Submitted:
24 June 2025
Posted:
25 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. History of Bisphosphonates
1.2. Types of BPs
1.3. Side Effects of N-BPs
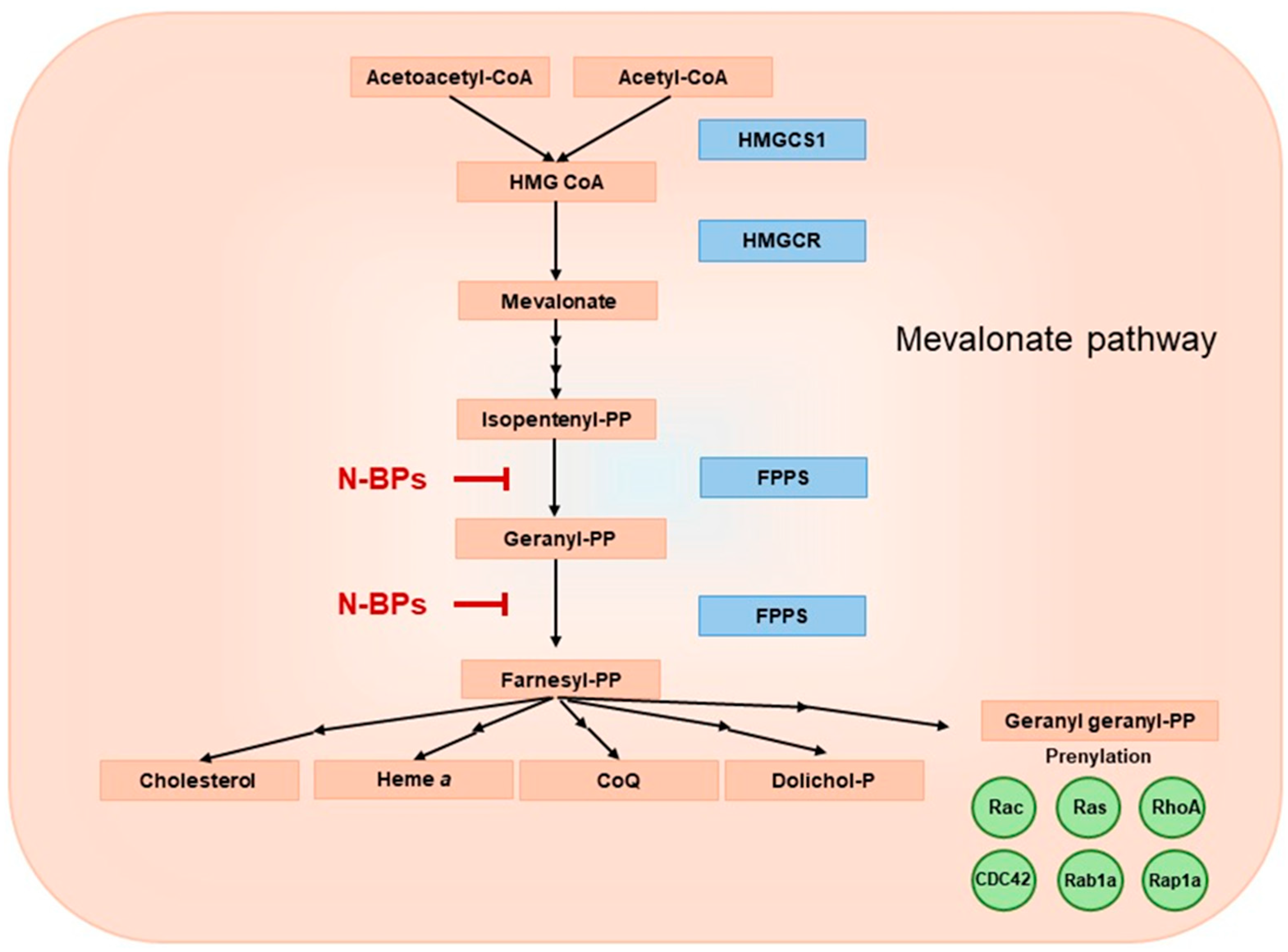
2. Mevalonate Pathway – A Biological Role and Products
2.1. Cholesterol
2.2. Dolichol
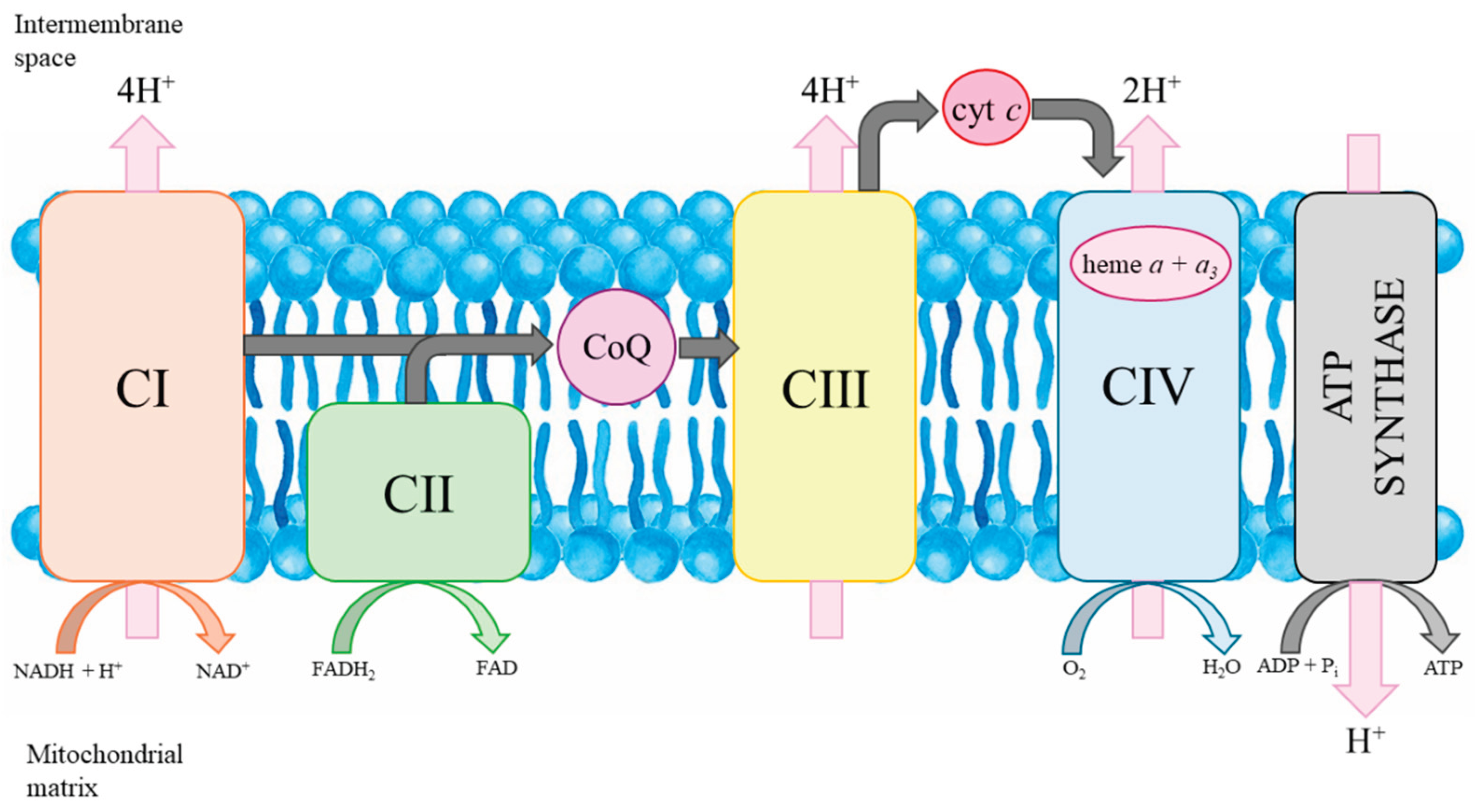
2.3. CoQ
2.4. Heme a
2.5. Protein Prenylation
3. Inhibition of the Mevalonate Pathway by N-BPs
3.1. Effects on the Cellular Level
3.1.1. Effects on Bone Cells
3.1.2. N-BP Effects on off-Target Cells
Anti-Cancer Effects, Cytoskeleton Alterations
Effects Related to Nephrotoxicity
Effects on Endothelial Cell Viability, Inflammation and Apoptosis
Effects on Endothelial Cell CoQ Homeostasis and Energy Metabolism
Effects on Lipid Metabolism
Effects on Authophagy, Mitochondrial Dynamics and Turnover
Effects on Calcium Homeostasis
3.2. N-BP Effects on the Mitochondrial Level
Effects on Mitochondria-Induced Apoptosis
Effects on Mitochondrial Calcium Homeostasis
N-BP-Induced Mitochondrial Heme a Decrease
N-BP-Induced Changes in Mitochondrial Respiratory Function
N-BP-Induced mtCoQ Deficiency
4. Conclusions and Future Perspectives
Author Contributions
Funding
References
- Cooper, C.; Campion, G.; Melton, L.J. 3rd Hip fractures in the elderly: a world-wide projection. Osteoporos. Int. a J. Establ. as result Coop. between Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 1992, 2, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Tu, K.N.; Lie, J.D.; Wan, C.K.V.; Cameron, M.; Austel, A.G.; Nguyen, J.K.; Van, K.; Hyun, D. Osteoporosis: A Review of Treatment Options. P T 2018, 43, 92–104. [Google Scholar]
- Barbosa, J.S.; Braga, S.S.; Almeida Paz, F.A. Empowering the Medicinal Applications of Bisphosphonates by Unveiling their Synthesis Details. Molecules 2020, 25. [Google Scholar] [CrossRef]
- Russell, R.G.G. Bisphosphonates: The first 40years. Bone 2011, 49, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Fleisch, H.; Russell, R.G.; Francis, M.D. Diphosphonates inhibit hydroxyapatite dissolution in vitro and bone resorption in tissue culture and in vivo. Science 1969, 165, 1262–1264. [Google Scholar] [CrossRef]
- Francis, M.D.; Russell, R.G.; Fleisch, H. Diphosphonates inhibit formation of calcium phosphate crystals in vitro and pathological calcification in vivo. Science 1969, 165, 1264–1266. [Google Scholar] [CrossRef]
- Russell, R.G.; Croucher, P.I.; Rogers, M.J. Bisphosphonates: pharmacology, mechanisms of action and clinical uses. Osteoporos. Int. a J. Establ. as result Coop. between Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 1999, 9 Suppl 2, S66–80. [Google Scholar] [CrossRef]
- Cremers, S.; Drake, M.T.; Ebetino, F.H.; Bilezikian, J.P.; Russell, R.G.G. Pharmacology of bisphosphonates. Br. J. Clin. Pharmacol. 2019, 85, 1052–1062. [Google Scholar] [CrossRef]
- Russell, G.R. Bisphosphonates: mode of action and pharmacology. Pediatrics 2007, 119, 150–162. [Google Scholar] [CrossRef]
- Frith, J.C.; Mönkkönen, J.; Blackburn, G.M.; Russell, R.G.; Rogers, M.J. Clodronate and liposome-encapsulated clodronate are metabolized to a toxic ATP analog, adenosine 5’-(beta, gamma-dichloromethylene) triphosphate, by mammalian cells in vitro. J. bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1997, 12, 1358–1367. [Google Scholar] [CrossRef] [PubMed]
- Mönkkönen, H.; Rogers, M.J.; Makkonen, N.; Niva, S.; Auriola, S.; Mönkkönen, J. The cellular uptake and metabolism of clodronate in RAW 264 macrophages. Pharm. Res. 2001, 18, 1550–1555. [Google Scholar] [CrossRef] [PubMed]
- Lehenkari, P.P.; Kellinsalmi, M.; Näpänkangas, J.P.; Ylitalo, K. V; Mönkkönen, J.; Rogers, M.J.; Azhayev, A.; Väänänen, H.K.; Hassinen, I.E. Further insight into mechanism of action of clodronate: inhibition of mitochondrial ADP/ATP translocase by a nonhydrolyzable, adenine-containing metabolite. Mol. Pharmacol. 2002, 61, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, K.L.; Guo, K.; Dunford, J.E.; Wu, X.; Knapp, S.; Ebetino, F.H.; Rogers, M.J.; Russell, R.G.G.; Oppermann, U. The molecular mechanism of nitrogen-containing bisphosphonates as antiosteoporosis drugs. Proc. Natl. Acad. Sci. 2006, 103, 7829–7834. [Google Scholar] [CrossRef]
- Giannasi, C.; Niada, S.; Farronato, D.; Lombardi, G.; Manfredi, B.; Farronato, G.; Brini, A.T. Nitrogen containing bisphosphonates impair the release of bone homeostasis mediators and matrix production by human primary pre-osteoblasts. Int. J. Med. Sci. 2019, 16, 23–32. [Google Scholar] [CrossRef]
- Diab, D.L.; Watts, N.B.; Miller, P.D. Chapter 80 - Bisphosphonates: Pharmacology and Use in the Treatment of Osteoporosis. In Osteoporosis (Fourth Edition); Marcus, R., Feldman, D., Dempster, D.W., Luckey, M., Cauley, J.A., Eds.; Academic Press: San Diego, 2013; ISBN 978-0-12-415853-5. [Google Scholar]
- Coleman, R.E. Risks and benefits of bisphosphonates. Br. J. Cancer 2008, 98, 1736–1740. [Google Scholar] [CrossRef]
- Jagpal, A.; Saag, K.G. How to use bisphosphonates safely and optimally. Rheumatology 2018, 57, 1875–1876. [Google Scholar] [CrossRef]
- Jha, S.; Wang, Z.; Laucis, N.; Bhattacharyya, T. Trends in Media Reports, Oral Bisphosphonate Prescriptions, and Hip Fractures 1996–2012: An Ecological Analysis. J. Bone Miner. Res. 2015, 30, 2179–2187. [Google Scholar] [CrossRef]
- market.us Global Osteoporosis Drug Market By Drug Class (Bisphosphonates, Parathyroid Hormone Therapy Drugs), By Route Administration (Oral and injectable), and By Distribution Channel (Retail Pharmacies, Hospital Pharmacies), By Region and Companies - Industry Seg; 2023;
- Kennel, K.A.; Drake, M.T. Adverse effects of bisphosphonates: implications for osteoporosis management. Mayo Clin. Proc. 2009, 84, 632–7, quiz 638. [Google Scholar] [CrossRef]
- Dunstan, C.R.; Felsenberg, D.; Seibel, M.J. Therapy insight: the risks and benefits of bisphosphonates for the treatment of tumor-induced bone disease. Nat. Clin. Pract. Oncol. 2007, 4, 42–55. [Google Scholar] [CrossRef]
- Kishimoto, H.; Noguchi, K.; Takaoka, K. Novel insight into the management of bisphosphonate-related osteonecrosis of the jaw (BRONJ). Jpn. Dent. Sci. Rev. 2019, 55, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Ziebart, T.; Koch, F.; Klein, M.O.; Guth, J.; Adler, J.; Pabst, A.; Al-Nawas, B.; Walter, C. Geranylgeraniol – A new potential therapeutic approach to bisphosphonate associated osteonecrosis of the jaw. Oral Oncol. 2011, 47, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.L.; Brown, M.S. Regulation of the mevalonate pathway. Nature 1990, 343, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Buhaescu, I.; Izzedine, H. Mevalonate pathway: a review of clinical and therapeutical implications. Clin. Biochem. 2007, 40, 575–584. [Google Scholar] [CrossRef]
- Hashemi, M.; Hoshyar, R.; Ande, S.R.; Chen, Q.M.; Solomon, C.; Zuse, A.; Naderi, M. Mevalonate Cascade and its Regulation in Cholesterol Metabolism in Different Tissues in Health and Disease. Curr. Mol. Pharmacol. 2017, 10, 13–26. [Google Scholar] [CrossRef]
- Lasunción, M.A.; Martínez-Botas, J.; Martín-Sánchez, C.; Busto, R.; Gómez-Coronado, D. Cell cycle dependence on the mevalonate pathway: Role of cholesterol and non-sterol isoprenoids. Biochem. Pharmacol. 2022, 196, 114623. [Google Scholar] [CrossRef]
- Shimano, H.; Sato, R. SREBP-regulated lipid metabolism: convergent physiology — divergent pathophysiology. Nat. Rev. Endocrinol. 2017, 13, 710–730. [Google Scholar] [CrossRef]
- Mullen, P.J.; Yu, R.; Longo, J.; Archer, M.C.; Penn, L.Z. The interplay between cell signalling and the mevalonate pathway in cancer. Nat. Rev. Cancer 2016, 16, 718–731. [Google Scholar] [CrossRef]
- Denecke, J.; Kranz, C. Hypoglycosylation due to dolichol metabolism defects. Biochim. Biophys. Acta - Mol. Basis Dis. 2009, 1792, 888–895. [Google Scholar] [CrossRef]
- Edlund, C.; Söderberg, M.; Kristensson, K. Isoprenoids in aging and neurodegeneration. Neurochem. Int. 1994, 25, 35–38. [Google Scholar] [CrossRef]
- Sood, B.; Patel, P.; Keenaghan, M. Coenzyme Q10. In; Treasure Island (FL), 2024.
- Pallotti, F.; Bergamini, C.; Lamperti, C.; Fato, R. The Roles of Coenzyme Q in Disease: Direct and Indirect Involvement in Cellular Functions. Int. J. Mol. Sci. 2021, 23. [Google Scholar] [CrossRef] [PubMed]
- de la Bella-Garzón, R.; Fernández-Portero, C.; Alarcón, D.; Amián, J.G.; López-Lluch, G. Levels of Plasma Coenzyme Q(10) Are Associated with Physical Capacity and Cardiovascular Risk in the Elderly. Antioxidants (Basel, Switzerland) 2022, 11. [Google Scholar] [CrossRef]
- Antonicka, H.; Mattman, A.; Carlson, C.G.; Glerum, D.M.; Hoffbuhr, K.C.; Leary, S.C.; Kennaway, N.G.; Shoubridge, E.A. Mutations in COX15 Produce a Defect in the Mitochondrial Heme Biosynthetic Pathway, Causing Early-Onset Fatal Hypertrophic Cardiomyopathy. Am. J. Hum. Genet. 2003, 72, 101–114. [Google Scholar] [CrossRef]
- Dwyer, B.E.; Stone, M.L.; Gorman, N.; Sinclair, P.R.; Perry, G.; Smith, M.A.; Zhu, X. Heme-a, the heme prosthetic group of cytochrome c oxidase, is increased in Alzheimer’s disease. Neurosci. Lett. 2009, 461, 302–305. [Google Scholar] [CrossRef]
- Palsuledesai, C.C.; Distefano, M.D. Protein prenylation: enzymes, therapeutics, and biotechnology applications. ACS Chem. Biol. 2015, 10, 51–62. [Google Scholar] [CrossRef]
- Walker, K.; Olson, M.F. Targeting Ras and Rho GTPases as opportunities for cancer therapeutics. Curr. Opin. Genet. Dev. 2005, 15, 62–68. [Google Scholar] [CrossRef]
- Vincenzi, B.; Santini, D.; Avvisati, G.; Baldi, A.; Cesa, A. La; Tonini, G. Statins may potentiate bisphosphonates anticancer properties: a new pharmacological approach? Med. Hypotheses 2003, 61, 98–101. [Google Scholar] [CrossRef]
- Rondeau, J.-M.; Bitsch, F.; Bourgier, E.; Geiser, M.; Hemmig, R.; Kroemer, M.; Lehmann, S.; Ramage, P.; Rieffel, S.; Strauss, A.; et al. Structural basis for the exceptional in vivo efficacy of bisphosphonate drugs. ChemMedChem 2006, 1, 267–273. [Google Scholar] [CrossRef]
- Russell, R.G.G. Determinants of structure–function relationships among bisphosphonates. Bone 2007, 40, S21–S25. [Google Scholar] [CrossRef]
- Dunford, J.E.; Thompson, K.; Coxon, F.P.; Luckman, S.P.; Hahn, F.M.; Poulter, C.D.; Ebetino, F.H.; Rogers, M.J. Structure-activity relationships for inhibition of farnesyl diphosphate synthase in vitro and inhibition of bone resorption in vivo by nitrogen-containing bisphosphonates. J. Pharmacol. Exp. Ther. 2001, 296, 235–242. [Google Scholar] [CrossRef]
- Xu, X.-L.; Gou, W.-L.; Wang, A.-Y.; Wang, Y.; Guo, Q.-Y.; Lu, Q.; Lu, S.-B.; Peng, J. Basic research and clinical applications of bisphosphonates in bone disease: what have we learned over the last 40 years? J. Transl. Med. 2013, 11, 303. [Google Scholar] [CrossRef] [PubMed]
- Holstein, S.A. A patent review of bisphosphonates in treating bone disease. Expert Opin. Ther. Pat. 2019, 29, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Shahi, A.K. Nitrogen containing bisphosphonates associated osteonecrosis of the jaws: A review for past 10 year literature. Dent. Res. J. (Isfahan). 2014, 11, 147–153. [Google Scholar] [PubMed]
- Russell, R.G.G.; Rogers, M.J. Bisphosphonates: from the laboratory to the clinic and back again. Bone 1999, 25, 97–106. [Google Scholar] [CrossRef]
- Wu, S.-N.; Huang, Y.-M.; Liao, Y.-K. Effects of Ibandronate Sodium, a Nitrogen-Containing Bisphosphonate, on Intermediate-Conductance Calcium-Activated Potassium Channels in Osteoclast Precursor Cells (RAW 264.7). J. Membr. Biol. 2015, 248, 103–115. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, Z.; Han, W.; Li, H. Zoledronate induces autophagic cell death in human umbilical vein endothelial cells via Beclin-1 dependent pathway activation. Mol. Med. Rep. 2016, 14, 4747–4754. [Google Scholar] [CrossRef]
- Dong, X.; He, Y.; An, J.; He, L.; Zheng, Y.; Wang, X.; Wang, J.; Chen, S.; Zhang, Y. Increased apoptosis of gingival epithelium is associated with impaired autophagic flux in medication-related osteonecrosis of the jaw. Autophagy 2023, 19, 2899–2911. [Google Scholar] [CrossRef]
- Budzinska, A.; Galganski, L.; Jarmuszkiewicz, W. The bisphosphonates alendronate and zoledronate induce adaptations of aerobic metabolism in permanent human endothelial cells. Sci. Rep. 2023, 13, 16205. [Google Scholar] [CrossRef]
- Clézardin, P.; Massaia, M. Nitrogen-containing bisphosphonates and cancer immunotherapy. Curr. Pharm. Des. 2010, 16, 2014–3007. [Google Scholar] [CrossRef]
- Virtanen, S.S.; Ishizu, T.; Sandholm, J.A.; Löyttyniemi, E.; Väänänen, H.K.; Tuomela, J.M.; Härkönen, P.L. Alendronate-induced disruption of actin cytoskeleton and inhibition of migration/invasion are associated with cofilin downregulation in PC-3 prostate cancer cells. Oncotarget 2018, 9, 32593–32608. [Google Scholar] [CrossRef]
- Lauterborn, J.C.; Gall, C.M. Chapter 14 - Defects in Rho GTPase Signaling to the Spine Actin Cytoskeleton in FMR1 Knockout Mice. In; Willemsen, R., Kooy, R.F.B.T.-F.X.S., Eds.; Academic Press, 2017; pp. 277–299 ISBN 978-0-12-804461-2.
- Oleinik, N. V; Helke, K.L.; Kistner-Griffin, E.; Krupenko, N.I.; Krupenko, S.A. Rho GTPases RhoA and Rac1 mediate effects of dietary folate on metastatic potential of A549 cancer cells through the control of cofilin phosphorylation. J. Biol. Chem. 2014, 289, 26383–26394. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Shen, W.; Zhu, H.; Lin, L.; Jiang, G.; Zhu, Y.; Song, H.; Wu, L. Zoledronate inhibits fibroblasts’ proliferation and activation via targeting TGF-β signaling pathway. Drug Des. Devel. Ther. 2018, 12, 3021–3031. [Google Scholar] [CrossRef]
- Wu, L.; Zhu, L.; Shi, W.-H.; Zhang, J.; Ma, D.; Yu, B. Zoledronate inhibits the proliferation, adhesion and migration of vascular smooth muscle cells. Eur. J. Pharmacol. 2009, 602, 124–131. [Google Scholar] [CrossRef]
- Tassone, P.; Tagliaferri, P.; Viscomi, C.; Palmieri, C.; Caraglia, M.; D’Alessandro, A.; Galea, E.; Goel, A.; Abbruzzese, A.; Boland, C.R.; et al. Zoledronic acid induces antiproliferative and apoptotic effects in human pancreatic cancer cells in vitro. Br. J. Cancer 2003, 88, 1971–1978. [Google Scholar] [CrossRef]
- Kara, M.; Boran, T.; Öztaş, E.; Jannuzzi, A.T.; Özden, S.; Özhan, G. Zoledronic acid-induced oxidative damage and endoplasmic reticulum stress-mediated apoptosis in human embryonic kidney (HEK-293) cells. J. Biochem. Mol. Toxicol. 2022, 36, e23083. [Google Scholar] [CrossRef] [PubMed]
- Lan, Z.; Chai, K.; Jiang, Y.; Liu, X. Characterization of urinary biomarkers and their relevant mechanisms of zoledronate-induced nephrotoxicity using rats and HK-2 cells. Hum. Exp. Toxicol. 2019, 38, 598–609. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, J.; Guo, T.; Liu, D.; Pan, J. Epidermal Growth Factor Reverses the Inhibitory Effects of the Bisphosphonate, Zoledronic Acid, on Human Oral Keratinocytes and Human Vascular Endothelial Cells In Vitro via the Epidermal Growth Factor Receptor (EGFR)/Akt/Phosphoinositide 3-Kinase (PI3K). Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2019, 25, 700–710. [Google Scholar] [CrossRef]
- Sharma, D.; Hamlet, S.M.; Petcu, E.B.; Ivanovski, S. The effect of bisphosphonates on the endothelial differentiation of mesenchymal stem cells. Sci. Rep. 2016, 6, 20580. [Google Scholar] [CrossRef]
- Treda, C.; Popeda, M.; Ksiazkiewicz, M.; Grzela, D.P.; Walczak, M.P.; Banaszczyk, M.; Peciak, J.; Stoczynska-Fidelus, E.; Rieske, P. EGFR Activation Leads to Cell Death Independent of PI3K/AKT/mTOR in an AD293 Cell Line. PLoS One 2016, 11, e0155230. [Google Scholar] [CrossRef]
- Inoue, R.; Matsuki, N.; Jing, G.; Kanematsu, T.; Abe, K.; Hirata, M. The inhibitory effect of alendronate, a nitrogen-containing bisphosphonate on the PI3K-Akt-NFkappaB pathway in osteosarcoma cells. Br. J. Pharmacol. 2005, 146, 633–641. [Google Scholar] [CrossRef]
- Hasmim, M.; Bieler, G.; Rüegg, C. Zoledronate inhibits endothelial cell adhesion, migration and survival through the suppression of multiple, prenylation-dependent signaling pathways. J. Thromb. Haemost. 2007, 5, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Maeng, Y.-S.; Min, J.-K.; Kim, J.H.; Yamagishi, A.; Mochizuki, N.; Kwon, J.-Y.; Park, Y.-W.; Kim, Y.-M.; Kwon, Y.-G. ERK is an anti-inflammatory signal that suppresses expression of NF-kappaB-dependent inflammatory genes by inhibiting IKK activity in endothelial cells. Cell. Signal. 2006, 18, 994–1005. [Google Scholar] [CrossRef] [PubMed]
- Açil, Y.; Arndt, M.L.; Gülses, A.; Wieker, H.; Naujokat, H.; Ayna, M.; Wiltfang, J. Cytotoxic and inflammatory effects of alendronate and zolendronate on human osteoblasts, gingival fibroblasts and osteosarcoma cells. J. cranio-maxillo-facial Surg. Off. Publ. Eur. Assoc. Cranio-Maxillo-Facial Surg. 2018, 46, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Broniarek, I.; Jarmuszkiewicz, W. [Statins and mitochondria]. Postepy Biochem. 2016, 62, 77–84. [Google Scholar]
- Jarmuszkiewicz, W.; Dominiak, K.; Budzinska, A.; Wojcicki, K.; Galganski, L. Mitochondrial coenzyme Q redox homeostasis and reactive oxygen species production. Front. Biosci. (Landmark Ed. 2023, 28, 61. [Google Scholar] [CrossRef]
- James, A.M.; Smith, R.A.J.; Murphy, M.P. Antioxidant and prooxidant properties of mitochondrial Coenzyme Q. Arch. Biochem. Biophys. 2004, 423, 47–56. [Google Scholar] [CrossRef]
- Kaya, Y.; Çebı, A.; Söylemez, N.; Demır, H.; Alp, H.H.; Bakan, E. Correlations between oxidative DNA damage, oxidative stress and coenzyme Q10 in patients with coronary artery disease. Int. J. Med. Sci. 2012, 9, 621–626. [Google Scholar] [CrossRef]
- Kalyan, S.; Huebbe, P.; Esatbeyoglu, T.; Niklowitz, P.; Côté, H.C.F.; Rimbach, G.; Kabelitz, D. Nitrogen-bisphosphonate therapy is linked to compromised coenzyme Q10 and vitamin E status in postmenopausal women. J. Clin. Endocrinol. Metab. 2014, 99, 1307–1313. [Google Scholar] [CrossRef]
- Budzinska, A.; Galganski, L.; Wojcicki, K.; Jarmuszkiewicz, W. Adaptation of mitochondrial bioenergetics to coenzyme Q deficiency in human endothelial cells after chronic exposure to bisphosphonates. Sci. Rep. 2025, 15, 17734. [Google Scholar] [CrossRef]
- Tang, Q.; Jiang, S.; Jia, W.; Shen, D.; Qiu, Y.; Zhao, Y.; Xue, B.; Li, C. Zoledronic acid, an FPPS inhibitor, ameliorates liver steatosis through inhibiting hepatic de novo lipogenesis. Eur. J. Pharmacol. 2017, 814, 169–177. [Google Scholar] [CrossRef]
- Cheng, L.; Ge, M.; Lan, Z.; Ma, Z.; Chi, W.; Kuang, W.; Sun, K.; Zhao, X.; Liu, Y.; Feng, Y.; et al. Zoledronate dysregulates fatty acid metabolism in renal tubular epithelial cells to induce nephrotoxicity. Arch. Toxicol. 2018, 92, 469–485. [Google Scholar] [CrossRef] [PubMed]
- Tricarico, P.M.; Crovella, S.; Celsi, F. Mevalonate Pathway Blockade, Mitochondrial Dysfunction and Autophagy: A Possible Link. Int. J. Mol. Sci. 2015, 16, 16067–16084. [Google Scholar] [CrossRef] [PubMed]
- Yamano, K.; Fogel, A.I.; Wang, C.; van der Bliek, A.M.; Youle, R.J. Mitochondrial Rab GAPs govern autophagosome biogenesis during mitophagy. Elife 2014, 3, e01612. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Yang, Q.; Guo, Y.; Liu, X.; Shen, D.; Jiang, C.; Wang, X.; Li, K.; Wang, H.; Yang, C.; et al. Oocytes orchestrate protein prenylation for mitochondrial function through selective inactivation of cholesterol biosynthesis in murine species. J. Biol. Chem. 2023, 299, 105183. [Google Scholar] [CrossRef]
- Kemeny-Suss, N.; Kasneci, A.; Rivas, D.; Afilalo, J.; Komarova, S. V; Chalifour, L.E.; Duque, G. Alendronate affects calcium dynamics in cardiomyocytes in vitro. Vascul. Pharmacol. 2009, 51, 350–358. [Google Scholar] [CrossRef]
- San-Millán, I. The Key Role of Mitochondrial Function in Health and Disease. Antioxidants 2023, 12. [Google Scholar] [CrossRef]
- Casanova, A.; Wevers, A.; Navarro-Ledesma, S.; Pruimboom, L. Mitochondria: It is all about energy. Front. Physiol. 2023, 14, 1114231. [Google Scholar] [CrossRef]
- Giorgi, C.; Marchi, S.; Pinton, P. The machineries, regulation and cellular functions of mitochondrial calcium. Nat. Rev. Mol. Cell Biol. 2018, 19, 713–730. [Google Scholar] [CrossRef]
- Mitrofan, L.M.; Castells, F.B.; Pelkonen, J.; Mönkkönen, J. Lysosomal-mitochondrial axis in zoledronic acid-induced apoptosis in human follicular lymphoma cells. J. Biol. Chem. 2010, 285, 1967–1979. [Google Scholar] [CrossRef]
- Guilland, D.F.; Sallis, J.D.; Fleisch, H. The effect of two diphosphonates on the handling of calcium by rat kidney mitochondria in vitro. Calcif. Tissue Res. 1974, 15, 303–314. [Google Scholar] [CrossRef]
- Guilland, D.F.; Fleisch, H. The effect of in vivo treatment with EHDP and/or 1,25-DHCC on calcium uptake and release in isolated kidney mitochondria. Biochem. Biophys. Res. Commun. 1974, 61, 906–911. [Google Scholar] [CrossRef] [PubMed]
- Greif, F.; Anais, D.; Frei, L.; Arbeit, L.; Sorroff, H.S. Blocking the calcium cascade in experimental acute renal failure. Isr. J. Med. Sci. 1990, 26, 301–305. [Google Scholar] [PubMed]
- Keyhani, J.; Keyhani, E. Mevalonic acid as a precursor of the alkyl sidechain of heme a of cytochrome c oxidase in yeast Saccharomyces cerevisiae. FEBS Lett. 1978, 93, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, H.; Rouhani, A.; Yüzügülen, J.; Ghaderi, F.; Fazlinezhad, R.; Kiafar, M.R.; Honarpishefard, Z.; Matinpour, P.; Arjmand, A.; Azarpira, N.; et al. Zoledronic acid-induced mitochondrial impairment, inflammation, and oxidative stress in the rat kidney. Trends Pharm. Sci. 2023, 9, 243–252. [Google Scholar] [CrossRef]
- Díaz-Casado, M.E.; Quiles, J.L.; Barriocanal-Casado, E.; González-García, P.; Battino, M.; López, L.C.; Varela-López, A. The Paradox of Coenzyme Q(10) in Aging. Nutrients 2019, 11, 2221. [Google Scholar] [CrossRef]
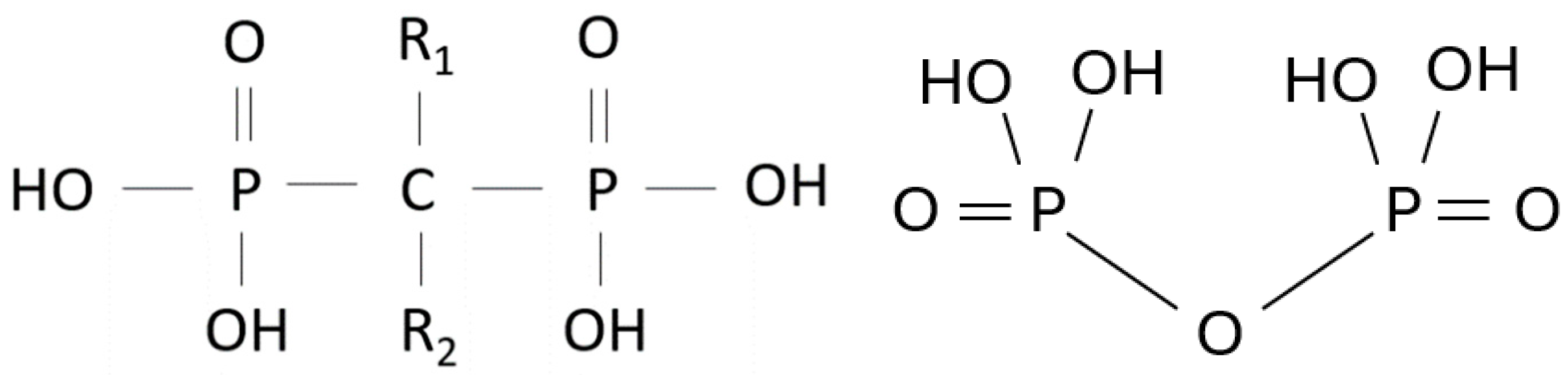
| Side effects | BPs |
| Osteomalacia | Etidronate |
| Hypocalcemia | Zoledronate, pamidronate, clodronate |
| Acute phase reaction | Zoledronate, ibandronate |
| Gastrointestinal side effects | Orally administered N-BPs, alendronate |
| Nephrotoxicity, renal failure | Zoledronate, pamidronate, ibandronate, alendronate |
| Ocular side effects | Pamidronate, zoledronate |
| Bisphosphonate-related osteonecrosis of the jaw (BRONJ) | Zoledronate |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




