Submitted:
30 June 2025
Posted:
02 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Recombinant Antibody Production
2.3. Flow Cytometry
2.4. ADCC
2.5. CDC
2.6. Antitumor Activity of humPcMab-60
3. Results
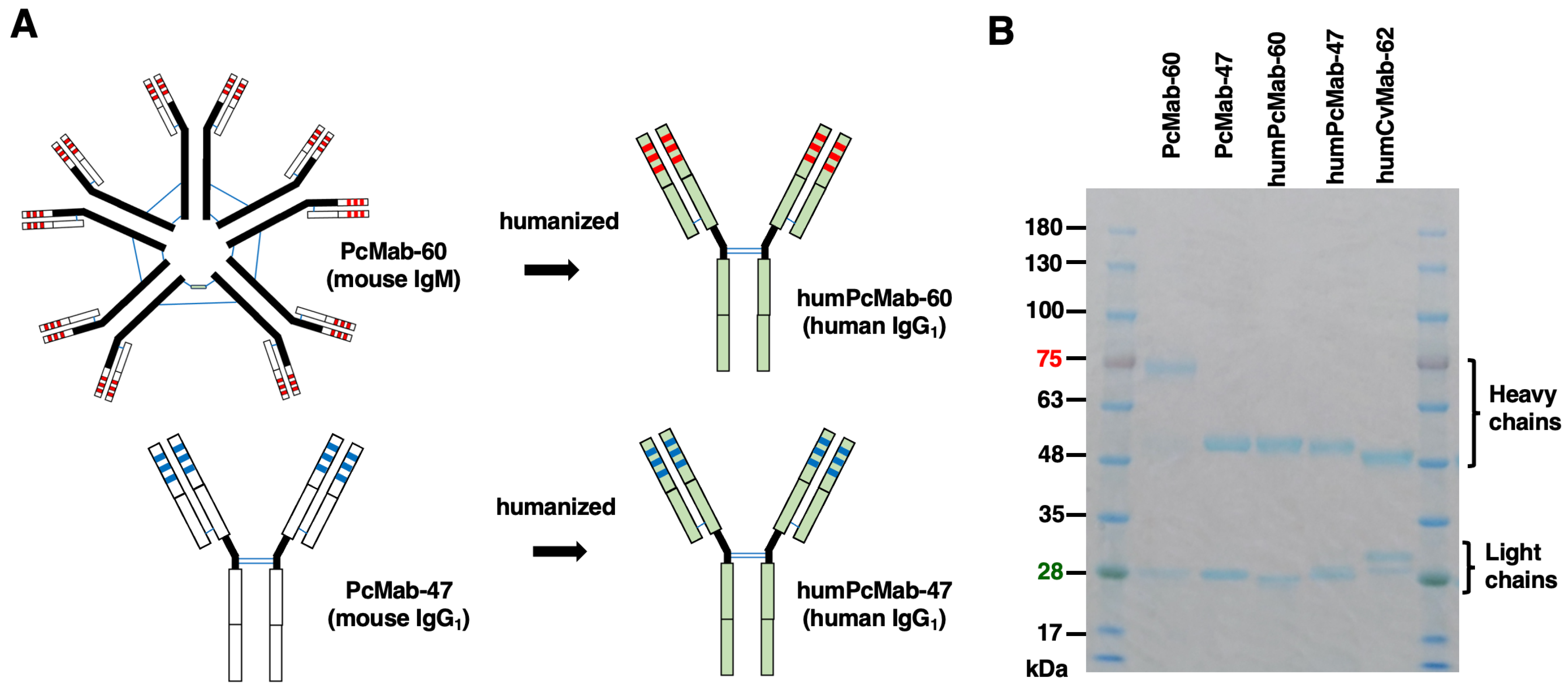
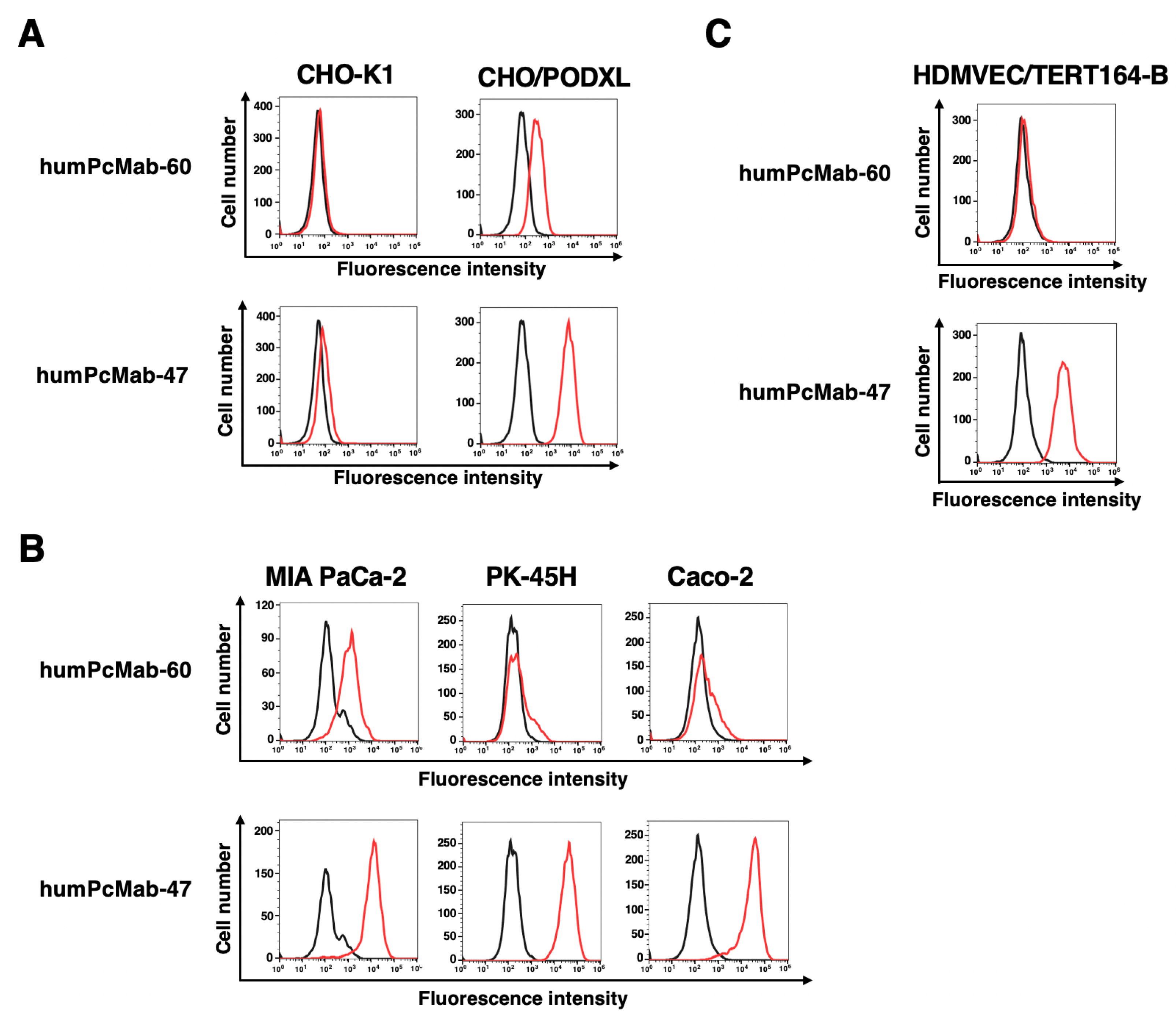
3.1. Production of a Humanized Anti-PODXL CasMab, humPcMab-60
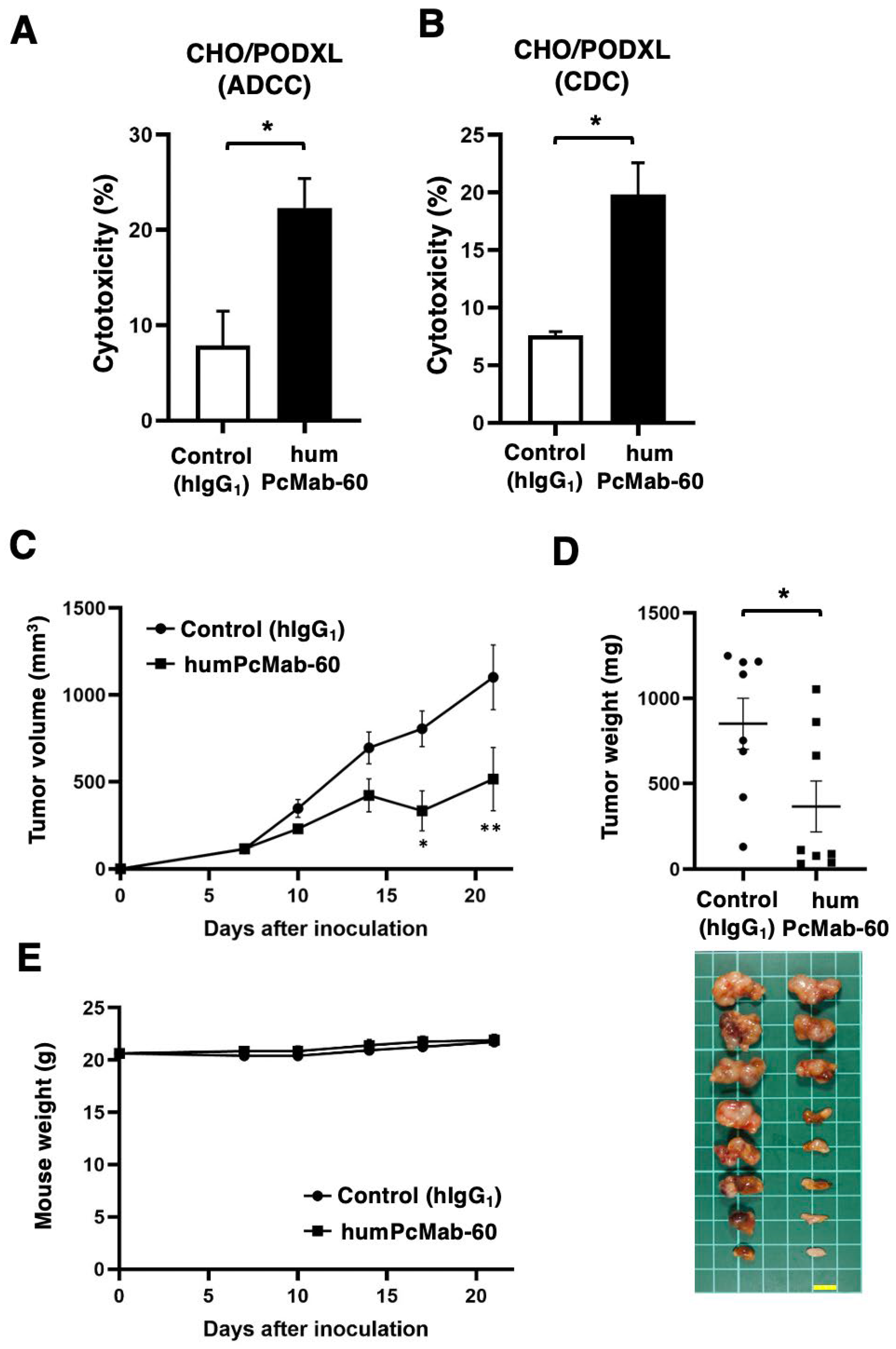
3.2. ADCC, CDC, and Antitumor Effect by humPcMab-60 Against CHO/PODXL.
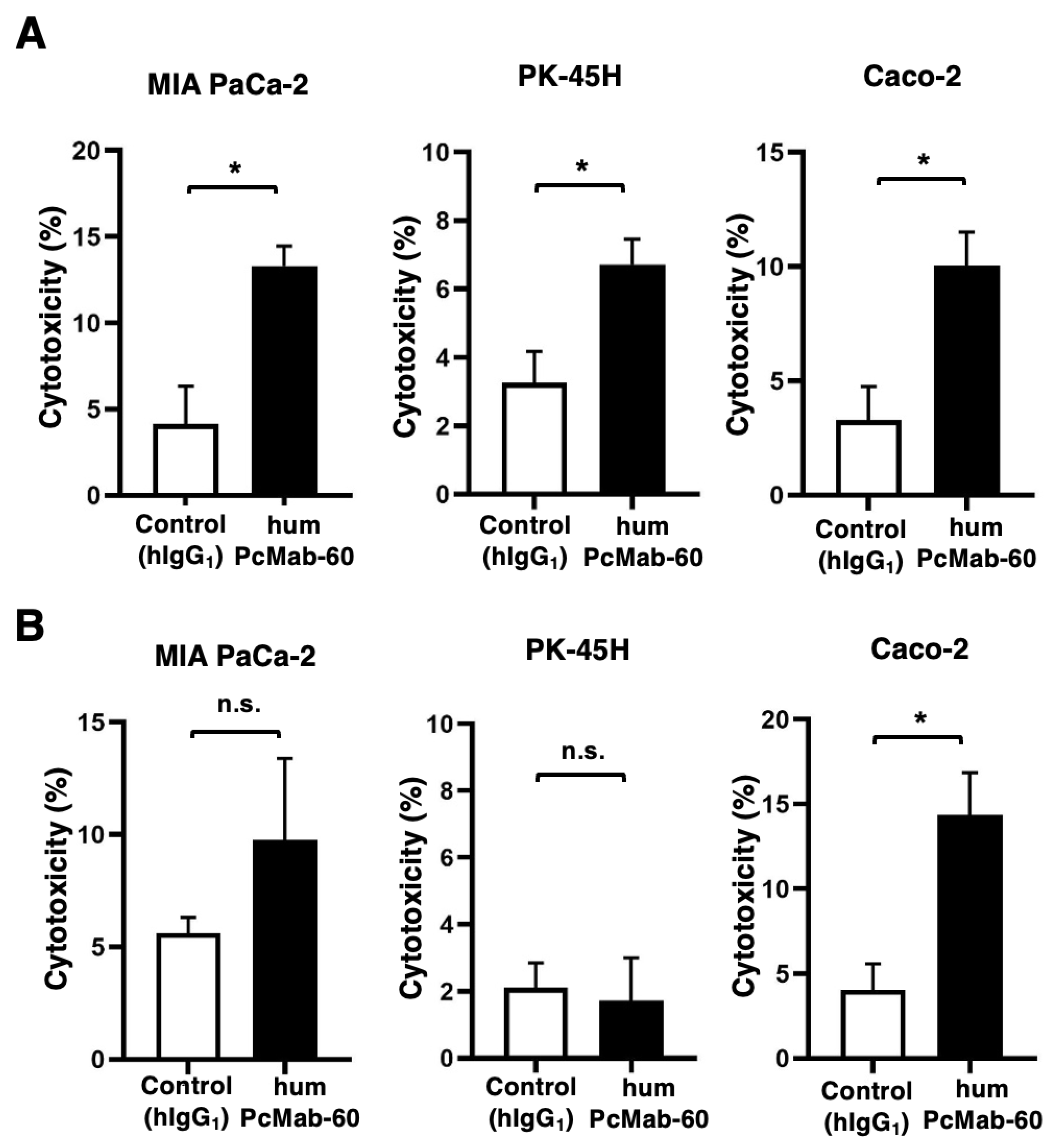
3.3. ADCC and CDC by humPcMab-60 Against Human Cancer Cells
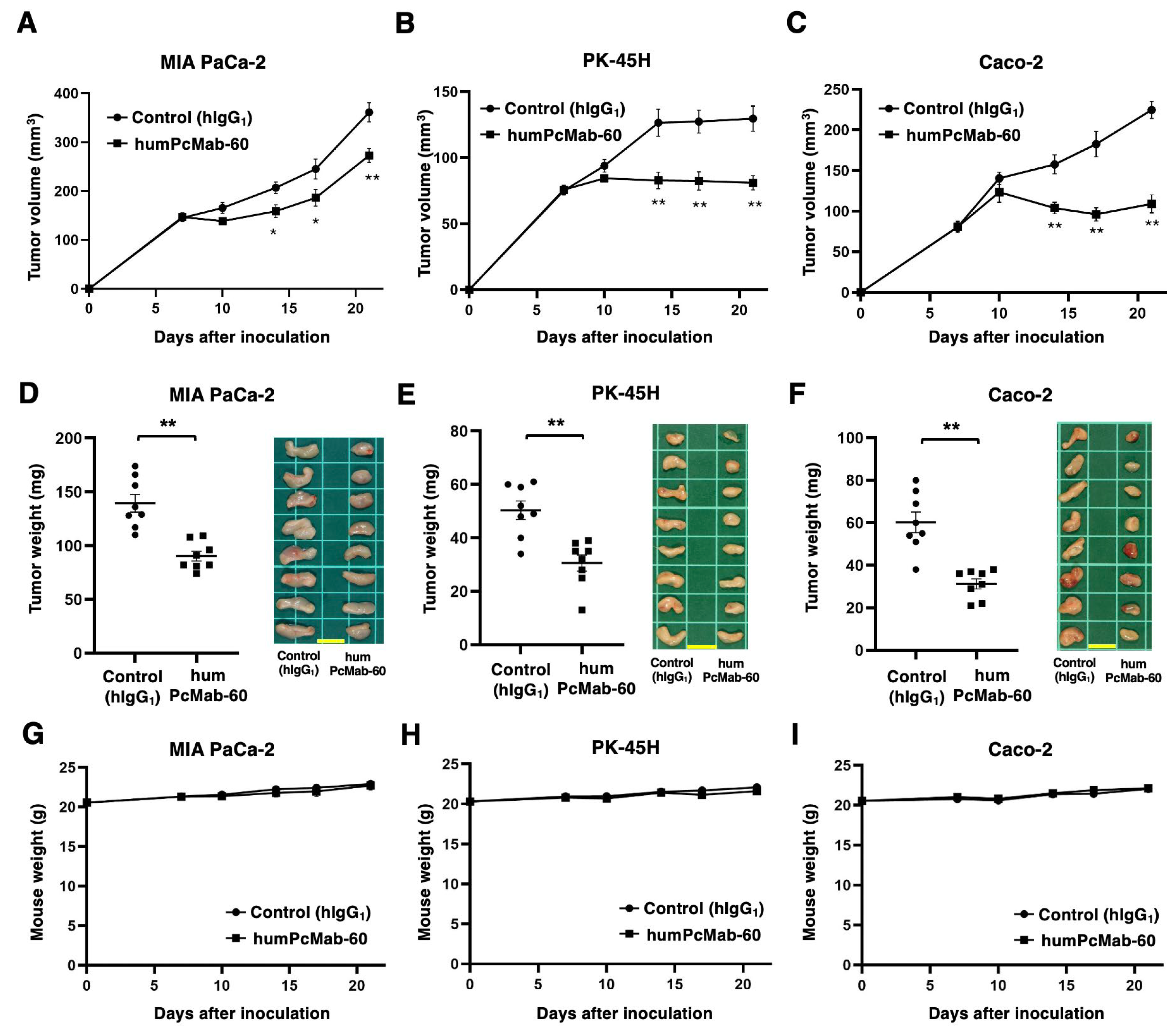
3.4. Antitumor Effect by humPcMab-60 Against Human Cancer Cells
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Doyonnas, R.; Nielsen, J.S.; Chelliah, S.; et al. Podocalyxin is a CD34-related marker of murine hematopoietic stem cells and embryonic erythroid cells. Blood 2005, 105, 4170–4178. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.S.; McNagny, K.M. Novel functions of the CD34 family. J Cell Sci 2008, 121, 3683–3692. [Google Scholar] [CrossRef] [PubMed]
- McNagny, K.M.; Pettersson, I.; Rossi, F.; et al. Thrombomucin, a novel cell surface protein that defines thrombocytes and multipotent hematopoietic progenitors. J Cell Biol 1997, 138, 1395–1407. [Google Scholar] [CrossRef] [PubMed]
- Kerjaschki, D.; Sharkey, D.J.; Farquhar, M.G. Identification and characterization of podocalyxin--the major sialoprotein of the renal glomerular epithelial cell. J Cell Biol 1984, 98, 1591–1596. [Google Scholar] [CrossRef]
- Kriehuber, E.; Breiteneder-Geleff, S.; Groeger, M.; et al. Isolation and characterization of dermal lymphatic and blood endothelial cells reveal stable and functionally specialized cell lineages. J Exp Med 2001, 194, 797–808. [Google Scholar] [CrossRef]
- Doyonnas, R.; Kershaw, D.B.; Duhme, C.; et al. Anuria, omphalocele, and perinatal lethality in mice lacking the CD34-related protein podocalyxin. J Exp Med 2001, 194, 13–27. [Google Scholar] [CrossRef]
- Ney, J.T.; Zhou, H.; Sipos, B.; et al. Podocalyxin-like protein 1 expression is useful to differentiate pancreatic ductal adenocarcinomas from adenocarcinomas of the biliary and gastrointestinal tracts. Hum Pathol 2007, 38, 359–364. [Google Scholar] [CrossRef]
- Hsu, Y.H.; Lin, W.L.; Hou, Y.T.; et al. Podocalyxin EBP50 ezrin molecular complex enhances the metastatic potential of renal cell carcinoma through recruiting Rac1 guanine nucleotide exchange factor ARHGEF7. Am J Pathol 2010, 176, 3050–3061. [Google Scholar] [CrossRef]
- Larsson, A.; Johansson, M.E.; Wangefjord, S.; et al. Overexpression of podocalyxin-like protein is an independent factor of poor prognosis in colorectal cancer. Br J Cancer 2011, 105, 666–672. [Google Scholar] [CrossRef]
- Forse, C.L.; Yilmaz, Y.E.; Pinnaduwage, D.; et al. Elevated expression of podocalyxin is associated with lymphatic invasion, basal-like phenotype, and clinical outcome in axillary lymph node-negative breast cancer. Breast Cancer Res Treat 2013, 137, 709–719. [Google Scholar] [CrossRef]
- Lin, C.W.; Sun, M.S.; Wu, H.C. Podocalyxin-like 1 is associated with tumor aggressiveness and metastatic gene expression in human oral squamous cell carcinoma. Int J Oncol 2014, 45, 710–718. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Du, W.; Li, M.; Yan, M.; Zheng, F. PODXL might be a new prognostic biomarker in various cancers: a meta-analysis and sequential verification with TCGA datasets. BMC Cancer 2020, 20, 620. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.J.; Gao, X.J.; Xu, L.N.; et al. Ezrin is required for epithelial-mesenchymal transition induced by TGF-β1 in A549 cells. Int J Oncol 2014, 45, 1515–1522. [Google Scholar] [CrossRef]
- Fröse, J.; Chen, M.B.; Hebron, K.E.; et al. Epithelial-Mesenchymal Transition Induces Podocalyxin to Promote Extravasation via Ezrin Signaling. Cell Rep 2018, 24, 962–972. [Google Scholar] [CrossRef]
- Le Tran, N.; Wang, Y.; Nie, G. Podocalyxin in Normal Tissue and Epithelial Cancer. Cancers (Basel).
- Dallas, M.R.; Chen, S.H.; Streppel, M.M.; et al. Sialofucosylated podocalyxin is a functional E- and L-selectin ligand expressed by metastatic pancreatic cancer cells. Am J Physiol Cell Physiol 2012, 303, C616–624. [Google Scholar] [CrossRef]
- Tamayo-Orbegozo, E.; Amo, L.; Díez-García, J.; et al. Emerging Role of Podocalyxin in the Progression of Mature B-Cell Non-Hodgkin Lymphoma. Cancers (Basel).
- Ogasawara, S.; Kaneko, M.K.; Yamada, S.; et al. PcMab-47: Novel Antihuman Podocalyxin Monoclonal Antibody for Immunohistochemistry. Monoclon Antib Immunodiagn Immunother 2017, 36, 50–56. [Google Scholar] [CrossRef]
- Itai, S.; Ohishi, T.; Kaneko, M.K.; et al. Anti-podocalyxin antibody exerts antitumor effects via antibody-dependent cellular cytotoxicity in mouse xenograft models of oral squamous cell carcinoma. Oncotarget 2018, 9, 22480–22497. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Kunita, A.; Yamada, S.; et al. Antipodocalyxin Antibody chPcMab-47 Exerts Antitumor Activity in Mouse Xenograft Models of Colorectal Adenocarcinomas. Monoclon Antib Immunodiagn Immunother 2017, 36, 157–162. [Google Scholar] [CrossRef]
- Snyder, K.A.; Hughes, M.R.; Hedberg, B.; et al. Podocalyxin enhances breast tumor growth and metastasis and is a target for monoclonal antibody therapy. Breast Cancer Res 2015, 17, 46. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Suzuki, H.; Kato, Y. Establishment of a Novel Cancer-Specific Anti-HER2 Monoclonal Antibody H(2)Mab-250/H(2)CasMab-2 for Breast Cancers. Monoclon Antib Immunodiagn Immunother 2024, 43, 35–43. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. Antitumor Activities of a Humanized Cancer-Specific Anti-HER2 Monoclonal Antibody, humH(2)Mab-250 in Human Breast Cancer Xenografts. Int J Mol Sci.
- Suzuki, H.; Ohishi, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Anti-HER2 Cancer-Specific mAb, H(2)Mab-250-hG(1), Possesses Higher Complement-Dependent Cytotoxicity than Trastuzumab. Int J Mol Sci.
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. A Cancer-Specific Monoclonal Antibody against HER2 Exerts Antitumor Activities in Human Breast Cancer Xenograft Models. Int J Mol Sci.
- Hosking, M.P.; Shirinbak, S.; Omilusik, K.; et al. Preferential tumor targeting of HER2 by iPSC-derived CAR T cells engineered to overcome multiple barriers to solid tumor efficacy. Cell Stem Cell 2025. [CrossRef] [PubMed]
- Kaneko, M.K.; Ohishi, T.; Kawada, M.; Kato, Y. A cancer-specific anti-podocalyxin monoclonal antibody (60-mG(2a)-f) exerts antitumor effects in mouse xenograft models of pancreatic carcinoma. Biochem Biophys Rep 2020, 24, 100826. [Google Scholar]
- Asano, T.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Epitope Mapping of a Cancer-Specific Anti-Podocalyxin Monoclonal Antibody (PcMab-60) Using Enzyme-Linked Immunosorbent Assay and Surface Plasmon Resonance. Monoclon Antib Immunodiagn Immunother 2021, 40, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Taniuchi, K.; Tsuboi, M.; Sakaguchi, M.; Saibara, T. Measurement of serum PODXL concentration for detection of pancreatic cancer. Onco Targets Ther 2018, 11, 1433–1445. [Google Scholar] [CrossRef]
- Siegel, R.L.; Kratzer, T.B.; Giaquinto, A.N.; Sung, H.; Jemal, A. Cancer statistics, 2025. CA Cancer J Clin 2025, 75, 10–45. [Google Scholar] [CrossRef]
- Kamisawa, T.; Wood, L.D.; Itoi, T.; Takaori, K. Pancreatic cancer. Lancet 2016, 388, 73–85. [Google Scholar] [CrossRef]
- Waddell, N.; Pajic, M.; Patch, A.M.; et al. Whole genomes redefine the mutational landscape of pancreatic cancer. Nature 2015, 518, 495–501. [Google Scholar] [CrossRef]
- Jones, S.; Zhang, X.; Parsons, D.W.; et al. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 2008, 321, 1801–1806. [Google Scholar] [CrossRef]
- Taherian, M.; Wang, H.; Wang, H. Pancreatic Ductal Adenocarcinoma: Molecular Pathology and Predictive Biomarkers. Cells.
- Bailey, P.; Chang, D.K.; Nones, K.; et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 2016, 531, 47–52. [Google Scholar] [CrossRef]
- Naishiro, Y.; Yamada, T.; Idogawa, M.; et al. Morphological and transcriptional responses of untransformed intestinal epithelial cells to an oncogenic beta-catenin protein. Oncogene 2005, 24, 3141–3153. [Google Scholar] [CrossRef]
- Lee, H.; Kong, J.S.; Lee, S.S.; Kim, A. Radiation-Induced Overexpression of TGFβ and PODXL Contributes to Colorectal Cancer Cell Radioresistance through Enhanced Motility. Cells.
- Eigenbrot, C.; Ultsch, M.; Dubnovitsky, A.; Abrahmsén, L.; Härd, T. Structural basis for high-affinity HER2 receptor binding by an engineered protein. Proc Natl Acad Sci U S A 2010, 107, 15039–15044. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.S.; Mason, K.; Ramyar, K.X.; et al. Structure of the extracellular region of HER2 alone and in complex with the Herceptin Fab. Nature 2003, 421, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Reis, C.A. Glycosylation in cancer: mechanisms and clinical implications. Nat Rev Cancer 2015, 15, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Canals Hernaez, D.; Hughes, M.R.; Dean, P.; et al. PODO447: a novel antibody to a tumor-restricted epitope on the cancer antigen podocalyxin. J Immunother Cancer.
- Itai, S.; Yamada, S.; Kaneko, M.K.; Kato, Y. Determination of critical epitope of PcMab-47 against human podocalyxin. Biochem Biophys Rep 2018, 14, 78–82. [Google Scholar] [CrossRef]
- Ring, A.; Nguyen-Sträuli, B.D.; Wicki, A.; Aceto, N. Biology, vulnerabilities and clinical applications of circulating tumour cells. Nat Rev Cancer 2023, 23, 95–111. [Google Scholar] [CrossRef]
- Dashzeveg, N.K.; Jia, Y.; Zhang, Y.; et al. Dynamic Glycoprotein Hyposialylation Promotes Chemotherapy Evasion and Metastatic Seeding of Quiescent Circulating Tumor Cell Clusters in Breast Cancer. Cancer Discov 2023, 13, 2050–2071. [Google Scholar] [CrossRef]
- Singh, A.; Liu, H.; El-Shennawy, L. Multi-omic features and clustering phenotypes of circulating tumor cells associated with metastasis and clinical outcomes. Int Rev Cell Mol Biol 2025, 392, 67–100. [Google Scholar]
- Li, F.; Ding, J. Sialylation is involved in cell fate decision during development, reprogramming and cancer progression. Protein Cell 2019, 10, 550–565. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



