Submitted:
07 May 2025
Posted:
08 May 2025
You are already at the latest version
Abstract
Keywords:
I. Introduction
What Does the Immune System Have to Learn?
Immune Computation
Evolution Rules Immune Computation
The Human Immune System Learns Well
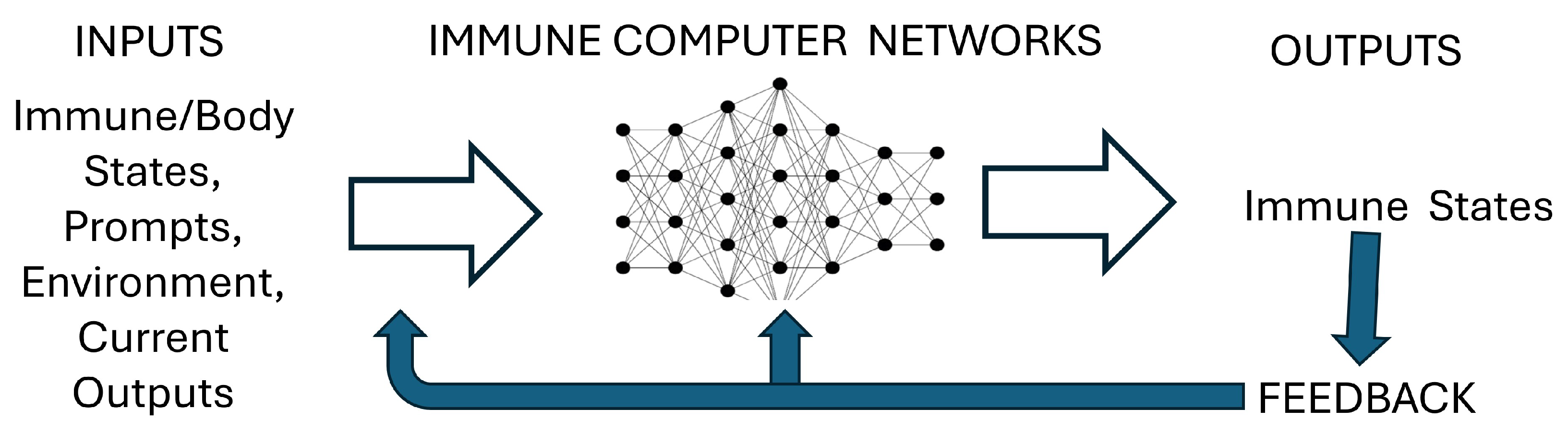
II. A Schematic Summary of Immune NML
Networks
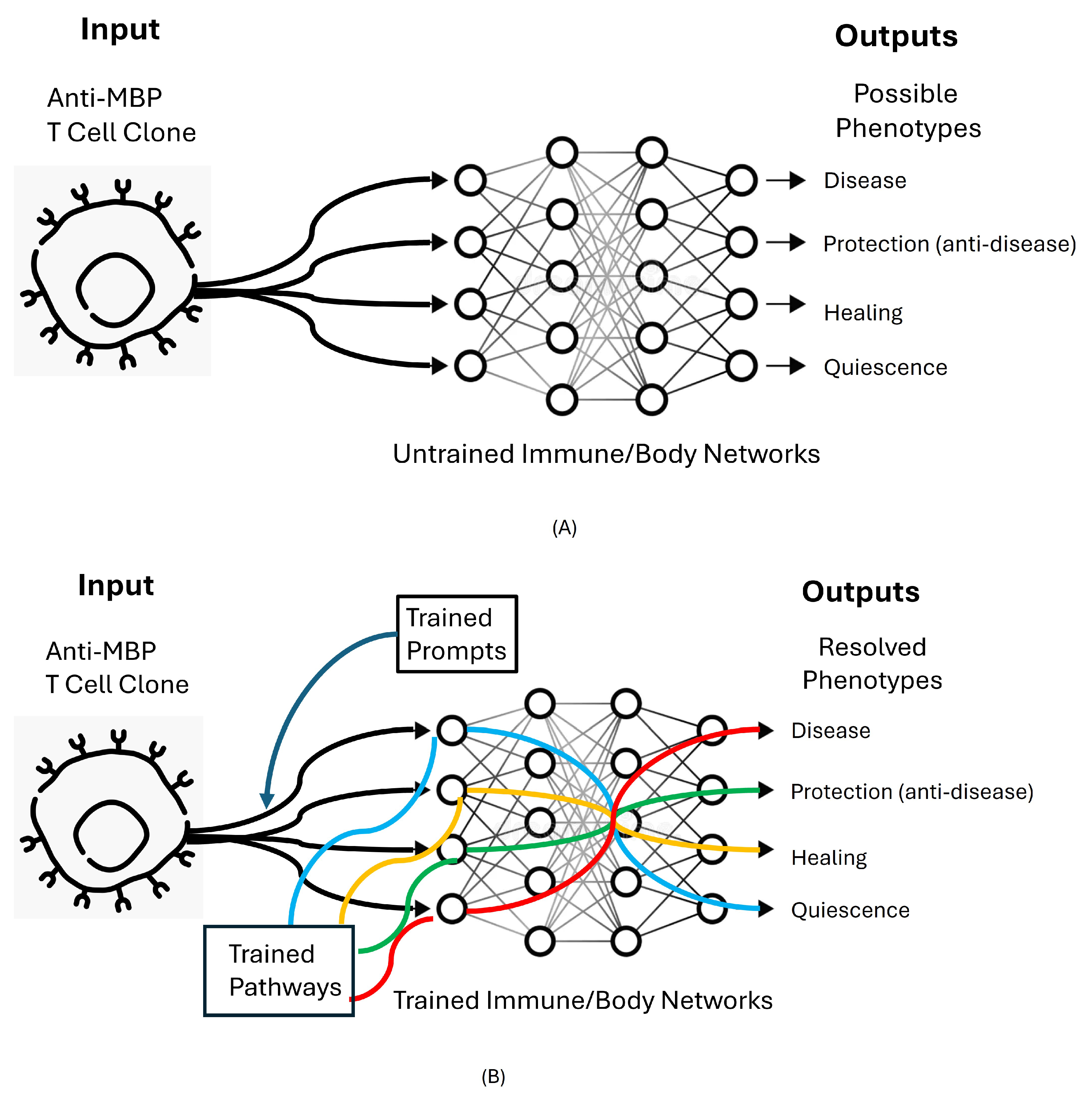
Phenotypes
Self-Immunity
Prompts
Microbiomes
III. Immune System Training in NML
Mother
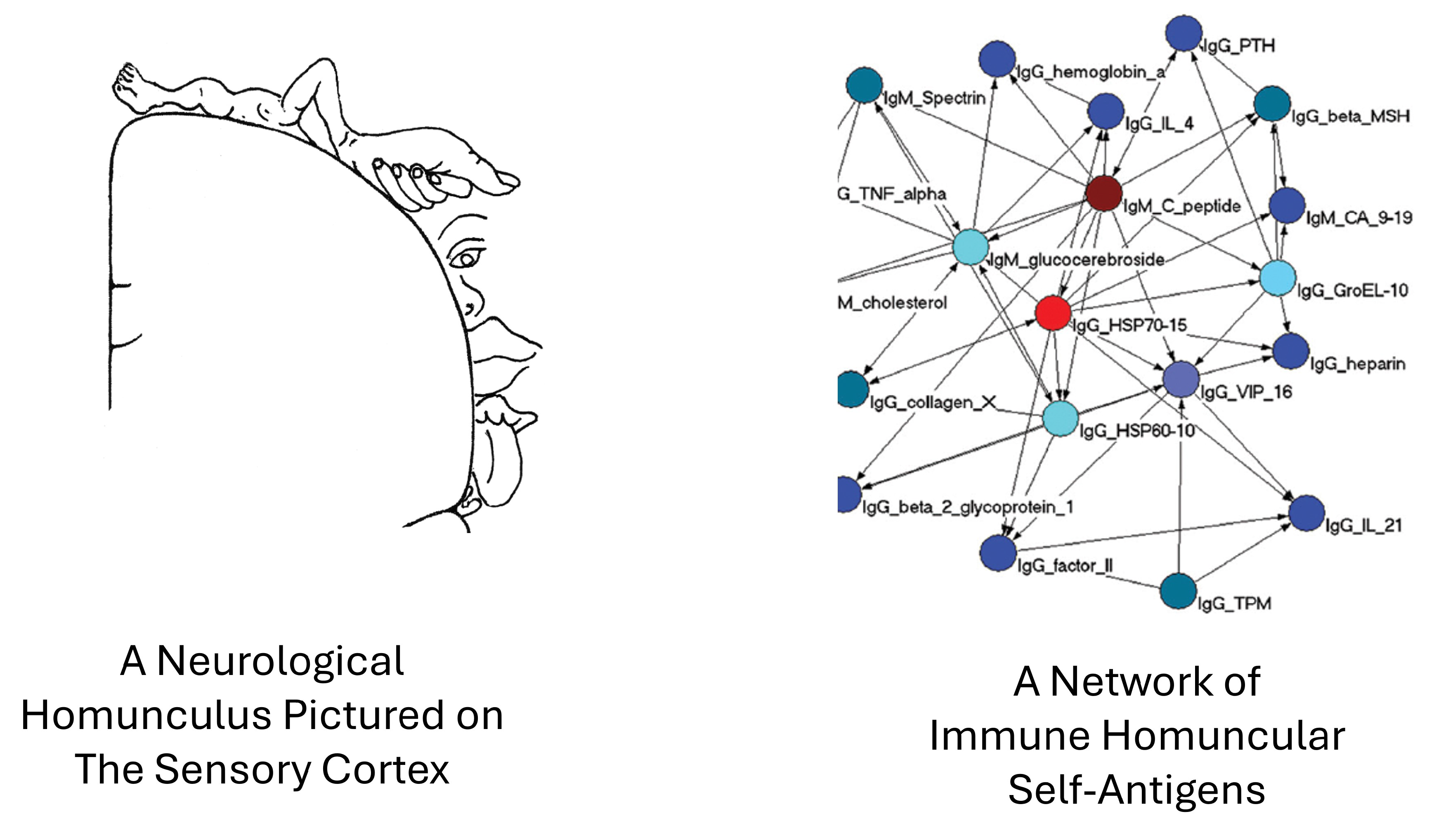
The Immunological Homunculus
Heat Shock Proteins
IV. The Organs of Immune Training
T-Cell Training in the Thymus
B-Cell Training
Lymphocyte Feedback to the Thymus
Ergotypic T-Cell Training
V. NML Feedback Regulation
VI. Training-Based Therapies
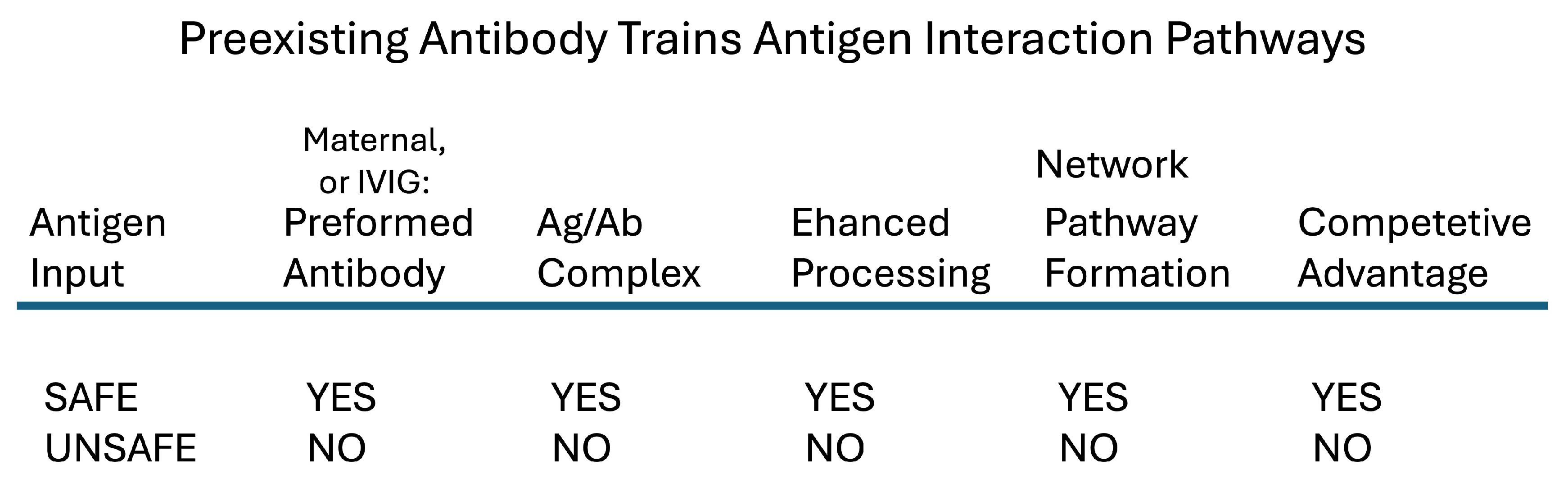
Therapeutic IVIg
T Cell Vaccinations
HSP Molecule Therapies
VII. Conclusions
References
- J Doyne Farmer, Norman H Packard, and Alan S Perelson. The immune system, adaptation, and machine learning. Physica D: Nonlinear Phenomena 1986, 22, 187–204. [Google Scholar] [CrossRef]
- Irun R Cohen. The cognitive paradigm and the immunological homunculus. Immunology today 1992, 13, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Irun R Cohen. Real and artificial immune systems: computing the state of the body. Nature Reviews Immunology 2007, 7, 569–574. [Google Scholar] [CrossRef]
- Irun R Cohen and Sol Efroni. The immune system computes the state of the body: crowd wisdom, machine learning, and immune cell reference repertoires help manage inflammation. Frontiers in immunology 2019, 10, 10. [Google Scholar] [CrossRef]
- Fabiola Curion and Fabian J Theis. Machine learning integrative approaches to advance computational immunology. Genome Medicine 2024, 80, 80. [Google Scholar]
- Irun R Cohen and Assaf Marron. The evolution of universal adaptations of life is driven by universal properties of matter: energy, entropy, and interaction. F1000Research 2020, 9, 626. [Google Scholar] [CrossRef] [PubMed]
- Jan De Houwer, Dermot Barnes-Holmes, and Agnes Moors. What is learning? on the nature and merits of a functional definition of learning. Psychonomic bulletin & review 2013, 20, 631–642. [Google Scholar]
- Sir Frank Macfarlane, Burnet; et al. The clonal selection theory of acquired immunity, volume 3. Vanderbilt University Press Nashville, 1959.
- Alfred I Tauber. Immunity: the evolution of an idea. Oxford University Press, 2017.
- Yifat Merbl, Merav Zucker-Toledano, Francisco J Quintana, Irun R Cohen, et al. Newborn humans manifest autoantibodies to defined self molecules detected by antigen microarray informatics. The Journal of clinical investigation 2007, 117, 712–718. [Google Scholar] [CrossRef]
- Eugene Rosenberg and Ilana Zilber-Rosenberg. The hologenome concept of evolution after 10 years. Microbiome 2018, 6, 1–14. [Google Scholar]
- Holden T Maecker, J Philip McCoy, and Robert Nussenblatt. Standardizing immunophenotyping for the human immunology project. Nature Reviews Immunology 2012, 12, 191–200. [Google Scholar] [CrossRef]
- Irun R Cohen. Tending Adam’s Garden: evolving the cognitive immune self. Elsevier, 2000.
- Charles RM Hay and Donna M DiMichele. The principal results of the international immune tolerance study: a randomized dose comparison. Blood, The Journal of the American Society of Hematology 2012, 119, 1335–1344. [Google Scholar]
- Frank W Marlowe. Hunter-gatherers and human evolution. Evolutionary Anthropology: Issues, News, and Reviews: Issues, News, and Reviews 2005, 14, 54–67. [Google Scholar] [CrossRef]
- Moustapha Mbow, Sanne E de Jong, Lynn Meurs, Souleymane Mboup, Tandakha Ndiaye Dieye, Katja Polman, and Maria Yazdanbakhsh. Changes in immunological profile as a function of urbanization and lifestyle. Immunology 2014, 143, 569–577. [Google Scholar] [CrossRef]
- Mary Muers. Edges, nodes and networks. Nature Reviews Genetics 2010, 11, 4–5. [Google Scholar] [CrossRef] [PubMed]
- Russell Lande. Natural selection and random genetic drift in phenotypic evolution. Evolution, pages 314–334, 1976.
- John Travis. On the origin of the immune system. American Association for the Advancement of Science, 2009.
- Avraham Ben-Nun, Hartmut Wekerle, and Irun R Cohen. The rapid isolation of clonable antigen-specific t lymphocyte lines capable of mediating autoimmune encephalomyelitis. European journal of immunology 1981, 11, 195–199. [Google Scholar] [CrossRef]
- Avraham Ben-Nun, Hartmut Wekerle, and Irun R Cohen. Vaccination against autoimmune encephalomyelitis with t-lymphocite line cells reactive against myelin basic protein. Nature 1981, 292, 60–81. [Google Scholar] [CrossRef]
- Irun R Cohen and Michal Schwartz. Autoimmune maintenance and neuroprotection of the central nervous system. Journal of neuroimmunology, 100(1-2):111–114, 1999.
- Yosef Yarom, Yaakov Naparstek, Varda Lev-Ram, Joseph Holoshitz, Avraham Ben-Nun, and Irun R Cohen. Immunospecific inhibition of nerve conduction by t lymphocytes reactive to basic protein of myelin. Nature, 303(5914):246–247, 1983.
- Yaakov Naparstek, Joseph Holoshitz, Steven Eisenstein, Tamara Reshef, Sara Rappaport, Juan Chemke, Avraham Ben-Nun, and Irun R Cohen. Effector t lymphocyte line cells migrate to the thymus and persist there. Nature 1982, 300, 262–264. [Google Scholar] [CrossRef]
- Ruth Maron, RACHEL Zerubavel, AHARON Friedman, and Irun R Cohen. T lymphocyte line specific for thyroglobulin produces or vaccinates against autoimmune thyroiditis in mice. Journal of immunology (Baltimore, Md.: 1950), 131(5):2316–2322, 1983.
- Joseph Holoshitz, Yaakov Naparstek, Avraham Ben-Nun, and Irun R Cohen. Lines of t lymphocytes induce or vaccinate against autoimmune arthritis. Science, 219(4580):56–58, 1983.
- Dana Elias, Yaron Tikochinski, Gad Frankel, and Irun R Cohen. Regulation of nod mouse autoimmune diabetes by t cells that recognize a tcr cdr3 peptide. International immunology, 11(6):957–966, 1999.
- Biao Zhang, Barry Haddow, and Alexandra Birch. Prompting large language model for machine translation: A case study. In International Conference on Machine Learning, pages 41092–41110. PMLR, 2023.
- Lin Guo. Using metacognitive prompts to enhance self-regulated learning and learning outcomes: A meta-analysis of experimental studies in computer-based learning environments. Journal of Computer Assisted Learning, 38(3):811–832, 2022.
- Matthew P Williams and Roy E Pounder. Helicobacter pylori: from the benign to the malignant. The American journal of gastroenterology, 94(11):S11–S16, 1999.
- Ming Ma, Wei Jiang, and Rongbin Zhou. Damps and damp-sensing receptors in inflammation and diseases. Immunity, 57(4):752–771, 2024.
- Marco E Bianchi. Damps, pamps and alarmins: all we need to know about danger. Journal of Leucocyte Biology 2007, 81, 1–5. [Google Scholar]
- Shizuo Akira and Kiyoshi Takeda. Toll-like receptor signalling. Nature reviews immunology 2004, 4, 499–511. [Google Scholar] [CrossRef]
- Grace Chen, Michael H Shaw, Yun-Gi Kim, and Gabriel Nuñez. Nod-like receptors: role in innate immunity and inflammatory disease. Annual Review of Pathology: Mechanisms of Disease 2009, 4, 365–398. [Google Scholar] [CrossRef]
- Ziv Frankenstein, Uri Alon, and Irun R Cohen. The immune-body cytokine network defines a social architecture of cell interactions. Biology direct, 1:1–15, 2006.
- Yair Neuman. AI for Understanding Context. Springer, 2024.
- Maria Gloria Dominguez-Bello, Filipa Godoy-Vitorino, Rob Knight, and Martin J Blaser. Role of the microbiome in human development. Gut, 68(6):1108–1114, 2019.
- Nita H Salzman. The role of the microbiome in immune cell development. Annals of Allergy, Asthma & Immunology 2014, 113, 593–598. [Google Scholar]
- Timothy R Sampson and Sarkis K Mazmanian. Control of brain development, function, and behavior by the microbiome. Cell host & microbe, 17(5):565–576, 2015.
- Gregory D Sepich-Poore, Laurence Zitvogel, Ravid Straussman, Jeff Hasty, Jennifer A Wargo, and Rob Knight. The microbiome and human cancer. Science, 371(6536):eabc4552, 2021.
- Eugene Rosenberg and Ilana Zilber-Rosenberg. Reconstitution and transmission of gut microbiomes and their genes between generations. Microorganisms, 10(1):70, 2021.
- Joseph M Pickard, Melody Y Zeng, Roberta Caruso, and Gabriel Núñez. Gut microbiota: Role in pathogen colonization, immune responses, and inflammatory disease. Immunological reviews 2017, 279, 70–89. [Google Scholar] [CrossRef]
- Guido Kroemer, Léa Montégut, Oliver Kepp, and Laurence Zitvogel. The danger theory of immunity revisited. Nature Reviews Immunology 2024, 1–17. [Google Scholar]
- Kurtis F Budden, Shaan L Gellatly, David LA Wood, Matthew A Cooper, Mark Morrison, Philip Hugenholtz, and Philip M Hansbro. Emerging pathogenic links between microbiota and the gut–lung axis. Nature Reviews Microbiology, 15(1):55–63, 2017.
- H Cody Meissner, Peter M Strebel, and Walter A Orenstein. Measles vaccines and the potential for worldwide eradication of measles. Pediatrics, 114(4):1065–1069, 2004.
- Susanne Rauch, Edith Jasny, Kim E Schmidt, and Benjamin Petsch. New vaccine technologies to combat outbreak situations. Frontiers in immunology, 9:1963, 2018.
- Patricia Palmeira, Camila Quinello, Ana Lúcia Silveira-Lessa, Cláudia Augusta Zago, and Magda Carneiro-Sampaio. Igg placental transfer in healthy and pathological pregnancies. Journal of Immunology Research, 2012(1):985646, 2012.
- Eddy Roosnek and Antonio Lanzavecchia. Efficient and selective presentation of antigen-antibody complexes by rheumatoid factor b cells. The Journal of experimental medicine, 173(2):487–489, 1991.
- Genevieve G Fouda, David R Martinez, Geeta K Swamy, and Sallie R Permar. The impact of igg transplacental transfer on early life immunity. Immunohorizons, 2(1):14–25, 2018.
- Emma Ronde, Maaike Alkema, Thomas Dierikx, Sam Schoenmakers, Clara Belzer, and Tim de Meij. The influence of maternal gut and vaginal microbiota on gastrointestinal colonization of neonates born vaginally and per caesarean section. BMC Pregnancy and Childbirth, 25(1):1–10, 2025.
- Stephanie B Orchanian, Elaine Y Hsiao, et al. The microbiome as a modulator of neurological health across the maternal-offspring interface. The Journal of Clinical Investigation, 135(4), 2025.
- Noora Carpén, Petter Brodin, Willem M de Vos, Anne Salonen, Kaija-Leena Kolho, Sture Andersson, and Otto Helve. Transplantation of maternal intestinal flora to the newborn after elective cesarean section (secflor): study protocol for a double blinded randomized controlled trial. BMC pediatrics, 22(1):565, 2022.
- Yong Joo Kim. Immunomodulatory effects of human colostrum and milk. Pediatric Gastroenterology, Hepatology & Nutrition, 24(4):337, 2021.
- Noel T Mueller, Elizabeth Bakacs, Joan Combellick, Zoya Grigoryan, and Maria G Dominguez-Bello. The infant microbiome development: mom matters. Trends in molecular medicine, 21(2):109–117, 2015.
- Asaf Madi, Sharron Bransburg-Zabary, Dror Y Kenett, Eshel Ben-Jacob, and Irun R Cohen. The natural autoantibody repertoire in newborns and adults: a current overview. Naturally Occurring Antibodies (NAbs 2012, 198–212. [Google Scholar]
- Franck-Emmanuel Roux, Imène Djidjeli, and Jean-Baptiste Durand. Functional architecture of the somatosensory homunculus detected by electrostimulation. The Journal of physiology, 596(5):941–956, 2018.
- Constantin Fesel and Antonio Coutinho. Dynamics of serum igm autoreactive repertoires following immunization: strain specificity, inheritance and association with autoimmune disease susceptibility. European journal of immunology, 28(11):3616–3629, 1998.
- Alexander Poletaev and Leeza Osipenko. General network of natural autoantibodies as immunological homunculus (immunculus). Autoimmunity Reviews, 2(5):264–271, 2003.
- Asaf Madi, Asaf Poran, Eric Shifrut, Shlomit Reich-Zeliger, Erez Greenstein, Irena Zaretsky, Tomer Arnon, Francois Van Laethem, Alfred Singer, Jinghua Lu, et al. T cell receptor repertoires of mice and humans are clustered in similarity networks around conserved public cdr3 sequences. Elife, 6:e22057, 2017.
- MARTIN E Munk, BERND Schoel, SUSANNE Modrow, ROBERT W Karr, RA Young, and SH Kaufmann. T lymphocytes from healthy individuals with specificity to self-epitopes shared by the mycobacterial and human 65-kilodalton heat shock protein. Journal of immunology (Baltimore, Md.: 1950), 143(9):2844–2849, 1989.
- Ulrich Zügel and Stefan HE Kaufmann. Role of heat shock proteins in protection from and pathogenesis of infectious diseases. Clinical Microbiology Reviews 1999, 39, 19–39. [Google Scholar]
- Martin E Feder and Gretchen E Hofmann. Heat-shock proteins, molecular chaperones, and the stress response: evolutionary and ecological physiology. Annual review of physiology, 61(1):243–282, 1999.
- Irun R Cohen and Douglas B Young. Autoimmunity, microbial immunity and the immunological homunculus. Immunology today, 12(4):105–110, 1991.
- Xolani Henry Makhoba. Two sides of the same coin: heat shock proteins as biomarkers and therapeutic targets for some complex diseases. Frontiers in Molecular Biosciences, 12:1491227, 2025.
- Francisco J Quintana, Avishai Mimran, Pnina Carmi, Felix Mor, and Irun R Cohen. Hsp60 as a target of anti-ergotypic regulatory t cells. PLoS One 2008, 3, e4026. [Google Scholar] [CrossRef]
- Francisco J Quintana and Irun R Cohen. The hsp60 immune system network. Trends in immunology, 32(2):89–95, 2011.
- Jagoda Mantej, Kinga Polasik, Ewa Piotrowska, and Stefan Tukaj. Autoantibodies to heat shock proteins 60, 70, and 90 in patients with rheumatoid arthritis. Cell Stress and Chaperones 2019, 24, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Aijing Liu, Concetta Ferretti, Fu-Dong Shi, Irun R Cohen, Francisco J Quintana, and Antonio La Cava. Dna vaccination with hsp70 protects against systemic lupus erythematosus in (nzb× nzw) f1 mice. Arthritis & Rheumatology, 72(6):997–1002, 2020.
- Ohad S Birk, Dana Elias, Alona S Weiss, Ada Rosen, Ruurd Van-Der Zee, Michael D Walker, and Irun R Cohen. Nod mouse diabetes: the ubiquitous mouse hsp60 is a β-cell target antigen of autoimmune t cells. Journal of Autoimmunity, 9(2):159–166, 1996.
- Marina Ferrarini, Silvia Heltai, M Raffaella Zocchi, and Claudio Rugarli. Unusual expression and localization of heat-shock proteins in human tumor cells. International Journal of Cancer, 51(4):613–619, 1992.
- David Margel, Meirav Pesvner-Fischer, Jack Baniel, Ofer Yossepowitch, and Irun R Cohen. Stress proteins and cytokines are urinary biomarkers for diagnosis and staging of bladder cancer. European urology 2011, 59, 113–119. [Google Scholar] [CrossRef]
- Jacques FAP Miller. The function of the thymus and its impact on modern medicine. Science 2020, 369, eaba2429. [Google Scholar] [CrossRef]
- Nara Vasconcelos Cavalcanti, Patricia Palmeira, Marcelo Biscegli Jatene, Mayra de Barros Dorna, and Magda Carneiro-Sampaio. Early thymectomy is associated with long-term impairment of the immune system: a systematic review. Frontiers in Immunology 2021, 12, 774780. [Google Scholar] [CrossRef]
- Dennis D Taub and Dan L Longo. Insights into thymic aging and regeneration. Immunological reviews 2005, 205, 72–93. [Google Scholar] [CrossRef] [PubMed]
- Richard R Hardy and Kyoko Hayakawa. B cell development pathways. Annual review of immunology, 19(1):595–621, 2001.
- David B Agus, Charles D Surh, and Jonathan Sprent. Reentry of t cells to the adult thymus is restricted to activated t cells. The Journal of experimental medicine, 173(5):1039–1046, 1991.
- Ansgar W Lohse, Felix Mor, Nathan Karin, and Irun R Cohen. Control of experimental autoimmune encephalomyelitis by t cells responding to activated t cells. Science 1989, 244, 820–822. [Google Scholar] [CrossRef] [PubMed]
- Patrizia Scapini and Marco A Cassatella. Social networking of human neutrophils within the immune system. Blood, The Journal of the American Society of Hematology, 124(5):710–719, 2014.
- Sheng Chen, Stephen A Billings, and PM Grant. Non-linear system identification using neural networks. International journal of control, 51(6):1191–1214, 1990.
- Katarzyna Janocha and Wojciech Marian Czarnecki. On loss functions for deep neural networks in classification. arXiv preprint , 2017. arXiv:1702.05659, 2017.
- David Furman, Judith Campisi, Eric Verdin, Pedro Carrera-Bastos, Sasha Targ, Claudio Franceschi, Luigi Ferrucci, Derek W Gilroy, Alessio Fasano, Gary W Miller, et al. Chronic inflammation in the etiology of disease across the life span. Nature medicine, 25(12):1822–1832, 2019.
- Claire Larroche, Youri Chanseaud, Paloma Garcia de la Pena-Lefebvre, and Luc Mouthon. Mechanisms of intravenous immunoglobulin action in the treatment of autoimmune disorders. BioDrugs, 16:47–55, 2002.
- A Achiron, R Gilad, Raanan Margalit, U Gabbay, I Sarova-Pinhas, Irun R Cohen, E Melamed, O Lider, S Noy, and I Ziv. Intravenous gammaglobulin treatment in multiple sclerosis and experimental autoimmune encephalomyelitis: delineation of usage and mode of action. Journal of Neurology, Neurosurgery & Psychiatry, 57(Suppl):57–61, 1994.
- Marinos C Dalakas. Intravenous immunoglobulin in autoimmune neuromuscular diseases. Jama, 291(19):2367–2375, 2004.
- Dimitrios Karussis, Hagai Shor, Julia Yachnin, Naama Lanxner, Merav Amiel, Keren Baruch, Yael Keren-Zur, Ofra Haviv, Massimo Filippi, Panayiota Petrou, et al. T cell vaccination benefits relapsing progressive multiple sclerosis patients: a randomized, double-blind clinical trial. PloS one, 7(12):e50478, 2012.
- A Achiron, G Lavie, I Kishner, Y Stern, I Sarova-Pinhas, T Ben-Aharon, Yoram Barak, H Raz, M Lavie, T Barliya, et al. T cell vaccination in multiple sclerosis relapsing–remitting nonresponders patients. Clinical immunology 2004, 113, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Itamar Raz, Dana Elias, Ann Avron, Merana Tamir, Muriel Metzger, and Irun R Cohen. β-cell function in new-onset type 1 diabetes and immunomodulation with a heat-shock protein peptide (diapep277): a randomised, double-blind, phase ii trial. The Lancet, 358(9295):1749–1753, 2001.
- Julia Spierings and Willem van Eden. Heat shock proteins and their immunomodulatory role in inflammatory arthritis. Rheumatology, 56(2):198–208, 2017.
- Michael D Rosenblum, Iris K Gratz, Jonathan S Paw, and Abul K Abbas. Treating human autoimmunity: current practice and future prospects. Science translational medicine, 4(125):125sr1–125sr1, 2012.
- Ofer N Gofrit, Benjamin Y Klein, Irun R Cohen, Tamir Ben-Hur, Charles L Greenblatt, and Hervé Bercovier. Bacillus calmette-guérin (bcg) therapy lowers the incidence of alzheimer’s disease in bladder cancer patients. PLoS One, 14(11):e0224433, 2019.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



