Submitted:
19 May 2025
Posted:
20 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Crampton, M. R. In Organic Reaction Mechanisms (Knipe, A. C.; Moloney, M. G., Eds), Wiley, New York, 2020, pp. 213-295.
- Koerner, W. Fatti per servire alla determinazione del luogo chimico nelle sostanze aromatiche. Giornale di Scienze Naturali ed Economiche 1869, 5, 212-256.
- Koerner, W. Studi sull’isomeria delle così dette sostanze aromatiche a sei atomi di carbonio. Gaz. Chim. Ital. 1874, 4, 305-446.
- Holleman, A. F. Trattato di Chimica Organica, Società Editrice Libraria, Milano, 1927.
- Kermack, W. O.; Robinson, R. An explanation of the property of induced polarity of atoms and an interpretation of the theory of partial valencies on an electronic basis. J. Chem. Soc. Trans. 1922, 121, 427-440. [CrossRef]
- Lapworth, A. A theoretical derivation of the principle of induces alternate polarities. J. Chem. Soc. Trans. 1922, 121, 416-427. [CrossRef]
- Saltzman, M. Arthur Lapworth. J. Chem. Ed. 1972, 49, 750-752.
- Karrer, P. Trattato di Chimica Organica, Sansoni, Firenze, 1965.
- Wheland, G. W. A quantum mechanical investigation of the orientation of substitution in aromatic molecules. J. Am. Chem. Soc. 1942, 64, 900-908. [CrossRef]
- Olah, G. A. Stable carbonium ions. IX. Methylbenzenonium hexafluoroantimonates. J. Am. Chem. Soc. 1965, 87, 1103-1108. [CrossRef]
- Olah, G. A.; Kiovsky, T. E. Stable carbonium ions. LI. Fluorobenzenonium ions. J. Am. Chem. Soc. 1967, 89, 5692-5694. [CrossRef]
- Olah, G. A.; Schlosberg, R. H.; Richard, D.; Porter, R. D.; Mo, Y. K.; Kelly, D. P.; Mateescu, G. D. Stable carbocations. CXXIV. Benzenium ion and monoalkylbenzenium ions. J. Am. Chem. Soc. 1972, 94, 2034-2043. [CrossRef]
- Corey, E. J.; Barcza, S.; Klotmann, G. Directed conversion of the phenoxy grouping into a variety of cyclic polyfunctional systems. J. Am. Chem. Soc. 1969, 91, 4782-4786. [CrossRef]
- Hahn, R. C.; Strack, D. L. Ipso nitration. II. Novel products and true positional selectivities in nitration of p-cymene. J. Am. Chem. Soc. 1974, 96, 4335-4337. [CrossRef]
- Carey, F. A.; Sundberg, R. J. Advanced Organic Chemistry, Part A: Structure and Mechanisms, Springer, New York, 2008.
- Fukui, K.; Yonezawa, T.; Shingu, H. A molecular orbital theory of reactivity of aromatic hydrocarbons. J. Chem. Phys. 1952, 20, 722-725. [CrossRef]
- Seeman, J. I. Kenichi Fukui, Frontier Molecular Orbital Theory, and the Woodward-Hoffmann Rules. Part II. A Sleeping Beauty in Chemistry. Chem. Rec. 2022, 22, e202100300.
- Elliott, R. J.; Sackwild, V.; Richards, W. G. Quantitative frontier orbital theory: Part I. Electrophilic aromatic substitution. J. Mol. Struct. 1982, 86, 301-314. [CrossRef]
- Klopman, G. Chemical reactivity and the concept of charge- and frontier-controlled reactions. J. Am. Chem. Soc. 1968, 90, 223-234.
- Salem, L. Intermolecular orbital theory of the interaction between conjugated systems. I. General theory. J. Am. Chem. Soc. 1968, 90, 543-552. [CrossRef]
- Salem, L. Intermolecular orbital theory of the interaction between conjugated systems. II. Thermal and photochemical cycloaddition. J. Am. Chem. Soc. 1968, 90, 553-566. [CrossRef]
- Domingo, L. R. Molecular Electron Density Theory: A Modern View of Reactivity in Organic Chemistry. Molecules 2016, 21, 1319. [CrossRef]
- Stuyver, T.; Danovich, D.; De Proft, F.; Shaik, S. Electrophilic aromatic substitution reactions: mechanistic landascape, electrostatic and electric-field control of reaction rates, and mechanistic crossovers, J. Am. Chem. Soc. 2019, 141, 9719-9730-.
- Galabov, B.; Nalbantova, D.; von R. Schleyer, P.; Schaefer, H. F. III, Electrophilic aromatic substitution: new insighta into an old class of reactions. Acc. Chem. Res. 2016, 49, 1191-1199.
- Liu, S. Where does the electron go? The nature of ortho/para and meta group directing in electrophilic aromatic substitution. J. Chem. Phys. 2014, 141, 194109. [CrossRef]
- Gaussian 09, Revision A.1, M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Na-katsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Son-nenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Nor-mand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Re-ga, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox, Gaussian, Inc., Wallingford CT, 2009.
- Parr, R. G.; Yang, W. Density Functional Theory of Atoms and Molecules, Oxford Universi-ty Press, Oxford, UK, 1989.
- Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange J. Chem. Phys. 1993, 98, 5648–5652.
- Pounder, F. E. The nitration of nitrobenzene. PhD Thesis, Durham University, 1935.
- Baciocchi, E.; Cacace, F.; Ciranni, G.; Illuminati, G. Isomeric distributions and relative reactivities in the uncatalyzed chlorination of benzonitrile, nitrobenzene, and benzotrifluoride. The directive effects of electron-withdrawing substituents as a function of reagent and solvent. J. Am. Chem. Soc. 1972, 94, 7030-7034. [CrossRef]
- Heidar-Zadeh, F.; Ayers, P. W.; Verstraelen, T.; Vinogradov, I.; Vöhringer-Martinez, E.; Bultinck, P. Information-theoretic approaches to atoms-in.molecules: Hirshfeld family of partitioning schemes. J. Phys. Chem. A 2018, 122, 4219-4245. [CrossRef]
- Liu, S. Quantifying reactivity of electrophilic aromatic substitution reactions with Hirshfeld charge. J. Phys. Chem. A 2015, 119, 3107-3111. [CrossRef]
- Johnstone, J. F.; Ridd, J. H.; Sandall, J. P. B. The borderline between the classical and the electron transfer process in nitration by the nitronium ion. Chem. Commun. 1989, 244-246. [CrossRef]
- Brown, J. J.; Cockroft, S. L. Aromatic reactivity revealed: beyond resonance theory and frontier orbitals. Chem. Sci. 2013, 4, 1772-1780. [CrossRef]
- Davey, W.; Gwilt, J. R. The preparation of mononitrobenzaldehdes. J. Chem. Soc. 1950, 204-208.
- Stock, L. M. Aromatic Substitution Reactions. Prentice-Hall, Englewood Cliffs, New Jersey, USA, 1968, p. 63.
- Kamm, O.; Segur, J. B. Methyl m-nitrobenzoate. Org. Synth. 1923, 3, 71-72.
- Yee, H. Y.; Boyle, A. J. The chlorination of benzoic acid in aqueous system by use of oxidizing acids. J. Chem. Soc. 1955, 4139-4140. [CrossRef]
- Pearson, D. E.; Stamper, W. E.; Suthers, B. R. The swamping catalyst effect. V. The halogenation of aromatic acid derivatives. J. Org. Chem. 1963, 28, 3147-3149. [CrossRef]

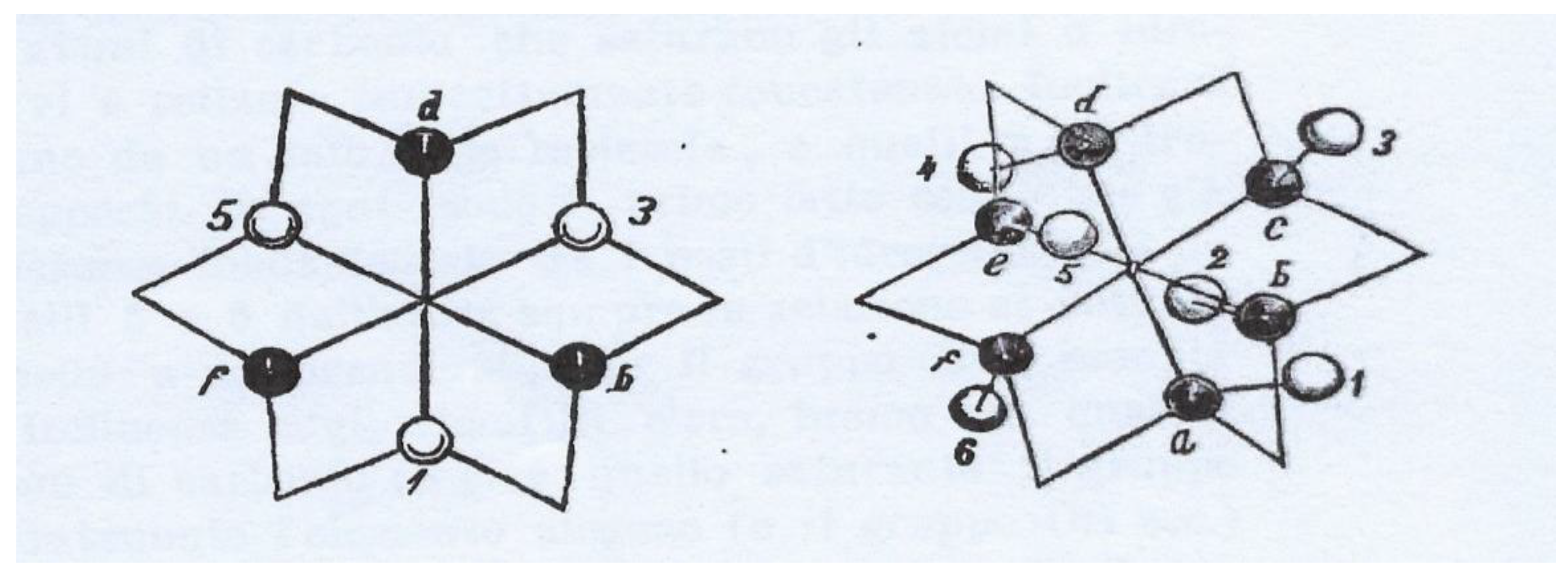
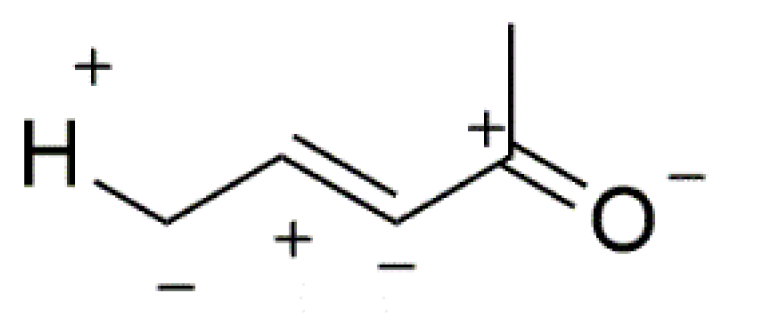
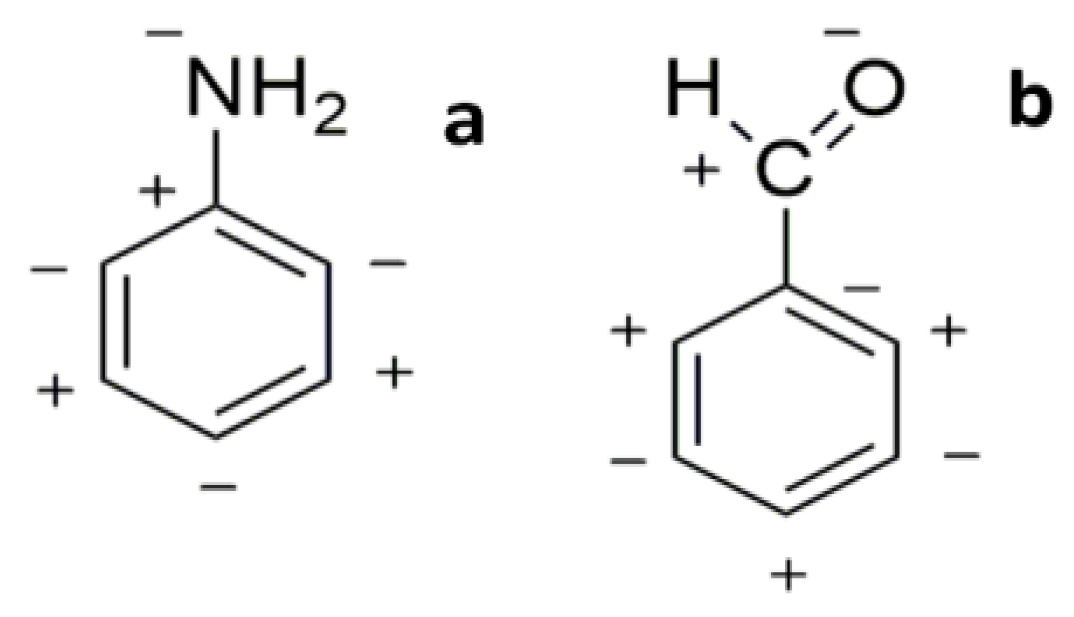
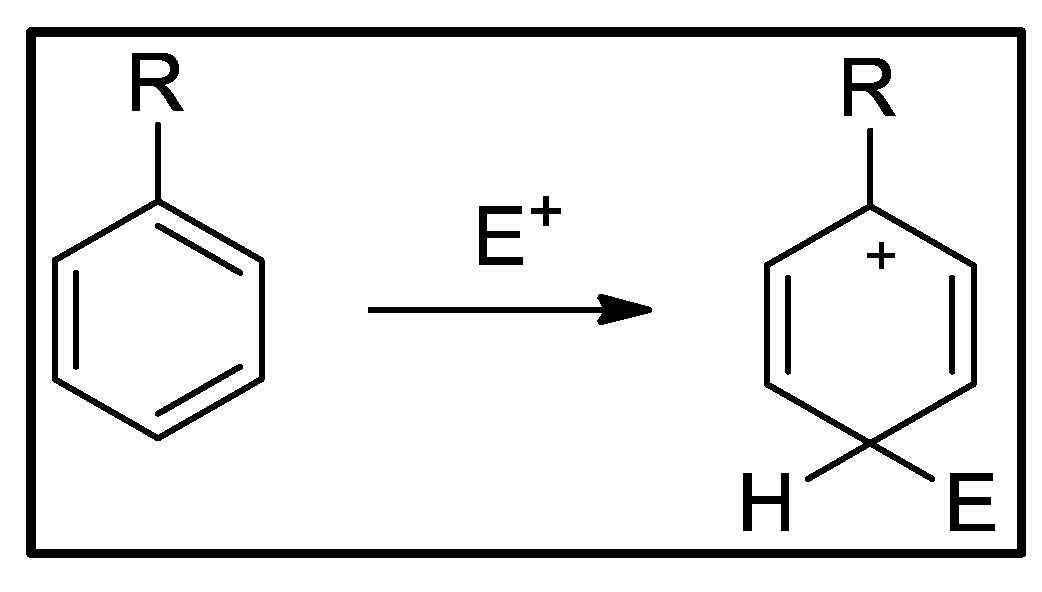
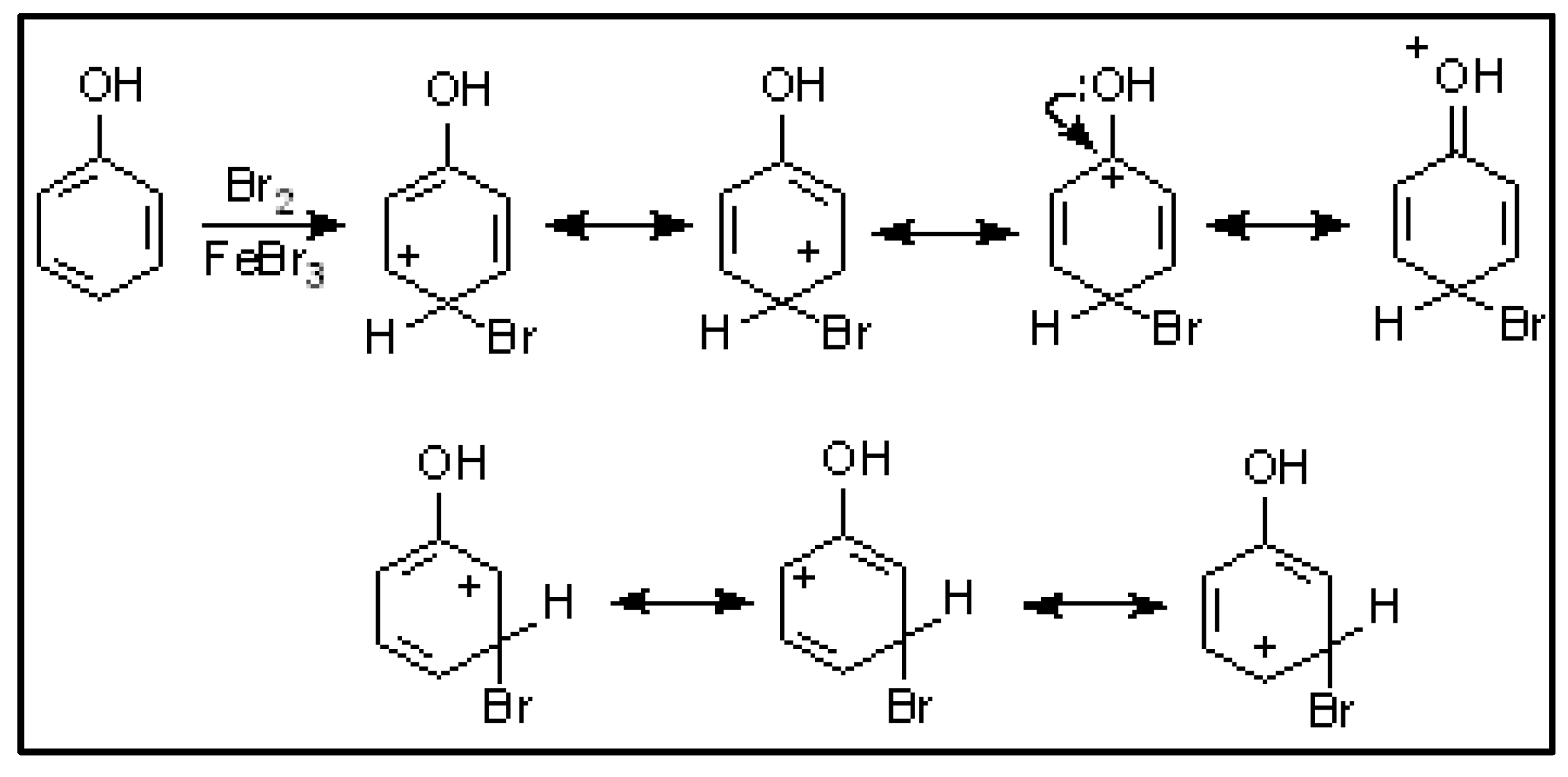
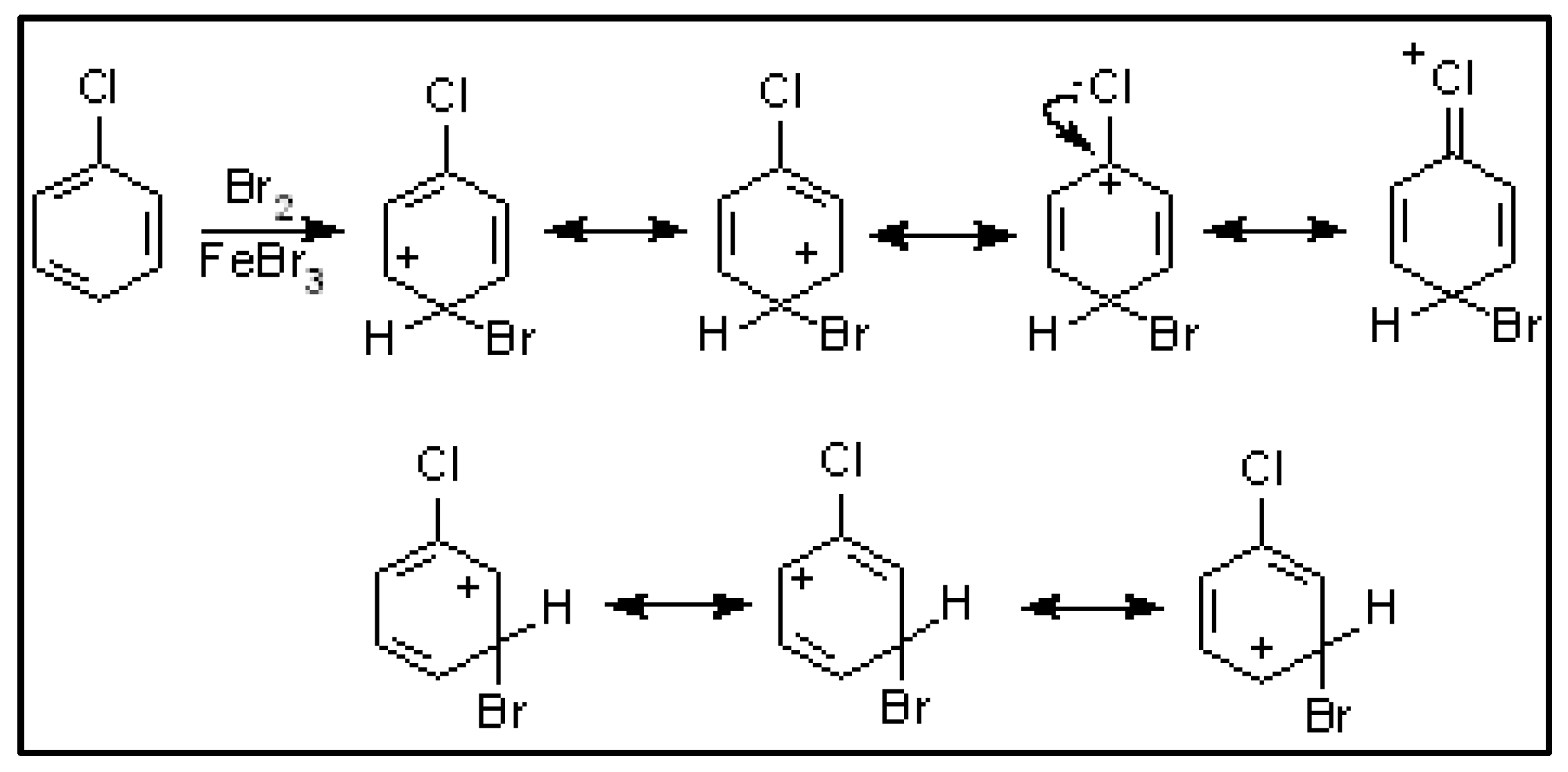
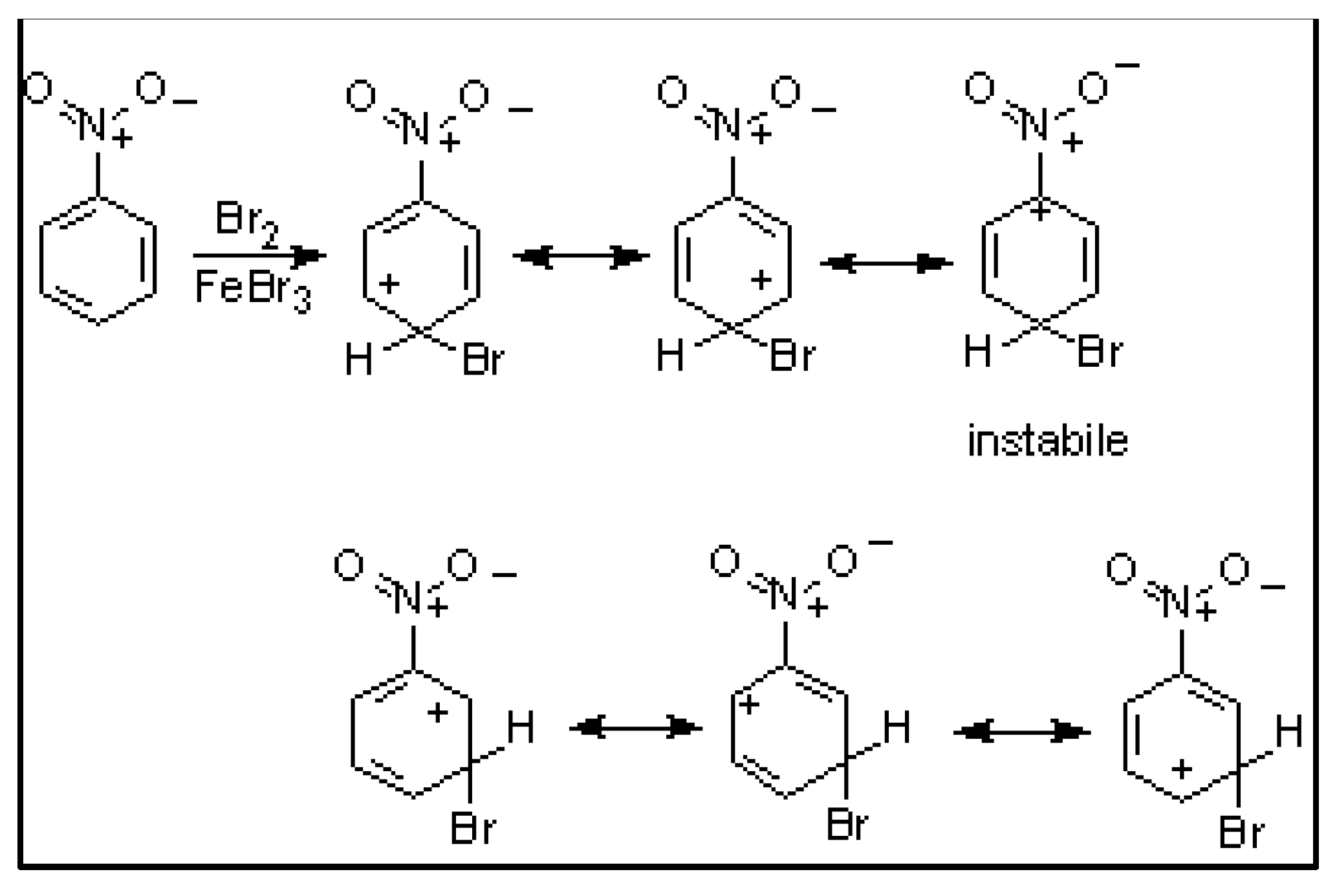
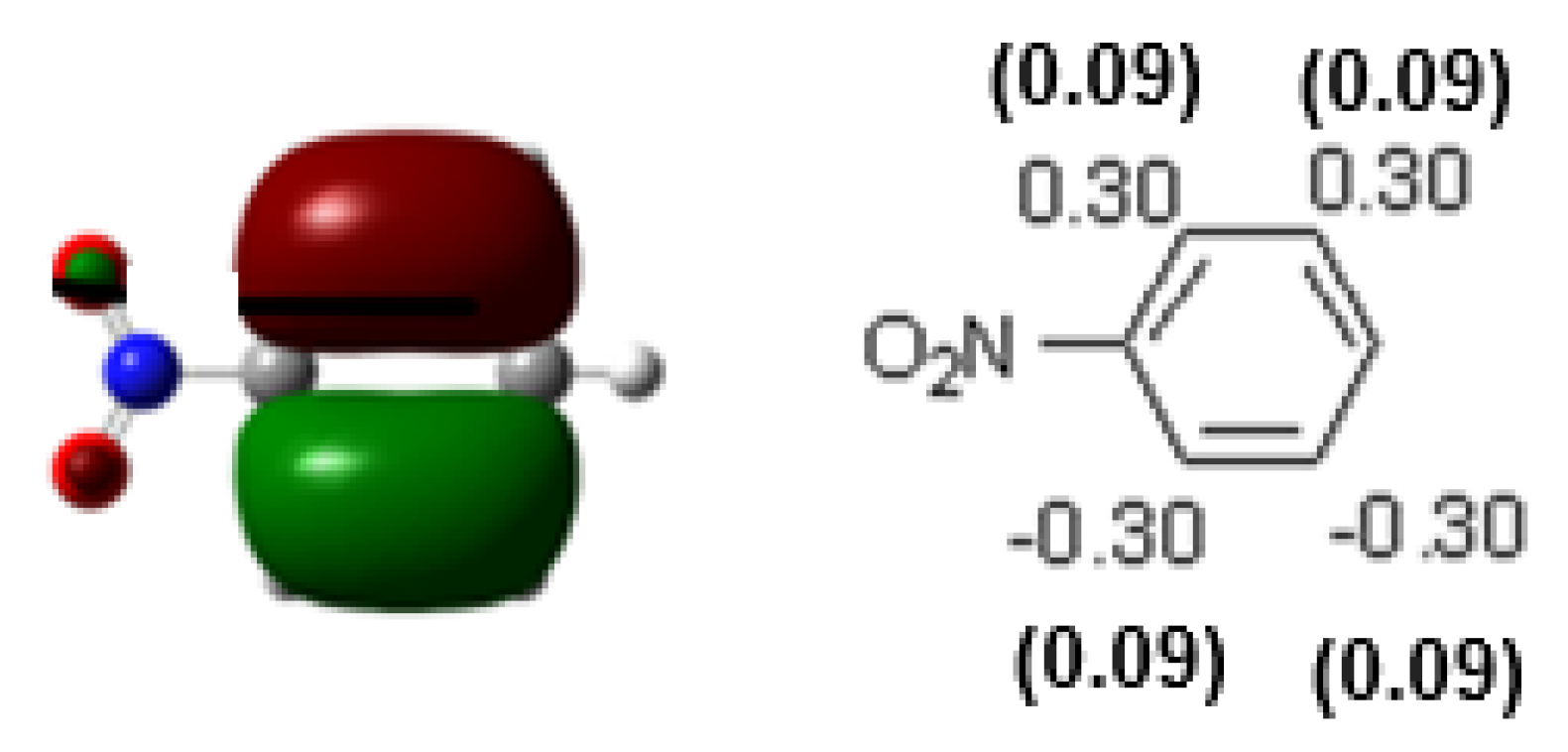
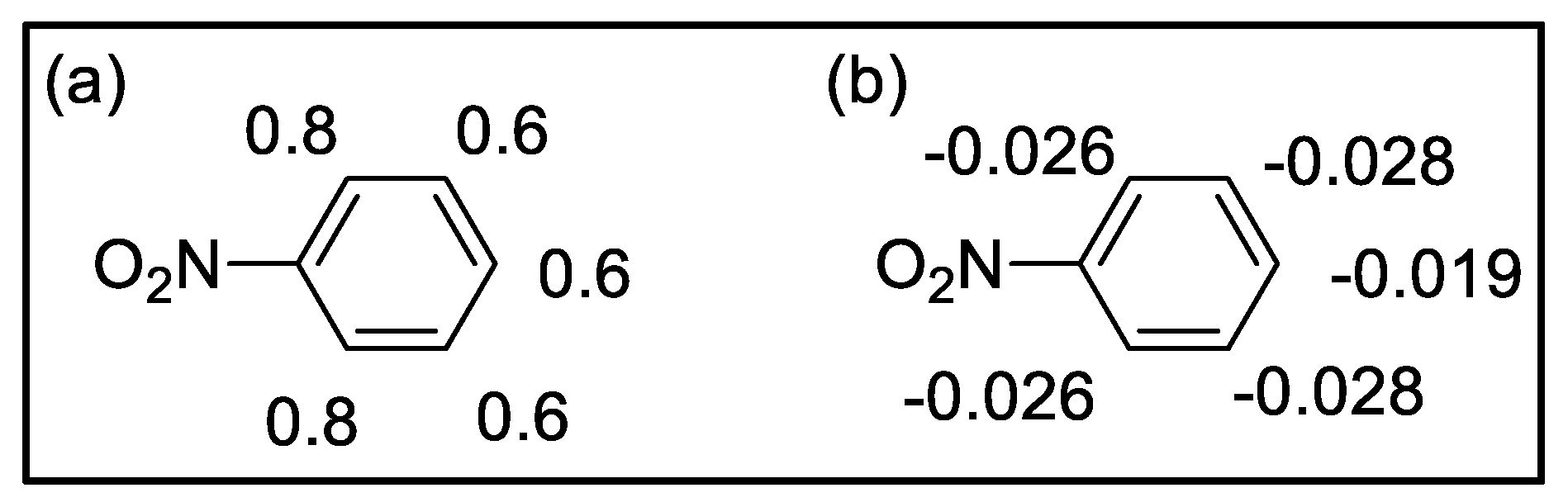
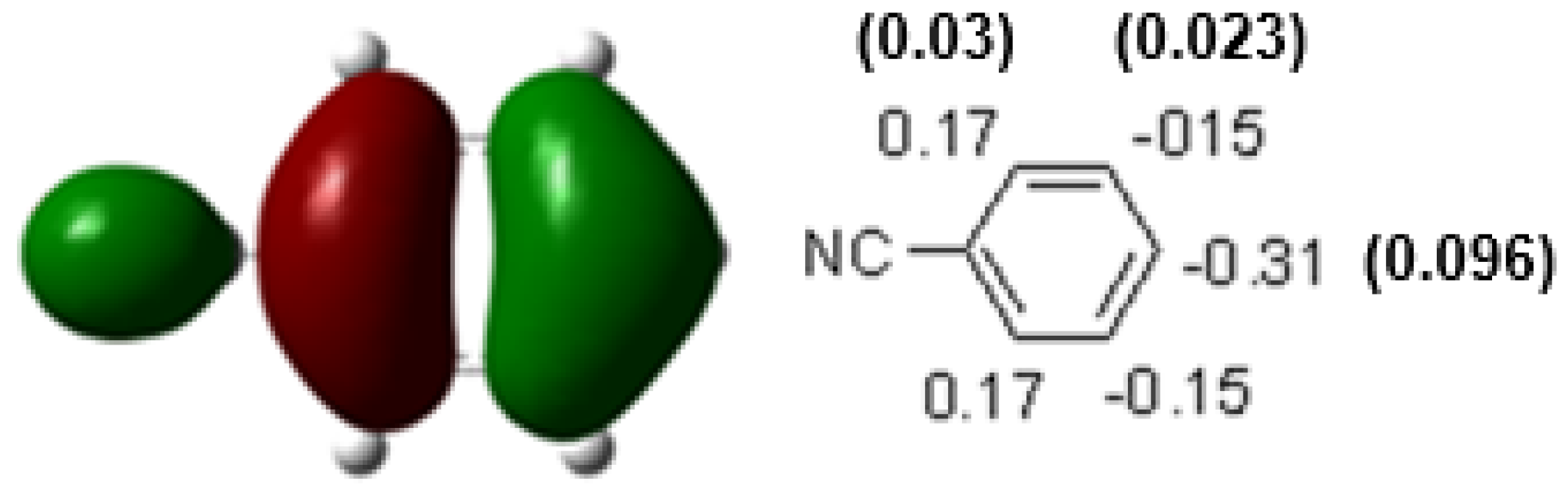
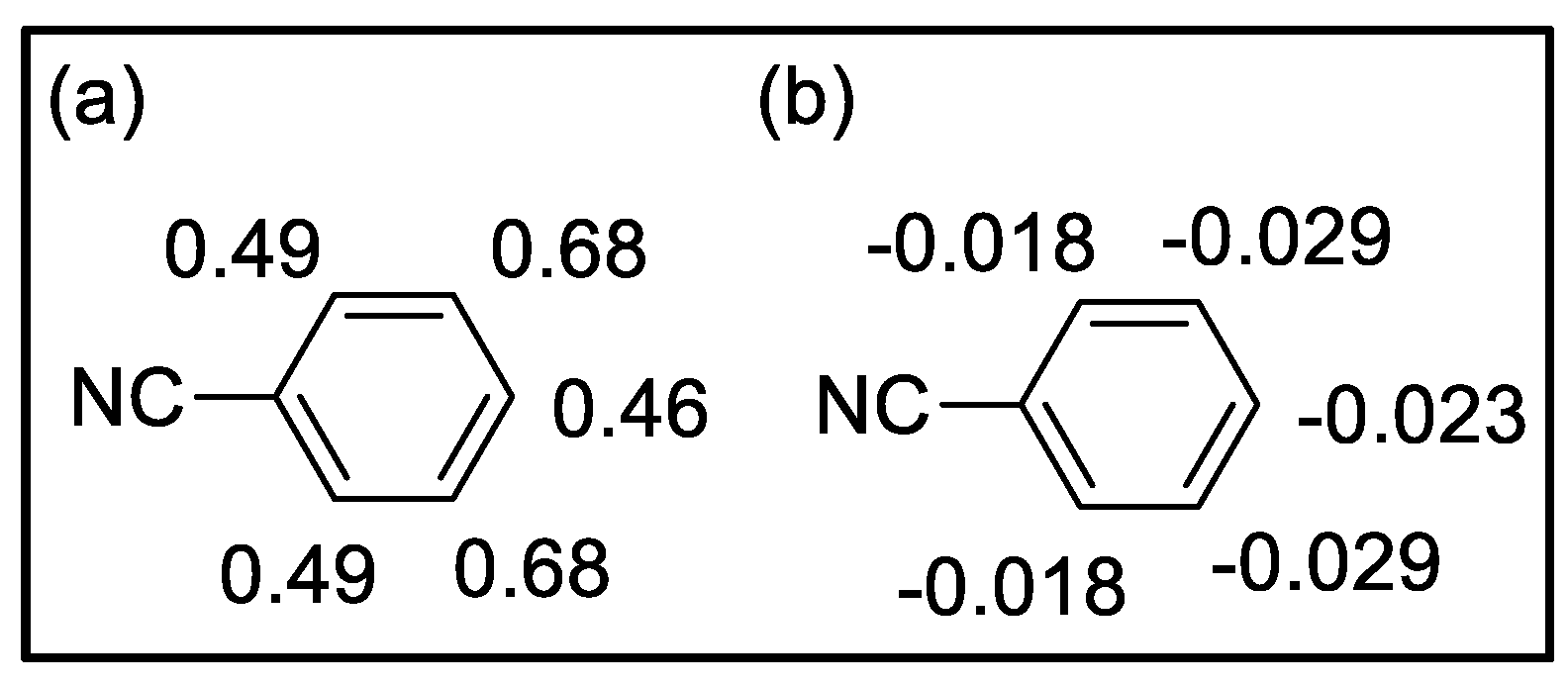
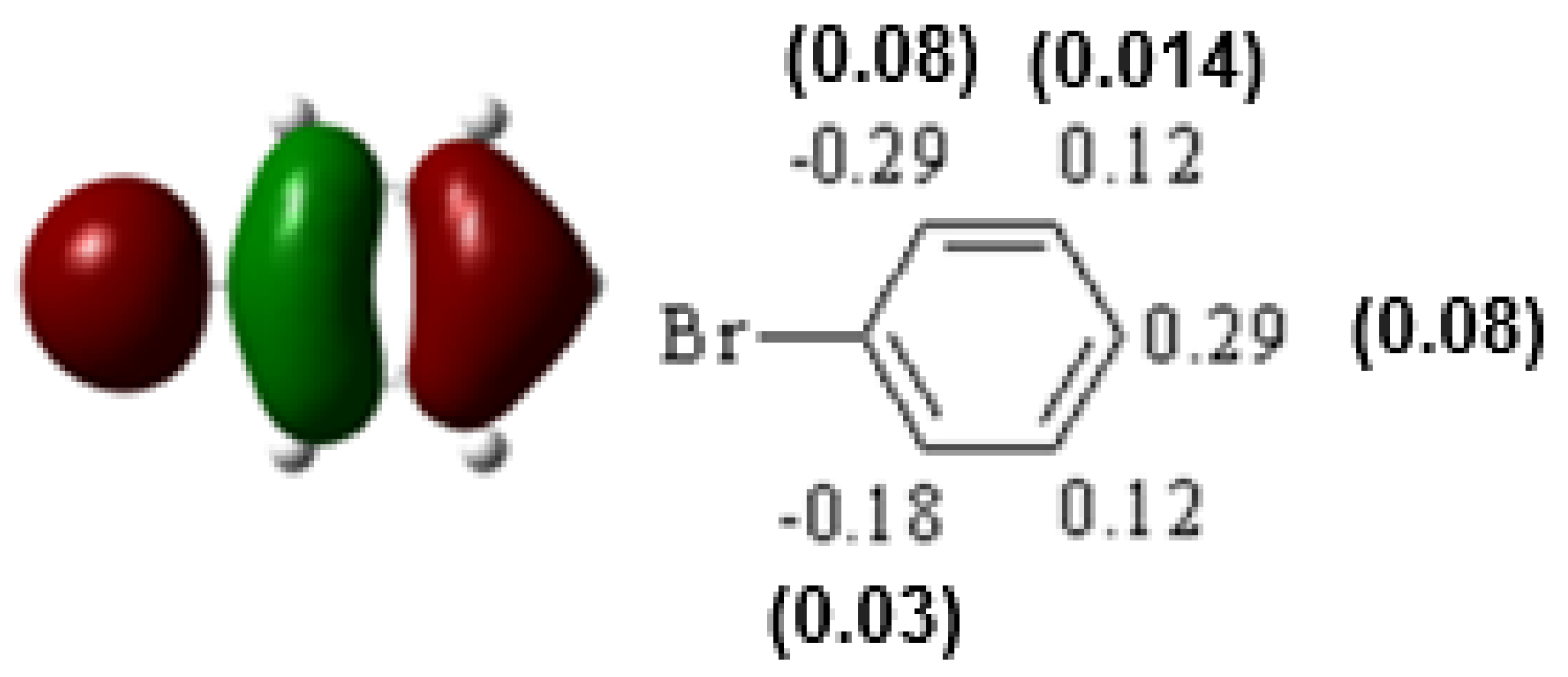

| Compound | HOMO | NHOMO | Energy [eV] |
Atomic coefficients (Electronic density) |
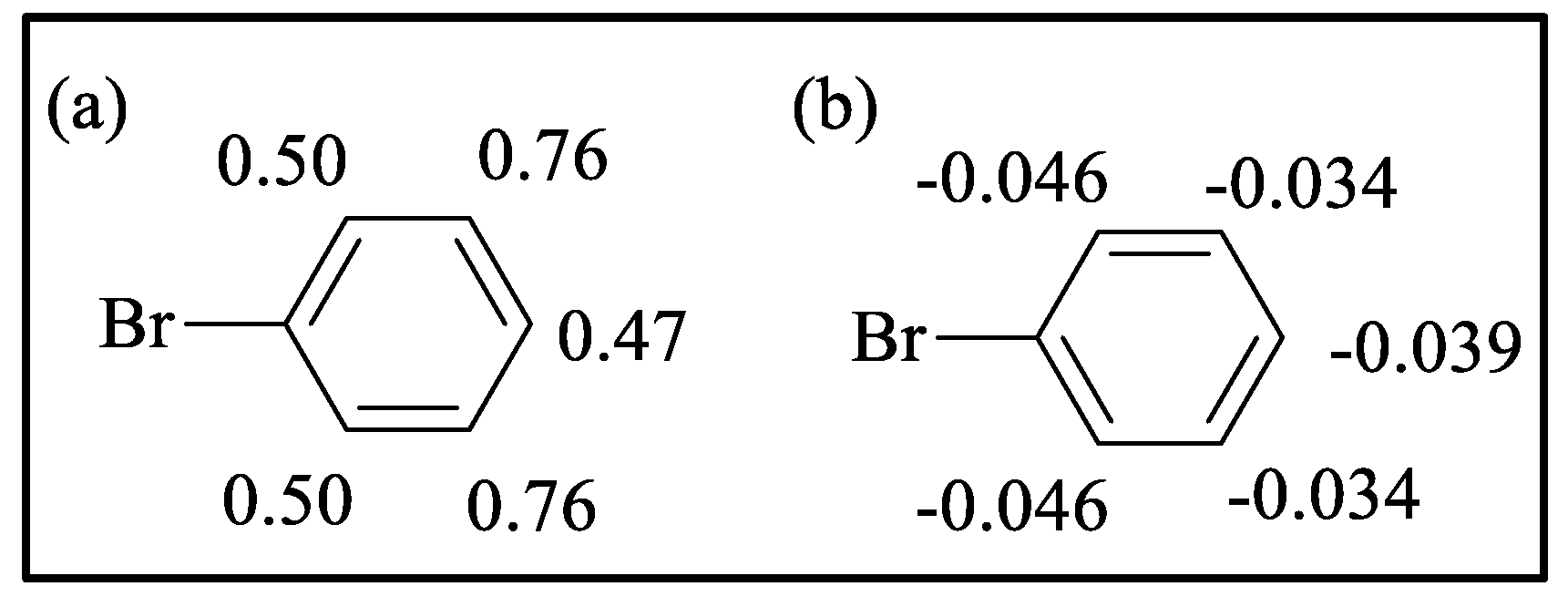
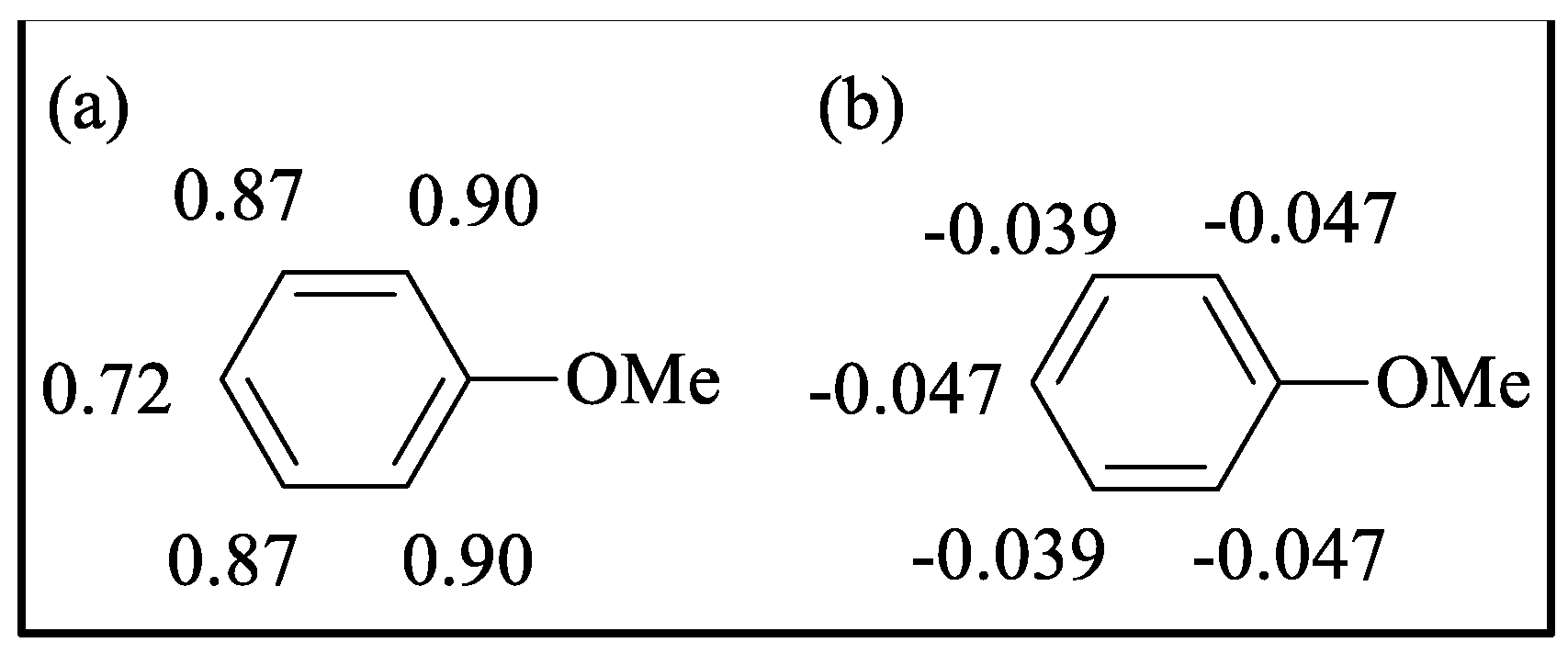
Mulliken charges | Hirshfeld charges | ||||||
| ortho | meta | para | ortho | meta | para | ortho | meta | para | ||||
| Benzaldehyde | 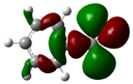 |
7.36 | 0.55 0.45 |
0.63 0.58 |
0.42 | -0.022 -0.031 |
-0.033 -0.037 |
-0.026 | ||||
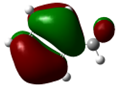 |
7.49 | 0.34 (0.12) -0.14 (0.02) |
0.15 (0.02) -0.34 (0.12) |
-0.19 (0.04) |
||||||||
| Benzoic acid | 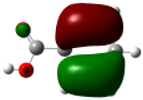 |
7.48 | -0.28 (0.08) 0.32 (0.10) |
-0.32 (0.10) 0.28 (0.08) |
-0.04 (0.002) |
0.58 0.45 |
0.52 0.64 |
0.49 | -0.028 -0.022 |
-0.034 -0.035 |
-0.027 | |
| Methyl benzoate |
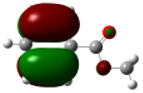 |
7.40 | 0.31 (0.10) -0.29 (0.08) |
0.29 (0.08) -0.31 (0.10) |
-0.02 (0.0004) |
0.49 0.78 |
0.55 0.33 |
0.58 | -0.024 -0.030 |
-0.036 -0.036 |
-0.029 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




