Submitted:
31 May 2025
Posted:
02 June 2025
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Circular Lunar Industrial Ecology
3. Thermal Sintering of Lunar-Derived Ceramics
4. 3D Printing of Lunar-Derived Ceramics
5. Problem of Polymers
6. Lunar-Derived Clays
7. Non-Clay Minerals and Their Uses
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mueller, R.; et al. Automated additive construction (AAC) for Earth and space using in-situ resources. Proc Biennial ASCE Conf on Engineering, Science, Construction & Operations in Challenging Environments; 2016. [Google Scholar]
- Fiske, M.; Ellery, A. On the development of an ISRU-based calcium sulphoaluminate concrete for 3D printed and cast lunar surface infrastructure applications. AIAA SciTech Forum, 2025; Orlando, FL, paper no 1863. [Google Scholar]
- Ellery, A. Leveraging in-situ resources for lunar base construction. Canadian J Civil Engineering 2021, 49, 657–674. [Google Scholar] [CrossRef]
- Azami, M.; Kazemi, Z.; Moazen, S.; Dube, M.; Potvin, M.-J.; Skonieczny, K. Comprehensive review of lunar-based manufacturing and construction. Progress in Aerospace Sciences 2024, 150, 101045. [Google Scholar] [CrossRef]
- Zocca, A.; Wilbig, J.; Gunster, J.; Widjaja, P.; Neumann, C.; Clozel, M.; Meyer, A.; Ding, J.; Zhou, Z.; Tian, X. Challenges in the technology development for additive manufacturing in space. Chinese J Mechanical Engineering: Additive Manufacturing Frontiers 2022, 1, 100018. [Google Scholar] [CrossRef]
- Ellery, A. Challenges of robotic milli-g operations for asteroid mining. In Proc Future Technologies Conference 4; Arai, K, Ed.; 2024; Volume 1157, pp. 45–64, Lecture Notes on Networks & Systems. [Google Scholar]
- Ellery, A. Sustainable in-situ resource utilisation on the Moon. Planetary & Space Science 2020, 184, 104870. [Google Scholar]
- Hou, Z.; Gong, Q.; Liu, N.; Jiang, B.; Li, J.; Wu, Y.; Huang, J.; Gu, W. Elemental abundances of Moon samples based on statistical distributions of analytical data. Applied Sciences 2023, 13, 360. [Google Scholar] [CrossRef]
- Heiken, G.; Vaniman, D.; French, B. Lunar Sourcebook: A Users Guide to the Moon. Cambridge University Press, 1991. Available online: https://www.lpi.usra.edu/publications/books/lunar_sourcebook/pdf/LunarSourceBook.pdf.
- Ellery, A. “Mining asteroid versus mining the Moon – can you have your cake and eat it?. extended abstract accepted by Space Resources Roundtable, Colorado School of Mines, CO. 2025. [Google Scholar]
- Unterclass, M. Geomimetics and extreme biomimetics inspired by hydrothermal systems – what can we learn from nature for materials synthesis? Biomimetics 2017, 2, 8. [Google Scholar] [CrossRef]
- Lasaga, A.; Soler, J.; Ganor, J.; Burch, T.; Nagy, K. Chemical weathering rate laws and global geochemical cycles. Geochimica et Cosmochimica Acta 1994, 58, 2361–2386. [Google Scholar] [CrossRef]
- Thibodeau, B.; Walls, X.; Ellery, A.; Cousens, B.; Marczenko, K. Extraction of silica and alumina from lunar highland simulant. Proc ASCE Earth & Space Conf 2024, 6962. [Google Scholar]
- Ellery, A.; Mellor, I.; Wanjara, P.; Conti, M. Metalysis FFC process as a strategic lunar in-situ resource utilisation technology. New Space J 2022, 10, 224–238. [Google Scholar] [CrossRef]
- Walls, X.; Ellery, A.; Marczenko, K.; Wanjara, P. Aluminium metal extraction from lunar highland simulant using electrochemistry. Proc ASCE Earth & Space Conf 2024, 7061. [Google Scholar]
- Hogue, M.; Mueller, R.; Sibille, L.; Hintze, P.; Rasky, D. Extraterrestrial regolith derived atmospheric entry heat shields. Proc 15th Biennial ASCE Conf Engineering, Science, Construction & Operations in Challenging Environments; 2016. [Google Scholar]
- Chen, Y.; Wang, X.; Yu, C.; Ding, J.; Deng, C.; Zhu, H. Properties of inorganic high temperature adhesive for high temperature furnace connection. Ceramics International 2019, 45, 8684–8689. [Google Scholar] [CrossRef]
- McAdam, A.; Zolotiv, M.; Sharp, T.; Leshkin, L. Preferential low pH dissolution of pyroxene in plagioclase-pyroxene mixtures: implications for martian surface materials. Icarus 2008, 196, 90–96. [Google Scholar] [CrossRef]
- Frankberg, E. Ceramic that bends instead of shattering. Science 2022, 378, 359–360. [Google Scholar] [CrossRef]
- Kuang, X.; Carotenuto, G.; Nicolais, L. Review of ceramic sintering and suggestions on reducing sintering temperatures. Advanced Performance Materials 1997, 4, 257–274. [Google Scholar] [CrossRef]
- Ellery, A. Generating and storing power on the Moon using in-situ resources. Proc IMechE J Aerospace Engineering 2021, 236, 1045–1063. [Google Scholar] [CrossRef]
- Vakifahmetoglu, C.; Karacasulu, L. Cold sintering of ceramics and glasses: a review. Current Opinion in Solid State & Materials Science 2020, 24, 100807. [Google Scholar]
- Yamasaki, N.; Yanagisawa, K.; Nishioka, M.; Kanahara, S. Hydrothermal hot-pressing method: apparatus and application. J Materials Science Letters 1986, 5, 355–356. [Google Scholar] [CrossRef]
- Yanagisawa, K.; Nishioka, M.; Ioku, K.; Yamasaki, N. Densification of silica gels by hydrothermal hot-pressing. J Materials Science Letters 1993, 12, 1073–1075. [Google Scholar] [CrossRef]
- Vakifahmetoglu, C.; Karacasulu, L. Cold sintering of ceramics and glasses: a review. Current Opinion in Solid State & Materials Science 2020, 24, 100807. [Google Scholar]
- Chen, W.; Shui, A.; Wang, C.; Li, J.; Ma, J.; Tian, W.; Ota, T.; Xi, X. Preparation of aluminium titanate flexible ceramic by solid phase sintering and its mechanical behaviour. J Alloys & Compounds 2019, 777, 119–126. [Google Scholar]
- Kishimoto, A.; Obata, M.; Asaoka, H.; Hayashi, H. Fabrication of alumina-based ceramic foams utilising superplasticity. J European Ceramic Society 2007, 27, 41–45. [Google Scholar] [CrossRef]
- Wakai, F.; Kondo, N.; Shinoda, Y. Ceramics superplasticity. Current Opinion in Solid State & Materials Science 1999, 4, 461–465. [Google Scholar]
- Pilling, J.; Payne, J. Superplasticity in Al2O3-ZrO2-Al2TiO5 ceramics. Scripta Metallurgica et Materiala 1995, 32, 1091–1097. [Google Scholar] [CrossRef]
- Shen, Z.; Peng, H.; Nygren, M. Formidable increase in the superplasticity of ceramics in the presence of an electric field. Advances in Materials 2003, 15, 1006–1009. [Google Scholar] [CrossRef]
- Dong, L.; Zhang, J.; Li, Z.; Gao, Y.; Wang, M.; Huang, M.; Wang, J.; Chen, K. Borrowed dislocations for ductility in ceramics. Science 2024, 385, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Cao, J.; Noda, K.; Han, K. Mechanical properties of ceramic-metal composites by pressure infiltration of metal into porous ceramics. Materials Science & Engineering A 2004, 374, 1–9. [Google Scholar]
- Wan, F.; Zuo, Y.; Pan, Y.; Yu, A.; Qi, J.; Lu, X. Iron-enhanced simulated lunar regolith sintered blocks: preparation, sintering and mechanical properties. Construction & Building Materials 2024, 439, 137395. [Google Scholar]
- Rahimian, M.; Ehsani, N.; Parvin, N.; Baharvandi, H. Effect of particle size, sintering temperature and sintering time on the properties of Al-Al2O3 composites made by powder metallurgy. J Materials Processing Technology 2009, 209, 5387–5393. [Google Scholar] [CrossRef]
- Azami, M.; Siahsarani, A.; Hadian, A.; Kazemi, Z.; Rahmatabadi, D.; Kashani-Bozorg, F.; Abrinia, K. Laser powder bed fusion of alumina/Fe-Ni ceramic matrix particulate composites impregnated with a polymeric resin. J Materials Research & Technology 2023, 24, 3133–3144. [Google Scholar]
- Laot, M.; Rich, B.; Cheibas, I.; Fu, J.; Zhu, J.-N.; Popovich, V. Additive manufacturing and spark plasma sintering of lunar regolith for functionally graded materials. Spool 2021, 8, 7–23. [Google Scholar]
- Ngo, T.; Kashani, A.; Imbalzano, G.; Nguyen, K.; Hui, D. Additive manufacturing (3D printing): a review of materials, methods, applications and challenges. Composites B: Engineering 2018, 143, 172–196. [Google Scholar] [CrossRef]
- Fang, W.; Mu, Z.; He, Y.; Kong, K.; Jiang, K.; Tang, R.; Liu, Z. Organic-inorganic covalent-ionic molecules for elastic ceramic plastic. Nature 2023, 619, 293–299. [Google Scholar] [CrossRef]
- Sun, S.; Mao, L.-B.; Lei, Z.; Yu, S.-H.; Colfen, H. Hydrogels from amorphous calcium carbonate and polyacrylic acid: bio-inspired materials for ‘mineral plastic’. Angewandte Chemie Int Edition 2016, 55, 11765–11769. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, T.; Huang, G. State-of-the-art research progress and challenge of the printing techniques, potential applications for advanced ceramic materials 3D printing. Materials Today Communications 2024, 40, 110001. [Google Scholar] [CrossRef]
- Clemens, F.; Sarraf, F.; Borzi, A.; Neels, A.; Hadian, A. Material extrusion additive manufacturing of advanced ceramics: towards the production of large components. J European Ceramic Society 2023, 43, 2752–2760. [Google Scholar] [CrossRef]
- Yeh, T.-S.; Sacks, M. Effect of particle size distribution on the sintering of alumina. Communications of American Ceramic Society 1988, 71, C484–C487. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, L.; Dong, X.; Wu, J.; Zhou, Q.; Li, S.; Shen, C.; Liu, W.; Wang, G.; He, R. Additive manufacturing of fibre reinforced ceramic matrix composites: advances, challenges and prospects. Ceramics International 2021, 48, 19542–19556. [Google Scholar] [CrossRef]
- Martin, J.; Fiore, B.; Erb, R. Designing bioinspired composite reinforcement architectures via 3D magnetic printing. Nature Communications 2015, 6, 8641. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Tang, D.; Sun, T.; Xiong, W.; Feng, Z.; Evans, K.; Li, Y. Direct ink writing of mineral materials – a review. Int J Precision Engineering & Manufacturing – Green Technology 2021, 8, 665–685. [Google Scholar]
- Lamnini, S.; Elsaed, H.; Lakhdar, Y.; Baino, F.; Smeacetto, F.; Bernado, E. Robocasting of advanced ceramics: ink optimization and protocol to predict the printing parameters – a review. Helyon 2022, 8, e10651. [Google Scholar] [CrossRef]
- Chen, Z.; Li, Z.; Li, J.; Liu, C.; Lao, C.; Fu, Y.; Liu, C.; Li, Y.; Wang, P.; He, Y. 3D printing of ceramics: a review. J European Ceramic Society 2019, 39, 661–687. [Google Scholar] [CrossRef]
- Shahzad, A.; Lazoglu, I. Direct ink writing of structural and functional ceramics: recent achievements and future challenges. Composites B 2021, 225, 109249. [Google Scholar] [CrossRef]
- Jakus, A.; Koube, K.; Geisendorfer, N.; Shah, R. Robust and elastic lunar and martian structures from 3D printed regolith inks. Scientific Reports 2017, 7, 44931. [Google Scholar] [CrossRef] [PubMed]
- Rocha, V.; Saiz, E.; Tirichenko, I.; Garcia-Tunon, E. Direct ink writing advances in multi-material structures for a sustainable future. J Materials Chemistry A 2020, 8, 15646–15657. [Google Scholar] [CrossRef]
- Bland, P.; Cintala, M.; Horz, F.; Cressey, G. Survivability of meteorite projectiles – results from impact experiments. 32nd Lunar & Planetary Science Conf; 2001. abstract no 1764. [Google Scholar]
- Bland, P.; Artemieva, N.; Collins, G.; Bottke, W.; Bussey, D.; Joy, K. (2008) “Asteroids on the Moon: projectile survival during low velocity impacts. 39th Lunar & Planetary Science Conf; 2008. abstract no 2045. [Google Scholar]
- Halim, S.; Crawford, I.; Collins, G.; Joy, K.; Davison, T. Assessing the survival of carbonaceous chondrites impacting the lunar surface as a potential resource. Planetary & Space Sciences 2024, 246, 105905. [Google Scholar]
- Joy, K.; Crawford, I.; Curran, N.; Zolensky, M.; Fagan, A.; King, D. Moon: an archive of small body migration in the solar system. Earth Moon & Planets 2016, 118, 133–158. [Google Scholar]
- Yang, Y.; Li, S.; Zhu, M.-H.; Liu, Y.; Wu, B.; Du, J.; Fa, W.; Xu, R.; He, Z.; Wang, C.; Xue, B.; Yang, J.; Zou, Y. Impact remnants rich in carbonaceous chondrites detected on the Moon by the Chang’e 4 rover. Nature Astronomy 2022, 6, 207–213. [Google Scholar] [CrossRef]
- Ellery, A. Trials and tribulations of asteroid mining. Proc ASCE Earth & Space Conf 2024, 8087. [Google Scholar]
- O’Lenick, A. Basic silicone chemistry – a review. Silicone Spectator 2009, 8087. [Google Scholar]
- Chaudhary, R.; Parameswaran, C.; Idrees, M.; Rasaki, A.; Liu, C.; Chen, Z.; Colombo, P. Additive manufacturing of polymer-derived ceramics: materials, technologies, properties and potential applications. Progress in Materials Science 2022, 128, 100969. [Google Scholar] [CrossRef]
- Davis, P.; Murugavel, R. Recent developments in the chemistry of molecular titanosiloxanes and titanophosphonates. Synthesis & Reactivity in Inorganic, Metal-Organic & Nano-Metal Chemistry 2005, 35, 591–622. [Google Scholar]
- Unny, R.; Gopinathan, S.; Gopinathan, C. Chelated aluminosiloxanes. Indian J Chemistry 1980, 19A, 484–486. [Google Scholar]
- Puxviel, J.; Boilot, J.; Poncelet, O.; Hubert-Pfalzgraf, L.; Lecomte, A.; Dauger, A.; Beloeil, J. Aluminosiloxane as a ceramic precursor. J Non-Crystalline Solids 1987, 93, 277–286. [Google Scholar] [CrossRef]
- Griffith, M.; Halloran, J. Freeform fabrication of ceramics vis stereolithography. J American Ceramics Society 1996, 79, 2601–2608. [Google Scholar] [CrossRef]
- Dou, R.; Tang, W.; Hu, K.; Wang, L. Ceramic paste for space stereolithography 3D printing technology in microgravity environments. J European Ceramic Society 2022, 42, 3968–3975. [Google Scholar] [CrossRef]
- Revelo, C.; Colorado, H. 3D printing of kaolinite clay ceramics using direct ink writing (DIW) technique. Ceramics International 2018, 44, 5673–5682. [Google Scholar] [CrossRef]
- Marquez, C.; Mata, J.; Renteria, A.; Gonzalez, D.; Gomez, G.; Lopez, A.; Baca, A.; Nunez, A.; Hassan, S.; Burke, V.; Perlasca, D.; Wang, Y.; Xiong, Y.; Kruichak, J.; Espalin, D.; Lin, Y. Direct ink-write printing of ceramic clay with an embedded wireless temperature and relative humidity sensor. Sensors 2023, 23, 3352. [Google Scholar] [CrossRef]
- Karl, D.; Kamutzki, F.; Lima, P.; Gili, A.; Duminy, T.; Zocca, A.; Gunster, J.; Gurlo, A. Sintering of ceramics for clay in situ resource utilisation of Mars. Open Ceramics 2020, 3, 100008. [Google Scholar] [CrossRef]
- Tong, W.; Jaw, W.; Pothunuri, L.; Soh, E.; Le Ferrand, H. Easily applicable protocol to formulate inks for extrusion-based 3D printing. Ceramica 2022, 68, 152–159. [Google Scholar] [CrossRef]
- Ellery, A. Supplementing closed ecological life support systems with in-situ resources on the Moon. Life 2021, 11, 770. [Google Scholar] [CrossRef]
- Lazorenko, G.; Kasprzhitskii, A. Geopolymer additive manufacturing: a review. Additive Manufacturing 2022, 55, 102782. [Google Scholar] [CrossRef]
- Ma, S.; Jiang, Z.; Fu, S.; He, P.; Sun, C.; Duan, X.; Jia, D.; Colombo, P.; Zhou, Y. 3D printed lunar regolith simulant-based geopolymer composites with bio-inspired sandwich architectures. J Advanced Ceramics 2023, 12, 510–525. [Google Scholar] [CrossRef]
- Abdulkareem, S.; Orkuma, G.; Apasi, A. Talc as a substitute for dry lubricant (an overview). AIP Proc 2011, 1315, 1400. [Google Scholar]
| Lunar Ilmenite | ||||
| Fe0 + H2O → ferrofluidic sealing | ||||
| FeTiO3 + H2 → TiO2 + H2O + Fe | ||||
| 2H2O→2H2+O2 | ||||
| 2Fe + 1.5O2 → Fe2O3/Fe2O3.CoO - ferrite magnets | ||||
| 3Fe2O3 + H2 ↔ Fe3O4 + H2O) – formation of magnetite at 350-750oC/1-2 kbar | ||||
| 4Fe2O3 + Fe ↔ 3Fe3O4 ) | ||||
| Nickel-Iron Meteorites | ||||
| W inclusions→ | Thermionic cathodic material | |||
| Mond process: | Alloy Ni Co Si C W | |||
| W(CO)6 ↔ 6CO + W | ||||
| Fe(CO)5 ↔ 5CO + Fe (175oC/100 bar) → | Tool steel | 2% | 9-18% | |
| Ni(CO)4 ↔ 4CO + Ni (55oC/1 bar) → | Electrical steel | 3% | ||
| Co2(CO)8 ↔ 8CO + 2Co (150oC/35 bar) → | Permalloy | 80% | ||
| S catalyst | Kovar 29% 17% 0.2% 0.01% ____ . | |||
| 4FeS + 7O2 → 2Fe2O3 + 4SO2 | ||||
| (Troilite) | SO2 + H2S → 3S + H2O | |||
| FeSe + Na2CO3 + 1.5O2 → FeO + Na2SeO3 + CO2 at 650oC | ||||
| KNO3 catalyst | Na2SeO3 + H2SO4 → Na2O + H2SO4 + Se → photosensitive Se | |||
| ↑____________| | ||||
| Na2O + H2O → 2NaOH | ||||
| NaOH + HCl → NaCl + H2O | ||||
| Lunar Orthoclase | ||||
| 3KAlSi3O8 + 2HCl + 12H2O → KAl3Si3O10(OH)2 + 6H4SiO4 + 2KCl | ||||
| orthoclase | illite | silicic acid (soluble silica) | ||
| 2KAl3Si3O10(OH)2 + 2HCl + 3H2O → 3Al2Si2O5(OH)4 + 2KCl | ||||
| kaolinite | ||||
| [2KAlSi3O8 + 2HCl + 2H2O → Al2Si2O5(OH)4 + 2KCl + SiO2 + H2O] | ||||
| KCl + NaNO3 → NaCl + KNO3 | ||||
| 2KCl + Na2SO4 → 2NaCl + K2SO4 | ||||
| Lunar Olivine | ||||
| Fe2SiO4 + 4 HCl + 4H2O → 2FeCl2+ SiO2 + 2H2O | ||||
| fayalite | ||||
| Mg2SiO4 + 4HCl + 4H2O→ 2MgCl2 + 2H4SiO4 | → Sorel cement | |||
| forsterite | MgCl2 +2NaOH → Mg(OH)2 + 2NaCl | |||
| Mg(OH)2 → MgO + H2O | → Sorel cement | |||
| 600-800oC | ||||
| Lunar Anorthite | ||||
| CaAl2Si2O8 + 4C → CO + CaO + Al2O3 + 2Si at 1650oC | → CaO cathode coatings | |||
| CaO + H2O → Ca(OH)2 | ||||
| Ca(OH)2 + CO2 → CaCO3 + H2O | ||||
| CaAl2SiO8 + 8HCl + 2H2O → CaCl2 + 2AlCl3.6H2O + SiO2 | → fused silica glass + FFC electrolyte | |||
| AlCl3.6H2O → Al(OH)3 + 3HCl + H2O at 100oC | ||||
| ↑________________________________________| | ||||
| 2Al(OH)3 → Al2O3 + 3H2O at 400oC → 2Al + Fe2O3 → 2Fe + Al2O3 (thermite) | ||||
| AlNiCo hard magnets | ||||
| Lunar Pyroxene | Al solar sail | |||
| Ca(Fe,Al)Si2O6 + HCl + H2O → Ca0.33(Al)2(Si4O10)(OH)2.nH2O + H4SiO4 + CaCl2 + Fe(OH)3 | ||||
| Augite | montmorillonite | silicic acid | iron hydroxide | |
| Lunar Volatiles | ||||
| CO + 0.5 O2 → CO2 | ||||
| CO2 + 4H2 → CH4 + 2H2O at 300oC (Sabatier reaction) | ||||
| Ni catalyst | ||||
| 850oC | 250oC | |||
| CH4 + H2O → CO + 3H2 → CH3OH 350oC | ||||
| Ni catalyst | Al2O3 | CH3OH + HCl → CH3Cl + H2O | 370oC | +nH2O |
| Al2O3 CH3Cl + Si → (CH3)2SiCl2 → ((CH3)2SiO)n + 2nHCl → silicone plastics/oils | ||||
| ↑_________________________________________| | ||||
| N2 + 3H2 → 2NH3 (Haber-Bosch process) | ||||
| Fe on CaO+SiO2+Al2O3 | ||||
| 4NH3 + 5O2 → 4NO + 6H2O | ||||
| WC on Ni | ||||
| 3NO + H2O → 2HNO3 + NO (Ostwald process) | ||||
| ↑__________________| | ||||
| 2SO2 + O2 ↔ 2SO3 (low temp) | ||||
| SO3 + H2O → H2SO4 | ||||
| Salt Reagent | ||||
| 2NaCl + CaCO3 ↔ Na2CO3 + CaCl2 (Solvay process) → FFC electrolyte | ||||
| 350oC/150 MPa | ||||
| Na2CO3 + SiO2(i) ↔ Na2SiO3 + CO2 → piezoelectric quartz crystal growth (40-80 days) | ||||
| 1000-1100oC | ||||
| CaCO3 → CaO + CO2 (calcination) | ||||
| NaCl(s) + HNO3(g) → HCl(g) + NaNO3(s) 2NaCl(s) + H2SO4(g) → 2HCl(g) + Na2SO4(s) |
||||
| Molten Salt (FFC) Process (CaCl2 electrolyte) | ||||
| MOx + xCa → M + xCaO → M + xCa + ½xO2 where M=Fe, Ti, Al, Mg, Si, etc | ||||
| CO + 0.5 O2 → CO2 | ||||
| CO2 + 4H2 → CH4 + 2H2O at 300oC (Sabatier reaction) → CH4 → C + 2H2 at 1400oC → FFC anode regeneration | ||||
| Ni catalyst | ||||
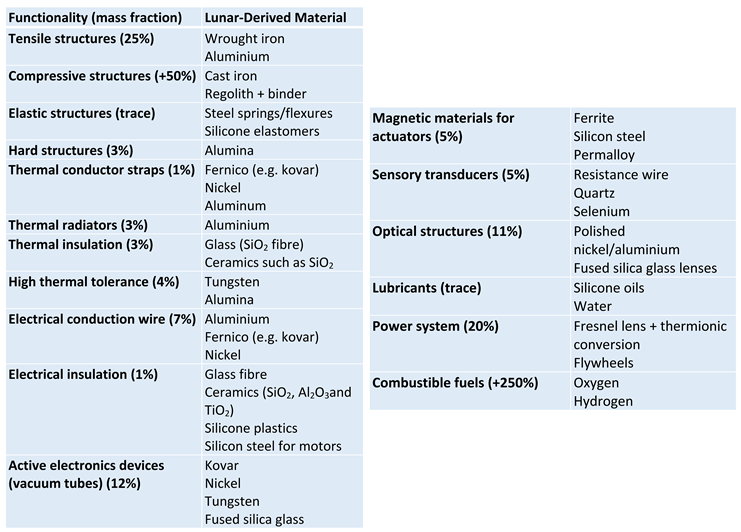 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




