Submitted:
30 June 2025
Posted:
01 July 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Search Strategy
3. Current and Emerging Therapeutics
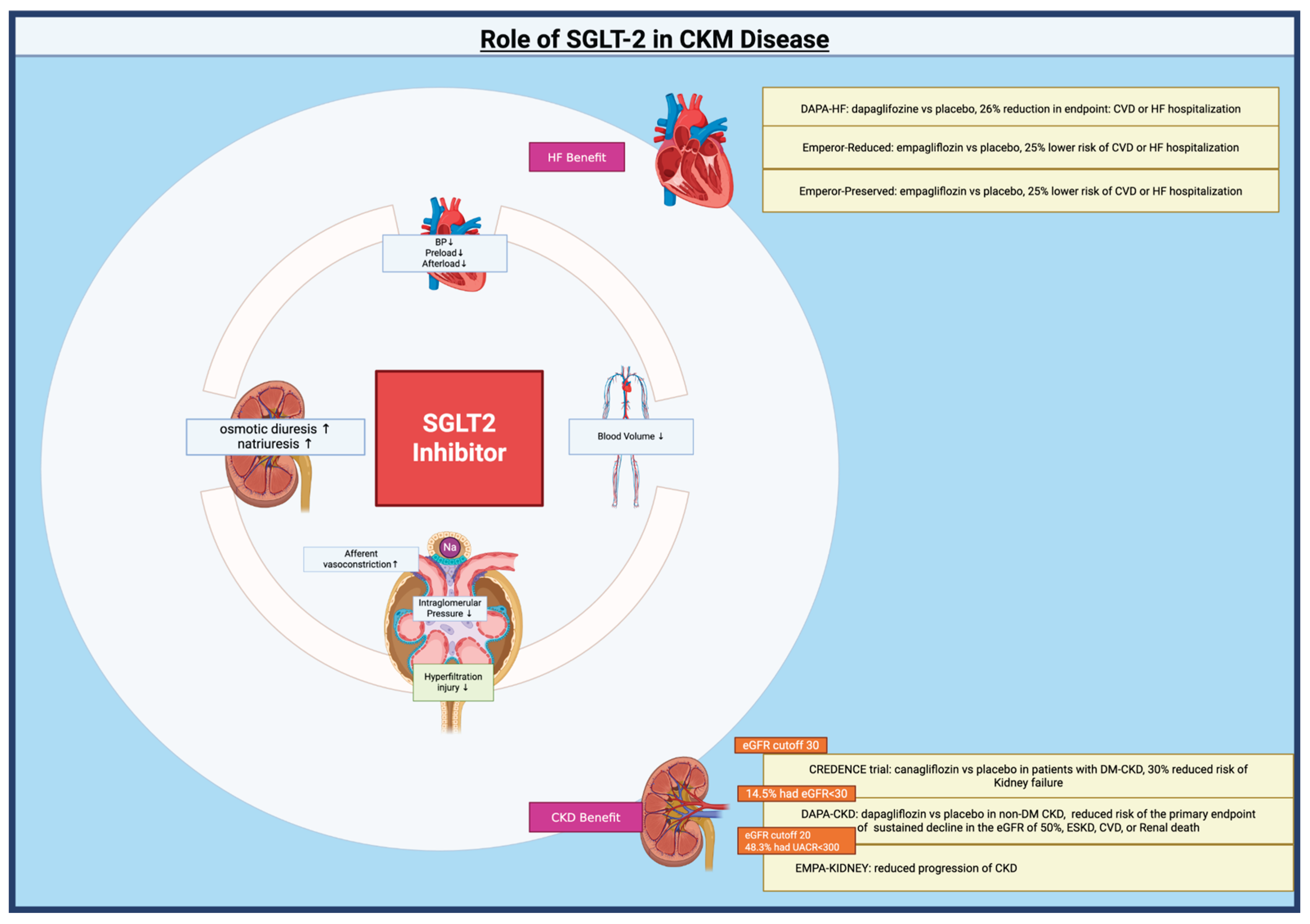
3.1. Sodium Glucose Co-Transport 2 Inhibitors:
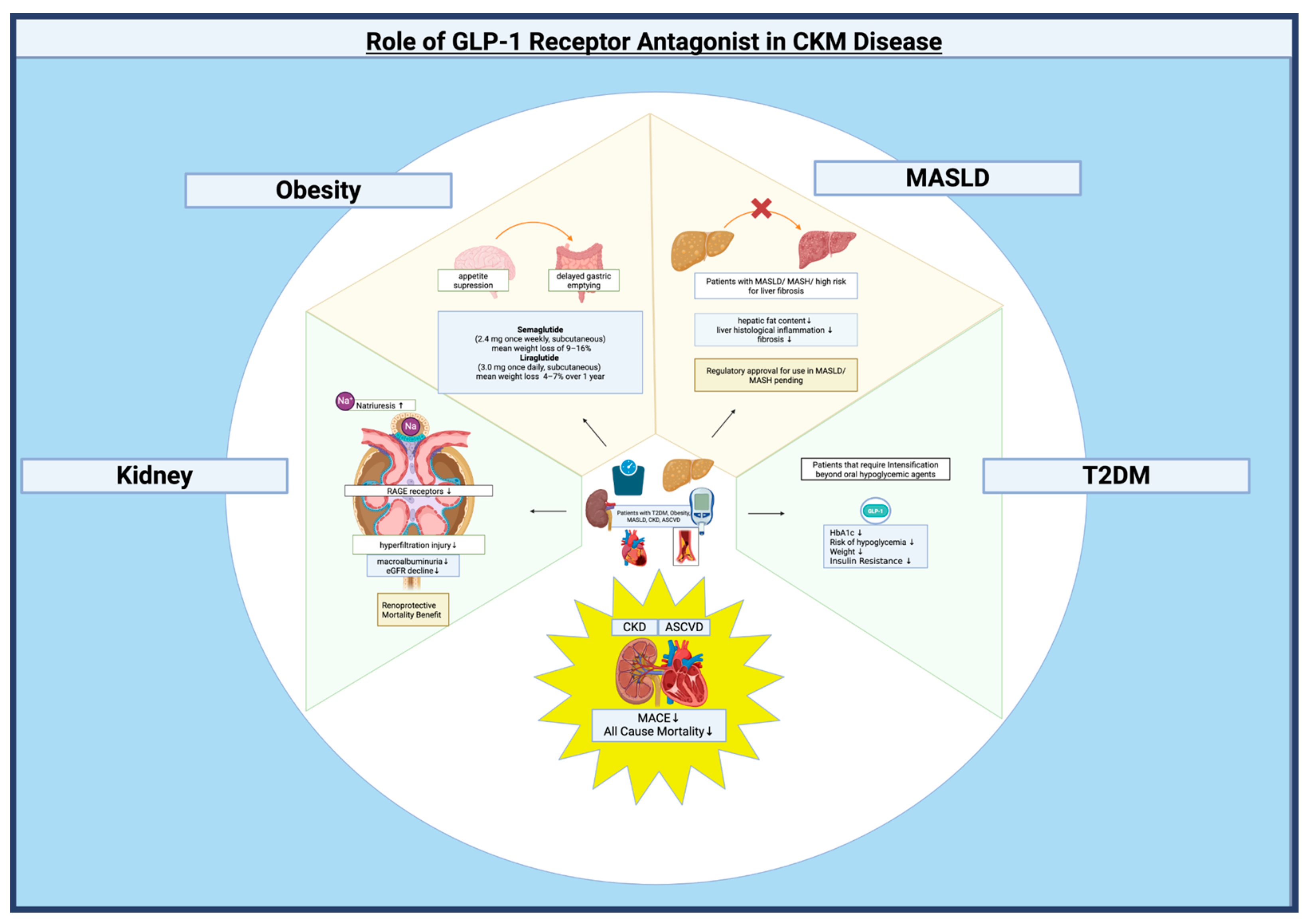
3.2. Glucagon-like-Peptide-1 Receptor Agonists:
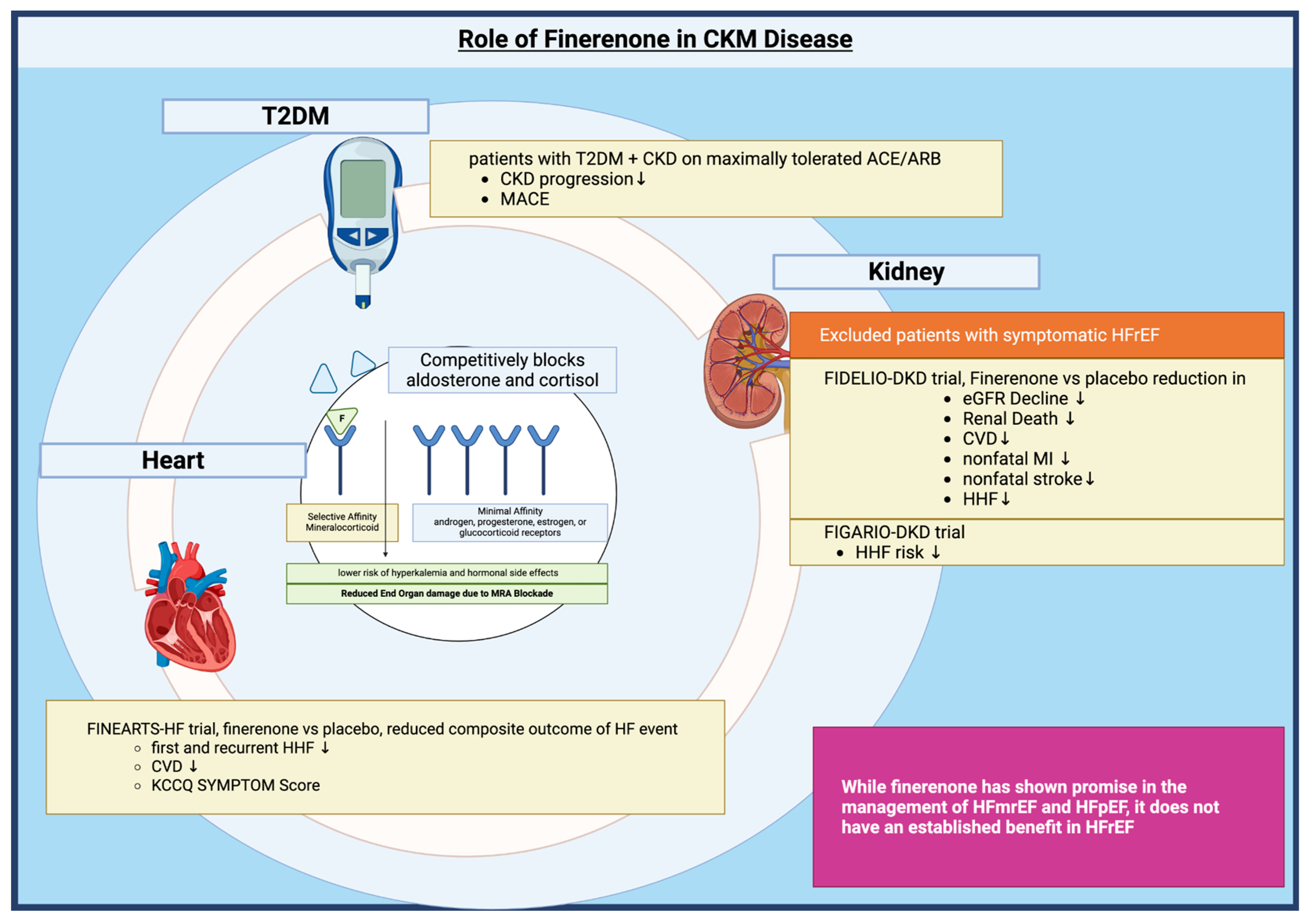
3.3. Mineralocorticoid Receptor Antagonist:
3.4. Thyroid Receptor Beta Agonists:
3.5. Lipoprotein-a (Lp(a))
3.6. Phase 3 Trial Drugs for CKM Syndrome
3.7. Role of Artificial Intelligence
| Paper Name | Authors* | Year | Study Type | Population Studied | Key Findings | Inclusion Criteria / Indication | Outcome Trial Results (with p-value if available) | Side Effect Profile* |
|---|---|---|---|---|---|---|---|---|
| Defining CKM Syndrome | ||||||||
| Cardiovascular-Kidney-Metabolic Health: A Presidential Advisory From the American Heart Association | AHA (Ndumele CE, et al.)[77] | 2023 | Presidential Advisory / Scientific Statement | General US population; focus on individuals with/at risk for CVD, CKD, T2D, Obesity. | Defines CKM syndrome as a health disorder linking obesity, diabetes, CKD, and CVD. Proposes staging (0-4) based on risk factors and disease presence. Emphasizes prevention, integrated care, and addressing social determinants of health (SDOH). | N/A (Definitional document) | N/A (Definitional document) | N/A (Recommends therapies like SGLT2i/GLP-1 RA for appropriate stages) |
| An Overview of Cardiovascular-Kidney-Metabolic Syndrome | Ferdinand KC et al.[84] | 2024 | Review | General overview of CKM syndrome patients. | Reinforces CKM definition, staging. Highlights role of excess/dysfunctional adipose tissue, inflammation, oxidative stress. Notes impact of SDOH and additional risk factors (chronic inflammation, family history, sleep/mental health). | N/A (Review) | N/A (Review) | N/A (Review) |
| Cardiovascular-Kidney-Metabolic (CKM) syndrome: A state-of-the-art review | Sebastian SA et al.[3] | 2024 | Review | Epidemiological data from NHANES and AHA reports, highlighting prevalence across different demographics | CKM syndrome involves interconnected metabolic, cardiovascular, and renal diseases. Key mechanisms include insulin resistance, RAAS activation, oxidative stress, chronic inflammation, and lipotoxicity. The syndrome progresses through five stages, from no risk factors to symptomatic cardiovascular disease with kidney failure. Management focuses on screening, early intervention, and multidisciplinary care to reduce adverse outcomes. | N/A (Review) | N/A (Review) | 1. GLP-1 RA: Primarily causes gastrointestinal issues like nausea, vomiting, and diarrhea. 2. SGLT2 inhibitors: Increase the risk of genital and urinary tract infections 3. Finerenone: May lead to hyperkalemia |
| SGLT2 Inhibitor Trials (Meta-Analyses) | ||||||||
| Effects of SGLT2 inhibitors on cardiovascular outcomes in patients with stage 3/4 CKD: A meta-analysis | Li N, et al.[85] | 2022 | Meta-analysis | 11 RCTs; 27,823 patients with stage 3/4 CKD. | SGLT2i significantly reduced primary CV outcomes (CV death/HHF) across stage 3a, 3b, and 4 CKD, irrespective of T2D or HF status. | Patients with stage 3/4 CKD included in RCTs comparing SGLT2i vs placebo. | Reduced primary CV outcome risk by 26% (HR 0.74, 95% CI 0.69–0.80, p<0.001 inferred). Consistent benefit across CKD stages (p interaction = 0.71). | General Class Effects: Genitourinary infections, potential for volume depletion/hypotension, rare risk of DKA. |
| Effect of SGLT2 Inhibitors on Cardiovascular Outcomes Across Various Patient Populations | Usman, et al.[86] | 2023 | Meta-analysis | 13 RCTs; >90,000 patients with HF, T2D, CKD or combinations. | SGLT2i consistently reduced the composite of first HHF or CV death (~23-24%) across HF, T2D, and CKD populations and combinations. Also reduced CV death (~12-16%) and HHF (~29-32%) separately. | Patients with HF, T2D, or CKD in large RCTs comparing SGLT2i vs placebo. | Reduced HHF/CV Death by ~24% (HR ~0.76-0.77, p<0.001 inferred). Reduced CV Death by ~12-16% (p<0.001 inferred). Reduced HHF by ~29-32% (p<0.001 inferred). |
General Class Effects: Genitourinary infections, potential for volume depletion/hypotension, rare risk of DKA. |
| GLP-1 Receptor Agonist Trials (Meta-Analyses) | ||||||||
| Kidney and Cardiovascular Outcomes Among Patients With CKD Receiving GLP-1 Receptor Agonists: A Systematic Review and Meta-Analysis of Randomized Trials | Chen et al.[32] | 2024 | Meta-analysis | 12 RCTs; 17,996 participants with baseline eGFR < 60 mL/min/1.73m2. | GLP-1 RAs significantly reduced composite kidney outcome, risk of >30/40/50% eGFR decline, all-cause mortality, and composite CV outcomes in patients with CKD. | Adults with varying kidney function (incl. CKD eGFR<60) in RCTs comparing GLP-1 RA vs control. | Reduced composite kidney outcome (OR 0.85, 95% CI 0.77-0.94, P=0.001). Reduced all-cause mortality (OR 0.77, 95% CI 0.60-0.98, P=0.03). Reduced composite CV outcomes (OR 0.86, 95% CI 0.74-0.99, P=0.03). | General Class Effects: Gastrointestinal side effects (nausea, vomiting, diarrhea), injection site reactions, rare risk of pancreatitis/thyroid tumors. |
| Effects of GLP-1 receptor agonists on kidney and cardiovascular disease outcomes: a meta-analysis of randomized controlled trials | Badve et al.[87] | 2024 | Meta-analysis (incl. SELECT trial) | 11 RCTs; 85,373 participants (mostly T2D, one trial non-diabetic obesity/CVD). | GLP-1 RAs reduced composite kidney outcome, kidney failure, MACE, and all-cause death in T2D patients. Similar effects when non-diabetic SELECT trial included. | Participants (mostly T2D, one non-diabetic obesity/CVD trial) in large RCTs comparing GLP-1 RA vs placebo. | Reduced composite kidney outcome by 18% (HR 0.82, 95% CI 0.73-0.93). Reduced kidney failure by 16% (HR 0.84, 95% CI 0.72-0.99). Reduced MACE by 13% (HR 0.87, 95% CI 0.81-0.93). Reduced all-cause death by 12% (HR 0.88, 95% CI 0.83-0.93). | Higher treatment discontinuation due to AEs (RR 1.51). No difference in serious AEs vs placebo. |
| Drugs | Phase 3 Trials | Principal investigator | Indication | MOA | Outcome | Adverse effects |
|---|---|---|---|---|---|---|
| Volanesorsen | NCT02211209 | Gaudet et al.[58] | Hypertriglyceridemia, Type 2 diabetes mellitus, Familial Chylomicronemia Syndrome (FCS) | ASO targeting ApoC-III | 77% decrease in mean triglyceride levels(TG). | Thrombocytopenia and injection site reaction. |
| Olezarsen | NCT04568434 | Stroes et al.[88] | Hypertriglyceridemia, Acute coronary syndrome (ACS), FCS | Gal- NAc3 conjugated ASO targeting ApoC-III | Reduction in the fasting triglyceride level of at least 70% at 6 months. | Abdominal pain, and diarrhea. |
| Mipomersen | NCT00794664 | McGowan et al.[89] | Hypercholesterolemia, Dyslipidemias | Induces ApoB100 degradation | Reduced LDL-C by 36% | Injection site reactions, flu-like symptoms. |
| Pelacarsen | NCT05305664 | Novartis Pharmaceuticals[90] | ACS, Hyperlipoproteinemia | ASO targeting Lp(a) | Pending results | Mild injection site reactions. |
| Plozasiran | NCT06347016 | Arrowhead Pharmaceuticals[91] | Mixed dyslipidemia, Hypertriglyceridemia, FCS | siRNA targeting apoC-III mRNA | Currently recruiting. | Worsening glycemic control, diarrhea, urinary tract infection. |
| Inclisiran | NCT03399370 | Ray KK et al.[92] | Coronary artery disease (CAD), Familial hypercholesterolemia (FHS), ACS | siRNA targeting PCSK9 | 50% reduction in low density lipoprotein (LDL) | Injection site reactions. |
| Lepodisiran | NCT06292013 | Ferdinand et al.[93] | Cardiovascular disorders (CVD), Metabolic disorders | siRNA targeting ApoA | Currently recruiting. | Injection site reactions, hypersensitivity reactions, hepatobiliary adverse events. |
| Olpasiran | NCT05581303 | UCSD Health[94] | CAD, elevated Lp(a) | siRNA targeting Lp(a) | Pre-recruitment stage | Injection-site reactions |
| Ziltivekimab | NCT05021835 | Ridker et al.[95] | CVD, Chronic kidney disease (CKD) | IL-6 Blocker | Currently recruiting | Injection-site reactions |
| Obicetrapib | NCT05142722 | Ditmarsch et al.[96] | Heterozygous FHS, CAD | CETP Inhibitors | Completed, pending publication of results. | Nausea, urinary tract infection, and headache. |
| Evinacumab | NCT05611528 | Gaudet et al.[97] | Homozygous Familial Hypercholesterolemia | ANGPTL3 Inhibition | 47.1% reduction in LDL | Nasopharyngitis, influenza-like illness, headache. |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADA | American Diabetes Association |
| AI | Artificial intelligence |
| ACEi | Angiotensin converting enzyme inhibitor |
| ARB | Angiotensin receptor blocker |
| ASO | Antisense Oligonucleotides |
| ANGPTL3 | angiopoietin-like 3 |
| ASCVD | Atherosclerotic cardiovascular disease |
| CETP | Cholesteryl ester transfer protein |
| CKM | Cardiovascular-kidney-metabolic syndrome |
| CVD | Cardiovascular disease |
| CKD | Chronic kidney disease |
| EASD | European Association for the Study of Diabetes |
| GLP1-RA | Glucagon-like peptide-1 receptor agonists |
| HFPEF | Heart failure with preserved ejection fraction |
| HFREF | Heart failure with reduced ejection fraction |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| KDOQI | National Kidney Foundation Kidney Disease Outcomes Quality Initiative |
| LDL | Low density lipoprotein |
| MACE | Major adverse cardiovascular events |
| MASLD | Metabolic dysfunction-associated steatotic liver disease |
| PCSK9 | Proprotein convertase subtilisin–kexin type 9 |
| RAS | Renin angiotensin system |
| SGLT2i | Sodium-glucose cotransporter 2 inhibitors |
| TRβ | Thyroid receptor beta |
References
- Marassi M, Fadini GP. The cardio-renal-metabolic connection: a review of the evidence. Cardiovasc Diabetol 2023; 22: 195. [CrossRef]
- Schnell O, Almandoz J, Anderson L, Barnard-Kelly K, Battelino T, Blüher M, Busetto L, Catrinou D, Ceriello A, Cos X, Danne T, Dayan CM, Del Prato S, Fernández-Fernández B, Fioretto P, Forst T, Gavin JR, Giorgino F, Groop P-H, Harsch IA, Heerspink HJL, Heinemann L, Ibrahim M, Jadoul M, Jarvis S, Ji L, Kanumilli N, Kosiborod M, Landmesser U, Macieira S, Mankovsky B, Marx N, Mathieu C, McGowan B, Milenkovic T, Moser O, Müller-Wieland D, Papanas N, Patel DC, Pfeiffer AFH, Rahelić D, Rodbard HW, Rydén L, Schaeffner E, Spearman CW, Stirban A, Tacke F, Topsever P, Van Gaal L, Standl E. CVOT summit report 2024: new cardiovascular, kidney, and metabolic outcomes. Cardiovasc Diabetol 2025; 24: 187. [CrossRef]
- Sebastian SA, Padda I, Johal G. Cardiovascular-Kidney-Metabolic (CKM) syndrome: A state-of-the-art review. Curr Probl Cardiol 2024; 49: 102344. [CrossRef]
- Ndumele CE, Neeland IJ, Tuttle KR, Chow SL, Mathew RO, Khan SS, Coresh J, Baker-Smith CM, Carnethon MR, Després J-P, Ho JE, Joseph JJ, Kernan WN, Khera A, Kosiborod MN, Lekavich CL, Lewis EF, Lo KB, Ozkan B, Palaniappan LP, Patel SS, Pencina MJ, Powell-Wiley TM, Sperling LS, Virani SS, Wright JT, Rajgopal Singh R, Elkind MSV, Rangaswami J, on behalf of the American Heart Association. A Synopsis of the Evidence for the Science and Clinical Management of Cardiovascular-Kidney-Metabolic (CKM) Syndrome: A Scientific Statement From the American Heart Association. Circulation 2023; 148: 1636–1664. [CrossRef]
- Januzzi JL, Van Kimmenade RRJ, Liu Y, Hu X, Browne A, Plutzky J, Tsimikas S, Blankstein R, Natarajan P. Lipoprotein(a), Oxidized Phospholipids, and Progression to Symptomatic Heart Failure: The CASABLANCA Study. J Am Heart Assoc 2024; 13: e034774. [CrossRef]
- Petta S, Targher G, Romeo S, Pajvani UB, Zheng M, Aghemo A, Valenti LVC. The first MASH drug therapy on the horizon: Current perspectives of resmetirom. Liver Int 2024; 44: 1526–1536. [CrossRef]
- Heerspink HJL, Perkins BA, Fitchett DH, Husain M, Cherney DZI. Sodium Glucose Cotransporter 2 Inhibitors in the Treatment of Diabetes Mellitus: Cardiovascular and Kidney Effects, Potential Mechanisms, and Clinical Applications. Circulation 2016; 134: 752–772. [CrossRef]
- Di Costanzo A, Esposito G, Indolfi C, Spaccarotella CAM. SGLT2 Inhibitors: A New Therapeutical Strategy to Improve Clinical Outcomes in Patients with Chronic Kidney Diseases. Int J Mol Sci 2023; 24: 8732. [CrossRef]
- Fonseca-Correa JI, Correa-Rotter R. Sodium-Glucose Cotransporter 2 Inhibitors Mechanisms of Action: A Review. Front Med 2021; 8: 777861. [CrossRef]
- Vallon V, Verma S. Effects of SGLT2 Inhibitors on Kidney and Cardiovascular Function. Annu Rev Physiol 2021; 83: 503–528. [CrossRef]
- Chen X, Hocher C-F, Shen L, Krämer BK, Hocher B. Reno- and cardioprotective molecular mechanisms of SGLT2 inhibitors beyond glycemic control: from bedside to bench. Am J Physiol-Cell Physiol 2023; 325: C661–C681. [CrossRef]
- Salvatore T, Galiero R, Caturano A, Rinaldi L, Di Martino A, Albanese G, Di Salvo J, Epifani R, Marfella R, Docimo G, Lettieri M, Sardu C, Sasso FC. An Overview of the Cardiorenal Protective Mechanisms of SGLT2 Inhibitors. Int J Mol Sci 2022; 23: 3651. [CrossRef]
- McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, Ponikowski P, Sabatine MS, Anand IS, Bělohlávek J, Böhm M, Chiang C-E, Chopra VK, De Boer RA, Desai AS, Diez M, Drozdz J, Dukát A, Ge J, Howlett JG, Katova T, Kitakaze M, Ljungman CEA, Merkely B, Nicolau JC, O’Meara E, Petrie MC, Vinh PN, Schou M, Tereshchenko S, Verma S, Held C, DeMets DL, Docherty KF, Jhund PS, Bengtsson O, Sjöstrand M, Langkilde A-M. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N Engl J Med 2019; 381: 1995–2008. [CrossRef]
- Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, Januzzi J, Verma S, Tsutsui H, Brueckmann M, Jamal W, Kimura K, Schnee J, Zeller C, Cotton D, Bocchi E, Böhm M, Choi D-J, Chopra V, Chuquiure E, Giannetti N, Janssens S, Zhang J, Gonzalez Juanatey JR, Kaul S, Brunner-La Rocca H-P, Merkely B, Nicholls SJ, Perrone S, Pina I, Ponikowski P, Sattar N, Senni M, Seronde M-F, Spinar J, Squire I, Taddei S, Wanner C, Zannad F. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med 2020; 383: 1413–1424. [CrossRef]
- Rangaswami J, Bhalla V, De Boer IH, Staruschenko A, Sharp JA, Singh RR, Lo KB, Tuttle K, Vaduganathan M, Ventura H, McCullough PA, On behalf of the American Heart Association Council on the Kidney in Cardiovascular Disease; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology; and Council on Lifestyle and Cardiometabolic Health. Cardiorenal Protection With the Newer Antidiabetic Agents in Patients With Diabetes and Chronic Kidney Disease: A Scientific Statement From the American Heart Association. Circulation 2020; 142. [CrossRef]
- Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers LR, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernandez AF, Khazanie P, Kittleson MM, Lee CS, Link MS, Milano CA, Nnacheta LC, Sandhu AT, Stevenson LW, Vardeny O, Vest AR, Yancy CW. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2022; 145. [CrossRef]
- Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, Brunner–La Rocca H-P, Choi D-J, Chopra V, Chuquiure-Valenzuela E, Giannetti N, Gomez-Mesa JE, Janssens S, Januzzi JL, Gonzalez-Juanatey JR, Merkely B, Nicholls SJ, Perrone SV, Piña IL, Ponikowski P, Senni M, Sim D, Spinar J, Squire I, Taddei S, Tsutsui H, Verma S, Vinereanu D, Zhang J, Carson P, Lam CSP, Marx N, Zeller C, Sattar N, Jamal W, Schnaidt S, Schnee JM, Brueckmann M, Pocock SJ, Zannad F, Packer M. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med 2021; 385: 1451–1461. [CrossRef]
- Patel SM, Kang YM, Im K, Neuen BL, Anker SD, Bhatt DL, Butler J, Cherney DZI, Claggett BL, Fletcher RA, Herrington WG, Inzucchi SE, Jardine MJ, Mahaffey KW, McGuire DK, McMurray JJV, Neal B, Packer M, Perkovic V, Solomon SD, Staplin N, Vaduganathan M, Wanner C, Wheeler DC, Zannad F, Zhao Y, Heerspink HJL, Sabatine MS, Wiviott SD. Sodium-Glucose Cotransporter-2 Inhibitors and Major Adverse Cardiovascular Outcomes: A SMART-C Collaborative Meta-Analysis. Circulation 2024; 149: 1789–1801. [CrossRef]
- Neuen BL, Ohkuma T, Neal B, Matthews DR, De Zeeuw D, Mahaffey KW, Fulcher G, Li Q, Jardine M, Oh R, Heerspink HL, Perkovic V. Effect of Canagliflozin on Renal and Cardiovascular Outcomes across Different Levels of Albuminuria: Data from the CANVAS Program. J Am Soc Nephrol 2019; 30: 2229–2242. [CrossRef]
- Heerspink HJL, Stefánsson BV, Correa-Rotter R, Chertow GM, Greene T, Hou F-F, Mann JFE, McMurray JJV, Lindberg M, Rossing P, Sjöström CD, Toto RD, Langkilde A-M, Wheeler DC. Dapagliflozin in Patients with Chronic Kidney Disease. N Engl J Med 2020; 383: 1436–1446. [CrossRef]
- The EMPA-KIDNEY Collaborative Group. Empagliflozin in Patients with Chronic Kidney Disease. N Engl J Med 2023; 388: 117–127. [CrossRef]
- Davies MJ, Aroda VR, Collins BS, Gabbay RA, Green J, Maruthur NM, Rosas SE, Del Prato S, Mathieu C, Mingrone G, Rossing P, Tankova T, Tsapas A, Buse JB. Management of Hyperglycemia in Type 2 Diabetes, 2022. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2022; 45: 2753–2786. [CrossRef]
- Blonde L, Umpierrez GE, Reddy SS, McGill JB, Berga SL, Bush M, Chandrasekaran S, DeFronzo RA, Einhorn D, Galindo RJ, Gardner TW, Garg R, Garvey WT, Hirsch IB, Hurley DL, Izuora K, Kosiborod M, Olson D, Patel SB, Pop-Busui R, Sadhu AR, Samson SL, Stec C, Tamborlane WV, Tuttle KR, Twining C, Vella A, Vellanki P, Weber SL. American Association of Clinical Endocrinology Clinical Practice Guideline: Developing a Diabetes Mellitus Comprehensive Care Plan—2022 Update. Endocr Pract 2022; 28: 923–1049. [CrossRef]
- Abdelmalek MF, Harrison SA, Sanyal AJ. The role of glucagon-like peptide-1 receptor agonists in metabolic dysfunction-associated steatohepatitis. Diabetes Obes Metab 2024; 26: 2001–2016. [CrossRef]
- De Boer IH, Khunti K, Sadusky T, Tuttle KR, Neumiller JJ, Rhee CM, Rosas SE, Rossing P, Bakris G. Diabetes management in chronic kidney disease: a consensus report by the American Diabetes Association (ADA) and Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 2022; 102: 974–989. [CrossRef]
- American Diabetes Association Professional Practice Committee, ElSayed NA, McCoy RG, Aleppo G, Bajaj M, Balapattabi K, Beverly EA, Briggs Early K, Bruemmer D, Cusi K, Echouffo-Tcheugui JB, Ekhlaspour L, Fleming TK, Garg R, Khunti K, Lal R, Levin SR, Lingvay I, Matfin G, Napoli N, Pandya N, Parish SJ, Pekas EJ, Pilla SJ, Pirih FQ, Polsky S, Segal AR, Jeffrie Seley J, Stanton RC, Verduzco-Gutierrez M, Younossi ZM, Bannuru RR. 4. Comprehensive Medical Evaluation and Assessment of Comorbidities: Standards of Care in Diabetes—2025. Diabetes Care 2025; 48: S59–S85. [CrossRef]
- Loomba R, Hartman ML, Lawitz EJ, Vuppalanchi R, Boursier J, Bugianesi E, Yoneda M, Behling C, Cummings OW, Tang Y, Brouwers B, Robins DA, Nikooie A, Bunck MC, Haupt A, Sanyal AJ. Tirzepatide for Metabolic Dysfunction–Associated Steatohepatitis with Liver Fibrosis. N Engl J Med 2024; 391: 299–310. [CrossRef]
- Sanyal AJ, Bedossa P, Fraessdorf M, Neff GW, Lawitz E, Bugianesi E, Anstee QM, Hussain SA, Newsome PN, Ratziu V, Hosseini-Tabatabaei A, Schattenberg JM, Noureddin M, Alkhouri N, Younes R. A Phase 2 Randomized Trial of Survodutide in MASH and Fibrosis. N Engl J Med 2024; 391: 311–319. [CrossRef]
- Newsome PN, Buchholtz K, Cusi K, Linder M, Okanoue T, Ratziu V, Sanyal AJ, Sejling A-S, Harrison SA. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N Engl J Med 2021; 384: 1113–1124. [CrossRef]
- Alkhouri N, Charlton M, Gray M, Noureddin M. Alkhouri, Naim, Michael Charlton, Meagan Gray, and Mazen Noureddin. “The Pleiotropic Effects of Glucagon-like Peptide-1 Receptor Agonists in Patients with Metabolic Dysfunction-Associated Steatohepatitis: A Review for Gastroenterologists.” Expert Opinion on Investigational Drugs 34, no. 3 (March 4, 2025): 169–95. Expert Opin Investig Drugs 2025; 34: 169–195. [CrossRef]
- Yabut JM, Drucker DJ. Glucagon-like Peptide-1 Receptor-based Therapeutics for Metabolic Liver Disease. Endocr Rev 2023; 44: 14–32. [CrossRef]
- Chen J-Y, Hsu T-W, Liu J-H, Pan H-C, Lai C-F, Yang S-Y, Wu V-C. Kidney and Cardiovascular Outcomes Among Patients With CKD Receiving GLP-1 Receptor Agonists: A Systematic Review and Meta-Analysis of Randomized Trials. Am J Kidney Dis 2025; 85: 555-569.e1. [CrossRef]
- Trevella P, Ekinci EI, MacIsaac RJ. Potential kidney protective effects of glucagon-like peptide-1 receptor agonists. Nephrol Carlton Vic 2024; 29: 457–469. PMID: 39030739. [CrossRef]
- Sourris KC, Ding Y, Maxwell SS, Al-Sharea A, Kantharidis P, Mohan M, Rosado CJ, Penfold SA, Haase C, Xu Y, Forbes JM, Crawford S, Ramm G, Harcourt BE, Jandeleit-Dahm K, Advani A, Murphy AJ, Timmermann DB, Karihaloo A, Knudsen LB, El-Osta A, Drucker DJ, Cooper ME, Coughlan MT. Glucagon-like peptide-1 receptor signaling modifies the extent of diabetic kidney disease through dampening the receptor for advanced glycation end products-induced inflammation. Kidney Int 2024; 105: 132–149. PMID: 38069998. [CrossRef]
- Grunvald E, Shah R, Hernaez R, Chandar AK, Pickett-Blakely O, Teigen LM, Harindhanavudhi T, Sultan S, Singh S, Davitkov P. AGA Clinical Practice Guideline on Pharmacological Interventions for Adults With Obesity. Gastroenterology 2022; 163: 1198–1225. [CrossRef]
- Moiz A, Filion KB, Tsoukas MA, Yu OHy, Peters TM, Eisenberg MJ. Mechanisms of GLP-1 Receptor Agonist-Induced Weight Loss: A Review of Central and Peripheral Pathways in Appetite and Energy Regulation. Am J Med 2025; 138: 934–940. [CrossRef]
- Taha MB, Yahya T, Satish P, Laird R, Agatston AS, Cainzos-Achirica M, Patel KV, Nasir K. Glucagon-Like Peptide 1 Receptor Agonists: A Medication for Obesity Management. Curr Atheroscler Rep 2022; 24: 643–654. [CrossRef]
- Ansari HUH, Qazi SU, Sajid F, Altaf Z, Ghazanfar S, Naveed N, Ashfaq AS, Siddiqui AH, Iqbal H, Qazi S. Efficacy and Safety of Glucagon-Like Peptide-1 Receptor Agonists on Body Weight and Cardiometabolic Parameters in Individuals With Obesity and Without Diabetes: A Systematic Review and Meta-Analysis. Endocr Pract 2024; 30: 160–171. [CrossRef]
- Xie W, Hong Z, Li B, Huang B, Dong S, Cai Y, Ruan L, Xu Q, Mou L, Zhang Y. Influence of glucagon-like peptide-1 receptor agonists on fat accumulation in patients with diabetes mellitus and non-alcoholic fatty liver disease or obesity: A systematic review and meta-analysis of randomized control trials. J Diabetes Complications 2024; 38: 108743. [CrossRef]
- Pandey AK, Bhatt DL, Cosentino F, Marx N, Rotstein O, Pitt B, Pandey A, Butler J, Verma S. Non-steroidal mineralocorticoid receptor antagonists in cardiorenal disease. Eur Heart J 2022; 43: 2931–2945. [CrossRef]
- Agarwal R, Kolkhof P, Bakris G, Bauersachs J, Haller H, Wada T, Zannad F. Agarwal, Rajiv, Peter Kolkhof, George Bakris, Johann Bauersachs, Hermann Haller, Takashi Wada, and Faiez Zannad. “Steroidal and Non-Steroidal Mineralocorticoid Receptor Antagonists in Cardiorenal Medicine.” European Heart Journal 42, no. 2 (January 7, 2021): 152–61. Eur Heart J 2021; 42: 152–161. [CrossRef]
- González-Juanatey JR, Górriz JL, Ortiz A, Valle A, Soler MJ, Facila L. Cardiorenal benefits of finerenone: protecting kidney and heart. Ann Med 2023; 55: 502–513. [CrossRef]
- Georgianos PI, Agarwal R. The Nonsteroidal Mineralocorticoid-Receptor-Antagonist Finerenone in Cardiorenal Medicine: A State-of-the-Art Review of the Literature. Am J Hypertens 2023; 36: 135–143. [CrossRef]
- Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, Kolkhof P, Nowack C, Schloemer P, Joseph A, Filippatos G. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med 2020; 383: 2219–2229. [CrossRef]
- Pitt B, Filippatos G, Agarwal R, Anker SD, Bakris GL, Rossing P, Joseph A, Kolkhof P, Nowack C, Schloemer P, Ruilope LM. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med 2021; 385: 2252–2263. [CrossRef]
- Solomon SD, McMurray JJV, Vaduganathan M, Claggett B, Jhund PS, Desai AS, Henderson AD, Lam CSP, Pitt B, Senni M, Shah SJ, Voors AA, Zannad F, Abidin IZ, Alcocer-Gamba MA, Atherton JJ, Bauersachs J, Chang-Sheng M, Chiang C-E, Chioncel O, Chopra V, Comin-Colet J, Filippatos G, Fonseca C, Gajos G, Goland S, Goncalvesova E, Kang S, Katova T, Kosiborod MN, Latkovskis G, Lee AP-W, Linssen GCM, Llamas-Esperón G, Mareev V, Martinez FA, Melenovský V, Merkely B, Nodari S, Petrie MC, Saldarriaga CI, Saraiva JFK, Sato N, Schou M, Sharma K, Troughton R, Udell JA, Ukkonen H, Vardeny O, Verma S, Von Lewinski D, Voronkov L, Yilmaz MB, Zieroth S, Lay-Flurrie J, Van Gameren I, Amarante F, Kolkhof P, Viswanathan P. Finerenone in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med 2024; 391: 1475–1485. [CrossRef]
- Ismahel H, Docherty KF. The role of finerenone in heart failure. Trends Cardiovasc Med 2025; : S1050173825000659. [CrossRef]
- Yang M, Henderson AD, Talebi A, Atherton JJ, Chiang C-E, Chopra V, Comin-Colet J, Kosiborod MN, Kerr Saraiva JF, Claggett BL, Desai AS, Kolkhof P, Viswanathan P, Lage A, Lam CSP, Senni M, Shah SJ, Rohwedder K, Voors AA, Zannad F, Pitt B, Vaduganathan M, Jhund PS, Solomon SD, McMurray JJV. Effect of Finerenone on the KCCQ in Patients With HFmrEF/HFpEF. J Am Coll Cardiol 2025; 85: 120–136. [CrossRef]
- Filippatos G, Anker SD, Agarwal R, Ruilope LM, Rossing P, Bakris GL, Tasto C, Joseph A, Kolkhof P, Lage A, Pitt B, on behalf of the FIGARO-DKD Investigators. Finerenone Reduces Risk of Incident Heart Failure in Patients With Chronic Kidney Disease and Type 2 Diabetes: Analyses From the FIGARO-DKD Trial. Circulation 2022; 145: 437–447. [CrossRef]
- Navaneethan SD, Bansal N, Cavanaugh KL, Chang A, Crowley S, Delgado C, Estrella MM, Ghossein C, Ikizler TA, Koncicki H, St. Peter W, Tuttle KR, William J. KDOQI US Commentary on the KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of CKD. Am J Kidney Dis 2025; 85: 135–176. [CrossRef]
- Li C, Wang T, Song J. A review regarding the article ‘Electrocardiographic abnormalities in patients with metabolic dysfunction-associated Steatotic liver disease: A systematic review and meta-analysis.’ Curr Probl Cardiol 2024; 49: 102626. [CrossRef]
- Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Castro Narro GE, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW-S, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. J Hepatol 2023; 79: 1542–1556. [CrossRef]
- Cho SW. Selective Agonists of Thyroid Hormone Receptor Beta: Promising Tools for the Treatment of Nonalcoholic Fatty Liver Disease. Endocrinol Metab 2024; 39: 285–287. [CrossRef]
- Harrison SA, Bedossa P, Guy CD, Schattenberg JM, Loomba R, Taub R, Labriola D, Moussa SE, Neff GW, Rinella ME, Anstee QM, Abdelmalek MF, Younossi Z, Baum SJ, Francque S, Charlton MR, Newsome PN, Lanthier N, Schiefke I, Mangia A, Pericàs JM, Patil R, Sanyal AJ, Noureddin M, Bansal MB, Alkhouri N, Castera L, Rudraraju M, Ratziu V. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N Engl J Med 2024; 390: 497–509. [CrossRef]
- Harrison SA, Bashir MR, Guy CD, Zhou R, Moylan CA, Frias JP, Alkhouri N, Bansal MB, Baum S, Neuschwander-Tetri BA, Taub R, Moussa SE. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. The Lancet 2019; 394: 2012–2024. [CrossRef]
- Younossi ZM, Stepanova M, Taub RA, Barbone JM, Harrison SA. Hepatic Fat Reduction Due to Resmetirom in Patients With Nonalcoholic Steatohepatitis Is Associated With Improvement of Quality of Life. Clin Gastroenterol Hepatol 2022; 20: 1354-1361.e7. [CrossRef]
- Karim G, Department of Medicine, Mount Sinai Israel, New York, NY, USA, Bansal MB, Division of Liver Diseases, Department of Medicine, Icahn School of Medicine at Mount Sinai, New York, NY, USA. Resmetirom: An Orally Administered, Small-molecule, Liver-directed, β-selective THR Agonist for the Treatment of Non-alcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis. Eur Endocrinol 2023; 19: 60. [CrossRef]
- A Randomized, Double-Blind, Placebo-Controlled, Phase 3 Study of ISIS 304801 Administered Subcutaneously to Patients with Familial Chylomicronemia Syndrome (FCS). 2022. Available from: https://clinicaltrials.gov/ct2/show/NCT02211209.
- Marino L, Kim A, Ni B, Celi FS. Thyroid hormone action and liver disease, a complex interplay. Hepatology 2025; 81: 651–669. [CrossRef]
- Reyes-Soffer G, Ginsberg HN, Berglund L, Duell PB, Heffron SP, Kamstrup PR, Lloyd-Jones DM, Marcovina SM, Yeang C, Koschinsky ML, on behalf of the American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Radiology and Intervention; and Council on Peripheral Vascular Disease. Lipoprotein(a): A Genetically Determined, Causal, and Prevalent Risk Factor for Atherosclerotic Cardiovascular Disease: A Scientific Statement From the American Heart Association. Arterioscler Thromb Vasc Biol 2022; 42. [CrossRef]
- Kamstrup PR, Nordestgaard BG. Elevated Lipoprotein(a) Levels, LPA Risk Genotypes, and Increased Risk of Heart Failure in the General Population. JACC Heart Fail 2016; 4: 78–87. [CrossRef]
- Li Z, Liu J, Shen J, Chen Y, He L, Li M, Xie X. Association of lipoprotein (a) and 1 year prognosis in patients with heart failure with reduced ejection fraction. ESC Heart Fail 2022; 9: 2399–2406. [CrossRef]
- Singh S, Baars DP, Aggarwal K, Desai R, Singh D, Pinto-Sietsma S-J. Association between lipoprotein (a) and risk of heart failure: A systematic review and meta-analysis of Mendelian randomization studies. Curr Probl Cardiol 2024; 49: 102439. [CrossRef]
- Steffen BT, Duprez D, Bertoni AG, Guan W, Tsai MY. Lp(a) [Lipoprotein(a)]-Related Risk of Heart Failure Is Evident in Whites but Not in Other Racial/Ethnic Groups: The Multi-Ethnic Study of Atherosclerosis. Arterioscler Thromb Vasc Biol 2018; 38: 2498–2504. [CrossRef]
- Wu B, Zhang Z, Long J, Zhao H, Zeng F. Association between lipoprotein (a) and heart failure with reduced ejection fraction development. J Clin Lab Anal 2022; 36: e24083. [CrossRef]
- Khera AV, Everett BM, Caulfield MP, Hantash FM, Wohlgemuth J, Ridker PM, Mora S. Lipoprotein(a) Concentrations, Rosuvastatin Therapy, and Residual Vascular Risk: An Analysis From the JUPITER Trial (Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin). Circulation 2014; 129: 635–642. [CrossRef]
- Tsimikas S, Viney NJ, Hughes SG, Singleton W, Graham MJ, Baker BF, Burkey JL, Yang Q, Marcovina SM, Geary RS, Crooke RM, Witztum JL. Antisense therapy targeting apolipoprotein(a): a randomised, double-blind, placebo-controlled phase 1 study. The Lancet 2015; 386: 1472–1483. [CrossRef]
- Mohammadi A, Karimian A, Shokri K, Mohammadi A, Hazhir-Karzar N, Bahar R, Radfar A, Pakyari M, Tehrani B. RNA Therapies in Cardio-Kidney-Metabolic Syndrome: Advancing Disease Management. J Cardiovasc Transl Res (e-pub ahead of print 13 March 2025; [CrossRef]
- Witztum JL, Gaudet D, Freedman SD, Alexander VJ, Digenio A, Williams KR, Yang Q, Hughes SG, Geary RS, Arca M, Stroes ESG, Bergeron J, Soran H, Civeira F, Hemphill L, Tsimikas S, Blom DJ, O’Dea L, Bruckert E. Volanesorsen and Triglyceride Levels in Familial Chylomicronemia Syndrome. N Engl J Med 2019; 381: 531–542. [CrossRef]
- Stitziel NO. Reducing the Risk of Pancreatitis by Inhibiting APOC3. N Engl J Med 2025; 392: 197–199. [CrossRef]
- Raal FJ, Kallend D, Ray KK, Turner T, Koenig W, Wright RS, Wijngaard PLJ, Curcio D, Jaros MJ, Leiter LA, Kastelein JJP. Inclisiran for the Treatment of Heterozygous Familial Hypercholesterolemia. N Engl J Med 2020; 382: 1520–1530. [CrossRef]
- O’Donoghue ML, Rosenson RS, López JAG, Lepor NE, Baum SJ, Stout E, Gaudet D, Knusel B, Kuder JF, Murphy SA, Wang H, Wu Y, Shah T, Wang J, Wilmanski T, Sohn W, Kassahun H, Sabatine MS. The Off-Treatment Effects of Olpasiran on Lipoprotein(a) Lowering. J Am Coll Cardiol 2024; 84: 790–797. [CrossRef]
- Ridker PM, Devalaraja M, Baeres FMM, Engelmann MDM, Hovingh GK, Ivkovic M, Lo L, Kling D, Pergola P, Raj D, Libby P, Davidson M. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. The Lancet 2021; 397: 2060–2069. [CrossRef]
- Chang B, Laffin LJ, Sarraju A, Nissen SE. Obicetrapib—the Rebirth of CETP Inhibitors? Curr Atheroscler Rep 2024; 26: 603–608. [CrossRef]
- Nicholls SJ, Ditmarsch M, Kastelein JJ, Rigby SP, Kling D, Curcio DL, Alp NJ, Davidson MH. Lipid lowering effects of the CETP inhibitor obicetrapib in combination with high-intensity statins: a randomized phase 2 trial. Nat Med 2022; 28: 1672–1678. [CrossRef]
- Zhu H, Qiao S, Zhao D, Wang K, Wang B, Niu Y, Shang S, Dong Z, Zhang W, Zheng Y, Chen X. Machine learning model for cardiovascular disease prediction in patients with chronic kidney disease. Front Endocrinol 2024; 15: 1390729. [CrossRef]
- Ndumele CE, Rangaswami J, Chow SL, Neeland IJ, Tuttle KR, Khan SS, Coresh J, Mathew RO, Baker-Smith CM, Carnethon MR, Despres J-P, Ho JE, Joseph JJ, Kernan WN, Khera A, Kosiborod MN, Lekavich CL, Lewis EF, Lo KB, Ozkan B, Palaniappan LP, Patel SS, Pencina MJ, Powell-Wiley TM, Sperling LS, Virani SS, Wright JT, Rajgopal Singh R, Elkind MSV, on behalf of the American Heart Association. Cardiovascular-Kidney-Metabolic Health: A Presidential Advisory From the American Heart Association. Circulation 2023; 148: 1606–1635. [CrossRef]
- Loftus TJ, Shickel B, Ozrazgat-Baslanti T, Ren Y, Glicksberg BS, Cao J, Singh K, Chan L, Nadkarni GN, Bihorac A. Artificial intelligence-enabled decision support in nephrology. Nat Rev Nephrol 2022; 18: 452–465. [CrossRef]
- Liu J, Liu Z, Liu C, Sun H, Li X, Yang Y. Integrating Artificial Intelligence in the Diagnosis and Management of Metabolic Syndrome: A Comprehensive Review. Diabetes Metab Res Rev 2025; 41: e70039. [CrossRef]
- Muse ED, Topol EJ. Transforming the cardiometabolic disease landscape: Multimodal AI-powered approaches in prevention and management. Cell Metab 2024; 36: 670–683. [CrossRef]
- Vrbaški D, Vrbaški M, Kupusinac A, Ivanović D, Stokić E, Ivetić D, Doroslovački K. Methods for algorithmic diagnosis of metabolic syndrome. Artif Intell Med 2019; 101: 101708. [CrossRef]
- Gill SK, Karwath A, Uh H-W, Cardoso VR, Gu Z, Barsky A, Slater L, Acharjee A, Duan J, Dall’Olio L, El Bouhaddani S, Chernbumroong S, Stanbury M, Haynes S, Asselbergs FW, Grobbee DE, Eijkemans MJC, Gkoutos GV, Kotecha D, BigData@Heart Consortium and the cardAIc group, Bunting KV, Tica O, Mobley AR, Wang X, Champsi A, Haider NA, Ventura M, Young A, McGreavy P, Castellani G, Bradlow W, O’Regan D, Center J. Artificial intelligence to enhance clinical value across the spectrum of cardiovascular healthcare. Eur Heart J 2023; 44: 713–725. [CrossRef]
- Khan SS, Coresh J, Pencina MJ, Ndumele CE, Rangaswami J, Chow SL, Palaniappan LP, Sperling LS, Virani SS, Ho JE, Neeland IJ, Tuttle KR, Rajgopal Singh R, Elkind MSV, Lloyd-Jones DM, on behalf of the American Heart Association. Novel Prediction Equations for Absolute Risk Assessment of Total Cardiovascular Disease Incorporating Cardiovascular-Kidney-Metabolic Health: A Scientific Statement From the American Heart Association. Circulation 2023; 148: 1982–2004. [CrossRef]
- An overview of cardiovascular-kidney-metabolic syndrome. Am J Manag Care 2024; 30: S181–S188. [CrossRef]
- Li N, Zhou G, Zheng Y, Lv D, Zhu X, Wei P, Zheng M, Liu S, Zhou E, Sun W, Zhang L. Effects of SGLT2 inhibitors on cardiovascular outcomes in patients with stage 3/4 CKD: A meta-analysis. PLOS ONE 2022; 17: e0261986. [CrossRef]
- Usman MS, Siddiqi TJ, Anker SD, Bakris GL, Bhatt DL, Filippatos G, Fonarow GC, Greene SJ, Januzzi JL, Khan MS, Kosiborod MN, McGuire DK, Piña IL, Rosenstock J, Vaduganathan M, Verma S, Zieroth S, Butler J. Usman, Muhammad Shariq, Tariq Jamal Siddiqi, Stefan D. Anker, George L. Bakris, Deepak L. Bhatt, Gerasimos Filippatos, Gregg C. Fonarow, et al. “Effect of SGLT2 Inhibitors on Cardiovascular Outcomes Across Various Patient Populations.” Journal of the American College of Cardiology 81, no. 25 (June 2023): 2377–87. J Am Coll Cardiol 2023; 81: 2377–2387. [CrossRef]
- Badve SV, Bilal A, Lee MMY, Sattar N, Gerstein HC, Ruff CT, McMurray JJV, Rossing P, Bakris G, Mahaffey KW, Mann JFE, Colhoun HM, Tuttle KR, Pratley RE, Perkovic V. Effects of GLP-1 receptor agonists on kidney and cardiovascular disease outcomes: a meta-analysis of randomised controlled trials. Lancet Diabetes Endocrinol 2025; 13: 15–28. [CrossRef]
- A Randomized, Double-Blind, Placebo-Controlled, Phase 3 Study of AKCEA-APOCIII-LRX (olezarsen) Administered Subcutaneously to Patients with Familial Chylomicronemia Syndrome (FCS). 2020. Available from: https://clinicaltrials.gov/ct2/show/NCT04568434.
- McGowan MP, Tardif J-C, Ceska R, Burgess LJ, Soran H, Gouni-Berthold I, Wagener G, Chasan-Taber S. Randomized, Placebo-Controlled Trial of Mipomersen in Patients with Severe Hypercholesterolemia Receiving Maximally Tolerated Lipid-Lowering Therapy. PLoS ONE 2012; 7: e49006. [CrossRef]
- A multicenter trial assessing the impact of lipoprotein(a) lowering with pelacarsen (TQJ230) on the rate of weekly lipoprotein apheresis sessions in patients with hyperlipoproteinemia(a) and established cardiovascular disease in Germany. 2022. Available from: https://clinicaltrials.gov/ct2/show/NCT05305664.
- Double-blind, placebo-controlled, phase 3 study to evaluate the efficacy and safety of plozasiran in adults with severe hypertriglyceridemia (SHTG). 2024. Available from: https://clinicaltrials.gov/ct2/show/NCT06347016.
- A placebo-controlled, double-blind, randomized trial to evaluate the effect of 300 mg of inclisiran sodium given as subcutaneous injections in subjects with atherosclerotic cardiovascular disease (ASCVD) and elevated low-density lipoprotein cholesterol (LDL-C) (ORION-10). 2017. Available from: https://clinicaltrials.gov/ct2/show/NCT03399370.
- A Phase 3, Randomized, Double-Blind, Placebo-Controlled Study to Investigate the Effect of Lepodisiran on the Reduction of Major Adverse Cardiovascular Events in Adults With Elevated Lipoprotein(a) Who Have Established Atherosclerotic Cardiovascular Disease or Are at Risk for a First Cardiovascular Event – ACCLAIM-Lp(a). 2024. Available from: https://clinicaltrials.gov/ct2/show/NCT06292013.
- Olpasiran Trials of Cardiovascular Events and Lipoprotein(a) Reduction (OCEAN(a)) - Outcomes Trial. 2022. Available from: https://clinicaltrials.ucsd.edu/trial/NCT05581303.
- ZEUS – Effects of ziltivekimab versus placebo on cardiovascular outcomes in participants with established atherosclerotic cardiovascular disease, chronic kidney disease and systemic inflammation. 2021. Available from: https://clinicaltrials.gov/ct2/show/NCT05021835.
- A Placebo-Controlled, Double-Blind, Randomized Phase 3 Study to Evaluate the Effect of 10 mg Obicetrapib in Participants with HeFH and/or ASCVD Who Are Not Adequately Controlled by Their Lipid Modifying Therapies (BROADWAY). 2021. Available from: https://clinicaltrials.gov/ct2/show/NCT05142722.
- Safety and Effectiveness of Evinacumab for the Treatment of Homozygous Familial Hypercholesterolemia in a Real Life Setting in Canada. 2023. Available from: https://clinicaltrials.gov/ct2/show/NCT05611528.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




