Submitted:
27 May 2025
Posted:
28 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
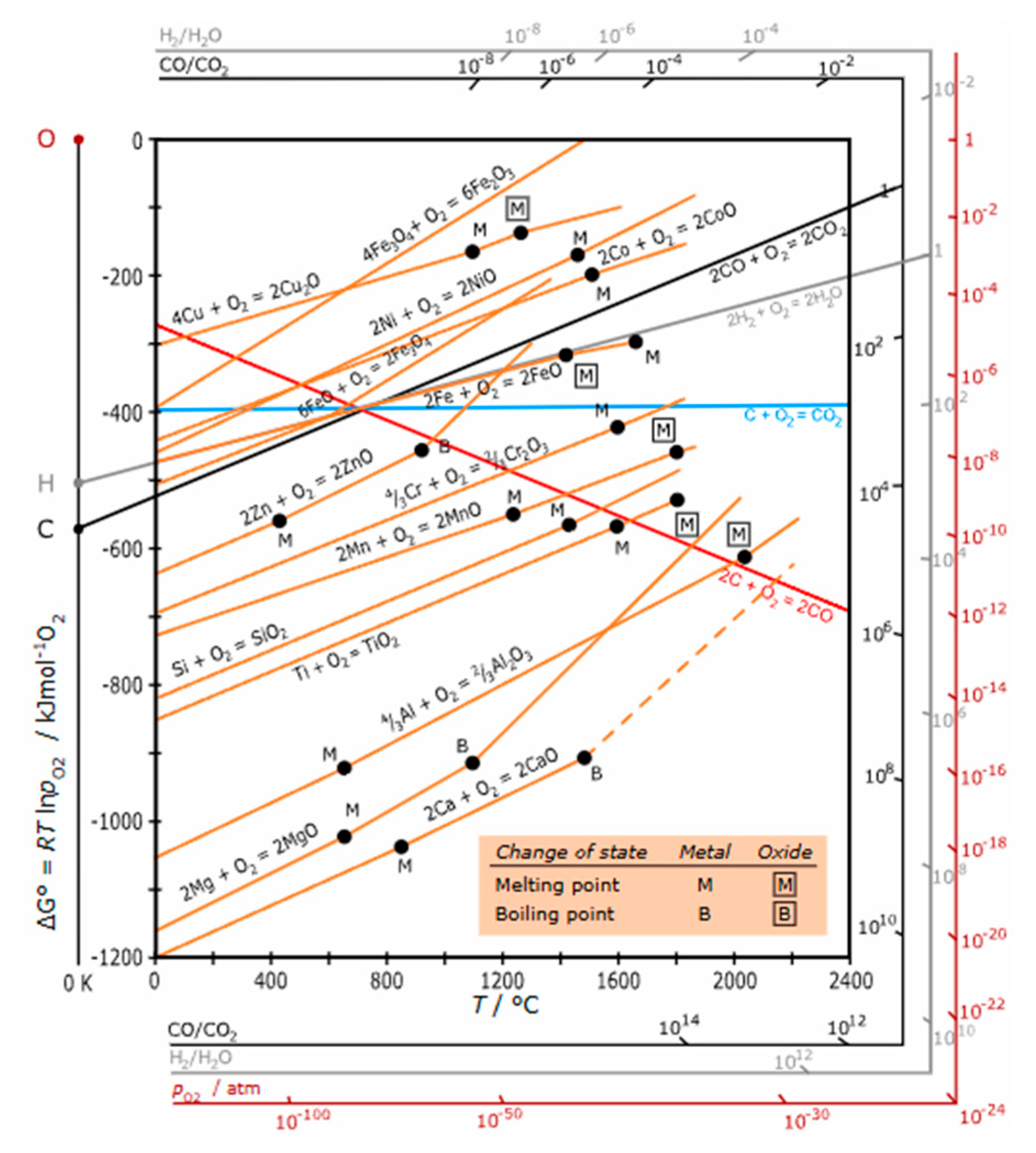
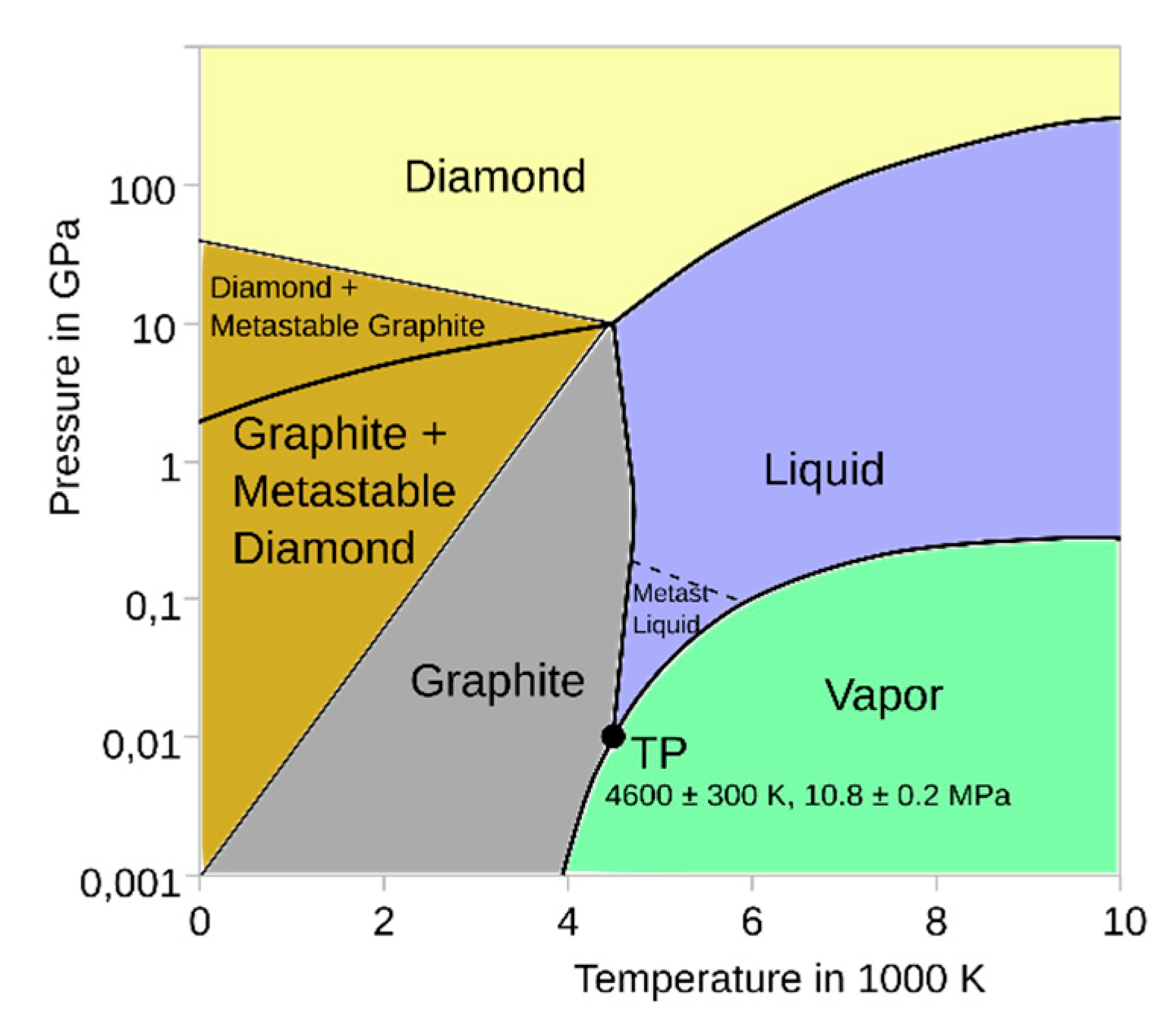
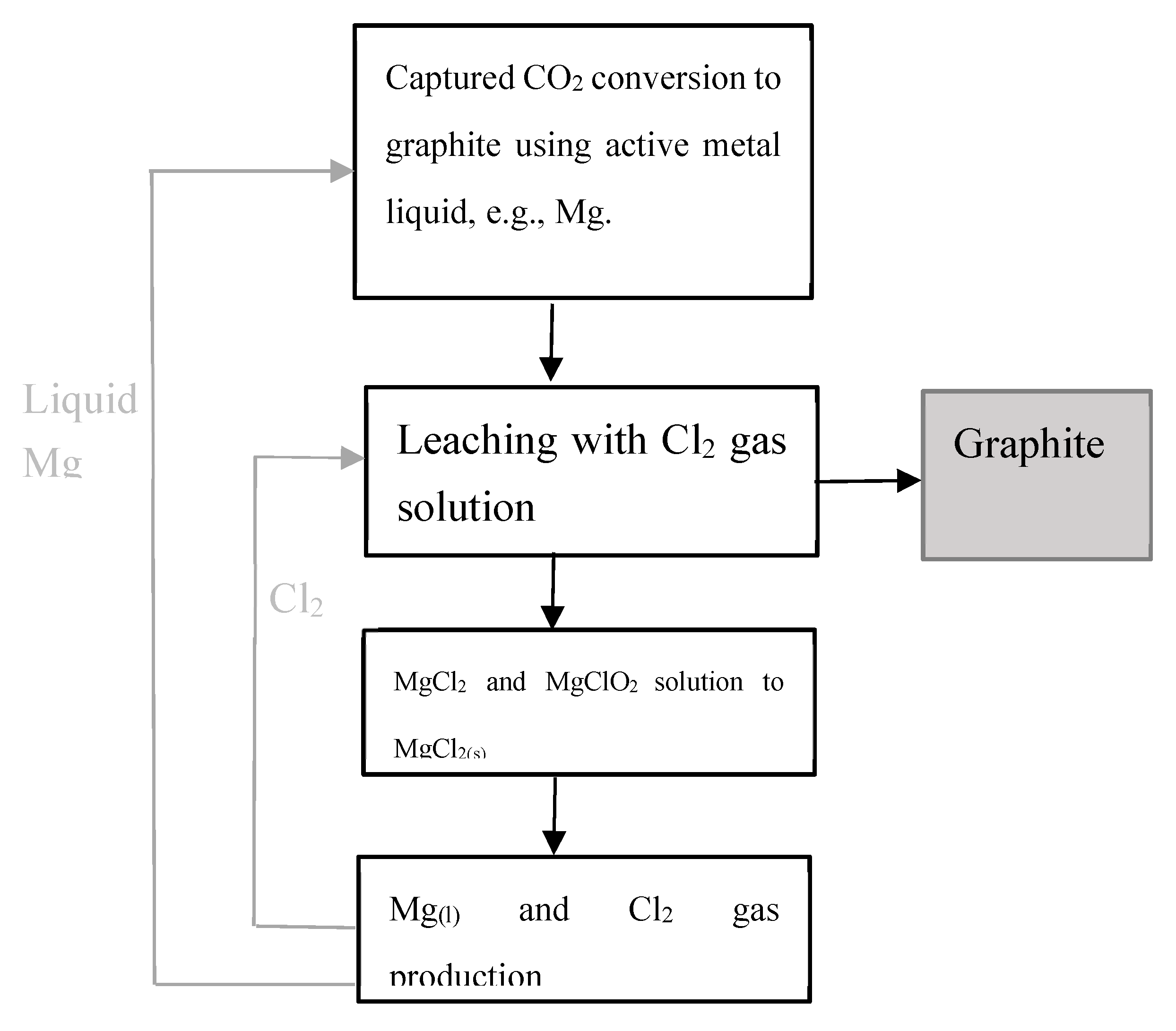
2. CO₂ removal and conversion to graphite via metallurgy process
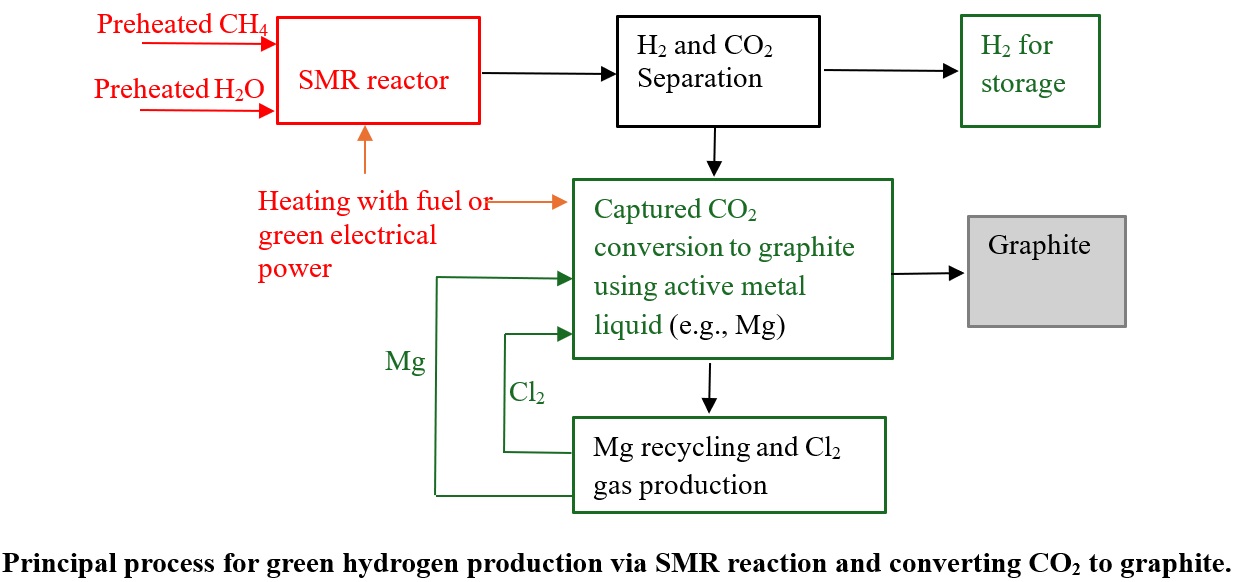
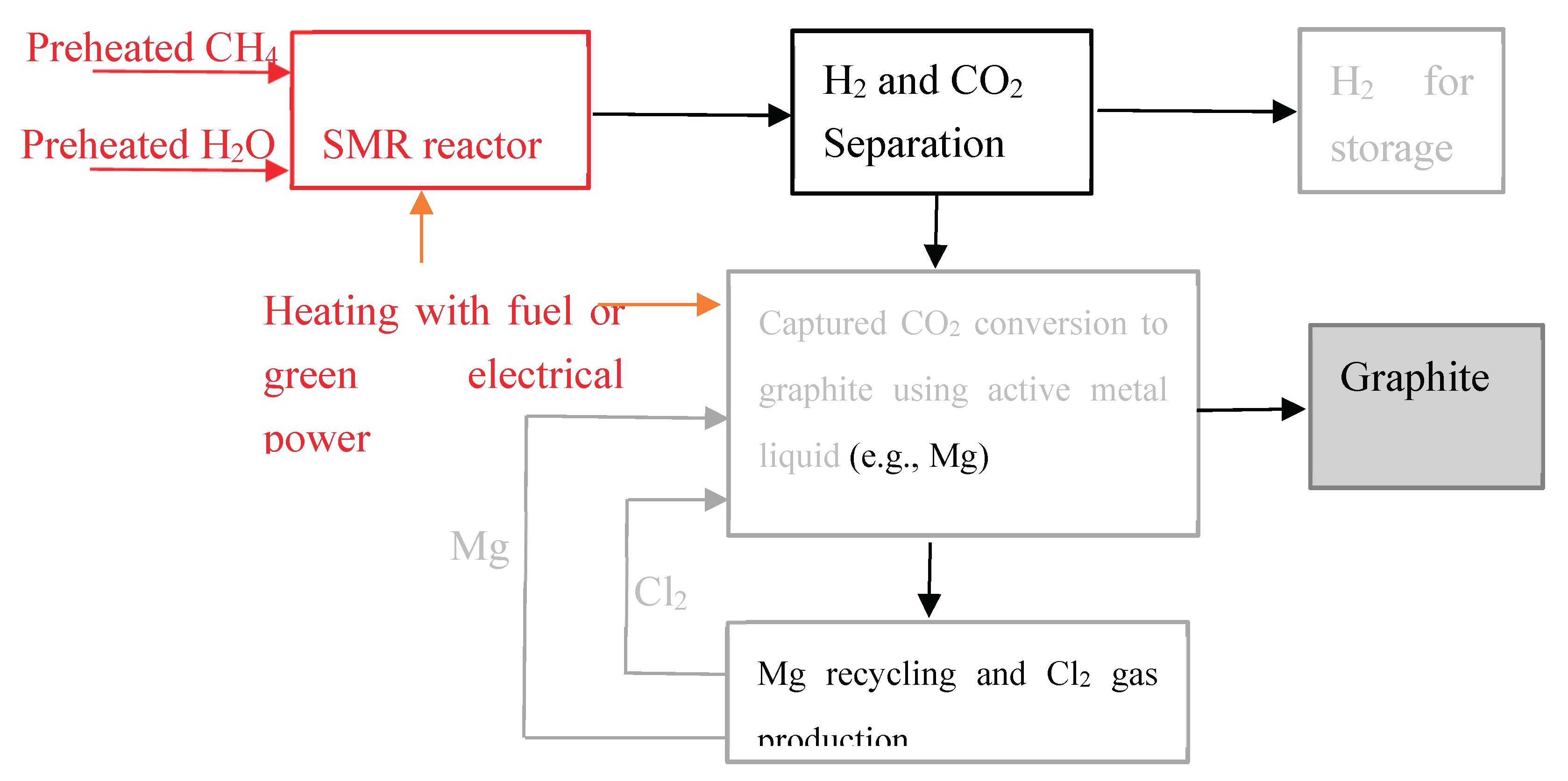
3. Green hydrogen production via SMR process combined with CO₂ conversion to graphite
4. Conclusions
Author Contributions
Acknowledgements
Disclosure Statement
References
- Monastersky, R. Global carbon dioxide levels near worrisome milestone. Nature 497, 13–14 (2013). [CrossRef]
- Yamasaki, A. An overview of CO2 mitigation options for global warming -Emphasizing CO2 sequestration options. Journal of Chemical Engineering of Japan 36, 361–375 (2003). [CrossRef]
- Global Carbon Budget Report 2023. https://globalcarbonbudget.org/fossil-co2-emissions-at-record-high-in-2023/.
- Lindsey, R. (2024) Climate change: Atmospheric carbon dioxide, NOAA Climate.gov. Available at: https://www.climate.gov/news-features/understanding-climate/climate-change-atmospheric-carbon-dioxide (accessed on 23 May 2025).
- Aini, N. and Shen, Z. The effect of tree planting within roadside green space on dispersion of CO2 from transportation. International Review for Spatial Planning and Sustainable Development 7, 97–112 (2019). [CrossRef]
- Fornaciari, M., Muscas, D., Rossi, F., Filipponi, M., Castellani, B., Di Giuseppe, A., Proietti, C., Ruga, L., Fabio O. CO2 Emission Compensation by Tree Species in Some Urban Green Areas. Sustainability. 6, p.3515 (2019). [CrossRef]
- Rasowo, J. O., Nyonje, B., Olendi, R., Orina, P., Odongo, S. Towards environmental sustainability: further evidences from decarbonization projects in Kenya’s Blue Economy. Frontiers in Marine Science. 11, 1239862 (2024). [CrossRef]
- Hurd, C. L., Law, C. S., Bach, L. T., Britton, D., Hovenden, M., Paine, E. R., John A. R., Veronica T., Philip W. B. Forensic carbon accounting: Assessing the role of seaweeds for carbon sequestration. Journal of phycology. 8, 347-363 (2022). [CrossRef]
- Awogbemi, O., Dawood A. D. Novel technologies for CO2 conversion to renewable fuels, chemicals, and value-added products. Discover nano. 20, 29-27(2025). [CrossRef]
- Porosoff, M. D., Yan, B., Chen, J. G. Catalytic Reduction of CO2 by H2 for Synthesis of CO, Methanol and Hydrocarbons: Challenges and Opportunities. Energy Environ. Sci. 9, 62–73 (2016). [CrossRef]
- Zhou, J., Huang, L., Yan, W., Li, J., Liu, C., Lu, X. Theoretical study of the mechanism for CO2 hydrogenation to methanol catalyzed by trans-RuH2(CO)(dpa), Catalysts. 8, 244-253 (2018). [CrossRef]
- Olah, G. A., Goeppert, A., Prakash, G. K. S. Chemical Recycling of Carbon Dioxide to Methanol and Dimethyl Ether: From Greenhouse Gas to Renewable, Environmentally Carbon Neutral Fuels and Synthetic Hydrocarbons. J. Org. Chem. 74, 487–498 (2009). [CrossRef]
- Kortlever R., Balemans C., Kwon Y., Koper M. T. M. Electrochemical CO2 reduction to formic acid on a Pd-based formic acid oxidation catalyst. Catal Today. 244, 58–62 (2015). [CrossRef]
- Moret, S., Dyson, P. J., Laurenczy, G. Direct Synthesis of Formic Acid from Carbon Dioxide by Hydrogenation in Acidic Media. Nat. Commun. 5, 1–7 (2014). [CrossRef]
- Cai, T. et al. Cell-free chemoenzymatic starch synthesis from carbon dioxide. Science. 373, 1523-1527 (2021). [CrossRef]
- Yang, J. et al. De novo artificial synthesis of hexoses from carbon dioxide. Science bulletin. 68, 2370-2381 (2023). [CrossRef]
- Rochelle, G.T. Amine scrubbing for CO2 capture. Science. 325, 1652–1654 (2009). [CrossRef]
- Luo, X., Guo, Y., Ding, F., Zhao, H., Cui, G., Li, H., Wang, C. Significant improvements in CO2 capture by pyridine-containing anion-functionalized ionic liquids through multiple-site cooperative interactions. Angew. Chem. Int. Ed. 53, 7053–7057 (2014). [CrossRef]
- Angelo, V., Davide, A., Nina-Luisa, M., Tomas, A., Kim, T., Gerald, B. Sorbent material for CO2 capture, uses thereof and methods for making same. Patent application No. WO-2023094386-A1 (2023).
- Querejeta, N., Gil, M.V., Rubiera, F., Pevida, C., Wawrzyn´czak, D., Panowski, M., Majchrzak-Kucęb, I. Bio-engineering of carbon adsorbents to capture CO2 from industrial sources: The cement case. Separation and Purification Technology. 330, 125407 (2024). [CrossRef]
- Bai, J. et al. Sulfur-Doped porous carbon Adsorbent: A promising solution for effective and selective CO2 capture. Chemical Engineering Journal. 479, 147667 (2023). [CrossRef]
- Rao, L., Ma, R., Liu, S., Wang, L., Wu, Z., Yang, J., Hu, X. Nitrogen enriched porous carbons from D-glucose with excellent CO2 capture performance. Chemical Engineering Journal. 362, 794-801 (2019). [CrossRef]
- Hou, M., Li, L., Xu, R., Lu, Y., Song, J., Jiang, Z., Wang, T., Jian, X. Precursor-chemistry engineering toward ultrapermeable carbon molecular sieve membrane for CO2 capture. Journal of energy chemistry. 102, 421-430 (2025). [CrossRef]
- Sabouni, R., Kazemian, H., Rohani, S. Carbon dioxide capturing technologies: a review focusing on metal organic framework materials (MOFs). Environ Sci Pollut Res. 21, 5427–5449 (2014). [CrossRef]
- Millward, A.R., Yaghi, O.M. Metal-organic frameworks with exceptionally high capacity for storage of carbon dioxide at room temperature. J. Am. Chem. Soc. 127, 17998–17999 (2005). [CrossRef]
- Peplow, M. The race to recycle carbon dioxide. Nature (London), 603 (7903), 780-783 (2022). [CrossRef]
- Daud, F. D. M., Ahmad, N. A. I., Mahmud, M. S., Sariffudin, N., Zaki, H. H. M. Two-Step Synthesis of Ca-Based MgO Hybrid Adsorbent for Potential CO2 Capturing Application. Materials science forum. 981, 369-374 (2020). [CrossRef]
- Pacciani, R., Müller, C. R., Davidson, J. F., Dennis, J. S., Hayhurst, A. N. Synthetic Ca-based solid sorbents suitable for capturing CO2 in a fluidized bed. Canadian journal of chemical engineering. 86, 356-366 (2008). [CrossRef]
- Zeng, H., Zhang, Y., Liu, Qi. Wang, K. Process for capture and conversion of carbon dioxide. US Patent Application No. 63/487,510; International Application No. PCT/CA2024/050248 (2024). https://worldwide.espacenet.com/patent/search/family/092589050/publication/WO2024178508A1?q=PCT%2FCA2024%2F050248.
- Ellingham diagram. Wikipedia. https://en.wikipedia.org/wiki/Ellingham_diagram (accessed on 7 May 2025).
- Ellingham, H. J. T. Reducibility of oxides and sulphides in metallurgical processes. Journal of the Society of Chemical Industry 63, 125-160 (1944).
- Carbon-phase-diagramp.svg. Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Carbon-phase-diagramp.svg (accessed on 7 May 2025).
- Weast, R. C. et al. CRC Handbook of Chemistry and Physics (59th ed.). West Palm Beach, FL: CRC Press. p. B-133. ISBN 0-8493-0549-8 (1978). [CrossRef]
- Kim, T. H., Merritt, C. R., Ducati, C., Bond, A. D., Bampos, N., Brown, C. L. Bulk synthesis of graphene-like materials possessing turbostratic graphite and graphene nanodomains via combustion of magnesium in carbon dioxide. Carbon. 149, 582-586 (2019). [CrossRef]
- Li, X., Shi, H., Wang, X., Hu, X., Xu, C., Shao, W. Direct synthesis and modification of graphene in Mg melt by converting CO2: A novel route to achieve high strength and stiffness in graphene/Mg composites. Carbon. 186, 632-643 (2022). [CrossRef]
- Kim, T. H., Merritt, C. R., Ducati, C., Bond, A. D., Bampos, N., Brown, C.L. Bulk synthesis of graphene-like materials possessing turbostratic graphite and graphene nanodomains via combustion of magnesium in carbon dioxide. Carbon. 149, 582–586 (2019). [CrossRef]
- Che, Y., Zhang, C., Song, J., Shang, X., Chen, X., He, J. The silicothermic reduction of magnesium in flowing argon and numerical simulation of novel technology. Journal of magnesium and alloys. 8, 752-760 (2020). [CrossRef]
- Hanusa, T. P., Phillips, C. S. G. Alkaline-earth metal. Britannica. https://www.britannica.com/science/alkaline-earth-metal.
- Zuraiqi, K. et al. Direct conversion of CO2 to solid carbon by Ga-based liquid metals. Energy & environmental science. 15, 595-600 (2022). [CrossRef]
- Jaganmohan, M. https://www.statista.com/statistics/1445336/production-of-gallium-worldwide/.
- Hu, L., Song, Y., Jiao, S., Liu, Y., Ge, J., Jiao, H., Zhu, J., Wang, J., Zhu, H., Fray, D. J. Direct conversion of greenhouse gas CO2 into graphene via molten salts electrolysis, ChemSusChem. 9 588–94 (2016). [CrossRef]
- Barelli, L., Bidini, G., Gallorini, F., Servili, S. Hydrogen production through sorption-enhanced steam methane reforming and membrane technology: A review. Energy. 33, 554-570 (2008). [CrossRef]
- Khan, A., Haider, M., Daiyan, R., Neal, P., Haque, N., MacGill, I., Amal, R. A framework for assessing economics of blue hydrogen production from steam methane reforming using carbon capture storage & utilisation. International journal of hydrogen energy. 46, 22685-22706 (2021). [CrossRef]
- Fulcheri, L., Rohani, V., Wyse, E., Hardman, N., Dames, E. An energy-efficient plasma methane pyrolysis process for high yields of carbon black and hydrogen. International journal of hydrogen energy. 48, 2920-2928 (2023). [CrossRef]
- Anwar, S., Khan, F., Zhang, Y., Djire, A. Recent Development in Electrocatalysts for Hydrogen Production through Water Electrolysis. International Journal of Hydrogen Energy. 46, 32284-32317 (2021). [CrossRef]
- Cermak, M., Perez, N., Collins, M., Bahrami, M. Material properties and structure of natural graphite sheet. Scientific reports. 10, 18672-18672 (2020). [CrossRef]
- Duan, S. et al. Preparation and properties of graphite-based “light–heat–electricity” conversion materials. Applied physics letters. 125, (2024). [CrossRef]
- Ghosh, A. K. Literature quest and survey on graphite beneficiation through flotation. Renewable & sustainable energy reviews. 189, 113980 (2024). [CrossRef]
- Zhang, X., Zhang, L., Qu, X., Qiu, Y. Beneficiation of a low-grade flaky graphite ore from australia by flotation. Advanced Materials Research. 1090, 188-192 (2015). [CrossRef]
- Nayak, P. K., Yang, L., Brehm, W., Adelhelm, P. From lithium-ion to sodium-ion batteries: advantages, challenges, and surprises. Angewandte Chemie International Edition. 57, 102-120 (2018). [CrossRef]
- He, X. et al. Sulfolane-based flame-retardant electrolyte for high-voltage sodium-ion batteries. Nano-micro letters. 17, 45 (2024). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



