Submitted:
27 May 2025
Posted:
28 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
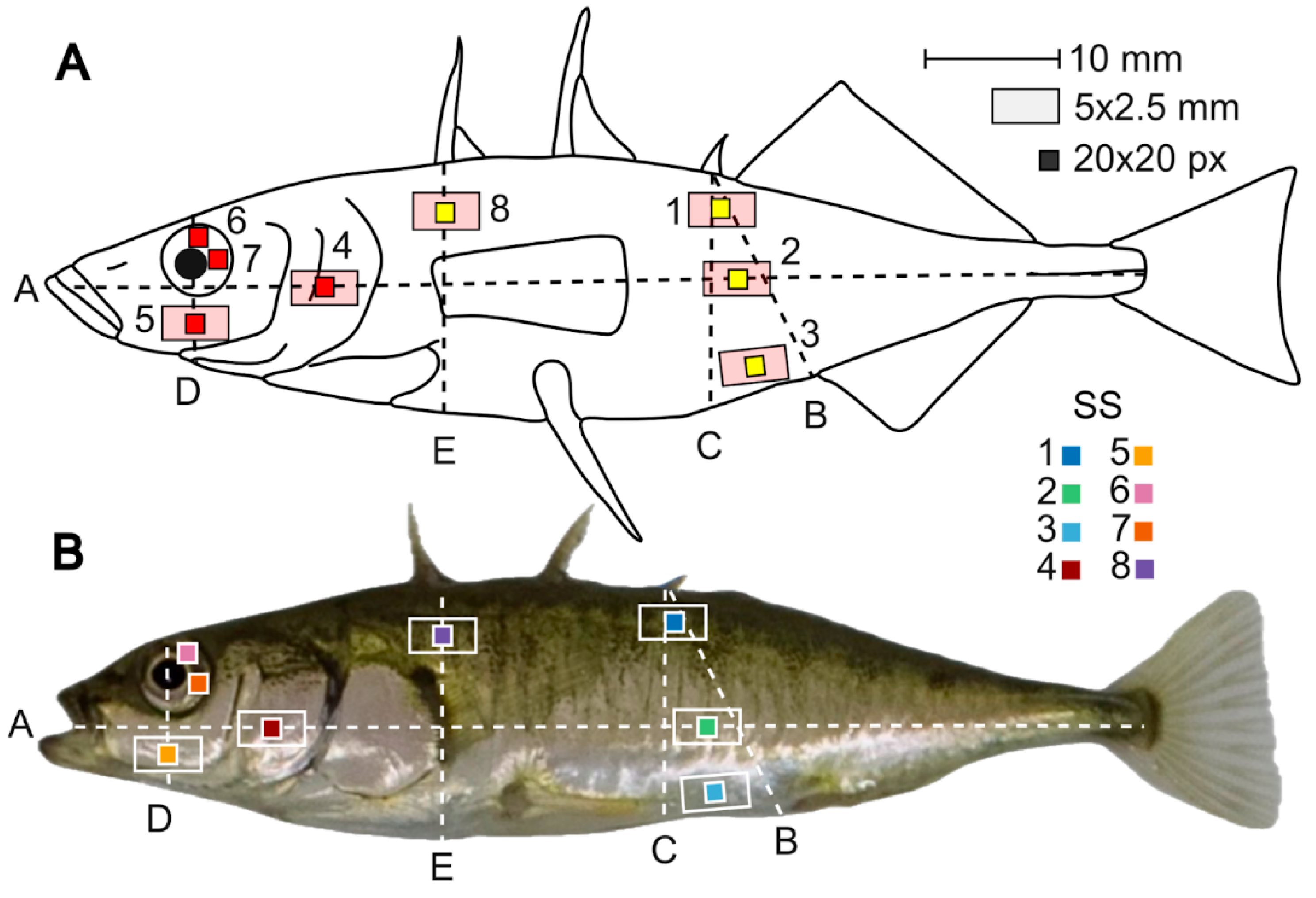
3.1. Method Developing: Standard Sites Selection and Coloration Description
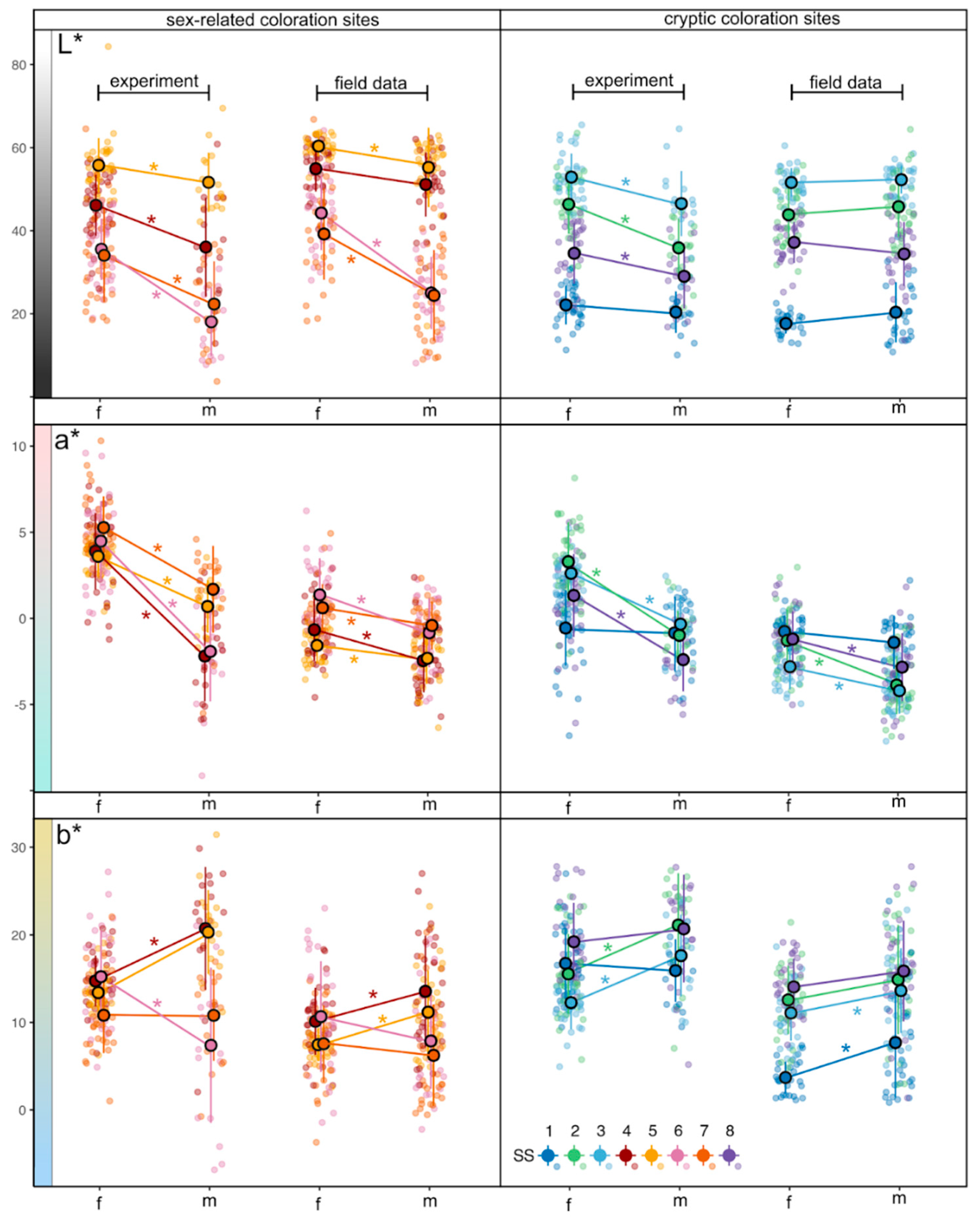
3.2. L*, a* and b* Variation
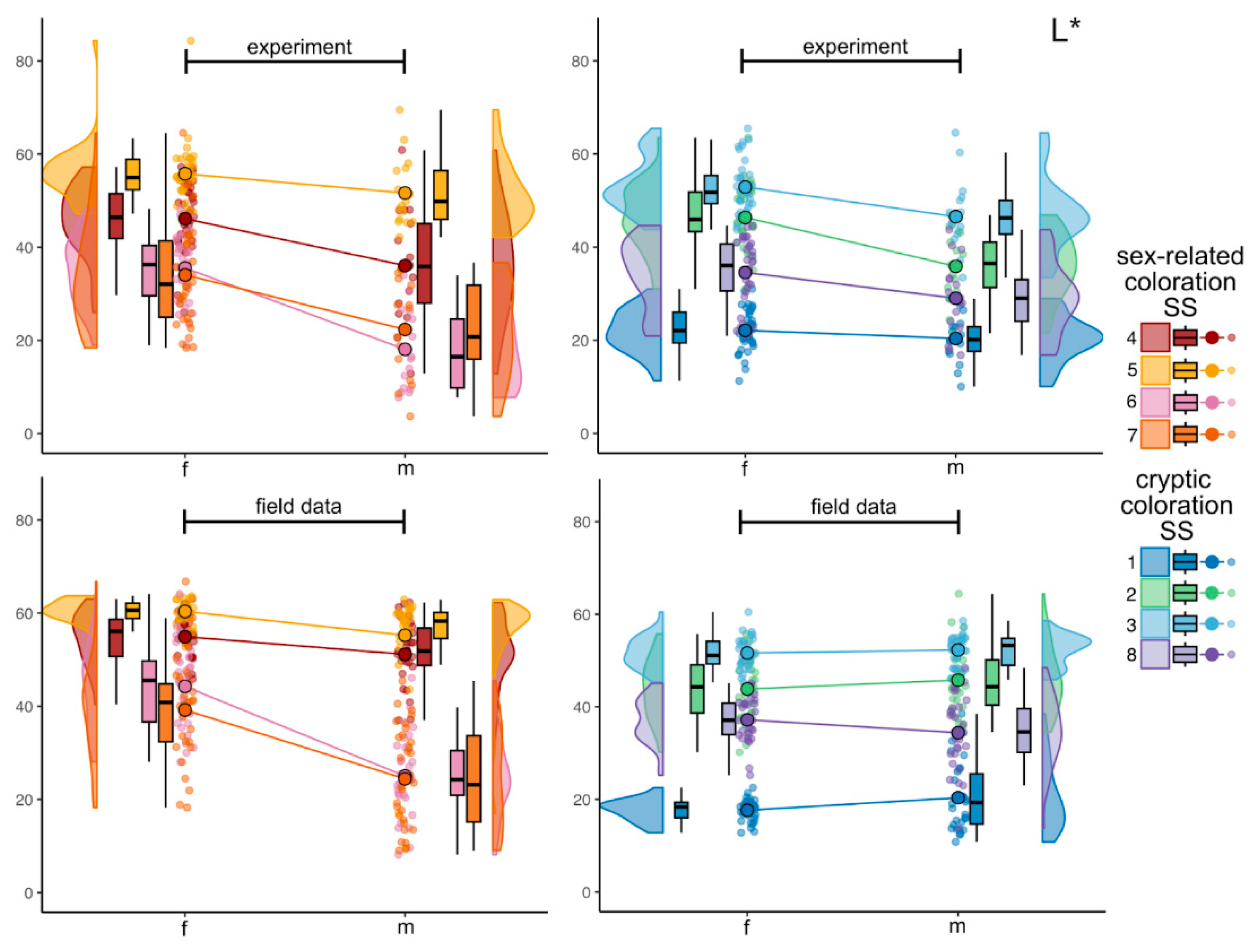
3.2.1. Lightness (L*)
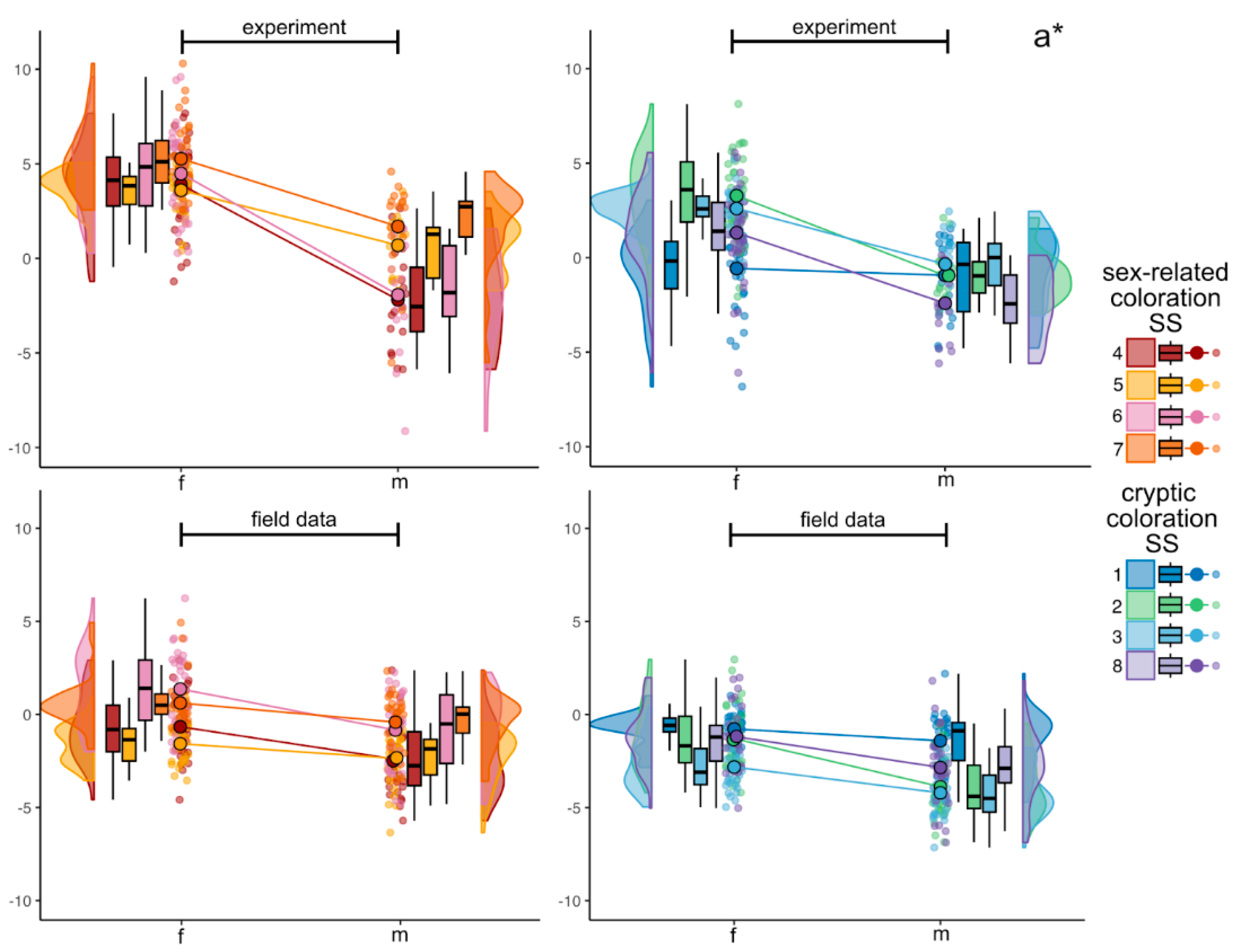
3.2.2. Green to Magenta Axis (a*)
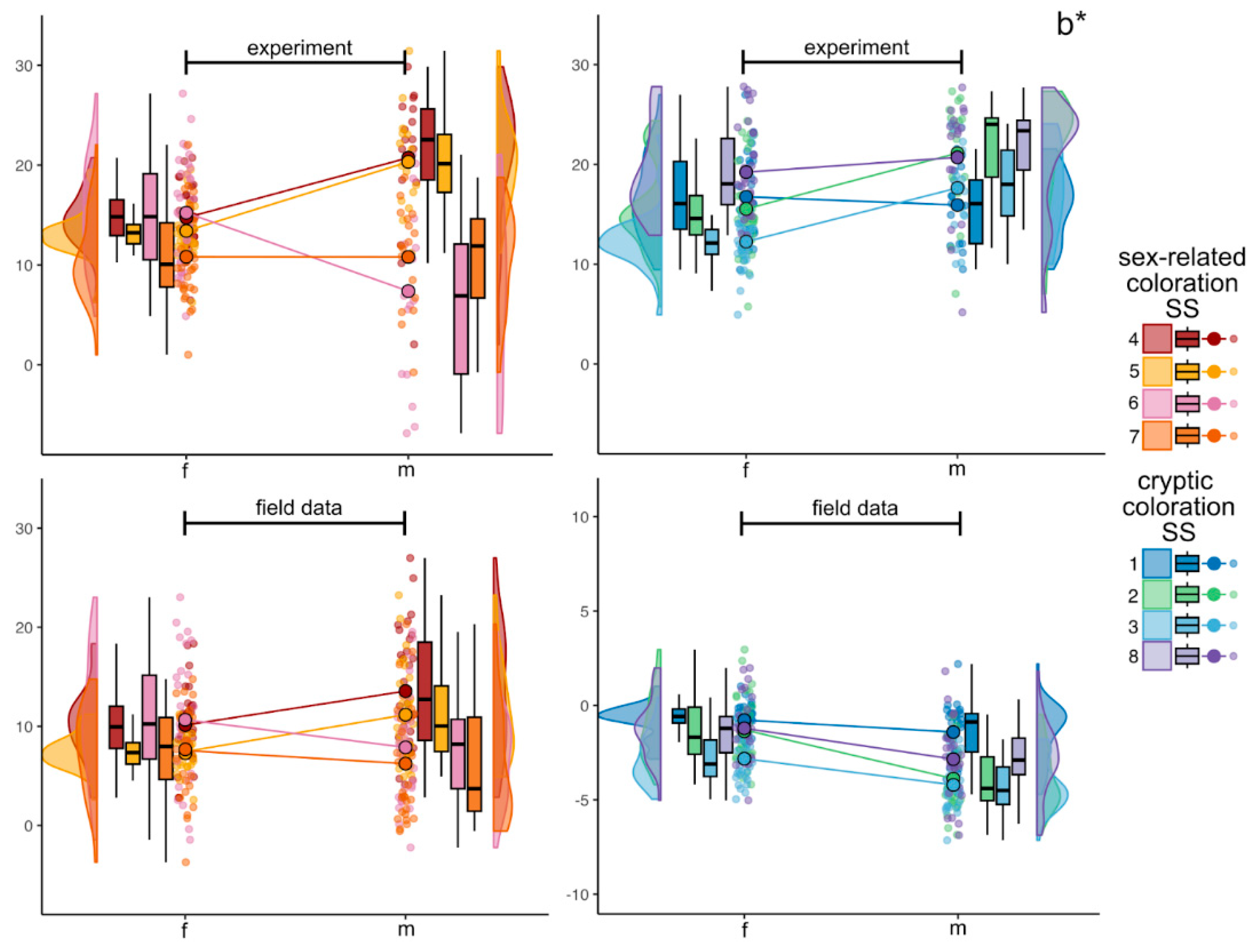
3.2.3. Blue to Yellow Axis (b*)
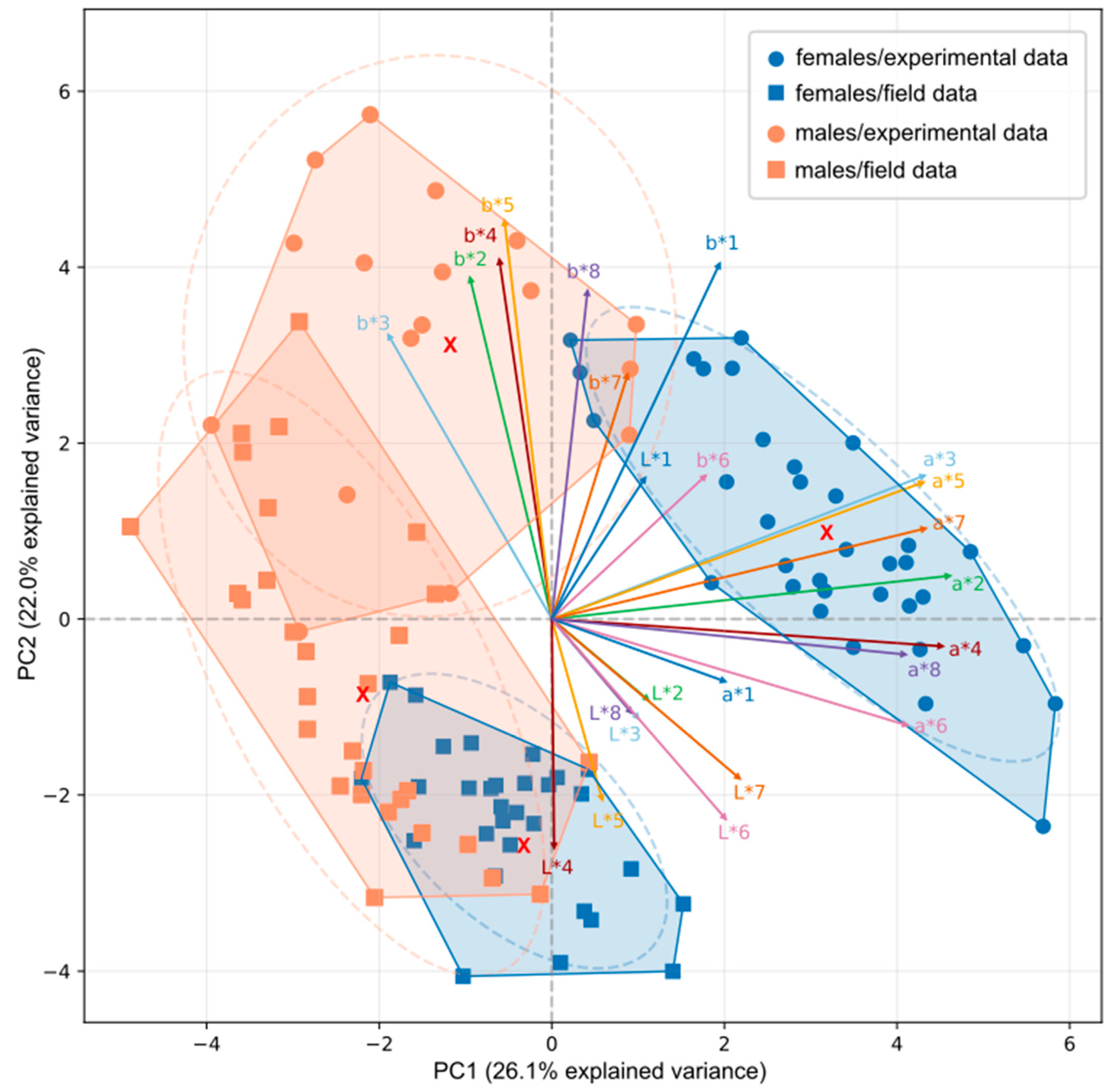
3.3. Principal Component Analysis
3.4. Pigmentation Index (∆E*)
4. Discussion
4.1. Image Capture Timing: Minimizing Color Shifts
4.2. Detection Thresholds: Human vs Analytical Sensitivity
4.3. Nuptial Coloration as a Baseline
4.4. Method Validation in Experiment and in the Field
4.5. Site-Specific Variation: Sex-Related vs Cryptic Standard Sites
4.6. Method Limitations and Future Developments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maan, M.E.; Seehausen, O.; Van Alphen, J.J. Female Mating Preferences and Male Coloration Covary with Water Transparency in a Lake Victoria Cichlid Fish. Biological Journal of the Linnean Society 2010, 99, 398–406. [CrossRef]
- Bergstrom, C.; Whiteley, A.; Tallmon, D. The Heritable Basis and Cost of Colour Plasticity in Coastrange Sculpins. Journal of Evolutionary Biology 2012, 25, 2526–2536. [CrossRef]
- Atsumi, K.; Koizumi, I. Web Image Search Revealed Large-Scale Variations in Breeding Season and Nuptial Coloration in a Mutually Ornamented Fish, Tribolodon Hakonensis. Ecological Research 2017, 32, 567–578.
- Morrongiello, J.; Bond, N.; Crook, D.; Wong, B. Nuptial Coloration Varies with Ambient Light Environment in a Freshwater Fish. Journal of Evolutionary Biology 2010, 23, 2718–2725. [CrossRef]
- Nguyen, C.-N.; Vo, V.-T.; Nhan, H.T.; Nguyen, C.-N.; others In Situ Measurement of Fish Color Based on Machine Vision: A Case Study of Measuring a Clownfish’s Color. Measurement 2022, 197. [CrossRef]
- Korzan, W.J.; Robison, R.R.; Zhao, S.; Fernald, R.D. Color Change as a Potential Behavioral Strategy. Hormones and behavior 2008, 54, 463–470.
- Guillot, R.; Ceinos, R.M.; Cal, R.; Rotllant, J.; Cerda-Reverter, J.M. Transient Ectopic Overexpression of Agouti-Signalling Protein 1 (Asip1) Induces Pigment Anomalies in Flatfish. PloS one 2012, 7. [CrossRef]
- Brady, P.C.; Gilerson, A.A.; Kattawar, G.W.; Sullivan, J.M.; Twardowski, M.S.; Dierssen, H.M.; Gao, M.; Travis, K.; Etheredge, R.I.; Tonizzo, A.; et al. Open-Ocean Fish Reveal an Omnidirectional Solution to Camouflage in Polarized Environments. Science 2015, 350, 965–969. [CrossRef]
- Ceinos, R.M.; Guillot, R.; Kelsh, R.N.; Cerdá-Reverter, J.M.; Rotllant, J. Pigment Patterns in Adult Fish Result from Superimposition of Two Largely Independent Pigmentation Mechanisms. Pigment Cell Melanoma Res. 2015, 28, 196–209. [CrossRef]
- Schweitzer, C.; Motreuil, S.; Dechaume-Moncharmont, F.-X. Coloration Reflects Behavioural Types in the Convict Cichlid, Amatitlania Siquia. Animal Behaviour 2015, 105, 201–209. [CrossRef]
- Morais, S.; Aragão, C.; Cabrita, E.; Conceição, L.E.; Constenla, M.; Costas, B.; Dias, J.; Duncan, N.; Engrola, S.; Estevez, A. New Developments and Biological Insights into the Farming of Solea Senegalensis Reinforcing Its Aquaculture Potential. Reviews in Aquaculture 2016, 8, 227–263. [CrossRef]
- Utagawa, U.; Higashi, S.; Kamei, Y.; Fukamachi, S. Characterization of Assortative Mating in Medaka: Mate Discrimination Cues and Factors That Bias Sexual Preference. Hormones and Behavior 2016, 84, 9–17. [CrossRef]
- Cal, L.; Suarez-Bregua, P.; Comesaña, P.; Owen, J.; Braasch, I.; Kelsh, R.; Cerdá-Reverter, J.M.; Rotllant, J. Countershading in Zebrafish Results from an Asip1 Controlled Dorsoventral Gradient of Pigment Cell Differentiation. Sci Rep 2019, 9.
- Sato, A.; Aihara, R.; Karino, K. Male Coloration Affects Female Gestation Period and Timing of Fertilization in the Guppy (Poecilia Reticulata). Plos one 2021, 16. [CrossRef]
- Boughman, J.W. Divergent Sexual Selection Enhances Reproductive Isolation in Sticklebacks. Nature 2001, 411, 944–948. [CrossRef]
- Lewandowski, E.; Boughman, J. Effects of Genetics and Light Environment on Colour Expression in Threespine Sticklebacks: Quantitative Genetics of Nuptial Colour. Biological Journal of the Linnean Society 2008, 94, 663–673.
- Tibblin, P.; Hall, M.; Svensson, P.A.; Merilä, J.; Forsman, A. Phenotypic Flexibility in Background-Mediated Color Change in Sticklebacks. Behavioral Ecology 2020, 31, 950–959. [CrossRef]
- Endler, J.A. Signals, Signal Conditions, and the Direction of Evolution. The American Naturalist 1992, 139, 125–153.
- Price, A.C.; Weadick, C.J.; Shim, J.; Rodd, F.H. Pigments, Patterns, and Fish Behavior. Zebrafish 2008, 5, 297–307.
- Phillips, B.L. The Evolution of Growth Rates on an Expanding Range Edge. Biol. Lett. 2009, 5, 802–804. [CrossRef]
- John, L.; Rick, I.P.; Vitt, S.; Thünken, T. Body Coloration as a Dynamic Signal during Intrasexual Communication in a Cichlid Fish. Bmc Zoology 2021, 6, 9. [CrossRef]
- Yoshioka, S.; Matsuhana, B.; Tanaka, S.; Inouye, Y.; Oshima, N.; Kinoshita, S. Mechanism of Variable Structural Colour in the Neon Tetra: Quantitative Evaluation of the Venetian Blind Model. Journal of the Royal Society Interface 2011, 8, 56–66.
- FitzGerald, G.J. The Reproductive Behavior of the Stickleback. Scientific American 1993, 268, 80–85.
- Bell, M.A.; Foster, S.A. Introduction to the Evolutionary Biology of the Threespine Stickleback. The evolutionary biology of the threespine stickleback 1994, 1–27.
- Hiermes, M.; Bakker, T.C.; Mehlis, M.; Rick, I.P. Context-Dependent Dynamic UV Signaling in Female Threespine Sticklebacks. Scientific reports 2015, 5. [CrossRef]
- Reimchen, T.E. Loss of Nuptial Color in Threespine Sticklebacks (Gasterosteus Aculeatus). Evolution 1989, 43, 450–460. [CrossRef]
- Clarke, J.M.; Schluter, D. Colour Plasticity and Background Matching in a Threespine Stickleback Species Pair: Colour Plasticity And Background Matching. Biological Journal of the Linnean Society 2011, 102, 902–914.
- Tapanes, E.; Rennison, D.J. The Genetic Basis of Divergent Melanic Pigmentation in Benthic and Limnetic Threespine Stickleback. Heredity 2024, 133, 207–215. [CrossRef]
- Sokołowska, E.; Kulczykowska, E. A New Insight into the Pigmentation of the Three-Spined Stickleback Exposed to Oxidative Stress: Day and Night Study. Frontiers in Marine Science 2024, 11.
- Bakker, T.C.; Milinski, M. The Advantages of Being Red: Sexual Selection in the Stickleback. Marine & Freshwater Behaviour & Phy 1993, 23, 287–300.
- Barber, I.; Arnott, S.A.; Braithwaite, V.; Andrew, J.; Mullen, W.; Huntingford, F. Carotenoid-Based Sexual Coloration and Body Condition in Nesting Male Sticklebacks. Journal of Fish Biology 2000, 57, 777–790.
- Candolin, U. Changes in Expression and Honesty of Sexual Signalling over the Reproductive Lifetime of Sticklebacks. Proceedings of the Royal Society of London. Series B: Biological Sciences 2000, 267, 2425–2430. [CrossRef]
- Chiara, V.; Velando, A.; Kim, S.-Y. Relationships between Male Secondary Sexual Traits, Physiological State and Offspring Viability in the Three-Spined Stickleback. BMC ecology and evolution 2022, 22, 4. [CrossRef]
- Wright, D.S.; Yong, L.; Pierotti, M.E.; McKinnon, J.S. Male Red Throat Coloration, Pelvic Spine Coloration, and Courtship Behaviours in Threespine Stickleback. Evolutionary Ecology Research 2016, 17, 407–418.
- Anderson, C.M.; McKinnon, J.S. Phenotypic Correlates of Pelvic Spine Coloration in the Threespine Stickleback (Gasterosteus Aculeatus): Implications for Function and Evolution. Behavioral ecology and sociobiology 2022, 76, 153. [CrossRef]
- Ivanova, T.; Ivanov, M.; Golovin, P.; Polyakova, N.; Lajus, D. The White Sea Threespine Stickleback Population: Spawning Habitats, Mortality, and Abundance. Evolutionary ecology research 2016, 17, 301–315.
- Lajus, D.L.; Golovin, P.V.; Zelenskaia, A.E.; Demchuk, A.S.; Dorgham, A.S.; Ivanov, M.V.; Ivanova, T.S.; Murzina, S.A.; Polyakova, N.V.; Rybkina, E.V.; et al. Threespine Stickleback of the White Sea: Population Characteristics and Role in the Ecosystem. Contemp. Probl. Ecol. 2020, 13, 132–145. [CrossRef]
- Lajus, D.; Ivanova, T.; Rybkina, E.; Lajus, J.; Ivanov, M. Multidecadal Fluctuations of Threespine Stickleback in the White Sea and Their Correlation with Temperature. ICES Journal of Marine Science 2021, 78, 653–665. [CrossRef]
- Ivanov, M. (St. Petersburg State University, St. Petersburg, Russian Federation). Personal communication 2021.
- Genelt-Yanovskaya, A.S.; Polyakova, N.V.; Ivanov, M.V.; Nadtochii, E.V.; Ivanova, T.S.; Genelt-Yanovskiy, E.A.; Tiunov, A.V.; Lajus, D.L. Tracing the Food Web of Changing Arctic Ocean: Trophic Status of Highly Abundant Fish, Gasterosteus Aculeatus (L.), in the White Sea Recovered Using Stomach Content and Stable Isotope Analyses. Diversity 2022, 14, 955. [CrossRef]
- Ivanov, M.; Zelenskaia, A.E.; Demchuk, A.S.; Ivanova, T.S.; Lajus, D.L. Threespine stickleback Gasterosteus aculeatus as a link between the inshore and offshore communities of the White Sea.; Novosibirsk State Agrarian University: Novosibirsk, 2021; pp. 99–103.
- Yurtseva, A.; Noreikiene, K.; Lajus, D.; Li, Z.; Alapassi, T.; Ivanova, T.; Ivanov, M.; Golovin, P.; Vesala, S.; Merilä, J. Aging Three-Spined Sticklebacks Gasterosteus Aculeatus: Comparison of Estimates from Three Structures. Journal of fish biology 2019, 95, 802–811. [CrossRef]
- Dorgham, A.; Candolin, U.; Ivanova, T.; Ivanov, M.; Nadtochii, E.; Yurtseva, A.; Lajus, D. Sexual Dimorphism Patterns of the White Sea Threespine Stickleback (Gasterosteus Aculeatus). Biological Communications 2021, 256–267. [CrossRef]
- Schewe, J. The Digital Print: Preparing Images in Lightroom and Photoshop for Printing; Peachpit Press, United States, 2013.
- Steeper, P. How to Achieve Accurate Color from Your Camera. PSA Journal 2012, 78, 18–20.
- Abràmoff, M.D.; Magalhães, P.J.; Ram, S.J. Image Processing with ImageJ. Biophotonics international 2004, 11, 36–42.
- Ferreira, T.; Rasband, W. ImageJ User Guide. ImageJ/Fiji 2012, 1, 155–161.
- Ly, B.C.K.; Dyer, E.B.; Feig, J.L.; Chien, A.L.; Del Bino, S. Research Techniques Made Simple: Cutaneous Colorimetry: A Reliable Technique for Objective Skin Color Measurement. Journal of Investigative Dermatology 2020, 140.
- Voss, D.H. Relating Colorimeter Measurement of Plant Color to the Royal Horticultural Society Colour Chart. HortScience HortSci 1992, 27, 1256–1260. [CrossRef]
- ChromaChecker Available online: https://chromachecker.com/manuals/en/show/chromaspot (accessed on 23 May 2025).
- Kekäläinen, J.; Huuskonen, H.; Kiviniemi, V.; Taskinen, J. Visual Conditions and Habitat Shape the Coloration of the Eurasian Perch (Perca Fluviatilis L.): A Trade-off between Camouflage and Communication? Biological Journal of the Linnean Society 2010, 99, 47–59.
- Martin, C.H. Strong Assortative Mating by Diet, Color, Size, and Morphology but Limited Progress toward Sympatric Speciation in a Classic Example: Cameroon Crater Lake Cichlids. Evolution 2013, 67, 2114–2123.
- Sundin, J.; Vossen, L.E.; Nilsson-Sköld, H.; Jutfelt, F. No Effect of Elevated Carbon Dioxide on Reproductive Behaviors in the Three-Spined Stickleback. Behavioral Ecology 2017, 28, 1482–1491. [CrossRef]
- Troscianko, J.; Stevens, M. Image Calibration and Analysis Toolbox–a Free Software Suite for Objectively Measuring Reflectance, Colour and Pattern. Methods in Ecology and Evolution 2015, 6, 1320–1331.
- Mahy, M.; Van Eycken, L.; Oosterlinck, A. Evaluation of Uniform Color Spaces Developed after the Adoption of CIELAB and CIELUV. Color Research & Application 1994, 19, 105–121. [CrossRef]
- Hammer, Ø.; Harper, D.A.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontologia electronica 2001, 4, 9.
- Love, J.; Selker, R.; Marsman, M.; Jamil, T.; Dropmann, D.; Verhagen, J.; Ly, A.; Gronau, Q.F.; Šmíra, M.; Epskamp, S. JASP: Graphical Statistical Software for Common Statistical Designs. Journal of Statistical Software 2019, 88, 1–17. [CrossRef]
- Python 3.13 Documentation. Python Software Foundation, Wilmington, Delaware, United States. Available online: https://docs.python.org/3/ (accessed on 23 May 2025).
- Anaconda.Org. Anaconda Inc., Austin, Texas, United States. Available online: https://www.anaconda.com/docs/tools/anaconda-org/main (accessed on 23 May 2025).
- R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Available online: https://www.R-project.org/ (accessed on 23 May 2025).
- RStudio: Integrated Development Environment for R. Posit Software, PBC, Boston, MA, United States. Available online: http://www.posit.co/ (accessed on 23 May 2025).
- Colihueque, N.; Parraguez, M.; Estay, F.J.; Diaz, N.F. Skin Color Characterization in Rainbow Trout by Use of Computer-Based Image Analysis. North American Journal of Aquaculture 2011, 73, 249–258. [CrossRef]
- Wedekind, C.; Meyer, P.; Frischknecht, M.; Niggli, U.A.; Pfander, H. Different Carotenoids and Potential Information Content of Red Coloration of Male Three-Spined Stickleback. Journal of Chemical Ecology 1998, 24, 787–801.
- Bolnick, D.I.; Shim, K.C.; Schmerer, M.; Brock, C.D. Population-Specific Covariation between Immune Function and Color of Nesting Male Threespine Stickleback. PloS one 2015, 10. [CrossRef]
- Franco-Belussi, L.; De Oliveira, C.; Sköld, H. Regulation of Eye and Jaw Colouration in Three-Spined Stickleback Gasterosteus Aculeatus. Journal of Fish Biology 2018, 92, 1788–1804. [CrossRef]
- Stevens, M.; Lown, A.E.; Denton, A.M. Rockpool Gobies Change Colour for Camouflage. PLoS One 2014, 9.
- Smithers, S.P.; Rooney, R.; Wilson, A.; Stevens, M. Rock Pool Fish Use a Combination of Colour Change and Substrate Choice to Improve Camouflage. Animal Behaviour 2018, 144, 53–65. [CrossRef]
- Ramachandran, V.; Tyler, C.; Gregory, R.; Rogers-Ramachandran, D.; Duensing, S.; Pillsbury, C.; Ramachandran, C. Rapid Adaptive Camouflage in Tropical Flounders. Nature 1996, 379, 815–818.
- Tyrie, E.K.; Hanlon, R.T.; Siemann, L.A.; Uyarra, M.C. Coral Reef Flounders, Bothus Lunatus, Choose Substrates on Which They Can Achieve Camouflage with Their Limited Body Pattern Repertoire. Biological Journal of the Linnean Society 2015, 114, 629–638. [CrossRef]
- John, L.; Santon, M.; Michiels, N.K. Scorpionfish Rapidly Change Colour in Response to Their Background. Frontiers in Zoology 2023, 20, 10.
- Candolin, U. The Relationship between Signal Quality and Physical Condition: Is Sexual Signalling Honest in the Three-Spined Stickleback? Animal behaviour 1999, 58, 1261–1267. [CrossRef]
- French, C.M.; Ingram, T.; Bolnick, D.I. Geographical Variation in Colour of Female Threespine Stickleback (Gasterosteus Aculeatus). PeerJ 2018, 6.
- MacAdam, D.L. Visual Sensitivities to Color Differences in Daylight. Journal of the Optical Society of America 1942, 32, 247–274. [CrossRef]
- Leinonen, T.; Cano, J.M.; Merilä, J. Genetics of Body Shape and Armour Variation in Threespine Sticklebacks. Journal of Evolutionary Biology 2011, 24, 206–218. [CrossRef]
- Reimchen, T.; Steeves, D.; Bergstrom, C. Sex Matters for Defence and Trophic Traits of Threespine Stickleback. Evolutionary Ecology Research 2016, 17, 459–485.
- Münzing, J. The Evolution of Variation and Distributional Patterns in European Populations of the Three-Spined Stickleback, Gasterosteus Aculeatus. Evolution 1963, 320–332.
- Bell, M.A. Evolution of Phenotypic Diversity in Gasterosteus Aculeatus Superspecies on the Pacific Coast of North America. Systematic Zoology 1976, 25, 211–227. [CrossRef]
- Jenck, C.S.; Lehto, W.R.; Ketterman, B.T.; Sloan, L.F.; Sexton, A.N.; Tinghitella, R.M. Phenotypic Divergence among Threespine Stickleback That Differ in Nuptial Coloration. Ecology and Evolution 2020, 10, 2900–2916. [CrossRef]
- Tinbergen, N. The Curious Behavior of the Stickleback. Scientific American 1952, 187, 22–27. [CrossRef]
- Milinski, M.; Bakker, T.C.M. Female Sticklebacks Use Male Coloration in Mate Choice and Hence Avoid Parasitized Males. Nature 1990, 344, 330–333. [CrossRef]
- Zyuganov, V.V. Fauna of the USSR. Fishes. The Sticklebacks (Gasterosteidae) of the World Fauna (Fauna SSSR. Ryby. T. 5. Vyp. 1. Semeistvo Koliushkovykh (Gasterosteidae) Mirovoi Fauny). 1991, 5.
- McKinnon, J.; Demayo, R.; Granquist, R.; Weggel, L. Female Red Throat Coloration in Two Populations of Threespine Stickleback. Behaviour 2000, 137, 947–963.
- Yong, L.; Guo, R.; Wright, D.S.; Mears, S.A.; Pierotti, M.; McKinnon, J.S. Correlates of Red Throat Coloration in Female Stickleback and Their Potential Evolutionary Significance. Evolutionary Ecology Research 2013, 15, 453–472.
- von Hippel, F.A. Black Male Bellies and Red Female Throats: Color Changes with Breeding Status in a Threespine Stickleback. Environmental Biology of Fishes 1999, 55, 237–244.
- Semler, D.E. Some Aspects of Adaptation in a Polymorphism for Breeding Colours in the Threespine Stickleback (Gasterosteus Aculeatus). Journal of Zoology 1971, 165, 291–302. [CrossRef]
- Yong, L.; Peichel, C.L.; McKinnon, J.S. Genetic Architecture of Conspicuous Red Ornaments in Female Threespine Stickleback. G3: Genes, Genomes, Genetics 2016, 6, 579–588. [CrossRef]
- Dorgham, A.S.A.; Golovin, P.; Ivanova, T.; Ivanov, M.; Saveliev, P.; Lajus, D. Morphological Variation of Threespine Stickleback (Gasterosteus Aculeatus) on Different Stages of Spawning Period. Proceedings of KarRC of RAS 2018, 59.
- Mclennan, D.A. Integrating Phylogenetic and Experimental Analyses: the Evolution of Male and Female Nuptial Coloration in the Stickleback Fishes (Gasterosteidae). Systematic Biology 1996, 45, 17.
- Hulslander, C.L. The Evolution of the Male Threespine Stickleback Color Signal. PhD Thesis, Clark University, Worcester, Massachusetts, United States, 2003.
- Kraak, S.; Bakker, T.; Hočevar, S. Stickleback Males, Especially Large and Red Ones, Are More Likely to Nest Concealed in Macrophytes. Behaviour 2000, 137, 907–919. [CrossRef]
- Levine, J.S.; MacNichol, E.F. Color Vision in Fishes. Scientific American 1982, 246, 140–149.
- Rennison, D.J.; Owens, G.L.; Heckman, N.; Schluter, D.; Veen, T. Rapid Adaptive Evolution of Colour Vision in the Threespine Stickleback Radiation. Proceedings of the Royal Society B: Biological Sciences 2016, 283. [CrossRef]
- Wyszecki, G.; Stiles, W.S. Color Science: Concepts and Methods, Quantitative Data and Formulae; John wiley & sons, Inc., Canada, 2000.
- Schnapf, J.; Kraft, T.; Baylor, D. Spectral Sensitivity of Human Cone Photoreceptors. Nature 1987, 325, 439–441. [CrossRef]
- Greenwood, A.K.; Jones, F.C.; Chan, Y.F.; Brady, S.D.; Absher, D.M.; Grimwood, J.; Schmutz, J.; Myers, R.M.; Kingsley, D.M.; Peichel, C.L. The Genetic Basis of Divergent Pigment Patterns in Juvenile Threespine Sticklebacks. Heredity 2011, 107, 155–166. [CrossRef]
- Greenwood, A.K.; Cech, J.N.; Peichel, C.L. Molecular and Developmental Contributions to Divergent Pigment Patterns in Marine and Freshwater Sticklebacks. Evolution & development 2012, 14, 351–362.
- Gygax, M.; Rentsch, A.K.; Rudman, S.M.; Rennison, D.J. Differential Predation Alters Pigmentation in Threespine Stickleback (Gasterosteus Aculeatus). Journal of evolutionary biology 2018, 31, 1589–1598. [CrossRef]
- van den Berg, C.P.; Troscianko, J.; Endler, J.A.; Marshall, N.J.; Cheney, K.L. Quantitative Colour Pattern Analysis (QCPA): A Comprehensive Framework for the Analysis of Colour Patterns in Nature. Methods in Ecology and Evolution 2020, 11, 316–332. [CrossRef]
- Bookstein, F.L. Morphometric Tools for Landmark Data; Cambridge University Press, England, 1992.
- Rohlf, F.J. The Tps Series of Software. Hystrix 2015, 26, 9–12. [CrossRef]
- Hand, D.M.; Brignon, W.R.; Olson, D.E.; Rivera, J. Comparing Two Methods Used to Mark Juvenile Chinook Salmon: Automated and Manual Marking. North American Journal of Aquaculture 2010, 72, 10–17. [CrossRef]
- Petrtýl, M.; Kalous, L.; Memi̇Ş, D. Comparison of Manual Measurements and Computer-Assisted Image Analysis in Fish Morphometry. Turk J Vet Anim Sci 2014, 38, 88–94. [CrossRef]

| SS | Comparisons | ||||||
|---|---|---|---|---|---|---|---|
| Females vs males, experiment | Females vs males, field | Experiment vs field, females | Experiment vs field, males | ||||
| 1 | 6.17±2.77 | 6.54±5.39 | 10.30±3.05 | 9.54±3.28 | |||
| (0.81-17.75) | (0.24-23.51) | (3.35-20.40) | (1.64-21.43) | ||||
| 2 | 13.72±6.54 | 8.99±3.81 | 11.33±4.49 | 13.12±6.55 | |||
| (0.87-38.58) | (0.43-23.64) | (1.11-33.19) | (1.89-41.06) | ||||
| 3 | 11.34±5.64 | 6.15±2.96 | 9.71±3.00 | 11.15±4.27 | |||
| (1.69-32.29) | (0.43-16.09) | (2.74-21.73) | (3.34-25.90) | ||||
| 4 | 14.66±4.49 | 9.00±4.13 | 13.19±5.93 | 17.94±8.31 | |||
| (2.15-26.12) | (0.56-23.43) | (2.69-35.39) | (2.31-44.77) | ||||
| 5 | 9.70±4.33 | 6.81±4.80 | 10.66±3.10 | 12.64±5.59 | |||
| (1.20-30.59) | (0.55-23.74) | (4.45-26.49) | (1.55-49.06) | ||||
| 6 | 17.00±5.96 | 14.81±6.19 | 13.85±6.51 | 11.91±5.58 | |||
| (1.60-31.21) | (1l.68-37.35) | (1.46-41.84) | (1.01-33.43) | ||||
| 7 | 12.12±6.55 | 12.47±6.73 | 14.64±7.28 | 12.15±5.30 | |||
| (1.39-37.82) | (0.22-35.37) | (3.04-44.43) | (1.61-31.25) | ||||
| 8 | 10.67±3.99 | 7.89±3.86 | 8.94±3.43 | 10.23±4.85 | |||
| (1.45-23.03) | (0.69-21.73) | (1.03-19.83) | (0.64-27.50) | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




