Submitted:
22 June 2025
Posted:
23 June 2025
You are already at the latest version
Abstract
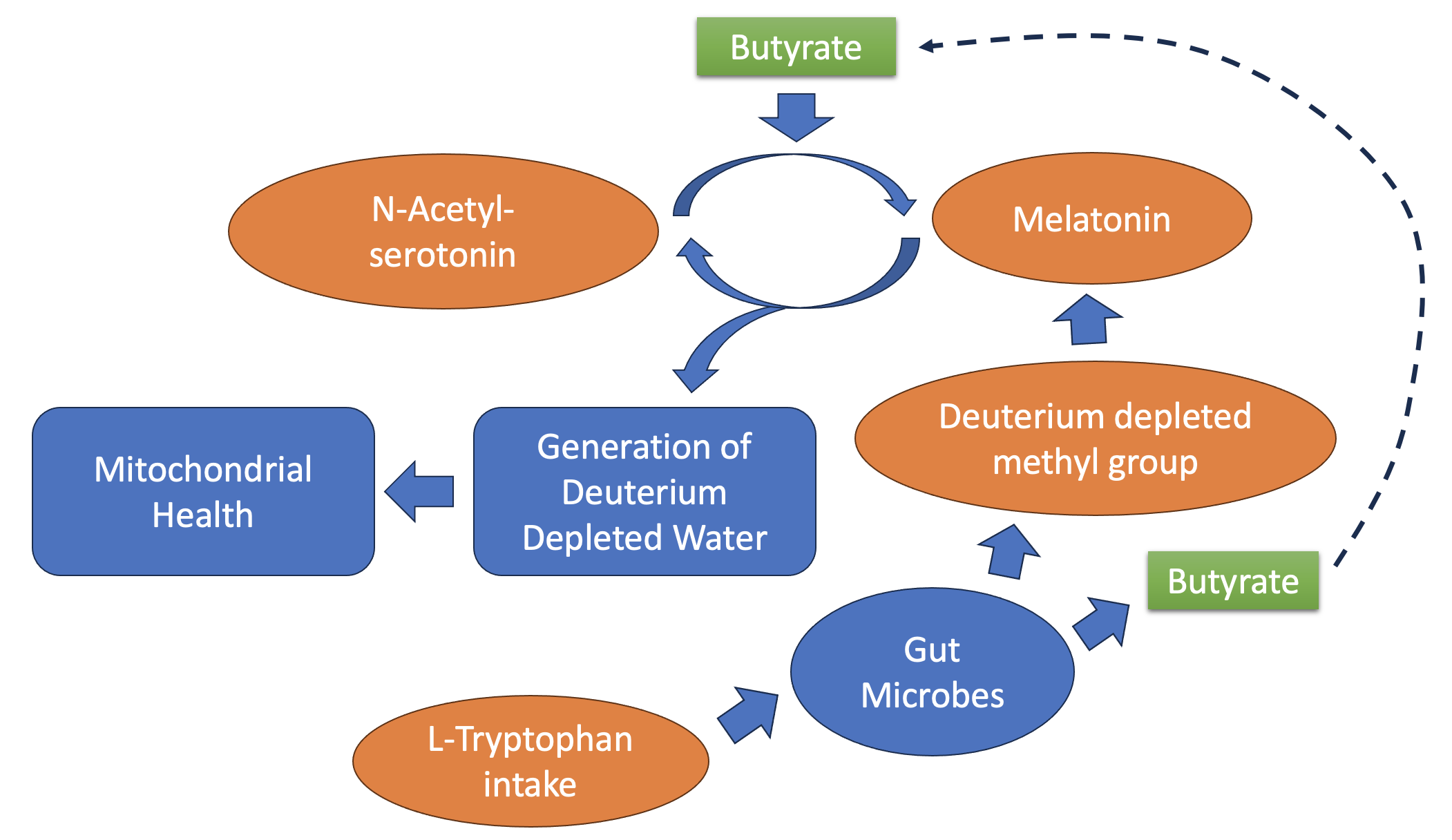
Keywords:
1. Introduction
2. The Co-Evolutionary Origin of the Mitochondrion and the Role of Melatonin Synthesis
3. S-Adenosylmethionine, Methylation Pathways, and Short Chain Fatty Acids
4. Butyrate is the Leading Short Chain Fatty Acid Offering Protection to Human Health
5. Does Hydrogen Peroxide Act as a Carrier Molecule for Deupleted Water in the Cell?
6. Cytochrome P450 Enzymes in the Endoplasmic Reticulum in the Gut and Liver
7. Considerations around Melatonin and Tryptophan Supplementation
8. Melatonin’s Role in Taming the Severity of SARS-CoV-2 Infection and Spike Protein-Related Pathology
8.1. Taming the Cytokine Storm
8.2. Protection from Coagulopathy
8.3. Protection from Neurodegeneration
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pal, P.K.; Sarkar, S.; Chattopadhyay, A.; Tan, D.X.; Bandyopadhyay, D. Enterochromaffin cells as the source of melatonin: key findings and functional relevance in mammals. Melatonin Res. 2019, 2, 61–82. [Google Scholar] [CrossRef]
- Zimmermann, P.; Kurth, S.; Pugin, B.; Bokulich, N.A. Microbial melatonin metabolism in the human intestine as a therapeutic target for dysbiosis and rhythm disorders. npj Biofilms Microbiomes 2024, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Raikhlin, N.T.; Kvetnoy, I.M.; Tolkachev, V.N. Melatonin may be synthesised in enterochromaffin cells. Nature 1975, 255, 344–345. [Google Scholar] [CrossRef] [PubMed]
- Raikhlin, N.T.; Kvetnoy, I.M. Melatonin and enterochromaffine cells. Acta Histochem. 1976, 55, 19–24. [Google Scholar] [CrossRef]
- Chen, C.-Q. Distribution, function and physiological role of melatonin in the lower gut. World J. Gastroenterol. 2011, 17, 3888–98. [Google Scholar] [CrossRef]
- Ahmadi, S.; Taghizadieh, M.; Mehdizadehfar, E.; Hasani, A.; Fard, J.K.; Feizi, H.; Hamishehkar, H.; Ansarin, M.; Yekani, M.; Memar, M.Y. Gut microbiota in neurological diseases: Melatonin plays an important regulatory role. Biomed. Pharmacother. 2024, 174, 116487. [Google Scholar] [CrossRef]
- Reigstad, C.S.; Salmonson, C.E.; Rainey, J.F., III; Szurszewski, J.H.; Linden, D.R.; Sonnenburg, J.L.; Farrugia, G.; Kashyap, P.C. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015, 29, 1395–1403. [Google Scholar] [CrossRef]
- Ge, X.; Pan, J.; Liu, Y.; Wang, H.; Zhou, W.; Wang, X. Intestinal Crosstalk between Microbiota and Serotonin and its Impact on Gut Motility. Curr. Pharm. Biotechnol. 2018, 19, 190–195. [Google Scholar] [CrossRef]
- Tan, D.-X.; Manchester, L.C.; Qin, L.; Reiter, R.J. Melatonin: A Mitochondrial Targeting Molecule Involving Mitochondrial Protection and Dynamics. Int. J. Mol. Sci. 2016, 17, 2124. [Google Scholar] [CrossRef]
- López, A.; García, J.A.; Escames, G.; Venegas, C.; Ortiz, F.; López, L.C.; Acuña-Castroviejo, D. Melatonin protects the mitochondria from oxidative damage reducing oxygen consumption, membrane potential, and superoxide anion production. J. Pineal Res. 2009, 46, 188–198. [Google Scholar] [CrossRef]
- Nasoni, M.G.; Carloni, S.; Canonico, B.; Burattini, S.; Cesarini, E.; Papa, S.; Pagliarini, M.; Ambrogini, P.; Balduini, W.; Luchetti, F. Melatonin reshapes the mitochondrial network and promotes intercellular mitochondrial transfer via tunneling nanotubes after ischemic-like injury in hippocampal HT22 cells. J. Pineal Res. 2021, 71, e12747. [Google Scholar] [CrossRef] [PubMed]
- Seneff, S.; Kyriakopoulos, A.M. Cancer, deuterium, and gut microbes: A novel perspective. Endocr. Metab. Sci. 2025, 17. [Google Scholar] [CrossRef]
- Kyriakopoulos, A.M.; Seneff, S. Explaining deuterium-depleted water as a cancer therapy: a narrative review. Eur. J. Cancer Prev. 2025. [Google Scholar] [CrossRef]
- Seneff, S.; Kyriakopoulos, A.M. Taurine prevents mitochondrial dysfunction and protects mitochondria from reactive oxygen species and deuterium toxicity. Amino Acids 2025, 57, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Seneff, S.; Nigh, G.; Kyriakopoulos, A.M. Is Deuterium Sequestering by Reactive Carbon Atoms an Important Mechanism to Reduce Deuterium Content in Biological Water? FASEB BioAdvances 2025, 7, e70019. [Google Scholar] [CrossRef]
- Seneff S, Nigh G, Kyriakopoulos AM. Mitochondrial dysfunction in Parkinson’s disease: is impaired deuterium-depleted nutrient supply by gut microbes a primary factor? Biocell 2025 [In Press].
- Hay, S.; Pudney, C.R.; Scrutton, N.S. Structural and mechanistic aspects of flavoproteins: probes of hydrogen tunnelling. FEBS J. 2009, 276, 3930–3941. [Google Scholar] [CrossRef]
- Tan, D.; Manchester, L.C.; Liu, X.; Rosales-Corral, S.A.; Acuna-Castroviejo, D.; Reiter, R.J. Mitochondria and chloroplasts as the original sites of melatonin synthesis: a hypothesis related to melatonin's primary function and evolution in eukaryotes. J. Pineal Res. 2012, 54, 127–138. [Google Scholar] [CrossRef]
- Esser, C.; Ahmadinejad, N.; Wiegand, C.; Rotte, C.; Sebastiani, F.; Gelius-Dietrich, G.; Henze, K.; Kretschmann, E.; Richly, E.; Leister, D.; et al. A Genome Phylogeny for Mitochondria Among -Proteobacteria and a Predominantly Eubacterial Ancestry of Yeast Nuclear Genes. Mol. Biol. Evol. 2004, 21, 1643–1660. [Google Scholar] [CrossRef]
- Rosengarten, H.; Meller, E.; Friedhoff, A.J. In vitro enzymatic formation of melatonin by human erythrocytes. . 1972, 4, 457–65. [Google Scholar]
- Manchester, L.C.; Poeggeler, B.; Alvares, F.L.; Ogden, G.B.; Reiter, R.J. Melatonin immunoreactivity in the photosynthetic prokaryote Rhodospirillum rubrum: implications for an ancient antioxidant system. Cell. Mol. Biol. Res. 1995, 41, 391–395. [Google Scholar]
- Bennett, G.M.; Kwak, Y.; Maynard, R.; Eyre-Walker, A. Endosymbioses Have Shaped the Evolution of Biological Diversity and Complexity Time and Time Again. Genome Biol. Evol. 2024, 16. [Google Scholar] [CrossRef] [PubMed]
- Mills, D.B.; Boyle, R.A.; Daines, S.J.; Sperling, E.A.; Pisani, D.; Donoghue, P.C.J.; Lenton, T.M. Eukaryogenesis and oxygen in Earth history. Nat. Ecol. Evol. 2022, 6, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Schultz, J.E.; Weaver, P.F. Fermentation and Anaerobic Respiration by Rhodospirillum rubrum and Rhodopseudomonas capsulata. J. Bacteriol. 1982, 149, 181–190. [Google Scholar] [CrossRef]
- Tan, D.-X.; Reiter, R.J. Mitochondria: the birth place, battle ground and the site of melatonin metabolism in cells. Melatonin Res. 2019, 2, 44–66. [Google Scholar] [CrossRef]
- Reiter, R.J.; Sharma, R.; Rosales-Corral, S.; Zuccari, D.A.P.d.C.; Chuffa, L.G.d.A. Melatonin: A mitochondrial resident with a diverse skill set. Life Sci. 2022, 301, 120612. [Google Scholar] [CrossRef]
- Coon, S.L.; Klein, D.C. Evolution of arylalkylamine N-acetyltransferase: Emergence and divergence. Mol. Cell. Endocrinol. 2006, 252, 2–10. [Google Scholar] [CrossRef]
- Quintela, T.; Gonçalves, I.; Silva, M.; Duarte, A.C.; Guedes, P.; Andrade, K.; Freitas, F.; Talhada, D.; Albuquerque, T.; Tavares, S.; et al. Choroid plexus is an additional source of melatonin in the brain. J. Pineal Res. 2018, 65, e12528. [Google Scholar] [CrossRef]
- Tan, D.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2006, 42, 28–42. [Google Scholar] [CrossRef]
- Menezo, Y.; Clement, P.; Clement, A.; Elder, K. Methylation: An Ineluctable Biochemical and Physiological Process Essential to the Transmission of Life. Int. J. Mol. Sci. 2020, 21, 9311. [Google Scholar] [CrossRef]
- Krichevsky, M.I.; Friedman, I.; Newell, M.F.; Sisler, F.D. Deuterium Fractionation during Molecular Hydrogen Formation in a Marine Pseudomonad. J. Biol. Chem. 1961, 236, 2520–2525. [Google Scholar] [CrossRef]
- Ducker, G.S.; Rabinowitz, J.D. One-Carbon Metabolism in Health and Disease. Cell Metab. 2017, 25, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Crowther, G.J.; KosálY, G.; Lidstrom, M.E. Formate as the Main Branch Point for Methylotrophic Metabolism in Methylobacterium extorquens AM1. J. Bacteriol. 2008, 190, 5057–5062. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Noor, E.; Ramos-Parra, P.A.; García-Valencia, L.E.; Patterson, J.A.; de la Garza, R.I.D.; Hanson, A.D.; Bar-Even, A. In Vivo Rate of Formaldehyde Condensation with Tetrahydrofolate. Metabolites 2020, 10, 65. [Google Scholar] [CrossRef] [PubMed]
- Mascarenhas, R.; Gouda, H.; Ruetz, M.; Banerjee, R.
- Schmidt, A.; Wu, H.; MacKenzie, R.E.; Chen, V.J.; Bewly, J.R.; Ray, J.E.; Toth, J.E.; Cygler, M. Structures of Three Inhibitor Complexes Provide Insight into the Reaction Mechanism of the Human Methylenetetrahydrofolate Dehydrogenase/Cyclohydrolase. Biochemistry 2000, 39, 6325–6335. [Google Scholar] [CrossRef]
- Xiong, R.-G.; Zhou, D.-D.; Wu, S.-X.; Huang, S.-Y.; Saimaiti, A.; Yang, Z.-J.; Shang, A.; Zhao, C.-N.; Gan, R.-Y.; Li, H.-B. Health Benefits and Side Effects of Short-Chain Fatty Acids. Foods 2022, 11, 2863. [Google Scholar] [CrossRef]
- Vincent, A.D.; Wang, X.-Y.; Parsons, S.P.; Khan, W.I.; Huizinga, J.D. Abnormal absorptive colonic motor activity in germ-free mice is rectified by butyrate, an effect possibly mediated by mucosal serotonin. Am. J. Physiol. Liver Physiol. 2018, 315, G896–G907. [Google Scholar] [CrossRef]
- Chen, X.-F.; Chen, X.; Tang, X. Short-chain fatty acid, acylation and cardiovascular diseases. Clin. Sci. 2020, 134, 657–676. [Google Scholar] [CrossRef]
- Pant, K.; Venugopal, S.K.; Pisarello, M.J.L.; Gradilone, S.A. The Role of Gut Microbiome-Derived Short-Chain Fatty Acid Butyrate in Hepatobiliary Diseases. Am. J. Pathol. 2023, 193, 1455–1467. [Google Scholar] [CrossRef]
- Liu, H.; Wang, J.; He, T.; Becker, S.; Zhang, G.; Li, D.; Ma, X. Butyrate: A Double-Edged Sword for Health? Adv. Nutr. 2018, 9, 21–29. [Google Scholar] [CrossRef]
- Sun, J.; Chen, S.; Zang, D.; Sun, H.; Sun, Y.; Chen, J. Butyrate as a promising therapeutic target in cancer: From pathogenesis to clinic (Review). Int. J. Oncol. 2024, 64, 1–16. [Google Scholar] [CrossRef]
- Eckschlager, T.; Plch, J.; Stiborova, M.; Hrabeta, J. Histone Deacetylase Inhibitors as Anticancer Drugs. Int. J. Mol. Sci. 2017, 18, 1414. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.V.; Sadler, R.K.; Llovera, G.; Singh, V.; Roth, S.; Heindl, S.; Monasor, L.S.; Verhoeven, A.; Peters, F.; Parhizkar, S.; et al. Microbiota-derived short chain fatty acids modulate microglia and promote Aβ plaque deposition. eLife 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Kim, S.J.; Ko, E.K.; Ahn, S.; Seo, H.; Sung, M. Gut microbiota-associated bile acid deconjugation accelerates hepatic steatosis in ob/ob mice. J. Appl. Microbiol. 2016, 121, 800–810. [Google Scholar] [CrossRef]
- Yano, J.M.; Yu, K.; Donaldson, G.P.; Shastri, G.G.; Ann, P.; Ma, L.; Nagler, C.R.; Ismagilov, R.F.; Mazmanian, S.K.; Hsiao, E.Y. Indigenous Bacteria from the Gut Microbiota Regulate Host Serotonin Biosynthesis. Cell 2015, 161, 264–276. [Google Scholar] [CrossRef]
- Gill, R.K.; Kumar, A.; Malhotra, P.; Maher, D.; Singh, V.; Dudeja, P.K.; Alrefai, W.; Saksena, S. Regulation of intestinal serotonin transporter expression via epigenetic mechanisms: role of HDAC2. Am. J. Physiol. Physiol. 2013, 304, C334–C341. [Google Scholar] [CrossRef]
- Zhou, D.; Chen, Y.-W.; Zhao, Z.-H.; Yang, R.-X.; Xin, F.-Z.; Liu, X.-L.; Pan, Q.; Zhou, H.; Fan, J.-G. Sodium butyrate reduces high-fat diet-induced non-alcoholic steatohepatitis through upregulation of hepatic GLP-1R expression. Exp. Mol. Med. 2018, 50, 1–12. [Google Scholar] [CrossRef]
- Yang, T.; Yang, H.; Heng, C.; Wang, H.; Chen, S.; Hu, Y.; Jiang, Z.; Yu, Q.; Wang, Z.; Qian, S.; et al. Amelioration of non-alcoholic fatty liver disease by sodium butyrate is linked to the modulation of intestinal tight junctions in db/db mice. Food Funct. 2020, 11, 10675–10689. [Google Scholar] [CrossRef]
- Amiri, P.; Arefhosseini, S.; Bakhshimoghaddam, F.; Gurvan, H.J.; Hosseini, S.A. Mechanistic insights into the pleiotropic effects of butyrate as a potential therapeutic agent on NAFLD management: A systematic review. Front. Nutr. 2022, 9, 1037696. [Google Scholar] [CrossRef]
- Hajjar, R.; Richard, C.S.; Santos, M.M. The role of butyrate in surgical and oncological outcomes in colorectal cancer. Am. J. Physiol. Liver Physiol. 2021, 320, G601–G608. [Google Scholar] [CrossRef]
- Zito, E. ERO1: A protein disulfide oxidase and H2O2 producer. Free. Radic. Biol. Med. 2015, 83, 299–304. [Google Scholar] [CrossRef]
- Bestetti, S.; Galli, M.; Sorrentino, I.; Pinton, P.; Rimessi, A.; Sitia, R.; Medraño-Fernandez, I. Human aquaporin-11 guarantees efficient transport of H2O2 across the endoplasmic reticulum membrane. Redox Biol. 2020, 28, 101326. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.P.; Weissman, J.S. The FAD- and O2-Dependent Reaction Cycle of Ero1-Mediated Oxidative Protein Folding in the Endoplasmic Reticulum. Mol. Cell 2002, 10, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Mailloux, R.J.; Mori, M. Mitochondrial Antioxidants and the Maintenance of Cellular Hydrogen Peroxide Levels. Oxidative Med. Cell. Longev. 2018, 2018, 7857251. [Google Scholar] [CrossRef]
- Seneff, S. Is deuterium fractionation a major controlling factor in human metabolism and cancer? An essential role for proline. Preprints , 2024. 19 June. [CrossRef]
- Mercedes-Camacho, A.Y.; Mullins, A.B.; Mason, M.D.; Xu, G.G.; Mahoney, B.J.; Wang, X.; Peng, J.W.; Etzkorn, F.A. Kinetic Isotope Effects Support the Twisted Amide Mechanism of Pin1 Peptidyl-Prolyl Isomerase. Biochemistry 2013, 52, 7707–7713. [Google Scholar] [CrossRef]
- Gharibi, H.; Chernobrovkin, A.L.; Eriksson, G.; Saei, A.A.; Timmons, Z.; Kitchener, A.C.; Kalthoff, D.C.; Lidén, K.; Makarov, A.A.; Zubarev, R.A. Abnormal (Hydroxy)proline Deuterium Content Redefines Hydrogen Chemical Mass. J. Am. Chem. Soc. 2022, 144, 2484–2487. [Google Scholar] [CrossRef]
- Stetten MR, Schoenheimerz R. The metabolism of I(-)-proline studied with the aid of deuterium and isotopic nitrogen. JBC 1944; 153(1): 113-132.
- Schönbrunner, E.R.; Schmid, F.X. Peptidyl-prolyl cis-trans isomerase improves the efficiency of protein disulfide isomerase as a catalyst of protein folding. Proc. Natl. Acad. Sci. 1992, 89, 4510–4513. [Google Scholar] [CrossRef]
- DeCoursey, T.E.; Cherny, V.V. Deuterium Isotope Effects on Permeation and Gating of Proton Channels in Rat Alveolar Epithelium. J. Gen. Physiol. 1997, 109, 415–434. [Google Scholar] [CrossRef]
- Werck-Reichhart, D.; Feyereisen, R. Cytochromes P450: a success story. Genome Biol. 2000, 1, 1–9. [Google Scholar] [CrossRef]
- Zhao, M.; Ma, J.; Li, M.; Zhang, Y.; Jiang, B.; Zhao, X.; Huai, C.; Shen, L.; Zhang, N.; He, L.; et al. Cytochrome P450 Enzymes and Drug Metabolism in Humans. Int. J. Mol. Sci. 2021, 22, 12808. [Google Scholar] [CrossRef]
- Ma, X.; Idle, J.R.; Krausz, K.W.; Gonzalez, F.J. Metabolism of melatonin by human cytochromes p450. Drug Metab. Dispos. 2005, 33, 489–494. [Google Scholar] [CrossRef]
- Gerecke, C.; Rodrigues, C.E.; Homann, T.; Kleuser, B. The Role of Ten-Eleven Translocation Proteins in Inflammation. Front. Immunol. 2022, 13, 861351. [Google Scholar] [CrossRef] [PubMed]
- Läpple, F.; Von Richter, O.; Fromm, M.F.; Richter, T.; Thon, K.P.; Wisser, H.; Griese, E.-U.; Eichelbaum, M.; Kivistö, K.T. Differential expression and function of CYP2C isoforms in human intestine and liver. Pharmacogenetics 2003, 13, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Somers, V.K.; Xu, H.; Lopez-Jimenez, F.; Covassin, N. Trends in Use of Melatonin Supplements Among US Adults, 1999-2018. JAMA 2022, 327, 483–485. [Google Scholar] [CrossRef] [PubMed]
- Future Market Insights. Melatonin Market Analysis—Size, Share, and Forecast from 2025 to 2035. [last accessed , 2025]. https://www.futuremarketinsights.com/reports/melatonin-market. 10 June.
- Arnao, M.B.; Giraldo-Acosta, M.; Castejón-Castillejo, A.; Losada-Lorán, M.; Sánchez-Herrerías, P.; El Mihyaoui, A.; Cano, A.; Hernández-Ruiz, J. Melatonin from Microorganisms, Algae, and Plants as Possible Alternatives to Synthetic Melatonin. Metabolites 2023, 13, 72. [Google Scholar] [CrossRef]
- Zimmermann, R.C.; McDougle, C.J.; Schumacher, M.; Olcese, J.; Mason, J.W.; Heninger, G.R.; Price, L.H. Effects of acute tryptophan depletion on nocturnal melatonin secretion in humans. J. Clin. Endocrinol. Metab. 1993, 76, 1160–1164. [Google Scholar] [CrossRef]
- Esteban, S.; Nicolaus, C.; Garmundi, A.; Rial, R.V.; Rodríguez, A.B.; Ortega, E.; Ibars, C.B. Effect of orally administered l-tryptophan on serotonin, melatonin, and the innate immune response in the rat. Mol. Cell. Biochem. 2004, 267, 39–46. [Google Scholar] [CrossRef]
- Kamfar, W.W.; Khraiwesh, H.M.; Ibrahim, M.O.; Qadhi, A.H.; Azhar, W.F.; Ghafouri, K.J.; Alhussain, M.H.; AlShahrani, A.M.; Alghannam, A.F.; Abdulal, R.H.; et al. Comprehensive review of melatonin as a promising nutritional and nutraceutical supplement. Heliyon 2024, 10, e24266. [Google Scholar] [CrossRef]
- Friedman, M. Analysis, Nutrition, and Health Benefits of Tryptophan. Int. J. Tryptophan Res. 2018, 11. [Google Scholar] [CrossRef]
- Tordjman, S.; Chokron, S.; Delorme, R.; Charrier, A.; Bellissant, E.; Jaafari, N.; Fougerou, C. Melatonin: Pharmacology, Functions and Therapeutic Benefits. Curr. Neuropharmacol. 2017, 15, 434–443. [Google Scholar] [CrossRef]
- Jackson, C.B.; Farzan, M.; Chen, B.; Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 2022, 23, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, Y.; Ma, J.; Zhang, Q.; Shao, J.; Liang, S.; Yu, Y.; Li, W.; Wang, C. The long-term health outcomes, pathophysiological mechanisms and multidisciplinary management of long COVID. Signal Transduct. Target. Ther. 2023, 8, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Cecon, E.; Fernandois, D.; Renault, N.; Coelho, C.F.F.; Wenzel, J.; Bedart, C.; Izabelle, C.; Gallet, S.; Le Poder, S.; Klonjkowski, B.; et al. Melatonin drugs inhibit SARS-CoV-2 entry into the brain and virus-induced damage of cerebral small vessels. Cell. Mol. Life Sci. 2022, 79, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Tamura, E.K.; Cecon, E.; Monteiro, A.W.A.; Silva, C.L.M.; Markus, R.P. Melatonin inhibits LPS-induced NO production in rat endothelial cells. J. Pineal Res. 2009, 46, 268–274. [Google Scholar] [CrossRef]
- Calvo, J.R.; González-Yanes, C.; Maldonado, M.D. The role of melatonin in the cells of the innate immunity: a review. J. Pineal Res. 2013, 55, 103–120. [Google Scholar] [CrossRef]
- Hosseinzadeh, A.; Bagherifard, A.; Koosha, F.; Amiri, S.; Karimi-Behnagh, A.; Reiter, R.J.; Mehrzadi, S. Melatonin effect on platelets and coagulation: Implications for a prophylactic indication in COVID-19. Life Sci. 2022, 307, 120866–120866. [Google Scholar] [CrossRef]
- Carrillo-Vico, A.; Calvo, J.R.; Abreu, P.; Lardone, P.J.; García-Mauriño, S.; Reiter, R.J.; Guerrero, J.M. Evidence of melatonin synthesis by human lymphocytes and its physiological significance: possible role as intracrine, autocrine, and/or paracrine substance. FASEB J. 2004, 18, 537–539. [Google Scholar] [CrossRef]
- Miller, S.C.; Pandi, P.S.R.; Esquifino, A.I.; Cardinali, D.P.; Maestroni, G.J.M. The role of melatonin in immuno-enhancement: potential application in cancer. Int. J. Exp. Pathol. 2006, 87, 81–87. [Google Scholar] [CrossRef]
- García-Mauriño, S.; Pozo, D.; Carrillo-Vico, A.; Calvo, J.R.; Guerrero, J.M. Melatonin activates Th1 lymphocytes by increasing IL-12 production. Life Sci. 1999, 65, 2143–2150. [Google Scholar] [CrossRef]
- Meyaard, L.; Hovenkamp, E.; A Otto, S.; Miedema, F. IL-12-induced IL-10 production by human T cells as a negative feedback for IL-12-induced immune responses. J. Immunol. 1996, 156, 2776–2782. [Google Scholar] [CrossRef]
- Chau, C.W.; Sugimura, R.; Kong, H. Locked in a pro-inflammatory state. eLife 2022, 11. [Google Scholar] [CrossRef]
- Salina, A.C.; Dos-Santos, D.; Rodrigues, T.S.; Fortes-Rocha, M.; Freitas-Filho, E.G.; Alzamora-Terrel, D.L.; Castro, I.M.; da Silva, T.F.F.; de Lima, M.H.; Nascimento, D.C.; et al. Efferocytosis of SARS-CoV-2-infected dying cells impairs macrophage anti-inflammatory functions and clearance of apoptotic cells. eLife 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Chen, S.; Zeng, S.; Zhao, Y.; Zhu, C.; Deng, B.; Zhu, G.; Yin, Y.; Wang, W.; Hardeland, R.; et al. Melatonin in macrophage biology: Current understanding and future perspectives. J. Pineal Res. 2018, 66, e12547. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.Y.; Ahn, S.; Cho, Y.-S.; Seo, S.-K.; Kim, D.H.; Shin, J.-G.; Lee, S.-J. CYP2C19 Contributes to THP-1-Cell-Derived M2 Macrophage Polarization by Producing 11,12- and 14,15-Epoxyeicosatrienoic Acid, Agonists of the PPARγ Receptor. Pharmaceuticals 2023, 16, 593. [Google Scholar] [CrossRef] [PubMed]
- Hasan, Z.T.; Al Atrakji, M.Q.Y.M.A.; Mehuaiden, A.K. The Effect of Melatonin on Thrombosis, Sepsis and Mortality Rate in COVID-19 Patients. Int. J. Infect. Dis. 2021, 114, 79–84. [Google Scholar] [CrossRef]
- Lehmann, A.; Prosch, H.; Zehetmayer, S.; Gysan, M.R.; Bernitzky, D.; Vonbank, K.; Idzko, M.; Gompelmann, D.; Sobh, E. Impact of persistent D-dimer elevation following recovery from COVID-19. PLOS ONE 2021, 16, e0258351. [Google Scholar] [CrossRef]
- Wirtz, P.H.; Bärtschi, C.; Spillmann, M.; Ehlert, U.; Von Känel, R. Effect of oral melatonin on the procoagulant response to acute psychosocial stress in healthy men: a randomized placebo-controlled study. J. Pineal Res. 2007, 44, 358–365. [Google Scholar] [CrossRef]
- Halma, M.T.J.; Plothe, C.; Marik, P.; Lawrie, T.A. Strategies for the Management of Spike Protein-Related Pathology. Microorganisms 2023, 11, 1308. [Google Scholar] [CrossRef]
- Kyriakopoulos, A.M.; Nigh, G.; A McCullough, P.; Seneff, S. Mitogen Activated Protein Kinase (MAPK) Activation, p53, and Autophagy Inhibition Characterize the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Spike Protein Induced Neurotoxicity. Cureus 2022, 14, e32361. [Google Scholar] [CrossRef]
- Khan, S.; Shafiei, M.S.; Longoria, C.; Schoggins, J.W.; Savani, R.C.; Zaki, H. SARS-CoV-2 spike protein induces inflammation via TLR2-dependent activation of the NF-κB pathway. eLife 2021, 10. [Google Scholar] [CrossRef]
- Parry, P.I.; Lefringhausen, A.; Turni, C.; Neil, C.J.; Cosford, R.; Hudson, N.J.; Gillespie, J. ‘Spikeopathy’: COVID-19 Spike Protein Is Pathogenic, from Both Virus and Vaccine mRNA. Biomedicines 2023, 11, 2287. [Google Scholar] [CrossRef]
- Seneff, S.; Kyriakopoulos, A.M.; Nigh, G.; A McCullough, P.; McCullough, P.A. A Potential Role of the Spike Protein in Neurodegenerative Diseases: A Narrative Review. Cureus 2023, 15, e34872. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Possible Application of Melatonin in Long COVID. Biomolecules 2022, 12, 1646. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef]
- Pan, H.-C.; Yang, C.-N.; Lee, W.-J.; Sheehan, J.; Wu, S.-M.; Chen, H.-S.; Lin, M.-H.; Shen, L.-W.; Lee, S.-H.; Shen, C.-C.; et al. Melatonin Enhanced Microglia M2 Polarization in Rat Model of Neuro-inflammation Via Regulating ER Stress/PPARδ/SIRT1 Signaling Axis. J. Neuroimmune Pharmacol. 2024, 19, 1–19. [Google Scholar] [CrossRef]
- Silvagno, F.; Vernone, A.; Pescarmona, G.P. The Role of Glutathione in Protecting against the Severe Inflammatory Response Triggered by COVID-19. Antioxidants 2020, 9, 624. [Google Scholar] [CrossRef]
- Morgan, M.J.; Liu, Z.-G. Crosstalk of reactive oxygen species and NF-kappaκB signaling. Cell Res. 2011, 21, 103–115. [Google Scholar] [CrossRef]
- Amioka, N.; Nakamura, K.; Kimura, T.; Ohta-Ogo, K.; Tanaka, T.; Toji, T.; Akagi, S.; Nakagawa, K.; Toh, N.; Yoshida, M.; et al. Pathological and clinical effects of interleukin-6 on human myocarditis. J. Cardiol. 2021, 78, 157–165. [Google Scholar] [CrossRef]
- Yonker, L.M.; Swank, Z.; Bartsch, Y.C.; Burns, M.D.; Kane, A.; Boribong, B.P.; Davis, J.P.; Loiselle, M.; Novak, T.; Senussi, Y.; et al. Circulating Spike Protein Detected in Post–COVID-19 mRNA Vaccine Myocarditis. Circulation 2023, 147, 867–876. [Google Scholar] [CrossRef]
- Buoninfante, A.; Andeweg, A.; Genov, G.; Cavaleri, M. Myocarditis associated with COVID-19 vaccination. npj Vaccines 2024, 9, 1–8. [Google Scholar] [CrossRef]
- Sun, E.; Motolani, A.; Campos, L.; Lu, T. The Pivotal Role of NF-kB in the Pathogenesis and Therapeutics of Alzheimer’s Disease. Int. J. Mol. Sci. 2022, 23, 8972. [Google Scholar] [CrossRef]
- Wu, J.; Huang, Y.; Zhan, C.; Chen, L.; Lin, Z.; Song, Z. Thioredoxin-1 promotes the restoration of alveolar bone in periodontitis with diabetes. iScience 2023, 26, 107618. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Cheng, Q.; Hu, Y.; Fan, X.; Liang, C.; Niu, C.; Kang, Q.; Wei, T. Melatonin antagonizes oxidative stress-induced apoptosis in retinal ganglion cells through activating the thioredoxin-1 pathway. Mol. Cell. Biochem. 2024, 479, 3393–3404. [Google Scholar] [CrossRef] [PubMed]
- Djavaheri-Mergny, M.; Javelaud, D.; Wietzerbin, J.; Besançon, F. NF-κB activation prevents apoptotic oxidative stress via an increase of both thioredoxin and MnSOD levels in TNFα-treated Ewing sarcoma cells. FEBS Lett. 2004, 578, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Smail, S.W.; Albarzinji, N.; Salih, R.H.; Taha, K.O.; Hirmiz, S.M.; Ismael, H.M.; Noori, M.F.; Azeez, S.S.; Janson, C. Microbiome dysbiosis in SARS-CoV-2 infection: implication for pathophysiology and management strategies of COVID-19. Front. Cell. Infect. Microbiol. 2025, 15, 1537456. [Google Scholar] [CrossRef]
- Bonmatí-Carrión, M.; Rol, M.-A. Melatonin as a Mediator of the Gut Microbiota–Host Interaction: Implications for Health and Disease. Antioxidants 2023, 13, 34. [Google Scholar] [CrossRef]
- Rubio-Casillas, A.; Fabrowski, M.; Brogna, C.; Cowley, D.; Redwan, E.M.; Uversky, V.N. Could the Spike Protein Derived from mRNA Vaccines Negatively Impact Beneficial Bacteria in the Gut? COVID 2024, 4, 1368–1378. [Google Scholar] [CrossRef]
- Boston, R.H.; Guan, R.; Kalmar, L.; Beier, S.; Horner, E.C.; Beristain-Covarrubias, N.; Yam-Puc, J.C.; Gerber, P.P.; Faria, L.; Kuroshchenkova, A.; et al. Stability of gut microbiome after COVID-19 vaccination in healthy and immuno-compromised individuals. Life Sci. Alliance 2024, 7, e202302529. [Google Scholar] [CrossRef]
- Yehia, A.; Abulseoud, O.A. Melatonin: a ferroptosis inhibitor with potential therapeutic efficacy for the post-COVID-19 trajectory of accelerated brain aging and neurodegeneration. Mol. Neurodegener. 2024, 19, 1–31. [Google Scholar] [CrossRef]
- Davis, H.E.; McCorkell, L.; Vogel, J.M.; Topol, E.J. Author Correction: Long COVID: major findings, mechanisms and recommendations. Nat. Rev. Microbiol. 2023, 21, 408–408. [Google Scholar] [CrossRef]
- Crivelli, L.; Palmer, K.; Calandri, I.; Guekht, A.; Beghi, E.; Carroll, W.; Frontera, J.; García-Azorín, D.; Westenberg, E.; Winkler, A.S.; et al. Changes in cognitive functioning after COVID-19: A systematic review and meta-analysis. Alzheimer's Dement. 2022, 18, 1047–1066. [Google Scholar] [CrossRef]
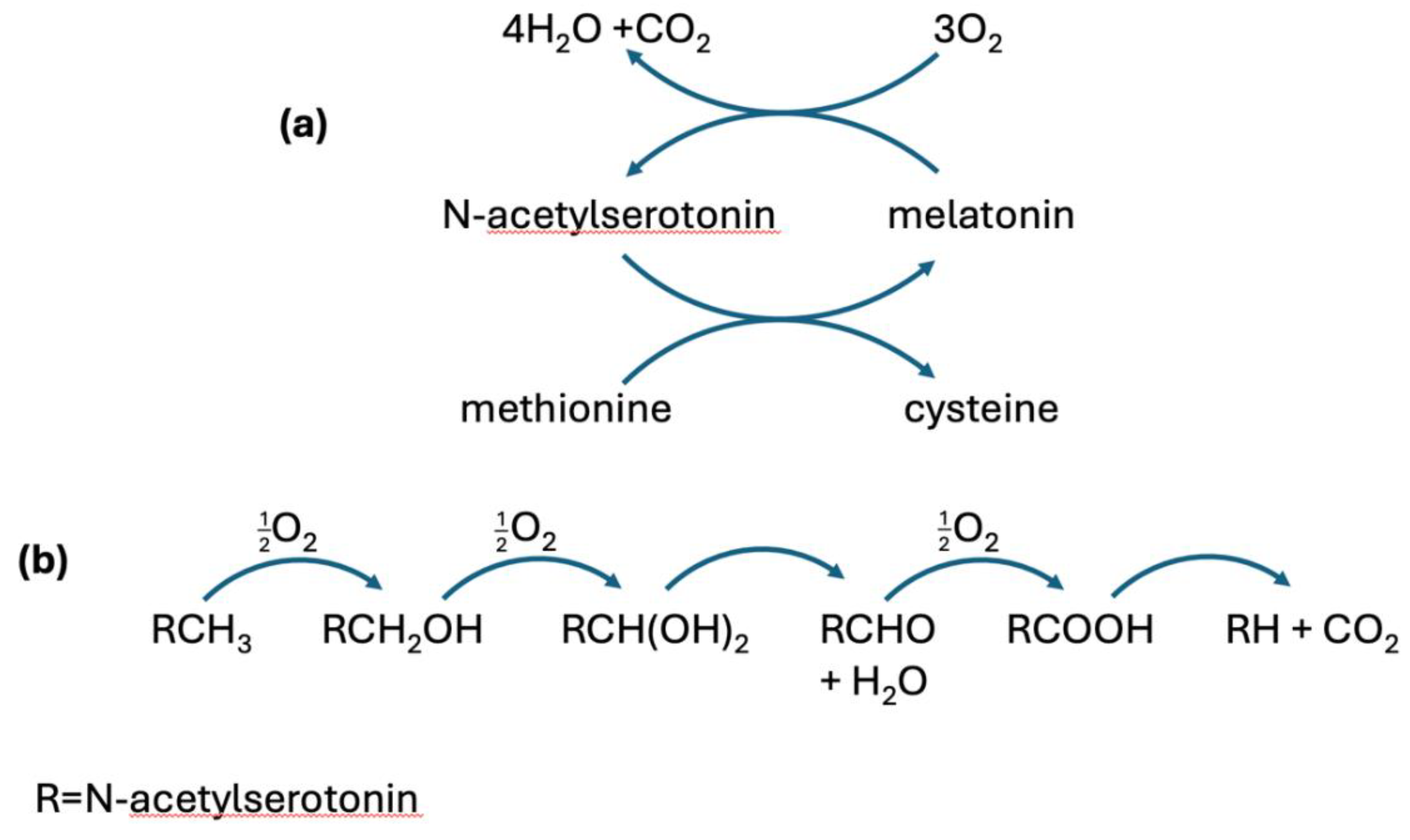
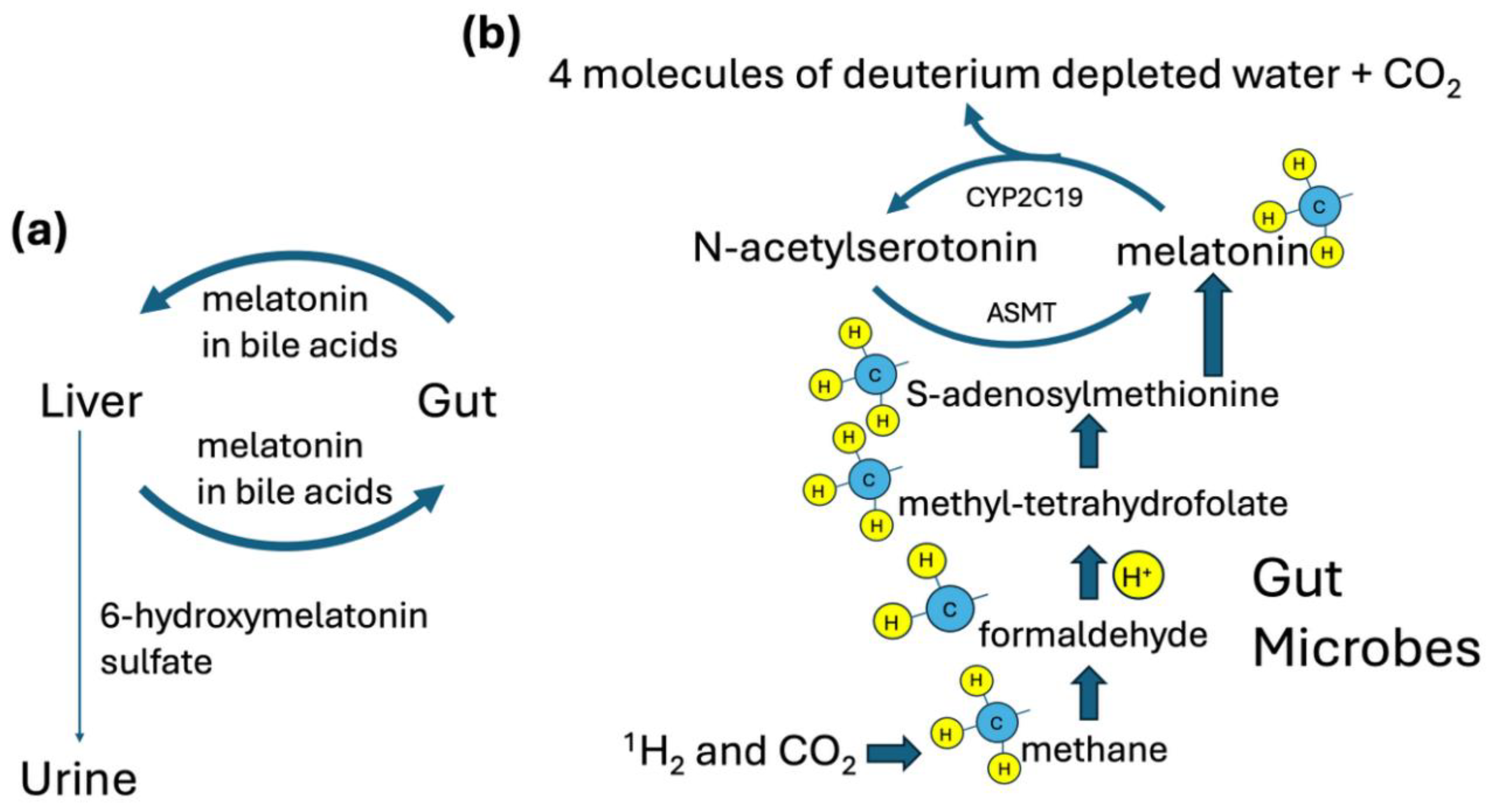
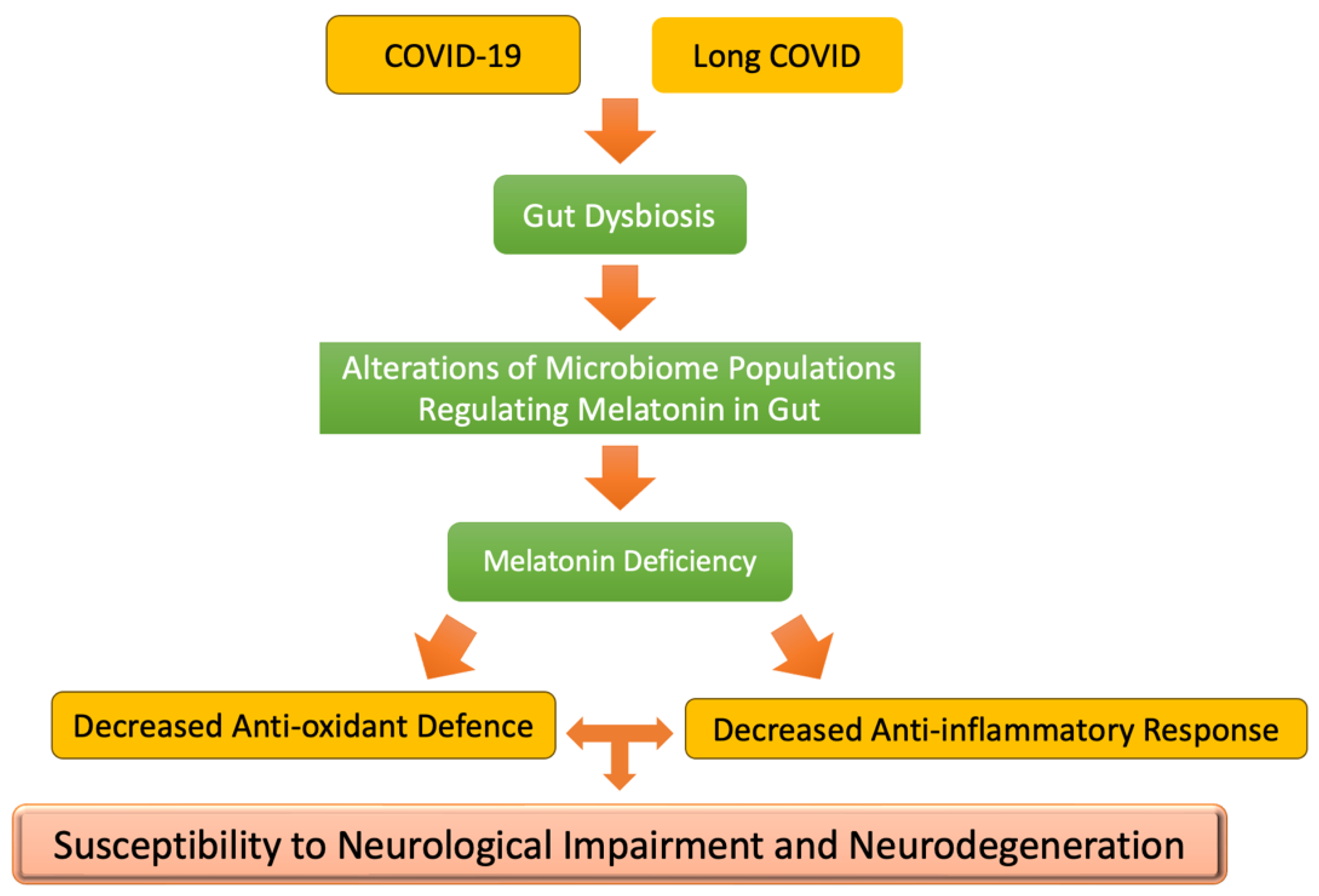
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



