Submitted:
26 June 2025
Posted:
27 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Investigational Products
2.3. Evaluations
- -
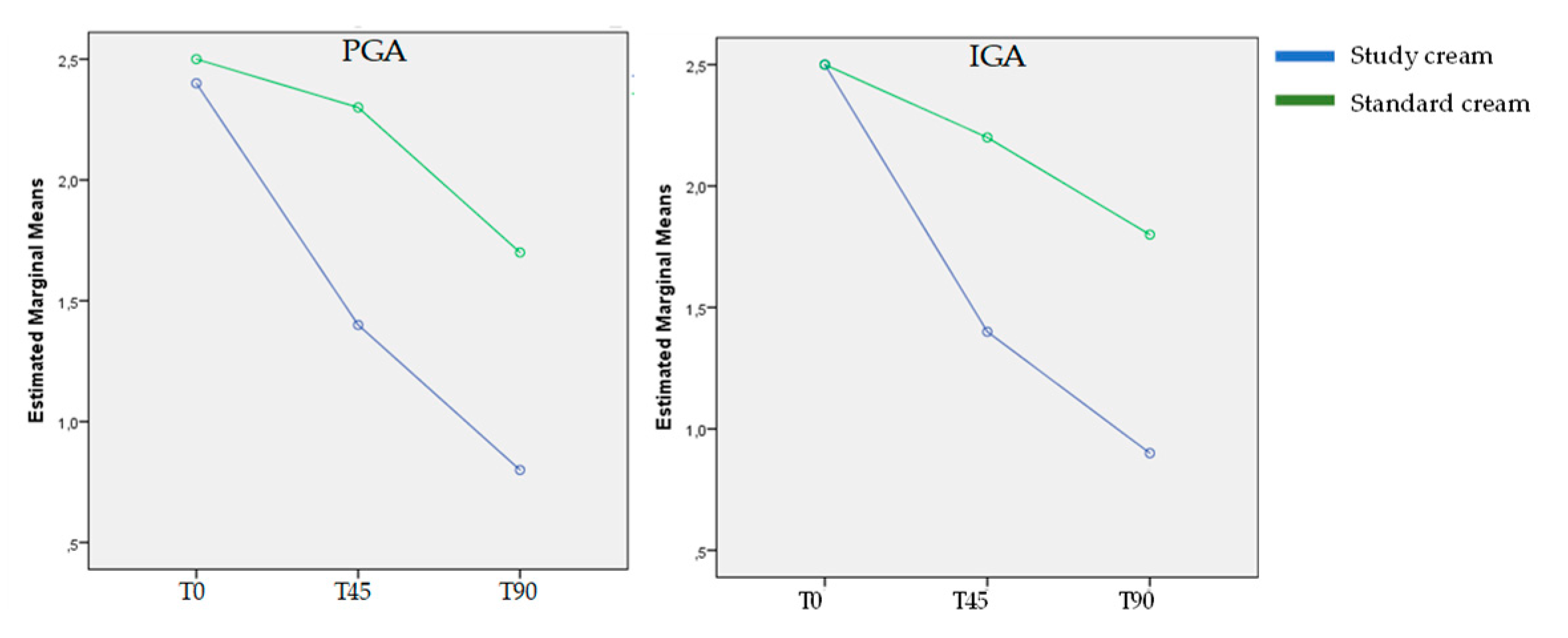
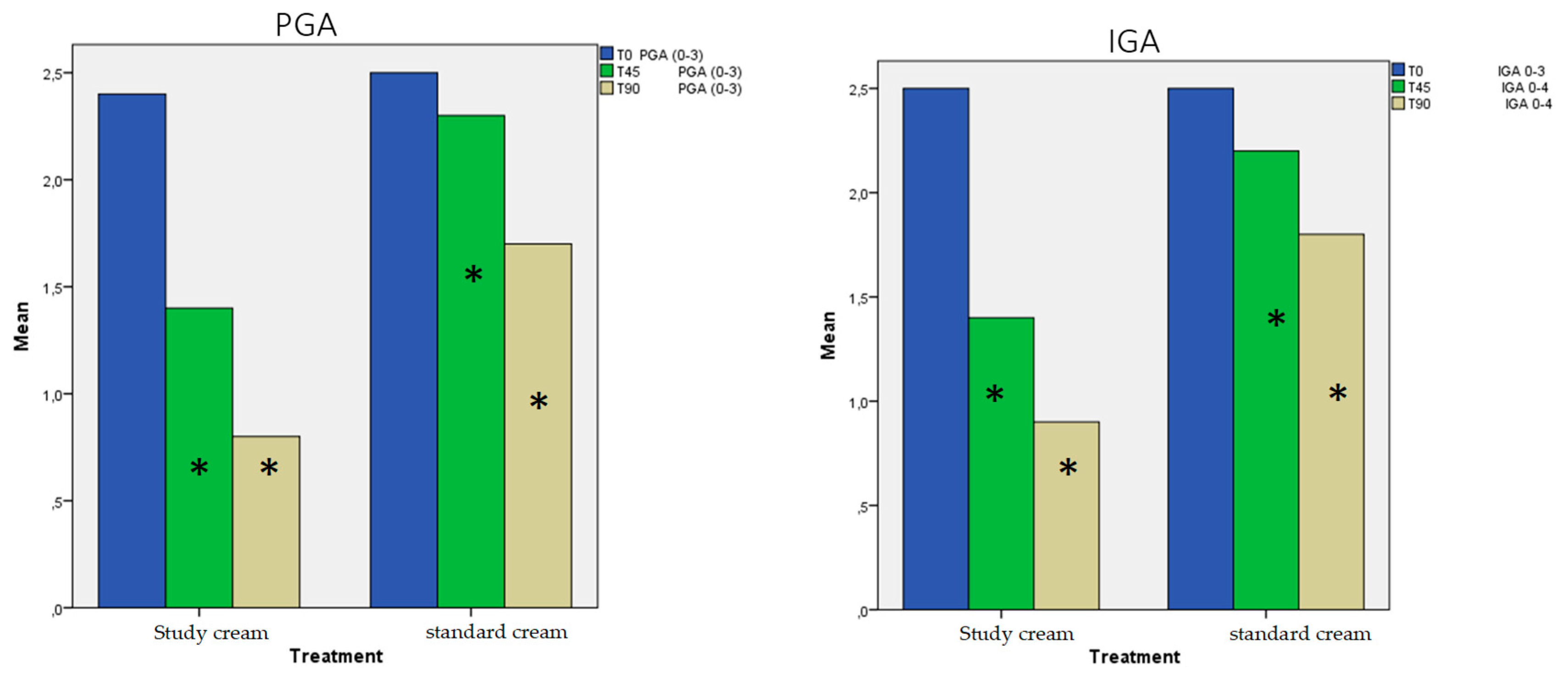
- PGA (Patient global assessment): clear (0), almost clear (1), mild (2), moderate (3), severe (4)
- -
- IGA (Investigator global assessment): 0: healthy clear skin with no evidence of acne vulgaris; 1: almost clear; rare noninflammatory lesions present; rare noninflamed re-solving papules (may be hyperpigmented but not pink-red); 2: some noninflammatory lesions present; few inflammatory lesions (papules/pustules only; no nodulocystic le-sions); 3: noninflammatory lesions predominate; multiple inflammatory lesions present; several to many comedones and papules/pustules; one small nodulocystic lesion; 4: in-flammatory lesions predominate; many comedones and papules/pustules; may or may not be a few nodulocystic lesions; 5:highly inflammatory lesions predominate; variable number of comedones; many papules/pustules and nodulocystic lesions. [20]
- -
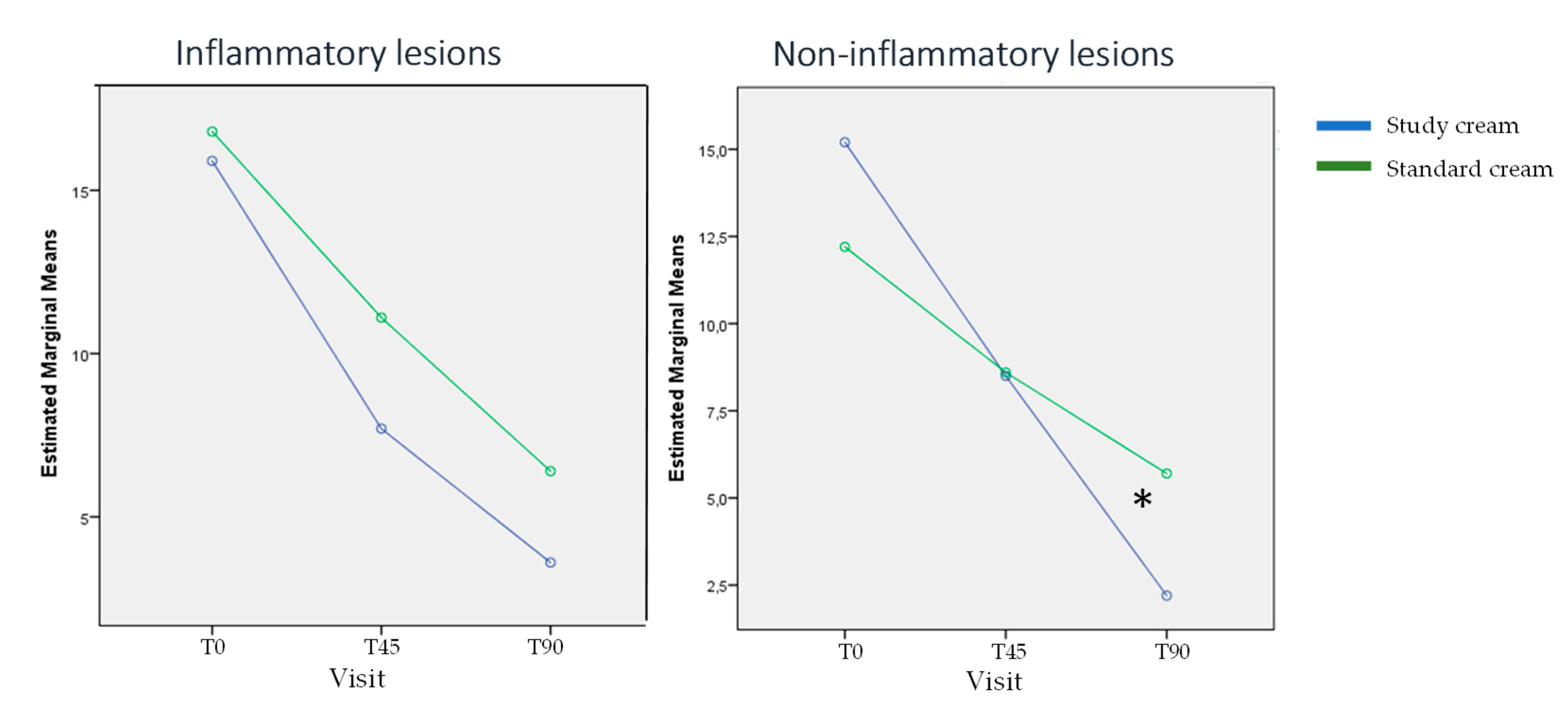
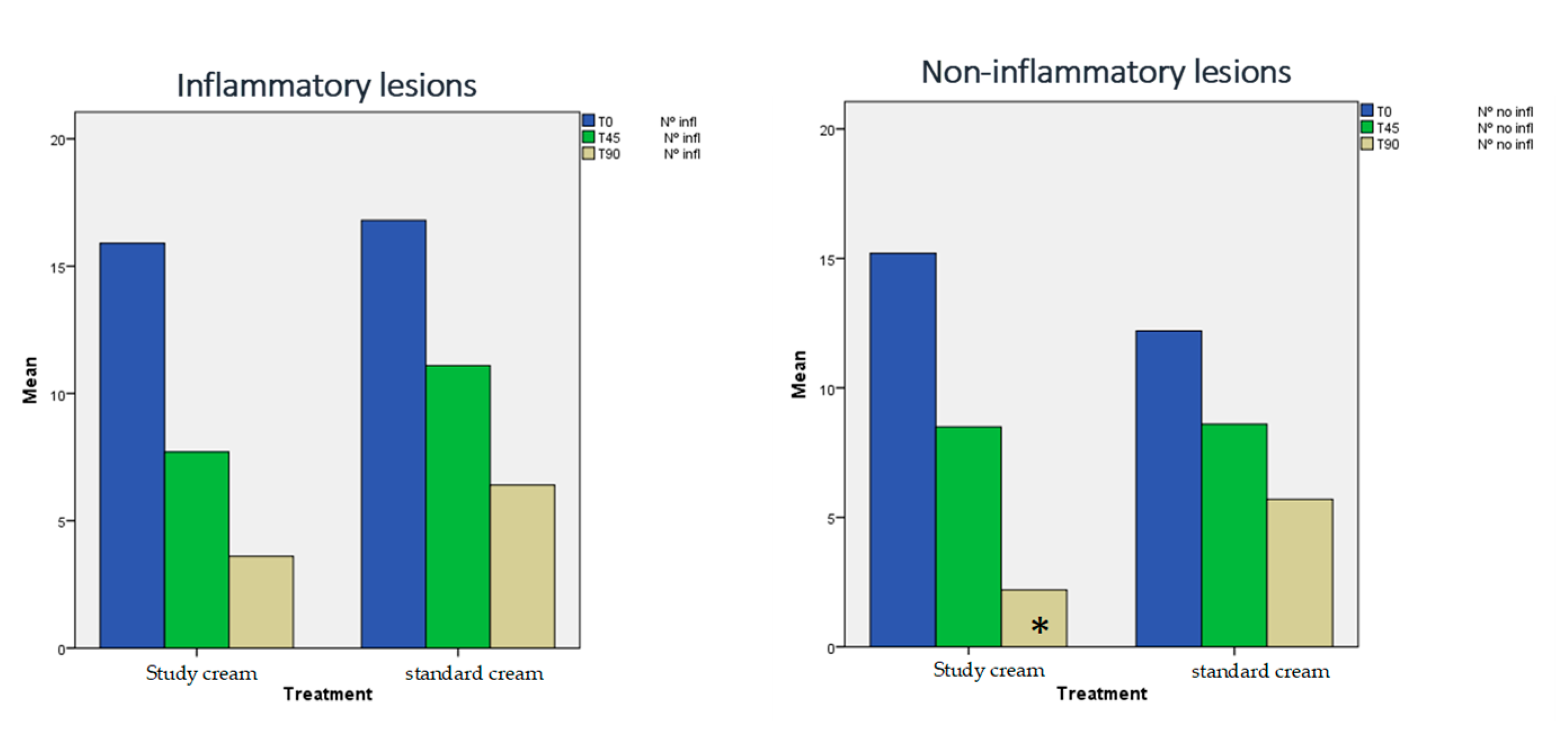
- Number of inflammatory lesions
- -
- Number of non-inflammatory lesions
- -
- Scarring (0: none, 1: mild, 2: moderate, 3: severe)
- -
- Semi-quantitative hydration measurement
- -
- PBI (Patient Benefit Index): Expectation Score (Annex I)
- -
- Skin (facial and extra-facial) and mucosal tolerance: Rated as -2 (very poor), -1 (poor), 0 (neutral), 1 (good), 2 (very good). Specifically, erythema, xerosis, desquamation, and pruritus were assessed on a scale of 0-3.
- -
- Treatment adherence: Rated as never (0), sometimes (1), always (2).
- -
- Patient global improvement assessment: Rated on a scale from 0 to 3 (0: no change, 1: mild improvement, 2: moderate improvement, 3: significant improvement).
- -
- Investigator global improvement assessment: Rated on a scale from 0 to 3 (0: no change, 1: mild improvement, 2: moderate improvement, 3: significant improvement).
- -
- Overall patient’s satisfaction: 0: none, 1: mild, 2: moderate, 3: intense.
- -
- Patient’s experience of product usage (Annex I)
2.4. Aim of the Study
2.5. Statistical Analysis
3. Results
3.1. Evolution of Acne Severity:
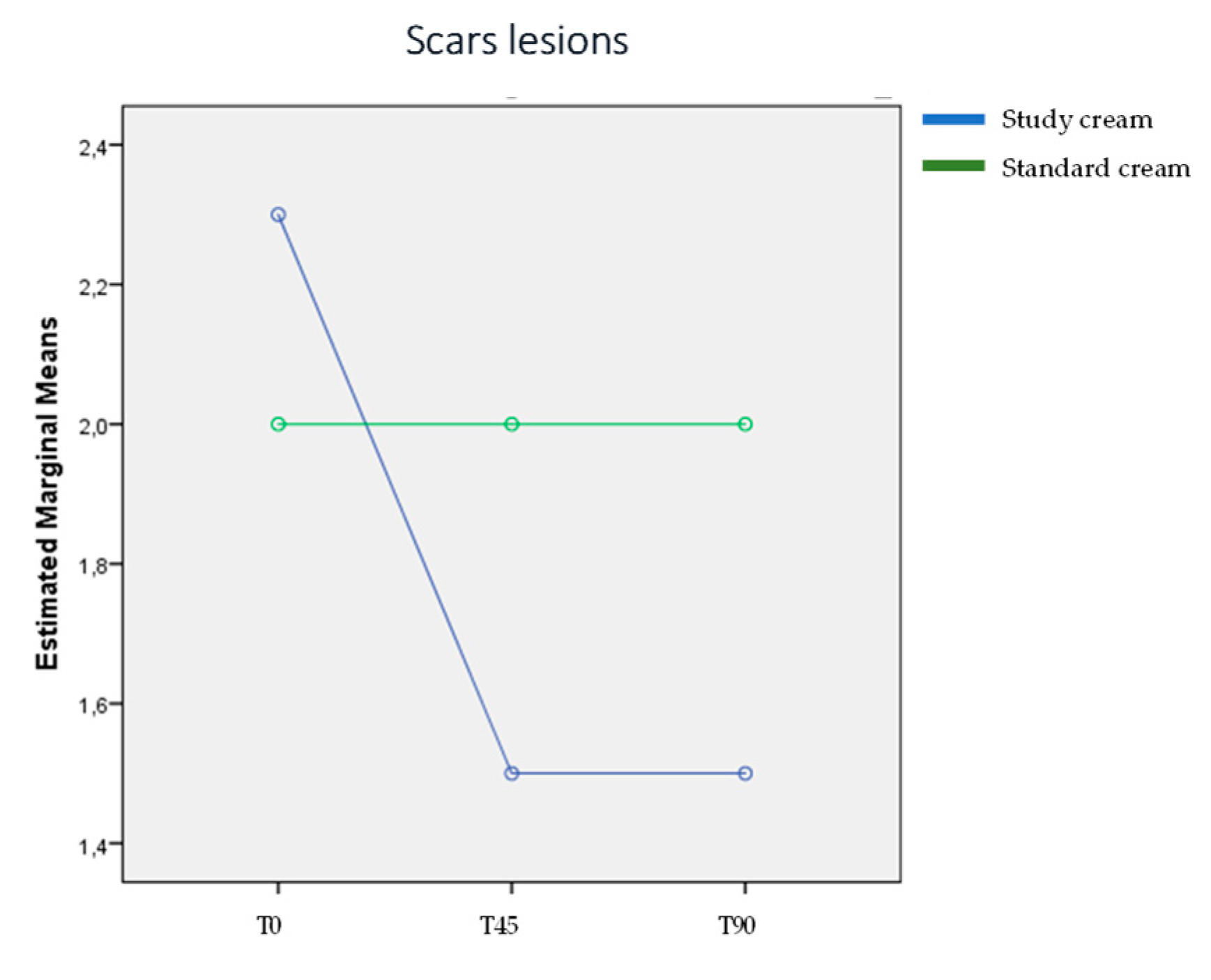
3.2. Scar Grading Evaluation:
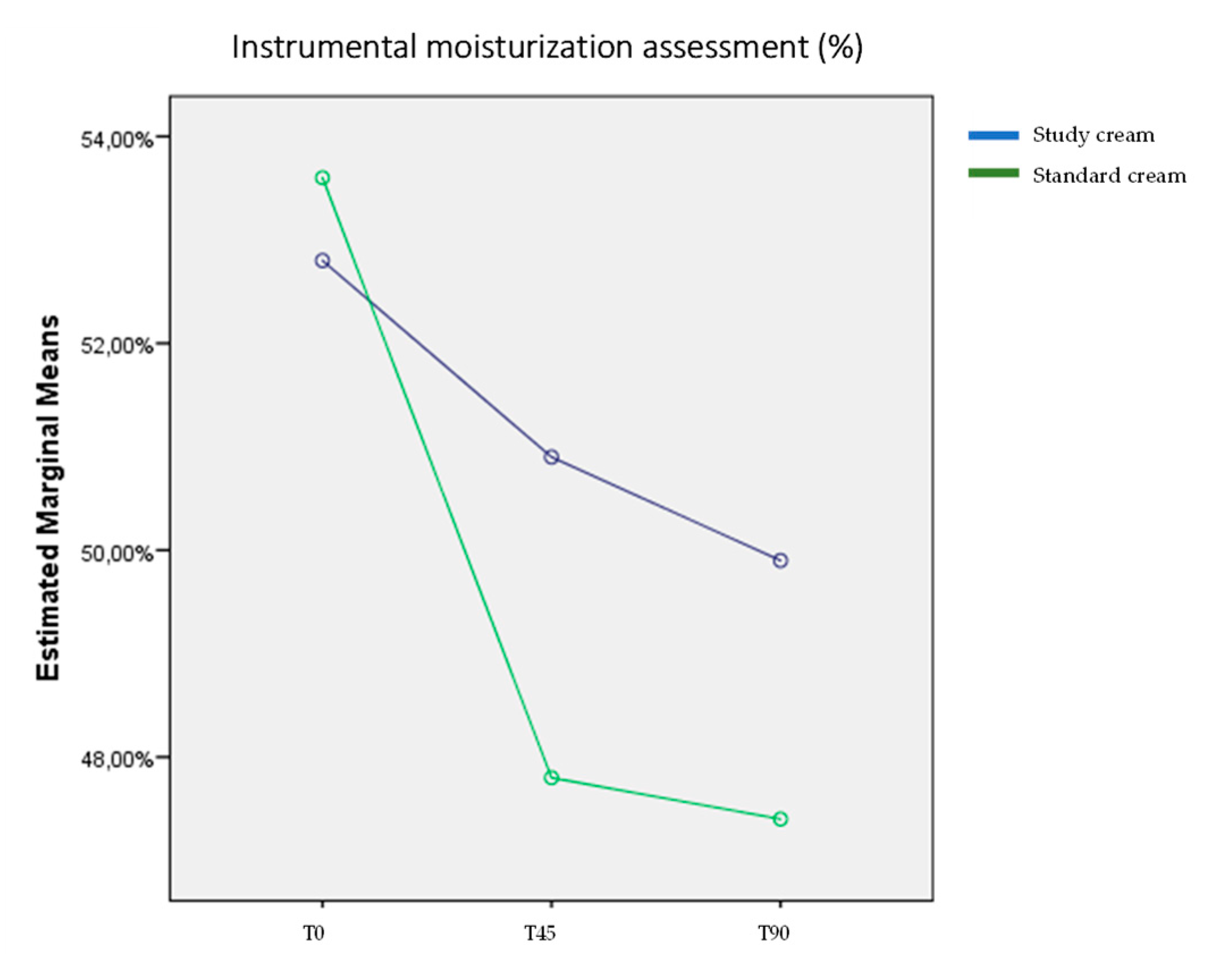
3.3. Instrumental Skin Moisturization
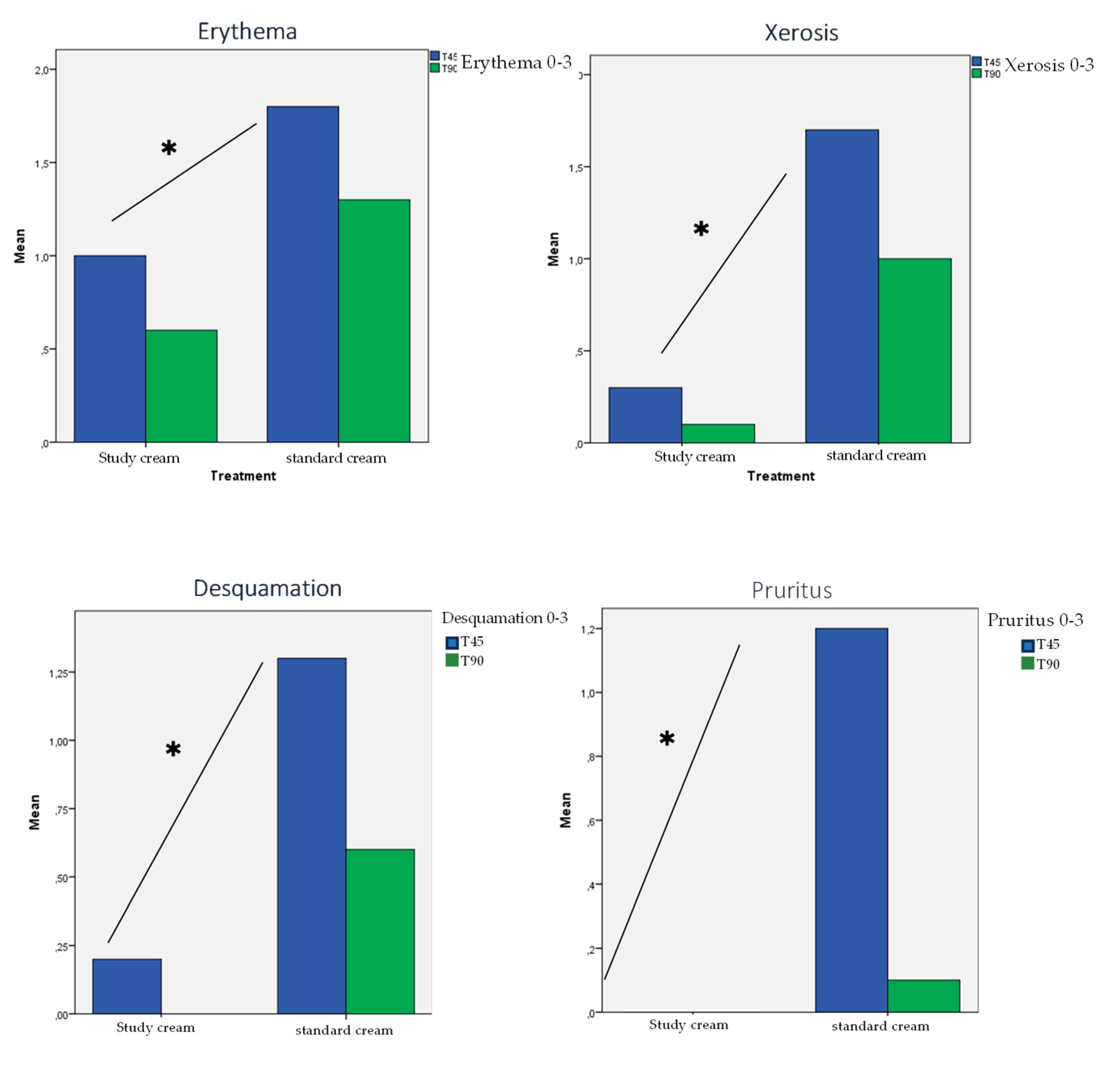
3.4. Skin Tolerance:
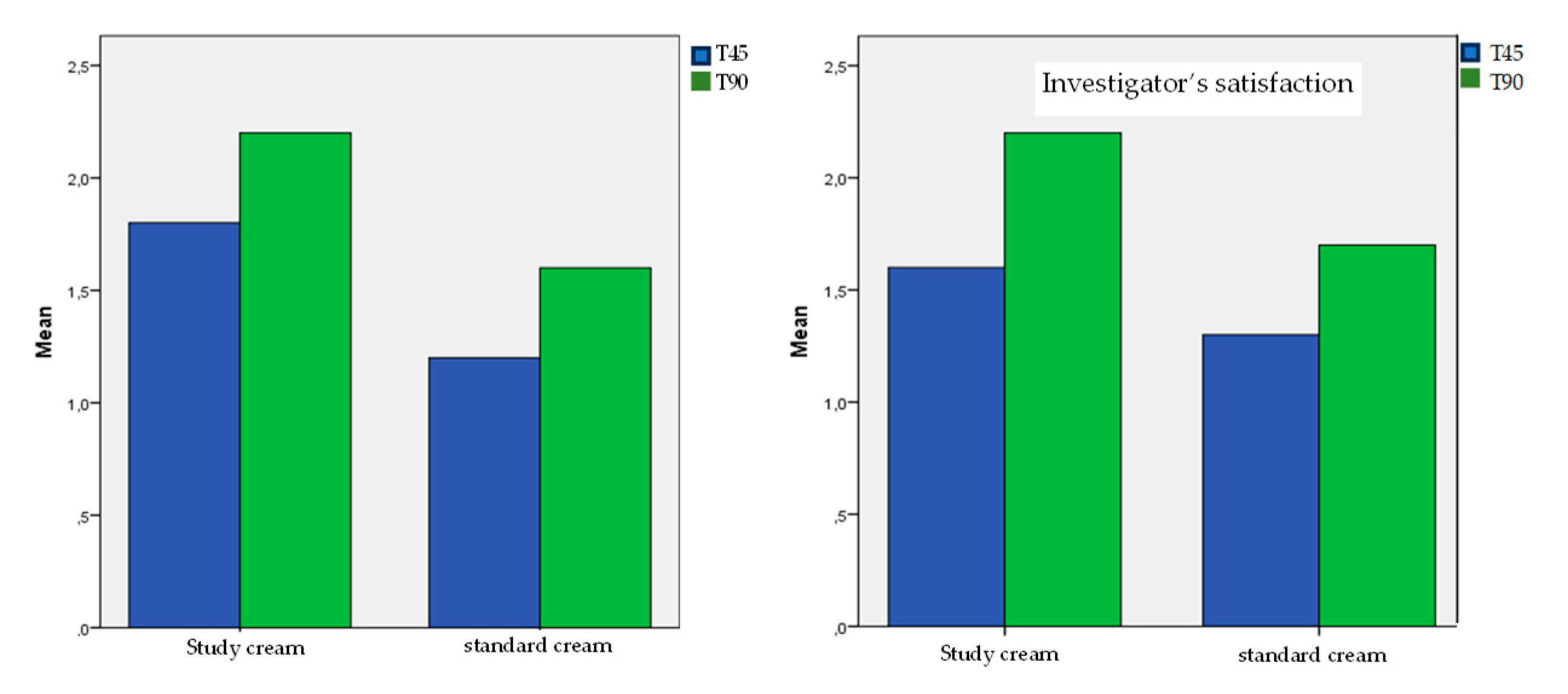
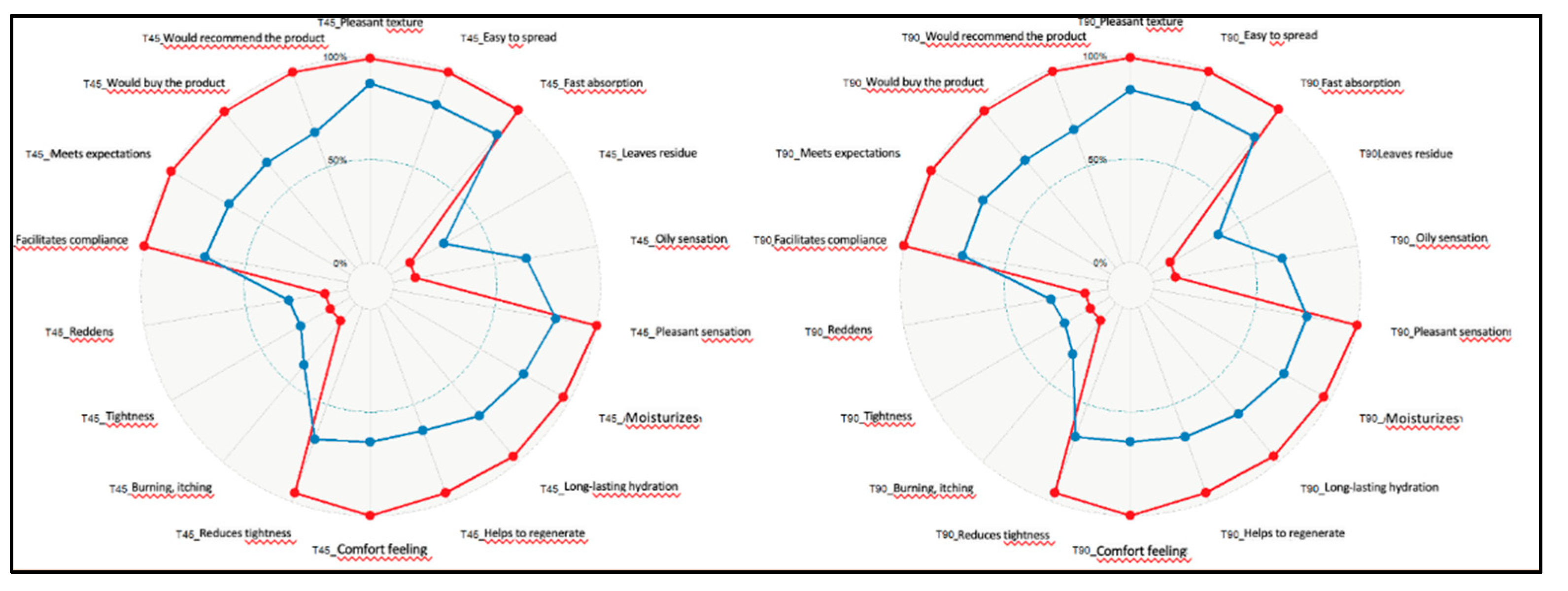
3.5. Subjective Evaluations Described by the Patient:
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1
- Do you expect the product to be pleasant?
- Do you expect it to have a soothing effect?
- Do you expect it to provide long-lasting hydration?
- Do you expect it to help with adherence to the treatment?
- The texture of the product is pleasant.
- The product spreads easily.
- The product is quickly absorbed.
- After absorption, the product leaves residue on the skin (white film, pilling).
- The product leaves a greasy feeling after application.
- The sensation on the skin after application is pleasant.
- The product provides skin hydration.
- The feeling of hydration is long-lasting.
- The product helps to regenerate/repair the skin.
- The product provides a feeling of comfort on the skin.
- The product reduces feelings of tightness and irritation.
- After application, a burning or stinging sensation is felt on the skin.
- After application, a feeling of tightness is felt on the skin.
- After application, the skin becomes red.
- The product supports adherence to the Dercutane (oral isotretinoin) treatment.
- The product has met your expectations.
- Would you purchase this product?
- Would you recommend this product?
- The texture of the product is pleasant.
- The product spreads easily.
- The product is quickly absorbed.
- After absorption, the product leaves residue on the skin (white film, pilling).
- The product leaves a greasy feeling after application.
- The sensation on the skin after application is pleasant.
- The product provides skin hydration.
- The feeling of hydration is long-lasting.
- The product helps to regenerate/repair the skin.
- The product provides a feeling of comfort on the skin.
- The product reduces feelings of tightness and irritation.
- After application, a burning or stinging sensation is felt on the skin.
- After application, a feeling of tightness is felt on the skin.
- After application, the skin becomes red.
- The product supports adherence to the Dercutane (oral isotretinoin) treatment.
- The product has met your expectations.
- Would you purchase this product?
- Would you recommend this product?
References
- Bettoli V, Guerra-Tapia A, Herane MI, Piquero-Martín J. Challenges and Solutions in Oral Isotretinoin in Acne: Reflections on 35 Years of Experience. Clin Cosmet Investig Dermatol. 2019 Dec 30;12:943-951. PMID: 32021364; PMCID: PMC6951028. [CrossRef]
- Ward A, Brogden RN, Heel RC, et al. Isotretinoin: a review of its pharmacologic properties and therapeutic efficacy in acne and other skin disorders. Drugs. 1984;28:6-37.
- Del Rosso JQ. Clinical relevance of skin barrier changes associated with the use of oral isotretinoin: the importance of barrier repair therapy in patient management. J Drugs Dermatol. 2013 Jun 1;12(6):626-31. PMID: 23839177.
- Williams ML, Elias PM. Nature of skin fragility in patients receving retinoids for systemic effect. Arch Dermatol. 1981;117(10):611-619.
- Elias PM. Epidermal effects of retinoids: supramolecular observations and clinical implications. J Am Acad Dermatol. 1986;15(4 Pt 2):797-809.
- Reyes-Hadsall S, Ju T, Keri JE. Use of Oral Supplements and Topical Adjuvants for Isotretinoin-Associated Side Effects: A Narrative Review. Skin Appendage Disord. 2024 Feb;10(1):1-9. Epub 2023 Oct 20. PMID: 38313565; PMCID: PMC10836938. [CrossRef]
- Herane MI, Fuenzalida H, Zegpi E, De Pablo C, Espadas MJ, Trullás C, Mirada A, Martin GG. Specific gel-cream as adjuvant to oral isotretinoin improved hydration and prevented TEWL increase--a double-blind, randomized, placebo-controlled study. J Cosmet Dermatol. 2009 Sep;8(3):181-5. PMID: 19735515. [CrossRef]
- Bertocchi M, Isani G, Medici F, Andreani G, Tubon Usca I, Roncada P, Forni M, Bernardini C. Anti-Inflammatory Activity of Boswellia serrata Extracts: An In Vitro Study on Porcine Aortic Endothelial Cells. Oxid Med Cell Longev. 2018 Jun 25;2018:2504305.. PMID: 30046370; PMCID: PMC6036794. [CrossRef]
- Danby SG, Andrew PV, Kay LJ, Pinnock A, Chittock J, Brown K, Williams SF, Cork MJ. Enhancement of stratum corneum lipid structure improves skin barrier function and protects against irritation in adults with dry, eczema-prone skin. Br J Dermatol. 2022 May;186(5):875-886. Epub 2022 Apr 3. PMID: 34921679; PMCID: PMC9321855. [CrossRef]
- Tempark T, Shem A, Lueangarun S. Efficacy of ceramides and niacinamide-containing moisturizer versus hydrophilic cream in combination with topical anti-acne treatment in mild to moderate acne vulgaris: A split face, double-blinded, randomized controlled trial. J Cosmet Dermatol. 2024 May;23(5):1758-1765. Epub 2024 Feb 1. PMID: 38299457. [CrossRef]
- Isoda K, Seki T, Inoue Y, et al. Efficacy of the combined use of a facial cleanser and moisturizers for the care of mild acne patients with sensitive skin. J Dermatol. 2015;42(2):181-188.
- Burke K, Clive J, Combs G, Commisso J, Keen C, Nakamura R. Effects of topical and oral vitamin E on pigmentation and skin can- cer induced by ultraviolet irradiation in Skh:2 hairless mice. Nutr Cancer. 2000;38(1):87-97.
- Keen MA, Hassan I. Vitamin E in dermatology. Indian Dermatol Online J. 2016 Jul-Aug;7(4):311-5. PMID: 27559512; PMCID: PMC4976416. [CrossRef]
- Truchuelo MT, Vitale M (2020) A cosmetic treatment based on the secretion of Cryptomphalus aspersa 40% improves the clinical results after the use of nonablative fractional laser in skin aging. J Cosmet Dermatol 19: 622-628.
- Fondevilla A, Moreno-Olmedo E, Bernal JM, Belmonte MJ, Nicolás A, López E. Prevention of radiation induced dermatitis in head and neck cancer patients using cryptomphalus aspersa secretion. Clin Transl Oncol. 2024 Sep 25. Epub ahead of print. PMID: 39322924. [CrossRef]
- Castro B, de Paz N, González S, Rodríguez-Luna A. SCA® Slows the Decline of Functional Parameters Associated with Senescence in Skin Cells. Int J Mol Sci. 2022 Jun 10;23(12):6538. PMID: 35742982; PMCID: PMC9224471. [CrossRef]
- Lim VZ, Yong AA, Tan WPM, Zhao X, Vitale M, Goh CL. Efficacy and Safety of a New Cosmeceutical Regimen Based on the Combination of Snail Secretion Filtrate and Snail Egg Extract to Improve Signs of Skin Aging. J Clin Aesthet Dermatol. 2020 Mar;13(3):31-36. Epub 2020 Mar 1. PMID: 32308795; PMCID: PMC7159309.
- Brieva A, Philips N, Tejedor R, Guerrero A, Pivel JP, et al. Molecular basis for the regenerative properties of a secretion of the mollusk Cryptomphalus aspersa. Skin Pharmacol Physiol 21: 15-22.
- Fernandez-Gonzalez P, Vitale M, Truchuelo MT. Early and maintained application of the secretion of Cryptomphalus aspersa (SCA) 40% improves cutaneous healing after ablative fractional laser in skin aging. J Cosmet Dermatol. 2021 Apr;20(4):1140-1145. Epub 2020 Sep 24. PMID: 32929855; PMCID: PMC8048427. [CrossRef]
- Del Rosso JQ. Defining criteria used to evaluate response to treatment of acne vulgaris. Cutis. 2006 Aug;78(2):117-21. PMID: 16983900.
- Piquero-Martin J, Misticone S, Piquero-Casals V, Piquero-Casals J. Topic therapy-mini isotretinoin doses vs topic therapy-systemic anti- biotics in the moderate acne patients. Ann Dermatol Venereol. 2002;129:S382.
- Park KY, Kim DH, Jeong MS, et al. Changes of antimicrobial peptides and transepidermal water loss after topical application of tacrolimus and ceramide-dominant emollient in patients with atopic dermatitis. J Korean Medi- cal Sci. 2010;25(5):766-771.
- Goh CL, Abad-Casintahan F, Aw DC, Baba R, Chan LC, Hung NT, Kulthanan K, Leong HN, Medina-Oblepias MS, Noppakun N, Sitohang IB, Sugito TL, Wong SN. South-East Asia study alliance guidelines on the management of acne vulgaris in South-East Asian patients. J Dermatol. 2015 Oct;42(10):945-53. Epub 2015 Jul 25. PMID: 26211507. [CrossRef]
- Lain E, Andriessen AE. Choosing the Right Partner: Complementing Prescription Acne Medication With Over-the-Counter Cleansers and Moisturizers. J Drugs Dermatol. 2020 Nov 1;19(11):1069-1075. PMID: 33196748. [CrossRef]
| T45 | T90 | |||
| Biretix® isorepair | standard cream | Biretix® isorepair | standard cream | |
| Erythema. T45 vs T90 p= 0.029. SD between treatments p 0.02 | 20% moderate | 60% moderate or severe | 20% moderate | 30% moderate or severe |
| Xerosis. T45 vs T90 p= 0.006. SD between treatments p <0.001 |
only 30% mild | 70% moderate or severe | only 10% mild | 30% moderate. |
| Desquamation. T45 vs T90 p=0.006. SD between treatments p =0.002 | only 20% mild | 40% moderate | None | Only 10% severe, 30% mild |
| Pruritus .T45 vs T90 p = 0.005. SD between treatments p =0.001 | None | 20% severe, 60% mild | None | 10% mild |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




