Submitted:
22 May 2025
Posted:
26 May 2025
Read the latest preprint version here
Abstract
Keywords:
Introduction
Materials and Methods
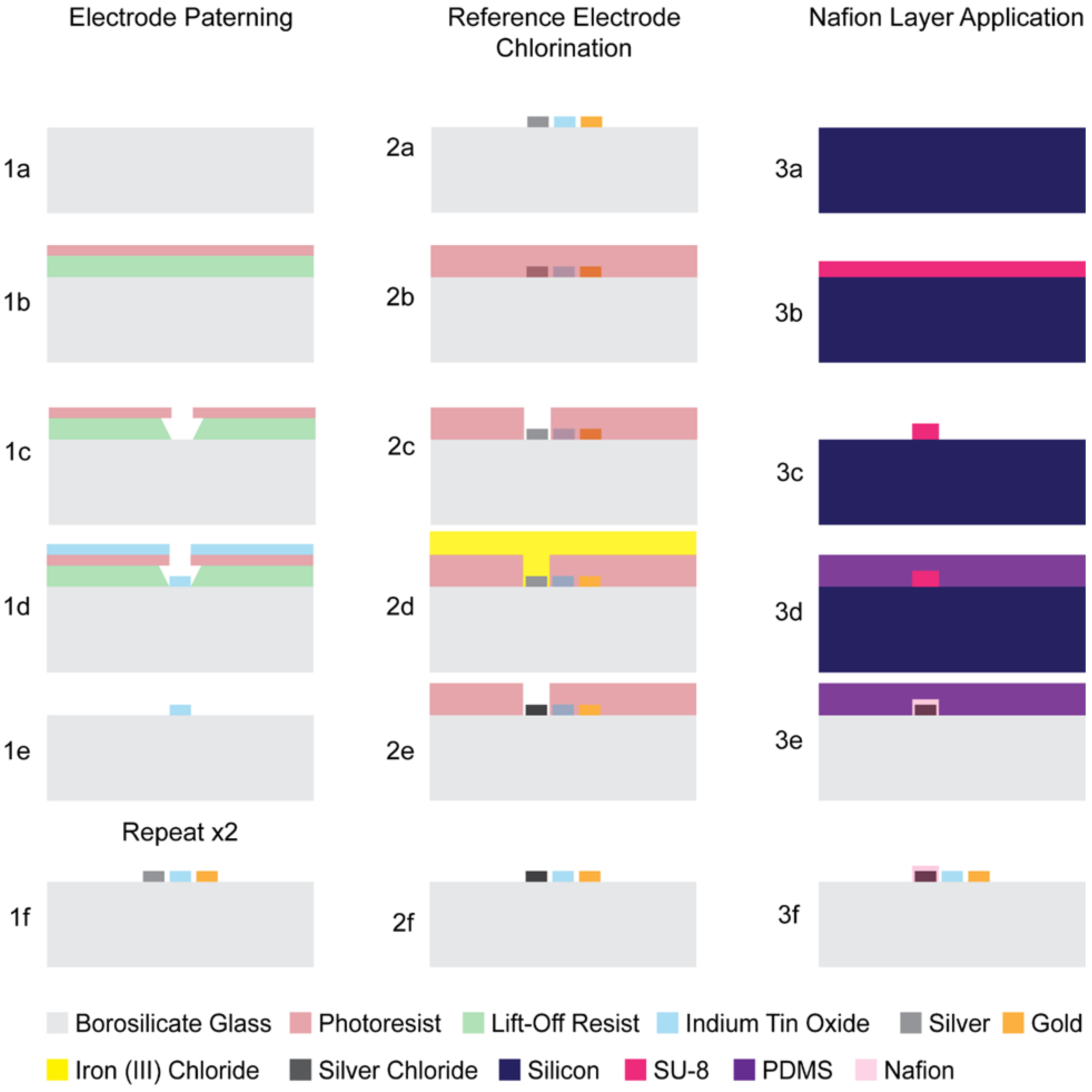
Electrode Array Lithography, Deposition, Lift-Off and Annealing
Chlorination of the Pseudoreference Electrodes
Application of a Patterned Proton Exchange Thin-Film Membrane on the Reference Electrode Surface
Microfluidic Channel Fabrication
Device Bonding and Packaging
Microfluidic Channel and Electrode Interfacing
Open Circuit Potential Measurements of the Pseudoreference Electrodes
Potential Calibration of the Pseudoreference Electrodes
Electrochemical Characterization of the µ-BEC Platform
Culture and Characterization of the Model EEU Organism Rhodopseudomonas palustris TIE-1 Inside the µ-BEC Platform
Results and Discussion
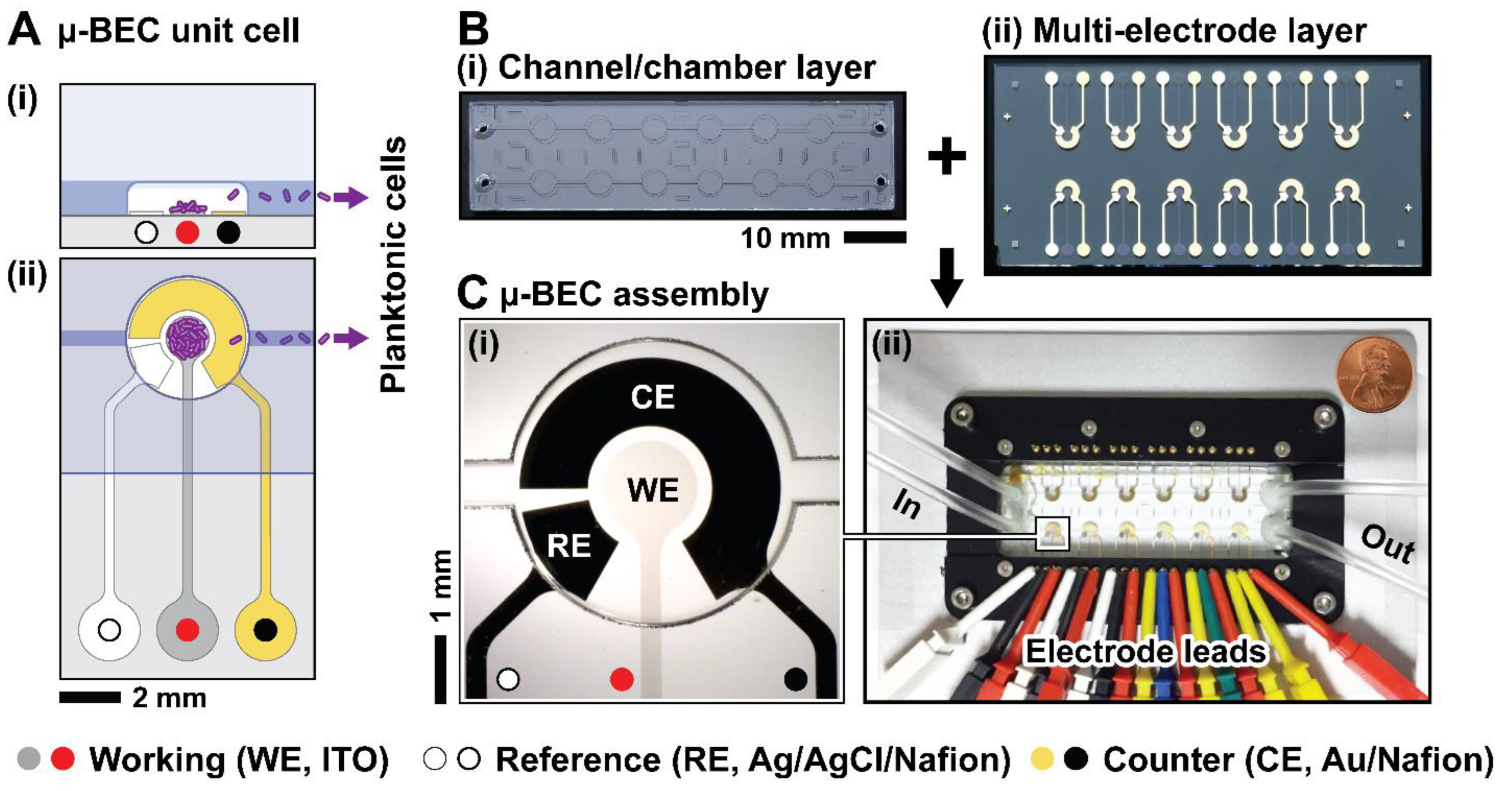
Device Design and Fabrication
Chlorination of the Pseudoreference Electrodes
Application of a Patterned Proton Exchange Thin-Film Membrane on the Reference Electrode Surface

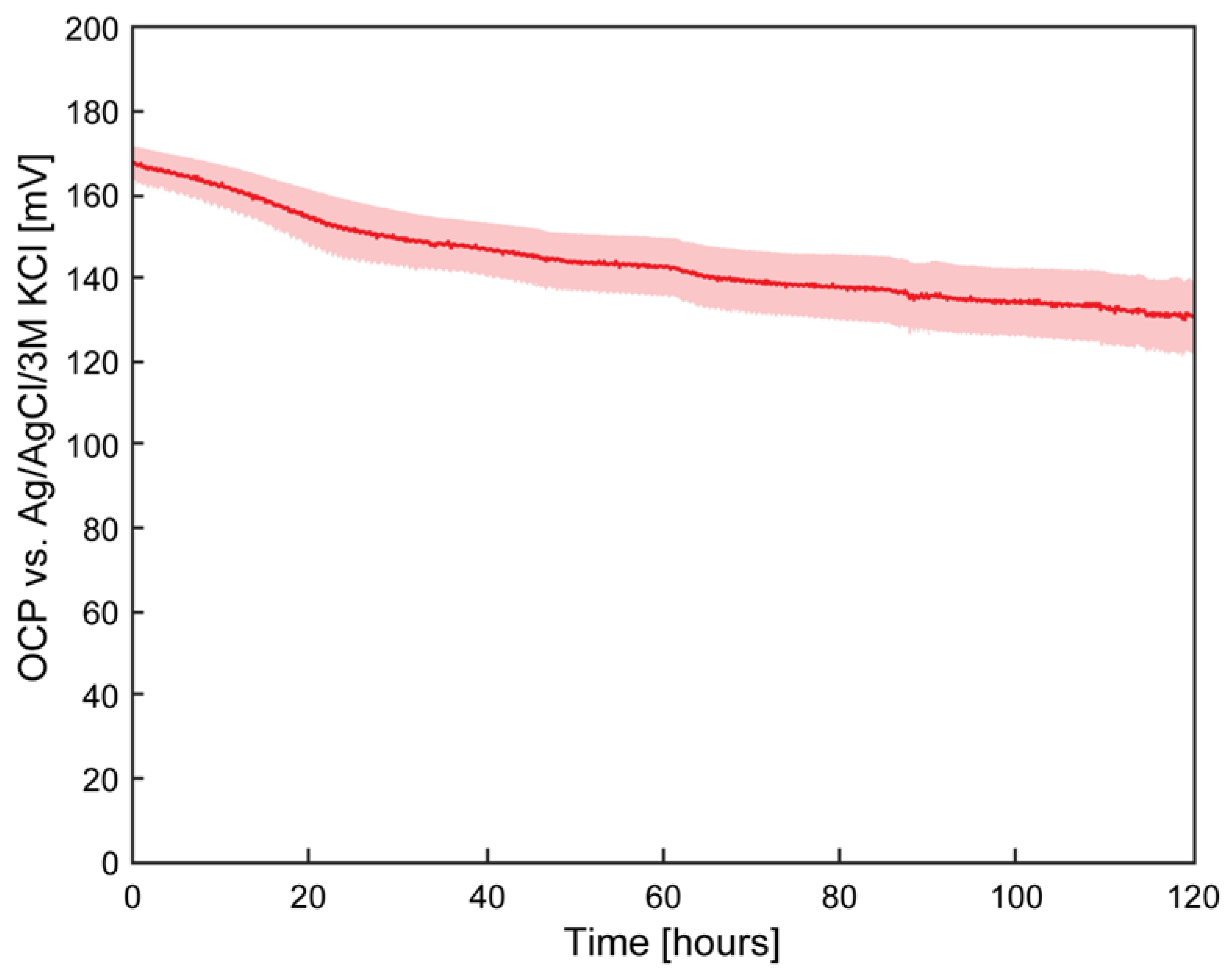
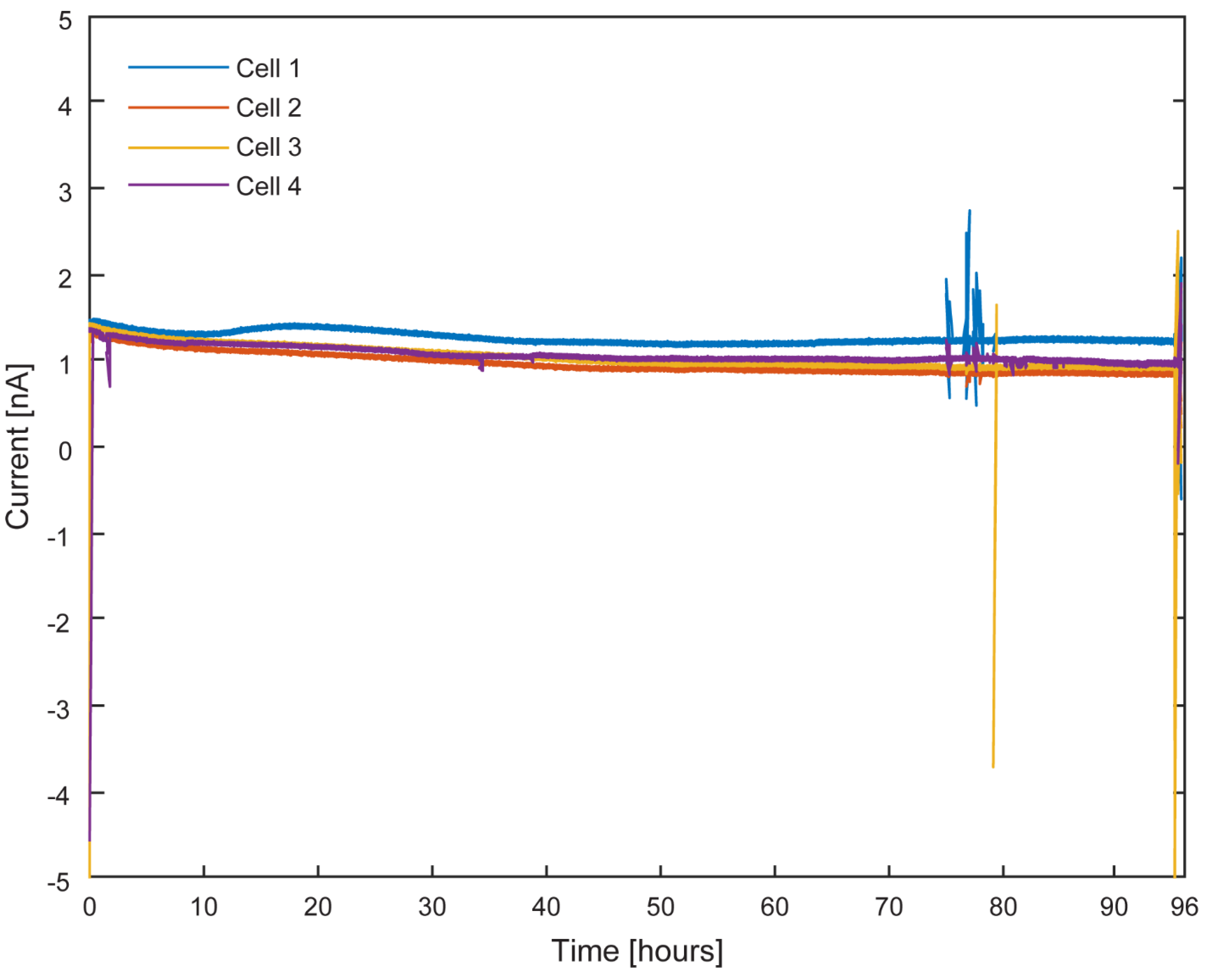
Testing the Stability of the Pseudoreference Electrodes
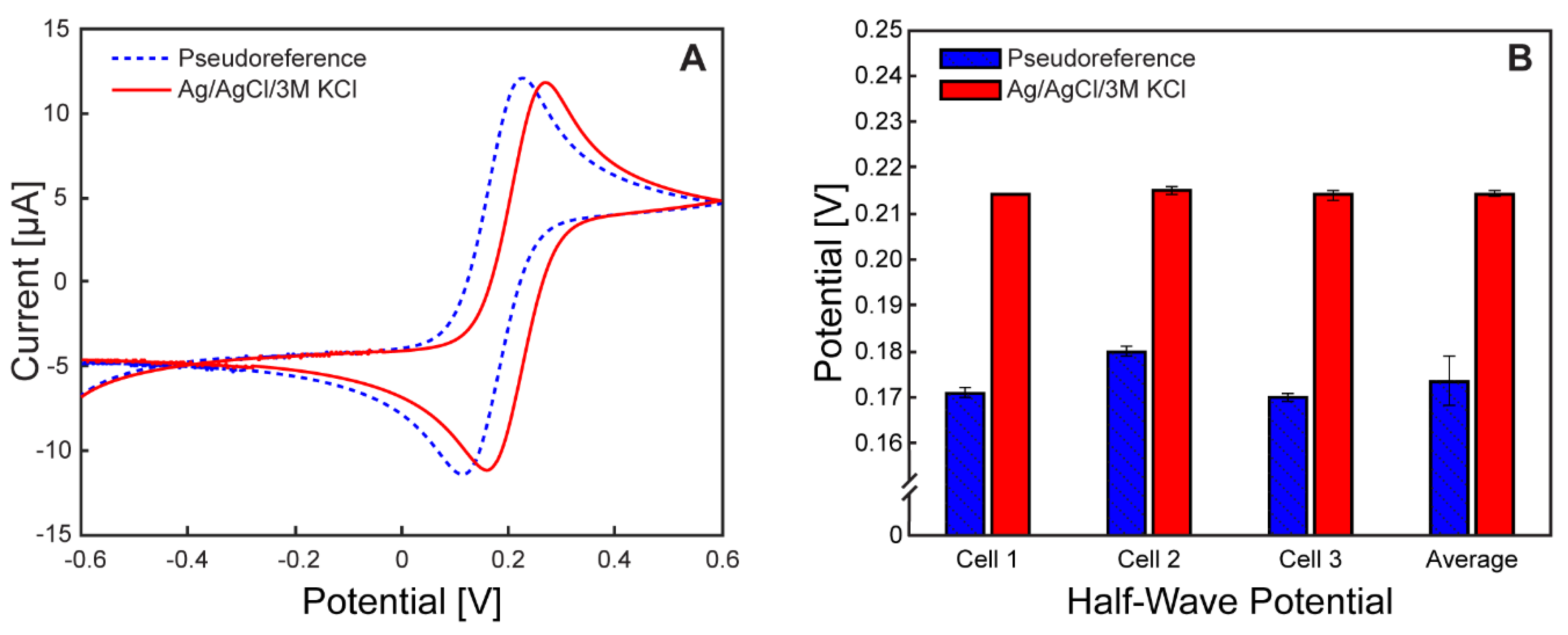
Potential Calibration of the Pseudoreference Electrodes
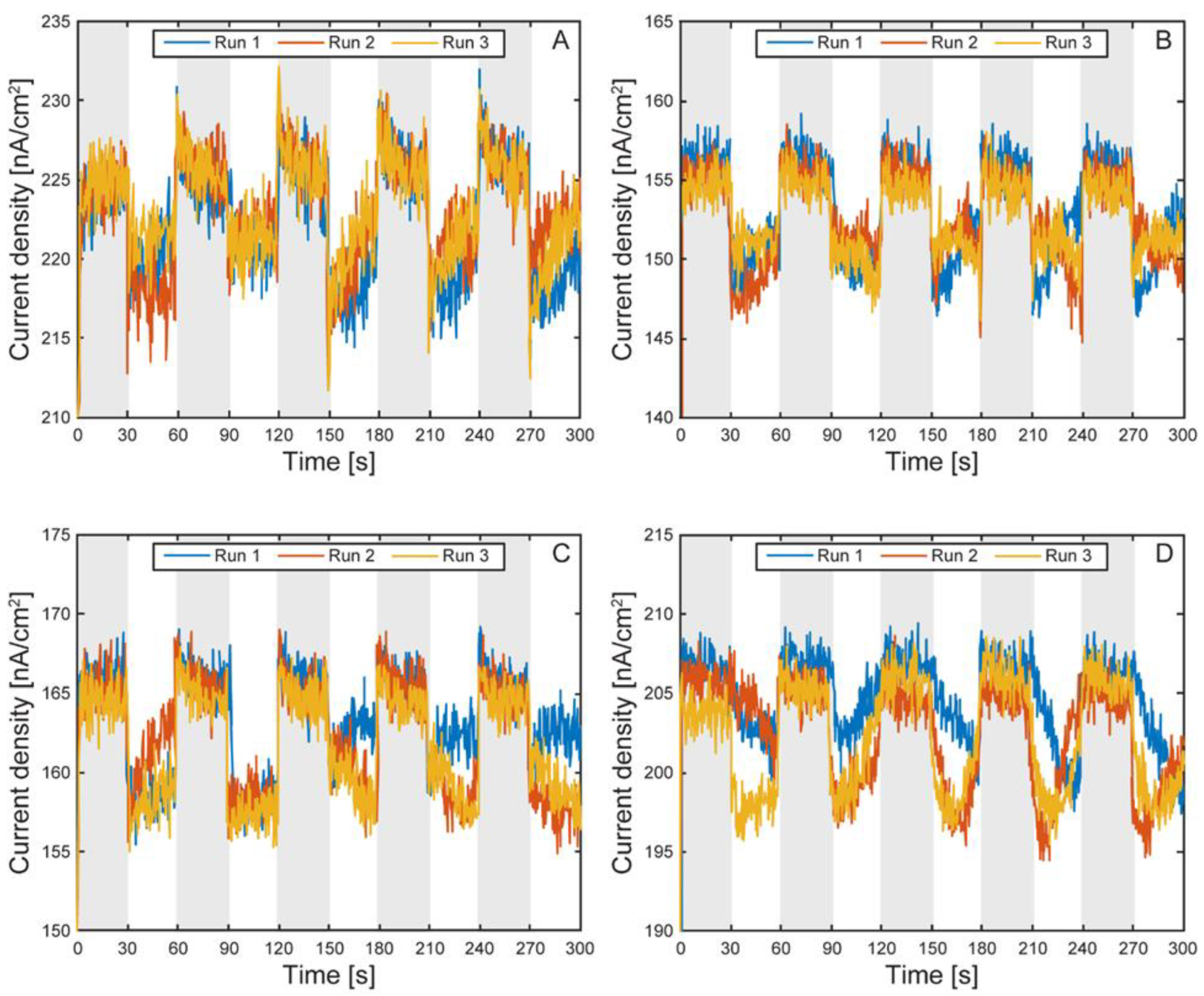
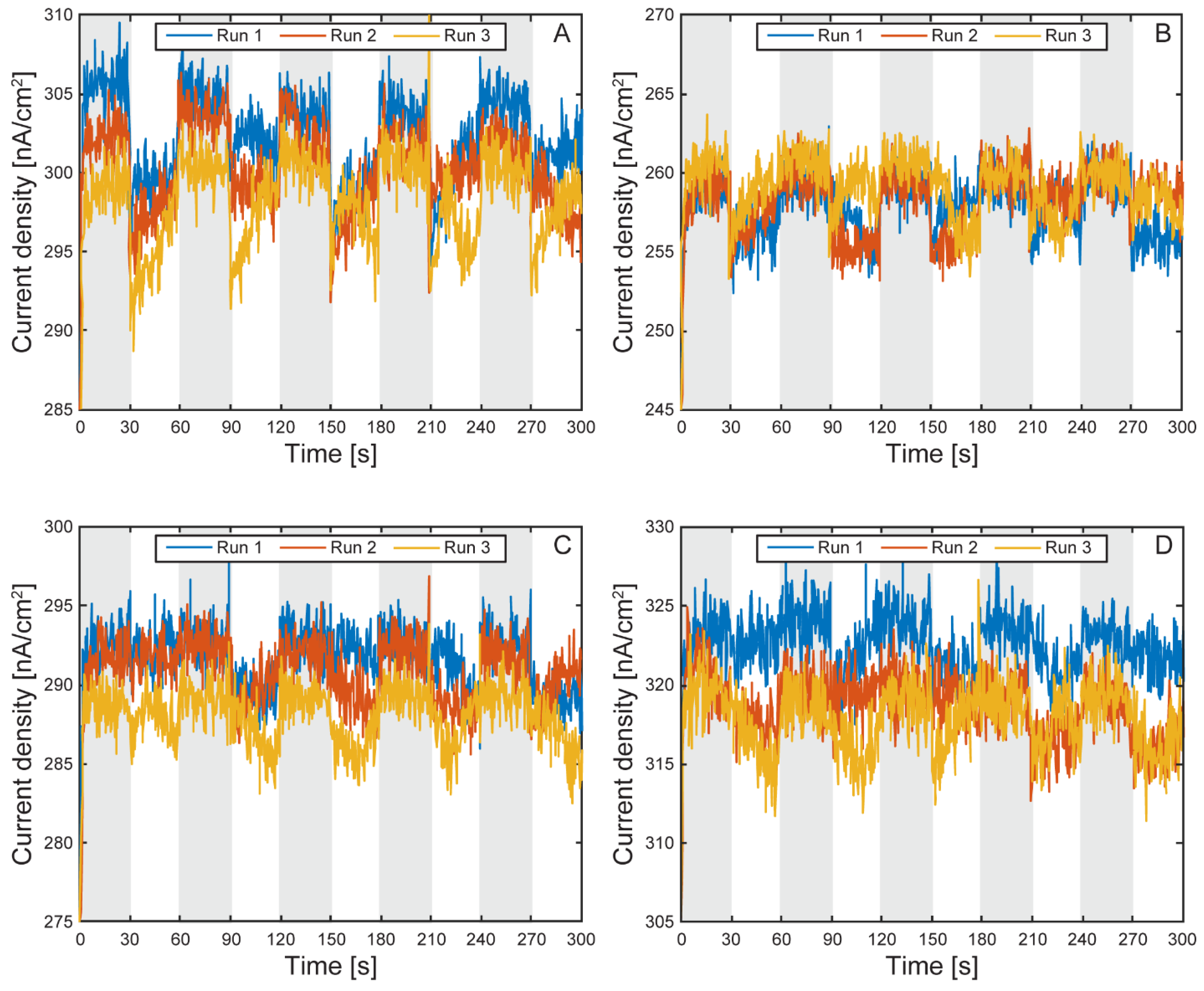
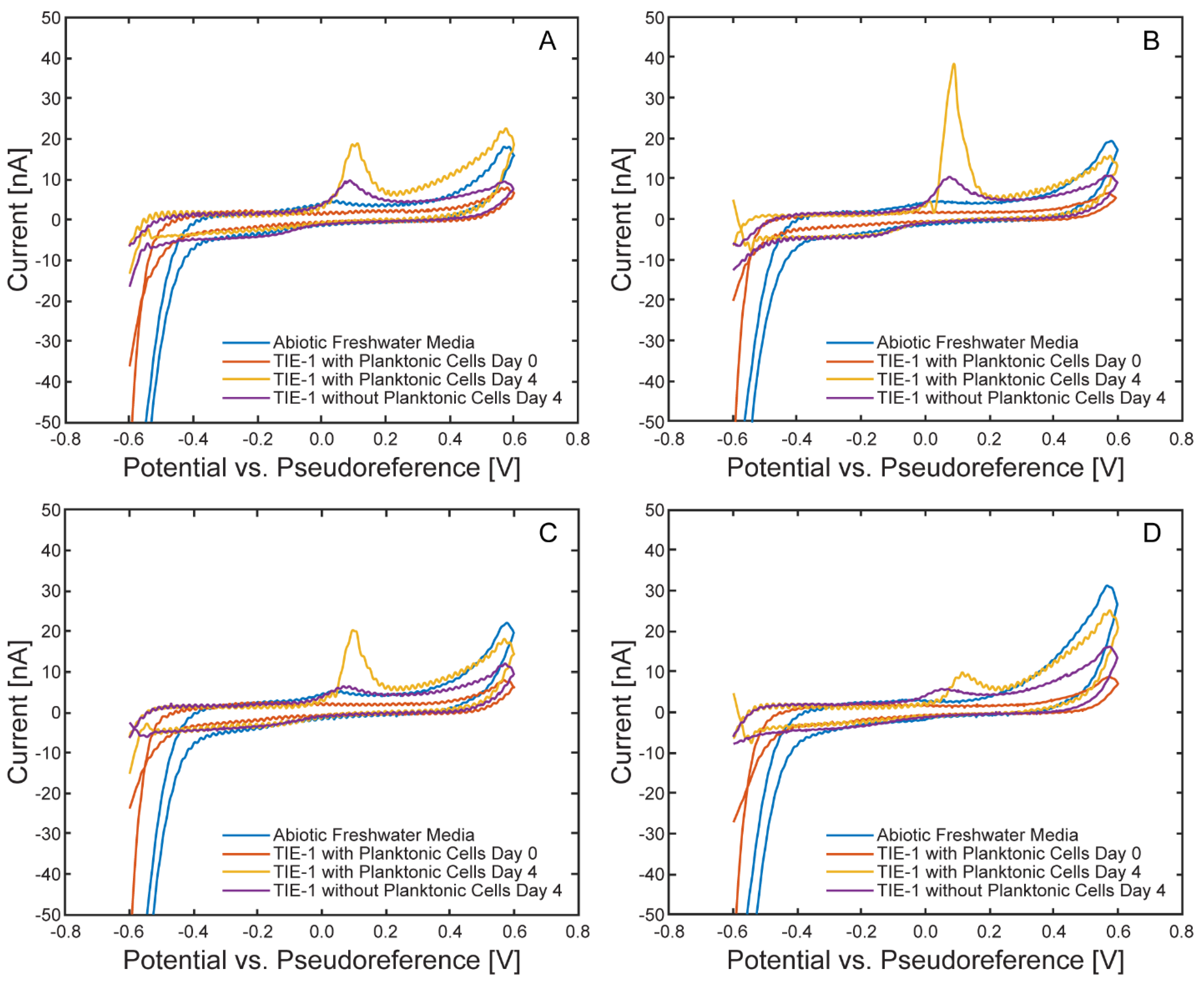
Electrochemical Characterization of the µ-BEC Platform
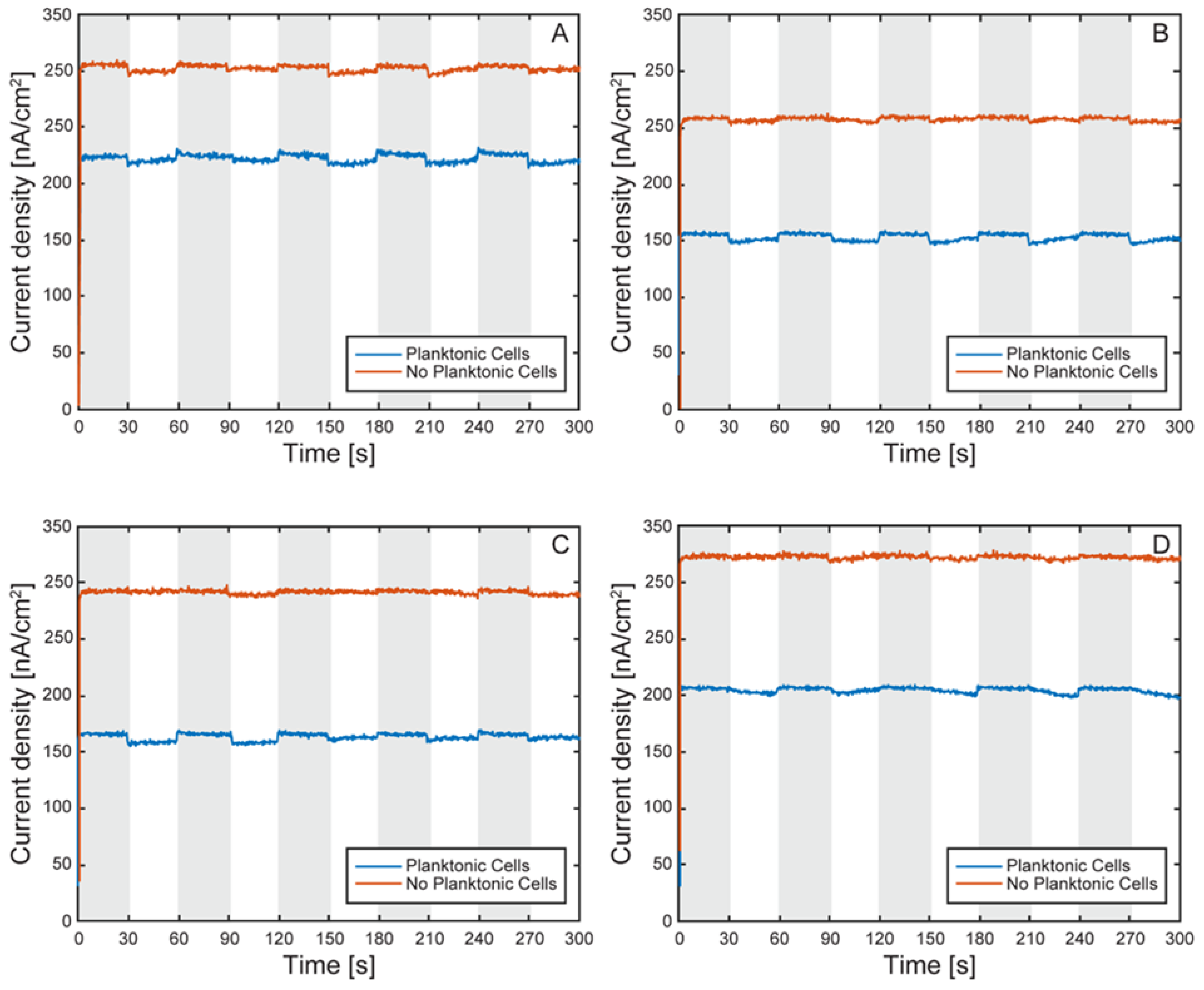
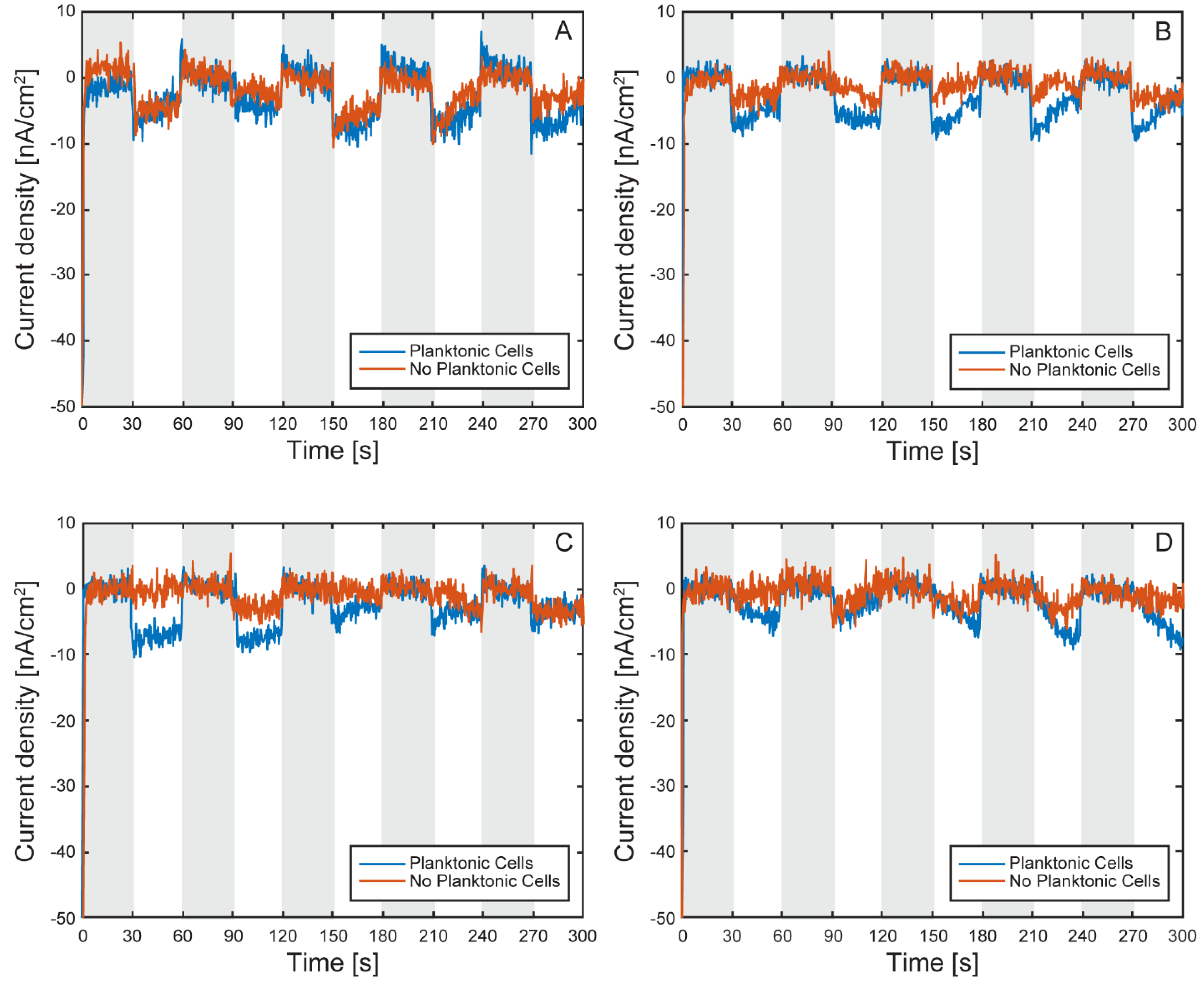
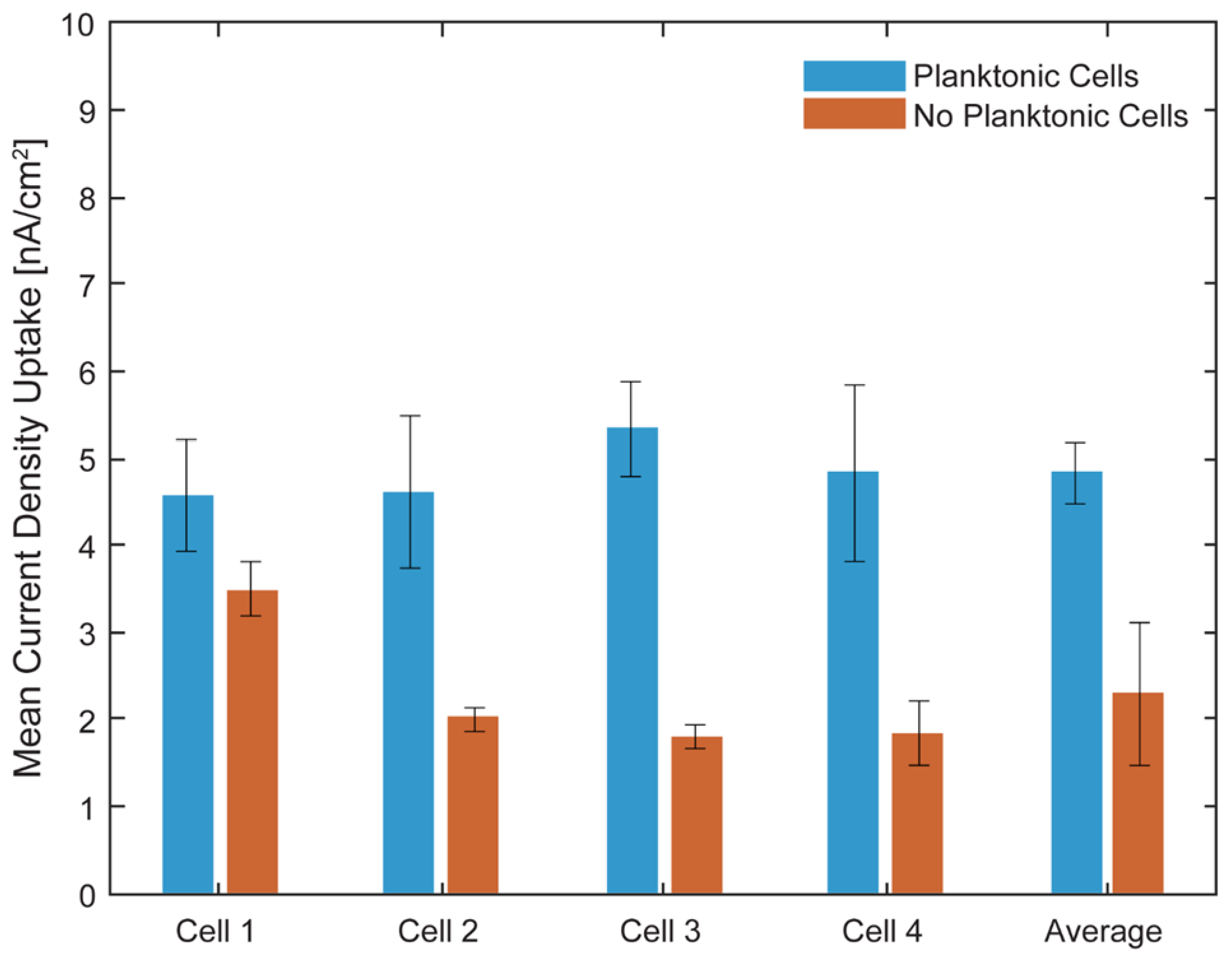
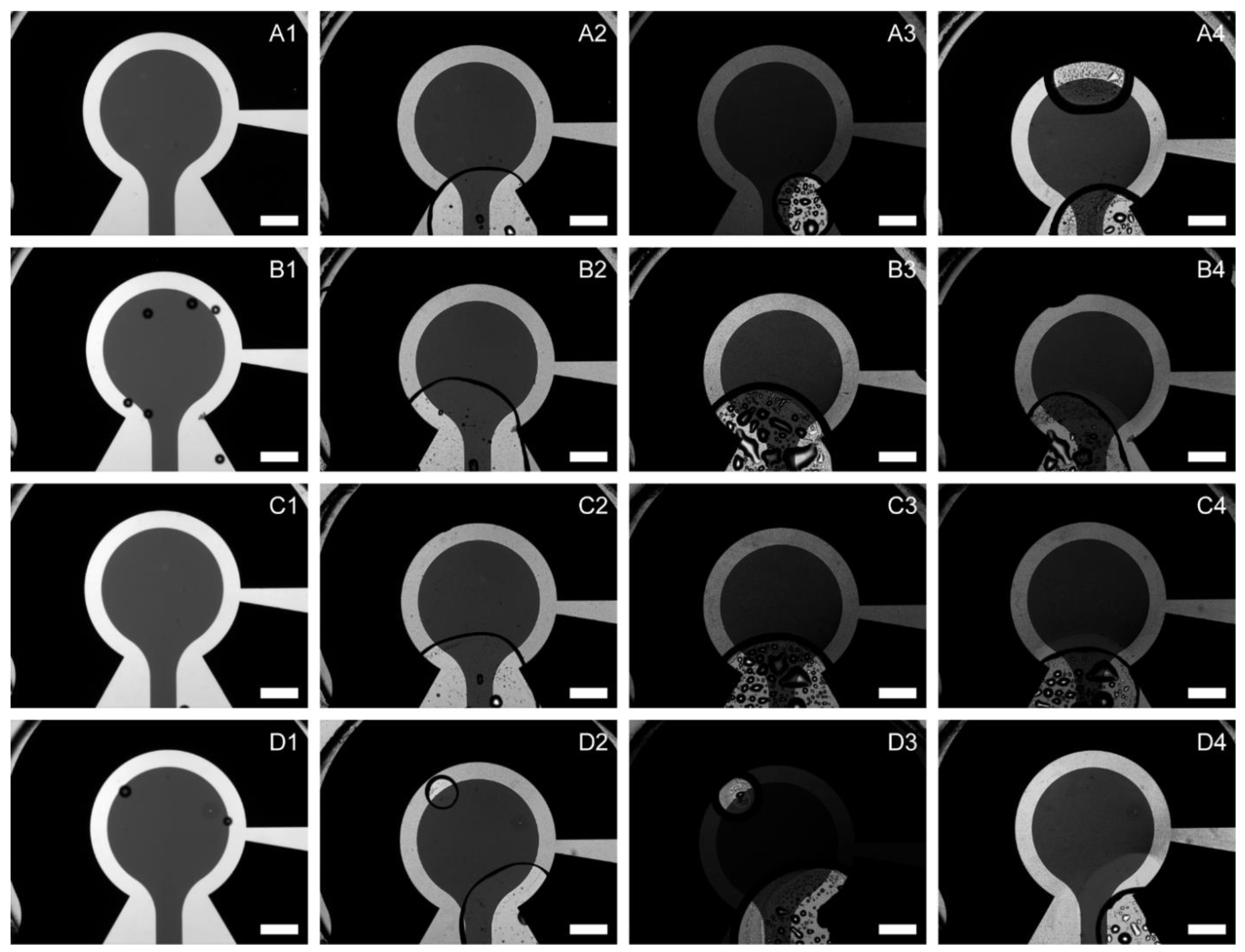
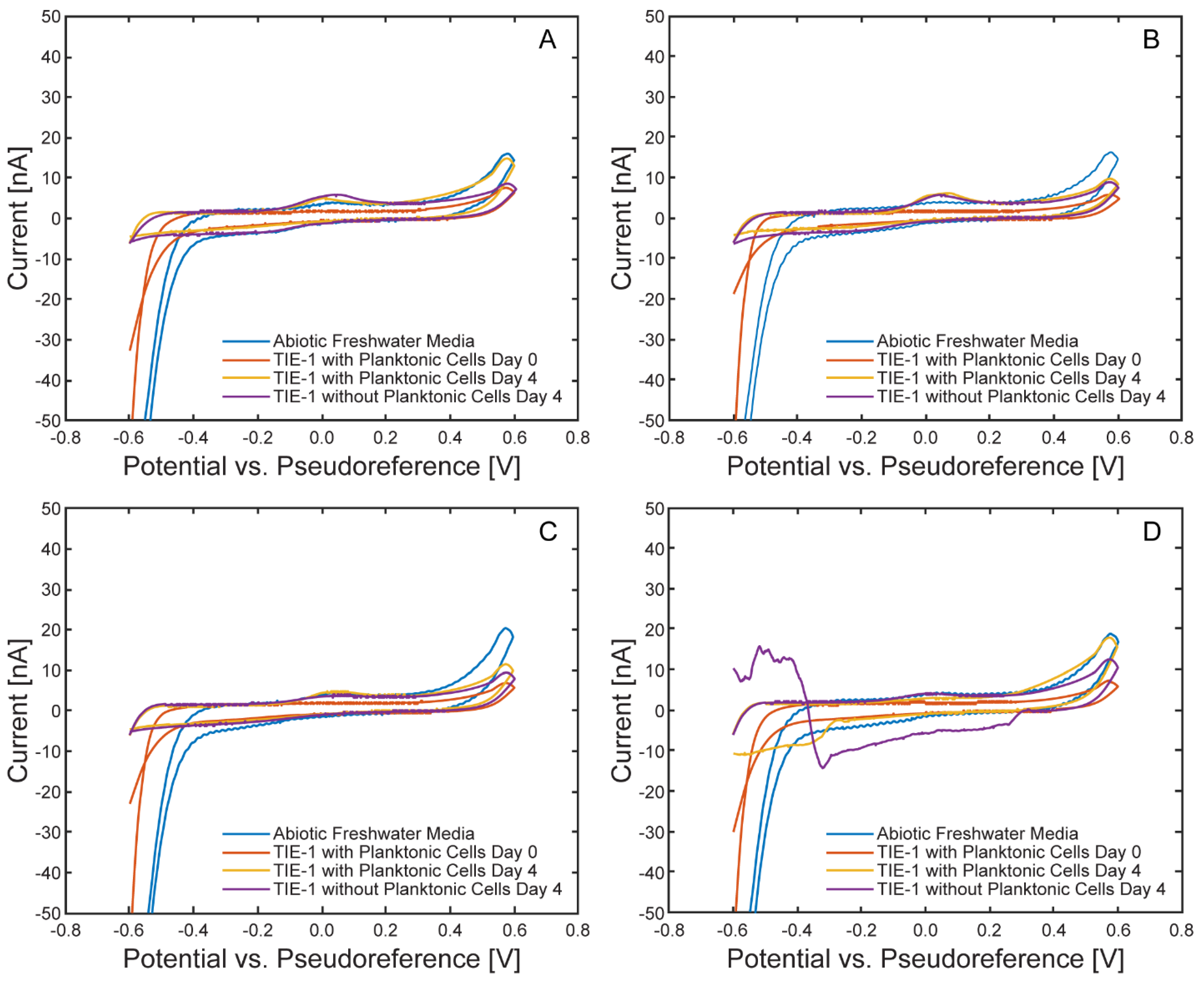
Characterization of the Model EEU Organism Rhodopseudomonas palustris TIE-1 Inside the µ-BEC Platform
Conclusions
Acknowledgments
References
- L. Shi et al., "Extracellular electron transfer mechanisms between microorganisms and minerals," Nature Reviews Microbiology, vol. 14, no. 10, pp. 651-662, 2016/10/01 2016. [CrossRef]
- D. R. Lovley and D. E. Holmes, "Electromicrobiology: the ecophysiology of phylogenetically diverse electroactive microorganisms," Nature Reviews Microbiology, vol. 20, no. 1, pp. 5-19, 2022/01/01 2022. [CrossRef]
- D. R. Lovley, J. F. Stolz, G. L. Nord, and E. J. P. Phillips, "Anaerobic production of magnetite by a dissimilatory iron-reducing microorganism," Nature, vol. 330, no. 6145, pp. 252-254, 1987/11/01 1987. [CrossRef]
- M. S. Guzman et al., "Phototrophic extracellular electron uptake is linked to carbon dioxide fixation in the bacterium Rhodopseudomonas palustris," Nature Communications, vol. 10, no. 1, p. 1355, 2019/03/22 2019. [CrossRef]
- D. R. Lovley, E. J. P. Phillips, Y. A. Gorby, and E. R. Landa, "Microbial reduction of uranium," Nature, vol. 350, no. 6317, pp. 413-416, 1991/04/01 1991. [CrossRef]
- L. Cologgi Dena, M. Speers Allison, A. Bullard Blair, D. Kelly Shelly, and G. Reguera, "Enhanced Uranium Immobilization and Reduction by Geobacter sulfurreducens Biofilms," Applied and Environmental Microbiology, vol. 80, no. 21, pp. 6638-6646, 2014/11/01 2014. [CrossRef]
- A. E. Franks and K. P. Nevin, "Microbial Fuel Cells, A Current Review," Energies, vol. 3, no. 5, pp. 899-919doi: 10.3390/en3050899.
- D. R. Lovley, "Bug juice: harvesting electricity with microorganisms," Nature Reviews Microbiology, vol. 4, no. 7, pp. 497-508, 2006/07/01 2006. [CrossRef]
- D. R. Lovley and K. P. Nevin, "Electrobiocommodities: powering microbial production of fuels and commodity chemicals from carbon dioxide with electricity," Current Opinion in Biotechnology, vol. 24, no. 3, pp. 385-390, 2013/06/01/ 2013. [CrossRef]
- P. Nevin Kelly, L. Woodard Trevor, E. Franks Ashley, M. Summers Zarath, and R. Lovley Derek, "Microbial Electrosynthesis: Feeding Microbes Electricity To Convert Carbon Dioxide and Water to Multicarbon Extracellular Organic Compounds," mBio, vol. 1, no. 2, pp. 10.1128/mbio.00103-10, 2010. [CrossRef]
- R. Karthikeyan, R. Singh, and A. Bose, "Microbial electron uptake in microbial electrosynthesis: a mini-review," Journal of Industrial Microbiology and Biotechnology, vol. 46, no. 9-10, pp. 1419-1426, 2019. [CrossRef]
- T. O. Ranaivoarisoa, R. Singh, K. Rengasamy, M. S. Guzman, and A. Bose, "Towards sustainable bioplastic production using the photoautotrophic bacterium Rhodopseudomonas palustris TIE-1," Journal of Industrial Microbiology and Biotechnology, vol. 46, no. 9-10, pp. 1401-1417, 2019. [CrossRef]
- M. Tahernia, M. Mohammadifar, Y. Gao, W. Panmanee, D. J. Hassett, and S. Choi, "A 96-well high-throughput, rapid-screening platform of extracellular electron transfer in microbial fuel cells," Biosensors and Bioelectronics, vol. 162, p. 112259, 2020/08/15/ 2020. [CrossRef]
- M. Tahernia, M. Mohammadifar, D. J. Hassett, and S. Choi, "A portable papertronic sensing system for rapid, high-throughput, and visual screening of bacterial electrogenicity," Biosensors and Bioelectronics, vol. 165, p. 112348, 2020/10/01/ 2020. [CrossRef]
- M. Tahernia, M. Mohammadifar, D. J. Hassett, and S. Choi, "A fully disposable 64-well papertronic sensing array for screening electroactive microorganisms," Nano Energy, vol. 65, p. 104026, 2019/11/01/ 2019. [CrossRef]
- N. H. Park, J. Kim, and Y. Ahn, "Fabric-based self-pumping, single-stream microfluidic fuel cell," Electrochimica Acta, vol. 446, p. 142106, 2023/04/01/ 2023. [CrossRef]
- J. Kim, H. G. Kong, and Y. Ahn, "Textile-Based Membraneless Microfluidic Double-Inlet Hybrid Microbial–Enzymatic Biofuel Cell," ACS Applied Materials & Interfaces, vol. 16, no. 33, pp. 43661-43669, 2024/08/21 2024. [CrossRef]
- Y. Kwon, D. Hong, and Y. Ahn, "Monolayer textile-based co-laminar flow biocompatible enzymatic biofuel cell," Energy Conversion and Management, vol. 301, p. 118042, 2024/02/01/ 2024. [CrossRef]
- M. P. Zarabadi, F. Paquet-Mercier, S. J. Charette, and J. Greener, "Hydrodynamic Effects on Biofilms at the Biointerface Using a Microfluidic Electrochemical Cell: Case Study of Pseudomonas sp," (in eng), Langmuir, vol. 33, no. 8, pp. 2041-2049, Feb 28 2017. [CrossRef]
- M. A. Amirdehi, L. Gong, N. Khodaparastasgarabad, J. M. Sonawane, B. E. Logan, and J. Greener, "Hydrodynamic interventions and measurement protocols to quantify and mitigate power overshoot in microbial fuel cells using microfluidics," Electrochimica Acta, vol. 405, p. 139771, 2022/02/10/ 2022. [CrossRef]
- L. Gong, N. Khodaparastasgarabad, D. M. Hall, and J. Greener, "A new angle to control concentration profiles at electroactive biofilm interfaces: Investigating a microfluidic perpendicular flow approach," Electrochimica Acta, vol. 431, p. 141071, 2022/11/01/ 2022. [CrossRef]
- N. Khodaparastasgarabad, M. Couture, and J. Greener, "A microfluidic study of acetate conversion kinetics in a microbial electrolysis cell: The role of age, concentration and flow on biofilm permeability," Sensors and Actuators B: Chemical, vol. 412, p. 135779, 2024/08/01/ 2024. [CrossRef]
- H.-M. Cho, H. Ha, and Y. Ahn, "Co-laminar Microfluidic Microbial Fuel Cell Integrated with Electrophoretically Deposited Carbon Nanotube Flow-Over Electrode," ACS Sustainable Chemistry & Engineering, vol. 10, no. 5, pp. 1839-1846, 2022/02/07 2022. [CrossRef]
- C. H. Lee, H. Ha, Y. Ahn, and H. Liu, "Performance of single-layer paper-based co-laminar flow microbial fuel cells," Journal of Power Sources, vol. 580, p. 233456, 2023/10/01/ 2023. [CrossRef]
- S.-C. Chang and J. M. Kempisty, "Lift-off Methods for MEMS Devices," MRS Online Proceedings Library, vol. 729, no. 1, p. 23, 2003/01/01 2003. [CrossRef]
- H.-R. Lim, N. Hillman, Y.-T. Kwon, Y.-S. Kim, Y.-H. Choa, and W.-H. Yeo, "Ultrathin, long-term stable, solid-state reference electrode enabled by enhanced interfacial adhesion and conformal coating of AgCl," Sensors and Actuators B: Chemical, vol. 309, p. 127761, 2020/04/15/ 2020. [CrossRef]
- C. Iliescu, F. E. H. Tay, and J. Miao, "Strategies in deep wet etching of Pyrex glass," Sensors and Actuators A: Physical, vol. 133, no. 2, pp. 395-400, 2007/02/12/ 2007. [CrossRef]
- C. Iliescu, J. Jing, F. E. H. Tay, J. Miao, and T. Sun, "Characterization of masking layers for deep wet etching of glass in an improved HF/HCl solution," Surface and Coatings Technology, vol. 198, no. 1, pp. 314-318, 2005/08/01/ 2005. [CrossRef]
- C. Iliescu and F. E. Tay, "Wet etching of glass," in CAS 2005 Proceedings. 2005 International Semiconductor Conference, 2005., 2005, vol. 1: IEEE, pp. 35-44.
- S. Satyanarayana, R. N. Karnik, and A. Majumdar, "Stamp-and-stick room-temperature bonding technique for microdevices," Journal of Microelectromechanical Systems, vol. 14, no. 2, pp. 392-399, 2005. [CrossRef]
- T. Y. Kim, S. A. Hong, and S. Yang, "A Solid-State Thin-Film Ag/AgCl Reference Electrode Coated with Graphene Oxide and Its Use in a pH Sensor," Sensors, vol. 15, no. 3, pp. 6469-6482, 2015. [Online]. Available: https://www.mdpi.com/1424-8220/15/3/6469.
- M. W. Shinwari, D. Zhitomirsky, I. A. Deen, P. R. Selvaganapathy, M. J. Deen, and D. Landheer, "Microfabricated reference electrodes and their biosensing applications," (in eng), Sensors (Basel), vol. 10, no. 3, pp. 1679-715, 2010. [CrossRef]
- D. Gupta et al., "Photoferrotrophy and phototrophic extracellular electron uptake is common in the marine anoxygenic phototroph Rhodovulum sulfidophilum," The ISME Journal, vol. 15, no. 11, pp. 3384-3398, 2021/11/01 2021. [CrossRef]
- R. Frank et al., "Electrochemical restructuring of thin layer indium tin oxide electrode arrays for optimized (bio)electrocatalysis," Electrochimica Acta, vol. 259, pp. 449-457, 2018/01/01/ 2018. [CrossRef]
- D. Xing, Y. Zuo, S. Cheng, J. M. Regan, and B. E. Logan, "Electricity Generation by Rhodopseudomonas palustris DX-1," Environmental Science & Technology, vol. 42, no. 11, pp. 4146-4151, 2008/06/01 2008. [CrossRef]
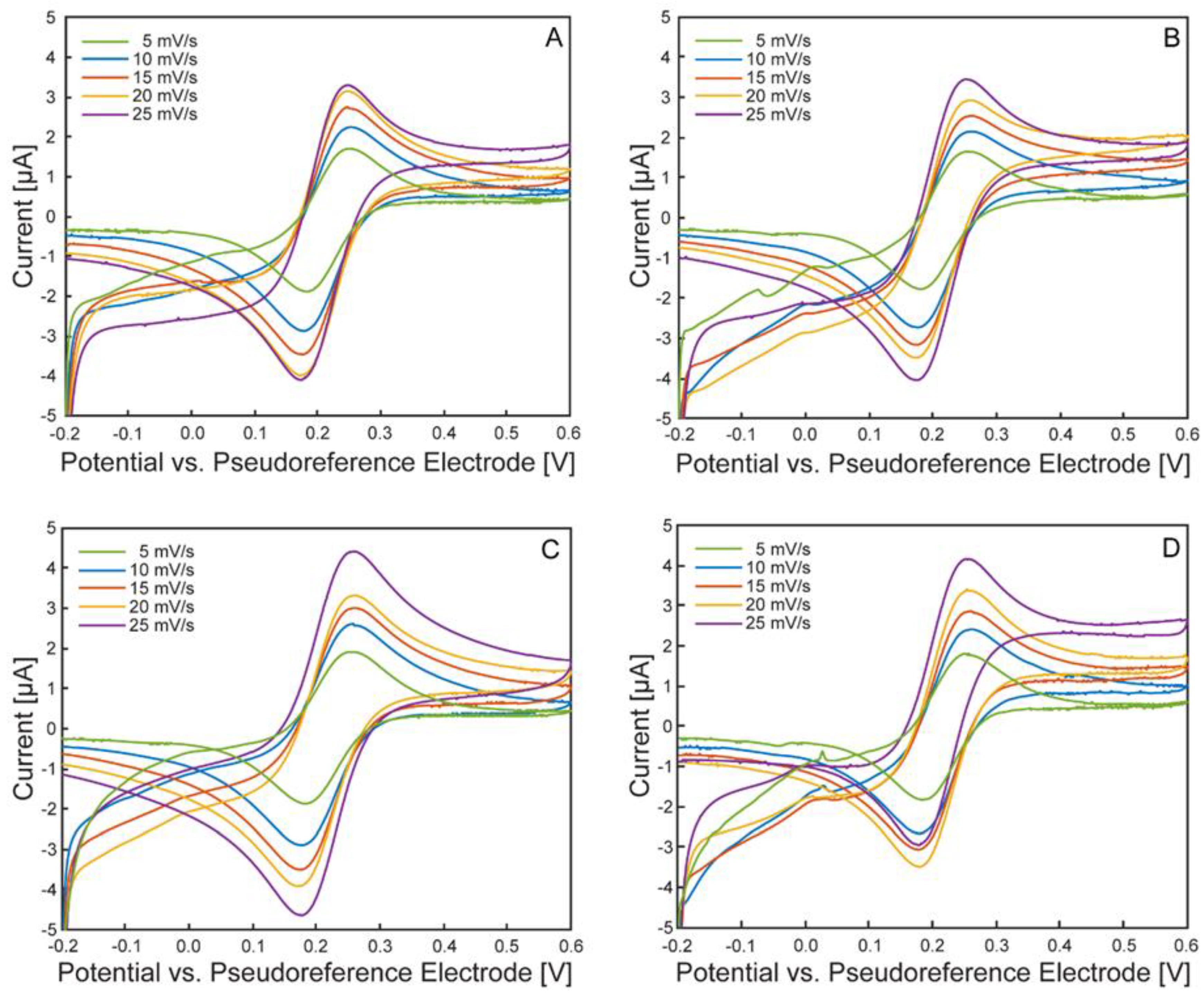
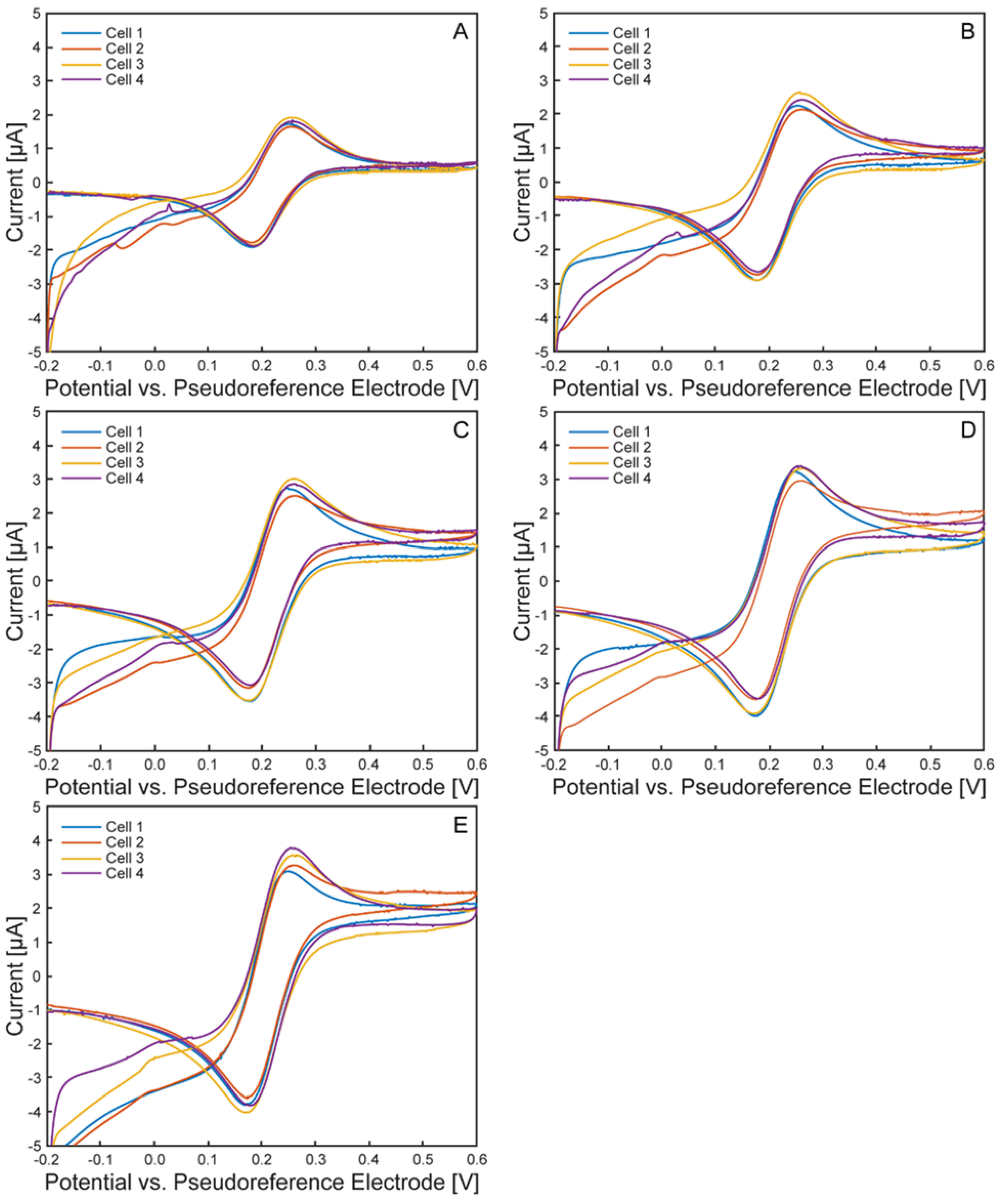
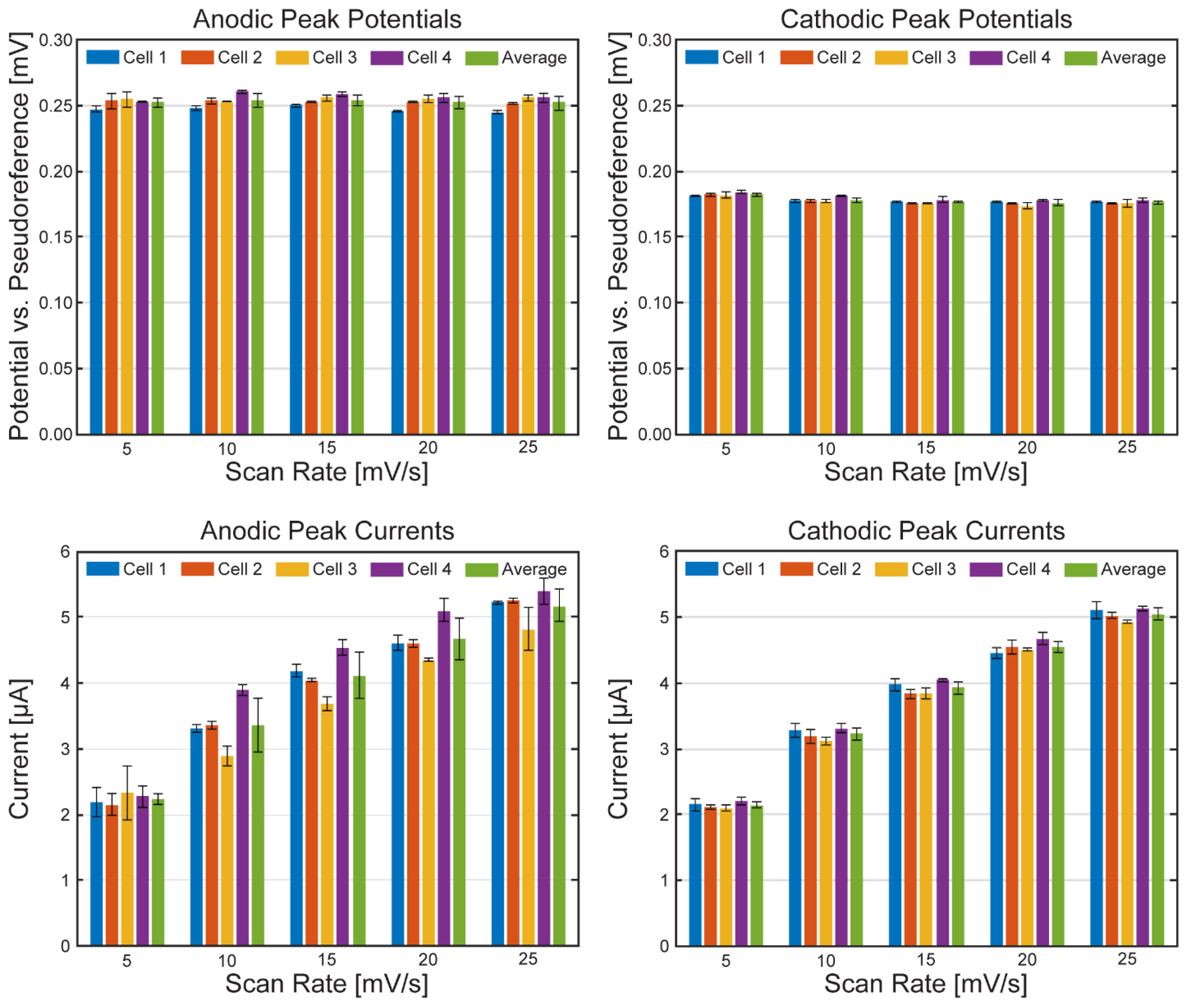
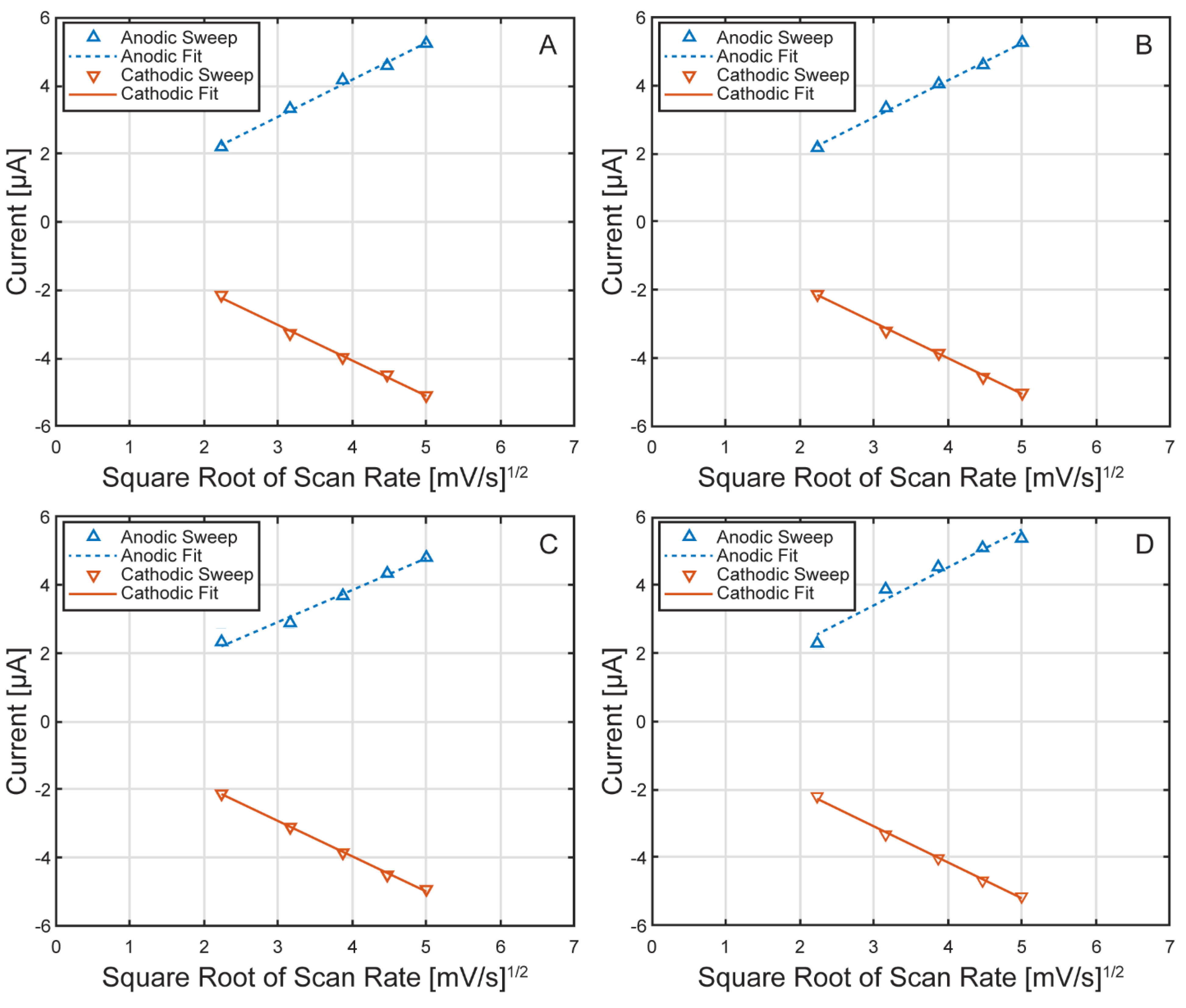
| Scan Rate | Anodic Peak Potential [mV] | Cathodic Peak Potential [mV] | ||
|---|---|---|---|---|
| Average | Standard Deviation | Average | Standard Deviation | |
| 5 mV/s | 252 | 3.25 | 182 | 1.29 |
| 10 mV/s | 254 | 5.21 | 178 | 1.90 |
| 15 mV/s | 254 | 3.75 | 177 | 1.12 |
| 20 mV/s | 252 | 4.50 | 176 | 2.05 |
| 25 mV/s | 252 | 5.20 | 176 | 1.13 |
| Scan Rate | Anodic Peak Current [µA] | Cathodic Peak Potential [µA] | ||
|---|---|---|---|---|
| Average | Standard Deviation | Average | Standard Deviation | |
| 5 mV/s | 2.24 | 0.08 | 2.15 | 0.05 |
| 10 mV/s | 3.36 | 0.41 | 3.23 | 0.09 |
| 15 mV/s | 4.12 | 0.35 | 3.93 | 0.10 |
| 20 mV/s | 4.67 | 0.31 | 4.55 | 0.09 |
| 25 mV/s | 5.17 | 0.25 | 5.05 | 0.09 |
| Cell Number | Anodic Fit Constant | Anodic R-Squared | Cathodic Fit Constant | Cathodic R-Squared |
|---|---|---|---|---|
| 1 | 3.30E-05 | 9.994E-01 | 3.21E-05 | 9.997E-01 |
| 2 | 3.29E-05 | 9.995E-01 | 3.17E-05 | 9.997E-01 |
| 3 | 3.05E-05 | 9.992E-01 | 3.13E-05 | 9.997E-01 |
| 4 | 3.58E-05 | 9.973E-01 | 3.27E-05 | 9.998E-01 |
| Average | 3.30E-05 | 9.989E-01 | 3.20E-05 | 9.997E-01 |
| Standard Deviation | 2.17E-06 | 1.041E-03 | 6.03E-07 | 5.00E-05 |
| CV (%) | 6.56 | 0.10 | 1.88 | 0.01 |
| Scan Rate | Coefficient of Variance [%] | |||
|---|---|---|---|---|
| Anodic Peak Potential | Anodic Peak Current | Cathodic Peak Potential | Cathodic Peak Current | |
| 5 mV/s | 1.29 | 3.46 | 0.71 | 2.33 |
| 10 mV/s | 2.05 | 12.25 | 1.06 | 2.65 |
| 15 mV/s | 1.47 | 8.51 | 0.64 | 2.61 |
| 20 mV/s | 1.79 | 6.74 | 1.17 | 2.01 |
| 25 mV/s | 2.06 | 4.76 | 0.64 | 1.77 |
| Average | 1.73 | 7.14 | 0.84 | 2.27 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




