Submitted:
21 May 2025
Posted:
22 May 2025
You are already at the latest version
Abstract
Keywords:
Introduction
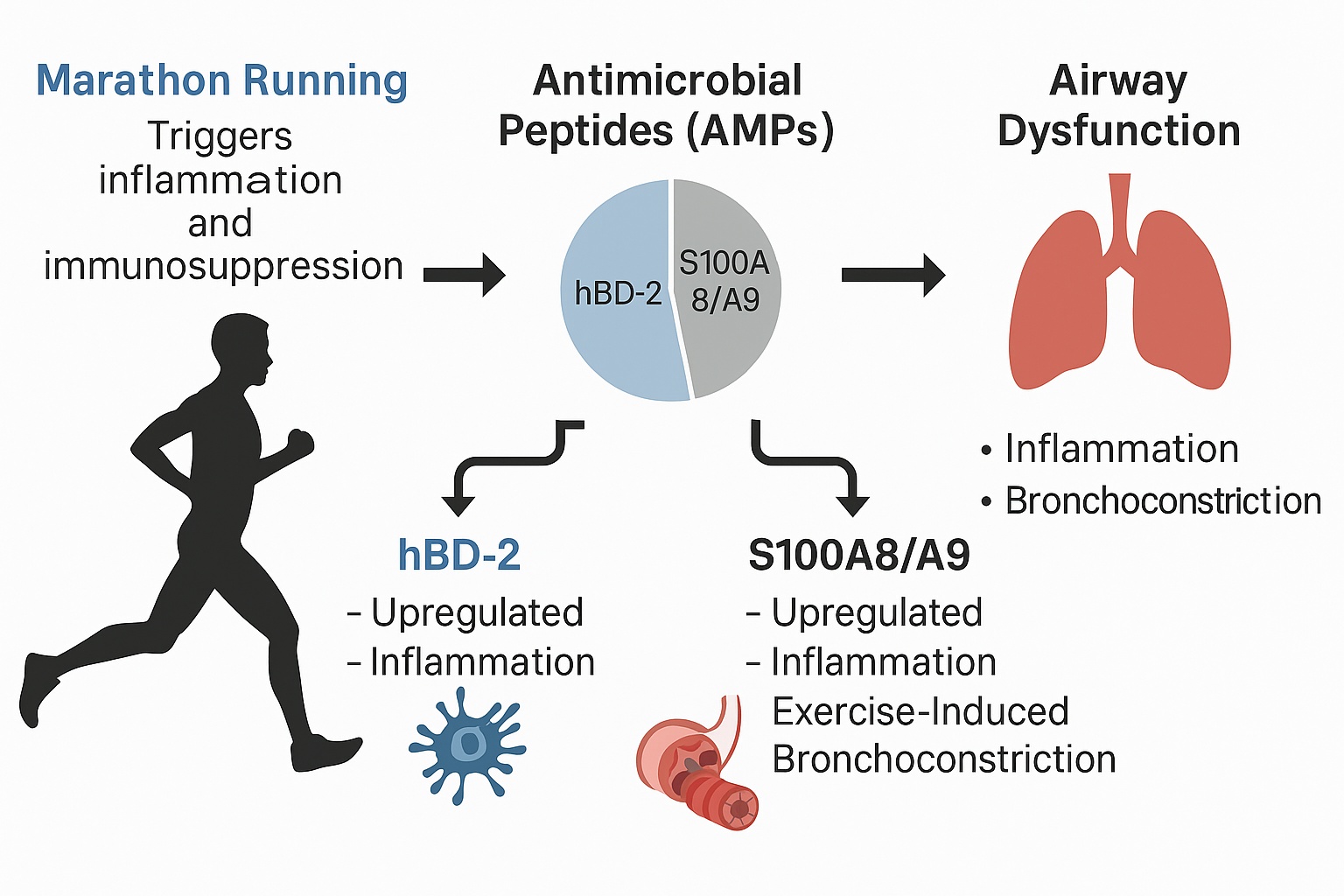
Marathon Running and Its Impact on Respiratory and Immune System
Antimicrobial Peptides (AMPs) and Respiratory Defense
Exercise-Induced Bronchoconstriction (EIB)
Human-Beta Defensin 2
S100A8/A9
Major Basic Protein (MBP)
Angiogenin
Material and Methods
Setting
Results
Demographic and Spirometry Data
Serum Measurement of Circulating Antimicrobial Peptides
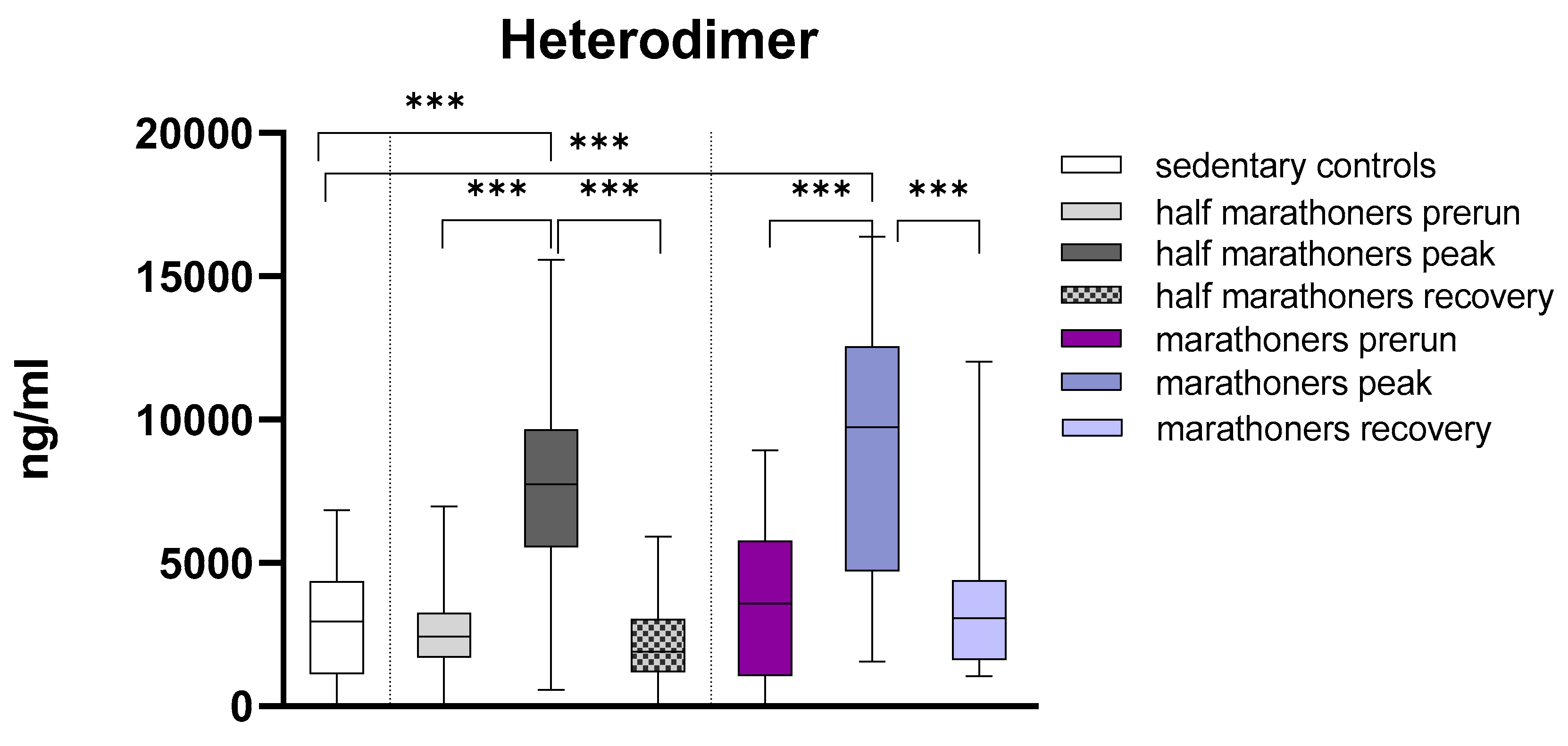
S100A8/S100A9 Heterodimer (Calprotectin)
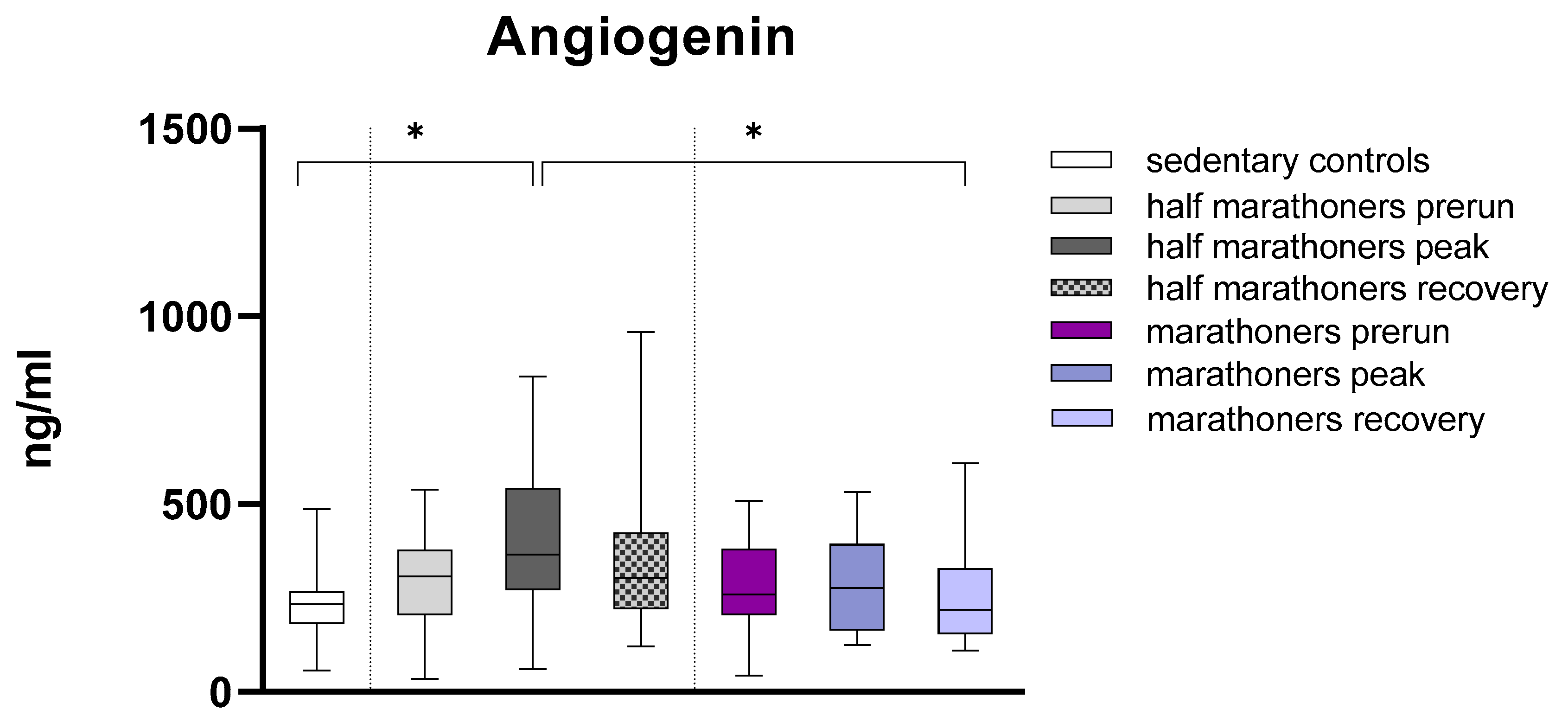
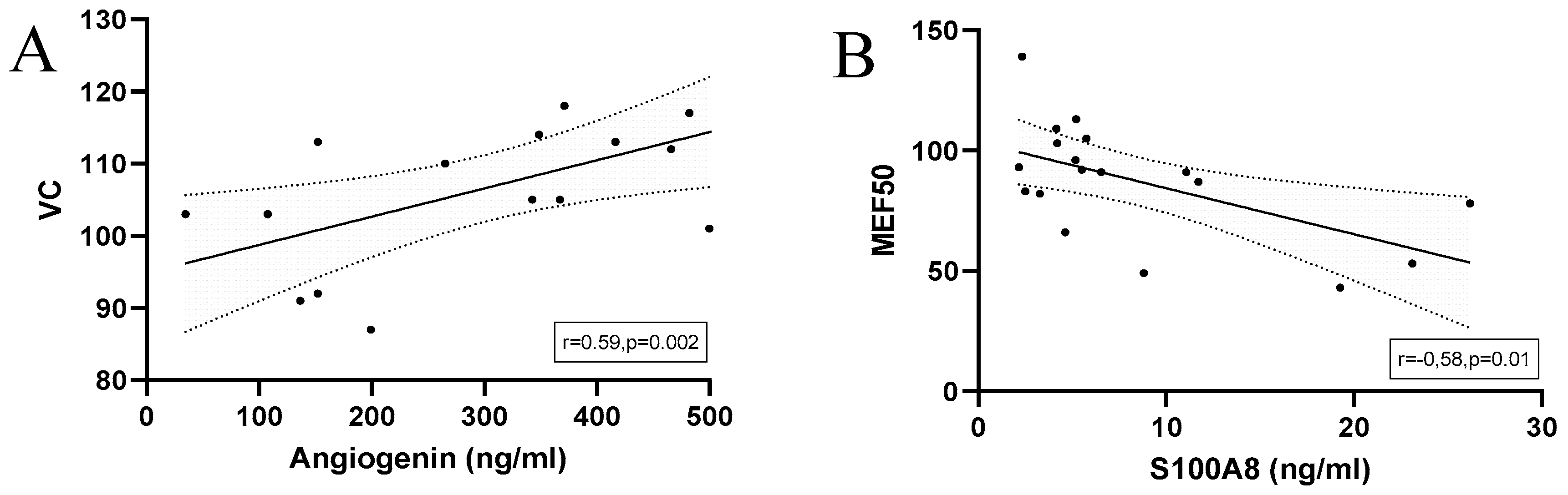
Angiogenin
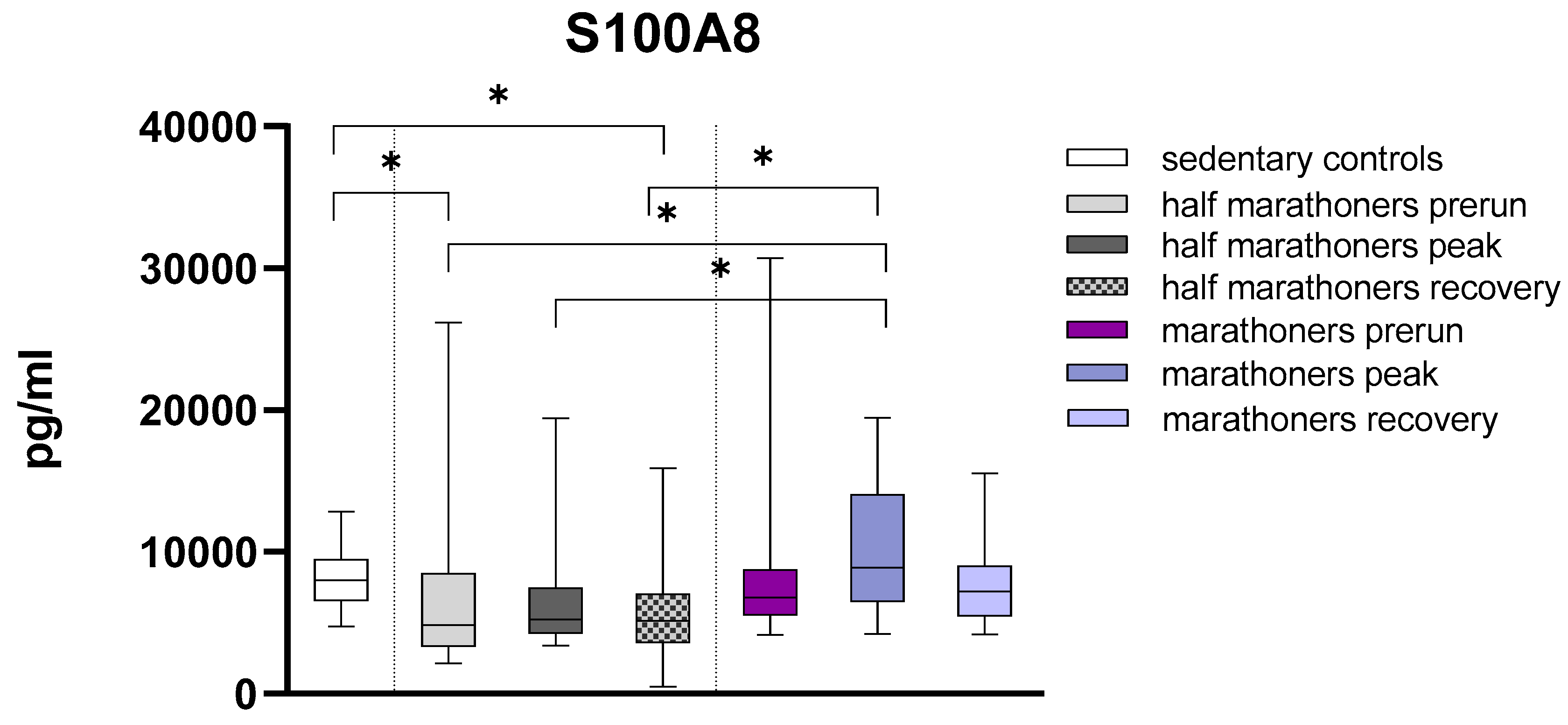
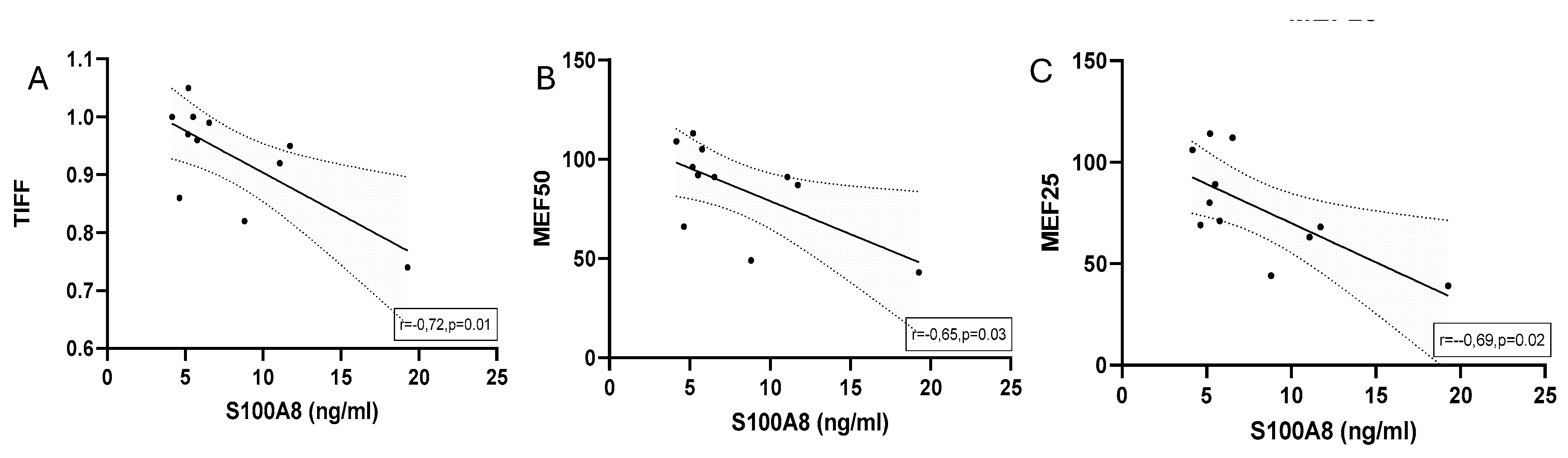
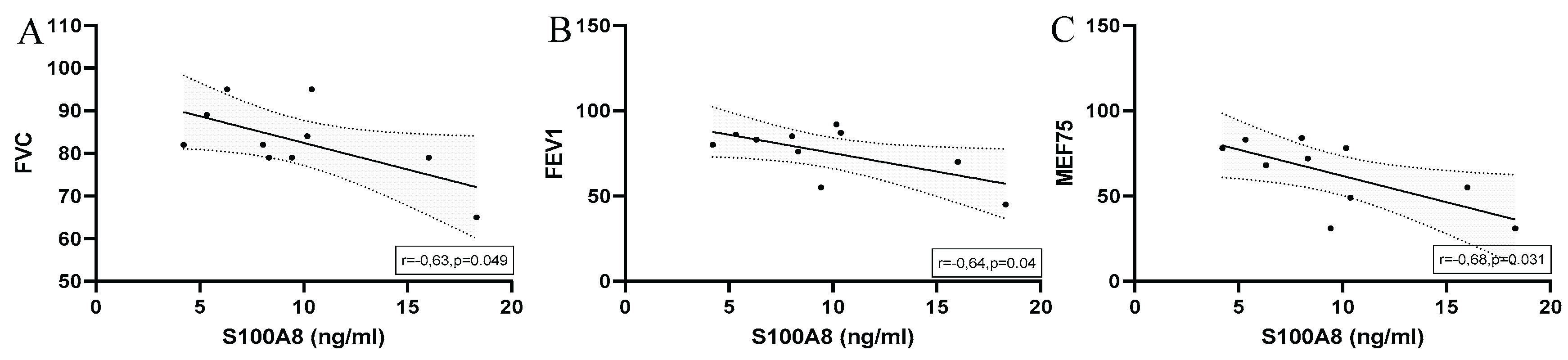
S100A8
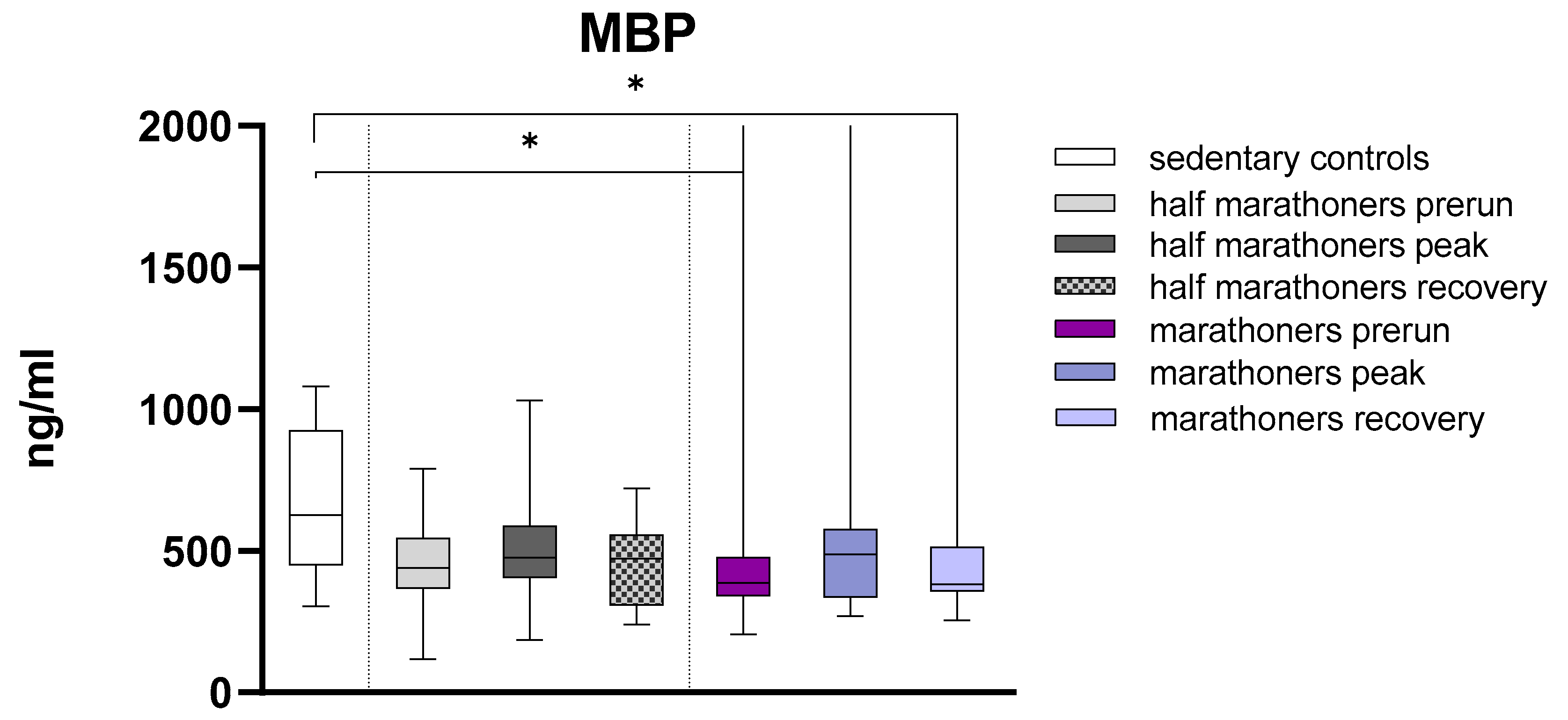
Major Basic Protein
Human Beta Defensin 2
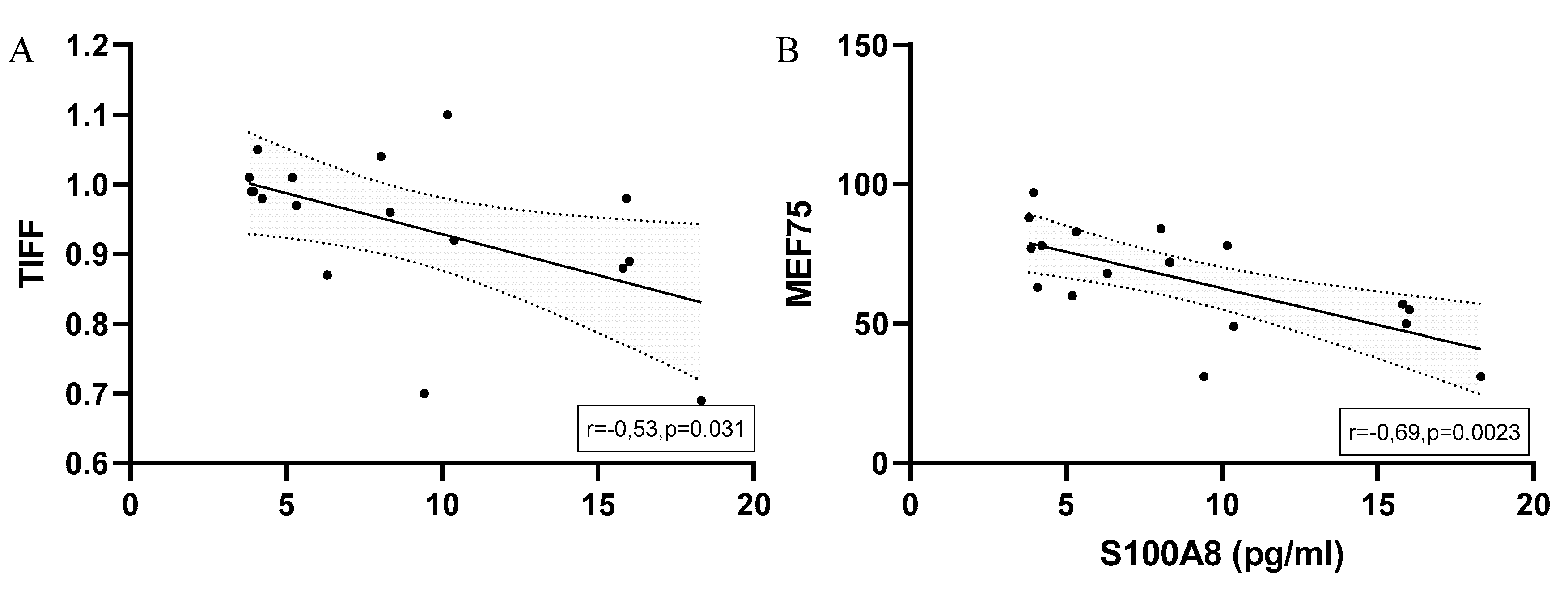
EIB
Correlations of Cytokines and Calculated Antimicrobial Peptides in Half-Marathoners and Marathoners with EIB
Discussion
Limitations
Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
List of Abbreviations
| AGECML | Advanced Glycation End Products Carboxymethyllysine |
| AMP | Antimicrobial Peptide |
| ARDS | Acute Respiratory Distress Syndrome |
| CRP | C-Reactive Protein |
| EIB | Exercise-Induced Bronchoconstriction |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| FEV1 | Forced Expiratory Volume in 1 Second |
| FVC | Forced Vital Capacity |
| HBDF2 / hBD-2 | Human Beta-Defensin 2 |
| HCT | Hematocrit |
| HM | Half-Marathoner(s) |
| HMGB1 | High Mobility Group Box 1 |
| ICU | Intensive Care Unit |
| IL-1RA | Interleukin 1 Receptor Antagonist |
| IL-33 | Interleukin 33 |
| IQR | Interquartile Range |
| ISS | Injury Severity Score |
| MBP | Major Basic Protein |
| MEF25/50/75 | Maximal Expiratory Flow at 25% / 50% / 75% of FVC |
| MPV | Mean Platelet Volume |
| MPO | Myeloperoxidase |
| MXD | Mixed Cell Percentage |
| NE | Neutrophil Elastase |
| PDW | Platelet Distribution Width |
| PLT | Platelet Count |
| RAGE | Receptor for Advanced Glycation End Products |
| r | Correlation Coefficient |
| SD | Standard Deviation |
| sRAGE | Soluble Receptor for Advanced Glycation End Products |
| ST2 | Suppression of Tumorigenicity 2 |
| TIFF | Tiffeneau Index (FEV1/FVC ratio) |
| WBC | White Blood Cell Count |
References
- Poirier, P.; Després, J.P. Exercise in weight management of obesity. Cardiol. Clin. 2001, 19, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Buresh, R. Exercise and glucose control. J. Sports Med. Phys. Fitness 2014, 54, 373–382. [Google Scholar] [PubMed]
- Marwaha, R.K.; Puri, S.; Tandon, N.; Dhir, S.; Agarwal, N.; Bhadra, K.; Saini, N. Effects of sports training & nutrition on bone mineral density in young Indian healthy females. Indian J. Med. Res. 2011, 134, 307–313. [Google Scholar] [PubMed]
- Thune, I.; Furberg, A.S. Physical activity and cancer risk: dose-response and cancer, all sites and site-specific. Med. Sci. Sports Exerc. 2001, 33, S530–50. [Google Scholar] [CrossRef]
- Borghini, A.; Giardini, G.; Tonacci, A.; Mastorci, F.; Mercuri, A.; Mrakic-Sposta, S.; Moretti, S.; Andreassi, M.G.; Pratali, L. Chronic and acute effects of endurance training on telomere length. Mutagenesis 2015, 30, 711–716. [Google Scholar] [CrossRef]
- Nieman, D.C.; Henson, D.A.; Smith, L.L.; Utter, A.C.; Vinci, D.M.; Davis, J.M.; Kaminsky, D.E.; Shute, M. Cytokine changes after a marathon race. J. Appl. Physiol. 2001, 91, 109–114. [Google Scholar] [CrossRef]
- Cantó, E.; Roca, E.; Perea, L.; Rodrigo-Troyano, A.; Suarez-Cuartin, G.; Giner, J.; Feliu, A.; Soria, J.M.; Nescolarde, L.; Vidal, S.; Sibila, O. Salivary immunity and lower respiratory tract infections in non-elite marathon runners. PLoS ONE 2018, 13, e0206059. [Google Scholar] [CrossRef]
- Predel, H.-G. Marathon run: cardiovascular adaptation and cardiovascular risk. Eur. Heart J. 2014, 35, 3091–3098. [Google Scholar] [CrossRef]
- Pasupuleti, M.; Schmidtchen, A.; Malmsten, M. Antimicrobial peptides: key components of the innate immune system. Crit. Rev. Biotechnol. 2012, 32, 143–171. [Google Scholar] [CrossRef]
- Wang, G. The antimicrobial peptide database is 20 years old: Recent developments and future directions. Protein Sci. 2023, 32, e4778. [Google Scholar] [CrossRef]
- Cieślik, M.; Bagińska, N.; Górski, A.; Jończyk-Matysiak, E. Human β-Defensin 2 and Its Postulated Role in Modulation of the Immune Response. Cells 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Siqueiros-Cendón, T.; Arévalo-Gallegos, S.; Iglesias-Figueroa, B.F.; García-Montoya, I.A.; Salazar-Martínez, J.; Rascón-Cruz, Q. Immunomodulatory effects of lactoferrin. Acta Pharmacol. Sin. 2014, 35, 557–566. [Google Scholar] [CrossRef] [PubMed]
- West, N.P.; Pyne, D.B.; Renshaw, G.; Cripps, A.W. Antimicrobial peptides and proteins, exercise and innate mucosal immunity. FEMS Immunol. Med. Microbiol. 2006, 48, 293–304. [Google Scholar] [CrossRef]
- Sonna, L.A.; Angel, K.C.; Sharp, M.A.; Knapik, J.J.; Patton, J.F.; Lilly, C.M. The prevalence of exercise-induced bronchospasm among US Army recruits and its effects on physical performance. Chest 2001, 119, 1676–1684. [Google Scholar] [CrossRef]
- Ng’ang’a, L.W.; Odhiambo, J.A.; Mungai, M.W.; Gicheha, C.M.; Nderitu, P.; Maingi, B.; Macklem, P.T.; Becklake, M.R. Prevalence of exercise induced bronchospasm in Kenyan school children: an urban-rural comparison. Thorax 1998, 53, 919–926. [Google Scholar] [CrossRef]
- Kukafka, D.S.; Lang, D.M.; Porter, S.; Rogers, J.; Ciccolella, D.; Polansky, M.; D’Alonzo, G.E. Exercise-induced bronchospasm in high school athletes via a free running test: incidence and epidemiology. Chest 1998, 114, 1613–1622. [Google Scholar] [CrossRef]
- Anderson, S.D.; Kippelen, P. Exercise-induced bronchoconstriction: pathogenesis. Curr. Allergy Asthma Rep. 2005, 5, 116–122. [Google Scholar] [CrossRef]
- Anderson, S.D.; Kippelen, P. Airway injury as a mechanism for exercise-induced bronchoconstriction in elite athletes. J. Allergy Clin. Immunol. 2008, 122, 225–35. [Google Scholar] [CrossRef]
- Schröder, J.M.; Harder, J. Human beta-defensin-2. Int. J. Biochem. Cell Biol. 1999, 31, 645–651. [Google Scholar] [CrossRef]
- Seo, E.S.; Blaum, B.S.; Vargues, T.; De Cecco, M.; Deakin, J.A.; Lyon, M.; Barran, P.E.; Campopiano, D.J.; Uhrín, D. Interaction of human β-defensin 2 (HBD2) with glycosaminoglycans. Biochemistry 2010, 49, 10486–10495. [Google Scholar] [CrossRef] [PubMed]
- Yanagi, S.; Ashitani, J.; Imai, K.; Kyoraku, Y.; Sano, A.; Matsumoto, N.; Nakazato, M. Significance of human beta-defensins in the epithelial lining fluid of patients with chronic lower respiratory tract infections. Clin. Microbiol. Infect. 2007, 13, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Sun, B.B.; Wang, T.; Wang, X.; Li, J.Q.; Wang, H.X.; Zhang, S.F.; Liu, D.S.; Liu, L.; Xu, D.; Ou, X.M.; Chen, Y.J.; Yang, T.; Zhou, H.; Wen, F.Q. Cigarette smoke enhances {beta}-defensin 2 expression in rat airways via nuclear factor-{kappa}B activation. Eur. Respir. J. 2010, 36, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Borchers, N.S.; Santos-Valente, E.; Toncheva, A.A.; Wehkamp, J.; Franke, A.; Gaertner, V.D.; Nordkild, P.; Genuneit, J.; Jensen, B.A.H.; Kabesch, M. Human β-Defensin 2 Mutations Are Associated With Asthma and Atopy in Children and Its Application Prevents Atopic Asthma in a Mouse Model. Front. Immunol. 2021, 12, 636061. [Google Scholar] [CrossRef]
- Patricia Rosete Olvera, D.; Cabello Gutiérrez, C. Multifunctional Activity of the β-Defensin-2 during Respiratory Infections. In Immune response activation and immunomodulation; K. Tyagi, R., S. Bisen, P., Eds.; IntechOpen, 2019 ISBN 978-1-78985-151-9.
- Liu, S.; He, L.-R.; Wang, W.; Wang, G.-H.; He, Z.-Y. Prognostic value of plasma human β-defensin 2 level on short-term clinical outcomes in patients with community-acquired pneumonia: a preliminary study. Respir. Care 2013, 58, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Menees, S.B.; Powell, C.; Kurlander, J.; Goel, A.; Chey, W.D. A meta-analysis of the utility of C-reactive protein, erythrocyte sedimentation rate, fecal calprotectin, and fecal lactoferrin to exclude inflammatory bowel disease in adults with IBS. Am. J. Gastroenterol. 2015, 110, 444–454. [Google Scholar] [CrossRef]
- Wang, S.; Song, R.; Wang, Z.; Jing, Z.; Wang, S.; Ma, J. S100A8/A9 in Inflammation. Front. Immunol. 2018, 9, 1298. [Google Scholar] [CrossRef]
- Ji, X.; Nie, C.; Yao, Y.; Ma, Y.; Huang, H.; Hao, C. S100A8/9 modulates perturbation and glycolysis of macrophages in allergic asthma mice. PeerJ 2024, 12, e17106. [Google Scholar] [CrossRef]
- Zhao, J.; Endoh, I.; Hsu, K.; Tedla, N.; Endoh, Y.; Geczy, C.L. S100A8 modulates mast cell function and suppresses eosinophil migration in acute asthma. Antioxid. Redox Signal. 2011, 14, 1589–1600. [Google Scholar] [CrossRef]
- Berry, A.; Busse, W.W. Biomarkers in asthmatic patients: Has their time come to direct treatment? J. Allergy Clin. Immunol. 2016, 137, 1317–1324. [Google Scholar] [CrossRef]
- Lee, L.-Y.; Gu, Q.; Lin, A.-H.; Khosravi, M.; Gleich, G. Airway hypersensitivity induced by eosinophil granule-derived cationic proteins. Pulm. Pharmacol. Ther. 2019, 57, 101804. [Google Scholar] [CrossRef]
- Plager, D.A.; Loegering, D.A.; Weiler, D.A.; Checkel, J.L.; Wagner, J.M.; Clarke, N.J.; Naylor, S.; Page, S.M.; Thomas, L.L.; Akerblom, I.; Cocks, B.; Stuart, S.; Gleich, G.J. A novel and highly divergent homolog of human eosinophil granule major basic protein. J. Biol. Chem. 1999, 274, 14464–14473. [Google Scholar] [CrossRef]
- Tai, P.C.; Ackerman, S.J.; Spry, C.J.; Dunnette, S.; Olsen, E.G.; Gleich, G.J. Deposits of eosinophil granule proteins in cardiac tissues of patients with eosinophilic endomyocardial disease. Lancet 1987, 1, 643–647. [Google Scholar] [CrossRef]
- Gleich, G.J.; Flavahan, N.A.; Fujisawa, T.; Vanhoutte, P.M. The eosinophil as a mediator of damage to respiratory epithelium: a model for bronchial hyperreactivity. J. Allergy Clin. Immunol. 1988, 81, 776–781. [Google Scholar] [CrossRef]
- Stelts, D.; Egan, R.W.; Falcone, A.; Garlisi, C.G.; Gleich, G.J.; Kreutner, W.; Kung, T.T.; Nahrebne, D.K.; Chapman, R.W.; Minnicozzi, M. Eosinophils retain their granule major basic protein in a murine model of allergic pulmonary inflammation. Am. J. Respir. Cell Mol. Biol. 1998, 18, 463–470. [Google Scholar] [CrossRef]
- Evans, C.M.; Fryer, A.D.; Jacoby, D.B.; Gleich, G.J.; Costello, R.W. Pretreatment with antibody to eosinophil major basic protein prevents hyperresponsiveness by protecting neuronal M2 muscarinic receptors in antigen-challenged guinea pigs. J. Clin. Invest. 1997, 100, 2254–2262. [Google Scholar] [CrossRef]
- Gundel, R.H.; Letts, L.G.; Gleich, G.J. Human eosinophil major basic protein induces airway constriction and airway hyperresponsiveness in primates. J. Clin. Invest. 1991, 87, 1470–1473. [Google Scholar] [CrossRef]
- Bonsignore, M.R.; Morici, G.; Riccobono, L.; Insalaco, G.; Bonanno, A.; Profita, M.; Paternò, A.; Vassalle, C.; Mirabella, A.; Vignola, A.M. Airway inflammation in nonasthmatic amateur runners. Am. J. Physiol. Lung Cell. Mol. Physiol. 2001, 281, L668–76. [Google Scholar] [CrossRef]
- Hu, H.; Gao, X.; Sun, Y.; Zhou, J.; Yang, M.; Xu, Z. Alpha-actinin-2, a cytoskeletal protein, binds to angiogenin. Biochem. Biophys. Res. Commun. 2005, 329, 661–667. [Google Scholar] [CrossRef]
- Tello-Montoliu, A.; Patel, J.V.; Lip, G.Y.H. Angiogenin: a review of the pathophysiology and potential clinical applications. J. Thromb. Haemost. 2006, 4, 1864–1874. [Google Scholar] [CrossRef]
- Fett, J.W.; Strydom, D.J.; Lobb, R.R.; Alderman, E.M.; Bethune, J.L.; Riordan, J.F.; Vallee, B.L. Isolation and characterization of angiogenin, an angiogenic protein from human carcinoma cells. Biochemistry 1985, 24, 5480–5486. [Google Scholar] [CrossRef]
- Strydom, D.J. The angiogenins. Cell. Mol. Life Sci. 1998, 54, 811–824. [Google Scholar] [CrossRef]
- Olson, K.A.; Verselis, S.J.; Fett, J.W. Angiogenin is regulated in vivo as an acute phase protein. Biochem. Biophys. Res. Commun. 1998, 242, 480–483. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Y.-P.; Kirsner, R.S. Angiogenesis in wound repair: angiogenic growth factors and the extracellular matrix. Microsc. Res. Tech. 2003, 60, 107–114. [Google Scholar] [CrossRef]
- Fallon, K.E. The acute phase response and exercise: the ultramarathon as prototype exercise. Clin. J. Sport Med. 2001, 11, 38–43. [Google Scholar] [CrossRef]
- Bekos, C.; Zimmermann, M.; Unger, L.; Janik, S.; Hacker, P.; Mitterbauer, A.; Koller, M.; Fritz, R.; Gäbler, C.; Kessler, M.; Nickl, S.; Didcock, J.; Altmann, P.; Haider, T.; Roth, G.; Klepetko, W.; Ankersmit, H.J.; Moser, B. Non-professional marathon running: RAGE axis and ST2 family changes in relation to open-window effect, inflammation and renal function. Sci. Rep. 2016, 6, 32315. [Google Scholar] [CrossRef]
- Bekos, C.; Zimmermann, M.; Unger, L.; Janik, S.; Mitterbauer, A.; Koller, M.; Fritz, R.; Gäbler, C.; Didcock, J.; Kliman, J.; Klepetko, W.; Ankersmit, H.J.; Moser, B. Exercise-induced bronchoconstriction, temperature regulation and the role of heat shock proteins in non-asthmatic recreational marathon and half-marathon runners. Sci. Rep. 2019, 9, 4168. [Google Scholar] [CrossRef]
- Nieman, D.C.; Wentz, L.M. The compelling link between physical activity and the body’s defense system. J. Sport Health Sci. 2019, 8, 201–217. [Google Scholar] [CrossRef]
- Mooren, F.C.; Lechtermann, A.; Fobker, M.; Brandt, B.; Sorg, C.; Völker, K.; Nacken, W. The response of the novel pro-inflammatory molecules S100A8/A9 to exercise. Int. J. Sports Med. 2006, 27, 751–758. [Google Scholar] [CrossRef]
- Górecka, M.; Krzemiński, K.; Buraczewska, M.; Kozacz, A.; Dąbrowski, J.; Ziemba, A.W. Effect of mountain ultra-marathon running on plasma angiopoietin-like protein 4 and lipid profile in healthy trained men. Eur. J. Appl. Physiol. 2020, 120, 117–125. [Google Scholar] [CrossRef]
- Legaz-Arrese, A.; George, K.; Carranza-García, L.E.; Munguía-Izquierdo, D.; Moros-García, T.; Serrano-Ostáriz, E. The impact of exercise intensity on the release of cardiac biomarkers in marathon runners. Eur. J. Appl. Physiol. 2011, 111, 2961–2967. [Google Scholar] [CrossRef]
- Gillum, T.L.; Kuennen, M.; Gourley, C.; Schneider, S.; Dokladny, K.; Moseley, P. Salivary antimicrobial protein response to prolonged running. Biol. Sport 2013, 30, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Usui, T.; Yoshikawa, T.; Orita, K.; Ueda, S.-Y.; Katsura, Y.; Fujimoto, S.; Yoshimura, M. Changes in salivary antimicrobial peptides, immunoglobulin A and cortisol after prolonged strenuous exercise. Eur. J. Appl. Physiol. 2011, 111, 2005–2014. [Google Scholar] [CrossRef]
- Shimizu, K.; Hanaoka, Y.; Akama, T.; Kono, I. Ageing and free-living daily physical activity effects on salivary beta-defensin 2 secretion. J. Sports Sci. 2017, 35, 617–623. [Google Scholar] [CrossRef]
- Ito, R.; Uchino, T.; Uchida, M.; Fujie, S.; Iemitsu, K.; Kojima, C.; Nakamura, M.; Shimizu, K.; Tanimura, Y.; Shinohara, Y.; Hashimoto, T.; Isaka, T.; Iemitsu, M. Acute salivary antimicrobial peptide secretion response to different exercise intensities and durations. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2024. [Google Scholar] [CrossRef]

| Sedentary controls | HM baseline | HM peak | HM recovery | M baseline | Mpeak | M recovery | p-value | |
| Angiogenin (ng/ml) | 233 (182-267) | 306 (204-379) | 365 (272-542) | 303 (221-424) | 260 (204-381) | 277 (164-393) | 218 (155-328) | 0.015 |
| S100A8/A9 (ng/ml) | 2958 (1121-4360) | 2431 (1710-3255) | 7746 (5561-9659) | 3042 (1196-3042) | 3579 (1052-5781) | 9750 (4719-12558) | 9750 (4719-12558) | <0.001 |
| S100A8 (pg/ml) | 8016 (6558-9492) | 4848 (3314-8478) | 5247 (4235-7489) | 5161 (3594-7036) | 6796 (5504-8751) | 8886 (6475-14038) | 7230 (5488-9029) | <0.001 |
| Major Basic Protein (ng/ml) | 627 (450-926) | 440 (368-547) | 476 (405-589) | 472 (307-558) | 388 (341-477) | 488 (335-578) | 383 (357-514) | 0.022 |
| HumanBetaDefensin2 (ng/ml) | 2.38 (1.15-3.16) | 2.18 (1.72-2.74) | 4.91(3.64-5.89) | 2.21 (1.60-2.64) | 2.02 (1.77-2.37) | 5.72 (5.10-6.61) | 2.27 (1.75-3.69) | <0.001 |
| GROUP | SUBGROUP | VARIABLES CORRELATED | CORRELATION COEFFICIENT (R) | P-VALUE |
|---|---|---|---|---|
| BASELINE | HM | ST2 ↔ Angiogenin | 0.70 | 0.002 |
| BASELINE | M | ST2 ↔ Angiogenin | 0.84 | 0.004 |
| BASELINE | HM | S100A8 ↔ esRAGE | 0.89 | 0.007 |
| BASELINE | M | MBP ↔ esRAGE | 0.95 | <0.001 |
| BASELINE | M | S100A8 ↔ CRP | 0.68 | 0.02 |
| PEAK | HM | S100A8/A9 ↔ MBP | 0.90 | 0.006 |
| PEAK | M | S100A8/A9 ↔ IL33 | 0.79 | 0.01 |
| PEAK | M | S100A8 ↔ AGECML | -0.64 | 0.05 |
| RECOVERY | HM | Angiogenin ↔ HBDF2 | 0.84 | 0.02 |
| RECOVERY | M | MBP ↔ esRAGE | 0.84 | 0.003 |
| RECOVERY | HM | Angiogenin ↔ HBDF2 | 0.85 | 0.032 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




