Submitted:
18 May 2025
Posted:
19 May 2025
You are already at the latest version
Abstract
Keywords:
Introduction
2. Materials and Methods
2.1. Synthesis of MitoGlow-ROS
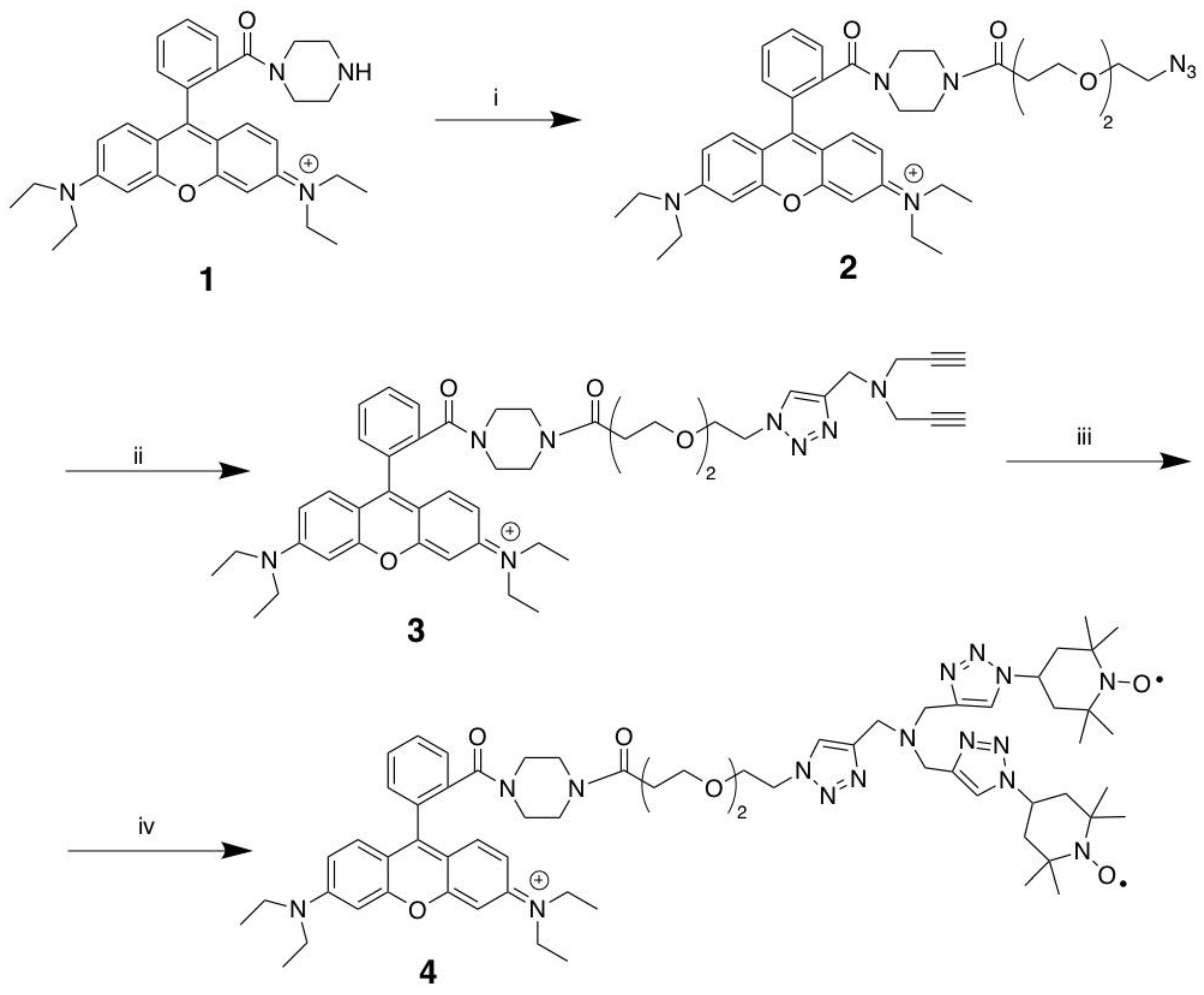
2.1.1. Rhodamine B 3-(2-(2-azidoethoxy)ethoxy)-1-(piperazin-1-yl)propan-1-one (2)
2.1.2. Rhodamine B 3-(2-(2-(4-((di(prop-2-yn-1-yl)amino)methyl)-lH-1,2,3-triazol-1-yl)ethoxy) ethoxy)-1-(piperazin-1-yl)propan-1-one (compound 3)
2.1.3. MitoGlow-ROS (compound 4)
2.2. Specificity Validation Assay
2.3. Sensitivity Validation Assay
2.4. Additional Validation Against Non-Specific Interactions
2.5. Cell Culture and Differentiation of PC12 Cells
2.6. In Vitro Model Using Primary Neuronal Cultures
2.6.1. Isolation and Culture of Primary Neuronal Cells from Neonatal SD Rat Pups
2.6.2. LPS Treatment to Induce Neuroinflammation
2.6.3. MitoGlow-ROS Probe Application and Fluorescence Intensity Measurement
2.6.4. Real-Time PCR for mRNA Expression Analysis
2.7. In Vivo Model of LPS-Induced Neuroinflammation in SD Rats
2.7.1. Administration of LPS via ICV Injection to Induce Systemic Neuroinflammation
2.7.2. Collection of Cortex and Hippocampus Tissues at 6 Hours Post-LPS Injection
2.7.3. Real-Time PCR for mRNA Expression Analysis
2.7.4. MitoGlow-ROS Probe Application and Mitochondrial ROS Measurement
3. Results
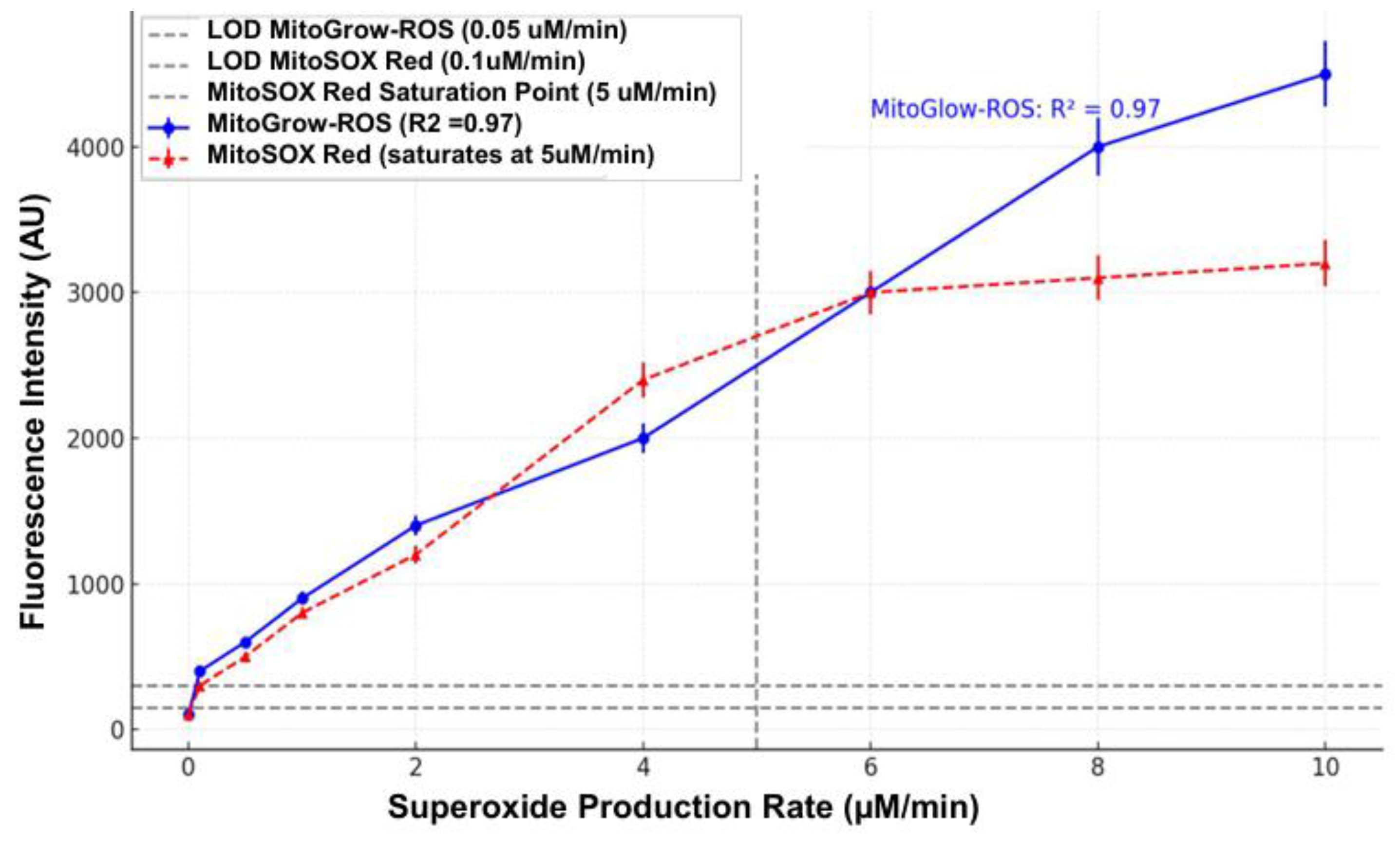
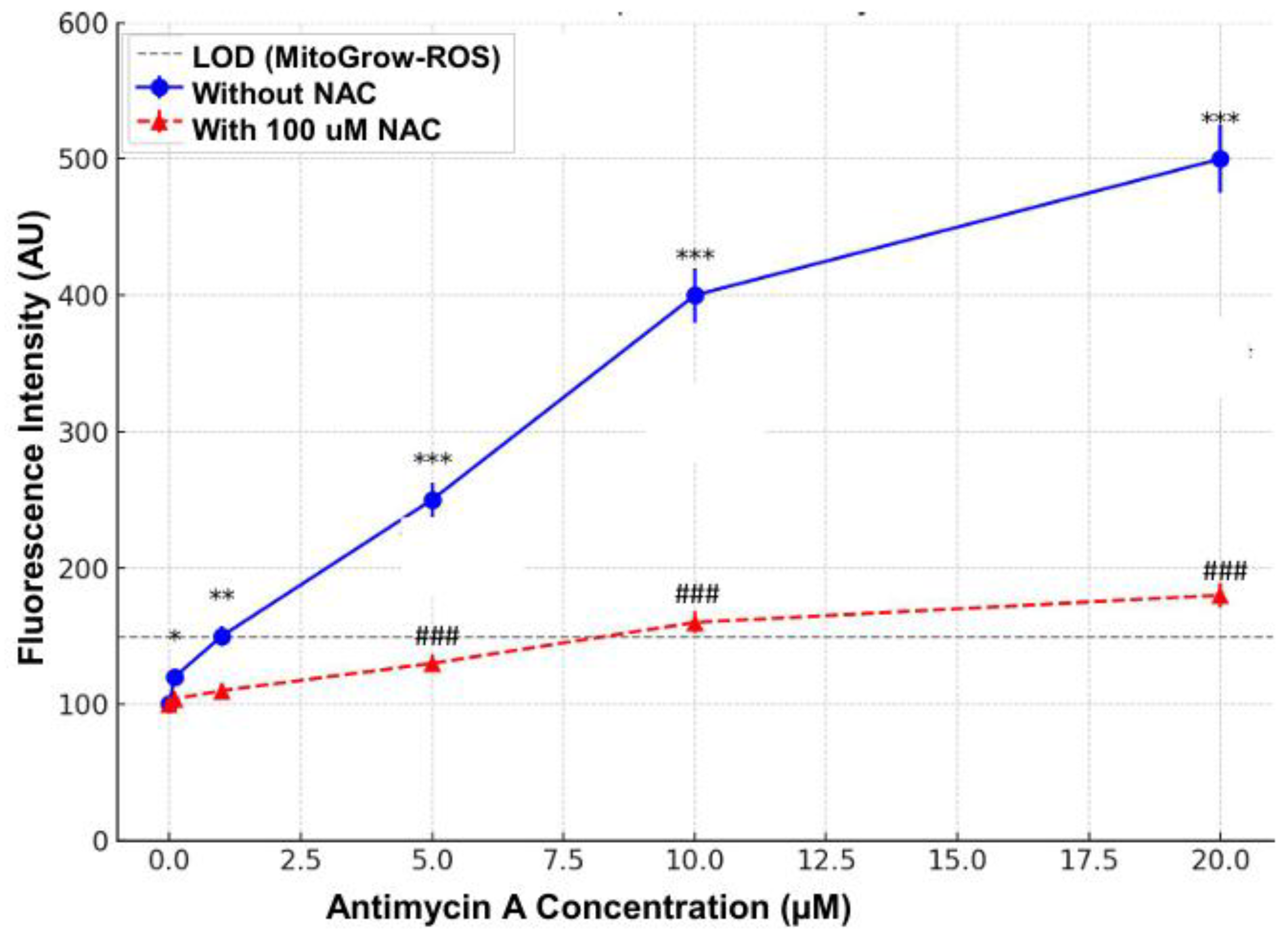
3.1. Cell-Free Validation of MitoGlow-ROS Sensitivity and Dynamic Range
3.2. Validation of MitoGlow-ROS Specificity and Sensitivity in PC12 Cells
3.3. Validation of MitoGlow-ROS Probe in Primary Neuronal Cells- A Cellular Model of Neuroinflammation
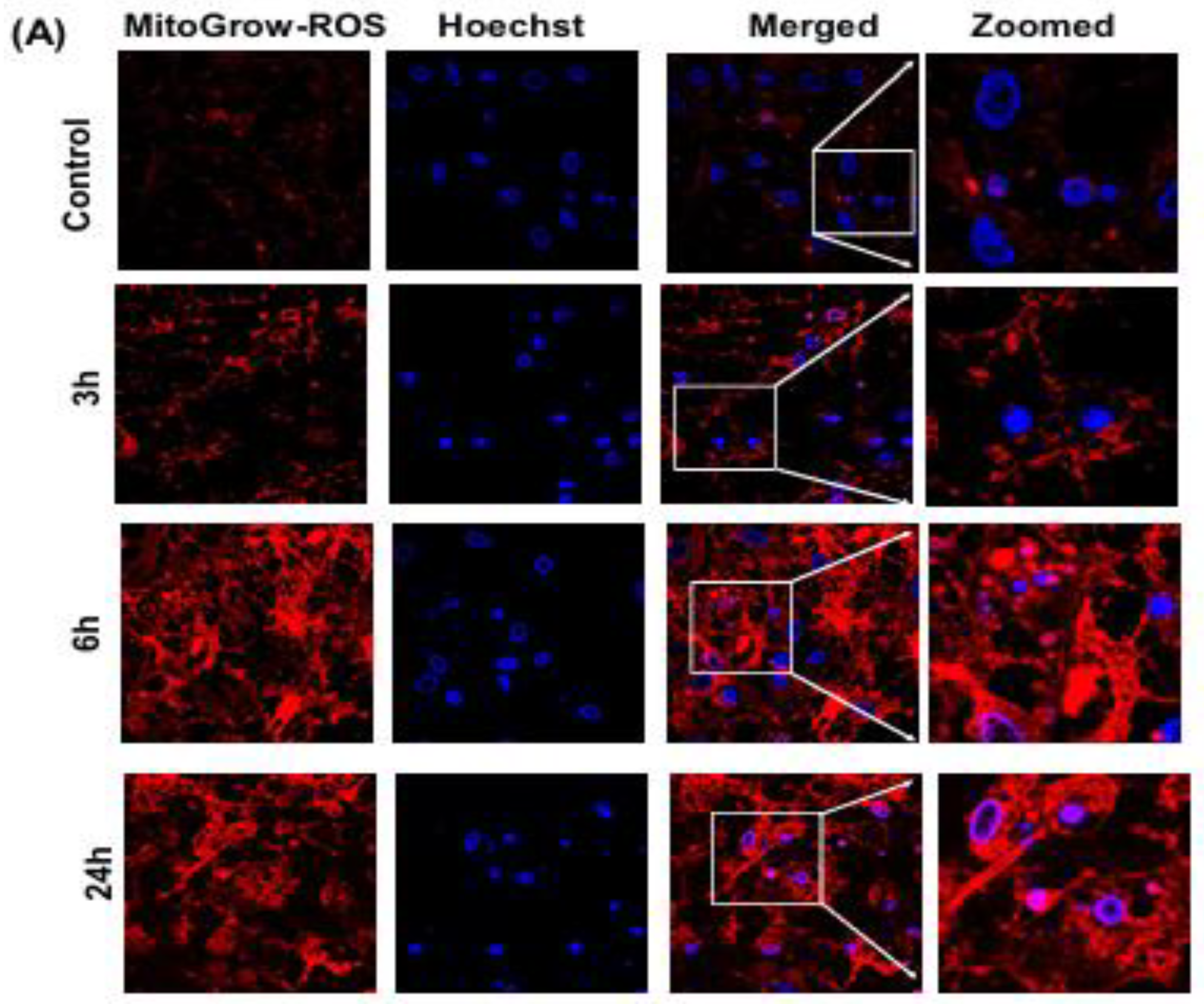
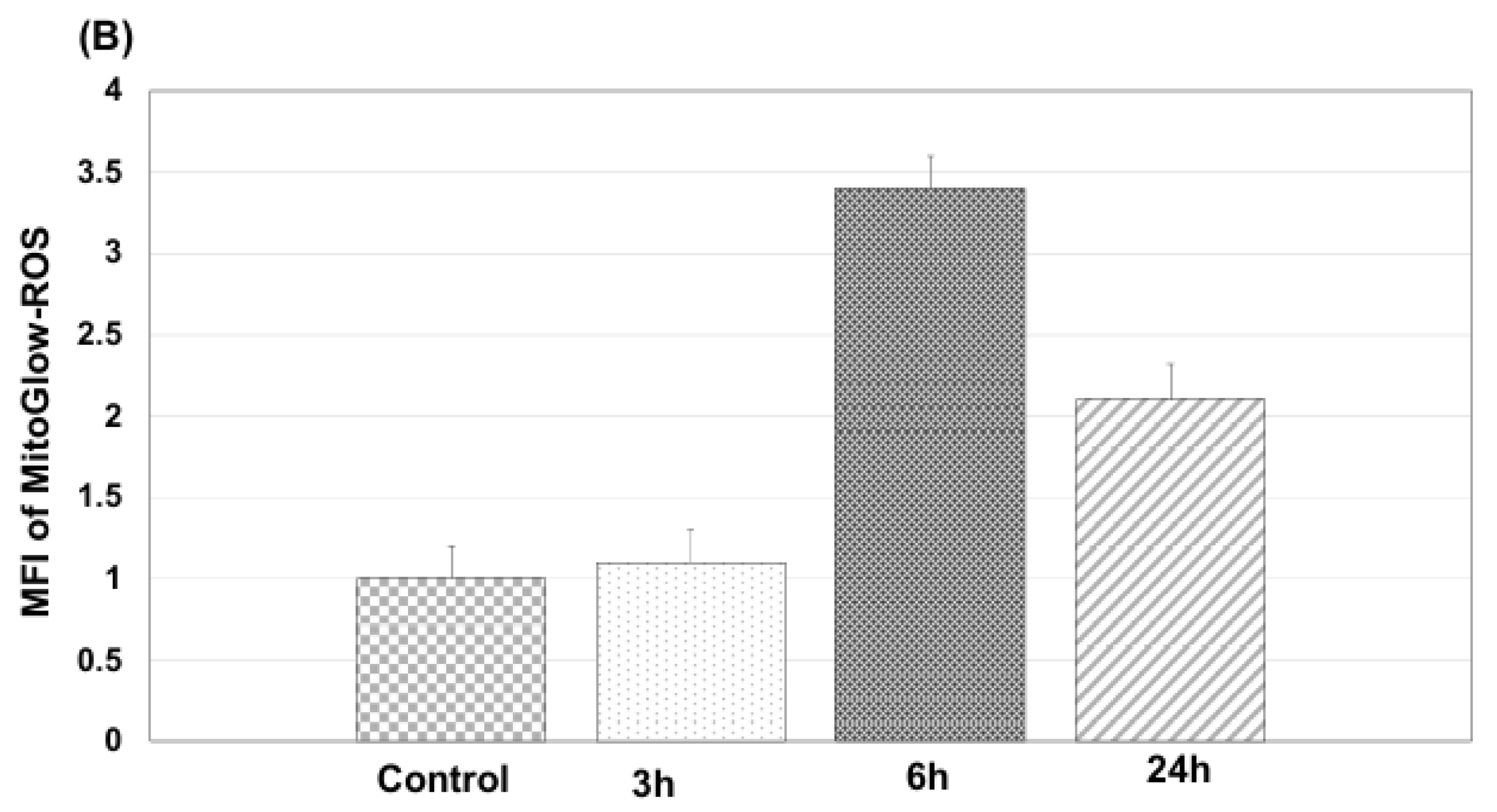
3.3.1. Mitochondrial ROS Generation and Morphology Changes in LPS-Treated Primary Neuronal Cells Using MitoGLow-ROS
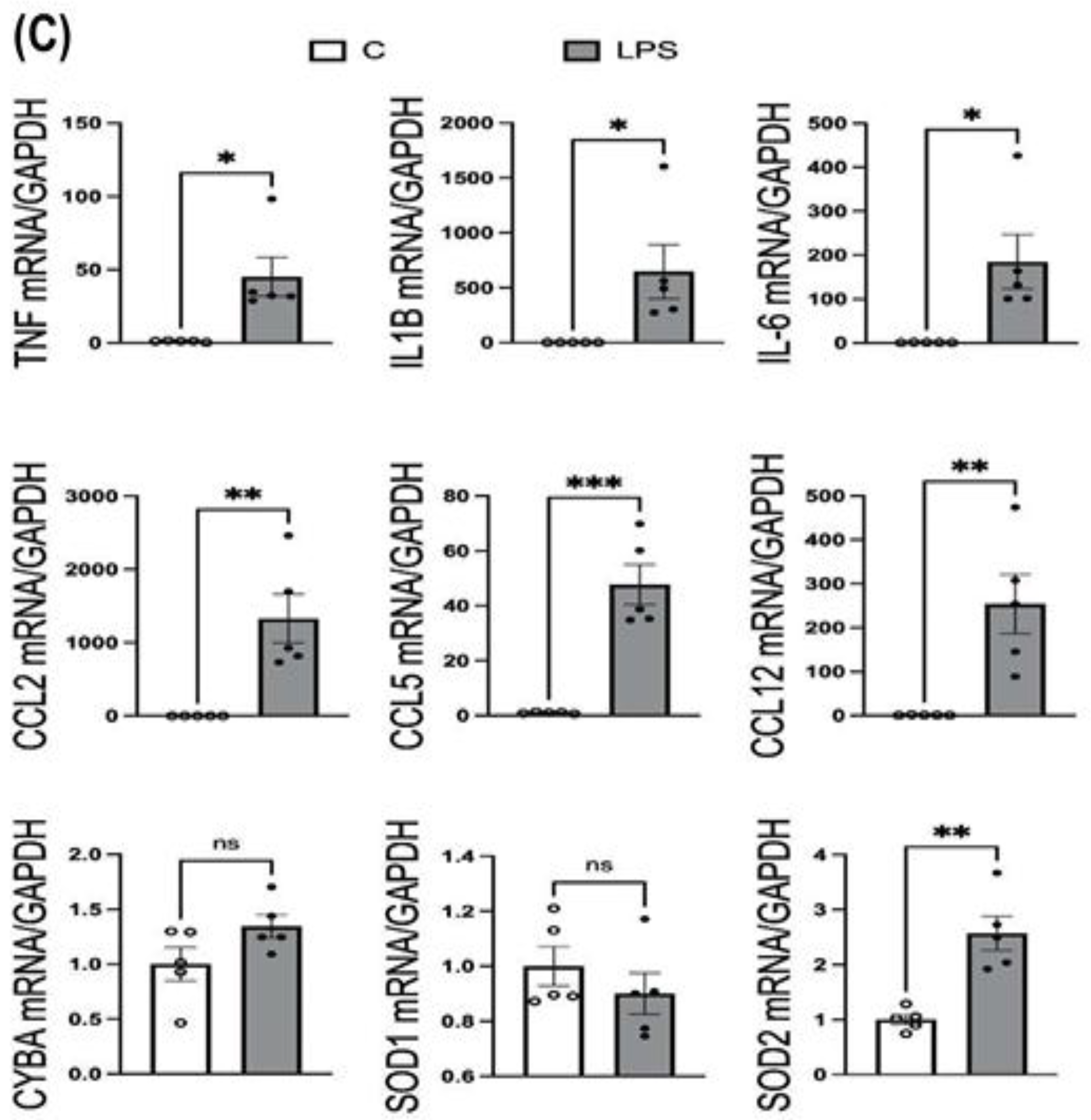
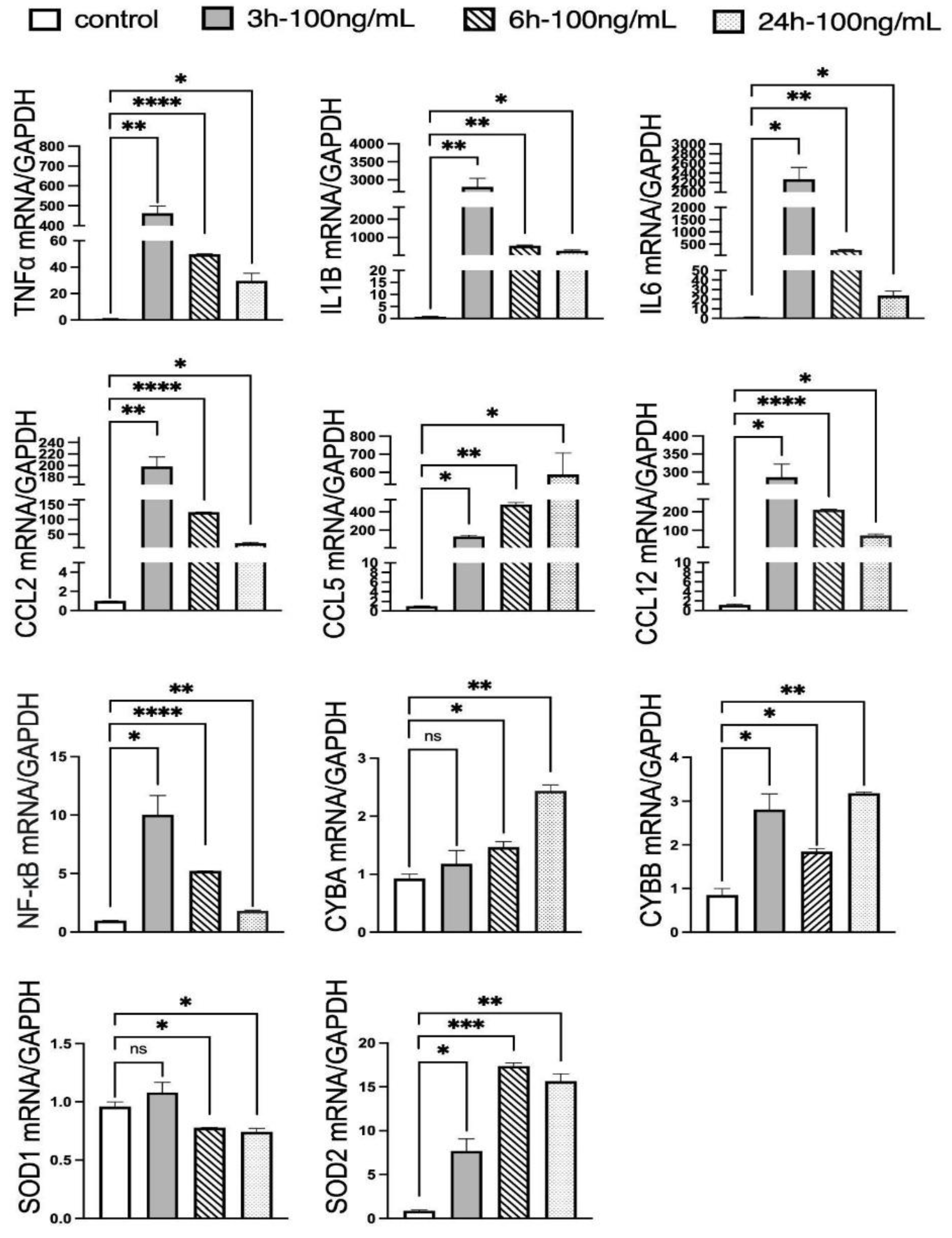
3.3.2. Time-Dependent Effects of LPS on Inflammatory and Oxidative Stress Gene Expression in Primary Neuronal Cultures
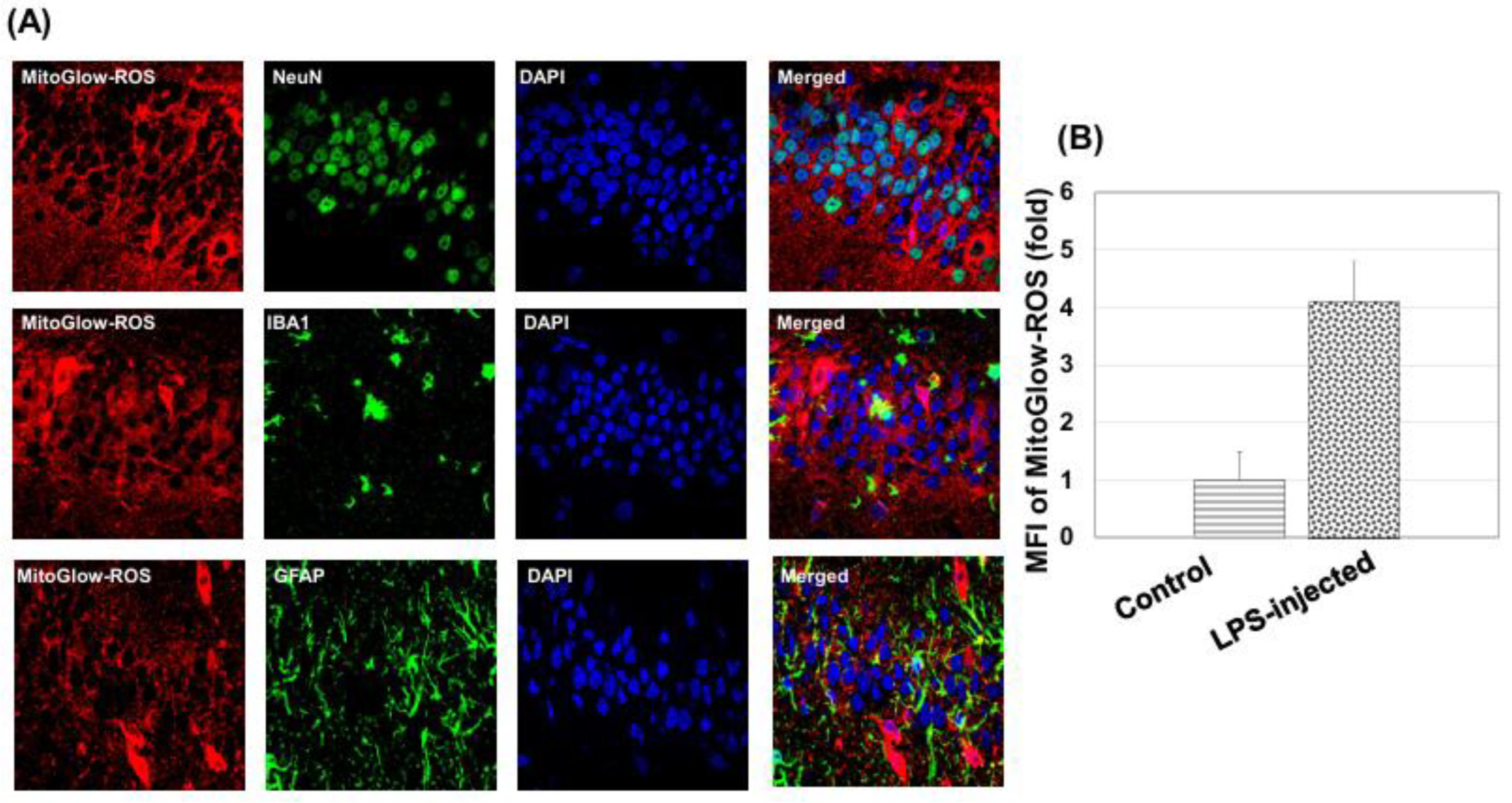
3.4. Validation of MitoGlow-ROS Probe in a Sprague-Dawley Rat Model of LPS-Induced Neuroinflammation
3.4.1. Mitochondrial ROS Generation
3.4.2. Hippocampal mRNA Expression: LPS treatment significantly upregulated hippocampal inflammatory mediators
3.4.3. Cortical mRNA Expression
3.4.4. Validation of MitoGlow-ROS Probe
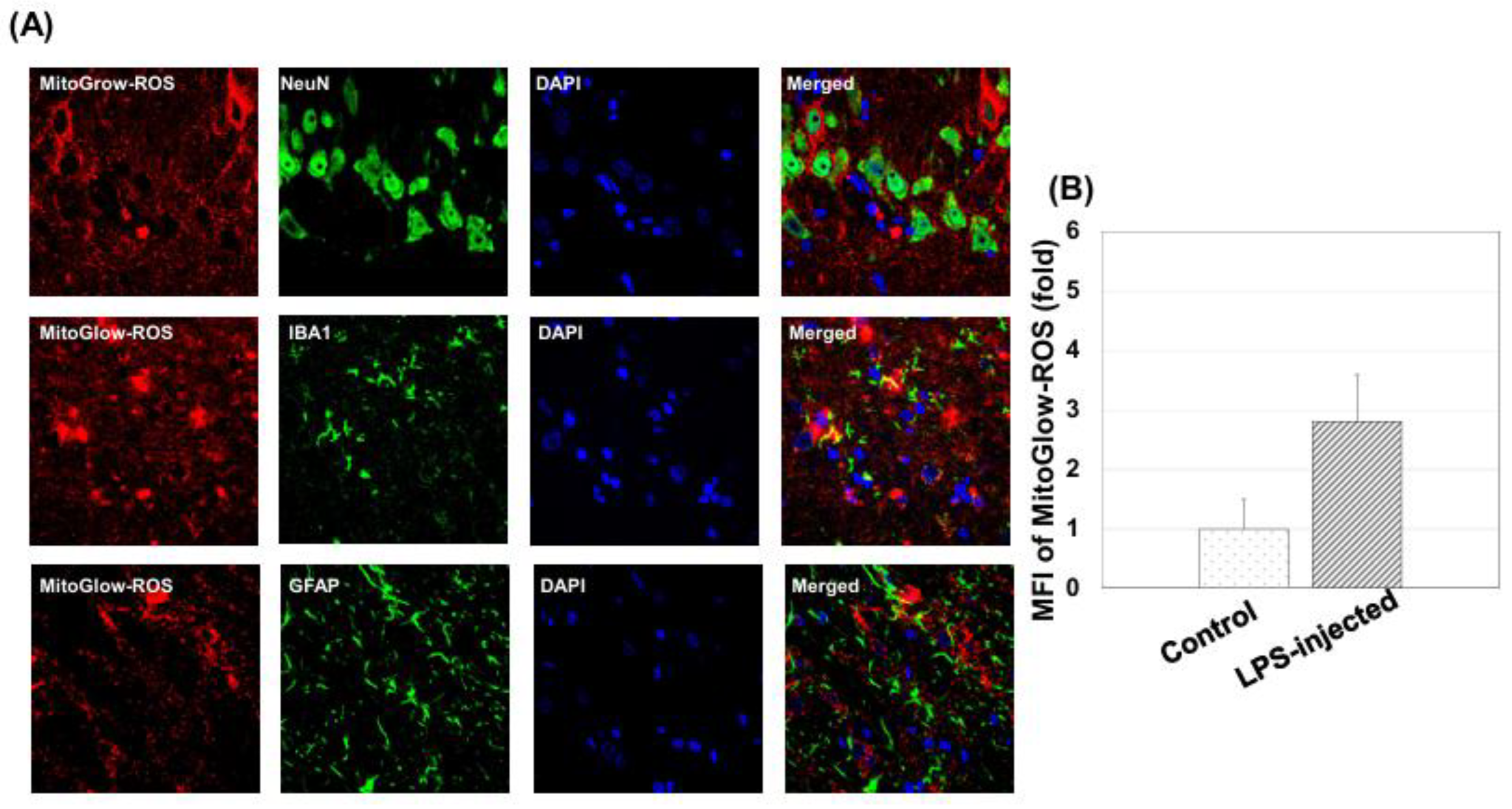
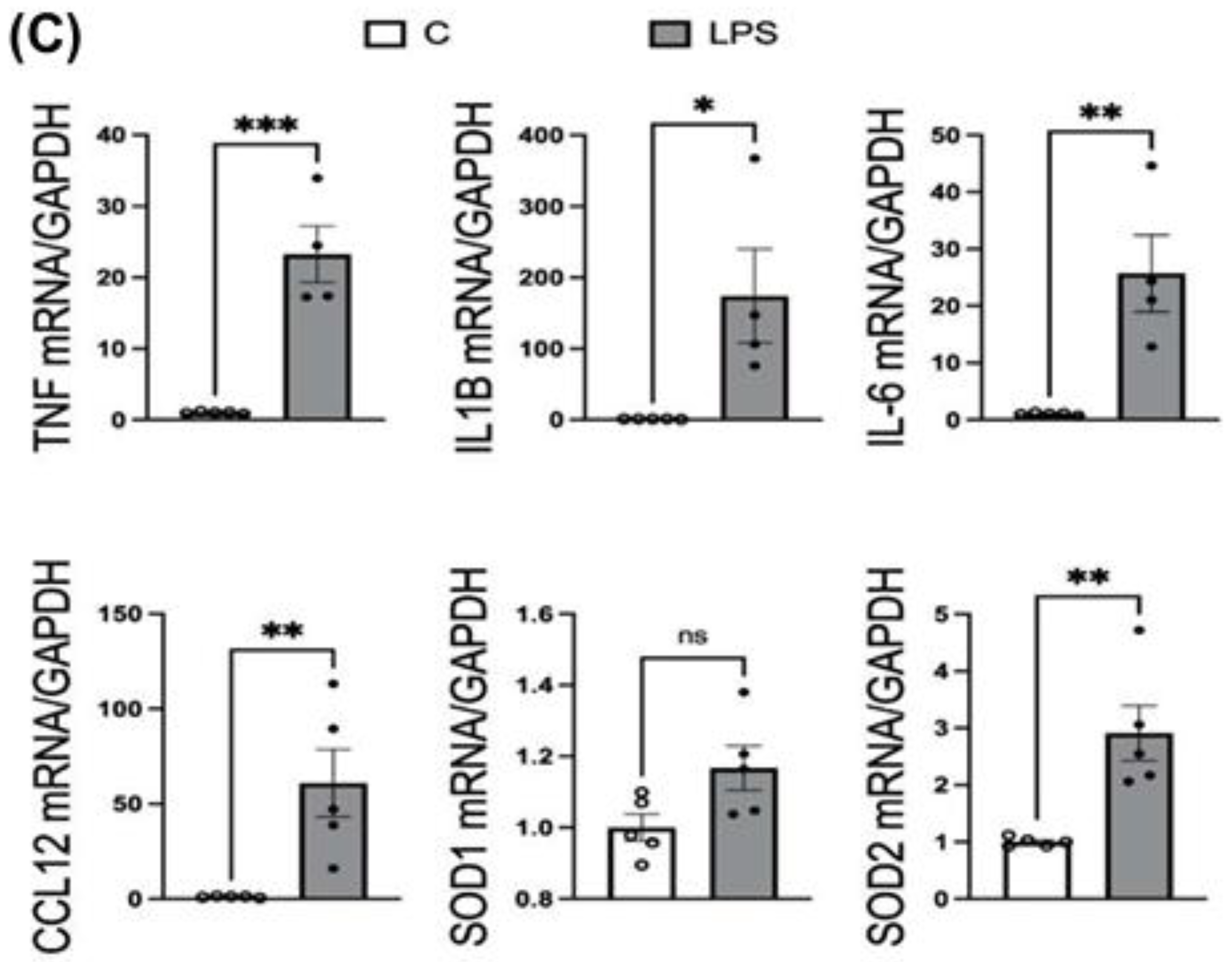
4. Discussion
5. Limitations
6. Conclusions
Funding
Acknowledgments
References
- Kishida, K.T.; Klann, E. Sources and targets of reactive oxygen species in synaptic plasticity and memory. Antioxid Redox Signal 2007, 9, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev 2014, 94, 909–950. [Google Scholar] [CrossRef]
- Quinlan, C.L.; et al. Sites of reactive oxygen species generation by mitochondria oxidizing different substrates. Redox Biol 2013, 1, 304–312. [Google Scholar] [CrossRef]
- Chan, P.H. Reactive oxygen radicals in signaling and damage in the ischemic brain. J Cereb Blood Flow Metab 2001, 21, 2–14. [Google Scholar] [CrossRef]
- Piantadosi, C.A.; Zhang, J. Mitochondrial generation of reactive oxygen species after brain ischemia in the rat. Stroke 1996, 27, 327–331. [Google Scholar] [CrossRef]
- Angelova, P.R.; Abramov, A.Y. Role of mitochondrial ROS in the brain: From physiology to neurodegeneration. FEBS Lett 2018, 592, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C.; Anrather, J. The immunology of stroke: From mechanisms to translation. Nat Med 2011, 17, 796–808. [Google Scholar] [CrossRef] [PubMed]
- Orellana-Urzúa, S.; et al. Pathophysiology of Ischemic Stroke: Role of Oxidative Stress. Curr Pharm Des 2020, 26, 4246–4260. [Google Scholar] [CrossRef]
- Chen, H.; et al. Therapeutic targets of oxidative/nitrosative stress and neuroinflammation in ischemic stroke: Applications for natural product efficacy with omics and systemic biology. Pharmacol Res 2020, 158, 104877. [Google Scholar] [CrossRef]
- Bai, R.; et al. Oxidative stress: The core pathogenesis and mechanism of Alzheimer's disease. Ageing Res Rev 2022, 77, 101619. [Google Scholar] [CrossRef]
- Ionescu-Tucker, A.; Cotman, C.W. Emerging roles of oxidative stress in brain aging and Alzheimer's disease. Neurobiol Aging 2021, 107, 86–95. [Google Scholar] [CrossRef]
- Tönnies, E.; Trushina, E. Oxidative Stress, Synaptic Dysfunction, and Alzheimer's Disease. J Alzheimers Dis 2017, 57, 1105–1121. [Google Scholar] [CrossRef]
- Cheignon, C.; et al. Oxidative stress and the amyloid beta peptide in Alzheimer's disease. Redox Biol 2018, 14, 450–464. [Google Scholar] [CrossRef]
- Reddy, P.H.; Beal, M.F. Amyloid beta, mitochondrial dysfunction and synaptic damage: Implications for cognitive decline in aging and Alzheimer's disease. Trends Mol Med 2008, 14, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Manczak, M.; et al. Mitochondria are a direct site of A beta accumulation in Alzheimer's disease neurons: Implications for free radical generation and oxidative damage in disease progression. Hum Mol Genet 2006, 15, 1437–1449. [Google Scholar] [CrossRef]
- Simpson, D.S.A.; Oliver, P.L. ROS Generation in Microglia: Understanding Oxidative Stress and Inflammation in Neurodegenerative Disease. Antioxidants 2020, 9, 743. [Google Scholar] [CrossRef] [PubMed]
- Miao, J.; et al. Microglia in Alzheimer's disease: Pathogenesis, mechanisms, and therapeutic potentials. Front Aging Neurosci 2023, 15, 1201982. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; et al. Inflammasomes in neurodegenerative diseases. Transl Neurodegener 2024, 13, 65. [Google Scholar] [CrossRef]
- Sho, T.; Xu, J. Role and mechanism of ROS scavengers in alleviating NLRP3-mediated inflammation. Biotechnol Appl Biochem 2019, 66, 4–13. [Google Scholar] [CrossRef]
- Xiao, B.; et al. Reactive oxygen species trigger Parkin/PINK1 pathway-dependent mitophagy by inducing mitochondrial recruitment of Parkin. J Biol Chem 2017, 292, 16697–16708. [Google Scholar] [CrossRef]
- Narendra, D.P.; et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol 2010, 8, e1000298. [Google Scholar] [CrossRef] [PubMed]
- Quinn, P.M.J.; et al. PINK1/PARKIN signalling in neurodegeneration and neuroinflammation. Acta Neuropathol Commun 2020, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, S.; et al. Dopamine induced neurodegeneration in a PINK1 model of Parkinson's disease. PLoS ONE 2012, 7, e37564. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.J.; et al. PINK1 deficiency impairs adult neurogenesis of dopaminergic neurons. Sci Rep 2021, 11, 6617. [Google Scholar] [CrossRef]
- Guo, C.; et al. Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen Res 2013, 8, 2003–2014. [Google Scholar]
- Wei, Y.; et al. Balanced basal-levels of ROS (redox-biology), and very-low-levels of pro-inflammatory cytokines (cold-inflammaging), as signaling molecules can prevent or slow-down overt-inflammaging, and the aging-associated decline of adaptive-homeostasis. Exp Gerontol 2023, 172, 112067. [Google Scholar] [CrossRef]
- Mosley, R.L.; et al. Neuroinflammation, Oxidative Stress and the Pathogenesis of Parkinson's Disease. Clin Neurosci Res 2006, 6, 261–281. [Google Scholar] [CrossRef]
- Isik, S.; et al. Microglia Mediated Neuroinflammation in Parkinson's Disease. Cells 2023, 12, 1012. [Google Scholar] [CrossRef]
- Anilkumar, S.; Wright-Jin, E. NF-κB as an Inducible Regulator of Inflammation in the Central Nervous System. Cells 2024, 13, 485. [Google Scholar] [CrossRef]
- Foley, P.; Riederer, P. Influence of neurotoxins and oxidative stress on the onset and progression of Parkinson's disease. J Neurol 2000, 247 (Suppl. S2), ii82–ii94. [Google Scholar] [CrossRef]
- Tiwari, P.C.; Pal, R. The potential role of neuroinflammation and transcription factors in Parkinson disease. Dialogues Clin Neurosci 2017, 19, 71–80. [Google Scholar] [CrossRef]
- Li, W.; et al. Crosstalk between ER stress, NLRP3 inflammasome, and inflammation. Appl Microbiol Biotechnol 2020, 104, 6129–6140. [Google Scholar] [CrossRef]
- Abais, J.M.; et al. Redox regulation of NLRP3 inflammasomes: ROS as trigger or effector? Antioxid Redox Signal 2015, 22, 1111–1129. [Google Scholar] [CrossRef] [PubMed]
- Kalyanaraman, B.; et al. Measuring reactive oxygen and nitrogen species with fluorescent probes: Challenges and limitations. Free Radic Biol Med 2012, 52, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Dikalov, S.I.; Harrison, D.G. Methods for detection of mitochondrial and cellular reactive oxygen species. Antioxid Redox Signal 2014, 20, 372–382. [Google Scholar] [CrossRef]
- Ribou, A.C. Synthetic Sensors for Reactive Oxygen Species Detection and Quantification: A Critical Review of Current Methods. Antioxid Redox Signal 2016, 25, 520–533. [Google Scholar] [CrossRef]
- Salma, F.; et al. Methods for detection of mitochondrial reactive oxygen species in senescent cells. Methods Cell Biol 2024, 181, 33–41. [Google Scholar] [PubMed]
- Zhang, X.; Gao, F. Imaging mitochondrial reactive oxygen species with fluorescent probes: Current applications and challenges. Free Radic Res 2015, 49, 374–382. [Google Scholar] [CrossRef]
- Mailloux, R.J. An update on methods and approaches for interrogating mitochondrial reactive oxygen species production. Redox Biol 2021, 45, 102044. [Google Scholar] [CrossRef]
- Rota, C.; Fann, Y.C.; Mason, R.P. Phenoxyl free radical formation during the oxidation of the fluorescent dye 2',7'-dichlorofluorescein by horseradish peroxidase. Possible consequences for oxidative stress measurements. J Biol Chem 1999, 274, 28161–28168. [Google Scholar] [CrossRef]
- Murphy, M.P.; et al. Guidelines for measuring reactive oxygen species and oxidative damage in cells and in vivo. Nat Metab 2022, 4, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, J.; Kalyanaraman, B. Hydroethidine- and MitoSOX-derived red fluorescence is not a reliable indicator of intracellular superoxide formation: Another inconvenient truth. Free Radic Biol Med 2010, 48, 983–1001. [Google Scholar] [CrossRef]
- Cobley, J.N.; et al. Ten "Cheat Codes" for Measuring Oxidative Stress in Humans. Antioxidants 2024, 13, 877. [Google Scholar] [CrossRef] [PubMed]
- Ghanian, Z.; et al. Quantitative optical measurement of mitochondrial superoxide dynamics in pulmonary artery endothelial cells. J Innov Opt Health Sci 2018, 11, 1750018. [Google Scholar] [CrossRef]
- Cazareth, J.; et al. Molecular and cellular neuroinflammatory status of mouse brain after systemic lipopolysaccharide challenge: Importance of CCR2/CCL2 signaling. J Neuroinflammation 2014, 11, 132. [Google Scholar] [CrossRef] [PubMed]
- Roelofs, B.A.; et al. Low micromolar concentrations of the superoxide probe MitoSOX uncouple neural mitochondria and inhibit complex IV. Free Radic Biol Med 2015, 86, 250–258. [Google Scholar] [CrossRef]
- Yapici, N.B.; et al. New rhodamine nitroxide based fluorescent probes for intracellular hydroxyl radical identification in living cells. Org Lett 2012, 14, 50–53. [Google Scholar] [CrossRef]
- Wilcox, C.S. Effects of tempol and redox-cycling nitroxides in models of oxidative stress. Pharmacol Ther 2010, 126, 119–145. [Google Scholar] [CrossRef]
- Battelli, M.G.; et al. Xanthine Oxidoreductase-Derived Reactive Species: Physiological and Pathological Effects. Oxid Med Cell Longev 2016, 2016, 3527579. [Google Scholar] [CrossRef]
- Halasi, M.; et al. ROS inhibitor N-acetyl-L-cysteine antagonizes the activity of proteasome inhibitors. Biochem J 2013, 454, 201–208. [Google Scholar] [CrossRef]
- Reiniers, M.J.; et al. 2',7'-Dichlorofluorescein is not a probe for the detection of reactive oxygen and nitrogen species. J Hepatol 2012, 56, 1214–1216. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; et al. Engineering Exosomes to Specifically Target the Mitochondria of Brain Cells. ACS Omega 2023, 8, 48984–48993. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Huber, M.J.; et al. Increased activity of the orexin system in the paraventricular nucleus contributes to salt-sensitive hypertension. Am J Physiol Heart Circ Physiol 2017, 313, H1075–H1086. [Google Scholar] [CrossRef]
- Fan, Y.; et al. Activation of Orexin System Stimulates CaMKII Expression. Front Physiol 2021, 12, 698185. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; et al. Crosstalk Between the Oxidative Stress and Glia Cells After Stroke: From Mechanism to Therapies. Front Immunol 2022, 13, 852416. [Google Scholar] [CrossRef]
- Yang, M.; et al. Mitochondrial Quality Control: A Pathophysiological Mechanism and Therapeutic Target for Stroke. Front Mol Neurosci 2021, 14, 786099. [Google Scholar] [CrossRef]
- Fang, C.; et al. Natural Compounds for SIRT1-Mediated Oxidative Stress and Neuroinflammation in Stroke: A Potential Therapeutic Target in the Future. Oxid Med Cell Longev 2022, 2022, 1949718. [Google Scholar] [CrossRef] [PubMed]
- Swanson, L.; et al. TLR4 signaling and macrophage inflammatory responses are dampened by GIV/Girdin. Proc Natl Acad Sci U S A 2020, 117, 26895–26906. [Google Scholar] [CrossRef]
- Lu, Y.C.; Yeh, W.C.; Ohashi, P.S. LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef]
- Park, J.S.; et al. Involvement of toll-like receptors 2 and 4 in cellular activation by high mobility group box 1 protein. J Biol Chem 2004, 279, 7370–7377. [Google Scholar] [CrossRef]
- Everhardt Queen, A.; et al. Differential Expression of Inflammatory Cytokines and Stress Genes in Male and Female Mice in Response to a Lipopolysaccharide Challenge. PLoS ONE 2016, 11, e0152289. [Google Scholar] [CrossRef] [PubMed]
- Voloboueva, L.A.; et al. Inflammatory response of microglial BV-2 cells includes a glycolytic shift and is modulated by mitochondrial glucose-regulated protein 75/mortalin. FEBS Lett 2013, 587, 756–762. [Google Scholar] [CrossRef]
- Dustin, C.M.; et al. NOX2 in Alzheimer's and Parkinson's disease. Redox Biol 2024, 78, 103433. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.A. The multifaceted profile of activated microglia. Mol Neurobiol 2009, 40, 139–156. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.S.; Cheng, G. Role of interleukin 10 transcriptional regulation in inflammation and autoimmune disease. Crit Rev Immunol 2012, 32, 23–63. [Google Scholar] [CrossRef]
- Cyktor, J.C.; Turner, J. Interleukin-10 and immunity against prokaryotic and eukaryotic intracellular pathogens. Infect Immun 2011, 79, 2964–2973. [Google Scholar] [CrossRef]
- Zhang, D.; et al. Ferulic acid alleviates sciatica by inhibiting neuroinflammation and promoting nerve repair via the TLR4/NF-κB pathway. CNS Neurosci Ther 2023, 29, 1000–1011. [Google Scholar] [CrossRef]
- Zhang, J.; et al. Curcumin inhibits LPS-induced neuroinflammation by promoting microglial M2 polarization via TREM2/ TLR4/ NF-κB pathways in BV2 cells. Mol Immunol 2019, 116, 29–37. [Google Scholar] [CrossRef]
- Jin, X.; et al. Baicalin mitigates cognitive impairment and protects neurons from microglia-mediated neuroinflammation via suppressing NLRP3 inflammasomes and TLR4/NF-κB signaling pathway. CNS Neurosci Ther 2019, 25, 575–590. [Google Scholar] [CrossRef]
- van Horssen, J.; van Schaik, P.; Witte, M. Inflammation and mitochondrial dysfunction: A vicious circle in neurodegenerative disorders? Neurosci Lett 2019, 710, 132931. [Google Scholar] [CrossRef]
- Arena, G.; et al. Neurodegeneration and Neuroinflammation in Parkinson's Disease: A Self-Sustained Loop. Curr Neurol Neurosci Rep 2022, 22, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; et al. Epigallocatechin-3-gallate Inhibits LPS/AβO-induced Neuroinflammation in BV2 Cells through Regulating the ROS/TXNIP/NLRP3 Pathway. J Neuroimmune Pharmacol 2024, 19, 31. [Google Scholar] [CrossRef] [PubMed]
- Shukuri, M.; et al. Ex vivo imaging and analysis of ROS generation correlated with microglial activation in rat model with acute neuroinflammation induced by intrastriatal injection of LPS. Biochem Biophys Res Commun 2021, 584, 101–106. [Google Scholar] [CrossRef]
- Flynn, J.M.; Melov, S. SOD2 in mitochondrial dysfunction and neurodegeneration. Free Radic Biol Med 2013, 62, 4–12. [Google Scholar] [CrossRef]
- Wang, Y.; et al. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol 2018, 217, 1915–1928. [Google Scholar] [CrossRef] [PubMed]
- Peters, O.; et al. Increased formation of reactive oxygen species after permanent and reversible middle cerebral artery occlusion in the rat. J Cereb Blood Flow Metab 1998, 18, 196–205. [Google Scholar] [CrossRef]
- Li, W.; Yang, S. Targeting oxidative stress for the treatment of ischemic stroke: Upstream and downstream therapeutic strategies. Brain Circ 2016, 2, 153–163. [Google Scholar]
- Briones-Valdivieso, C.; et al. Novel Multi-Antioxidant Approach for Ischemic Stroke Therapy Targeting the Role of Oxidative Stress. Biomedicines 2024, 12, 501. [Google Scholar] [CrossRef]
- Karki, R.; Kanneganti, T.D. The 'cytokine storm': Molecular mechanisms and therapeutic prospects. Trends Immunol 2021, 42, 681–705. [Google Scholar] [CrossRef]
- Puleo, M.G.; et al. Molecular Mechanisms of Inflammasome in Ischemic Stroke Pathogenesis. Pharmaceuticals 2022, 15, 1168. [Google Scholar] [CrossRef]
- Alves, J.L.; et al. Exploring different mechanisms of reactive oxygen species formation in hypoxic conditions at the hippocampal CA3 area. Mol Cell Endocrinol 2025, 601, 112517. [Google Scholar] [CrossRef] [PubMed]
- Patergnani, S.; et al. Mitochondrial Oxidative Stress and "Mito-Inflammation": Actors in the Diseases. Biomedicines 2021, 9, 216. [Google Scholar] [CrossRef]
- Adusumilli, V.S.; et al. ROS Dynamics Delineate Functional States of Hippocampal Neural Stem Cells and Link to Their Activity-Dependent Exit from Quiescence. Cell Stem Cell 2021, 28, 300–314.e6. [Google Scholar] [CrossRef]
- Popa-Wagner, A.; et al. ROS and brain diseases: The good, the bad, and the ugly. Oxid Med Cell Longev 2013, 2013, 963520. [Google Scholar] [CrossRef] [PubMed]
- MohanKumar, S.M.J.; et al. Role of cytokines and reactive oxygen species in brain aging. Mech Ageing Dev 2023, 214, 111855. [Google Scholar] [CrossRef]
- Batista, C.R.A.; et al. Lipopolysaccharide-Induced Neuroinflammation as a Bridge to Understand Neurodegeneration. Int. J. Mol. Sci. 2019, 20, 2293. [Google Scholar] [CrossRef] [PubMed]
- Skrzypczak-Wiercioch, A.; Sałat, K. Lipopolysaccharide-Induced Model of Neuroinflammation: Mechanisms of Action, Research Application and Future Directions for Its Use. Molecules 2022, 27, 5481. [Google Scholar] [CrossRef]
- Ganesan, K.; et al. Hippocampal neuroinflammation induced by lipopolysaccharide causes sex-specific disruptions in action selection, food approach memories, and neuronal activation. Brain Behav Immun 2025, 124, 9–27. [Google Scholar] [CrossRef]
- Zhou, L.; et al. Remimazolam attenuates lipopolysaccharide-induced neuroinflammation and cognitive dysfunction. Behav Brain Res 2025, 476, 115268. [Google Scholar] [CrossRef]
- Leal, M.C.; et al. Interleukin-1β and tumor necrosis factor-α: Reliable targets for protective therapies in Parkinson's Disease? Front Cell Neurosci 2013, 7, 53. [Google Scholar] [CrossRef]
- Peruzzotti-Jametti, L.; et al. Mitochondrial complex I activity in microglia sustains neuroinflammation. Nature 2024, 628, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; et al. Mitochondrial complex I: The key to sustained microglia activation and neuroinflammation maintenance. Mil Med Res 2024, 11, 47. [Google Scholar] [CrossRef]
- Palma, F.R.; et al. Mitochondrial Superoxide Dismutase: What the Established, the Intriguing, and the Novel Reveal About a Key Cellular Redox Switch. Antioxid Redox Signal 2020, 32, 701–714. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, G.; Fasciolo, G.; Venditti, P. Mitochondrial Management of Reactive Oxygen Species. Antioxidants 2021, 10, 1824. [Google Scholar] [CrossRef]
- Wang, X.; Michaelis, E.K. Selective neuronal vulnerability to oxidative stress in the brain. Front Aging Neurosci 2010, 2, 12. [Google Scholar] [CrossRef]
- Feng, S.; et al. Oxidative stress as a bridge between age and stroke: A narrative review. J Intensive Med 2023, 3, 313–319. [Google Scholar] [CrossRef]
- Nishino, H.; et al. The striatum is the most vulnerable region in the brain to mitochondrial energy compromise: A hypothesis to explain its specific vulnerability. J Neurotrauma 2000, 17, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Davidson, T.L.; Stevenson, R.J. Vulnerability of the Hippocampus to Insults: Links to Blood-Brain Barrier Dysfunction. Int. J. Mol. Sci. 2024, 25, 1991. [Google Scholar] [CrossRef]
- Anthony, S.; et al. Neuroinflammation, Stem Cells, and Stroke. Stroke 2022, 53, 1460–1472. [Google Scholar] [CrossRef]
- Liu, Z.; et al. Oxidative Stress in Neurodegenerative Diseases: From Molecular Mechanisms to Clinical Applications. Oxid Med Cell Longev 2017, 2017, 2525967. [Google Scholar] [CrossRef]
- Klein, J.A.; Ackerman, S.L. Oxidative stress, cell cycle, and neurodegeneration. J Clin Invest 2003, 111, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, N.R.; et al. Oxidative Stress and Redox Imbalance: Common Mechanisms in Cancer Stem Cells and Neurodegenerative Diseases. Cells 2025, 14, 511. [Google Scholar] [CrossRef] [PubMed]
- Żamojć, K.; et al. Fluorescence Quenching Studies on the Interactions between Chosen Fluoroquinolones and Selected Stable TEMPO and PROXYL Nitroxides. Int J Mol Sci 2021, 22, 885. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Clamme, J.P.; Deniz, A.A. Fluorescence quenching by TEMPO: A sub-30 A single-molecule ruler. Biophys J 2005, 89, L37–L39. [Google Scholar] [CrossRef]
- Pandya, V.A.; Patani, R. Region-specific vulnerability in neurodegeneration: Lessons from normal ageing. Ageing Res Rev 2021, 67, 101311. [Google Scholar] [CrossRef]
- Choi, E.H.; Kim, M.H.; Park, S.J. Targeting Mitochondrial Dysfunction and Reactive Oxygen Species for Neurodegenerative Disease Treatment. Int. J. Mol. Sci. 2024, 25, 7952. [Google Scholar] [CrossRef]
- Sousa, T.; Moreira, P.I.; Cardoso, S. Current Advances in Mitochondrial Targeted Interventions in Alzheimer's Disease. Biomedicines 2023, 11, 2331. [Google Scholar] [CrossRef]
- Wadan, A.S.; et al. Mitochondrial-based therapies for neurodegenerative diseases: A review of the current literature. Naunyn Schmiedebergs Arch Pharmacol 2025. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



