Submitted:
10 June 2025
Posted:
11 June 2025
Read the latest preprint version here
Abstract
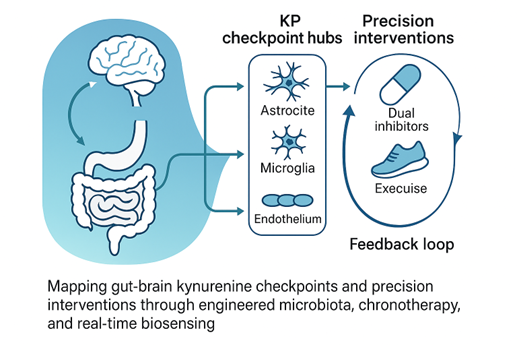
Keywords:
1. Introduction
2. Microbiota-Driven Modulation of Indoleamine-2,3-Dioxygenase-1 (IDO1) and Tryptophan-2,3-Dioxygenase (TDO2) Signaling
2.1. Literature Review: Microbial Metabolites as Modulators of Intestinal Integrity and Systemic Disease
2.2. Research Gaps: Gaps in Dosing Strategies, Longitudinal Efficacy, and Mechanistic Insights
2.3. Time-Stamped Isotope-Tracing in Gnotobiotic Mice Can Tag Flux Through Indoleamine-2,3-Dioxygenase-1 (IDO1) Versus Tryptophan-2,3-Dioxygenase (TDO2)
2.4. Single-Cell Proteomics in Intestinal Organoids Could Reveal Which Epithelial or Immune Subsets Sense Each “Metabokine.”
2.5. Synthetic Consortia with Inducible Kyn Operons Would Let Us Dial Metabolite Output like a Volume Knob
3. Kynurenine (KYN) Metabolic Pathway “Checkpoints” in the Brain’s Cellular Grid
3.1. Literature Review: Mapping Kynurenine (KYN) Dynamics Across Neurovascular and Immune Landscapes
3.2. Research Gaps: Mapping, Monitoring, and Modulating Kynurenine (KYN) Checkpoints Across Systems
3.3. CRISPRi “Zip-Codes” Delivered by Adeno-Associated Virus (AAV) Can Silence Kynurenine 3-Monooxygenase (KMO) or Kynureninase (KYNU) only in Perivascular Endothelium and Watch Downstream Glutamatergic Sync Crash—or Not
3.4. Light-Addressable Riboswitches Could Let Us Pulse KP Enzymes in Astrocytes and Read Real-Time Calcium Waves
4. Sex and the Circadian City: Hidden Modifiers
4.1. Literature Review: Circadian Misalignment, Mood Vulnerability, and Emerging Chronotherapeutics
4.2. Research Gaps: Timing, Sex, and Biomarker Integration for Precision Kynurenine (KYN) Intervention
4.3. Multi-Time-Point Plasma Kynurenine (KYN) Profiles Stratified by Sex and Hormonal Phase
4.4. Wearable Light-Exposure + Metabolite Logging to See If Circadian Misalignment Exaggerates the Quinolinic Spike
4.5. Adaptive Trial Designs that Randomize Dose-Timing Rather Than Just Dose Size
5. Microbiota Engineering as a Precision Switch
5.1. Literature Review: Microbiota-Targeted Strategies for Modulating Mood and Inflammation
5.2. Research Gaps: Live Biotherapeutic Products Against Multi-Drug Resistant (MDR) Enteric Pathogens: Research Gaps
5.3. Designer Strains with Kill-Switches and Inducible Kynurenine Aminotransferase (KAT) Expression
5.4. Encapsulated “Post-Biotics” (e.g., Stabilized KYNA) to Bypass Colonization
5.5. Cloud-Linked Stool Metabolomics Dashboards to Guide Weekly Probiotic Titration
| Key Clues | Implication | |
|---|---|---|
| 1. Multispecies probiotics shorten gut transit time and shift microbiota in constipation meta-analyses, with response stratified by baseline diet and Lactobacillus colonization predictors like cheese and n-3 fatty acid intake | Personalize probiotic therapy based on individual dietary patterns and colonization potential | |
| 2. Probiotics modulate clock gene expression and the gut–lung axis, suggesting time of day and symptom phase windows for optimized dosing | Incorporate circadian timing in probiotic administration to enhance therapeutic efficacy | |
| 3. Large-scale genome scans map foodborne lactic acid bacteria, offering candidates to seed personalized probiotic consortia | Develop customized probiotic blends from identified foodborne strains for targeted microbiome modulation |
| Next Steps | Purpose | |
|---|---|---|
| 1. Build a reference library of weekly stool metabolomes from diverse cohorts on fixed probiotic regimens | Establish a baseline for microbiome metabolite variation under controlled probiotic interventions | |
| 2. Train adaptive Bayesian models to recommend titration when normalized SCFA or indole scores drift beyond control limits | Enable dynamic adjustment of probiotic dosing based on real-time metabolic biomarkers | |
| 3. Integrate wearable-captured feeding rhythms to optimize capsule timing | Personalize probiotic administration schedules to individual feeding patterns | |
| 4. Run N-of-1 cross-over trials to benchmark dashboard-guided titration against static dosing | Validate the superiority of adaptive, dashboard-driven interventions over conventional static dosing | |
| 5. Build a reference library of weekly stool metabolomes from diverse cohorts on fixed probiotic regimens | Establish a baseline for microbiome metabolite variation under controlled probiotic interventions |
6. Intervention 2.0: Dual Inhibitors, Exercise, and Real-Time Biosensing
6.1. Literature Review. Dual Inhibition and KP Modulation
6.2. Research Gaps: Adaptive Dose-Timing and Real-Time Monitoring
6.3. Phase-Ib “Smart Protocols”: Micro-Dosed Dual Inhibitors Guided by Saliva KYNA Sensors
6.4. AI-Driven Feedback Loops That Auto-Adjust Evening Treadmill Sessions or Probiotic Cocktails Based on Morning KYN/TRP Slope.AI-Driven KYN/TRP Feedback Loops
7. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAV | adeno-associated virus |
| AD | Alzheimer’s disease |
| AhR | aryhydorcarbon receptor |
| AI | artificial intelligence |
| BBB | blood–brain barrier |
| CM | Circadian misalignment |
| COVID-19 | coronavirus disease 2019 |
| CRISPR | clustered regularly interspaced short palindromic repeats |
| CRISPRi | clustered regularly interspaced short palindromic repeats interference |
| IDO1 | indoleamine-2,3-dioxygenase 1 |
| KMO | kynurenine 3-monooxygenase |
| KYN | kynurenine |
| KYNU | kynureninase |
| KYNA | kynurenic acid |
| KAT | kynurenine aminotransferase |
| LBPs | biotherapeutic products |
| LC-MS | liquid chromatography–mass spectrometry |
| LD | Linear dichroism |
| NAD | nicotinamide adenine dinucleotide |
| QUIN | quinolinic acid |
| TDO2 | tryptophan-2,3-dioxygenase-2 |
| TLA | Three letter acronym |
| Trp | tryptophan |
| ZIM3 | zinc finger protein 3 |
References
- Sun, M.; Ma, N.; He, T.; Johnston, L.J.; Ma, X. Tryptophan (Trp) modulates gut homeostasis via aryl hydrocarbon receptor (AhR). Critical reviews in food science and nutrition 2020, 60, 1760-1768.
- Roth, W.; Zadeh, K.; Vekariya, R.; Ge, Y.; Mohamadzadeh, M. Tryptophan metabolism and gut-brain homeostasis. International journal of molecular sciences 2021, 22, 2973.
- Su, X.; Gao, Y.; Yang, R. Gut microbiota-derived tryptophan metabolites maintain gut and systemic homeostasis. Cells 2022, 11, 2296.
- Hyland, N.P.; Cavanaugh, C.R.; Hornby, P.J. Emerging effects of tryptophan pathway metabolites and intestinal microbiota on metabolism and intestinal function. Amino Acids 2022, 54, 57-70.
- Figueiredo Godoy, A.C.; Frota, F.F.; Araújo, L.P.; Valenti, V.E.; Pereira, E.; Detregiachi, C.R.P.; Galhardi, C.M.; Caracio, F.C.; Haber, R.S.A.; Fornari Laurindo, L.; et al. Neuroinflammation and Natural Antidepressants: Balancing Fire with Flora. Biomedicines 2025, 13. [CrossRef]
- Chen, X.; Xu, D.; Yu, J.; Song, X.-J.; Li, X.; Cui, Y.-L. Tryptophan metabolism disorder-triggered diseases, mechanisms, and therapeutic strategies: a scientometric review. Nutrients 2024, 16, 3380.
- Vécsei, L.; Szalárdy, L.; Fülöp, F.; Toldi, J. Kynurenines in the CNS: recent advances and new questions. Nat Rev Drug Discov 2013, 12, 64-82. [CrossRef]
- Tanaka, M.; Vécsei, L. A Decade of Dedication: Pioneering Perspectives on Neurological Diseases and Mental Illnesses. Biomedicines 2024, 12. [CrossRef]
- Tanaka, M.; Battaglia, S.; Giménez-Llort, L.; Chen, C.; Hepsomali, P.; Avenanti, A.; Vécsei, L. Innovation at the Intersection: Emerging Translational Research in Neurology and Psychiatry. Cells 2024, 13. [CrossRef]
- Tanaka, M.; Battaglia, S.; Liloia, D. Navigating Neurodegeneration: Integrating Biomarkers, Neuroinflammation, and Imaging in Parkinson's, Alzheimer's, and Motor Neuron Disorders. Biomedicines 2025, 13. [CrossRef]
- de Lima, E.P.; Laurindo, L.F.; Catharin, V.C.S.; Direito, R.; Tanaka, M.; Jasmin Santos German, I.; Lamas, C.B.; Guiguer, E.L.; Araújo, A.C.; Fiorini, A.M.R.; et al. Polyphenols, Alkaloids, and Terpenoids Against Neurodegeneration: Evaluating the Neuroprotective Effects of Phytocompounds Through a Comprehensive Review of the Current Evidence. Metabolites 2025, 15. [CrossRef]
- Barbalho, S.M.; Leme Boaro, B.; da Silva Camarinha Oliveira, J.; Patočka, J.; Barbalho Lamas, C.; Tanaka, M.; Laurindo, L.F. Molecular Mechanisms Underlying Neuroinflammation Intervention with Medicinal Plants: A Critical and Narrative Review of the Current Literature. Pharmaceuticals (Basel) 2025, 18. [CrossRef]
- Pagotto, G.L.O.; Santos, L.; Osman, N.; Lamas, C.B.; Laurindo, L.F.; Pomini, K.T.; Guissoni, L.M.; Lima, E.P.; Goulart, R.A.; Catharin, V.; et al. Ginkgo biloba: A Leaf of Hope in the Fight against Alzheimer's Dementia: Clinical Trial Systematic Review. Antioxidants (Basel) 2024, 13. [CrossRef]
- Valotto Neto, L.J.; Reverete de Araujo, M.; Moretti Junior, R.C.; Mendes Machado, N.; Joshi, R.K.; Dos Santos Buglio, D.; Barbalho Lamas, C.; Direito, R.; Fornari Laurindo, L.; Tanaka, M.; et al. Investigating the Neuroprotective and Cognitive-Enhancing Effects of Bacopa monnieri: A Systematic Review Focused on Inflammation, Oxidative Stress, Mitochondrial Dysfunction, and Apoptosis. Antioxidants (Basel) 2024, 13. [CrossRef]
- Xue, C.; Li, G.; Zheng, Q.; Gu, X.; Shi, Q.; Su, Y.; Chu, Q.; Yuan, X.; Bao, Z.; Lu, J. Tryptophan metabolism in health and disease. Cell Metabolism 2023, 35, 1304-1326.
- Miyamoto, K.; Sujino, T.; Kanai, T. The tryptophan metabolic pathway of the microbiome and host cells in health and disease. International immunology 2024, 36, 601-616.
- Cellini, B.; Zelante, T.; Dindo, M.; Bellet, M.M.; Renga, G.; Romani, L.; Costantini, C. Pyridoxal 5′-phosphate-dependent enzymes at the crossroads of host–microbe tryptophan metabolism. International Journal of Molecular Sciences 2020, 21, 5823.
- Tanaka, M.; Szabó, Á.; Vécsei, L. Redefining Roles: A Paradigm Shift in Tryptophan-Kynurenine Metabolism for Innovative Clinical Applications. Int J Mol Sci 2024, 25. [CrossRef]
- Vandereyken, K.; Sifrim, A.; Thienpont, B.; Voet, T. Methods and applications for single-cell and spatial multi-omics. Nature Reviews Genetics 2023, 24, 494-515.
- Bressan, D.; Battistoni, G.; Hannon, G.J. The dawn of spatial omics. Science 2023, 381, eabq4964.
- Hrovatin, K.; Fischer, D.S.; Theis, F.J. Toward modeling metabolic state from single-cell transcriptomics. Molecular metabolism 2022, 57, 101396.
- Lok, R.; Qian, J.; Chellappa, S.L. Sex differences in sleep, circadian rhythms, and metabolism: implications for precision medicine. Sleep medicine reviews 2024, 101926.
- Lévi, F.A.; Okyar, A.; Hadadi, E.; Innominato, P.F.; Ballesta, A. Circadian regulation of drug responses: toward sex-specific and personalized chronotherapy. Annual review of pharmacology and toxicology 2024, 64, 89-114.
- Weger, M.; Weger, B.D.; Gachon, F. Understanding circadian dynamics: current progress and future directions for chronobiology in drug discovery. Expert Opinion on Drug Discovery 2023, 18, 893-901.
- Liang, Y.; Truong, T.; Saxton, A.J.; Boekweg, H.; Payne, S.H.; Van Ry, P.M.; Kelly, R.T. HyperSCP: combining isotopic and isobaric labeling for higher throughput single-cell proteomics. Analytical chemistry 2023, 95, 8020-8027.
- Fernández-García, J.; Altea-Manzano, P.; Pranzini, E.; Fendt, S.-M. Stable isotopes for tracing mammalian-cell metabolism in vivo. Trends in biochemical sciences 2020, 45, 185-201.
- Rottinghaus, A.G.; Ferreiro, A.; Fishbein, S.R.; Dantas, G.; Moon, T.S. Genetically stable CRISPR-based kill switches for engineered microbes. Nature communications 2022, 13, 672.
- Zhang, Y.; Hu, Y.; Jiang, N.; Yetisen, A.K. Wearable artificial intelligence biosensor networks. Biosensors and Bioelectronics 2023, 219, 114825.
- Qiu, Y.; Li, M. A Bayesian Dynamic Model-Based Adaptive Design for Oncology Dose Optimization in Phase I/II Clinical Trials. Pharm Stat 2025, 24, e2451. [CrossRef]
- Joisten, N.; Kummerhoff, F.; Koliamitra, C.; Schenk, A.; Walzik, D.; Hardt, L.; Knoop, A.; Thevis, M.; Kiesl, D.; Metcalfe, A.J.; et al. Exercise and the Kynurenine pathway: Current state of knowledge and results from a randomized cross-over study comparing acute effects of endurance and resistance training. Exerc Immunol Rev 2020, 26, 24-42.
- Jankovskaja, S.; Engblom, J.; Rezeli, M.; Marko-Varga, G.; Ruzgas, T.; Björklund, S. Non-invasive skin sampling of tryptophan/kynurenine ratio in vitro towards a skin cancer biomarker. Sci Rep 2021, 11, 678. [CrossRef]
- Cordaillat-Simmons, M.; Rouanet, A.; Pot, B. Live biotherapeutic products: the importance of a defined regulatory framework. Exp Mol Med 2020, 52, 1397-1406. [CrossRef]
- Cerqueira, F.P.; Jesus, A.M.C.; Cotrim, M.D. Adaptive Design: A Review of the Technical, Statistical, and Regulatory Aspects of Implementation in a Clinical Trial. Ther Innov Regul Sci 2020, 54, 246-258. [CrossRef]
- Hou, Y.; Li, J.; Ying, S. Tryptophan Metabolism and Gut Microbiota: A Novel Regulatory Axis Integrating the Microbiome, Immunity, and Cancer. Metabolites 2023, 13. [CrossRef]
- Su, X.; Gao, Y.; Yang, R. Gut Microbiota-Derived Tryptophan Metabolites Maintain Gut and Systemic Homeostasis. Cells 2022, 11. [CrossRef]
- Gao, J.; Xu, K.; Liu, H.; Liu, G.; Bai, M.; Peng, C.; Li, T.; Yin, Y. Impact of the Gut Microbiota on Intestinal Immunity Mediated by Tryptophan Metabolism. Front Cell Infect Microbiol 2018, 8, 13. [CrossRef]
- Agus, A.; Planchais, J.; Sokol, H. Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease. Cell Host Microbe 2018, 23, 716-724. [CrossRef]
- Mostafavi Abdolmaleky, H.; Zhou, J.R. Gut Microbiota Dysbiosis, Oxidative Stress, Inflammation, and Epigenetic Alterations in Metabolic Diseases. Antioxidants (Basel) 2024, 13. [CrossRef]
- Peña-Durán, E.; García-Galindo, J.J.; López-Murillo, L.D.; Huerta-Huerta, A.; Balleza-Alejandri, L.R.; Beltrán-Ramírez, A.; Anaya-Ambriz, E.J.; Suárez-Rico, D.O. Microbiota and Inflammatory Markers: A Review of Their Interplay, Clinical Implications, and Metabolic Disorders. Int J Mol Sci 2025, 26. [CrossRef]
- Hand, T.W.; Vujkovic-Cvijin, I.; Ridaura, V.K.; Belkaid, Y. Linking the Microbiota, Chronic Disease, and the Immune System. Trends Endocrinol Metab 2016, 27, 831-843. [CrossRef]
- Potrykus, M.; Czaja-Stolc, S.; Stankiewicz, M.; Kaska, Ł.; Małgorzewicz, S. Intestinal Microbiota as a Contributor to Chronic Inflammation and Its Potential Modifications. Nutrients 2021, 13. [CrossRef]
- Rizzetto, L.; Fava, F.; Tuohy, K.M.; Selmi, C. Connecting the immune system, systemic chronic inflammation and the gut microbiome: The role of sex. J Autoimmun 2018, 92, 12-34. [CrossRef]
- Wang, Q.; Liu, D.; Song, P.; Zou, M.H. Tryptophan-kynurenine pathway is dysregulated in inflammation, and immune activation. Front Biosci (Landmark Ed) 2015, 20, 1116-1143. [CrossRef]
- Scott, S.A.; Fu, J.; Chang, P.V. Microbial tryptophan metabolites regulate gut barrier function via the aryl hydrocarbon receptor. Proc Natl Acad Sci U S A 2020, 117, 19376-19387. [CrossRef]
- Gasaly, N.; de Vos, P.; Hermoso, M.A. Impact of Bacterial Metabolites on Gut Barrier Function and Host Immunity: A Focus on Bacterial Metabolism and Its Relevance for Intestinal Inflammation. Front Immunol 2021, 12, 658354. [CrossRef]
- Mayengbam, S.; Chleilat, F.; Reimer, R.A. Dietary Vitamin B6 Deficiency Impairs Gut Microbiota and Host and Microbial Metabolites in Rats. Biomedicines 2020, 8. [CrossRef]
- Hou, C.; Shi, H.; Xiao, J.; Song, X.; Luo, Z.; Ma, X.; Shi, L.; Wei, H.; Li, J. Pomegranate Juice Supplemented with Inulin Modulates Gut Microbiota and Promotes the Production of Microbiota-Associated Metabolites in Overweight/Obese Individuals: A Randomized, Double-Blind, Placebo-Controlled Trial. J Agric Food Chem 2024, 72, 14663-14677. [CrossRef]
- Hiel, S.; Gianfrancesco, M.A.; Rodriguez, J.; Portheault, D.; Leyrolle, Q.; Bindels, L.B.; Gomes da Silveira Cauduro, C.; Mulders, M.; Zamariola, G.; Azzi, A.S.; et al. Link between gut microbiota and health outcomes in inulin -treated obese patients: Lessons from the Food4Gut multicenter randomized placebo-controlled trial. Clin Nutr 2020, 39, 3618-3628. [CrossRef]
- Zhang, J.; Zhu, S.; Ma, N.; Johnston, L.J.; Wu, C.; Ma, X. Metabolites of microbiota response to tryptophan and intestinal mucosal immunity: A therapeutic target to control intestinal inflammation. Med Res Rev 2021, 41, 1061-1088. [CrossRef]
- Gupta, S.K.; Vyavahare, S.; Duchesne Blanes, I.L.; Berger, F.; Isales, C.; Fulzele, S. Microbiota-derived tryptophan metabolism: Impacts on health, aging, and disease. Exp Gerontol 2023, 183, 112319. [CrossRef]
- Meghani, S.; Frishkopf, M.; Park, T.; Montgomery, C.L.; Norris, C.; Papathanassoglou, E. Music-based interventions and theoretical mechanisms in post-ICU survivors: A critical narrative synthesis. Intensive Crit Care Nurs 2025, 86, 103777. [CrossRef]
- Dimitriadis, T.; Della Porta, D.; Perschl, J.; Evers, A.W.M.; Magee, W.L.; Schaefer, R.S. Motivation and music interventions in adults: A systematic review. Neuropsychol Rehabil 2024, 34, 649-678. [CrossRef]
- Kuuse, A.K.; Paulander, A.S.; Eulau, L. Characteristics and impacts of live music interventions on health and wellbeing for children, families, and health care professionals in paediatric hospitals: a scoping review. Int J Qual Stud Health Well-being 2023, 18, 2180859. [CrossRef]
- Pakdeesatitwara, N.; Clark, I.; Tamplin, J. A mixed-studies systematic review of self-administered music interventions (SAMIs) for psychological wellbeing in people with chronic health conditions: Meta-analysis and narrative summary. Patient Educ Couns 2024, 118, 108006. [CrossRef]
- Chang, E.X.; Brooker, J.; Hiscock, R.; O'Callaghan, C. Music-based intervention impacts for people with eating disorders: A narrative synthesis systematic review. J Music Ther 2023, 60, 202-231. [CrossRef]
- Aslamkhan, A.G.; Xu, Q.; Loughlin, A.; Vu, H.; Pacchione, S.; Bhatt, B.; Garfinkel, I.; Styring, T.G.; LaFranco-Scheuch, L.; Pearson, K.; et al. Characterization of indoleamine-2,3-dioxygenase 1, tryptophan-2,3-dioxygenase, and Ido1/Tdo2 knockout mice. Toxicol Appl Pharmacol 2020, 406, 115216. [CrossRef]
- Tijono, S.M.; Palmer, B.D.; Tomek, P.; Flanagan, J.U.; Henare, K.; Gamage, S.; Braun, L.; Ching, L.M. Evaluation of Novel Inhibitors of Tryptophan Dioxygenases for Enzyme and Species Selectivity Using Engineered Tumour Cell Lines Expressing Either Murine or Human IDO1 or TDO2. Pharmaceuticals (Basel) 2022, 15. [CrossRef]
- Naing, A.; Eder, J.P.; Piha-Paul, S.A.; Gimmi, C.; Hussey, E.; Zhang, S.; Hildebrand, V.; Hosagrahara, V.; Habermehl, C.; Moisan, J.; et al. Preclinical investigations and a first-in-human phase I trial of M4112, the first dual inhibitor of indoleamine 2,3-dioxygenase 1 and tryptophan 2,3-dioxygenase 2, in patients with advanced solid tumors. J Immunother Cancer 2020, 8. [CrossRef]
- Juhász, L.; Spisák, K.; Szolnoki, B.Z.; Nászai, A.; Szabó, Á.; Rutai, A.; Tallósy, S.P.; Szabó, A.; Toldi, J.; Tanaka, M.; et al. The Power Struggle: Kynurenine Pathway Enzyme Knockouts and Brain Mitochondrial Respiration. J Neurochem 2025, 169, e70075. [CrossRef]
- Szabó, Á.; Galla, Z.; Spekker, E.; Szűcs, M.; Martos, D.; Takeda, K.; Ozaki, K.; Inoue, H.; Yamamoto, S.; Toldi, J.; et al. Oxidative and Excitatory Neurotoxic Stresses in CRISPR/Cas9-Induced Kynurenine Aminotransferase Knockout Mice: A Novel Model for Despair-Based Depression and Post-Traumatic Stress Disorder. Front Biosci (Landmark Ed) 2025, 30, 25706. [CrossRef]
- Tanaka, M.; Szatmári, I.; Vécsei, L. Quinoline Quest: Kynurenic Acid Strategies for Next-Generation Therapeutics via Rational Drug Design. Pharmaceuticals (Basel) 2025, 18. [CrossRef]
- Capochiani de Iudicibus, R.; Tomek, P.; Palmer, B.D.; Tijono, S.M.; Flanagan, J.U.; Ching, L.M. Parallel discovery of selective and dual inhibitors of tryptophan dioxygenases IDO1 and TDO2 with a newly-modified enzymatic assay. Bioorg Med Chem 2021, 39, 116160. [CrossRef]
- MacCannell, A.D.; Roberts, L.D. Metabokines in the regulation of systemic energy metabolism. Curr Opin Pharmacol 2022, 67, 102286. [CrossRef]
- Choi, M.J.; Jung, S.B.; Chang, J.Y.; Shong, M. Cellular and Intercellular Homeostasis in Adipose Tissue with Mitochondria-Specific Stress. Endocrinol Metab (Seoul) 2021, 36, 1-11. [CrossRef]
- Whitehead, A.; Krause, F.N.; Moran, A.; MacCannell, A.D.V.; Scragg, J.L.; McNally, B.D.; Boateng, E.; Murfitt, S.A.; Virtue, S.; Wright, J.; et al. Brown and beige adipose tissue regulate systemic metabolism through a metabolite interorgan signaling axis. Nat Commun 2021, 12, 1905. [CrossRef]
- Qian, X.; Chen, L.; Sui, Y.; Chen, C.; Zhang, W.; Zhou, J.; Dong, W.; Jiang, M.; Xin, F.; Ochsenreither, K. Biotechnological potential and applications of microbial consortia. Biotechnol Adv 2020, 40, 107500. [CrossRef]
- Hampton, T. From the Literature. Circulation 2020, 142, 1491-1493.
- Benninghaus, L.; Schwardmann, L.S.; Jilg, T.; Wendisch, V.F. Establishment of synthetic microbial consortia with Corynebacterium glutamicum and Pseudomonas putida: Design, construction, and application to production of γ-glutamylisopropylamide and l-theanine. Microb Biotechnol 2024, 17, e14400. [CrossRef]
- Peng, H.; Darlington, A.P.S.; South, E.J.; Chen, H.H.; Jiang, W.; Ledesma-Amaro, R. A molecular toolkit of cross-feeding strains for engineering synthetic yeast communities. Nat Microbiol 2024, 9, 848-863. [CrossRef]
- Gasparek, M.; Steel, H.; Papachristodoulou, A. Deciphering mechanisms of production of natural compounds using inducer-producer microbial consortia. Biotechnol Adv 2023, 64, 108117. [CrossRef]
- Alnahhas, R.N.; Sadeghpour, M.; Chen, Y.; Frey, A.A.; Ott, W.; Josić, K.; Bennett, M.R. Majority sensing in synthetic microbial consortia. Nat Commun 2020, 11, 3659. [CrossRef]
- Bodapati, S.; Daley, T.P.; Lin, X.; Zou, J.; Qi, L.S. A benchmark of algorithms for the analysis of pooled CRISPR screens. Genome Biol 2020, 21, 62. [CrossRef]
- Li, Y.; Tang, J.; Jiang, J.; Chen, Z. Metabolic checkpoints and novel approaches for immunotherapy against cancer. International Journal of Cancer 2022, 150, 195-207.
- Yang, S.; Yuan, Z.; Zhu, Y.; Liang, C.; Chen, Z.; Zhang, J.; Leng, L. Multi-omics analysis reveals GAPDH posttranscriptional regulation of IFN-γ and PHGDH as a metabolic checkpoint of microglia polarization. Brain, behavior, and immunity 2024, 117, 155-166.
- Fang, J.; Lu, Y.; Zheng, J.; Jiang, X.; Shen, H.; Shang, X.; Lu, Y.; Fu, P. Exploring the crosstalk between endothelial cells, immune cells, and immune checkpoints in the tumor microenvironment: new insights and therapeutic implications. Cell death & disease 2023, 14, 586.
- Liu, M.; Hong, L.; Sridhar, S.; Jaynes, P.; Tipgomut, C.; Poon, L.; De Mel, S.; Lee, J.S.X.; Ng, S.-B.; Tan, C.L. Spatial-Resolved Transcriptomics Reveals Immune Landscape Variations in Primary Central Nervous System Lymphoma (PCNSL) and Diffuse Large B-Cell Lymphoma (DLBCL). Blood 2024, 144, 3004.
- Tanaka, M.; Vécsei, L. Revolutionizing our understanding of Parkinson's disease: Dr. Heinz Reichmann's pioneering research and future research direction. J Neural Transm (Vienna) 2024, 131, 1367-1387. [CrossRef]
- Abdel-Rahman, S.A.; Gabr, M. Small molecule immunomodulators as next-generation therapeutics for glioblastoma. Cancers 2024, 16, 435.
- de Lima, E.P.; Tanaka, M.; Lamas, C.B.; Quesada, K.; Detregiachi, C.R.P.; Araújo, A.C.; Guiguer, E.L.; Catharin, V.; de Castro, M.V.M.; Junior, E.B.; et al. Vascular Impairment, Muscle Atrophy, and Cognitive Decline: Critical Age-Related Conditions. Biomedicines 2024, 12. [CrossRef]
- Barbalho, S.M.; Laurindo, L.F.; de Oliveira Zanuso, B.; da Silva, R.M.S.; Gallerani Caglioni, L.; Nunes Junqueira de Moraes, V.B.F.; Fornari Laurindo, L.; Dogani Rodrigues, V.; da Silva Camarinha Oliveira, J.; Beluce, M.E.; et al. AdipoRon's Impact on Alzheimer's Disease-A Systematic Review and Meta-Analysis. Int J Mol Sci 2025, 26. [CrossRef]
- Kim, J.E.; Patel, K.; Jackson, C.M. The potential for immune checkpoint modulators in cerebrovascular injury and inflammation. Expert opinion on therapeutic targets 2021, 25, 101-113.
- Battaglia, S.; Fazio, C.D.; Borgomaneri, S.; Avenanti, A. Cortisol Imbalance and Fear Learning in PTSD: Therapeutic Approaches to Control Abnormal Fear Responses. Curr Neuropharmacol 2025, 23, 835-846. [CrossRef]
- Battaglia, S.; Di Fazio, C.; Mazzà, M.; Tamietto, M.; Avenanti, A. Targeting Human Glucocorticoid Receptors in Fear Learning: A Multiscale Integrated Approach to Study Functional Connectivity. Int J Mol Sci 2024, 25. [CrossRef]
- Tortora, F.; Hadipour, A.L.; Battaglia, S.; Falzone, A.; Avenanti, A.; Vicario, C.M. The Role of Serotonin in Fear Learning and Memory: A Systematic Review of Human Studies. Brain Sci 2023, 13. [CrossRef]
- Tanaka, M.; Tóth, F.; Polyák, H.; Szabó, Á.; Mándi, Y.; Vécsei, L. Immune influencers in action: metabolites and enzymes of the tryptophan-kynurenine metabolic pathway. Biomedicines 2021, 9, 734.
- Fujigaki, H.; Yamamoto, Y.; Saito, K. L-Tryptophan-kynurenine pathway enzymes are therapeutic target for neuropsychiatric diseases: Focus on cell type differences. Neuropharmacology 2017, 112, 264-274.
- Cervenka, I.; Agudelo, L.Z.; Ruas, J.L. Kynurenines: Tryptophan’s metabolites in exercise, inflammation, and mental health. Science 2017, 357, eaaf9794.
- Klaessens, S.; Stroobant, V.; De Plaen, E.; Van den Eynde, B.J. Systemic tryptophan homeostasis. Frontiers in molecular biosciences 2022, 9, 897929.
- Cheong, J.E.; Sun, L. Targeting the IDO1/TDO2–KYN–AhR pathway for cancer immunotherapy–challenges and opportunities. Trends in pharmacological sciences 2018, 39, 307-325.
- Stone, T.W.; Williams, R.O. Tryptophan metabolism as a ‘reflex’feature of neuroimmune communication: sensor and effector functions for the indoleamine-2, 3-dioxygenase kynurenine pathway. Journal of Neurochemistry 2024, 168, 3333-3357.
- Labadie, B.W.; Bao, R.; Luke, J.J. Reimagining IDO pathway inhibition in cancer immunotherapy via downstream focus on the tryptophan–kynurenine–aryl hydrocarbon axis. Clinical Cancer Research 2019, 25, 1462-1471.
- Tanaka, M.; Vécsei, L. From Lab to Life: Exploring Cutting-Edge Models for Neurological and Psychiatric Disorders. Biomedicines 2024, 12. [CrossRef]
- Platten, M.; von Knebel Doeberitz, N.; Oezen, I.; Wick, W.; Ochs, K. Cancer immunotherapy by targeting IDO1/TDO and their downstream effectors. Frontiers in immunology 2015, 5, 673.
- Triplett, T.A.; Garrison, K.C.; Marshall, N.; Donkor, M.; Blazeck, J.; Lamb, C.; Qerqez, A.; Dekker, J.D.; Tanno, Y.; Lu, W.-C. Reversal of indoleamine 2, 3-dioxygenase–mediated cancer immune suppression by systemic kynurenine depletion with a therapeutic enzyme. Nature biotechnology 2018, 36, 758-764.
- Stone, T.W.; Williams, R.O. Modulation of T cells by tryptophan metabolites in the kynurenine pathway. Trends in Pharmacological Sciences 2023, 44, 442-456.
- Williams, H.L.; Frei, A.L.; Koessler, T.; Berger, M.D.; Dawson, H.; Michielin, O.; Zlobec, I. The current landscape of spatial biomarkers for prediction of response to immune checkpoint inhibition. NPJ precision oncology 2024, 8, 178.
- Ala, M. The footprint of kynurenine pathway in every cancer: a new target for chemotherapy. European Journal of Pharmacology 2021, 896, 173921.
- Liu, M.; Wang, X.; Wang, L.; Ma, X.; Gong, Z.; Zhang, S.; Li, Y. Targeting the IDO1 pathway in cancer: from bench to bedside. J Hematol Oncol 2018, 11, 100. [CrossRef]
- Battaglia, S.; Nazzi, C.; Di Fazio, C.; Borgomaneri, S. The role of pre-supplementary motor cortex in action control with emotional stimuli: A repetitive transcranial magnetic stimulation study. Ann N Y Acad Sci 2024, 1536, 151-166. [CrossRef]
- Battaglia, S.; Nazzi, C.; Fullana, M.A.; di Pellegrino, G.; Borgomaneri, S. 'Nip it in the bud': Low-frequency rTMS of the prefrontal cortex disrupts threat memory consolidation in humans. Behav Res Ther 2024, 178, 104548. [CrossRef]
- Krolak, T.; Chan, K.Y.; Kaplan, L.; Huang, Q.; Wu, J.; Zheng, Q.; Kozareva, V.; Beddow, T.; Tobey, I.G.; Pacouret, S.; et al. A High-Efficiency AAV for Endothelial Cell Transduction Throughout the Central Nervous System. Nat Cardiovasc Res 2022, 1, 389-400. [CrossRef]
- Chen, X.; Wolfe, D.A.; Bindu, D.S.; Zhang, M.; Taskin, N.; Goertsen, D.; Shay, T.F.; Sullivan, E.E.; Huang, S.F.; Ravindra Kumar, S.; et al. Functional gene delivery to and across brain vasculature of systemic AAVs with endothelial-specific tropism in rodents and broad tropism in primates. Nat Commun 2023, 14, 3345. [CrossRef]
- Bolanos-Palmieri, P.; Kotb, A.; Schenk, H.; Bähre, H.; Schroder, P.; Schiffer, M. MO006 CHANGES IN THE KYNURENINE PATHWAY LEAD TO ALTERATIONS IN NAD BALANCE AND BIOENERGETICS PARAMETERS IN GLOMERULAR CELLS IN VITRO AND CONTRIBUTE TO PROTEINURIA IN A ZEBRAFISH MODEL. Nephrology Dialysis Transplantation 2021, 36, gfab079. 002.
- Kampmann, M. CRISPRi and CRISPRa Screens in Mammalian Cells for Precision Biology and Medicine. ACS Chem Biol 2018, 13, 406-416. [CrossRef]
- Battaglia, S.; Avenanti, A.; Vécsei, L.; Tanaka, M. Neurodegeneration in Cognitive Impairment and Mood Disorders for Experimental, Clinical and Translational Neuropsychiatry. Biomedicines 2024, 12. [CrossRef]
- Späth, M.R.; Hoyer-Allo, K.J.R.; Seufert, L.; Höhne, M.; Lucas, C.; Bock, T.; Isermann, L.; Brodesser, S.; Lackmann, J.W.; Kiefer, K.; et al. Organ Protection by Caloric Restriction Depends on Activation of the De Novo NAD+ Synthesis Pathway. J Am Soc Nephrol 2023, 34, 772-792. [CrossRef]
- Flickinger, K.M.; Cantor, J.R. Uncovering the Conditionally Essential Roles of NAD Kinases in Human Cells. The FASEB Journal 2022, 36.
- Walsh, S.; Gardner, L.; Deiters, A.; Williams, G.J. Intracellular light-activation of riboswitch activity. Chembiochem 2014, 15, 1346-1351. [CrossRef]
- Borrachero-Conejo, A.I.; Adams, W.R.; Saracino, E.; Mola, M.G.; Wang, M.; Posati, T.; Formaggio, F.; De Bellis, M.; Frigeri, A.; Caprini, M.; et al. Stimulation of water and calcium dynamics in astrocytes with pulsed infrared light. Faseb j 2020, 34, 6539-6553. [CrossRef]
- Spennato, D.; Leone, J.; Gundhardt, C.; Varnavski, O.; Fabbri, R.; Caprini, M.; Zamboni, R.; Benfenati, V.; Goodson, T., 3rd. Investigations of Astrocyte Calcium Signaling and Imaging with Classical and Nonclassical Light. J Phys Chem B 2024, 128, 7966-7977. [CrossRef]
- Qiao, L.; Niu, L.; Wang, M.; Wang, Z.; Kong, D.; Yu, G.; Ye, H. A sensitive red/far-red photoswitch for controllable gene therapy in mouse models of metabolic diseases. Nature Communications 2024, 15, 10310.
- Walton, J.C.; Bumgarner, J.R.; Nelson, R.J. Sex Differences in Circadian Rhythms. Cold Spring Harb Perspect Biol 2022, 14. [CrossRef]
- Tanaka, M.; Tuka, B.; Vécsei, L. Navigating the Neurobiology of Migraine: From Pathways to Potential Therapies. Cells 2024, 13. [CrossRef]
- Minbay, M.; Khan, A.; Ghasemi, A.R.; Ingram, K.K.; Ay, A.A. Sex-specific associations between circadian-related genes and depression in UK Biobank participants highlight links to glucose metabolism, inflammation and neuroplasticity pathways. Psychiatry Res 2024, 337, 115948. [CrossRef]
- Abo, S.M.; Layton, A.T. Modeling the circadian regulation of the immune system: Sexually dimorphic effects of shift work. PLoS computational biology 2021, 17, e1008514.
- Logan, R.W.; Xue, X.; Ketchesin, K.D.; Hoffman, G.; Roussos, P.; Tseng, G.; McClung, C.A.; Seney, M.L. Sex Differences in Molecular Rhythms in the Human Cortex. Biol Psychiatry 2022, 91, 152-162. [CrossRef]
- Liloia, D.; Zamfira, D.A.; Tanaka, M.; Manuello, J.; Crocetta, A.; Keller, R.; Cozzolino, M.; Duca, S.; Cauda, F.; Costa, T. Disentangling the role of gray matter volume and concentration in autism spectrum disorder: A meta-analytic investigation of 25 years of voxel-based morphometry research. Neurosci Biobehav Rev 2024, 164, 105791. [CrossRef]
- Bailey, M.; Silver, R. Sex differences in circadian timing systems: implications for disease. Front Neuroendocrinol 2014, 35, 111-139. [CrossRef]
- Tanaka, M. Beyond the boundaries: Transitioning from categorical to dimensional paradigms in mental health diagnostics. Adv Clin Exp Med 2024, 33, 1295-1301. [CrossRef]
- Wu, F.; Langer, P.; Shim, J.; Fleisch, E.; Barata, F. Comparative Efficacy of Commercial Wearables for Circadian Rhythm Home Monitoring From Activity, Heart Rate, and Core Body Temperature. IEEE J Biomed Health Inform 2025, 29, 900-908. [CrossRef]
- Zisapel, N. New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. Br J Pharmacol 2018, 175, 3190-3199. [CrossRef]
- Lightman, S.L.; Conway-Campbell, B.L. Circadian and ultradian rhythms: Clinical implications. J Intern Med 2024, 296, 121-138. [CrossRef]
- Steele, T.A.; St Louis, E.K.; Videnovic, A.; Auger, R.R. Circadian Rhythm Sleep-Wake Disorders: a Contemporary Review of Neurobiology, Treatment, and Dysregulation in Neurodegenerative Disease. Neurotherapeutics 2021, 18, 53-74. [CrossRef]
- Tordjman, S.; Chokron, S.; Delorme, R.; Charrier, A.; Bellissant, E.; Jaafari, N.; Fougerou, C. Melatonin: Pharmacology, Functions and Therapeutic Benefits. Curr Neuropharmacol 2017, 15, 434-443. [CrossRef]
- Lévi, F.A.; Okyar, A.; Hadadi, E.; Innominato, P.F.; Ballesta, A. Circadian Regulation of Drug Responses: Toward Sex-Specific and Personalized Chronotherapy. Annu Rev Pharmacol Toxicol 2024, 64, 89-114. [CrossRef]
- Karaboué, A.; Innominato, P.F.; Wreglesworth, N.I.; Duchemann, B.; Adam, R.; Lévi, F.A. Why does circadian timing of administration matter for immune checkpoint inhibitors' efficacy? Br J Cancer 2024, 131, 783-796. [CrossRef]
- Ohdo, S.; Koyanagi, S.; Matsunaga, N. Chronopharmacological strategies focused on chrono-drug discovery. Pharmacol Ther 2019, 202, 72-90. [CrossRef]
- Wang, C.; Zeng, Q.; Gül, Z.M.; Wang, S.; Pick, R.; Cheng, P.; Bill, R.; Wu, Y.; Naulaerts, S.; Barnoud, C.; et al. Circadian tumor infiltration and function of CD8(+) T cells dictate immunotherapy efficacy. Cell 2024, 187, 2690-2702.e2617. [CrossRef]
- Ye, Y.; Xiang, Y.; Ozguc, F.M.; Kim, Y.; Liu, C.J.; Park, P.K.; Hu, Q.; Diao, L.; Lou, Y.; Lin, C.; et al. The Genomic Landscape and Pharmacogenomic Interactions of Clock Genes in Cancer Chronotherapy. Cell Syst 2018, 6, 314-328.e312. [CrossRef]
- Ephraim, A.; Leatheng, C.; Lu, Z.E.; Xia, X.; Pirruccello, J.P.; Marotti, J.D.; MacKenzie, T.; Chamberlin, M.D. Association of plasma kynurenine (KYN) with plasma osteopontin (OPN) in patients with locally invasive breast cancer. 2024.
- Chang, K.H.; Cheng, M.L.; Tang, H.Y.; Huang, C.Y.; Wu, Y.R.; Chen, C.M. Alternations of Metabolic Profile and Kynurenine Metabolism in the Plasma of Parkinson's Disease. Mol Neurobiol 2018, 55, 6319-6328. [CrossRef]
- Chantrapanichkul, P.; Stevenson, M.O.; Suppakitjanusant, P.; Goodman, M.; Tangpricha, V. SERUM HORMONE CONCENTRATIONS IN TRANSGENDER INDIVIDUALS RECEIVING GENDER-AFFIRMING HORMONE THERAPY: A LONGITUDINAL RETROSPECTIVE COHORT STUDY. Endocr Pract 2021, 27, 27-33. [CrossRef]
- Kervezee, L.; Cermakian, N.; Boivin, D.B. Individual metabolomic signatures of circadian misalignment during simulated night shifts in humans. PLoS Biol 2019, 17, e3000303. [CrossRef]
- Skene, D.J.; Skornyakov, E.; Chowdhury, N.R.; Gajula, R.P.; Middleton, B.; Satterfield, B.C.; Porter, K.I.; Van Dongen, H.P.A.; Gaddameedhi, S. Separation of circadian- and behavior-driven metabolite rhythms in humans provides a window on peripheral oscillators and metabolism. Proc Natl Acad Sci U S A 2018, 115, 7825-7830. [CrossRef]
- Whittaker, D.S.; Akhmetova, L.; Carlin, D.; Romero, H.; Welsh, D.K.; Colwell, C.S.; Desplats, P. Circadian modulation by time-restricted feeding rescues brain pathology and improves memory in mouse models of Alzheimer's disease. Cell Metab 2023, 35, 1704-1721.e1706. [CrossRef]
- Emens, J.S.; Burgess, H.J. Effect of Light and Melatonin and Other Melatonin Receptor Agonists on Human Circadian Physiology. Sleep Med Clin 2015, 10, 435-453. [CrossRef]
- Chen, X.; He, R.; Chen, X.; Jiang, L.; Wang, F. Optimizing dose-schedule regimens with bayesian adaptive designs: opportunities and challenges. Front Pharmacol 2023, 14, 1261312. [CrossRef]
- Lu, M.; Yuan, Y.; Liu, S. A Bayesian pharmacokinetics integrated phase I-II design to optimize dose-schedule regimes. Biostatistics 2024, 26. [CrossRef]
- Ballesta, A.; Innominato, P.F.; Dallmann, R.; Rand, D.A.; Lévi, F.A. Systems Chronotherapeutics. Pharmacol Rev 2017, 69, 161-199. [CrossRef]
- Jiang, L.; Li, R.; Yan, F.; Yap, T.A.; Yuan, Y. Shotgun: A Bayesian seamless phase I-II design to accelerate the development of targeted therapies and immunotherapy. Contemp Clin Trials 2021, 104, 106338. [CrossRef]
- Bai, X.; Huang, Z.; Duraj-Thatte, A.M.; Ebert, M.P.; Zhang, F.; Burgermeister, E.; Liu, X.; Scott, B.M.; Li, G.; Zuo, T. Engineering the gut microbiome. Nature Reviews Bioengineering 2023, 1, 665-679.
- Sorbara, M.T.; Pamer, E.G. Microbiome-based therapeutics. Nat Rev Microbiol 2022, 20, 365-380. [CrossRef]
- Mousa, W.K.; Al Ali, A. The Gut Microbiome Advances Precision Medicine and Diagnostics for Inflammatory Bowel Diseases. Int J Mol Sci 2024, 25. [CrossRef]
- Dodd, D.; Spitzer, M.H.; Van Treuren, W.; Merrill, B.D.; Hryckowian, A.J.; Higginbottom, S.K.; Le, A.; Cowan, T.M.; Nolan, G.P.; Fischbach, M.A.; et al. A gut bacterial pathway metabolizes aromatic amino acids into nine circulating metabolites. Nature 2017, 551, 648-652. [CrossRef]
- Han, S.; Van Treuren, W.; Fischer, C.R.; Merrill, B.D.; DeFelice, B.C.; Sanchez, J.M.; Higginbottom, S.K.; Guthrie, L.; Fall, L.A.; Dodd, D.; et al. A metabolomics pipeline for the mechanistic interrogation of the gut microbiome. Nature 2021, 595, 415-420. [CrossRef]
- Nunes, Y.C.; Mendes, N.M.; Pereira de Lima, E.; Chehadi, A.C.; Lamas, C.B.; Haber, J.F.S.; Dos Santos Bueno, M.; Araújo, A.C.; Catharin, V.C.S.; Detregiachi, C.R.P.; et al. Curcumin: A Golden Approach to Healthy Aging: A Systematic Review of the Evidence. Nutrients 2024, 16. [CrossRef]
- Lamichhane, S.; Sen, P.; Dickens, A.M.; Orešič, M.; Bertram, H.C. Gut metabolome meets microbiome: A methodological perspective to understand the relationship between host and microbe. Methods 2018, 149, 3-12. [CrossRef]
- Guo, H.; Liu, X.; Chen, T.; Wang, X.; Zhang, X. Akkermansia muciniphila Improves Depressive-Like Symptoms by Modulating the Level of 5-HT Neurotransmitters in the Gut and Brain of Mice. Mol Neurobiol 2024, 61, 821-834. [CrossRef]
- Ding, Y.; Bu, F.; Chen, T.; Shi, G.; Yuan, X.; Feng, Z.; Duan, Z.; Wang, R.; Zhang, S.; Wang, Q.; et al. A next-generation probiotic: Akkermansia muciniphila ameliorates chronic stress-induced depressive-like behavior in mice by regulating gut microbiota and metabolites. Appl Microbiol Biotechnol 2021, 105, 8411-8426. [CrossRef]
- Liu, J.Y.; Lin, T.L.; Chiu, C.Y.; Hsieh, P.F.; Lin, Y.T.; Lai, L.Y.; Wang, J.T. Decolonization of carbapenem-resistant Klebsiella pneumoniae from the intestinal microbiota of model mice by phages targeting two surface structures. Front Microbiol 2022, 13, 877074. [CrossRef]
- Medlock, G.; Felix, C.; Alsharif, W.; Cornacchione, L.; Schinn, M.; Watson, A.; Bedard-Shurtleff, S.; Norman, J.; Faith, J.; Kuijper, E.J. 2521. VE707, a defined live biotherapeutic product for prevention of infection by multidrug-resistant gram-negative bacteria. In Proceedings of the Open Forum Infectious Diseases, 2023; p. ofad500. 2139.
- Tavoukjian, V. Faecal microbiota transplantation for the decolonization of antibiotic-resistant bacteria in the gut: a systematic review and meta-analysis. J Hosp Infect 2019, 102, 174-188. [CrossRef]
- Macareño-Castro, J.; Solano-Salazar, A.; Dong, L.T.; Mohiuddin, M.; Espinoza, J.L. Fecal microbiota transplantation for Carbapenem-Resistant Enterobacteriaceae: A systematic review. J Infect 2022, 84, 749-759. [CrossRef]
- Mortzfeld, B.M.; Palmer, J.D.; Bhattarai, S.K.; Dupre, H.L.; Mercado-Lubio, R.; Silby, M.W.; Bang, C.; McCormick, B.A.; Bucci, V. Microcin MccI47 selectively inhibits enteric bacteria and reduces carbapenem-resistant Klebsiella pneumoniae colonization in vivo when administered via an engineered live biotherapeutic. Gut Microbes 2022, 14, 2127633. [CrossRef]
- Osbelt, L.; Wende, M.; Almási, É.; Derksen, E.; Muthukumarasamy, U.; Lesker, T.R.; Galvez, E.J.C.; Pils, M.C.; Schalk, E.; Chhatwal, P.; et al. Klebsiella oxytoca causes colonization resistance against multidrug-resistant K. pneumoniae in the gut via cooperative carbohydrate competition. Cell Host Microbe 2021, 29, 1663-1679.e1667. [CrossRef]
- Agarwal, S.; Tiwari, P.; Deep, A.; Kidwai, S.; Gupta, S.; Thakur, K.G.; Singh, R. System-Wide Analysis Unravels the Differential Regulation and In Vivo Essentiality of Virulence-Associated Proteins B and C Toxin-Antitoxin Systems of Mycobacterium tuberculosis. J Infect Dis 2018, 217, 1809-1820. [CrossRef]
- Lin, M.; Kussell, E. Inferring bacterial recombination rates from large-scale sequencing datasets. Nat Methods 2019, 16, 199-204. [CrossRef]
- Martin, G.; Kolida, S.; Marchesi, J.R.; Want, E.; Sidaway, J.E.; Swann, J.R. In Vitro Modeling of Bile Acid Processing by the Human Fecal Microbiota. Front Microbiol 2018, 9, 1153. [CrossRef]
- Cheng, F.; Wu, A.; Liu, C.; Cao, X.; Wang, R.; Shu, X.; Wang, L.; Zhang, Y.; Xiang, H.; Li, M. The toxin-antitoxin RNA guards of CRISPR-Cas evolved high specificity through repeat degeneration. Nucleic Acids Res 2022, 50, 9442-9452. [CrossRef]
- Wiechert, J.; Gätgens, C.; Wirtz, A.; Frunzke, J. Inducible Expression Systems Based on Xenogeneic Silencing and Counter-Silencing and Design of a Metabolic Toggle Switch. ACS Synth Biol 2020, 9, 2023-2038. [CrossRef]
- Schwarz, S.; Gildemeister, D.; Hein, A.; Schröder, P.; Bachmann, J. Environmental fate and effects assessment of human pharmaceuticals: lessons learnt from regulatory data. Environmental Sciences Europe 2021, 33, 68.
- Richard, E.; Darracq, B.; Littner, E.; Vit, C.; Whiteway, C.; Bos, J.; Fournes, F.; Garriss, G.; Conte, V.; Lapaillerie, D.; et al. Cassette recombination dynamics within chromosomal integrons are regulated by toxin-antitoxin systems. Sci Adv 2024, 10, eadj3498. [CrossRef]
- Yu, L.; Sun, Q.; Hui, Y.; Seth, A.; Petrovsky, N.; Zhao, C.X. Microfluidic formation of core-shell alginate microparticles for protein encapsulation and controlled release. J Colloid Interface Sci 2019, 539, 497-503. [CrossRef]
- Omer, A.M.; Ahmed, M.S.; El-Subruiti, G.M.; Khalifa, R.E.; Eltaweil, A.S. pH-Sensitive Alginate/Carboxymethyl Chitosan/Aminated Chitosan Microcapsules for Efficient Encapsulation and Delivery of Diclofenac Sodium. Pharmaceutics 2021, 13. [CrossRef]
- George, M.; Abraham, T.E. Polyionic hydrocolloids for the intestinal delivery of protein drugs: alginate and chitosan--a review. J Control Release 2006, 114, 1-14. [CrossRef]
- Qu, Q.; Yang, A.; Wang, J.; Xie, M.; Zhang, X.; Huang, D.; Xiong, R.; Pei, D.; Huang, C. Responsive and biocompatible chitosan-phytate microparticles with various morphology for antibacterial activity based on gas-shearing microfluidics. J Colloid Interface Sci 2023, 649, 68-75. [CrossRef]
- Feng, R.; Wang, L.; Zhou, P.; Luo, Z.; Li, X.; Gao, L. Development of the pH responsive chitosan-alginate based microgel for encapsulation of Jughans regia L. polyphenols under simulated gastrointestinal digestion in vitro. Carbohydr Polym 2020, 250, 116917. [CrossRef]
- Liao, P.; Dai, S.; Lian, Z.; Tong, X.; Yang, S.; Chen, Y.; Qi, W.; Peng, X.; Wang, H.; Jiang, L. The Layered Encapsulation of Vitamin B(2) and β-Carotene in Multilayer Alginate/Chitosan Gel Microspheres: Improving the Bioaccessibility of Vitamin B(2) and β-Carotene. Foods 2021, 11. [CrossRef]
- Liu, X.; Liu, L.; Huang, F.; Meng, Y.; Chen, Y.; Wang, J.; Wang, S.; Luo, Y.; Li, J.; Liang, Y. pH-sensitive chitosan/sodium alginate/calcium chloride hydrogel beads for potential oral delivery of rice bran bioactive peptides. Food Chem 2025, 470, 142618. [CrossRef]
- Tian, Y.; Ran, H.; Wen, X.; Fu, G.; Zhou, X.; Liu, R.; Pan, T. Probiotics improve symptoms of patients with COVID-19 through gut-lung axis: a systematic review and meta-analysis. Front Nutr 2023, 10, 1179432. [CrossRef]
- Schaub, A.C.; Schneider, E.; Vazquez-Castellanos, J.F.; Schweinfurth, N.; Kettelhack, C.; Doll, J.P.K.; Yamanbaeva, G.; Mählmann, L.; Brand, S.; Beglinger, C.; et al. Clinical, gut microbial and neural effects of a probiotic add-on therapy in depressed patients: a randomized controlled trial. Transl Psychiatry 2022, 12, 227. [CrossRef]
- Horvath, A.; Habisch, H.; Prietl, B.; Pfeifer, V.; Balazs, I.; Kovacs, G.; Foris, V.; John, N.; Kleinschek, D.; Feldbacher, N.; et al. Alteration of the Gut-Lung Axis After Severe COVID-19 Infection and Modulation Through Probiotics: A Randomized, Controlled Pilot Study. Nutrients 2024, 16. [CrossRef]
- Xu, L.; Yang, C.S.; Liu, Y.; Zhang, X. Effective Regulation of Gut Microbiota With Probiotics and Prebiotics May Prevent or Alleviate COVID-19 Through the Gut-Lung Axis. Front Pharmacol 2022, 13, 895193. [CrossRef]
- Kazemi, A.; Noorbala, A.A.; Azam, K.; Eskandari, M.H.; Djafarian, K. Effect of probiotic and prebiotic vs placebo on psychological outcomes in patients with major depressive disorder: A randomized clinical trial. Clin Nutr 2019, 38, 522-528. [CrossRef]
- Rudzki, L.; Ostrowska, L.; Pawlak, D.; Małus, A.; Pawlak, K.; Waszkiewicz, N.; Szulc, A. Probiotic Lactobacillus Plantarum 299v decreases kynurenine concentration and improves cognitive functions in patients with major depression: A double-blind, randomized, placebo controlled study. Psychoneuroendocrinology 2019, 100, 213-222. [CrossRef]
- Platten, M.; Nollen, E.A.A.; Röhrig, U.F.; Fallarino, F.; Opitz, C.A. Tryptophan metabolism as a common therapeutic target in cancer, neurodegeneration and beyond. Nat Rev Drug Discov 2019, 18, 379-401. [CrossRef]
- Liang, H.; Li, T.; Fang, X.; Xing, Z.; Zhang, S.; Shi, L.; Li, W.; Guo, L.; Kuang, C.; Liu, H.; et al. IDO1/TDO dual inhibitor RY103 targets Kyn-AhR pathway and exhibits preclinical efficacy on pancreatic cancer. Cancer Lett 2021, 522, 32-43. [CrossRef]
- Agudelo, L.Z.; Ferreira, D.M.S.; Dadvar, S.; Cervenka, I.; Ketscher, L.; Izadi, M.; Zhengye, L.; Furrer, R.; Handschin, C.; Venckunas, T.; et al. Skeletal muscle PGC-1α1 reroutes kynurenine metabolism to increase energy efficiency and fatigue-resistance. Nat Commun 2019, 10, 2767. [CrossRef]
- Martos, D.; Lőrinczi, B.; Szatmári, I.; Vécsei, L.; Tanaka, M. The Impact of C-3 Side Chain Modifications on Kynurenic Acid: A Behavioral Analysis of Its Analogs in the Motor Domain. Int J Mol Sci 2024, 25. [CrossRef]
- Fan, Q.Z.; Zhou, J.; Zhu, Y.B.; He, L.J.; Miao, D.D.; Zhang, S.P.; Liu, X.P.; Zhang, C. Design, synthesis, and biological evaluation of a novel indoleamine 2,3-dioxigenase 1 (IDO1) and thioredoxin reductase (TrxR) dual inhibitor. Bioorg Chem 2020, 105, 104401. [CrossRef]
- Di Gregorio, F.; Steinhauser, M.; Maier, M.E.; Thayer, J.F.; Battaglia, S. Error-related cardiac deceleration: Functional interplay between error-related brain activity and autonomic nervous system in performance monitoring. Neurosci Biobehav Rev 2024, 157, 105542. [CrossRef]
- Nazzi, C.; Avenanti, A.; Battaglia, S. The Involvement of Antioxidants in Cognitive Decline and Neurodegeneration: Mens Sana in Corpore Sano. Antioxidants (Basel) 2024, 13. [CrossRef]
- Battaglia, S.; Nazzi, C.; Lonsdorf, T.B.; Thayer, J.F. Neuropsychobiology of fear-induced bradycardia in humans: progress and pitfalls. Mol Psychiatry 2024, 29, 3826-3840. [CrossRef]
- Heyes, M.P. Metabolism and neuropathologic significance of quinolinic acid and kynurenic acid. Biochemical Society Transactions 1993, 21, 83-89.
- Damerell, V.; Klaassen-Dekker, N.; Brezina, S.; Ose, J.; Ulvik, A.; van Roekel, E.H.; Holowatyj, A.N.; Baierl, A.; Böhm, J.; Bours, M.J.L.; et al. Circulating tryptophan-kynurenine pathway metabolites are associated with all-cause mortality among patients with stage I-III colorectal cancer. Int J Cancer 2025, 156, 552-565. [CrossRef]
- Chiu, L.C.; Tang, H.Y.; Fan, C.M.; Lo, C.J.; Hu, H.C.; Kao, K.C.; Cheng, M.L. Kynurenine Pathway of Tryptophan Metabolism Is Associated with Hospital Mortality in Patients with Acute Respiratory Distress Syndrome: A Prospective Cohort Study. Antioxidants (Basel) 2022, 11. [CrossRef]
- Hoong, C.W.S.; Chua, M.W.J. SGLT2 Inhibitors as Calorie Restriction Mimetics: Insights on Longevity Pathways and Age-Related Diseases. Endocrinology 2021, 162. [CrossRef]
- Palmer, B.F.; Clegg, D.J. Euglycemic Ketoacidosis as a Complication of SGLT2 Inhibitor Therapy. Clin J Am Soc Nephrol 2021, 16, 1284-1291. [CrossRef]
- Hoeflich, K.P.; Merchant, M.; Orr, C.; Chan, J.; Den Otter, D.; Berry, L.; Kasman, I.; Koeppen, H.; Rice, K.; Yang, N.Y.; et al. Intermittent administration of MEK inhibitor GDC-0973 plus PI3K inhibitor GDC-0941 triggers robust apoptosis and tumor growth inhibition. Cancer Res 2012, 72, 210-219. [CrossRef]
- Yu, M.; Chen, J.; Xu, Z.; Yang, B.; He, Q.; Luo, P.; Yan, H.; Yang, X. Development and safety of PI3K inhibitors in cancer. Arch Toxicol 2023, 97, 635-650. [CrossRef]
- Wu, T.; Zhang, C.; Lv, R.; Qin, Q.; Liu, N.; Yin, W.; Wang, R.; Sun, Y.; Wang, X.; Sun, Y.; et al. Design, synthesis, biological evaluation and pharmacophore model analysis of novel tetrahydropyrrolo[3,4-c]pyrazol derivatives as potential TRKs inhibitors. Eur J Med Chem 2021, 223, 113627. [CrossRef]
- Yan, W.; Lakkaniga, N.R.; Carlomagno, F.; Santoro, M.; McDonald, N.Q.; Lv, F.; Gunaganti, N.; Frett, B.; Li, H.Y. Insights into Current Tropomyosin Receptor Kinase (TRK) Inhibitors: Development and Clinical Application. J Med Chem 2019, 62, 1731-1760. [CrossRef]
- Altzerinakou, M.A.; Paoletti, X. An adaptive design for the identification of the optimal dose using joint modeling of continuous repeated biomarker measurements and time-to-toxicity in phase I/II clinical trials in oncology. Stat Methods Med Res 2020, 29, 508-521. [CrossRef]
- Pinsker, J.E.; Dassau, E.; Deshpande, S.; Raghinaru, D.; Buckingham, B.A.; Kudva, Y.C.; Laffel, L.M.; Levy, C.J.; Church, M.M.; Desrochers, H.; et al. Outpatient Randomized Crossover Comparison of Zone Model Predictive Control Automated Insulin Delivery with Weekly Data Driven Adaptation Versus Sensor-Augmented Pump: Results from the International Diabetes Closed-Loop Trial 4. Diabetes Technol Ther 2022, 24, 635-642. [CrossRef]
- Iasonos, A.; O'Quigley, J. Adaptive dose-finding studies: a review of model-guided phase I clinical trials. J Clin Oncol 2014, 32, 2505-2511. [CrossRef]
- Visser, M.M.; Charleer, S.; Fieuws, S.; De Block, C.; Hilbrands, R.; Van Huffel, L.; Maes, T.; Vanhaverbeke, G.; Dirinck, E.; Myngheer, N.; et al. Comparing real-time and intermittently scanned continuous glucose monitoring in adults with type 1 diabetes (ALERTT1): a 6-month, prospective, multicentre, randomised controlled trial. Lancet 2021, 397, 2275-2283. [CrossRef]
- Perry, M.W.D.; Abdulai, R.; Mogemark, M.; Petersen, J.; Thomas, M.J.; Valastro, B.; Westin Eriksson, A. Evolution of PI3Kγ and δ Inhibitors for Inflammatory and Autoimmune Diseases. J Med Chem 2019, 62, 4783-4814. [CrossRef]
- Bornaei, M.; Khajehsharifi, H.; Shahrokhian, S.; Sheydaei, O.; Zarnegarian, A. Differential pulse voltammetric quantitation of kynurenic acid in human plasma using carbon-paste electrode modified with metal-organic frameworks. Materials Chemistry and Physics 2023, 295, 127016.
- Beatty, G.L.; O'Dwyer, P.J.; Clark, J.; Shi, J.G.; Bowman, K.J.; Scherle, P.A.; Newton, R.C.; Schaub, R.; Maleski, J.; Leopold, L.; et al. First-in-Human Phase I Study of the Oral Inhibitor of Indoleamine 2,3-Dioxygenase-1 Epacadostat (INCB024360) in Patients with Advanced Solid Malignancies. Clin Cancer Res 2017, 23, 3269-3276. [CrossRef]
- Wang, S.; Liu, H.; Wu, D.; Wang, X. Temperature and pH dual-stimuli-responsive phase-change microcapsules for multipurpose applications in smart drug delivery. J Colloid Interface Sci 2021, 583, 470-486. [CrossRef]
- Lin, R.; Zhou, Y.; Yan, F.; Li, D.; Yuan, Y. BOIN12: Bayesian Optimal Interval Phase I/II Trial Design for Utility-Based Dose Finding in Immunotherapy and Targeted Therapies. JCO Precis Oncol 2020, 4. [CrossRef]
- Liu, D.; Xin, Z.; Guo, S.; Li, S.; Cheng, J.; Jiang, H. Blood and Salivary MicroRNAs for Diagnosis of Oral Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis. J Oral Maxillofac Surg 2021, 79, 1082.e1081-1082.e1013. [CrossRef]
- Zahran, F.; Ghalwash, D.; Shaker, O.; Al-Johani, K.; Scully, C. Salivary microRNAs in oral cancer. Oral Dis 2015, 21, 739-747. [CrossRef]
- Hare, S.M.; Adhikari, B.M.; Mo, C.; Chen, S.; Wijtenburg, S.A.; Seneviratne, C.; Kane-Gerard, S.; Sathyasaikumar, K.V.; Notarangelo, F.M.; Schwarcz, R.; et al. Tryptophan challenge in individuals with schizophrenia and healthy controls: acute effects on circulating kynurenine and kynurenic acid, cognition and cerebral blood flow. Neuropsychopharmacology 2023, 48, 1594-1601. [CrossRef]
- Huang, J.; Tong, J.; Zhang, P.; Zhou, Y.; Li, Y.; Tan, S.; Wang, Z.; Yang, F.; Kochunov, P.; Chiappelli, J.; et al. Elevated salivary kynurenic acid levels related to enlarged choroid plexus and severity of clinical phenotypes in treatment-resistant schizophrenia. Brain Behav Immun 2022, 106, 32-39. [CrossRef]
- Badawy, A.A. Tryptophan availability for kynurenine pathway metabolism across the life span: Control mechanisms and focus on aging, exercise, diet and nutritional supplements. Neuropharmacology 2017, 112, 248-263. [CrossRef]
- Tanaka, M. From Serendipity to Precision: Integrating AI, Multi-Omics, and Human-Specific Models for Personalized Neuropsychiatric Care. Biomedicines 2025, 13. [CrossRef]
| Challenge | Core Issue | Implication | ||
|---|---|---|---|---|
| 1. Causal Mapping (Conceptual Challenge) |
Despite thousands of disease associations, we lack a coherent causal framework linking microbiota, host enzymes (e.g., IDO1, TDO2), and metabolites (e.g., KYNA, QUIN) to physiological outcomes. | Limits precision design of interventions (e.g., probiotics, enzyme inhibitors, lifestyle prescriptions). | ||
| 2. Spatial Resolution (Anatomical Challenge) | Bulk assays obscure localized metabolic activity in specialized niches (e.g., astrocytes, microglia, BBB endothelial cells). | Demands a paradigm shift: from homogeneous pathways to cell-type-specific “switchboards” requiring targeted modulation. | ||
| 3. Temporal Dynamics (Chronobiological Challenge) | Trp–KYN flux oscillates with circadian rhythms and is modulated by sex hormones, but time-sensitive dosing is understudied. | Missing chronotherapeutic windows may reduce efficacy or increase toxicity of interventions. |
| Objective | Description | |
|---|---|---|
| 1. Map Spatial “Checkpoints” | Survey localized KYN metabolism niches in brain and periphery, detailing how enzyme activity in astrocytes, microglia, BBB, and peripheral hubs interfaces with immunity and circuitry. | |
| 2. Characterize Sex & Circadian Modifiers | Examine how sex hormones and circadian rhythms influence Trp–KYN flux, identifying periods tilting toward neurotoxicity or resilience to inform time- and sex-specific interventions. | |
| 3. Develop Microbiota-Based Precision Switches | Explore designer microbial consortia and encapsulated post-biotics to steer Trp flux, addressing manufacturing, safety, and regulatory considerations for clinical translation. | |
| 4. Outline Integrated “Intervention 2.0” Platform | Propose a closed-loop therapeutic framework combining dual enzyme inhibitors, structured exercise regimens, and AI-driven biosensing for adaptive modulation of the Trp–KYN axis. | |
| 5. Build Dynamic, Multiscale Predictive Models & Networks | Weave spatial, temporal, and microbiome data into predictive tools; foster collaborations among bench scientists, clinicians, data engineers, and regulators to guide personalized interventions. |
| Next Steps | Purpose | |
|---|---|---|
| 1. Build bar-coded AAV libraries to refine endothelial specificity | Enhance targeting precision | |
| 2. Validate knock-down efficiency and KYN metabolite flux in brain-slice co-culture | Confirm functional impact on Trp metabolism | |
| 3. Monitor glutamate dynamics with optogenetic reporters in vivo | Track real-time neurotransmitter changes | |
| 4. Assess effects on tumor infiltration and behavior in KMO-high breast-cancer metastasis models | Evaluate therapeutic impact on metastasis | |
| 5. Conduct parallel safety screens to chart NAD pools and mitochondrial stress in non-target tissues | Ensure systemic safety and minimize off-target effects |
| Key Concept | Mechanism | Application | Advantages | |||
|---|---|---|---|---|---|---|
| Riboswitches with Z-lock or Photocleavable Linker | Light-controlled conformational changes in RNA elements regulate gene expression. | Precise gating of target gene expression in living cells | High specificity; reversible; minimal background activation | |||
| Pulsed Light-Driven Calcium Oscillations in Astrocytes | Tunable light pulses induce calcium signaling cascades without causing photodamage. | Real-time monitoring of cellular metabolism and signaling | Non-invasive; tunable frequency/amplitude; low cytotoxicity |
| Research Gap | Description | |
|---|---|---|
| 1. Absence of Chrono-pharmacology Trials | No clinical studies have evaluated time-of-day effects for IDO1/TDO2 inhibitors, KMO inhibitors, or KAT activators. Optimal administration schedules remain unexplored, impeding evidence-based chrono-dosing strategies. | |
| 2. Inadequate Stratification by Circadian Phase and Sex | Trials and PK/PD analyses seldom disaggregate data by both circadian timing and biological sex. Female-specific pharmacokinetic profiles are largely unreported, obscuring mechanistic reasons for sex-dependent toxicity reductions (e.g., afternoon regimens in lymphoma). | |
| 3. Undefined Mechanistic Links between Clock Genes, Immune Oscillations, and KYN Enzyme Activity | The interactions among peripheral circadian regulators, immune cell rhythmicity, and drug metabolism—especially modulation by estrogen and glucocorticoids on daily fluctuations of KYN pathway enzymes—remain mechanistically uncharacterized. | |
| 4. Lack of Wearable-Derived Chronotype Integration in Trial Design | Personalized chrono-type metrics from wearable sensors are not incorporated into clinical protocols. Without individual rhythm profiling, dosing algorithms cannot be tailored to synchronize drug exposure with each patient’s endogenous rhythm. | |
| 5. Sex and Circadian Biases in Preclinical Models | Preclinical experiments predominantly use male rodents under fixed light–dark schedules, neglecting sex-dimorphic and circadian-variant responses. This undermines translational relevance for patients exhibiting divergent chrono-biological profiles. | |
| 6. Absence of Validated Real-Time Biomarkers Coupling KYN Dynamics to Efficacy | There is a shortage of dynamic biomarkers that track KYN metabolite oscillations in real time and correlate these fluctuations with therapeutic outcomes. This gap limits the implementation of adaptive dosing regimens based on metabolic feedback. |
| Next Steps | Purpose | |
|---|---|---|
| 1. Pilot a cross-over study where shift workers wear light and activity trackers and collect hourly capillary samples across two work cycles | Capture real-time physiological and circadian data in shift workers | |
| 2. Model QUIN dynamics versus lux-derived phase angle using mixed-effects chronobiology | Analyze the relationship between light exposure patterns and QUIN fluctuations | |
| 3. Overlay cortisol and melatonin rhythms to disentangle stress versus circadian effects | Separate stress-induced effects from circadian-driven changes in biomarkers | |
| 4. Test whether timed blue light blockers, melatonin, or time-restricted feeding blunt QUIN spikes | Evaluate interventions to mitigate QUIN elevations linked to circadian misalignment | |
| 5. Pilot a cross-over study where shift workers wear light and activity trackers and collect hourly capillary samples across two work cycles | Capture real-time physiological and circadian data in shift workers |
| Next Steps | Purpose | |
|---|---|---|
| 1. Simulate Bayesian hierarchical designs co-randomizing dose level and dosing hour, borrowing strength across adjacent time bins | Enhance efficiency and robustness in chrono-dose finding | |
| 2. Integrate wearable-captured chronotype to stratify randomization and inform priors | Personalize treatment timing based on individual circadian profiles | |
| 3. Embed rolling interim analyses that drop unfavorable time windows rather than doses | Optimize trial adaptation by focusing on optimal dosing windows | |
| 4. Pilot such designs in drugs with known chronotoxicities, using point-of-care melatonin or cortisol assays as safety triggers | Validate design feasibility and safety in chronotherapy-prone drugs | |
| 5. Simulate Bayesian hierarchical designs co-randomizing dose level and dosing hour, borrowing strength across adjacent time bins | Enhance efficiency and robustness in chrono-dose finding |
| Next Steps | Purpose | |
|---|---|---|
| 1. Unpredictable Engraftment and Lack of Comparative Trials | Engraftment remains erratic (e.g., VE303 only after antibiotic conditioning; VE707’s murine efficacy lacks human PK analogues). No head-to-head studies versus FMT)exist, so key efficacy drivers (bacteriocins, phages, niche competition) are unclear. | |
| 2. Scarce Durability and Mechanistic Data | Longitudinal sequencing hints at phage-mediated suppression post-FMT, but detailed mechanistic dissection of phage–bacteria–host interactions over time is missing, leaving durability of decolonization unpredictable. | |
| 3. Undercharacterized Safety and Horizontal Gene Transfer Risks | Potential for engineered strains (e.g., E. coli secreting microcins) to acquire resistance cassettes in vivo is insufficiently studied, so safety profiles and mitigation strategies for HGT remain undefined. |
|
| 4. Manufacturing and Quality Control Deficits | Current frameworks lag pharmaceutical standards: multi- LBPs lack verified batch-to-batch consistency in metabolite outputs and stability, hindering reproducibility and scale-up. | |
| 5. Absence of Adaptive Trial Designs Integrating Timing Factors | Trials rarely modulate dosing relative to antibiotic schedules, feeding rhythms, or bile acid fluctuations, despite evidence these factors gate colonization resistance; person-alized timing algorithms remain unexplored. | |
| 6. Fragmented Regulatory Pathways for Genetically Modified LBPs | Regulatory requirements differ across jurisdictions for engineered or defined consortia, deterring investment and delaying standardization; clear, harmonized guidelines are needed to accelerate safe, predictable, and durable applications. |
| Next Steps | Purpose | |
|---|---|---|
| 1. Screen GRAS-grade polymers for KYNA compatibility under accelerated aging | Identify stable encapsulation materials suitable for KYNA under storage conditions | |
| 2. Map release profiles in simulated gastrointestinal fluids and pig colonic explants | Characterize release dynamics in physiologically relevant gut environments | |
| 3. Employ near-infrared–triggered nanocapsules to test on-demand bursts during inflammation | Enable controlled KYNA release in response to inflammation using light-triggered mechanisms | |
| 4. Quantify systemic versus luminal KYNA using LC-MS in gnotobiotic mice, benchmarking against Bifidobacterium-produced levels | Evaluate bioavailability and distribution of KYNA compared to natural microbial production | |
| 5. Screen GRAS-grade polymers for KYNA compatibility under accelerated aging | Identify stable encapsulation materials suitable for KYNA under storage conditions |
| Key Gap Description | Implication | |
|---|---|---|
| 1. Adaptive randomization models that incorporate dosing clock time as a modifiable arm | Enables dynamic optimization of treatment timing to enhance therapeutic efficacy and reduce toxicity | |
| 2. Validated software bridges between CGM, lactate, or KYN sensors and electronic trial master files | Facilitates seamless integration of real-time biomarker data into clinical trial records, improving data fidelity and regulatory compliance | |
| 3. Safety rules for rapid dose time shifts in outpatient settings | Ensures patient safety during temporal treatment adjustments, especially outside controlled environments | |
| 4. Patient-reported outcome measures (PROMs) sensitive to circadian toxicity | Captures subjective patient experiences related to time-dependent side effects, enhancing the assessment of tolerability and quality of life |
| Immediate Next Steps | Objective | |
|---|---|---|
| 1. Run a crossover pharmacokinetic study comparing saliva, plasma, and tumor microdialysate KYNA after RY103 micro-dosing | Evaluate the relationship between KYNA levels in different biological matrices post micro-dosing | |
| 2. Calibrate the Bayesian control algorithm using simulated patient data | Optimize the Bayesian algorithm for real-time adaptive control of dosing schedules | |
| 3. Embed patient-reported fatigue and cognitive scores to test whether KYNA-targeted pacing improves tolerability compared to fixed BID regimens | Assess the clinical benefit of KYNA-guided dosing on patient fatigue and cognitive outcomes relative to standard dosing practices |
| Clues Supporting Feasibility | Objective | |
|---|---|---|
| 1. Clinical artificial intelligence operations (ClinAIOps) frameworks for continuous therapeutic monitoring in hypertension and diabetes | Demonstrate the capability of AI frameworks to adaptively manage complex biological feedback loops | |
| 2. Kinect- or sensor-driven treadmills that auto-adjust speed based on user position | Validate the use of real-time biomechanical data to modulate physical activity interventions | |
| 3. Murine and human studies showing tailored probiotic blends reduce intestinal inflammation and modulate TRP metabolism | Provide proof of principle that microbiome-based therapies can influence gut–brain biochemical pathways |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




