Submitted:
06 May 2025
Posted:
09 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Section
2.1. Synthesis of the Corrosion Inhibitors
2.2. Weight Loss Measurements
2.3. Electrochemical Measurements
2.4. Computational Methods
2.4.1. MD Simulations
2.5. Surface Analyses
2.5.1. FTIR Analysis
2.5.2. Scanning Electron Microscopy (SEM)
3. Results and Discussion
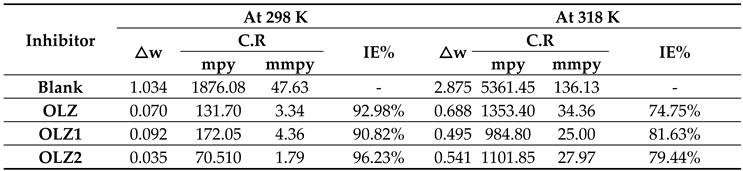
3.1. Weight Loss Measurements
3.2. Open Circuit Potential Measurements
3.3. Electrochemical Impedance Spectroscopy (EIS)
3.4. Potentiodynamic Polarization Studies (PDP)
3.5. Linear Polarization Resistance (LPR)
3.6. Quantum Chemistry Calculations
3.7. Adsorption Behavior
3.8. Diffusion Study
3.9. Surface Analysis
3.9.1. SEM-EDX Analysis
3.9.2. FT-IR Spectroscopy
3.10. Inhibition Mechanism
| Parameters | Inhibitor molecules | ||
|---|---|---|---|
| OLZ | OLZ1 | OLZ2 | |
| EHOMO (eV) | −4.643 | −4.637 | −4.669 |
| ELUMO (eV) | −1.865 | −1.852 | −1.879 |
| I (eV) | 4.643 | 4.637 | 4.669 |
| A (eV) | 1.865 | 1.852 | 1.879 |
| ΔE (eV) | 2.778 | 2.785 | 2.790 |
| χ (eV) | 3.254 | 3.245 | 3.274 |
| η (eV) | 1.389 | 1.393 | 1.395 |
| ΔN (eV) | −2.697 | −2.697 | −2.671 |
| μ (Debye) | 4.603 | 6.510 | 5.160 |
| Inhibitor film | D (x10⁻⁹ m²․s⁻¹) | ||
|---|---|---|---|
| H2O | H3O+ | Cl− | |
| Blank | 2.79 | 2.57 | 1.24 |
| OLZ | 0.39 | 0.35 | 0.07 |
| OLZ1 | 0.66 | 0.87 | 0.25 |
| OLZ2 | 0.35 | 0.17 | 0.19 |
| NO. | Band | Absorbance Peak | Reason |
|---|---|---|---|
| 1 | Fe-O | 450 | The presence of a sharp, intense peak indicates the formation of ferrous and ferric oxides or hydroxides |
| 2 | O-H | 3300 | The presence of a hydroxyl group is an indication of the formation of ferric oxides or hydroxides |
| 3 | C-Cl | 1000 | Presence of chloride ions as corrosion products |
| 4 | Fe-O | 490 | Reduction in peak intensity due to the presence of the inhibitor |
| 5 | C-Cl | 1000 | Reduction in peak intensity due to the presence of the inhibitor |
| 6 | O-H | 3300 | Disappearance of the hydroxyl group in the presence of the inhibitor |
4. Conclusions
- The three inhibition properties of OLZ and its derivatives on C1018 carbon steel in 1M HCl solution were very high at 300 ppm, where inhibition efficiencies exceeded 88%. However, inhibition efficiency was temperature and concentration-dependent.
- Electrochemical studies indicate that the three inhibitors are predominantly anodic. PDP-derived inhibition efficiencies were in agreement with those obtained from EIS measurements.
- At 298 K, the inhibition efficiency followed the order OLZ2> OLZ1> OLZ, while at 318 K, it followed the order OLZ1 > OLZ2 > OLZ, which confirms that the structural modifications enhanced the corrosion inhibition properties of the parent molecule.
- The SEM-EDX analysis demonstrates that OLZ and its derivatives effectively inhibit steel corrosion in 1M HCl compared to the uninhibited solution.
- The FTIR spectroscopy results show that the metal surface active sites include -NH, -C=N, C=C and aromatic ring structures.
Supplementary Materials
References
- Jacobson, G.A. NACE international’s IMPACT study breaks new ground in corrosion management research and practice. The Bridge 2016, 46. [Google Scholar]
- Fayomi, O.; Akande, I.; Odigie, S. Economic impact of corrosion in oil sectors and prevention: an overview. Journal of Physics: Conference Series, IOP Publishing 2019, 022037. [Google Scholar] [CrossRef]
- Al-Moubaraki, A.H.; Obot, I.B. Corrosion challenges in petroleum refinery operations: Sources, mechanisms, mitigation, and future outlook. Journal of Saudi Chemical Society 2021, 25, 101370. [Google Scholar] [CrossRef]
- Raghavendra, N. Green compounds to attenuate aluminum corrosion in HCl activation: a necessity review. Chemistry Africa 2020, 3, 21–34. [Google Scholar] [CrossRef]
- Baari, M.J.; Sabandar, C.W. A Review on expired drug-based corrosion inhibitors: Chemical composition, structural effects, inhibition mechanism, current challenges, and future prospects. Indonesian Journal of Chemistry 2021, 21, 1316–1336. [Google Scholar] [CrossRef]
- Narang, R.; Vashishth, P.; Bairagi, H.; Sehrawat, R.; Shukla, S.; Mangla, B. Applicability of drugs as sustainable corrosion inhibitors. Adv Mater Lett 2023, 14, 2304–1732. [Google Scholar] [CrossRef]
- Tanwer, S.; Shukla, S.K. Recent advances in the applicability of drugs as corrosion inhibitor on metal surface: A review. Current Research in Green and Sustainable Chemistry 2022, 5, 100227. [Google Scholar] [CrossRef]
- Piggott, T.; Moja, L.; Huttner, B.; Okwen, P.; Raviglione, M.C.B.; Kredo, T.; Schünemann, H.J. WHO Model list of essential medicines: Visions for the future. Bulletin of the World Health Organization 2024, 102, 722. [Google Scholar] [CrossRef]
- Rehan, S.T.; Siddiqui, A.H.; Khan, Z.; Imran, L.; Syed, A.A.; Tahir, M.J.; Jassani, Z.; Singh, M.; Asghar, M.S.; Ahmed, A. Samidorphan/olanzapine combination therapy for schizophrenia: Efficacy, tolerance and adverse outcomes of regimen, evidence-based review of clinical trials. Annals of medicine and surgery 2022, 79, 104115–104115. [Google Scholar] [CrossRef]
- ClinCalc DrugStats Database. The Top 300 Drugs of 2022 in the United States by Prescription. ClinCalc LLC; 2022. 2022.
- Obermayer, D.; Glasnov, T.N.; Kappe, C.O. Microwave-assisted and continuous flow multistep synthesis of 4-(pyrazol-1-yl) carboxanilides. The Journal of Organic Chemistry 2011, 76, 6657–6669. [Google Scholar] [CrossRef]
- Obermayer, D.; Kappe, C.O. On the importance of simultaneous infrared/fiber-optic temperature monitoring in the microwave-assisted synthesis of ionic liquids. Organic & biomolecular chemistry 2010, 8, 114–121. [Google Scholar]
- Gunawan, R.; Nandiyanto, A.B.D. How to read and interpret 1H-NMR and 13C-NMR spectrums. Indonesian Journal of Science and Technology 2021, 6, 267–298. [Google Scholar] [CrossRef]
- Standard, A. G31-72: Standard Practice for Laboratory Immersion Corrosion Testing of Metals. Annual Book of ASTM Standards; ASTM: West Conshohocken, PA, USA, 2004. [Google Scholar]
- Satapathy, A.; Gunasekaran, G.; Sahoo, S.; Amit, K.; Rodrigues, P. Corrosion inhibition by Justicia gendarussa plant extract in hydrochloric acid solution. Corrosion science 2009, 51, 2848–2856. [Google Scholar] [CrossRef]
- Testing, A.S. f.; Materials In ASTM G1-03: Standard Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens, ASTM: 2004.
- Shankar, U.; Gogoi, R.; Sethi, S.K.; Verma, A. Introduction to materials studio software for the atomistic-scale simulations. In Forcefields for atomistic-scale simulations: materials and applications; Springer, 2022; pp. 299–299. [Google Scholar]
- Klamt, A.; Schüürmann, G. COSMO: a new approach to dielectric screening in solvents with explicit expressions for the screening energy and its gradient. Journal of the Chemical Society, Perkin Transactions 2 1993, 799–805. [Google Scholar] [CrossRef]
- Qiang, Y.; Zhi, H.; Guo, L.; Fu, A.; Xiang, T.; Jin, Y. Experimental and molecular modeling studies of multi-active tetrazole derivative bearing sulfur linker for protecting steel from corrosion. Journal of Molecular Liquids 2022, 351, 118638. [Google Scholar] [CrossRef]
- Obot, I.; Bahraq, A.A.; Alamri, A.H. Density functional theory and molecular dynamics simulation of the corrosive particle diffusion in pyrimidine and its derivatives films. Computational Materials Science 2022, 210, 111428. [Google Scholar] [CrossRef]
- Sokolov, I.M.; Klafter, J. From diffusion to anomalous diffusion: a century after Einstein’s Brownian motion. Chaos: An Interdisciplinary Journal of Nonlinear Science 2005, 15. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.-Q.; Guo, A.-L.; Yan, Y.-G.; Jia, X.-L.; Geng, Y.-F.; Guo, W.-Y. Computer simulation of diffusion of corrosive particle in corrosion inhibitor membrane. Computational and Theoretical Chemistry 2011, 964, 176–181. [Google Scholar] [CrossRef]
- Dutta, A. Fourier transform infrared spectroscopy. Spectroscopic methods for nanomaterials characterization 2017, 73–93. [Google Scholar]
- Mohammed, A.; Abdullah, A. In Scanning electron microscopy (SEM): A review, Proceedings of the 2018 international conference on hydraulics and pneumatics—HERVEX, Băile Govora, Romania, 2018; pp 7-9.
- Nahlé, A.; Abu-Abdoun, I.I.; Abdel-Rahman, I. Effect of Temperature on the Corrosion Inhibition of Trans-4-Hydroxy-4′-Stilbazole on Mild Steel in HCl Solution. International Journal of corrosion 2012, 2012, 380329. [Google Scholar] [CrossRef]
- Obot, I.; Obi-Egbedi, N.; Umoren, S. Adsorption characteristics and corrosion inhibitive properties of clotrimazole for aluminium corrosion in hydrochloric acid. International Journal of Electrochemical Science 2009, 4, 863–877. [Google Scholar] [CrossRef]
- Sarkar, T.K.; Saraswat, V.; Mitra, R.K.; Obot, I.; Yadav, M. Mitigation of corrosion in petroleum oil well/tubing steel using pyrimidines as efficient corrosion inhibitor: Experimental and theoretical investigation. Materials Today Communications 2021, 26, 101862. [Google Scholar] [CrossRef]
- Tang, J.; Wang, H.; Jiang, X.; Zhu, Z.; Xie, J.; Tang, J.; Wang, Y.; Chamas, M.; Zhu, Y.; Tian, H. Electrochemical behavior of jasmine tea extract as corrosion inhibitor for carbon steel in hydrochloric acid solution. International Journal of Electrochemical Science 2018, 13, 3625–3642. [Google Scholar] [CrossRef]
- Prasanna, B.; Praveen, B.; Hebbar, N.; Venkatesha, T. Anticorrosion potential of hydralazine for corrosion of mild steel in 1m hydrochloric acid solution. Journal of Fundamental and Applied Sciences 2015, 7, 222–243. [Google Scholar] [CrossRef]
- Oguzie, E.; Li, Y.; Wang, F. Corrosion inhibition and adsorption behavior of methionine on mild steel in sulfuric acid and synergistic effect of iodide ion. Journal of colloid and interface science 2007, 310, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Haruna, K.; Saleh, T.A.; Quraishi, M. Expired metformin drug as green corrosion inhibitor for simulated oil/gas well acidizing environment. Journal of Molecular Liquids 2020, 315, 113716. [Google Scholar] [CrossRef]
- Mobin, M.; Zamindar, S.; Banerjee, P. Mechanistic insight into adsorption and anti-corrosion capability of a novel surfactant-derived ionic liquid for mild steel in HCl medium. Journal of Molecular Liquids 2023, 385, 122403. [Google Scholar] [CrossRef]
- Touir, R.; Errahmany, N.; Rbaa, M.; Benhiba, F.; Doubi, M.; Kafssaoui, E.E.; Lakhrissi, B. Experimental and computational chemistry investigation of the molecular structures of new synthetic quinazolinone derivatives as acid corrosion inhibitors for mild steel. Journal of Molecular Structure 2024, 1303, 137499. [Google Scholar] [CrossRef]
- Fernandes, C.M.; Costa, A.R.; Leite, M.C.; Martins, V.; Lee, H.-S.; da CS Boechat, F.; de Souza, M.C.; Batalha, P.N.; Lgaz, H.; Ponzio, E.A. A detailed experimental performance of 4-quinolone derivatives as corrosion inhibitors for mild steel in acid media combined with first-principles DFT simulations of bond breaking upon adsorption. Journal of Molecular Liquids 2023, 375, 121299. [Google Scholar] [CrossRef]
- Acidi, A.; Sedik, A.; Rizi, A.; Bouasla, R.; Rachedi, K.O.; Berredjem, M.; Delimi, A.; Abdennouri, A.; Ferkous, H.; Yadav, K.K. Examination of the main chemical components of essential oil of Syzygium aromaticum as a corrosion inhibitor on the mild steel in 0.5 M HCl medium. Journal of Molecular Liquids 2023, 391, 123423. [Google Scholar] [CrossRef]
- Millard, S.; Law, D.; Bungey, J.; Cairns, J. Environmental influences on linear polarisation corrosion rate measurement in reinforced concrete. Ndt & E International 2001, 34, 409–417. [Google Scholar]
- Olasunkanmi, L.O.; Obot, I.B.; Kabanda, M.M.; Ebenso, E.E. Some quinoxalin-6-yl derivatives as corrosion inhibitors for mild steel in hydrochloric acid: experimental and theoretical studies. The Journal of Physical Chemistry C 2015, 119, 16004–16019. [Google Scholar] [CrossRef]
- Umoren, S.A. Polypropylene glycol: A novel corrosion inhibitor for× 60 pipeline steel in 15% HCl solution. Journal of Molecular Liquids 2016, 219, 946–958. [Google Scholar] [CrossRef]
- Zhang, J.; Qiao, G.; Hu, S.; Yan, Y.; Ren, Z.; Yu, L. Theoretical evaluation of corrosion inhibition performance of imidazoline compounds with different hydrophilic groups. Corrosion Science 2011, 53, 147–152. [Google Scholar] [CrossRef]
- Guo, L.; Kaya, S.; Obot, I.B.; Zheng, X.; Qiang, Y. Toward understanding the anticorrosive mechanism of some thiourea derivatives for carbon steel corrosion: A combined DFT and molecular dynamics investigation. Journal of colloid and interface science 2017, 506, 478–485. [Google Scholar] [CrossRef]
- Zhang, X.; Kang, Q.; Wang, Y. Theoretical study of N-thiazolyl-2-cyanoacetamide derivatives as corrosion inhibitor for aluminum in alkaline environments. Computational and Theoretical Chemistry 2018, 1131, 25–32. [Google Scholar] [CrossRef]
- Obi-Egbedi, N.; Obot, I.; El-Khaiary, M.I. Quantum chemical investigation and statistical analysis of the relationship between corrosion inhibition efficiency and molecular structure of xanthene and its derivatives on mild steel in sulphuric acid. Journal of Molecular Structure 2011, 1002, 89–96. [Google Scholar] [CrossRef]
- Verma, C.; Quraishi, M.; Singh, A. 5-Substituted 1H-tetrazoles as effective corrosion inhibitors for mild steel in 1 M hydrochloric acid. Journal of Taibah University for Science 2016, 10, 718–733. [Google Scholar] [CrossRef]
- Ouakki, M.; Galai, M.; Rbaa, M.; Abousalem, A.S.; Lakhrissi, B.; Rifi, E.; Cherkaoui, M. Investigation of imidazole derivatives as corrosion inhibitors for mild steel in sulfuric acidic environment: experimental and theoretical studies. Ionics 2020, 26, 5251–5272. [Google Scholar] [CrossRef]
- Verma, C.; Quraishi, M.; Rhee, K.Y. Electronic effect vs. molecular size effect: experimental and computational based designing of potential corrosion inhibitors. Chemical Engineering Journal 2022, 430, 132645. [Google Scholar] [CrossRef]
- Hou, B.; Zhang, Q.; Li, Y.; Zhu, G.; Zhang, G. Influence of corrosion products on the inhibition effect of pyrimidine derivative for the corrosion of carbon steel under supercritical CO2 conditions. Corrosion Science 2020, 166, 108442. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Z.; Zhang, L.; Lu, M. Theoretical insights into the inhibition performance of three neonicotine derivatives as novel type of inhibitors on carbon steel. Journal of Renewable Materials 2020, 8, 819–832. [Google Scholar] [CrossRef]
- Yan, Y.; Wang, X.; Zhang, Y.; Wang, P.; Cao, X.; Zhang, J. Molecular dynamics simulation of corrosive species diffusion in imidazoline inhibitor films with different alkyl chain length. Corrosion science 2013, 73, 123–129. [Google Scholar] [CrossRef]
- Aziz, I.A.A.; Abdulkareem, M.H.; Annon, I.A.; Hanoon, M.M.; Al-Kaabi, M.H.; Shaker, L.M.; Alamiery, A.A.; Isahak, W.N.R.W.; Takriff, M.S. Weight loss, thermodynamics, SEM, and electrochemical studies on N-2-methylbenzylidene-4-antipyrineamine as an inhibitor for mild steel corrosion in hydrochloric acid. Lubricants 2022, 10, 23. [Google Scholar] [CrossRef]
- Obot, I.; Umoren, S.; Ankah, N. Pyrazine derivatives as green oil field corrosion inhibitors for steel. Journal of Molecular Liquids 2019, 277, 749–761. [Google Scholar] [CrossRef]
- Dao, D.Q.; Hieu, T.D.; Le Minh Pham, T.; Tuan, D.; Nam, P.C.; Obot, I.B. DFT study of the interactions between thiophene-based corrosion inhibitors and an Fe 4 cluster. Journal of Molecular Modeling 2017, 23, 1–15. [Google Scholar] [CrossRef]
- Saranya, J.; Sounthari, P.; Parameswari, K.; Chitra, S. Acenaphtho [1, 2-b] quinoxaline and acenaphtho [1, 2-b] pyrazine as corrosion inhibitors for mild steel in acid medium. Measurement 2016, 77, 175–186. [Google Scholar] [CrossRef]
- Singh, P.; Srivastava, V.; Quraishi, M. Novel quinoline derivatives as green corrosion inhibitors for mild steel in acidic medium: electrochemical, SEM, AFM, and XPS studies. Journal of Molecular Liquids 2016, 216, 164–173. [Google Scholar] [CrossRef]
 |
| Inh. | Rs Ω⋅cm2 |
CPEdl |
Rct Ω⋅cm2 |
CPEf |
Rf Ω⋅cm2 |
Cdl μF·cm⁻² | Rp Ω⋅cm2 |
IE % |
||
|---|---|---|---|---|---|---|---|---|---|---|
| Y01 (mΩsncm−2) |
n 1 |
Y02 (mΩsncm−2) | n 2 | |||||||
| Blank | 131.15 | 89.380 | 0.867 | 1.74 | 70.465 | 0.958 | 8.005 | 193.850 | 140.897 | - |
| OLZ | 0.74 | 63.657 | 0.967 | 10.89 | 92.736 | 0.882 | 1189.333 | 79.578 | 1200.97 | 88.28 |
| OLZ1 | 1.64 | 47.53 | 0.972 | 1416.3 | 20.820 | 0.984 | 12.449 | 65.471 | 1430.42 | 90.15 |
| OLZ2 | 1.32 | 50.41 | 0.996 | 898.06 | 29.413 | 0.904 | 747.033 | 51.416 | 1646.42 | 91.45 |
| Inhibitor | PDP | LPR | |||||
|---|---|---|---|---|---|---|---|
| Ecorr (mV/SCE) |
icorr (μA cm−2) |
βa (mV/dec) |
βc (mV/dec) |
IE% | Rp | IE% | |
| Blank | -447.00 | 200.00 | 160.80 | 120.00 | - | 132.20 | - |
| OLZ | -401.000 | 20.100 | 100.00 | 132.700 | 89.95 | 1135.66 | 88.35 |
| OLZ1 | -397.000 | 15.900 | 89.000 | 218.600 | 92.05 | 1473.00 | 91.01 |
| OLZ2 | -422.000 | 14.600 | 91.400 | 241.600 | 92.70 | 1760.66 | 92.47 |
| Element | Metal | Metal with HCl | Metal with OLZ | Metal with OLZ1 | Metal with OLZ2 |
|---|---|---|---|---|---|
| N | 1.57 | 0.91 | 1.74 | 1.68 | 1.68 |
| O | 1.69 | 20.76 | 1.62 | 1.78 | 2.24 |
| C | 9.69 | 14.42 | 11.41 | 10.75 | 10.81 |
| S | 0.46 | 0.36 | 0.43 | 0.43 | 0.39 |
| P | 0.65 | 0.51 | 0.59 | 0.73 | 0.67 |
| Mn | 0.38 | 0.33 | 0.31 | 0.45 | 0.40 |
| Fe | 85.56 | 62.71 | 83.90 | 84.19 | 85.49 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




