Submitted:
10 June 2025
Posted:
10 June 2025
You are already at the latest version
Abstract

Keywords:
Highlight
- ▪ Fifteen new charged triazoles with quaternized triazole cores were synthesized with high yields and confirmed structural integrity.
- ▪ Derivatives 9 and 14 exhibited nanomolar BChE inhibition, outperforming the reference drug donepezil.
- ▪ Compound 9 also showed potent anti-inflammatory activity by inhibiting TNF-α production in LPS-stimulated PBMCs.
- ▪ In silico predictions and molecular docking confirmed stable and specific enzyme binding with low mutagenic potential.
1. Introduction
2. Materials and Methods
2.1. General Remarks
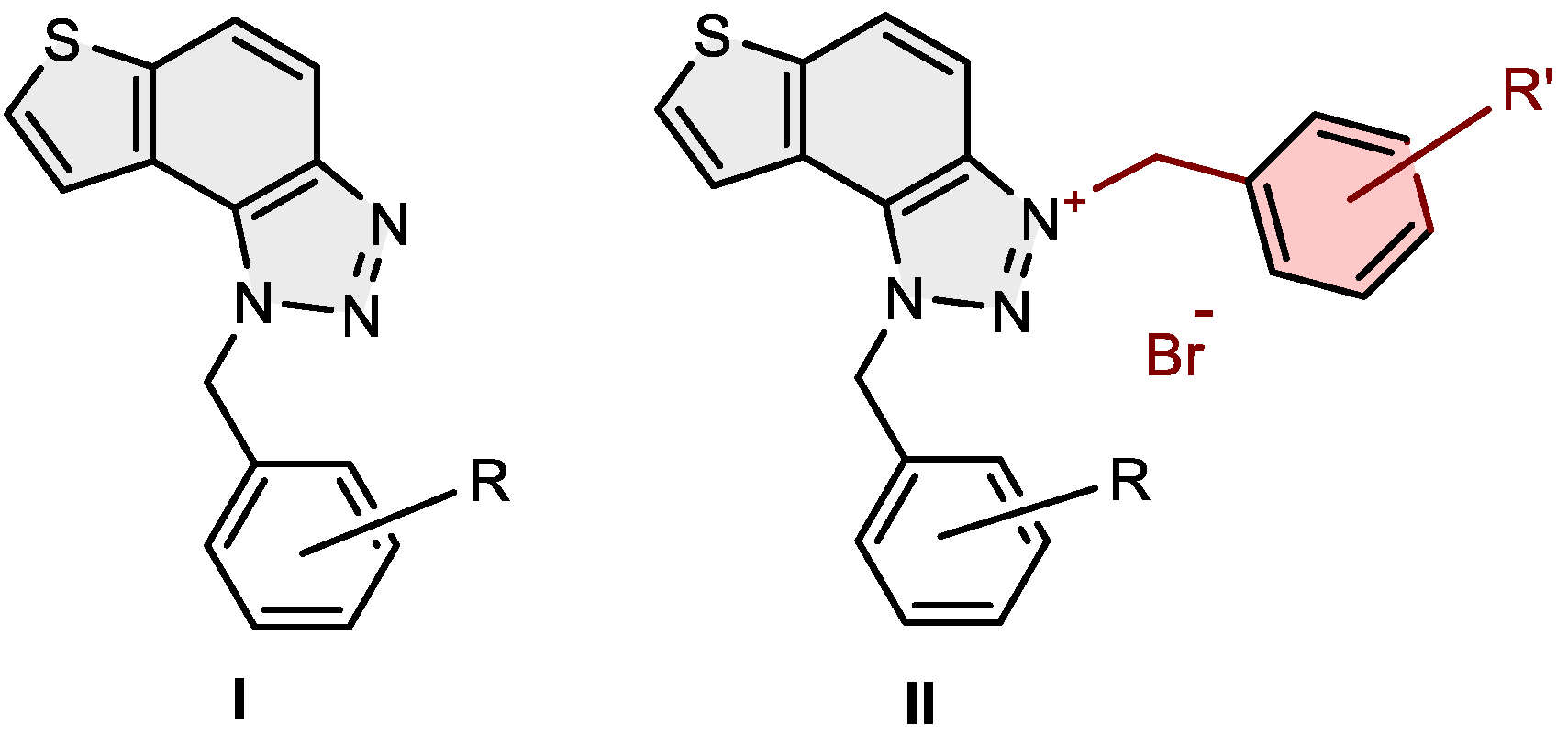
2.2. Synthesis of Bromide Salts 1–15
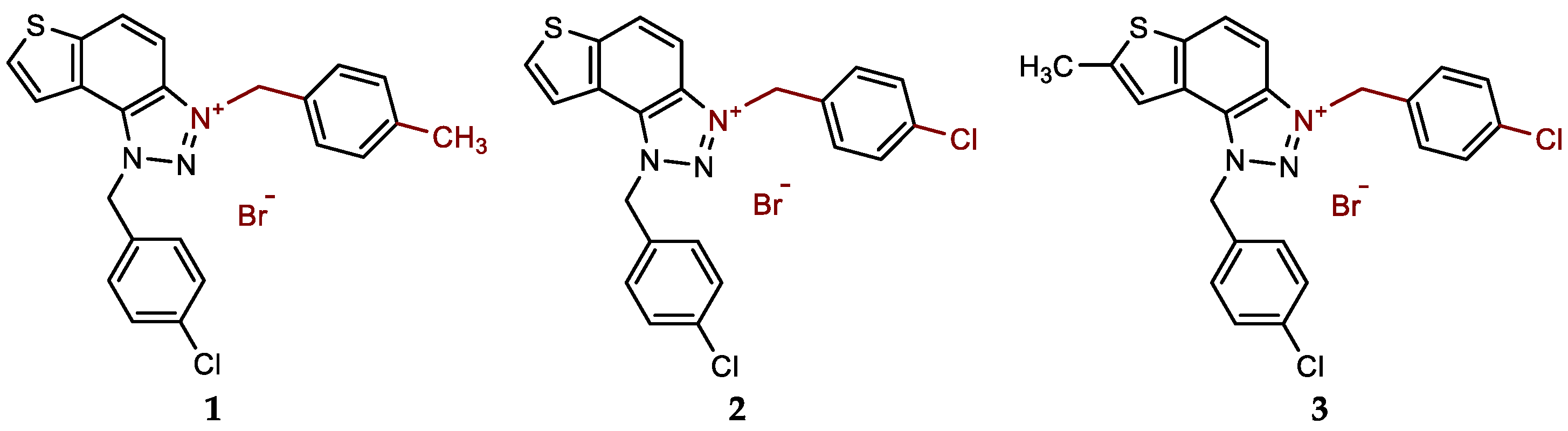
- 1-(4-chlorobenzyl)-3-(4-methylbenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (1): 9 mg (isolated 69%), white powder; m.p. 117-118 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.51 (d, J = 9.2 Hz, 1H), 8.23 (d, J = 5.5 Hz, 1H), 8.10–8.06 (m, 2H), 7.47–7.43 (m, 6H), 7.28 (d, J = 8.1 Hz, 2H), 6.50 (s, 2H), 6.25 (s, 2H), 2.36 (s, 3H); 13C NMR (CD3OD, 150 MHz) δ/ppm: 143.0, 139.7, 135.1, 134.0, 133.1, 130.9, 130.7, 129.7, 129.4, 129.2, 129.0, 128.5, 126.9, 123.1, 120.4, 107.9, 55.3, 55.2, 19.8; HRMS (ESI) (m/z) for C23H19ClN3S+ Br-: [M + H]+calcd = 404.0988, and [M + H]+measured = 404.0997.
- 1,3-bis(4-chlorobenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (2): 13 mg (isolated 96%), white powder; m.p. 108-110 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.54 (d, J = 9.2 Hz, 1H), 8.25 (d, J = 5.7 Hz, 1H), 8.13–8.08 (m, 2H), 7.57 (d, J = 8.6 Hz, 2H), 7.50–7.43 (m, 6H), 6.50 (s, 2H), 6.30 (s, 2H); 13C NMR (CD3OD, 150 MHz) δ/ppm: 143.1, 135.1, 135.40, 134.2, 133.2, 131.0, 130.7, 130.6, 130.3, 129.5, 129.4, 129.2, 127.2, 123.2, 120.4, 107.7, 55.4, 54.4.; HRMS (ESI) (m/z) for C22H16Cl2N3S+Br-: [M + H]+calcd = 424.0442, and [M + H]+measured = 424.0448.
- 1,3-bis(4-chlorobenzyl)-7-methyl-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (3): 10 mg (isolated 95%), white powder; m.p. 105-107 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.39 (d, J = 9.2 Hz, 1H), 8.00 (d, J = 9.2 Hz, 1H), 7.83 (s, 1H), 7.54 (d, J = 8.7 Hz, 2H), 7.49–7.43 (m, 6H), 6.45 (s, 2H), 6.26 (s, 2H), 2.77 (s, 3H); 13C NMR (CD3OD, 75 MHz) δ/ppm: 150.2, 131.7, 131.0, 130.6, 129.8, 129.4, 119.9, 108.0, 55.7, 56.7, 16.1 (several signals are missing due to the small quantity of the sample); HRMS (ESI) (m/z) for C23H18Cl2N3S+Br-: [M + H]+calcd = 438.0598, and [M + H]+measured = 438.0610.
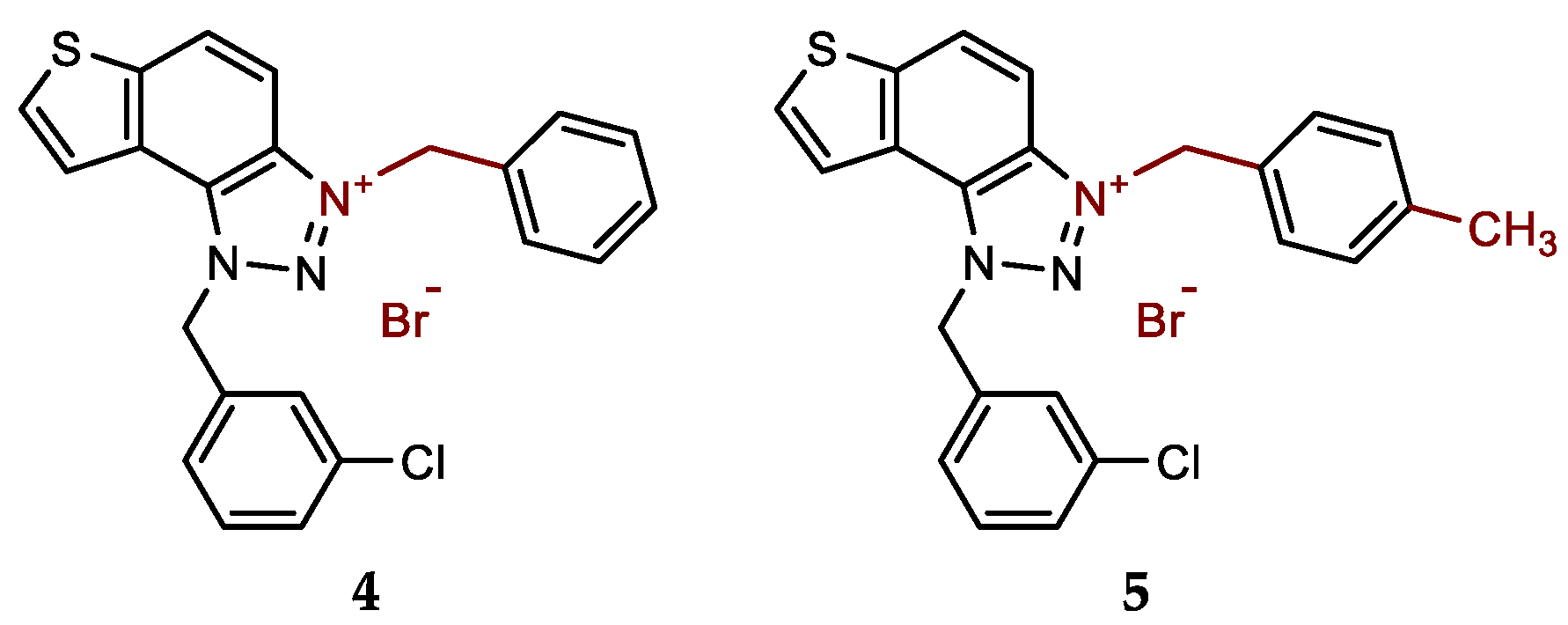
- 3-benzyl-1-(3-chlorobenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (4): 5 mg (isolated 27%), white powder; m.p. 119-120 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.53 (d, J = 9.1 Hz, 1H), 8.25 (d, J = 5.7 Hz, 1H), 8.13–8.09 (m, 3H), 7.57 (d, J = 7.8 Hz, 2H), 7.52 (s, 1H), 7.49–7.40 (m, 5H), 6.52 (s, 2H), 6.31 (s, 2H); 13C NMR (CD3OD, 150 MHz) δ/ppm: 143.1, 135.0, 134.1, 133.2, 132.1, 131.0, 130.6, 129.4, 129.3, 129.1, 128.5, 127.8, 127.0, 126.1, 123.1, 120.4, 107.8, 55.3; MS (ESI) (m/z, %) for C22H17ClN3S+Br-: [M + H]+ 391 (100), 250 (85).
- 1-(3-chlorobenzyl)-3-(4-methylbenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (5): 11 mg (isolated 57%), white powder; m.p. 104-105 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.51 (d, J = 9.2 Hz, 1H), 8.24 (d, J = 5.4 Hz, 1H), 8.12–8.08 (m, 2H), 7.53–7.36 (m, 6H), 7.28 (d, J = 8.0 Hz, 2H), 6.51 (s, 2H), 6.26 (s, 2H), 2.36 (s, 3H); 13C NMR (CD3OD, 150 MHz) δ/ppm: 144.5, 141.1, 136.4, 135.5, 135.4, 134.5, 132.4, 132.0, 131.1, 130.7, 130.0, 129.2, 128.4, 127.5, 124.5, 121.8, 109.3, 56.7, 56.6, 21.2; HRMS (ESI) (m/z) for C23H19ClN3S+Br-: [M + H]+calcd = 404.0988, and [M + H]+measured = 404.0998.
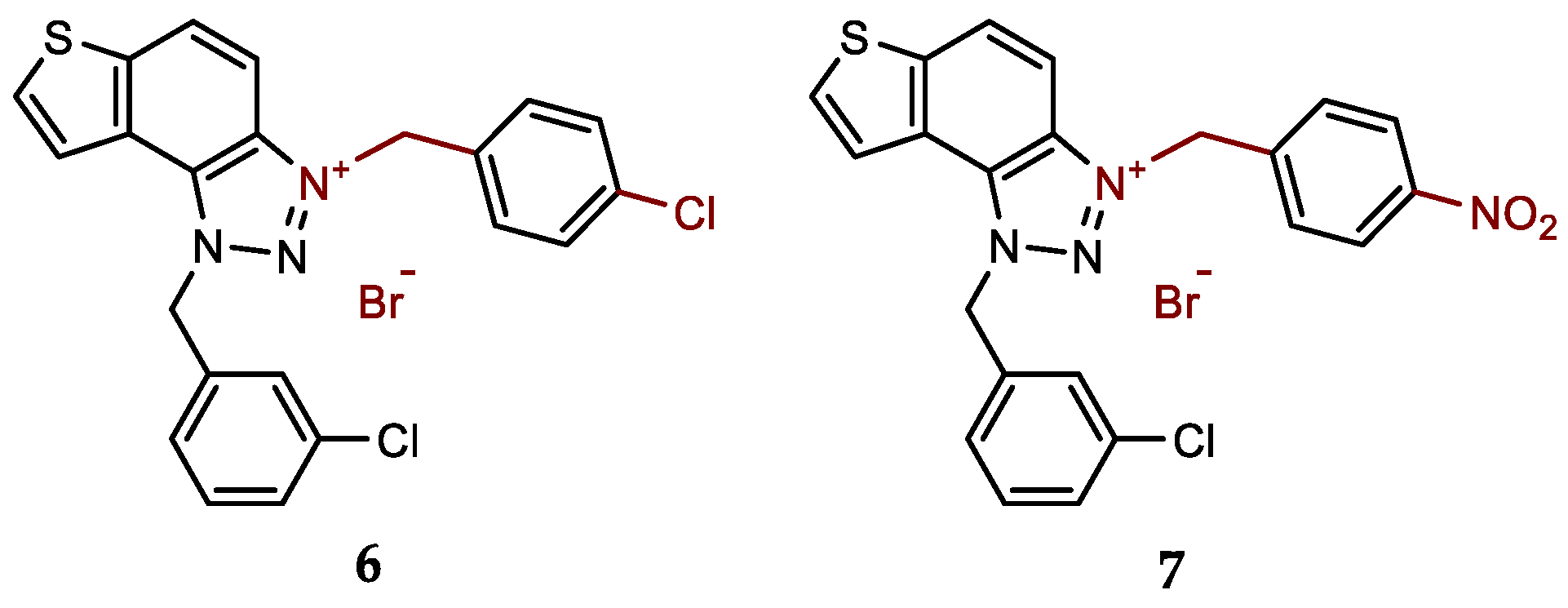
- 1-(3-chlorobenzyl)-3-(4-chlorobenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (6): 8 mg (isolated 40%), white powder; m.p. 111-113 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.55 (d, J = 9.2 Hz, 1H), 8.26 (d, J = 5.5 Hz, 1H), 8.14–8.10 (m, 2H), 7.58 (d, J = 8.8 Hz, 2H), 7.52 (t, J = 1.5, 3.4 Hz, 1H), 7.50–7.41 (m, 4H), 7.38 (dt, J = 1.5, 3.3 Hz, 1H), 6.49 (s, 2H), 6.28 (s, 2H); 13C NMR (CD3OD, 150 MHz) δ/ppm: 143.2, 135.4, 134.9, 134.1, 133.8, 133.2, 130.6, 130.3, 129.2, 129.3, 127.9, 127.2, 126.2, 123.1, 120.3 55.3, 54.3; HRMS (ESI) (m/z) for C22H16Cl2N3S+Br-: [M + H]+calcd = 424.0442, and [M + H]+measured = 424.0447.
- 1-(3-chlorobenzyl)-3-(4-nitrobenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (7): 9 mg (isolated 42%), white powder; m.p. 125-126 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.57 (d, J = 9.2 Hz, 1H), 8.32 (d, J = 8.7 Hz, 2H), 8.27 (d, J = 5.7 Hz, 1H), 8.15–8.11 (m, 3H), 7.80–7.77 (m, 2H), 7.47–7.38 (m, 3H) 6.53 (s, 2H), 6.47 (s, 2H); 13C NMR (CD3OD, 75 MHz) δ/ppm: 140.3, 136.4, 135.9, 135.3, 134.8, 132.1, 131.1, 130.6, 130.7, 129.4, 128.8, 127.7, 125.4, 124.6, 123.9, 121.8, 110.0, 56.9, 55.4; HRMS (ESI) (m/z) for C22H16ClN4O2S+Br-: [M + H]+calcd = 435.0682, and [M + H]+measured = 435.0690.
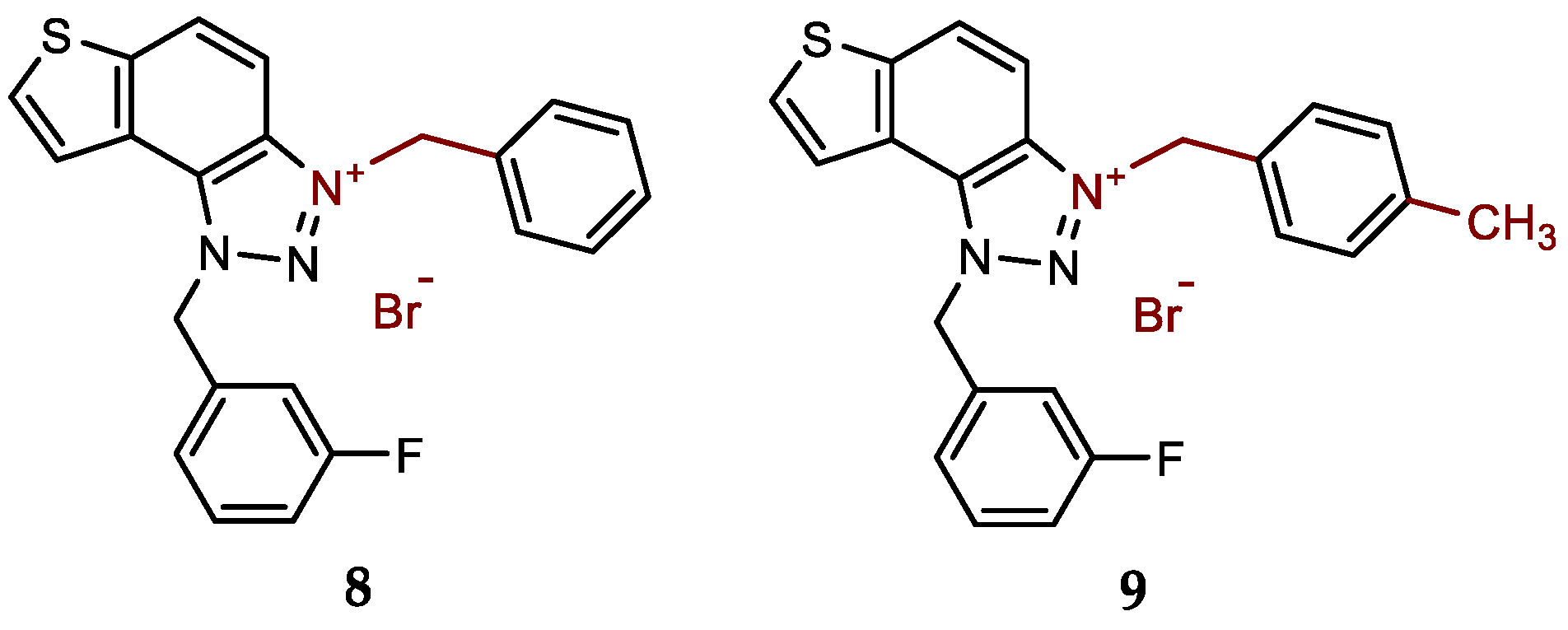
- 3-benzyl-1-(3-fluorobenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (8): 7 mg (isolated 48%), white powder; m.p. 103-104 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.53 (d, J = 9.1 Hz, 1H), 8.23 (d, J = 5.5 Hz, 1H), 8.13–8.05 (m, 3H), 7.58 (dd, J = 1.8, 8.0 Hz, 2H), 7.49–7.42 (m, 4H), 7.27–7.21 (m, 2H), 6.53 (s, 2H), 6.31 (s, 2H); 13C NMR (CD3OD, 75 MHz) δ/ppm: 145.3 (d, JCF = 236.7 Hz), 143.7, 139.1, 138.0, 137.1, 134.9, 134.6, 133.6, 133.4, 133.1, 132.9, 132.5, 130.9, 127.1, 124.3, 111.8, 59.3, 59.2 (signals for four quaternary C are missing); HRMS (ESI) (m/z) for C22H17FN3S+Br-: [M + H]+calcd = 374.1127, and [M + H]+measured = 374.1131.
- 1-(3-fluorobenzyl)-3-(4-methylbenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (9): 14 mg (isolated 93%), white powder; m.p. 116-117 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.51 (dd, J = 0.7, 9.2 Hz, 1H), 8.22 (d, J = 5.2 Hz, 1H), 8.12–8.05 (m, 2H), 7.50–7.43 (m, 3H), 7.30 – 7.22 (m, 4H), 7.19–7.15 (m, 1H), 6.53 (s, 2H), 6.27 (s, 2H), 2.36 (s, 3H); 13C NMR (CD3OD, 75 MHz) δ/ppm: 167.1 (d, JCF = 246.2 Hz), 147.0, 143.7, 138.4 (d, JCF = 8.0 Hz), 138.0, 137.0, 135.0 (d, JCF = 8.7 Hz), 133.6, 132.9, 132.5, 130.9, 127.4, 127.3, 127.0, 124.3, 119.9 (d, JCF = 21.7 Hz), 118.5 (d, JCF = 23.3 Hz), 59.3, 59.2, 23.7; HRMS (ESI) (m/z) for C23H19FN3S+Br-: [M + H]+calcd = 388.1284, and [M + H]+measured = 388.1292.
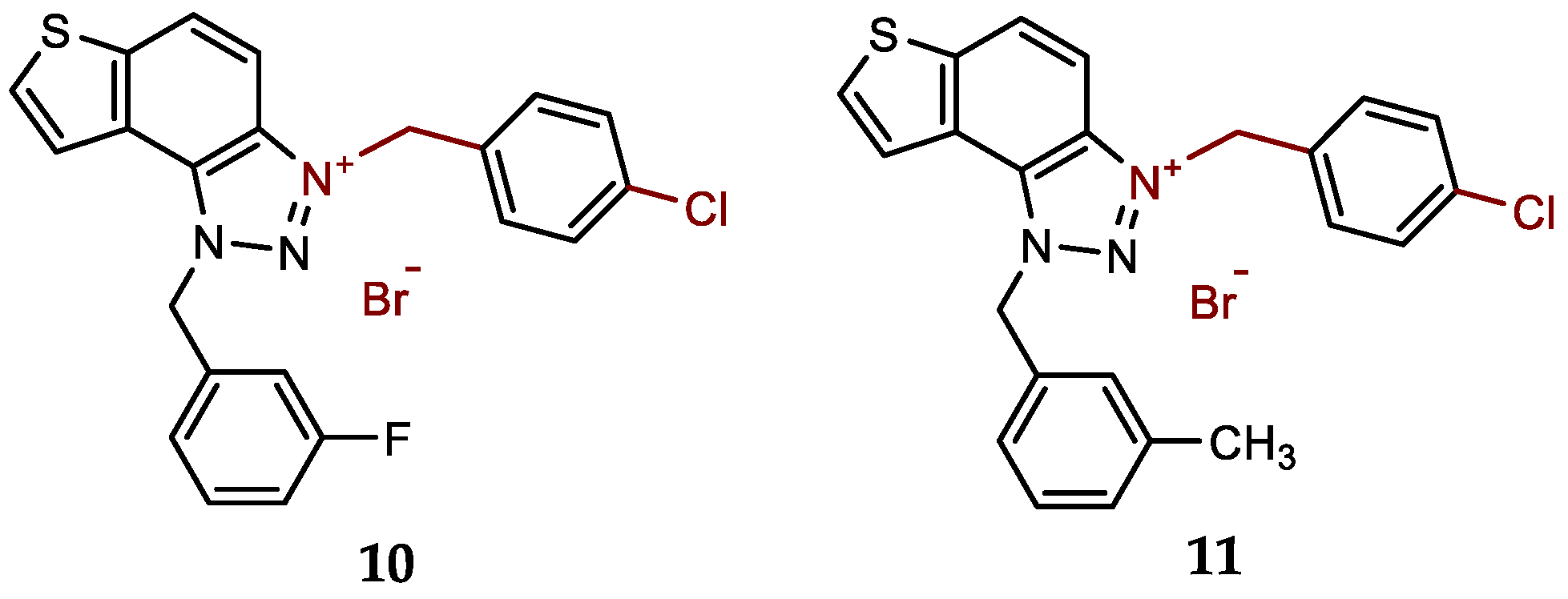
- 3-(4-chlorobenzyl)-1-(3-fluorobenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (10): 7 mg (isolated 45%), white powder; m.p. 110-112 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.55 (dd, J = 0.7, 9.2 Hz, 1H), 8.24 (d, J = 5.5 Hz, 1H), 8.14–8.10 (m, 1H), 8.08–8.06 (m, 1H), 7.58 (d, J = 8.6 Hz, 2H), 7.50–7.44 (m, 3H), 7.29–7.15 (m, 3H), 6.53 (s, 2H), 6.31 (s, 2H); 13C NMR (CD3OD, 150 MHz) δ/ppm: 143.2, 136.3 (d, JCF = 216.2 Hz), 135.4 , 134.2, 133.6, 133.2, 132.7, 131.1 (d, JCF = 8.1 Hz), 130.7, 130.3, 129.2, 127.2, 123.5, 123.1, 120.4, 115.9 (d, JCF = 21.9 Hz), 114.6 (d, JCF = 23.2 Hz), 107.7, 55.4, 54.5 (signals for 2 quaternary C are missing); HRMS (ESI) (m/z) for C22H16ClFN3S+Br-: [M + H]+calcd = 408.0738, and [M + H]+measured = 408.0745.
- 3-(4-chlorobenzyl)-1-(3-methylbenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (11): 8 mg (isolated 94%), white powder; m.p. 122-123 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.53 (d, J = 9.2 Hz, 1H), 8.22 (d, J =5.5 Hz, 1H), 8.14–8.06 (m, 2H), 7.58 (d, J = 8.6 Hz, 2H), 7.48 (d, J = 8.5 Hz, 2H), 7.32–7.17 (m, 4H), 6.46 (s, 2H), 6.32 (s, 2H), 2.32 (s, 3H); 13C NMR (CD3OD, 75 MHz) δ/ppm: 144.5, 140.7, 136.8, 135.6, 134.5, 133.2, 132.4, 131.7, 131.2, 130.6, 130.4, 129.4, 128.5, 125.9, 124.6, 122.0, 109.1, 55.8, 57.5, 21.3; HRMS (ESI) (m/z) for C23H19ClN3S+Br-: [M + H]+calcd = 404.0988, and [M + H]+measured = 404.0996.
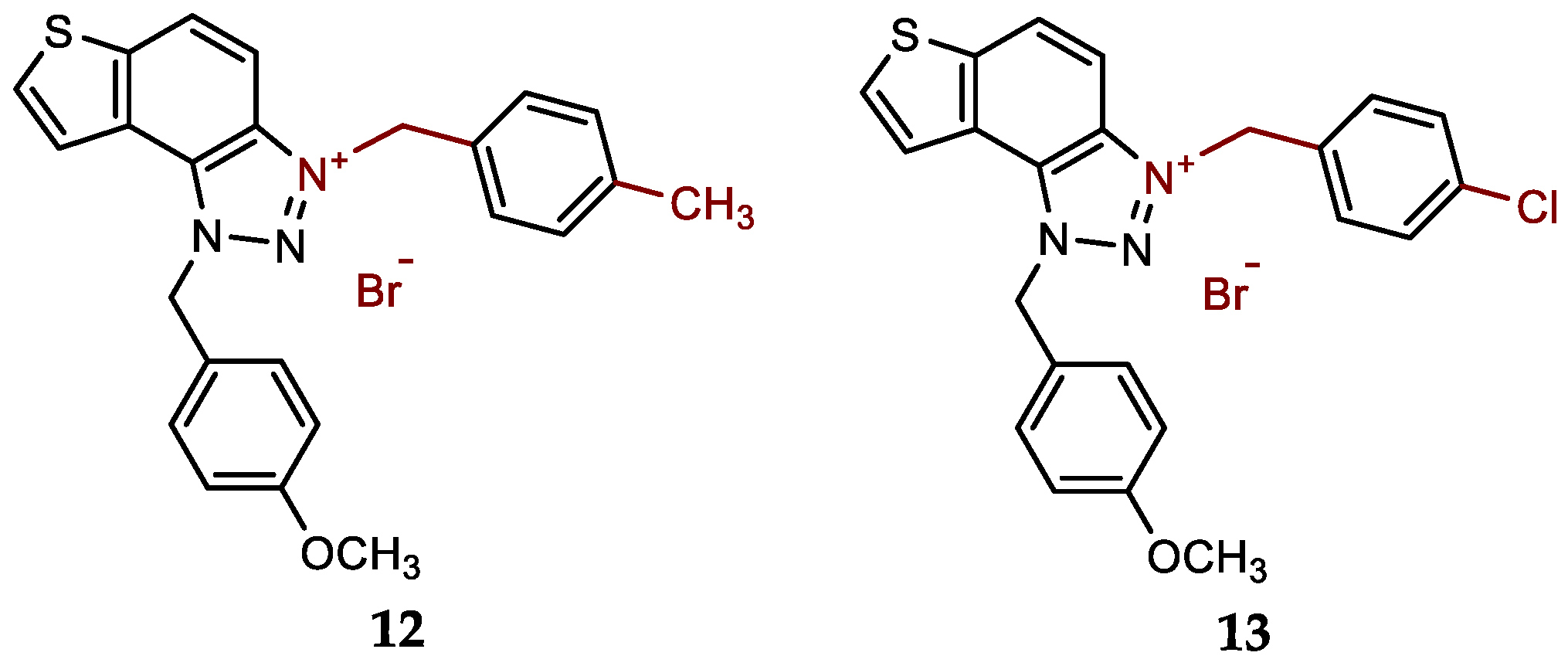
- 1-(4-methoxybenzyl)-3-(4-methylbenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (12): 9 mg (isolated 95%), white powder; m.p. 120-122 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.49 (d, J = 9.5 Hz, 1H), 8.22 (d, J = 5.5 Hz, 1H), 8.15–8.11 (m, 1H), 8.06 (d, J = 9.2 Hz, 1H), 7.44 (d, J = 8.2 Hz, 2H), 7.39 (d, J = 8.9 Hz, 2H), 7.27 (d, J = 8.2 Hz, 2H), 6.97 (d, J = 8.9 Hz, 2H), 6.42 (s, 2H), 6.24 (s, 2H), 3.79 (s, 3H), 2.35 (s, 3H); 13C NMR (CD3OD, 150 MHz) δ/ppm: 142.9, 139.7, 134.3, 133.9, 132.9, 130.8, 129.7, 129.2, 129.1, 128.4, 126.8, 123.6, 123.2, 120.6, 114.3, 107.8, 65.4, 54.3, 53.4, 19.8; HRMS (ESI) (m/z) for C24H22N3OS+Br-: [M + H]+calcd = 400.1484, and [M + H]+measured = 400.1487.
- 3-(4-chlorobenzyl)-1-(4-methoxybenzyl)-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (13): 7 mg (isolated 74%), white powder; m.p. 124-125 °C; 1H NMR (CD3OD, 600 MHz) δ/ppm: 8.52 (dd, J = 0.7, 9.2 Hz, 1H), 8.24 (d, J = 5.5 Hz, 1H), 8.15 (d, J = 5.5 Hz, 1H), 8.09 (d, J = 9.2 Hz, 1H), 7.36 (d, J = 8.6 Hz, 2H), 7.47 (d, J = 8.6 Hz, 2H), 7.40 (d, J = 8.8 Hz, 2H), 7.00 (d, J = 8.7 Hz, 2H), 6.42 (s, 2H), 6.29 (s, 2H), 3.80 (s, 3H); 13C NMR (CD3OD, 75 MHz) δ/ppm: 134.5, 132.3, 131.6, 130.7, 130.6, 128.5, 125.5, 124.9, 122.1, 115.8, 109.1, 57.3, 55.8, 55.7 (signals for 5 quaternary C are missing); HRMS (ESI) (m/z) for C23H19ClN3OS+Br-: [M + H]+calcd = 420.0937, and [M + H]+measured = 420.0943.
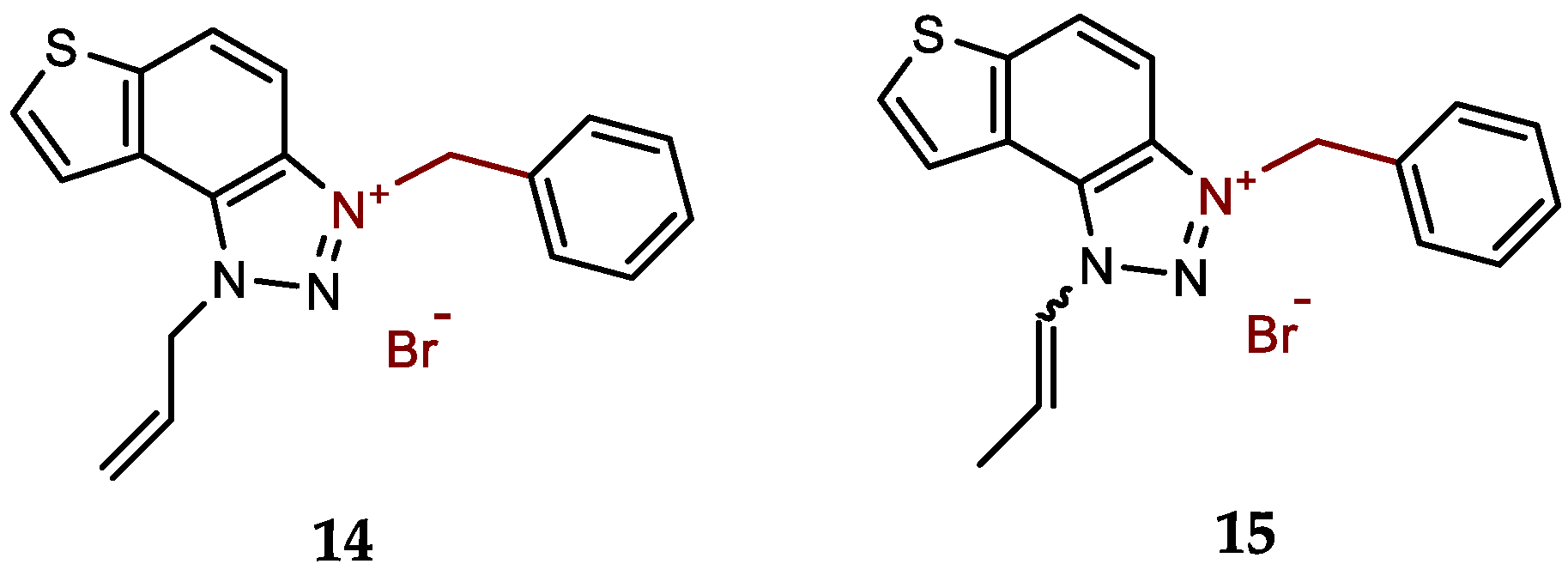
- 1-allyl-3-benzyl-1H-thieno[3',2':3,4]benzo[1,2-d][1,2,3]triazol-3-ium bromide (14): 8 mg (isolated 64%), white powder; m.p. 101-102 °C; 1H NMR (CD3OD, 300 MHz) δ/ppm: 8.52 (d, J = 9.2 Hz, 1H), 8.29 (d, J = 5.5 Hz, 1H), 8.18 (d, J = 5.6 Hz, 1H), 8.14–8.05 (m, 2H), 7.61–7.53 (m, 2H), 7.49–7.41 (m, 2H), 6.40–6.32 (m, 1H), 6.30 (s, 2H), 5.92 (d, J = 5.5 Hz, 2H), 5.52 (d, J = 10.4 Hz, 1H), 5.41 (d, J = 17.1 Hz, 1H); 13C NMR (CD3OD, 75 MHz) δ/ppm: 142.9, 133.9, 133.02, 132.1, 129.3, 129.1, 128.8, 128.5, 126.9, 123.2, 120.7, 120.2, 114.5, 107.7, 55.2, 54.9; MS (EI) (m/z, %) for C18H16N3S+Br-: 306 (100).
2.3. In Vitro Cholinesterase Inhibition Activity Measurements
2.4. Anti-Inflammatory Activity
2.5. Computational Details
2.6. ADME-Tox Predictions
3. Results and Discussion
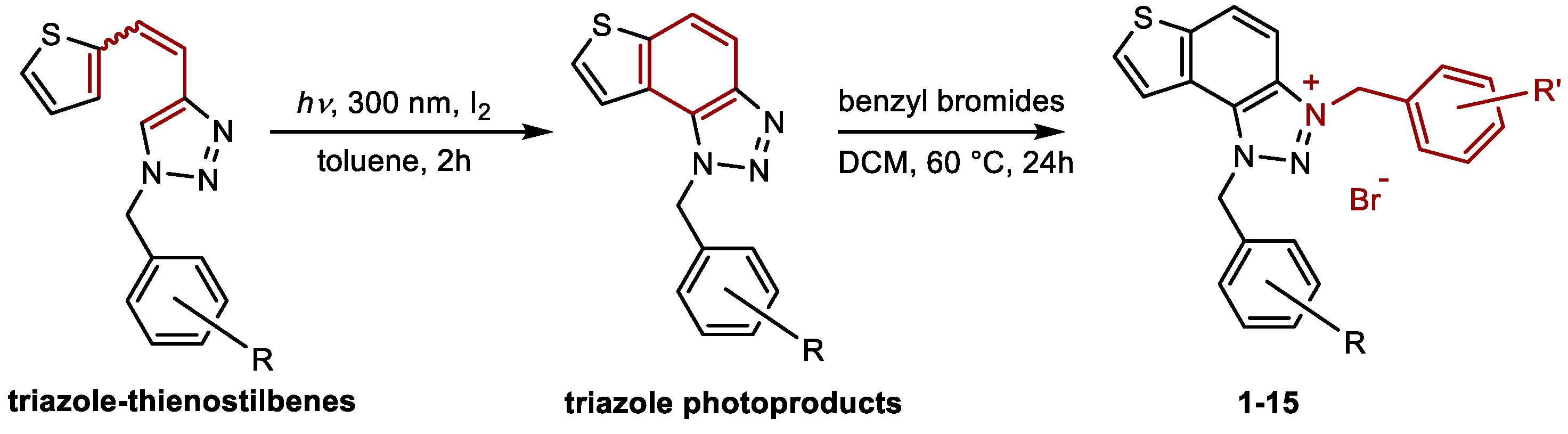
3.1. Synthesis of Charged Thienobenzo-1,2,3-triazoles 1–15
3.2. Cholinesterase Inhibition Activity of Triazole Salts 1-15
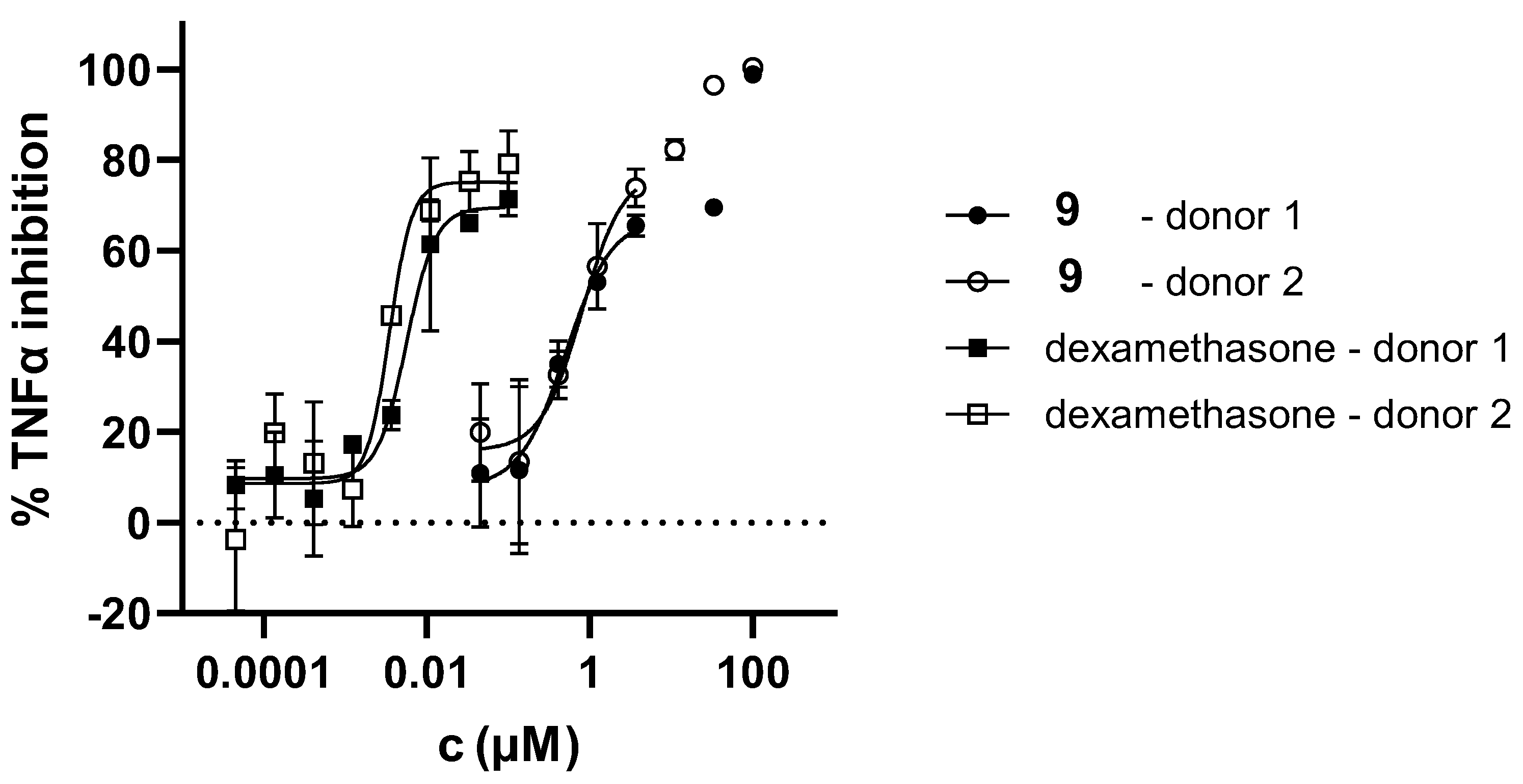
3.3. Anti-Inflammatory Activity of Triazole Salts 1‒15
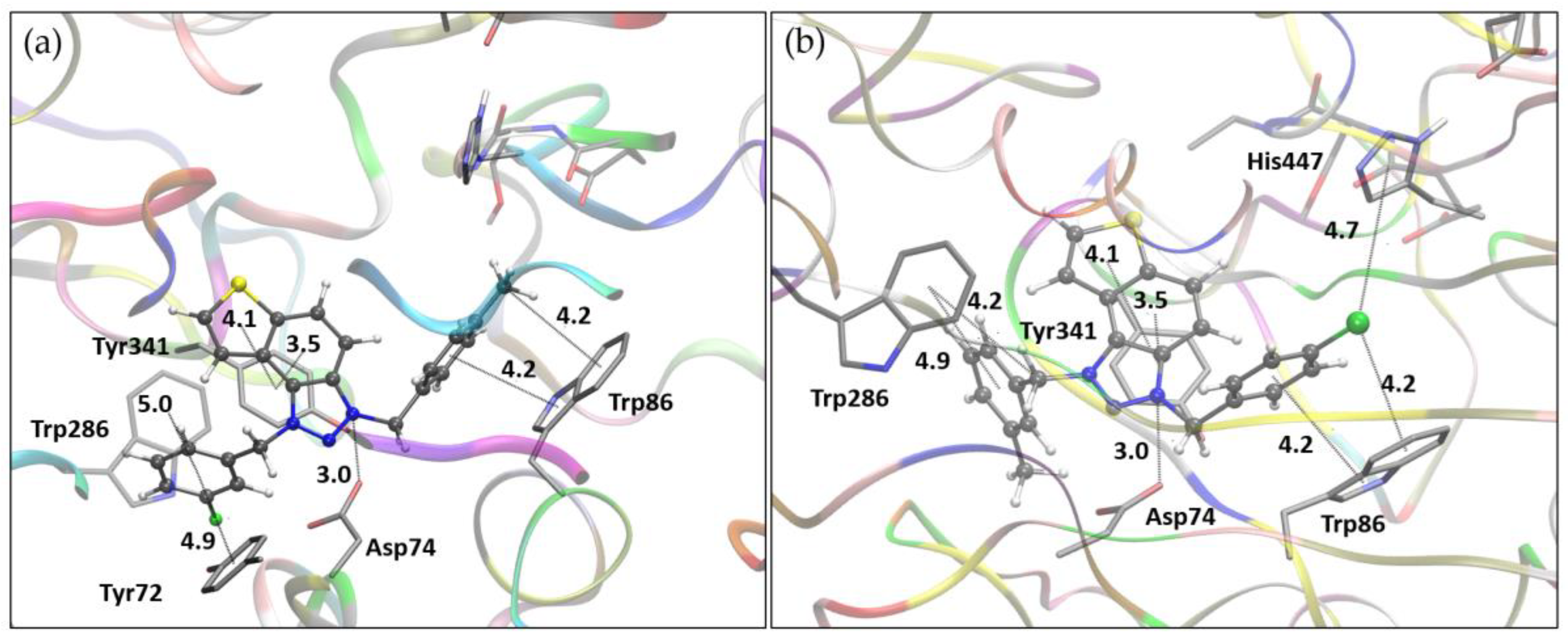
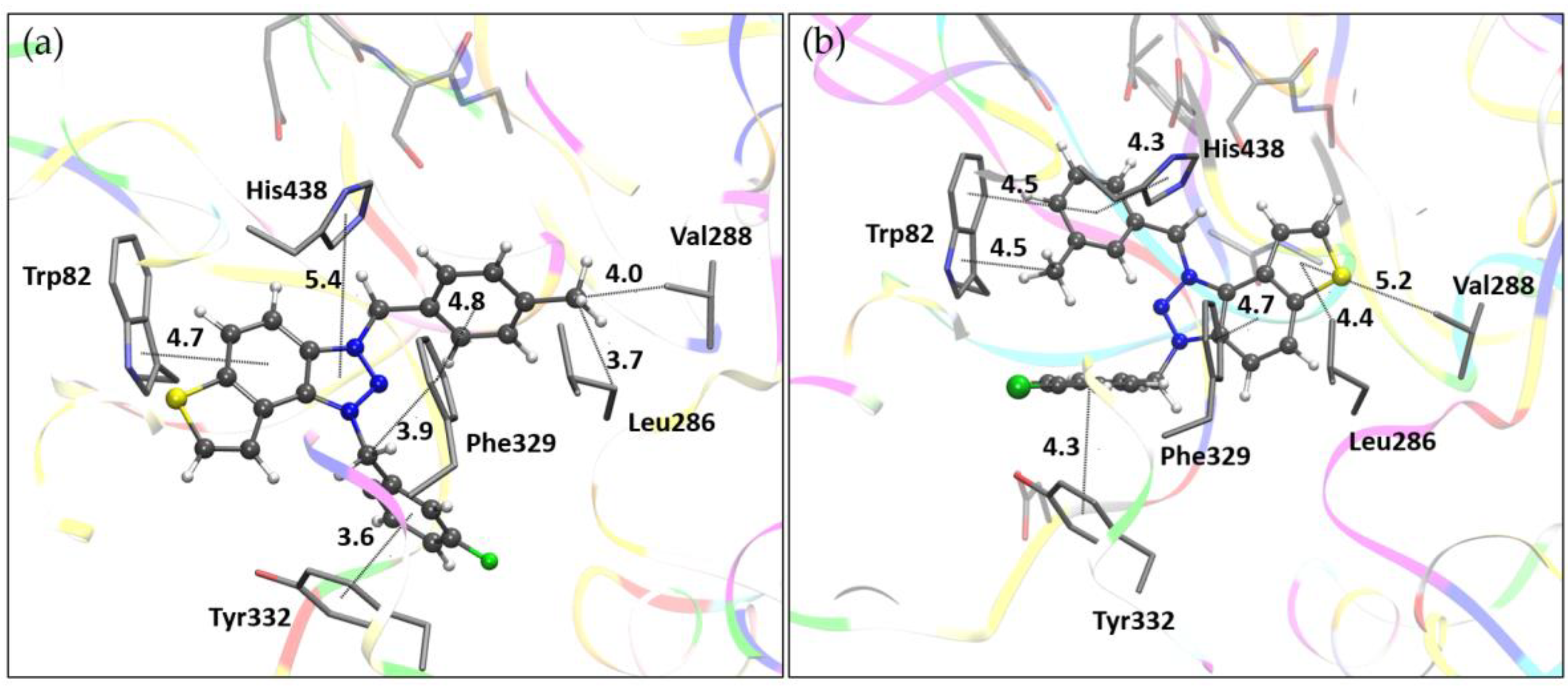
3.4. Molecular Docking of Bioactive Triazole Salts 9 and 11
3.5. Genotoxicity of Triazole Salts 1‒15
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Masson, P.; Lockridge, O. Butyrylcholinesterase for protection from organophosphorus poisons: Catalytic complexities and therapeutic promises. British Journal of Pharmacology 2010, 2010 160, 344–365. [Google Scholar] [CrossRef]
- Colović, M.B.; Krstić, D.Z.; Lazarević-Pašti, T.D.; Bondžić, A.M.; Vasić, V.M. Acetylcholinesterase inhibitors: Pharmacology and toxicology. Current Neuropharmacology 2013, 11, 315–335. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Singh, B. A review on cholinesterase inhibitors for Alzheimer’s disease. Archives of Pharmacal Research 2013, 36, 375–399. [Google Scholar] [CrossRef] [PubMed]
- Talesa, V.N. Acetylcholinesterase in Alzheimer’s disease. Current Drug Targets 2001, 2, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Greig, N.H.; Utsuki, T.; Yu, Q.S.; Holloway, H.W. A new therapeutic target in Alzheimer's disease treatment: Selective butyrylcholinesterase inhibition. Current Medicinal Chemistry 2005, 12, 237–243. [Google Scholar]
- Mesulam, M.M.; Guillozet, A.; Shaw, P.; Levey, A. Acetylcholinesterase knockouts establish centrality of cholinergic networks. Annals of Neurology 2002, 52, 253–256. [Google Scholar]
- Lane, R.M.; Potkin, S.G.; Enz, A. Galantamine: A novel cholinergic modulator. Expert Opinion on Investigational Drugs 2017, 16, 1487–1504. [Google Scholar] [CrossRef]
- Waghmare, A.A.; Kadam, P.; Sharma, H. Clinical application of neostigmine and its peripheral selectivity. Indian Journal of Anaesthesia 2022, 66, 738–745. [Google Scholar]
- Silman, I.; Sussman, J.L. Acetylcholinesterase: 'Classical' and 'non-classical' functions and pharmacology. Current Opinion in Pharmacology 2005, 5, 293–302. [Google Scholar] [CrossRef]
- Taylor, P. Anticholinesterase agents. In Basic & Clinical Pharmacology, 12th ed.; Katzung B.G., Ed.; McGraw-Hill, New York, USA, 2011; pp. 123–134.
- Younus, M. , & Raza, S. (2023). Blood-brain barrier permeability of cholinergic drugs: Prodrug strategies. Drug Development Research, 84. [CrossRef]
- Bajgar, J.; Kuca, K.; Fusek, J.; Jun, D. Pharmacology and toxicology of cholinesterase reactivators: Influence of molecular charge. Neurotoxicity Research 2020, 38, 567–579. [Google Scholar] [CrossRef]
- Bousquet, M.; Nguyen, L.; Bergeron, R. Cholinesterase inhibitors: Clinical applications and molecular mechanisms. Journal of Neurochemistry 2021, 158, 1301–1315. [Google Scholar] [CrossRef]
- Zhou, L.; Wu, Z.; Song, L. Peripheral-targeted cholinesterase inhibitors: A safer path. Frontiers in Pharmacology 2021, 12, 678233. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, C.; Wu, Y.; Zhang, L. Advances in peripheral-selective cholinesterase inhibitors: From chemistry to clinical implications. Current Medicinal Chemistry 2022, 29, 2050–2065. [Google Scholar] [CrossRef]
- Singh, M.; Kaur, R.; Virdi, R. Strategies to optimize CNS delivery of cholinesterase inhibitors. Pharmaceutical Research 2022, 39, 1975–1990. [Google Scholar] [CrossRef]
- Roth, B.L.; Driscol, J.; Glennon, R.A. Drugs with anticholinergic properties: Functional roles and side effects. Pharmacological Reviews 2002, 54, 364–385. [Google Scholar]
- Krátký, M.; Vinšová, J.; Buchta, V.; Stolaříková, J. Quaternary ammonium-based cholinesterase inhibitors: Synthesis and biological evaluation. Bioorganic & Medicinal Chemistry Letters 2016, 26, 1125–1130. [Google Scholar]
- Mlakić, M.; Sviben, M.; Ratković, A.; Raspudić, A.; Barić, D.; Šagud, I.; Lasić, Z.; Odak, I.; Škorić, I. Efficient Access to New Thienobenzo-1,2,3-Triazolium Salts as Preferred Dual Cholinesterase Inhibitors. Biomolecules 2024, 14, 1391. [Google Scholar] [CrossRef]
- Ratković, A.; Mlakić, M.; Dehaen, W.; Opsomer, T.; Barić, D.; Škorić, I. Synthesis and photochemistry of novel 1,2,3-triazole di-heterostilbenes. An experimental and computational study. Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy 2021, 261, 120056. [Google Scholar] [CrossRef]
- Mlakić, M.; Barić, D.; Ratković, A.; Šagud, I.; Čipor, I.; Piantanida, I.; Odak, I.; Škorić, I. New Charged Cholinesterase Inhibitors: Design, Synthesis, and Characterization. Molecules 2024, 29, 1622. [Google Scholar] [CrossRef]
- Mlakić, M.; Odak, I.; Faraho, I.; Talić, S.; Bosnar, M.; Lasić, K.; Barić, D.; Škorić, I. New naphtho/thienobenzo-triazoles with interconnected anti-inflammatory and cholinesterase inhibitory activity. Eur. J. Med. Chem. 2022, 241, 114616. [Google Scholar] [CrossRef]
- Mlakić, M.; Selec, I.; Ćaleta, I.; Odak, I.; Barić, D.; Ratković, A.; Molčanov, K.; Škorić, I. New Thienobenzo/Naphtho-Triazoles as Butyrylcholinesterase Inhibitors: Design, Synthesis and Computational Study. Int. J. Mol. Sci. 2023, 24, 5879. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Courtnex, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcho-linesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Mlakić, M.; Faraho, I.; Odak, I.; Kovačević, B.; Raspudić, A.; Šagud, I.; Bosnar, M.; Škorić, I.; Barić, D. Cholinesterase Inhibitory and Anti-Inflammatory Activity of the Naphtho- and Thienobenzo-Triazole Photoproducts: Experimental and Computational Study. Int. J. Mol. Sci. 2023, 24, 14676. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision C01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDock-Tools4: Automated Docking with Selective Receptor Flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- Cheung, J.; Rudolph, M.; Burshteyn, F.; Cassidy, M.; Gary, E.; Love, J.; Height, J.; Franklin, M. Crystal Structure of Recombinant Human Acetylcholinesterase in Complex with Donepezil. J. Med. Chem. 2012, 55, 10282–10286. [Google Scholar] [CrossRef]
- Nicolet, Y.; Lockridge, O.; Masson, P.; Fontecilla-Camps, J.C.; Nachon, F. Crystal Structure of Human Butyrylcholinesterase. J. Biol. Chem. 2003, 278, 41141–41147. [Google Scholar] [CrossRef]
- Yang, H.; Lou, Ch.; Sun, L.; Li, J.; Cai, Y.; Wang, Zh.; Li, W.; Liu, G.; Tang, Y. AdmetSAR 2.0: web-service for prediction and optimization of chemical ADMET properties. Bioinformatics 2019, 35, 1067–1069. [Google Scholar] [CrossRef]
- Hasselgren, C.; Bercu, J.; Cayley, A.; Cross, K.; Glowienke, S.; Kruhlak, N.; Muster, W.; Nicolette, J.; Vijayaraj Reddy, M.; Saiakhov, R.; Dobo, K. Management of Pharmaceutical ICH M7 (Q)SAR Predictions – The Impact of Model Updates. Regul. Toxicol. Pharmacol. 2020, 118, 104807. [Google Scholar] [CrossRef]


| Compound | AChE IC50 / μM |
BChE IC50 / μM |
Selectivity index |
|---|---|---|---|
| 1 | 12.74 ± 1.85 | 2.55 ± 0.58 | 5.0 |
| 2 | 10.71 ± 1.46 | 4.99 ± 1.10 | 2.1 |
| 3 | 8.89 ± 3.26 | 7.60 ± 0.82 | 1.2 |
| 5 | 55.62 ± 2.36 | 8.13 ± 0.83 | 6.8 |
| 6 | 12.90 ± 0.87 | 0.96 ± 0.14 | 13.4 |
| 7 | 11.42 ± 0.39 | 10.03 ± 1.64 | 1.1 |
| 8 | 22.23 ± 2.83 | 4.52 ± 0.17 | 4.9 |
| 9 | 4.80 ± 0.71 | 0.363 ± 0.038 | 13.2 |
| 10 | 9.96 ± 0.42 | 2.45 ± 0.43 | 4.1 |
| 11 | 6.23 ± 0.58 | 1.11 ± 0.77 | 5.6 |
| 12 | 11.81 ± 2.15 | 3.98 ± 1.25 | 3.0 |
| 13 | 7.40 ± 0.94 | 4.05 ± 1.05 | 1.8 |
| 14 | 2.13 ± 0.13 | 0.098 ± 0.018 | 21.7 |
| 15 | 3.62 ± 0.42 | 0.732 ± 0.137 | 4.9 |
| Donepezil | 0.023 ±0.004 | 4.25 ±0.09 | 0.005 |
| Structure | ICH M7 Class | DerekPrediction | Sarah Prediction |
Experimental Data | Overall In Silico |
|---|---|---|---|---|---|
| 1 | Class 5 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 2 | Class 5 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 3 | Class 5 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 4 | Class 5 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 5 | Class 3 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Positive |
| 6 | Inconclusive |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 7 | Class 3 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Positive |
| 8 | Class 5 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 9 | Class 5 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 10 | Class 5 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 11 | Inconclusive |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 12 | Class 3 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Positive |
| 13 | Class 3 |  |
 |
Carc: UnspecifiedAmes: Unspecified | Positive |
| 14 | Inconclusive |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| 15 | Inconclusive |  |
 |
Carc: UnspecifiedAmes: Unspecified | Negative |
| Property | Model Name | 9 | 11 | Unit |
|---|---|---|---|---|
| Absorption | Water solubility | -5.103 | -5.31 | log mol/L |
| Caco2 | 0.814 | 0.812 | log Papp in 10−6 cm/s | |
| Intestinal absorption | 100 | 100 | % Absorbed | |
| Skin permeability | -2.732 | -2.732 | log Kp | |
| P−glycoprotein substrate | Yes | Yes | ||
| P−glycoprotein I inhibitor | No | No | ||
| P−glycoprotein II inhibitor | Yes | Yes | ||
| Distribution | VDss (human) | -0.333 | -0.186 | log L/kg |
| Fraction unbound | 0.299 | 0.289 | Fu | |
| BBB permeability | 0.883 | 0.81 | log BB | |
| CNS permeability | -0.91 | -0.888 | log PS | |
| Metabolism | CYP2D6 substrate | No | No | |
| CYP3A4 substrate | Yes | Yes | ||
| CYP1A2 inhibitor | Yes | Yes | ||
| CYP2C19 inhibitor | Yes | No | ||
| CYP2C9 inhibitor | No | No | ||
| CYP2D6 inhibitor | Yes | Yes | ||
| CYP3A4 inhibitor | No | No | ||
| Excretion | Total clearance | 0.215 | 1.231 | log ml/min/kg |
| Renal OCT2 substrate | No | No | Yes/No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




