Submitted:
06 May 2025
Posted:
07 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction to Advanced Drug Delivery Technologies
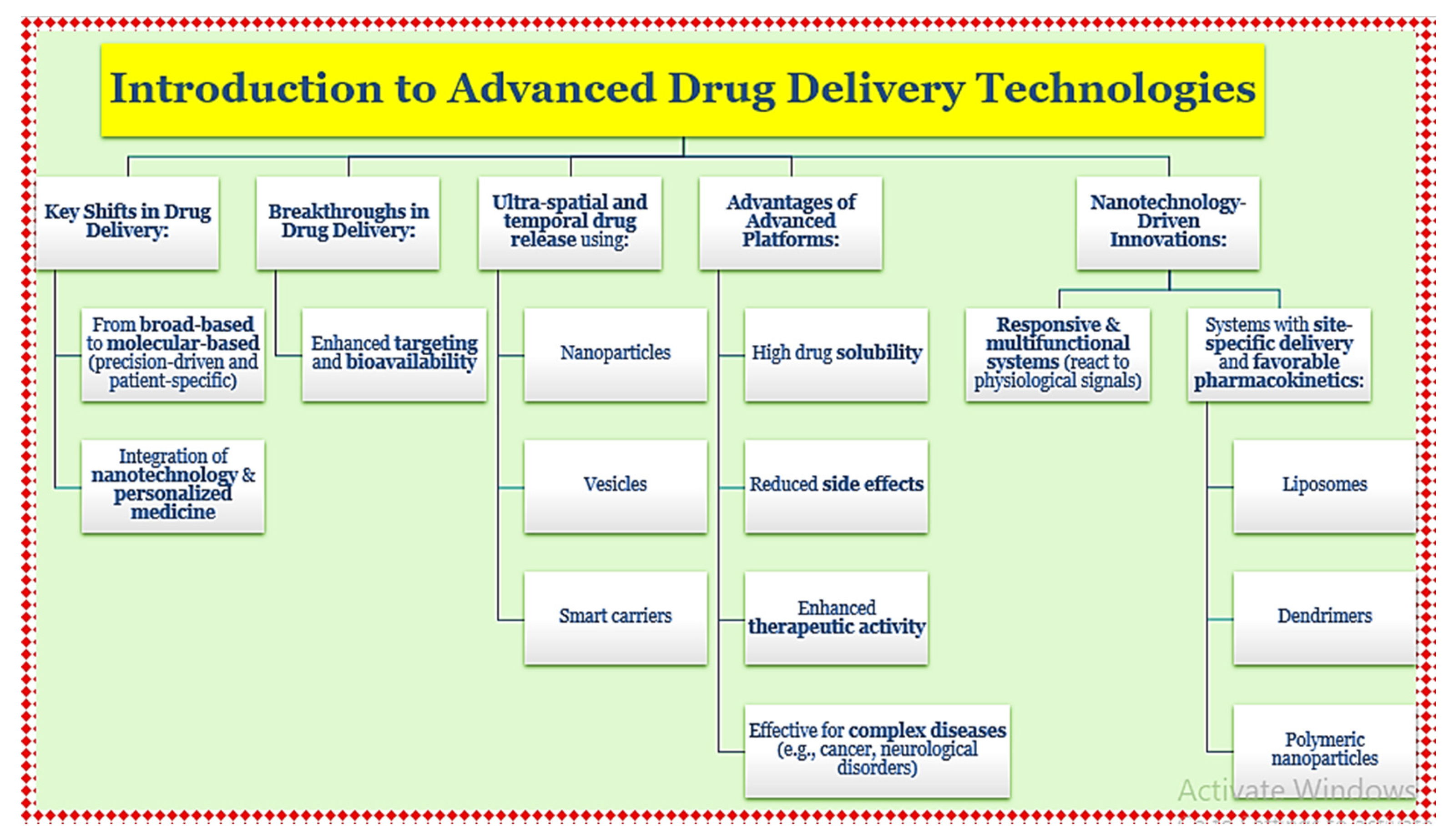
| Topic | Details |
|---|---|
| Revolution in Drug Delivery | The field has shifted from traditional dosage forms to precision-based, patient-profiled therapeutics, enabled by advances in nanotechnology and personalized medicine. |
| Targeting & Bioavailability | New platforms like nanoparticles and smart carriers allow for ultra-specific spatial and temporal drug release, enhancing solubility and reducing side effects. |
| Complex Diseases Addressed | These systems are especially beneficial in treating complex conditions such as cancer and neurological disorders. |
| Responsive Systems | Nanotechnology has led to systems that respond to physiological signals (e.g., pH, temperature) for targeted drug release. |
| Types of Carriers | Includes liposomes, dendrimers, and polymeric nanoparticles with favorable pharmacokinetics and improved drug targeting compared to conventional routes. |
| Shift in Formulation Approach | Represents a paradigm shift from broad-based drug delivery to molecular-level, responsive systems tailored to individual patient needs. |
2. Smart Nanocarriers for Precision Medicine
| Topic | Details |
|---|---|
| Foundation of Precision Medicine | Smart nanocarriers enable site-specific drug delivery with minimal off-target toxicity, forming a core part of precision medicine. |
| Types of Delivery Systems | Includes liposomal, vesicular, and nanoparticle-based systems designed for superior pharmacokinetics and targeting specificity. |
| Therapeutic Engineering | These nano-scale carriers are bioengineered for controlled release, high biocompatibility, and are ideal for localized therapy. |
| Ligand-Targeted Liposomes | Surface-engineered to bind cellular receptors, allowing precise targeting of diseased tissue and enhancing therapeutic selectivity. |
| Challenges and Benefits | While immune evasion and stability remain concerns, such systems offer a promising route for selective and effective drug delivery. |
| Programmable Lipid Nanoparticles | Modular, four-domain structures enable functional targeting and responsive release, proving effective for both systemic and organ-specific drug delivery. |
3. Transdermal and Mucosal Delivery Innovations
| Aspect | Details |
|---|---|
| Overview | Microneedling offers a minimally invasive, pain-free method for mucosal and transdermal drug and vaccine delivery. |
| Mechanism of Action | Microscopic needle tips penetrate the stratum corneum to deliver drugs into the dermis, increasing bioavailability and improving patient compliance. |
| Advantages for Large Molecules | Particularly effective for biologics and large molecules that cannot passively permeate the skin. |
| Second-Generation Innovations | Comprise advanced materials and structural designs allowing controlled release, improved mechanical strength, and higher therapeutic effectiveness. |
| Versatility | Capable of delivering a broad spectrum of therapeutic agents, including small molecules, peptides, proteins, and nucleic acids (e.g., DNA). |
4. Biodegradable and Sustainable Systems in Drug Delivery
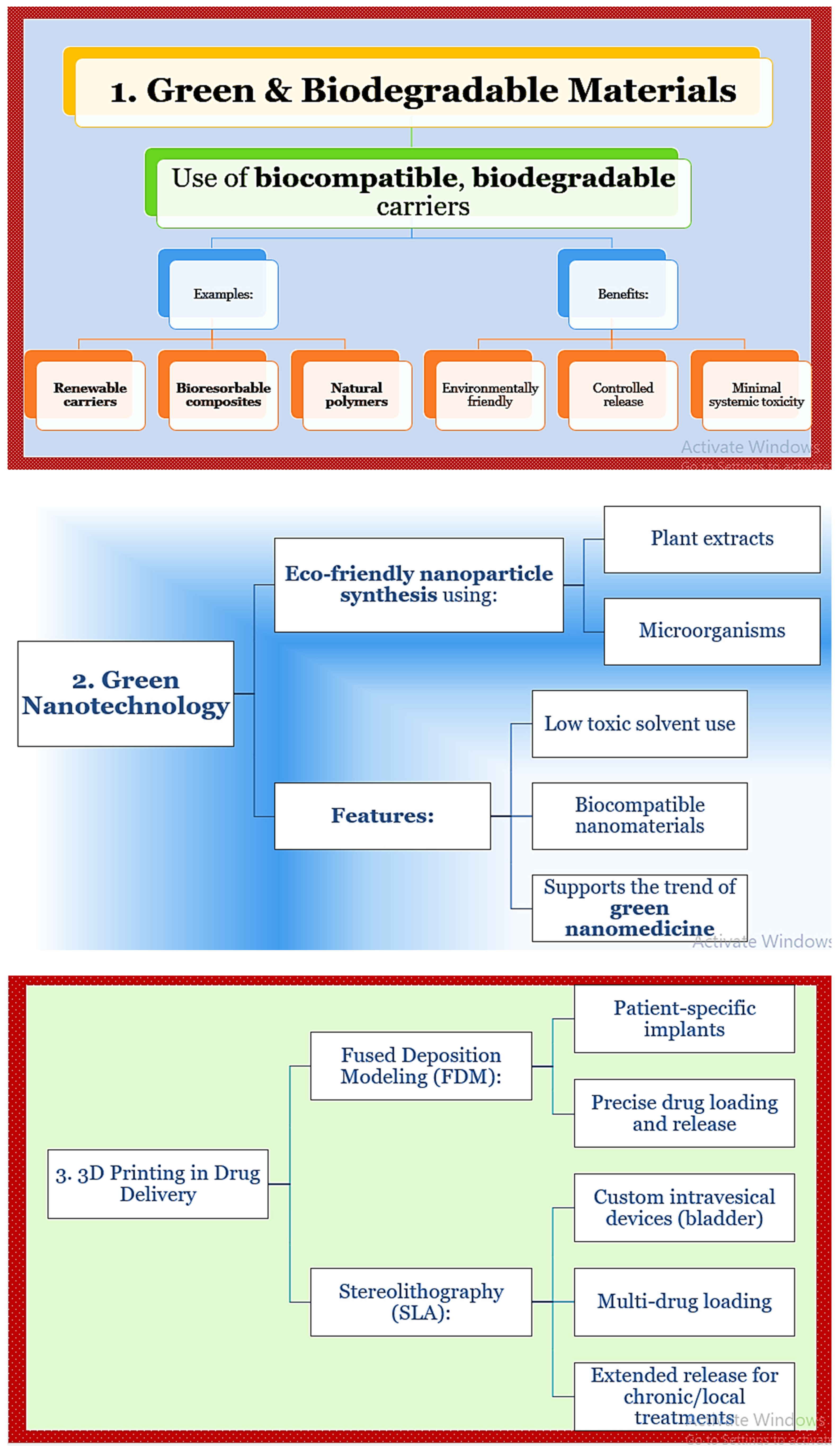
5. Role of AI and Smart Systems in Drug Targeting
| Category | Description |
|---|---|
| AI in Drug Delivery | AI facilitates data-driven therapeutic decisions, predicts pharmacokinetics, and optimizes nanocarrier design for enhanced specificity and reduced side effects. |
| Smart Nanocarriers | AI-designed nanocarriers are tailored for precise physicochemical and targeting properties, representing dynamic and responsive delivery platforms. |
| SORT Nanoparticles | Engineered to exploit organ-level biodistribution for tissue-specific delivery (e.g., mRNA to liver/lungs), enhancing efficacy and minimizing systemic toxicity. |
| Physical Targeting Techniques | Image-guided approaches like percutaneous ablation integrate with nanomedicine for localized drug delivery to tumors and lesions. |
6.3. D Bioprinting and Implantable Delivery Devices
Conclusions
References
- Sengar, A. (2025). Personalized medicine and nanotechnology: Transforming modern therapeutics. Preprints. [CrossRef]
- Sengar, A. (2025). Next-gen drug delivery: Redefining precision, bioavailability, and therapeutic outcomes. Preprints. [CrossRef]
- Sengar, A. (2025). Advancements in drug delivery systems: Targeting strategies, nanotechnology, and vesicular innovations. Preprints. [CrossRef]
- Sengar, A. (2025). Advancements in targeted drug delivery: Innovations in liposomal, nanoparticle, and vesicular systems. Preprints. [CrossRef]
- Noble, G. T., et al. (2014). Ligand-targeted liposome design: Challenges and fundamental considerations. Trends in Biotechnology, 32(1), 32–45. [CrossRef]
- Liu, Z., et al. (2024). Advancements in programmable lipid nanoparticles: Exploring the four-domain model for targeted drug delivery. arXiv. https://arxiv.org/abs/2408.05695.
- Sengar, A. (2025). The role of nanotechnology in revolutionizing cancer treatment. Preprints. [CrossRef]
- Sanchez-Moreno, P., et al. (2024). Smart drug-delivery systems for cancer nanotherapy. arXiv. https://arxiv.org/abs/2401.11192.
- Chacko, I. A., et al. (2020). Lipid vesicles: A versatile drug delivery platform for dermal and transdermal applications. Colloids and Surfaces B: Biointerfaces, 190, 110948. [CrossRef]
- Babaie, S., et al. (2020). Invasome: A novel nanocarrier for transdermal drug delivery. Nanomaterials, 10(5), 1–19.
- Jain, S., et al. (2021). Invasomes: Potential vesicular systems for transdermal delivery of drug molecules. JDDST, 61, 102265. [CrossRef]
- Paris, J. L., et al. (2021). Nanoparticles for multimodal antivascular therapeutics. arXiv. https://arxiv.org/abs/2103.09236.
- Kim, Y.-C., et al. (2012). Microneedles for drug and vaccine delivery. Advanced Drug Delivery Reviews, 64(14), 1547–1568. [CrossRef]
- Larrañeta, E., et al. (2016). Microneedles: A new frontier in nanomedicine delivery. Pharmaceutical Research, 33(5), 1055–1073. [CrossRef]
- El-Tokhy, F. S., et al. (2021). Design of long-acting invasomal nanovesicles. International Journal of Pharmaceutics, 603, 120679. [CrossRef]
- Teaima, M. H., et al. (2022). Propranolol mucoadhesive invasomes. Drug Delivery, 29(1), 1–14. [CrossRef]
- Souto, E. B., et al. (2021). Elastic and ultradeformable liposomes. International Journal of Molecular Sciences, 22(2), 974. [CrossRef]
- Sahu, T., et al. (2016). Skin cream as topical drug delivery system: A review. Journal of Pharmaceutical and Biological Sciences, 4(1), 1–9.
- Ashutosh, S. (2025). Sustainable drug delivery systems with biodegradable innovations. Preprints. [CrossRef]
- Noah, N., & Ndangili, P. (2021). Green synthesis of nanomaterials. arXiv. https://arxiv.org/abs/2112.04740.
- Kempin, W., et al. (2017). Polymers and drug loads for FDM 3D-printed implants. EJPB, 115, 84–93. [CrossRef]
- Goyanes, A., et al. (2015). SLA 3D printing of a bladder device. Materials Science and Engineering: C, 61, 931–938. [CrossRef]
- Sengar, A. (2025). Smart drug delivery: AI, nanotech & future innovations. Preprints. [CrossRef]
- Dilliard, S. A., et al. (2021). Tissue-specific mRNA delivery. PNAS, 118(52), e2109256118. [CrossRef]
- Ahmed, M., et al. (2011). Principles and advances in percutaneous ablation. Radiology, 258(2), 351–369. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




