Submitted:
26 June 2025
Posted:
27 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Fluorescent-Based Sensors
2.1. Fluorescent RNA Aptamers
2.2. Fluorescence-Quenching RNA Aptamers
2.3. Molecular Beacon Biosensors
2.4. FRET-Based RNA Sensors
3. Enzymatic and Electrochemical Sensors
3.1. Catalytic RNA Biosensors
3.2. Split RNA Aptamers and Ribozymes
3.3. Electrochemical RNA Sensors
3.4. Field-Effect Transistor (FET)-Based RNA Biosensors:
4. Environmental and Metabolite-Responsive Sensors
4.1. Riboswitch biosensors
4.2. RNA Thermometers
4.3. RNA Sensors for Reactive Oxygen Species (ROS)
4.4. tRNA-Based Biosensors
5. Sequence-Specific Detection Sensors
5.1. CRISPR-Cas13/ and -Cas12a RNA Sensors
5.2. Toehold Switches
5.3. RNA aptamer-Based Sensors for Post-Transcriptional Modifications
6. Structural and Inter-Cellular Communication Sensors
6.1. Circular RNA Sensors
6.2. MS2 System
6.3. Exosomal RNA Sensors
6.4. miRNA Sensors
6.5. LncRNA Sensors
7. Limitations and Challenges
8. Conclusion and Future Directions
References
- Abudayyeh, O.O., Gootenberg, J.S., Essletzbichler, P., Han, S., Joung, J., Belanto, J.J., Verdine, V., Cox, D.B.T., Kellner, M.J., Regev, A., Lander, E.S., Voytas, D.F., Ting, A.Y., Zhang, F., 2017. RNA targeting with CRISPR–Cas13. Nat. 2017 5507675 550, 280–284. [CrossRef]
- Aich, A., Boshnakovska, A., Witte, S., Gall, T., Unthan-Fechner, K., Yousefi, R., Chowdhury, A., Dahal, D., Methi, A., Kaufmann, S., Silbern, I., Prochazka, J., Nichtova, Z., Palkova, M., Raishbrook, M., Koubkova, G., Sedlacek, R., Tröder, S.E., Zevnik, B., Riedel, D., Michanski, S., Möbius, W., Ströbel, P., Lüchtenborg, C., Giavalisco, P., Urlaub, H., Fischer, A., Brügger, B., Jakobs, S., Rehling, P., 2024. Defective mitochondrial COX1 translation due to loss of COX14 function triggers ROS-induced inflammation in mouse liver. Nat. Commun. 2024 151 15, 1–20. [CrossRef]
- Alam, K.K., Tawiah, K.D., Lichte, M.F., Porciani, D., Burke, D.H., 2017. A Fluorescent Split Aptamer for Visualizing RNA-RNA Assembly in Vivo. ACS Synth. Biol. 6, 1710–1721. [CrossRef]
- Alkhamis, O., Canoura, J., Willis, C., Wang, L., Perry, J., Xiao, Y., 2023. Comparison of Aptamer Signaling Mechanisms Reveals Disparities in Sensor Response and Strategies to Eliminate False Signals. J. Am. Chem. Soc. 145, 12407–12422. [CrossRef]
- Aman, R., Mahas, A., Mahfouz, M., 2020. Nucleic Acid Detection Using CRISPR/Cas Biosensing Technologies. ACS Synth. Biol. 9, 1226–1233. [CrossRef]
- Arora, A., Sunbul, M., Jäschke, A., 2015. Dual-colour imaging of RNAs using quencher- and fluorophore-binding aptamers. Nucleic Acids Res. 43, e144–e144. [CrossRef]
- Barckmann, B., Simonelig, M., 2013. Control of maternal mRNA stability in germ cells and early embryos. Biochim. Biophys. Acta - Gene Regul. Mech. 1829, 714–724. [CrossRef]
- Barta, A., Jantsch, M.F., 2017. RNA in Disease and development. RNA Biol. 14, 457–459. [CrossRef]
- Beadle, L.F., Sutcliffe, C., Ashe, H.L., 2024. A simple MiMIC-based approach for tagging endogenous genes to visualise live transcription in Drosophila. Development 151. [CrossRef]
- Bidar, N., Amini, M., Oroojalian, F., Baradaran, B., Hosseini, S.S., Shahbazi, M.A., Hashemzaei, M., Mokhtarzadeh, A., Hamblin, M.R., de la Guardia, M., 2021. Molecular beacon strategies for sensing purpose. TrAC Trends Anal. Chem. 134, 116143. [CrossRef]
- Boussebayle, A., Torka, D., Ollivaud, S., Braun, J., Bofill-Bosch, C., Dombrowski, M., Groher, F., Hamacher, K., Suess, B., 2019. Next-level riboswitch development—implementation of Capture-SELEX facilitates identification of a new synthetic riboswitch. Nucleic Acids Res. 47, 4883–4895. [CrossRef]
- Braselmann, E., Rathbun, C., Richards, E.M., Palmer, A.E., 2020. Illuminating RNA Biology: Tools for Imaging RNA in Live Mammalian Cells. Cell Chem. Biol. 27, 891–903. [CrossRef]
- Braselmann, E., Wierzba, A.J., Polaski, J.T., Chromiński, M., Holmes, Z.E., Hung, S.T., Batan, D., Wheeler, J.R., Parker, R., Jimenez, R., Gryko, D., Batey, R.T., Palmer, A.E., 2018. A multicolor riboswitch-based platform for imaging of RNA in live mammalian cells. Nat. Chem. Biol. 2018 1410 14, 964–971. [CrossRef]
- Bruckhoff, R.W., Becker, O., Steinhilber, D., Suess, B., 2025. Potential and Optimization of Mammalian Splice Riboswitches for the Regulation of Exon Skipping-Dependent Gene Expression and Isoform Switching within the ALOX5 Gene. ACS Synth. Biol. 14. [CrossRef]
- Buchser, R., Sweet, P., Anantharaman, A., Contreras, L., 2023. RNAs as Sensors of Oxidative Stress in Bacteria. Annu. Rev. Chem. Biomol. Eng. 14, 265–281. [CrossRef]
- Buentello, D.C., Garcia-Corral, M., Trujillo-De Santiago, G., Alvarez, M.M., 2024. Neuron(s)-on-a-Chip: A Review of the Design and Use of Microfluidic Systems for Neural Tissue Culture. IEEE Rev. Biomed. Eng. 17, 243–263. [CrossRef]
- Campuzano, S., Pedrero, M., Yánez-Sedeño, P., Pingarrón, J.M., 2019. Advances in Electrochemical (Bio)Sensing Targeting Epigenetic Modifications of Nucleic Acids. Electroanalysis 31, 1816–1832. [CrossRef]
- Catrina, I.E., Bayer, L. V., Omar, O.S., Bratu, D.P., 2019. Visualizing and Tracking Endogenous mRNAs in Live <em>Drosophila melanogaster</em> Egg Chambers. JoVE (Journal Vis. Exp. 2019, e58545. [CrossRef]
- Cawte, A.D., Unrau, P.J., Rueda, D.S., 2020. Live cell imaging of single RNA molecules with fluorogenic Mango II arrays. Nat. Commun. 2020 111 11, 1–11. [CrossRef]
- Chau, T.H.T., Mai, D.H.A., Pham, D.N., Le, H.T.Q., Lee, E.Y., 2020. Developments of Riboswitches and Toehold Switches for Molecular Detection-Biosensing and Molecular Diagnostics. Int. J. Mol. Sci. 21. [CrossRef]
- Chen, W., Zhao, X., Yang, N., Li, X., 2023. Single mRNA Imaging with Fluorogenic RNA Aptamers and Small-molecule Fluorophores. Angew. Chemie 135, e202209813. [CrossRef]
- Chen, X., Zhang, D., Su, N., Bao, B., Xie, X., Zuo, F., Yang, L., Wang, H., Jiang, L., Lin, Q., Fang, M., Li, N., Hua, X., Chen, Z., Bao, C., Xu, J., Du, W., Zhang, L., Zhao, Y., Zhu, L., Loscalzo, J., Yang, Y., 2019. Visualizing RNA dynamics in live cells with bright and stable fluorescent RNAs. Nat. Biotechnol. 2019 3711 37, 1287–1293. [CrossRef]
- Chen, Z., Chen, W., Xu, C., Song, H., Ji, X., Jiang, H., Duan, H., Li, Z., Gao, W., Yao, T., Zhang, Z., He, L., Yin, Y., Yang, N., Tian, W., Wu, J., Li, X., 2025. Near-infrared fluorogenic RNA for in vivo imaging and sensing. Nat. Commun. 2025 161 16, 1–15. [CrossRef]
- Choi, H.M.T., Chang, J.Y., Trinh, L.A., Padilla, J.E., Fraser, S.E., Pierce, N.A., 2010. Programmable in situ amplification for multiplexed imaging of mRNA expression. Nat. Biotechnol. 2010 2811 28, 1208–1212. [CrossRef]
- Chu, J., Ejaz, A., Lin, K.M., Joseph, M.R., Coraor, A.E., Drummond, D.A., Squires, A.H., 2024. Single-molecule fluorescence multiplexing by multi-parameter spectroscopic detection of nanostructured FRET labels. Nat. Nanotechnol. 2024 198 19, 1150–1157. [CrossRef]
- Debiais, M., Lelievre, A., Smietana, M., Müller, S., 2020. Splitting aptamers and nucleic acid enzymes for the development of advanced biosensors. Nucleic Acids Res. 48, 3400–3422. [CrossRef]
- Dodbele, S., Mutlu, N., Wilusz, J.E., 2021. Best practices to ensure robust investigation of circular RNAs: pitfalls and tips. EMBO Rep. 22. [CrossRef]
- Du, J., Dartawan, R., Rice, W., Gao, F., Zhou, J.H., Sheng, J., 2022. Fluorescent Platforms for RNA Chemical Biology Research. Genes 2022, Vol. 13, Page 1348 13, 1348. [CrossRef]
- Dykstra, P.B., Kaplan, M., Smolke, C.D., 2022. Engineering synthetic RNA devices for cell control. Nat. Rev. Genet. 2022 234 23, 215–228. [CrossRef]
- Eck, E., Moretti, B., Schlomann, B.H., Bragantini, J., Lange, M., Zhao, X., VijayKumar, S., Valentin, G., Loureiro, C., Soroldoni, D., Royer, L.A., Oates, A.C., Garcia, H.G., 2024. Single-cell transcriptional dynamics in a living vertebrate. bioRxiv 2024.01.03.574108. [CrossRef]
- Englert, D., Burger, E.M., Grün, F., Verma, M.S., Lackner, J., Lampe, M., Bühler, B., Schokolowski, J., Nienhaus, G.U., Jäschke, A., Sunbul, M., 2023. Fast-exchanging spirocyclic rhodamine probes for aptamer-based super-resolution RNA imaging. Nat. Commun. 2023 141 14, 1–13. [CrossRef]
- Eroshkin, F.M., Fefelova, E.A., Bredov, D. V., Orlov, E.E., Kolyupanova, N.M., Mazur, A.M., Sokolov, A.S., Zhigalova, N.A., Prokhortchouk, E.B., Nesterenko, A.M., Zaraisky, A.G., 2024. Mechanical Tensions Regulate Gene Expression in the Xenopus laevis Axial Tissues. Int. J. Mol. Sci. 25, 870. [CrossRef]
- Falgenhauer, E., Mückl, A., Schwarz-Schilling, M., Simmel, F.C., 2022. Transcriptional Interference in Toehold Switch-Based RNA Circuits. ACS Synth. Biol. 11, 1735–1745. [CrossRef]
- Fan, J., Liu, X., Duan, Z., Zhao, H., Chang, Z., Li, L., 2024. The Regulatory Role of miRNAs in Zebrafish Fin Regeneration. Int. J. Mol. Sci. 25, 10542. [CrossRef]
- Fang, J., Wang, J., Wang, Y., Liu, X., Chen, B., Zou, W., 2023. Ribo-On and Ribo-Off tools using a self-cleaving ribozyme allow manipulation of endogenous gene expression in C. elegans. Commun. Biol. 2023 61 6, 1–13. [CrossRef]
- Fasciano, S., Luo, S., Wang, S., 2023. Long non-coding RNA (lncRNA) MALAT1 in regulating osteogenic and adipogenic differentiation using a double-stranded gapmer locked nucleic acid nanobiosensor. Analyst 148, 6261–6273. [CrossRef]
- Frommer, J., Appel, B., Müller, S., 2015. Ribozymes that can be regulated by external stimuli. Curr. Opin. Biotechnol. 31, 35–41. [CrossRef]
- Furukawa, A., Tanaka, T., Furuta, H., Matsumura, S., Ikawa, Y., 2016. Use of a Fluorescent Aptamer RNA as an Exonic Sequence to Analyze Self-Splicing Ability of a Group I Intron from Structured RNAs. Biol. 2016, Vol. 5, Page 43 5, 43. [CrossRef]
- Galizi, R., Jaramillo, A., 2019. Engineering CRISPR guide RNA riboswitches for in vivo applications. Curr. Opin. Biotechnol. 55, 103–113. [CrossRef]
- Gambill, L., Staubus, A., Mo, K.W., Ameruoso, A., Chappell, J., 2023. A split ribozyme that links detection of a native RNA to orthogonal protein outputs. Nat. Commun. 2023 141 14, 1–12. [CrossRef]
- Gao, Y., Zhang, Y., Zhang, D., Dai, X., Estelle, M., Zhao, Y., 2015. Auxin binding protein 1 (ABP1) is not required for either auxin signaling or Arabidopsis development. Proc. Natl. Acad. Sci. U. S. A. 112, 2275–2280. [CrossRef]
- Gupta, R., Ghosh, A., Chakravarti, R., Singh, R., Ravichandiran, V., Swarnakar, S., Ghosh, D., 2022. Cas13d: A New Molecular Scissor for Transcriptome Engineering. Front. Cell Dev. Biol. 10, 866800. [CrossRef]
- Gupta, R., Laxman, S., 2020. tRNA wobble-uridine modifications as amino acid sensors and regulators of cellular metabolic state. Curr. Genet. 66, 475–480. [CrossRef]
- Hallberg, Z.F., Su, Y., Kitto, R.Z., Hammond, M.C., 2017. Engineering and in vivo applications of riboswitches. Annu. Rev. Biochem. 86, 515–539. [CrossRef]
- Halstead, J.M., Lionnet, T., Wilbertz, J.H., Wippich, F., Ephrussi, A., Singer, R.H., Chao, J.A., 2015. An RNA biosensor for imaging the first round of translation from single cells to living animals. Science (80-. ). 347, 1367–1370. [CrossRef]
- Hamidi-Asl, E., Palchetti, I., Hasheminejad, E., Mascini, M., 2013. A review on the electrochemical biosensors for determination of microRNAs. Talanta 115, 74–83. [CrossRef]
- Han, Y., Yang, D., Jiang, S., Zhao, S., Ma, F., Zhang, C. yang, 2024. Recent advance in optical single-molecule detection of methylation modification and methyl-modifying enzymes. TrAC Trends Anal. Chem. 172, 117553. [CrossRef]
- Hani, S., Cuyas, L., David, P., Secco, D., Whelan, J., Thibaud, M.C., Merret, R., Mueller, F., Pochon, N., Javot, H., Faklaris, O., Maréchal, E., Bertrand, E., Nussaume, L., 2021. Live single-cell transcriptional dynamics via RNA labelling during the phosphate response in plants. Nat. plants 7, 1050–1064. [CrossRef]
- Hashem, A., Hossain, M.A.M., Marlinda, A.R., Mamun, M. Al, Sagadevan, S., Shahnavaz, Z., Simarani, K., Johan, M.R., 2022. Nucleic acid-based electrochemical biosensors for rapid clinical diagnosis: advances, challenges, and opportunities. Crit. Rev. Clin. Lab. Sci. 59, 156–177. [CrossRef]
- Hu, Y., Xu, J., Gao, E., Fan, X., Wei, J., Ye, B., Xu, S., Ma, W., 2023. Enhanced single RNA imaging reveals dynamic gene expression in live animals. Elife 12. [CrossRef]
- Huang, J., Xue, S., Teixeira, A.P., Fussenegger, M., 2024. A Gene-Switch Platform Interfacing with Reactive Oxygen Species Enables Transcription Fine-Tuning by Soluble and Volatile Pharmacologics and Food Additives. Adv. Sci. 11, 2306333. [CrossRef]
- Huang, K., Doyle, F., Wurz, Z.E., Tenenbaum, S.A., Hammond, R.K., Caplan, J.L., Meyers, B.C., 2017. FASTmiR: an RNA-based sensor for in vitro quantification and live-cell localization of small RNAs. Nucleic Acids Res. 45, e130–e130. [CrossRef]
- Ino, K., Nashimoto, Y., Taira, N., Azcon, J.R., Shiku, H., 2018. Intracellular Electrochemical Sensing. Electroanalysis 30, 2195–2209. [CrossRef]
- Islam, M.N., Masud, M.K., Haque, M.H., Hossain, M.S. Al, Yamauchi, Y., Nguyen, N.T., Shiddiky, M.J.A., 2017. RNA Biomarkers: Diagnostic and Prognostic Potentials and Recent Developments of Electrochemical Biosensors. Small Methods 1, 1700131. [CrossRef]
- Ji, D., Lyu, K., Zhao, H., Kwok, C.K., 2021. Circular L-RNA aptamer promotes target recognition and controls gene activity. Nucleic Acids Res. 49, 7280–7291. [CrossRef]
- Jiang, H.K., Ambrose, N.L., Chung, C.Z., Wang, Y.S., Söll, D., Tharp, J.M., 2023. Split aminoacyl-tRNA synthetases for proximity-induced stop codon suppression. Proc. Natl. Acad. Sci. U. S. A. 120, e2219758120. [CrossRef]
- Jones, K.A., Zinkus-Boltz, J., Dickinson, B.C., 2019. Recent advances in developing and applying biosensors for synthetic biology. Nano Futur. 3, 042002. [CrossRef]
- Juan, T., Molina, T., Xie, L., Papadopoulou, S., Cardoso, B., Jha, S.G., Stainier, D.Y.R., 2025. A recombinase-activated ribozyme to knock down endogenous gene expression in zebrafish. PLOS Genet. 21, e1011594. [CrossRef]
- Kang, D., Zuo, X., Yang, R., Xia, F., Plaxco, K.W., White, R.J., 2009. Comparing the properties of electrochemical-based DNA sensors employing different redox tags. Anal. Chem. 81, 9109–9113. [CrossRef]
- Kang, W.J., Cho, Y.L., Chae, J.R., Lee, J.D., Choi, K.J., Kim, S., 2011. Molecular beacon-based bioimaging of multiple microRNAs during myogenesis. Biomaterials 32, 1915–1922. [CrossRef]
- Keenan, S.E., Avdeeva, M., Yang, L., Alber, D.S., Wieschaus, E.F., Shvartsman, S.Y., 2022. Dynamics of Drosophila endoderm specification. Proc. Natl. Acad. Sci. U. S. A. 119, e2112892119. [CrossRef]
- Khodarev, N.N., 2019. Intracellular RNA Sensing in Mammalian Cells: Role in Stress Response and Cancer Therapies. Int. Rev. Cell Mol. Biol. 344, 31–89. [CrossRef]
- Kornienko, I. V., Aramova, O.Y., Tishchenko, A.A., Rudoy, D. V., Chikindas, M.L., 2024. RNA Stability: A Review of the Role of Structural Features and Environmental Conditions. Molecules 29, 5978. [CrossRef]
- Kuang, T., Chang, L., Peng, X., Hu, X., Gallego-Perez, D., 2017. Molecular Beacon Nano-Sensors for Probing Living Cancer Cells. Trends Biotechnol. 35, 347–359. [CrossRef]
- Kudłak, B., Wieczerzak, M., 2020. Aptamer based tools for environmental and therapeutic monitoring: A review of developments, applications, future perspectives. Crit. Rev. Environ. Sci. Technol. 50, 816–867. [CrossRef]
- Larkey, N.E., Phillips, J.L., Jang, H.S., Kolluri, S.K., Burrows, S.M., 2020. Small RNA Biosensor Design Strategy to Mitigate Off-Analyte Response. ACS Sensors 5, 377–384. [CrossRef]
- Lateef, O.M., Akintubosun, M.O., Olaoba, O.T., Samson, S.O., Adamczyk, M., 2022. Making Sense of “Nonsense” and More: Challenges and Opportunities in the Genetic Code Expansion, in the World of tRNA Modifications. Int. J. Mol. Sci. 2022, Vol. 23, Page 938 23, 938. [CrossRef]
- Lefebvre, F.A., Lécuyer, É., 2018. Flying the RNA Nest: Drosophila Reveals Novel Insights into the Transcriptome Dynamics of Early Development. J. Dev. Biol. 2018, Vol. 6, Page 5 6, 5. [CrossRef]
- Li, W., Xu, Y., Zhang, Y., Li, P., Zhu, X., Feng, C., 2023. Cell-Free Biosensing Genetic Circuit Coupled with Ribozyme Cleavage Reaction for Rapid and Sensitive Detection of Small Molecules. ACS Synth. Biol. 12, 1657–1666. [CrossRef]
- Litke, J.L., Jaffrey, S.R., 2023. Designing and Expressing Circular RNA Aptamers to Regulate Mammalian Cell Biology. Methods Mol. Biol. 2570, 223–234. [CrossRef]
- Litke, J.L., Jaffrey, S.R., 2019. Highly efficient expression of circular RNA aptamers in cells using autocatalytic transcripts. Nat. Biotechnol. 2019 376 37, 667–675. [CrossRef]
- Liu, M. hao, Zhao, N. ning, Yu, W. tong, Qiu, J.G., Jiang, B.H., Zhang, Y., Zhang, C.Y., 2024. Construction of a label-free fluorescent biosensor for homogeneous detection of m6A eraser FTO in breast cancer tissues. Talanta 272, 125784. [CrossRef]
- Liu, R., Zhong, Y., Chen, R., Chu, C., Liu, G., Zhou, Y., Huang, Y., Fang, Z., Liu, H., 2022. m6A reader hnRNPA2B1 drives multiple myeloma osteolytic bone disease. Theranostics 12, 7760–7774. [CrossRef]
- Liu, X., Wang, T., Wu, Y., Tan, Y., Jiang, T., Li, K., Lou, B., Chen, L., Liu, Y., Liu, Z., 2022. Aptamer based probes for living cell intracellular molecules detection. Biosens. Bioelectron. 208, 114231. [CrossRef]
- Liu, Y., Zhu, Z., Ho, I.H.T., Shi, Y., Li, J., Wang, X., Chan, M.T.V., Cheng, C.H.K., 2020. Genetic Deletion of miR-430 Disrupts Maternal-Zygotic Transition and Embryonic Body Plan. Front. Genet. 11, 568845. [CrossRef]
- Loffet, E.A., Durel, J.F., Nerurkar, N.L., 2023. Evo-Devo Mechanobiology: The Missing Link. Integr. Comp. Biol. 63, 1455–1473. [CrossRef]
- Lu, X., Kong, K.Y.S., Unrau, P.J., 2023. Harmonizing the growing fluorogenic RNA aptamer toolbox for RNA detection and imaging. Chem. Soc. Rev. 52, 4071–4098. [CrossRef]
- Manna, S., Kellenberger, C.A., Hallberg, Z.F., Hammond, M.C., 2021. Live Cell Imaging Using Riboswitch–Spinach tRNA Fusions as Metabolite-Sensing Fluorescent Biosensors. Methods Mol. Biol. 2323, 121–140. [CrossRef]
- Markey, F.B., Parashar, V., Batish, M., 2021. Methods for spatial and temporal imaging of the different steps involved in RNA processing at single-molecule resolution. Wiley Interdiscip. Rev. RNA 12. [CrossRef]
- Mau, C., Rudolf, H., Strobl, F., Schmid, B., Regensburger, T., Palmisano, R., Stelzer, E.H.K., Taher, L., El-Sherif, E., 2023. How enhancers regulate wavelike gene expression patterns. Elife 12. [CrossRef]
- McNerney, M.P., Zhang, Y., Steppe, P., Silverman, A.D., Jewett, M.C., Styczynski, M.P., 2019. Point-of-care biomarker quantification enabled by sample-specific calibration. Sci. Adv. 5. [CrossRef]
- Micura, R., Höbartner, C., 2020. Fundamental studies of functional nucleic acids: aptamers, riboswitches, ribozymes and DNAzymes. Chem. Soc. Rev. 49, 7331–7353. [CrossRef]
- Mikaeeli Kangarshahi, B., Naghib, S.M., Rabiee, N., 2024. DNA/RNA-based electrochemical nanobiosensors for early detection of cancers. Crit. Rev. Clin. Lab. Sci. 61, 473–495. [CrossRef]
- Moraldo, C., Vuille-dit-Bille, E., Shkodra, B., Kloter, T., Nakatsuka, N., 2022. Aptamer-modified biosensors to visualize neurotransmitter flux. J. Neurosci. Methods 365, 109386. [CrossRef]
- Moro, A., Driscoll, T.P., Boraas, L.C., Armero, W., Kasper, D.M., Baeyens, N., Jouy, C., Mallikarjun, V., Swift, J., Ahn, S.J., Lee, D., Zhang, J., Gu, M., Gerstein, M., Schwartz, M., Nicoli, S., 2019. MicroRNA-dependent regulation of biomechanical genes establishes tissue stiffness homeostasis. Nat. Cell Biol. 2019 213 21, 348–358. [CrossRef]
- Moros, M., Fergola, E., Marchesano, V., Mutarelli, M., Tommasini, G., Miedziak, B., Palumbo, G., Ambrosone, A., Tino, A., Tortiglione, C., 2021. The Aquatic Invertebrate Hydra vulgaris Releases Molecular Messages Through Extracellular Vesicles. Front. Cell Dev. Biol. 9, 788117. [CrossRef]
- Morris, K. V., Mattick, J.S., 2014. The rise of regulatory RNA. Nat. Rev. Genet. 2014 156 15, 423–437. [CrossRef]
- Murata, Y., Jo, J.I., Tabata, Y., 2021. Molecular Beacon Imaging to Visualize Ki67 mRNA for Cell Proliferation Ability. Tissue Eng. Part A 27, 526–535. [CrossRef]
- Murphy, M.P., Bayir, H., Belousov, V., Chang, C.J., Davies, K.J.A., Davies, M.J., Dick, T.P., Finkel, T., Forman, H.J., Janssen-Heininger, Y., Gems, D., Kagan, V.E., Kalyanaraman, B., Larsson, N.G., Milne, G.L., Nyström, T., Poulsen, H.E., Radi, R., Van Remmen, H., Schumacker, P.T., Thornalley, P.J., Toyokuni, S., Winterbourn, C.C., Yin, H., Halliwell, B., 2022. Guidelines for measuring reactive oxygen species and oxidative damage in cells and in vivo. Nat. Metab. 2022 46 4, 651–662. [CrossRef]
- Myers, J.M., Sullivan, J.M., 2025. Enhanced hammerhead ribozyme turnover rates: Reevaluating therapeutic space for small catalytic RNAs. Mol. Ther. Nucleic Acids 36, 102431. [CrossRef]
- Napoletano, S., Battista, E., Netti, P.A., Causa, F., 2024. MicroLOCK: Highly stable microgel biosensor using locked nucleic acids as bioreceptors for sensitive and selective detection of let-7a. Biosens. Bioelectron. 260, 116406. [CrossRef]
- Narberhaus, F., Waldminghaus, T., Chowdhury, S., 2006. RNA thermometers. FEMS Microbiol. Rev. 30, 3–16. [CrossRef]
- Nesterova, I. V., Erdem, S.S., Pakhomov, S., Hammer, R.P., Soper, S.A., 2009. Phthalocyanine dimerization-based molecular beacons using near-IR fluorescence. J. Am. Chem. Soc. 131, 2432–2433. [CrossRef]
- Neubacher, S., Hennig, S., 2019. RNA Structure and Cellular Applications of Fluorescent Light-Up Aptamers. Angew. Chemie Int. Ed. 58, 1266–1279. [CrossRef]
- Nyberg, K.G., Navales, F.G., Keles, E., Nguyen, J.Q., Hertz, L.M., Carthew, R.W., 2024. Robust and heritable knockdown of gene expression using a self-cleaving ribozyme in Drosophila. Genetics 227. [CrossRef]
- Osman, E.A., Rynes, T.P., Wang, Y.L., Mruk, K., McKeague, M., McKeague, M., 2024. Non-invasive single cell aptasensing in live cells and animals. Chem. Sci. 15. [CrossRef]
- Palchetti, I., Mascini, M., 2008. Nucleic acid biosensors for environmental pollution monitoring. Analyst 133, 846–854. [CrossRef]
- Panahi, A., Ghafar-Zadeh, E., 2023. Emerging Field-Effect Transistor Biosensors for Life Science Applications. Bioeng. 2023, Vol. 10, Page 793 10, 793. [CrossRef]
- Pavani, K.C., Meese, T., Pascottini, O.B., Guan, X.F., Lin, X., Peelman, L., Hamacher, J., van Nieuwerburgh, F., Deforce, D., Boel, A., Heindryckx, B., Tilleman, K., van Soom, A., Gadella, B.M., Hendrix, A., Smits, K., 2022. Hatching is modulated by microRNA-378a-3p derived from extracellular vesicles secreted by blastocysts. Proc. Natl. Acad. Sci. U. S. A. 119, e2122708119. [CrossRef]
- Peng, Y., Shu, L., Deng, X., Huang, X., Mo, X., Du, F., Tang, Z., 2023. Live-Cell Imaging of Endogenous RNA with a Genetically Encoded Fluorogenic Allosteric Aptamer. Anal. Chem. 95, 13762–13768. [CrossRef]
- Pichon, X., Robert, M.C., Bertrand, E., Singer, R.H., Tutucci, E., 2020. New Generations of MS2 Variants and MCP Fusions to Detect Single mRNAs in Living Eukaryotic Cells. Methods Mol. Biol. 2166, 121–144. [CrossRef]
- Pitchiaya, S., Heinicke, L.A., Custer, T.C., Walter, N.G., 2014. Single molecule fluorescence approaches shed light on intracellular RNAs. Chem. Rev. 114, 3224–3265. [CrossRef]
- Qian, Y., Li, J., Zhao, S., Matthews, E.A., Adoff, M., Zhong, W., An, X., Yeo, M., Park, C., Yang, X., Wang, B.S., Southwell, D.G., Huang, Z.J., 2022. Programmable RNA sensing for cell monitoring and manipulation. Nat. 2022 6107933 610, 713–721. [CrossRef]
- Rosa, A., Ciaudo, C., Sumazin, P., Fazi, F., 2021. Editorial: The RNA Revolution in Embryonic Development and Cell Differentiation in Health and Disease. Front. Cell Dev. Biol. 9, 715341. [CrossRef]
- Rovira, E., Moreno, B., Razquin, N., Blázquez, L., Hernández-Alcoceba, R., Fortes, P., Pastor, F., 2023. Engineering U1-Based Tetracycline-Inducible Riboswitches to Control Gene Expression in Mammals. ACS Nano 17, 23331–23346. [CrossRef]
- Sadeghzade, S., Hooshiar, M.H., Akbari, H., Tajer, M.H.M., Sahneh, K.K., Ziaei, S.Y., Jalali, F., Akouchakian, E., 2024. Recent advances in Organ-on-a-Chip models: How precision engineering integrates cutting edge technologies in fabrication and characterization. Appl. Mater. Today 38, 102231. [CrossRef]
- Salinas, S., Jayasundara, N., 2022. Thermal plasticity in marine organisms. Mar. Biol. A Funct. Approach to Ocean. their Org. 327–348. [CrossRef]
- Santos-Cancel, M., Simpson, L.W., Leach, J.B., White, R.J., 2019. Direct, Real-Time Detection of Adenosine Triphosphate Release from Astrocytes in Three-Dimensional Culture Using an Integrated Electrochemical Aptamer-Based Sensor. ACS Chem. Neurosci. 10, 2070–2079. [CrossRef]
- Sarfraz, N., Lee, H.J., Rice, M.K., Moscoso, E., Shafik, L.K., Glasgow, E., Ranjit, S., Lambeck, B.J., Braselmann, E., 2023. Establishing Riboglow-FLIM to visualize noncoding RNAs inside live zebrafish embryos. Biophys. Reports 3. [CrossRef]
- Saw, P.E., Song, E., 2025. RNA-Based Imaging System. RNA Ther. Hum. Dis. 459–488. [CrossRef]
- Schmidt, C.M., Smolke, C.D., 2021. A convolutional neural network for the prediction and forward design of ribozyme-based gene-control elements. Elife 10. [CrossRef]
- Sharma, P., Mondal, K., Kumar, S., Tamang, S., Najar, I.N., Das, S., Thakur, N., 2022. RNA thermometers in bacteria: Role in thermoregulation. Biochim. Biophys. Acta - Gene Regul. Mech. 1865, 194871. [CrossRef]
- Sheng, J., Gong, J., Shi, Y., Wang, X., Liu, D., 2022. MicroRNA-22 coordinates vascular and motor neuronal pathfinding via sema4 during zebrafish development. Open Biol. 12. [CrossRef]
- Sherwood, A. V., Henkin, T.M., 2016. Riboswitch-Mediated Gene Regulation: Novel RNA Architectures Dictate Gene Expression Responses. Annu. Rev. Microbiol. 70, 361–374. [CrossRef]
- Shui, B., Ozer, A., Zipfel, W., Sahu, N., Singh, A., Lis, J.T., Shi, H., Kotlikoff, M.I., 2012. RNA aptamers that functionally interact with green fluorescent protein and its derivatives. Nucleic Acids Res. 40. [CrossRef]
- Somero, G.N., 2018. RNA thermosensors: How might animals exploit their regulatory potential? J. Exp. Biol. 221. [CrossRef]
- Song, Y., Xu, Z., Wang, F., 2020. Genetically Encoded Reporter Genes for MicroRNA Imaging in Living Cells and Animals. Mol. Ther. Nucleic Acids 21, 555–567. [CrossRef]
- Srinivas, T., Siqueira, E., Guil, S., 2023. Techniques for investigating lncRNA transcript functions in neurodevelopment. Mol. Psychiatry 2023 294 29, 874–890. [CrossRef]
- Su, Y., Hammond, M.C., 2020. RNA-based fluorescent biosensors for live cell imaging of small molecules and RNAs. Curr. Opin. Biotechnol. 63, 157–166. [CrossRef]
- Sun, X., Li, Q., Wang, Y., Zhou, W., Guo, Y., Chen, J., Zheng, P., Sun, J., Ma, Y., 2020. Isoleucyl-tRNA synthetase mutant based whole-cell biosensor for high-throughput selection of isoleucine overproducers. Biosens. Bioelectron. 172, 112783–112783. [CrossRef]
- Sunbul, M., Lackner, J., Martin, A., Englert, D., Hacene, B., Grün, F., Nienhaus, K., Nienhaus, G.U., Jäschke, A., 2021. Super-resolution RNA imaging using a rhodamine-binding aptamer with fast exchange kinetics. Nat. Biotechnol. 2021 396 39, 686–690. [CrossRef]
- Sung, D., Koo, J., 2021. A review of BioFET’s basic principles and materials for biomedical applications. Biomed. Eng. Lett. 11, 85–96. [CrossRef]
- Terzi, A., Alam, S.M.S., Suter, D.M., 2021. ROS Live Cell Imaging During Neuronal Development. J. Vis. Exp. 2021, 10.3791/62165. [CrossRef]
- Thomas, S.E., Balcerowicz, M., Chung, B.Y.W., 2022. RNA structure mediated thermoregulation: What can we learn from plants? Front. Plant Sci. 13, 938570. [CrossRef]
- Tian, M., Xu, S., Zhang, J., Wang, X., Li, Z., Liu, H., Song, R., Yu, Z., Wang, J., 2018. RNA Detection Based on Graphene Field-Effect Transistor Biosensor. Adv. Condens. Matter Phys. 2018, 8146765. [CrossRef]
- Tomescu, A.I., Robb, N.C., Hengrung, N., Fodor, E., Kapanidis, A.N., 2014. Single-molecule FRET reveals a corkscrew RNA structure for the polymerase-bound influenza virus promoter. Proc. Natl. Acad. Sci. U. S. A. 111, E3335–E3342. [CrossRef]
- Trachman, R.J., Cojocaru, R., Wu, D., Piszczek, G., Ryckelynck, M., Unrau, P.J., Ferré-D’Amaré, A.R., 2020. Structure-Guided Engineering of the Homodimeric Mango-IV Fluorescence Turn-on Aptamer Yields an RNA FRET Pair. Structure 28, 776-785.e3. [CrossRef]
- Tutucci, E., Vera, M., Biswas, J., Garcia, J., Parker, R., Singer, R.H., 2017. An improved MS2 system for accurate reporting of the mRNA life cycle. Nat. Methods 2017 151 15, 81–89. [CrossRef]
- Verma, A.K., Noumani, A., Yadav, A.K., Solanki, P.R., 2023. FRET Based Biosensor: Principle Applications Recent Advances and Challenges. Diagnostics (Basel, Switzerland) 13. [CrossRef]
- Vong, Y.H., Sivashanmugam, L., Leech, R., Zaucker, A., Jones, A., Sampath, K., 2021. The RNA-binding protein Igf2bp3 is critical for embryonic and germline development in zebrafish. PLOS Genet. 17, e1009667. [CrossRef]
- Wang, J., Cui, C., Nan, H., Yu, Y., Xiao, Y., Poon, E., Yang, G., Wang, X., Wang, C., Li, L., Boheler, K.R., Ma, X., Cheng, X., Ni, Z., Chen, M., 2017. Graphene Sheet-Induced Global Maturation of Cardiomyocytes Derived from Human Induced Pluripotent Stem Cells. ACS Appl. Mater. Interfaces 9, 25929–25940. [CrossRef]
- Wang, L., Hast, K., Aggarwal, T., Baci, M., Hong, J., Izgu, E.C., 2022. MicroRNA detection in biologically relevant media using a split aptamer platform. Bioorg. Med. Chem. 69, 116909. [CrossRef]
- Wang, L., Xu, W., Zhang, S., Gundberg, G.C., Zheng, C.R., Wan, Z., Mustafina, K., Caliendo, F., Sandt, H., Kamm, R., Weiss, R., 2024. Sensing and guiding cell-state transitions by using genetically encoded endoribonuclease-mediated microRNA sensors. Nat. Biomed. Eng. 2024 812 8, 1730–1743. [CrossRef]
- Wang, S., Emery, N.J., Liu, A.P., 2019. A Novel Synthetic Toehold Switch for MicroRNA Detection in Mammalian Cells. ACS Synth. Biol. 8, 1079–1088. [CrossRef]
- Wang, X.W., Hu, L.F., Hao, J., Liao, L.Q., Chiu, Y.T., Shi, M., Wang, Y., 2019. A microRNA-inducible CRISPR–Cas9 platform serves as a microRNA sensor and cell-type-specific genome regulation tool. Nat. Cell Biol. 2019 214 21, 522–530. [CrossRef]
- Wang, Y., Li, Z., Weber, T.J., Hu, D., Lin, C.T., Li, J., Lin, Y., 2013. In situ live cell sensing of multiple nucleotides exploiting DNA/RNA aptamers and graphene oxide nanosheets. Anal. Chem. 85, 6775–6782. [CrossRef]
- Wang, Z., Sun, X., Zhang, X., Dong, B., Yu, H., 2021. Development of a miRNA Sensor by an Inducible CRISPR-Cas9 Construct in Ciona Embryogenesis. Mol. Biotechnol. 63, 613–620. [CrossRef]
- Wani, A.K., Akhtar, N., Mir, T. ul G., Chopra, C., Singh, R., Hong, J.C., Kadam, U.S., 2024. CRISPR/Cas12a-based biosensors for environmental monitoring and diagnostics. Environ. Technol. Innov. 34, 103625. [CrossRef]
- Wei, J., Zhang, W., Jiang, A., Peng, H., Zhang, Q., Li, Y., Bi, J., Wang, L., Liu, P., Wang, J., Ge, Y., Zhang, L., Yu, H., Li, L., Wang, S., Leng, L., Chen, K., Dong, B., 2024. Temporospatial hierarchy and allele-specific expression of zygotic genome activation revealed by distant interspecific urochordate hybrids. Nat. Commun. 2024 151 15, 1–11. [CrossRef]
- Weldemichael, T., Asemoloye, M.D., Marchisio, M.A., 2022. Feedforward Loops: Evolutionary Conserved Network Motifs Redesigned for Synthetic Biology Applications. Appl. Sci. 2022, Vol. 12, Page 8292 12, 8292. [CrossRef]
- Wile, B.M., Ban, K., Yoon, Y.S., Bao, G., 2014. Molecular beacon–enabled purification of living cells by targeting cell type–specific mRNAs. Nat. Protoc. 2014 910 9, 2411–2424. [CrossRef]
- Winkler, W.C., Nahvi, A., Roth, A., Collins, J.A., Breaker, R.R., 2004. Control of gene expression by a natural metabolite-responsive ribozyme. Nat. 2004 4286980 428, 281–286. [CrossRef]
- Wójcik, A.M., 2020. Research Tools for the Functional Genomics of Plant miRNAs During Zygotic and Somatic Embryogenesis. Int. J. Mol. Sci. 2020, Vol. 21, Page 4969 21, 4969. [CrossRef]
- Wong, C., Jurczak, E.M., Correspondence, R.R., Roy, R., 2024. Neuronal exosomes transport an miRISC cargo to preserve stem cell integrity during energy stress. Cell Rep. 43, 114851. [CrossRef]
- Wu, Y., Zhu, L., Li, S., Chu, H., Wang, X., Xu, W., 2023. High content design of riboswitch biosensors: All-around rational module-by-module design. Biosens. Bioelectron. 220, 114887. [CrossRef]
- Wu, Y.X., Kwon, Y.J., 2016. Aptamers: The “evolution” of SELEX. Methods 106, 21–28. [CrossRef]
- Xia, C., Colognori, D., Jiang, X.S., Xu, K., Doudna, J.A., 2025. Single-molecule live-cell RNA imaging with CRISPR–Csm. Nat. Biotechnol. 2025 1–8. [CrossRef]
- Xiang, J.S., Kaplan, M., Dykstra, P., Hinks, M., McKeague, M., Smolke, C.D., 2019. Massively parallel RNA device engineering in mammalian cells with RNA-Seq. Nat. Commun. 2019 101 10, 1–16. [CrossRef]
- Yang, L.Z., Wang, Y., Li, S.Q., Yao, R.W., Luan, P.F., Wu, H., Carmichael, G.G., Chen, L.L., 2019. Dynamic Imaging of RNA in Living Cells by CRISPR-Cas13 Systems. Mol. Cell 76, 981-997.e7. [CrossRef]
- Yao, Y., Yin, F., He, M., Cheng, W., Wang, Z., Xiang, Y., 2025. Multi-target recognition and DNAzyme signal amplification integrated functional nucleic acid framework for lncRNA in situ live cell imaging. Sensors Actuators B Chem. 422, 136599. [CrossRef]
- Yarra, S.S., Ashok, G., Mohan, U., 2023. “Toehold Switches; a foothold for Synthetic Biology.” Biotechnol. Bioeng. 120, 932–952. [CrossRef]
- Yin, S., Chen, A., Ding, Y., Song, J., Chen, R., Zhang, P., Yang, C., 2023. Recent advances in exosomal RNAs analysis towards diagnostic and therapeutic applications. TrAC Trends Anal. Chem. 158, 116840. [CrossRef]
- Yoo, H., Jo, H., Oh, S.S., 2020. Detection and beyond: challenges and advances in aptamer-based biosensors. Mater. Adv. 1, 2663–2687. [CrossRef]
- Yu, S., Lei, X., Qu, C., 2024. MicroRNA Sensors Based on CRISPR/Cas12a Technologies: Evolution From Indirect to Direct Detection. Crit. Rev. Anal. Chem. [CrossRef]
- Zhang, C., Miao, P., Sun, M., Yan, M., Liu, H., 2019. Progress in miRNA Detection Using Graphene Material–Based Biosensors. Small 15, 1901867. [CrossRef]
- Zhang, L., Xie, X., Djokovic, N., Nikolic, K., Kosenkov, D., Abendroth, F., Vázquez, O., 2023. Reversible Control of RNA Splicing by Photoswitchable Small Molecules. J. Am. Chem. Soc. 145, 12783–12792. [CrossRef]
- Zhang, Q., Ma, D., Wu, F., Standage-Beier, K., Chen, X., Wu, K., Green, A.A., Wang, X., 2021. Predictable control of RNA lifetime using engineered degradation-tuning RNAs. Nat. Chem. Biol. 2021 177 17, 828–836. [CrossRef]
- Zhang, Q., Su, C., Tian, X., Zhang, C.Y., 2023a. Corn-Based Fluorescent Light-Up Biosensors with Improved Signal-to-Background Ratio for Label-Free Detection of Long Noncoding RNAs. Anal. Chem. 95, 8097–8104. [CrossRef]
- Zhang, Q., Zhao, S., Su, C., Han, Q., Han, Y., Tian, X., Li, Y., Zhang, C.Y., 2023b. Construction of a Quantum-Dot-Based FRET Nanosensor through Direct Encoding of Streptavidin-Binding RNA Aptamers for N6-Methyladenosine Demethylase Detection. Anal. Chem. 95, 13201–13210. [CrossRef]
- Zhang, Z., Hu, J.J., Lin, S., Wu, J., Xia, F., Lou, X., 2024. Field effect transistor biosensors for healthcare monitoring. Interdiscip. Med. 2, e20240032. [CrossRef]
- Zhao, E.M., Mao, A.S., de Puig, H., Zhang, K., Tippens, N.D., Tan, X., Ran, F.A., Han, I., Nguyen, P.Q., Chory, E.J., Hua, T.Y., Ramesh, P., Thompson, D.B., Oh, C.Y., Zigon, E.S., English, M.A., Collins, J.J., 2021. RNA-responsive elements for eukaryotic translational control. Nat. Biotechnol. 2021 404 40, 539–545. [CrossRef]
- Zhao, N.N., Li, F.Z., Zhang, X., Liu, M., Cao, H., Zhang, C.Y., 2023. Construction of Genetically Encoded Light-Up RNA Aptamers for Label-free and Ultrasensitive Detection of CircRNAs in Cancer Cells and Tissues. Anal. Chem. 95, 8728–8734. [CrossRef]
- Zheng, L., Li, J., Li, Y., Sun, W., Ma, L. Le, Qu, F., Tan, W., 2024. Empowering Exosomes with Aptamers for Precision Theranostics. Small Methods 2400551. [CrossRef]
- Zheng, R., Xue, Z., You, M., 2024. Optogenetic Tools for Regulating RNA Metabolism and Functions. ChemBioChem 25, e202400615. [CrossRef]
- Zhong, W., Sczepanski, J.T., 2019. A Mirror Image Fluorogenic Aptamer Sensor for Live-Cell Imaging of MicroRNAs. ACS Sensors 4, 566–570. [CrossRef]
- Zhou, H., Zhang, S., 2022. Recent Development of Fluorescent Light-Up RNA Aptamers. Crit. Rev. Anal. Chem. 52, 1644–1661. [CrossRef]
- Zhou, Xiaolu, Gao, H., Xiang, L., Liu, H., Zhou, Xiang, 2025. Developments of the mDZ-RhoBAST Probe for Super-Resolution Imaging of FTO Demethylation in Live Cells. J. Am. Chem. Soc. [CrossRef]
- Zhou, Zhixin, Han, B., Wang, Y., Lin, N., Zhou, Zhongqiu, Zhang, Yuan, Bai, Y., Shen, L., Shen, Y., Zhang, Yuanjian, Yao, H., 2024. Fast and sensitive multivalent spatial pattern-recognition for circular RNA detection. Nat. Commun. 2024 151 15, 1–15. [CrossRef]
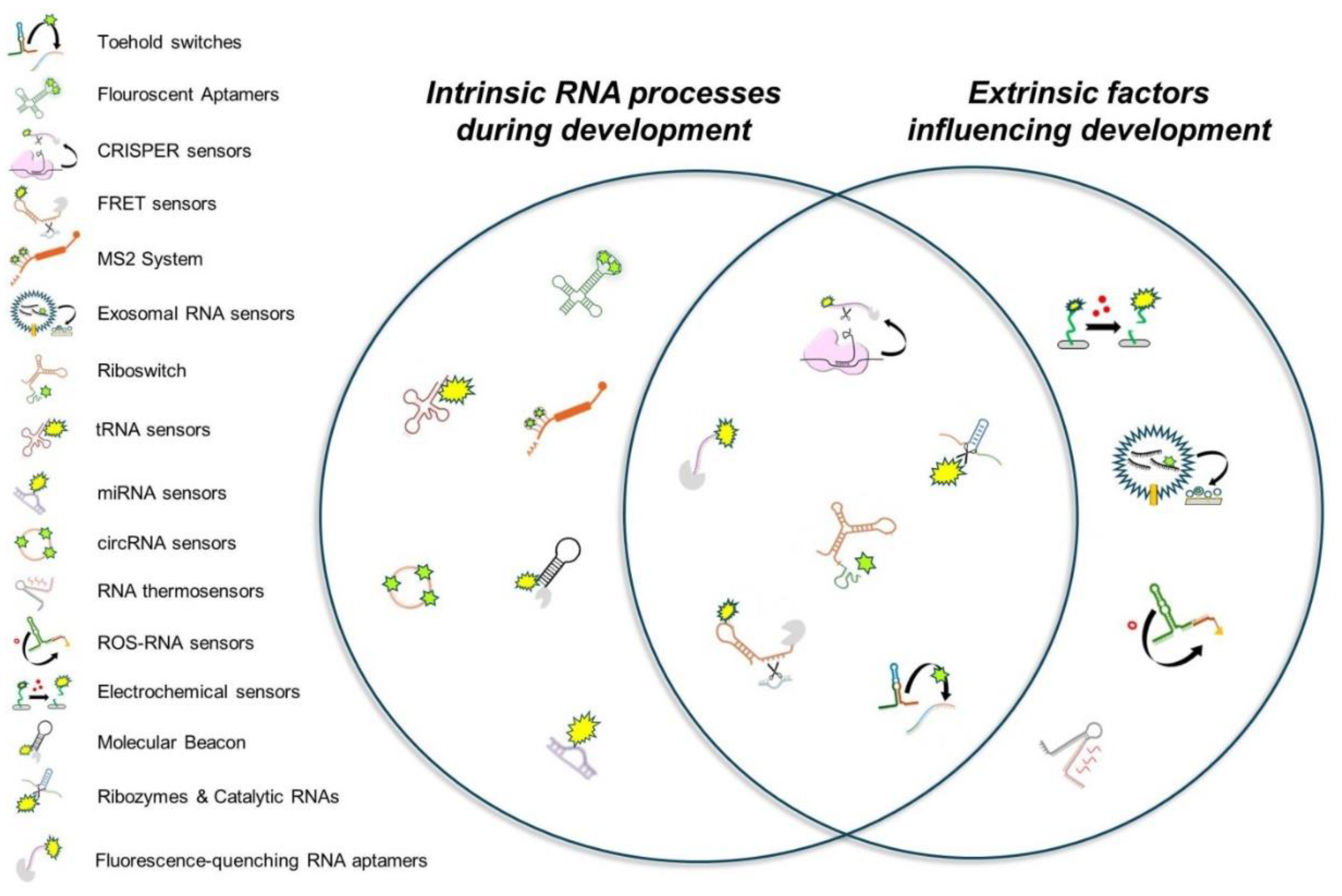
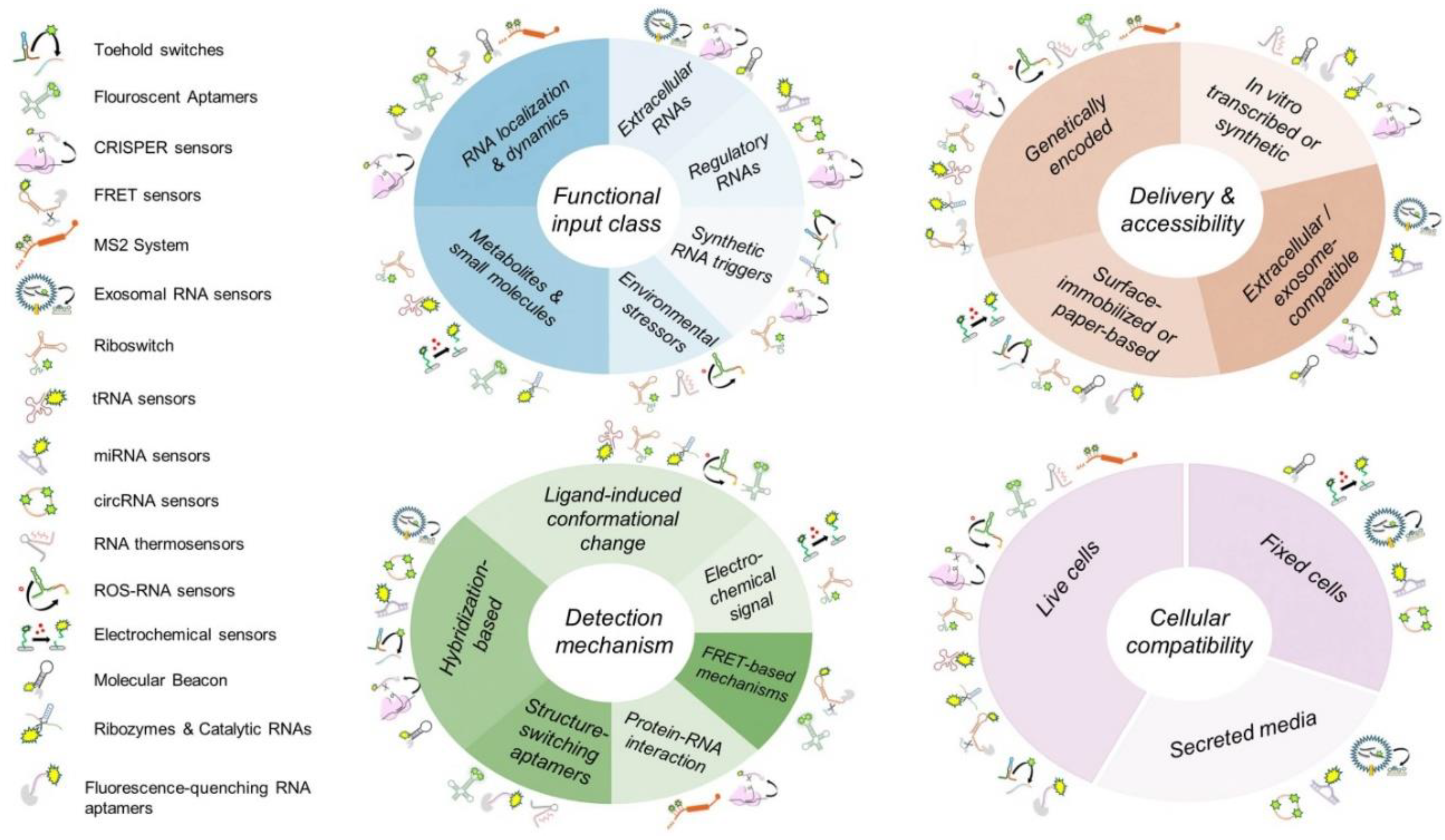
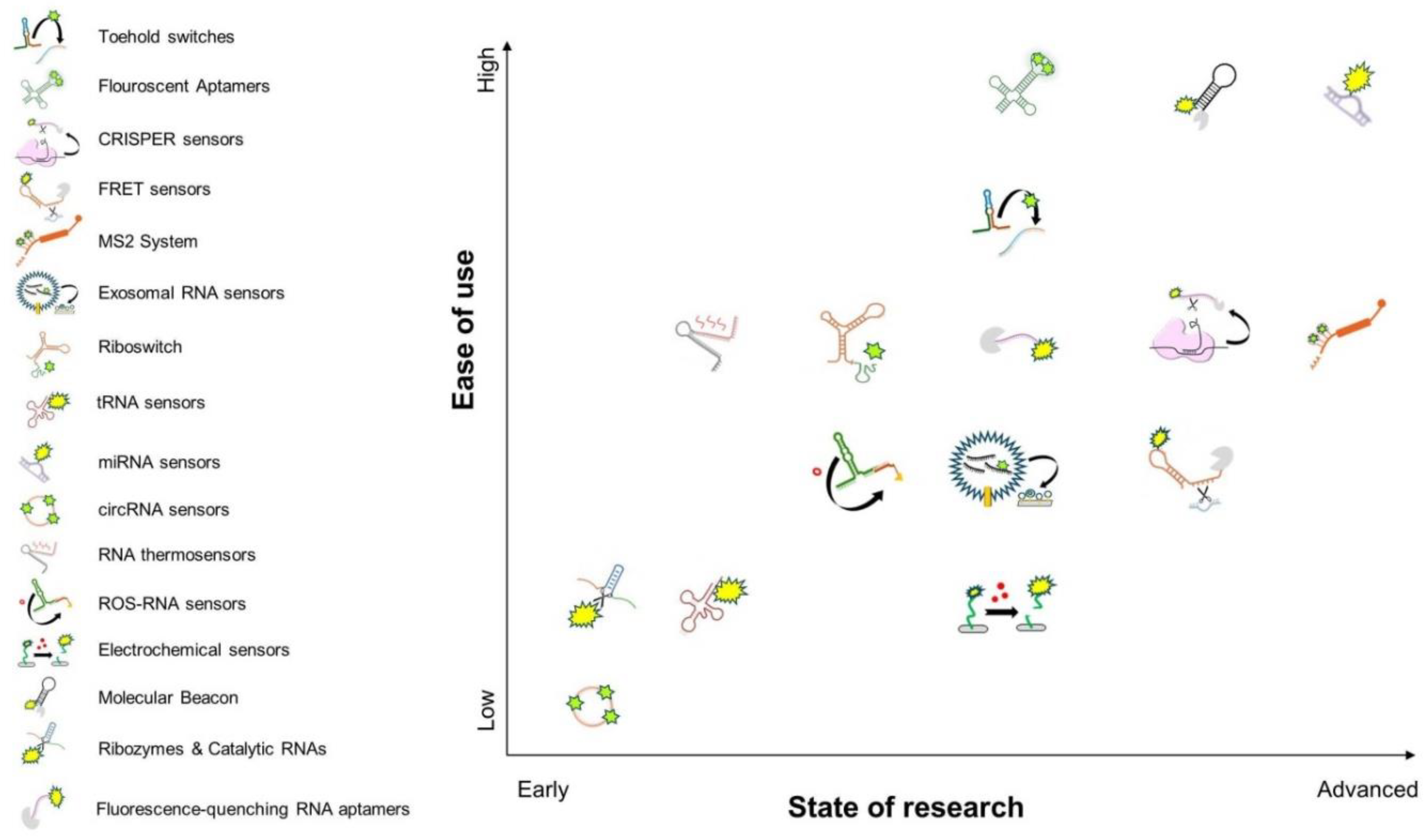
| RNA-Based Biosensors | Function | Most Common Usage | Potential Developmental Applications |
|---|---|---|---|
| Spinach, Broccoli, Mango, and Pepper aptamers | Fluoresce upon binding small molecules for RNA visualization | Live-cell RNA imaging | Real-time tracking of developmental RNAs |
| CRISPR-Cas13/Cas12a RNA sensors | Detect RNA sequences via guide RNAs and produce signals | Diagnostics for RNA biomarkers | Detecting transient RNAs in gene networks (e.g., mapping m6A during development) |
| Riboswitch biosensors | Bind small molecules and regulate gene expression structurally | Metabolite sensing and gene regulation | Studying conserved metabolic pathways during development |
| Toehold switches | Regulate translation by hairpin unwinding upon RNA triggers | Pathogen detection and synthetic biology | Monitoring developmental stage-specific RNA triggers |
| Molecular Beacon biosensors | Fluoresce when hybridizing with specific RNA sequences | RNA quantification in diagnostics | Visualizing developmental RNA patterns |
| MS2 system | Tag RNA for visualization and tracking with phage coat proteins | Studying RNA localization and stability | Tracking RNA transport in cell differentiation |
| miRNA sensors | Hybridize with microRNAs to produce measurable signals | Cancer diagnostics and monitoring biomarkers | Identifying roles of miRNAs in development |
| RNA thermometers | Change conformation with temperature to regulate translation | Studying bacterial thermal stress responses | Exploring species-specific thermal effects on development |
| Fluorescence-quenching RNA aptamers | Quench or enhance fluorescence based on environmental stimuli | Environmental monitoring and metabolite tracking | Examining metabolite-driven developmental changes |
| FRET-based RNA sensors | Detect RNA conformational changes using FRET | Visualizing RNA folding and interactions (e.g. real-time detection of m6A effects on RNA structure) | Tracking RNA structural changes during development (e.g., m6A modifications effects on RNA folding) |
| Catalytic RNA biosensors (ribozymes) | Catalyze reactions upon ligand binding for signal generation | Detecting small molecules in RNA studies | Detecting conserved signaling molecules in development |
| Exosomal RNA sensors | Hybridize with RNA in exosomes for fluorescence or signals | Cancer diagnostics and cell communication studies | Studying RNA communication in development |
| Split RNA aptamers and ribozymes | Activate fluorescence or catalysis when RNA fragments join | Studying splicing and RNA-protein interactions | Detecting splicing events at developmental level |
| RNA sensors for reactive oxygen species (ROS) | Detect reactive oxygen species and fluoresce in response | Researching oxidative stress in diseases | Monitoring stress in regeneration or embryogenesis |
| Electrochemical RNA sensors | Produce electrical signals upon RNA binding at electrodes | Point-of-care diagnostics for RNA targets | Detecting RNA biomarkers in development |
| Circular RNA sensors | Resist degradation while binding targets to emit signals | Long-term RNA expression monitoring | Tracking stable RNAs in developmental contexts |
| tRNA-based biosensors | Detect metabolites or amino acids via modified tRNA | Exploring translation and metabolic regulation | Studying amino acid-driven translation shifts |
| RNA-Based Biosensors | Delivery Challenge | Stability Issue | Signal Limitations | Tissue/Imaging Constraints | Target Specificity Concern |
|---|---|---|---|---|---|
| Fluorescence-quenching RNA aptamers | Synthetic RNA delivery required | Moderate; prone to degradation in vivo | Environmental sensitivity can cause false signals | Auto-fluorescence interference in deep tissues | Limited tuning for new targets |
| Ribozymes & Catalytic RNAs | Often require stable expression or microinjection | Variable, depends on structure and sequence | Can produce background activity | Requires amplification or reporter coupling | Context-dependent activation |
| Molecular Beacon | Challenging to deliver into intact embryos | Sensitive to nucleases | High background if improperly designed | Difficult deep-tissue detection | High for known targets, limited for novel RNAs |
| Electrochemical sensors | Difficult in vivo; mostly external use | Good extracellularly; intracellular stability is low | Requires external instrumentation | Limited spatial resolution | High when using validated probes |
| ROS-RNA sensors | Synthetic delivery or transfection | Rapid degradation under stress conditions | ROS fluctuation may confound signal | Unspecific spatial resolution | Low; generally detects ROS rather than RNA |
| RNA thermosensors | Genetically encoded or synthetic | Good if encoded; moderate if synthetic | Temperature range may limit precision | Affected by tissue heat gradients | Low; responds to temperature not sequence |
| circRNA sensors | Requires probe design or expression vectors | Stable once formed | Indirect; depends on backsplicing detection | Tissue-specific expression complicates interpretation | High if designed properly |
| miRNA sensors | Requires careful delivery (e.g., nanoparticles) | miRNA-probe interactions stable | Signal influenced by miRNA abundance | Tissue heterogeneity affects readout | High with optimized probes |
| tRNA sensors | Requires intracellular delivery | Moderate; depends on structural mimicry | Signal can be influenced by amino acid levels | Requires normalization in complex tissues | Moderate; depends on reporter design |
| Riboswitch | Needs expression in host cells | Good when chromosomally integrated | Ligand availability may limit output | May not work uniformly across tissues | High if ligand is specific |
| Exosomal RNA sensors | Requires vesicle isolation or extracellular sampling | Stable in extracellular fluids | Dependent on sample prep and purity | No spatial resolution in tissue | High for known exosomal RNAs |
| MS2 System | Genetic tagging required | Stable in vivo when fused with coat protein | Signal dilution during division | Limited in deep tissues unless optimized | High for tagged transcripts |
| FRET sensors | Genetic fusion or co-expression required | Susceptible to photobleaching | Sensitive to microenvironment changes | Shallow imaging unless NIR optimized | High for structured RNAs |
| CRISPR sensors | Requires Cas protein and guide delivery | Good if properly designed | Collateral activity may cause noise | Depends on delivery method and tissue | High; programmable specificity |
| Fluorescent Aptamers | Requires fluorophore and RNA co-expression | Can be stabilized via aptamer design | Background fluorescence possible | Autofluorescence in thick tissues | High when sequence-targeted |
| Toehold switches | Needs transcription in cells | Stable when encoded | Minimal background with rational design | Hard to tune expression across tissues | High; modular and programmable |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




