Submitted:
18 June 2025
Posted:
19 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals
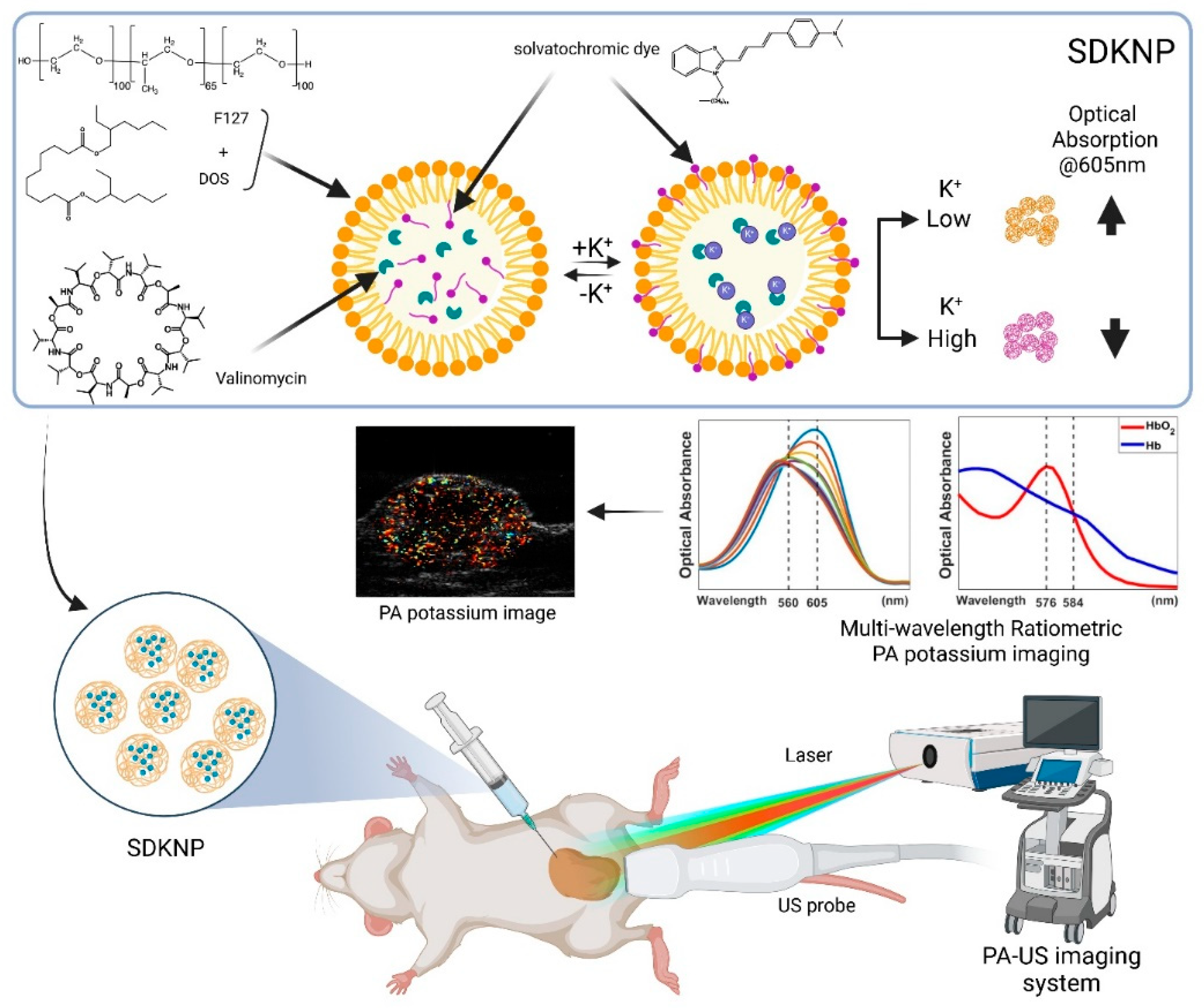
2.2. Synthesis of SDKNP
2.2.1. 2-methyl-3-octadecylbenzo[d]thiazol-3-ium (SD2a)
2.2.2. 2-((1E,3E)-4-(4-(dimethylamino)phenyl)buta-1,3-dien-1-yl)-3-octadecylbenzo[d]thiazol-3-ium iodide (SD2)
2.2.3. SDKNP
2.3. Animal Models
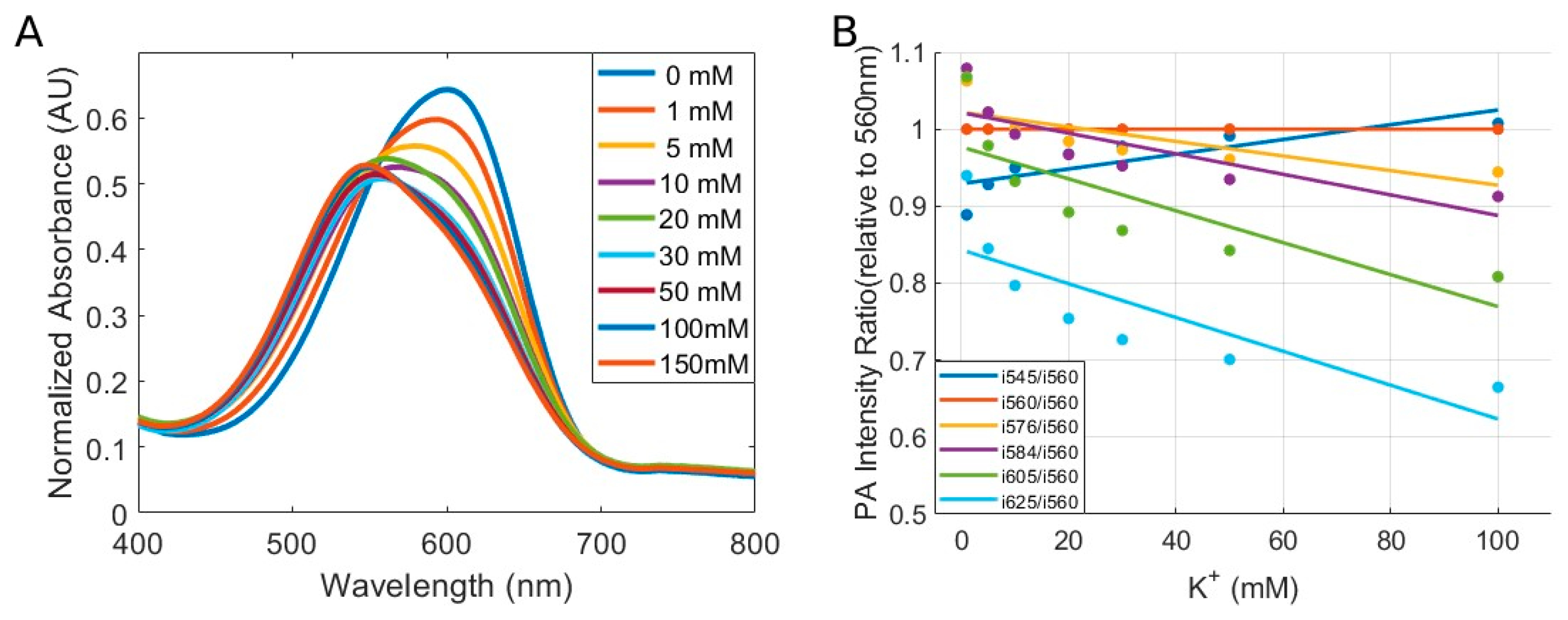
2.4. Multi-Wavelength Photoacoustic Ratiometric Imaging of K⁺
2.5. Collection of Tumor Interstitial Fluid
2.6. Inductively Coupled Plasma Mass Spectrometry.
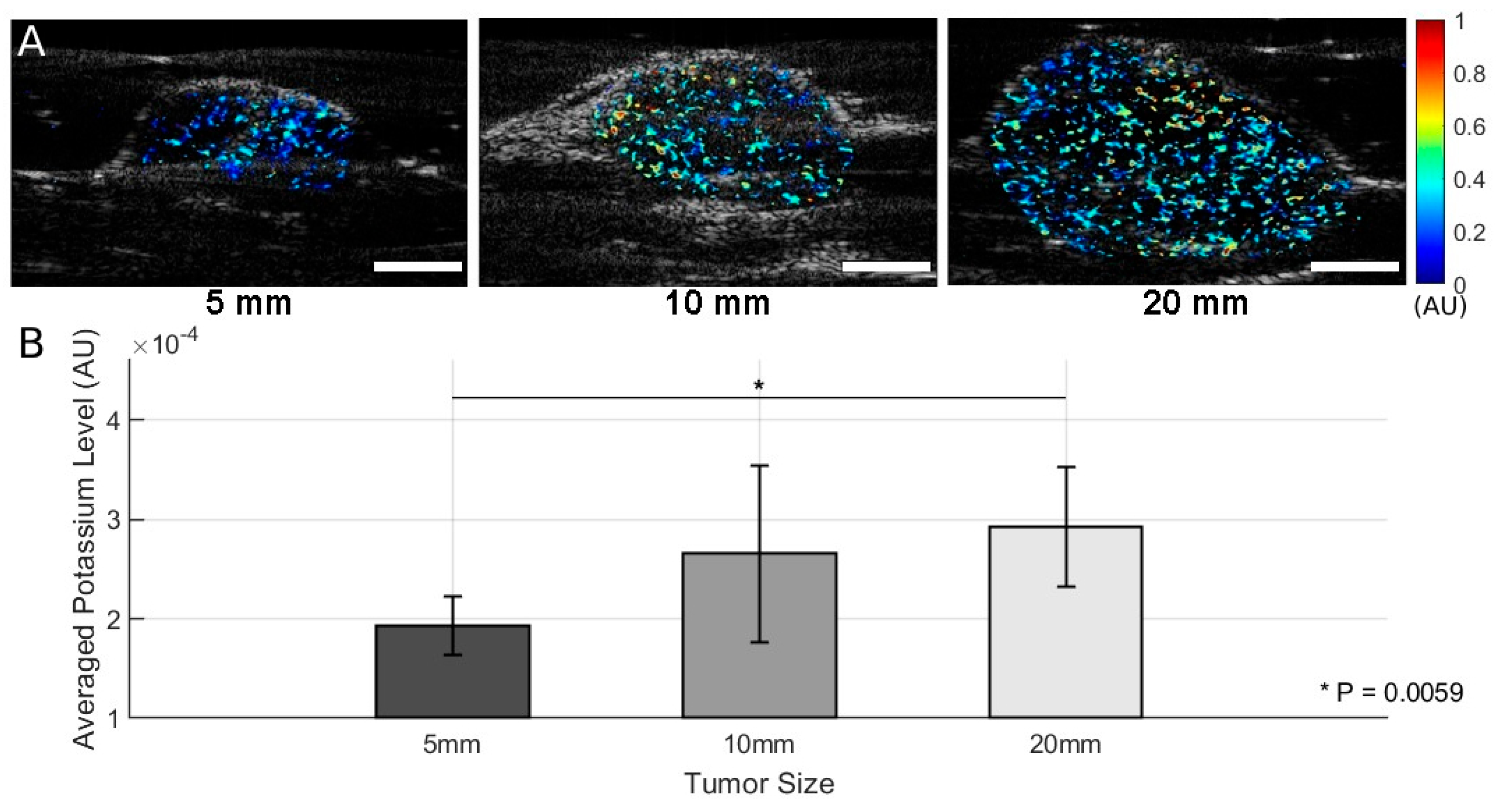
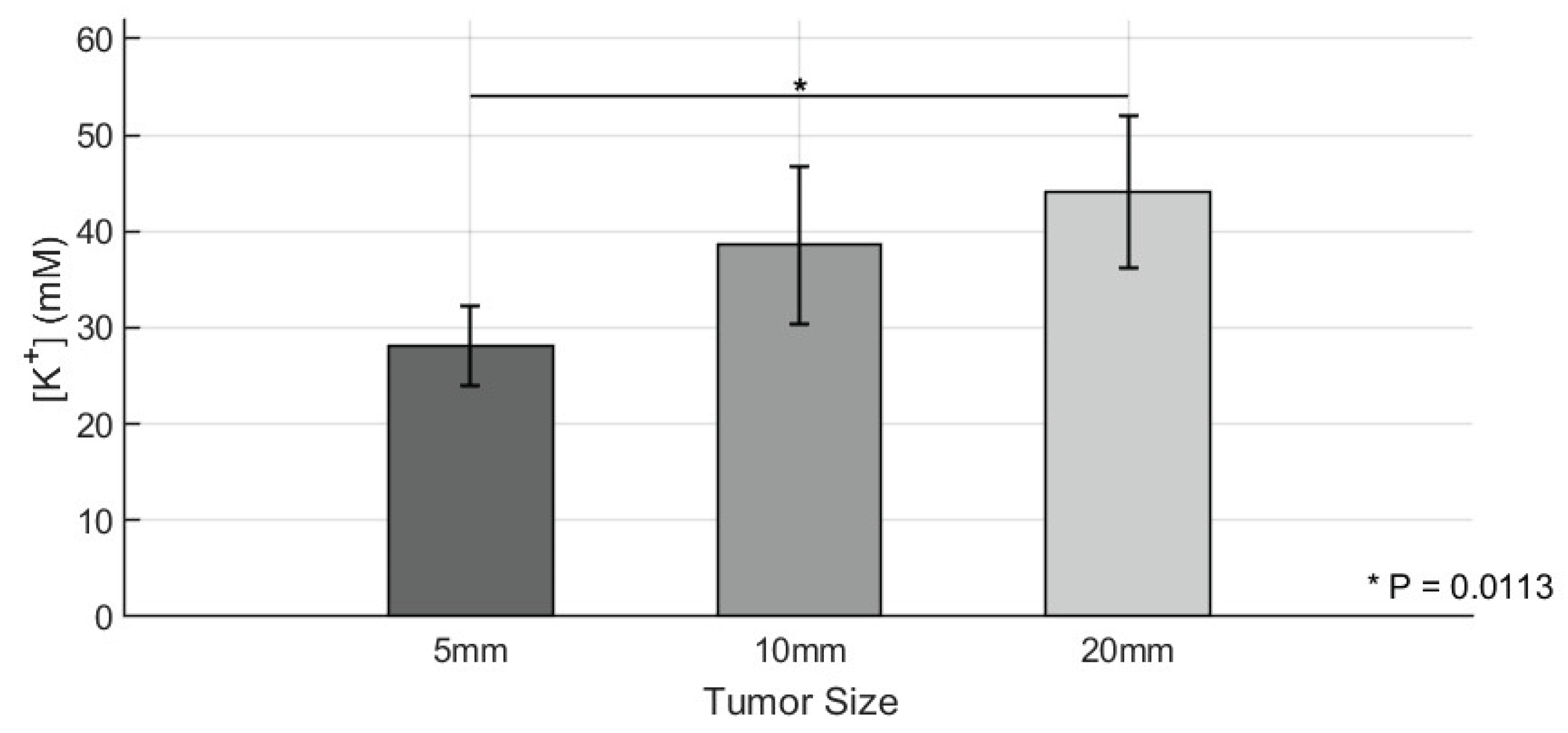
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ICP-MS | Inductively coupled plasma mass spectrometry |
| K+ | Potassium |
| PACI | Photoacoustic chemical imaging |
| SDKNP | Solvatochromic dye-based, potassium-sensitive nanoprobe |
| TME | Tumor microenvironment |
| UCUCA | University committee on the use and care of animals |
References
- Rockwell, S.; Dobrucki, I.T.; Kim, E.Y.; Marrison, S.T.; Vu, V.T. Hypoxia and radiation therapy: past history, ongoing research, and future promise. Curr Mol Med 2009, 9, 442-458. [CrossRef]
- Harrison, L.B.; Chadha, M.; Hill, R.J.; Hu, K.; Shasha, D. Impact of tumor hypoxia and anemia on radiation therapy outcomes. Oncologist 2002, 7, 492-508. [CrossRef]
- Horsman, M.R.; Mortensen, L.S.; Petersen, J.B.; Busk, M.; Overgaard, J. Imaging hypoxia to improve radiotherapy outcome. Nat Rev Clin Oncol 2012, 9, 674-687. [CrossRef]
- Bortner, C.D.; Hughes, F.M., Jr.; Cidlowski, J.A. A primary role for K+ and Na+ efflux in the activation of apoptosis. J Biol Chem 1997, 272, 32436-32442.
- Sigel, A., Sigel, H., & Sigel R. K. Interrelations between essential metal ions and human diseases, 1 ed.; Springer.
- Eil, R.; Vodnala, S.K.; Clever, D.; Klebanoff, C.A.; Sukumar, M.; Pan, J.H.; Palmer, D.C.; Gros, A.; Yamamoto, T.N.; Patel, S.J.; et al. Ionic immune suppression within the tumour microenvironment limits T cell effector function. Nature 2016, 537, 539-543. [CrossRef]
- Vodnala, S.K.; Eil, R.; Kishton, R.J.; Sukumar, M.; Yamamoto, T.N.; Ha, N.H.; Lee, P.H.; Shin, M.; Patel, S.J.; Yu, Z.; et al. T cell stemness and dysfunction in tumors are triggered by a common mechanism. Science 2019, 363. [CrossRef]
- Elabyad, I.A.; Kalayciyan, R.; Shanbhag, N.C.; Schad, L.R. First Potassium-39 (K) MRI at 9.4 T Using Conventional Copper Radio Frequency Surface Coil Cooled to 77 K. Ieee T Bio-Med Eng 2014, 61, 334-345. [CrossRef]
- Jo, J.; Lee, C.H.; Kopelman, R.; Wang, X. In vivo quantitative imaging of tumor pH by nanosonophore assisted multispectral photoacoustic imaging. Nat Commun 2017, 8, 471. [CrossRef]
- Song, J.; Zhai, T.; Hahm, H.S.; Li, Y.; Mao, H.; Wang, X.; Jo, J.; Chang, J.W. Development of a Dual Factor Activatable Covalent Targeted Photoacoustic Imaging Probe for Tumor Imaging. Angew Chem Int Ed Engl 2024, e202410645. [CrossRef]
- Folz, J.; Jo, J.; Gonzalez, M.E.; Eido, A.; Zhai, T.; Caruso, R.; Kleer, C.G.; Wang, X.; Kopelman, R. Photoacoustic lifetime oxygen imaging of radiotherapy-induced tumor reoxygenation In Vivo. J Photochem Photobiol 2024, 21. [CrossRef]
- Jo, J.; Folz, J.; Gonzalez, M.E.; Paoli, A.; Eido, A.; Salfi, E.; Tekula, S.; Ando, S.; Caruso, R.; Kleer, C.G.; et al. Personalized Oncology by In Vivo Chemical Imaging: Photoacoustic Mapping of Tumor Oxygen Predicts Radiotherapy Efficacy. ACS Nano 2023, 17, 4396-4403. [CrossRef]
- Folz, J.; Wasserman, J.H.; Jo, J.; Wang, X.; Kopelman, R. Photoacoustic Chemical Imaging Sodium Nano-Sensor Utilizing a Solvatochromic Dye Transducer for In Vivo Application. Biosensors (Basel) 2023, 13. [CrossRef]
- Lee, C.H.; Folz, J.; Tan, J.W.Y.; Jo, J.; Wang, X.; Kopelman, R. Chemical Imaging in Vivo: Photoacoustic-Based 4-Dimensional Chemical Analysis. Anal Chem 2019, 91, 2561-2569. [CrossRef]
- Jo, J.; Lee, C.H.; Folz, J.; Tan, J.W.Y.; Wang, X.; Kopelman, R. In Vivo Photoacoustic Lifetime Based Oxygen Imaging with Tumor Targeted G2 Polyacrylamide Nanosonophores. ACS Nano 2019, 13, 14024-14032. [CrossRef]
- Tan, J.W.Y.; Folz, J.; Kopelman, R.; Wang, X. In vivo photoacoustic potassium imaging of the tumor microenvironment. Biomed Opt Express 2020, 11, 3507-3522. [CrossRef]
- Xie, X.; Szilagyi, I.; Zhai, J.; Wang, L.; Bakker, E. Ion-Selective Optical Nanosensors Based on Solvatochromic Dyes of Different Lipophilicity: From Bulk Partitioning to Interfacial Accumulation. ACS Sens 2016, 1, 516-520. [CrossRef]
- Tran, M.N.; Kleer, C.G. Matricellular CCN6 (WISP3) protein: a tumor suppressor for mammary metaplastic carcinomas. J Cell Commun Signal 2018, 12, 13-19. [CrossRef]
- Huang, W.; Martin, E.E.; Burman, B.; Gonzalez, M.E.; Kleer, C.G. The matricellular protein CCN6 (WISP3) decreases Notch1 and suppresses breast cancer initiating cells. Oncotarget 2016, 7, 25180-25193. [CrossRef]
- Pal, A.; Huang, W.; Toy, K.A.; Kleer, C.G. CCN6 knockdown disrupts acinar organization of breast cells in three-dimensional cultures through up-regulation of type III TGF-beta receptor. Neoplasia 2012, 14, 1067-1074. [CrossRef]
- Huang, W.; Pal, A.; Kleer, C.G. On how CCN6 suppresses breast cancer growth and invasion. J Cell Commun Signal 2012, 6, 5-10. [CrossRef]
- Lorenzatti, G.; Huang, W.; Kleer, C.G. The emerging role of CCN6 in breast cancer invasion. Cellscience 2009, 6, 146-157.
- Kleer, C.G.; Zhang, Y.; Merajver, S.D. CCN6 (WISP3) as a new regulator of the epithelial phenotype in breast cancer. Cells Tissues Organs 2007, 185, 95-99. [CrossRef]
- Zhang, Y.; Pan, Q.; Zhong, H.; Merajver, S.D.; Kleer, C.G. Inhibition of CCN6 (WISP3) expression promotes neoplastic progression and enhances the effects of insulin-like growth factor-1 on breast epithelial cells. Breast Cancer Res 2005, 7, R1080-1089. [CrossRef]
- Gonzalez, M.E.; Brophy, B.; Eido, A.; Leonetti, A.E.; Djomehri, S.I.; Augimeri, G.; Carruthers, N.J.; Cavalcante, R.G.; Giordano, F.; Ando, S.; et al. CCN6 Suppresses Metaplastic Breast Carcinoma by Antagonizing Wnt/beta-Catenin Signaling to Inhibit EZH2-Driven EMT. Cancer Res 2024, 84, 3235-3249. [CrossRef]
- Haslene-Hox, H.; Oveland, E.; Berg, K.C.; Kolmannskog, O.; Woie, K.; Salvesen, H.B.; Tenstad, O.; Wiig, H. A new method for isolation of interstitial fluid from human solid tumors applied to proteomic analysis of ovarian carcinoma tissue. PLoS One 2011, 6, e19217. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



