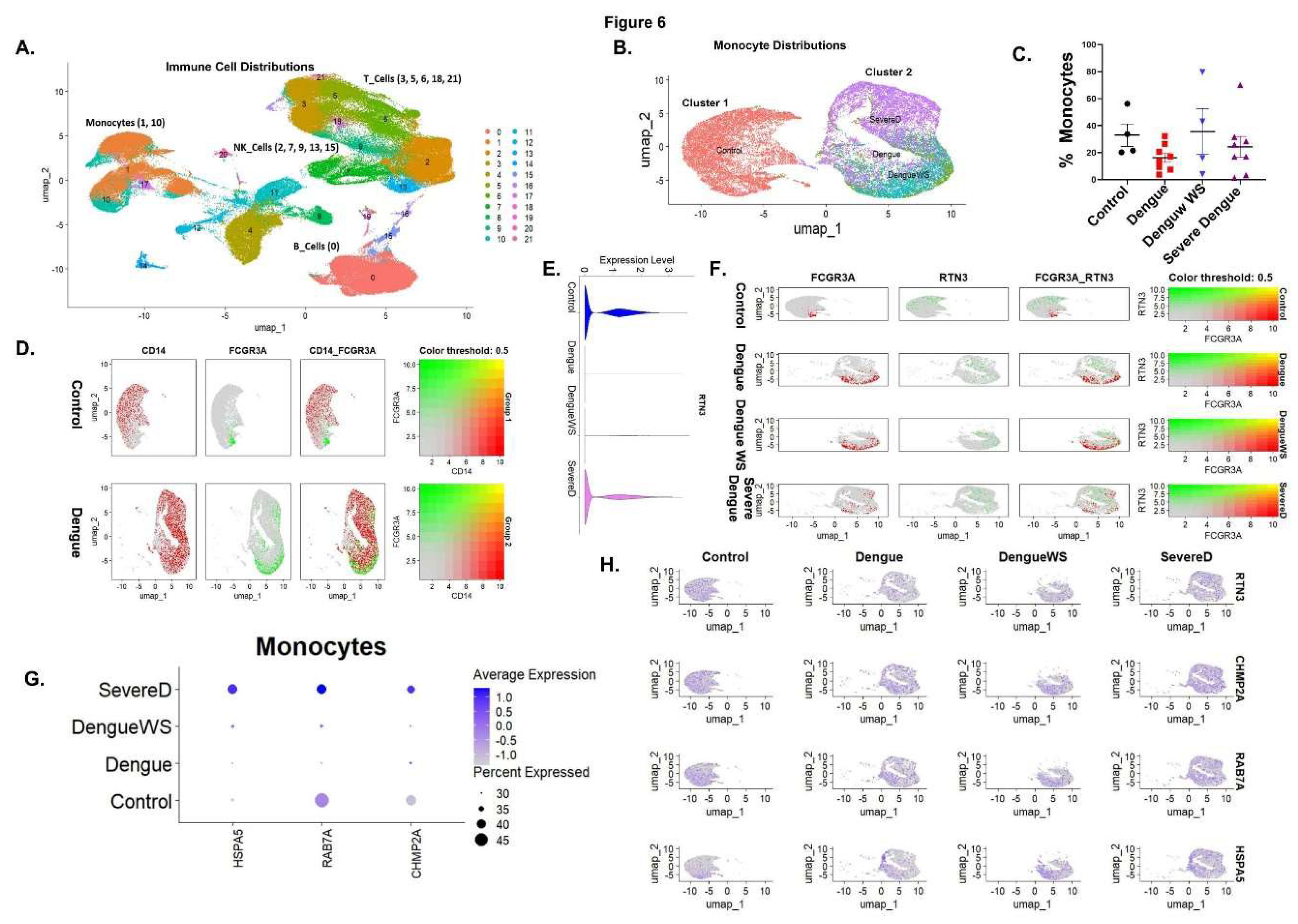
Monocyte Infection Triggers RTN3-Linked Exosome Pathways and Immune Activation
Single-Cell Transcriptomics Links RTN3-High FCGR3A+ Monocytes with Exosome-Related Pathways and Clinical Severity in Dengue Virus Infection
Discussion
Supplementary Materials
Funding
Contributions
Ethics Declaration
Acknowledgements
Competing Interests
References
- Venkatesan, P., Global upsurge in dengue in 2024. Lancet Infect Dis 2024, 24, (10), e620. [CrossRef]
- eClinicalMedicine, Dengue as a growing global health concern. EClinicalMedicine 2024, 77, 102975.
- (WHO), W. H. O., Dengue and severe dengue. In 2025.
- Bhatt, S.; Gething, P. W.; Brady, O. J.; Messina, J. P.; Farlow, A. W.; Moyes, C. L.; Drake, J. M.; Brownstein, J. S.; Hoen, A. G.; Sankoh, O.; Myers, M. F.; George, D. B.; Jaenisch, T.; Wint, G. R.; Simmons, C. P.; Scott, T. W.; Farrar, J. J.; Hay, S. I., The global distribution and burden of dengue. Nature 2013, 496, (7446), 504-7. [CrossRef]
- Reinhold, J. M.; Lazzari, C. R.; Lahondere, C., Effects of the Environmental Temperature on Aedes aegypti and Aedes albopictus Mosquitoes: A Review. Insects 2018, 9, (4). [CrossRef]
- Khanam, A.; Gutierrez-Barbosa, H.; Lyke, K. E.; Chua, J. V., Immune-Mediated Pathogenesis in Dengue Virus Infection. Viruses 2022, 14, (11). [CrossRef]
- Benda, R.; Hala, S.; Dostalova, J.; Vojtiskova-Tuckova, E.; Weidenhoffer, Z.; Rehn, F., [Preventive effect of 1-aminoadamantane of domestic origin on experimental asiatic influenza]. Cesk Epidemiol Mikrobiol Imunol 1970, 19, (2), 72-9.
- Fernandes-Santos, C.; Azeredo, E. L., Innate Immune Response to Dengue Virus: Toll-like Receptors and Antiviral Response. Viruses 2022, 14, (5).
- Lee, M. F.; Voon, G. Z.; Lim, H. X.; Chua, M. L.; Poh, C. L., Innate and adaptive immune evasion by dengue virus. Front Cell Infect Microbiol 2022, 12, 1004608. [CrossRef]
- Schmid, M. A.; Diamond, M. S.; Harris, E., Dendritic cells in dengue virus infection: targets of virus replication and mediators of immunity. Front Immunol 2014, 5, 647. [CrossRef]
- Castillo Ramirez, J. A.; Urcuqui-Inchima, S., Dengue Virus Control of Type I IFN Responses: A History of Manipulation and Control. J Interferon Cytokine Res 2015, 35, (6), 421-30. [CrossRef]
- Thurmond, S.; Wang, B.; Song, J.; Hai, R., Suppression of Type I Interferon Signaling by Flavivirus NS5. Viruses 2018, 10, (12). [CrossRef]
- Tian, J.; Xu, Z.; Smith, J. S.; Hofherr, S. E.; Barry, M. A.; Byrnes, A. P., Adenovirus activates complement by distinctly different mechanisms in vitro and in vivo: indirect complement activation by virions in vivo. J Virol 2009, 83, (11), 5648-58. [CrossRef]
- Green, A. M.; Beatty, P. R.; Hadjilaou, A.; Harris, E., Innate immunity to dengue virus infection and subversion of antiviral responses. J Mol Biol 2014, 426, (6), 1148-60. [CrossRef]
- Kao, Y. T.; Lai, M. M. C.; Yu, C. Y., How Dengue Virus Circumvents Innate Immunity. Front Immunol 2018, 9, 2860. [CrossRef]
- Teo, A.; Tan, H. D.; Loy, T.; Chia, P. Y.; Chua, C. L. L., Understanding antibody-dependent enhancement in dengue: Are afucosylated IgG1s a concern? PLoS Pathog 2023, 19, (3), e1011223. [CrossRef]
- Latanova, A.; Karpov, V.; Starodubova, E., Extracellular Vesicles in Flaviviridae Pathogenesis: Their Roles in Viral Transmission, Immune Evasion, and Inflammation. Int J Mol Sci 2024, 25, (4).
- Zhou, W.; Woodson, M.; Neupane, B.; Bai, F.; Sherman, M. B.; Choi, K. H.; Neelakanta, G.; Sultana, H., Exosomes serve as novel modes of tick-borne flavivirus transmission from arthropod to human cells and facilitates dissemination of viral RNA and proteins to the vertebrate neuronal cells. PLoS Pathog 2018, 14, (1), e1006764. [CrossRef]
- Sultana, H.; Ahmed, W.; Neelakanta, G., GW4869 inhibitor affects vector competence and tick-borne flavivirus acquisition and transmission by blocking exosome secretion. iScience 2024, 27, (8), 110391. [CrossRef]
- Vora, A.; Zhou, W.; Londono-Renteria, B.; Woodson, M.; Sherman, M. B.; Colpitts, T. M.; Neelakanta, G.; Sultana, H., Arthropod EVs mediate dengue virus transmission through interaction with a tetraspanin domain containing glycoprotein Tsp29Fb. Proc Natl Acad Sci U S A 2018, 115, (28), E6604-E6613. [CrossRef]
- Martinez-Rojas, P. P.; Monroy-Martinez, V.; Ruiz-Ordaz, B. H., Role of extracellular vesicles in the pathogenesis of mosquito-borne flaviviruses that impact public health. J Biomed Sci 2025, 32, (1), 4. [CrossRef]
- Mishra, R.; Lahon, A.; Banerjea, A. C., Dengue Virus Degrades USP33-ATF3 Axis via Extracellular Vesicles to Activate Human Microglial Cells. J Immunol 2020, 205, (7), 1787-1798. [CrossRef]
- Mishra, R.; Lata, S.; Ali, A.; Banerjea, A. C., Dengue haemorrhagic fever: a job done via exosomes? Emerg Microbes Infect 2019, 8, (1), 1626-1635.
- Aktepe, T. E.; Liebscher, S.; Prier, J. E.; Simmons, C. P.; Mackenzie, J. M., The Host Protein Reticulon 3.1A Is Utilized by Flaviviruses to Facilitate Membrane Remodelling. Cell Rep 2017, 21, (6), 1639-1654. [CrossRef]
- Ci, Y.; Shi, L., Compartmentalized replication organelle of flavivirus at the ER and the factors involved. Cell Mol Life Sci 2021, 78, (11), 4939-4954. [CrossRef]
- Li, J.; Abosmaha, E.; Coffin, C. S.; Labonte, P.; Bukong, T. N., Reticulon-3 modulates the incorporation of replication competent hepatitis C virus molecules for release inside infectious exosomes. PLoS One 2020, 15, (9), e0239153. [CrossRef]
- Bukong, T. N.; Momen-Heravi, F.; Kodys, K.; Bala, S.; Szabo, G., Exosomes from hepatitis C infected patients transmit HCV infection and contain replication competent viral RNA in complex with Ago2-miR122-HSP90. PLoS Pathog 2014, 10, (10), e1004424. [CrossRef]
- Kratzel, A.; Thiel, V., RTN3 and RTN4: Architects of SARS-CoV-2 replication organelles. J Cell Biol 2023, 222, (7). [CrossRef]
- Wilson, A.; McCormick, C., Reticulophagy and viral infection. Autophagy 2025, 21, (1), 3-20.
- Park, J.; Kim, J.; Park, H.; Kim, T.; Lee, S., ESCRT-III: a versatile membrane remodeling machinery and its implications in cellular processes and diseases. Anim Cells Syst (Seoul) 2024, 28, (1), 367-380. [CrossRef]
- Dixit, U.; Pandey, A. K.; Mishra, P.; Sengupta, A.; Pandey, V. N., Staufen1 promotes HCV replication by inhibiting protein kinase R and transporting viral RNA to the site of translation and replication in the cells. Nucleic Acids Res 2016, 44, (11), 5271-87. [CrossRef]
- Tabata, K.; Arimoto, M.; Arakawa, M.; Nara, A.; Saito, K.; Omori, H.; Arai, A.; Ishikawa, T.; Konishi, E.; Suzuki, R.; Matsuura, Y.; Morita, E., Unique Requirement for ESCRT Factors in Flavivirus Particle Formation on the Endoplasmic Reticulum. Cell Rep 2016, 16, (9), 2339-47. [CrossRef]
- McKnight, K. L.; Xie, L.; Gonzalez-Lopez, O.; Rivera-Serrano, E. E.; Chen, X.; Lemon, S. M., Protein composition of the hepatitis A virus quasi-envelope. Proc Natl Acad Sci U S A 2017, 114, (25), 6587-6592. [CrossRef]
- Effantin, G.; Dordor, A.; Sandrin, V.; Martinelli, N.; Sundquist, W. I.; Schoehn, G.; Weissenhorn, W., ESCRT-III CHMP2A and CHMP3 form variable helical polymers in vitro and act synergistically during HIV-1 budding. Cell Microbiol 2013, 15, (2), 213-26. [CrossRef]
- Duhaini, M.; Fares, P.; Hafezi, L.; El-Zein, H.; Kondapalli, K. C., Sodium proton exchanger NHE9 pHine-tunes exosome production by impairing Rab7 activity. J Biol Chem 2025, 301, (3), 108264. [CrossRef]
- Liang, W.; Sagar, S.; Ravindran, R.; Najor, R. H.; Quiles, J. M.; Chi, L.; Diao, R. Y.; Woodall, B. P.; Leon, L. J.; Zumaya, E.; Duran, J.; Cauvi, D. M.; De Maio, A.; Adler, E. D.; Gustafsson, A. B., Mitochondria are secreted in extracellular vesicles when lysosomal function is impaired. Nat Commun 2023, 14, (1), 5031. [CrossRef]
- Song, P.; Trajkovic, K.; Tsunemi, T.; Krainc, D., Parkin Modulates Endosomal Organization and Function of the Endo-Lysosomal Pathway. J Neurosci 2016, 36, (8), 2425-37. [CrossRef]
- Wati, S.; Soo, M. L.; Zilm, P.; Li, P.; Paton, A. W.; Burrell, C. J.; Beard, M.; Carr, J. M., Dengue virus infection induces upregulation of GRP78, which acts to chaperone viral antigen production. J Virol 2009, 83, (24), 12871-80. [CrossRef]
- Schneider, W. M.; Hoffmann, H. H., Flavivirus-host interactions: an expanding network of proviral and antiviral factors. Curr Opin Virol 2022, 52, 71-77. [CrossRef]
- Kohli, E.; Causse, S.; Baverel, V.; Dubrez, L.; Borges-Bonan, N.; Demidov, O.; Garrido, C., Endoplasmic Reticulum Chaperones in Viral Infection: Therapeutic Perspectives. Microbiol Mol Biol Rev 2021, 85, (4), e0003521. [CrossRef]
| Primer | Sequence 5’ to 3’ |
|---|---|
| RTN3S |
F: GGAGAGATGTGAAGAAGACTGCC R: AGATCCTGAAGCTGATGGTGA |
| RTN3L |
F: GTAGGGAGGCTAAAACTGCA R: CTCCTGAAACTTTGGATGGAGA |
| DENV |
F: TTATCAGTTCAAAATCCAATGTTGGT R: AGGAGGAAGCTGGGTTGACA |
| CHMP2A |
F: GAAGACGCCAGAGGAGCTACTGC R: GCTTGGCCATCTTCTTAATGTCTGC |
| RAB7A |
F: GTCGGGAAGACATCACTCA R: CTAGCCTGTCATCCACCAT |
| HSPA5 |
F: GGGAGGTGTCATGACCAAAC R: GCAGGAGGAATTCCAGTCAG |
| CXCL10 |
F: GTGGATGTTCTGACCCTGCT R: GGAGGATGGCAGTGGAAGTC |
| IFNB |
F: TGGGAGGCTTGAATACTGCCTCAA R: TCCTTGGCCTTCAGGTAATGCAGA |
| 18S |
F: GTAACCCGTTGAACCCCATT R: CCATCCAATCGGTAGTAGCG |
| gRNA cloning verification |
F: AGCTGCAGCTCTTCGTC R: TCGGCGGCCACTCAGTC |
| gRNA sequence targeting RTN3S | CTCGGCTCCGAAGGACGACG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




