Submitted:
30 April 2025
Posted:
02 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
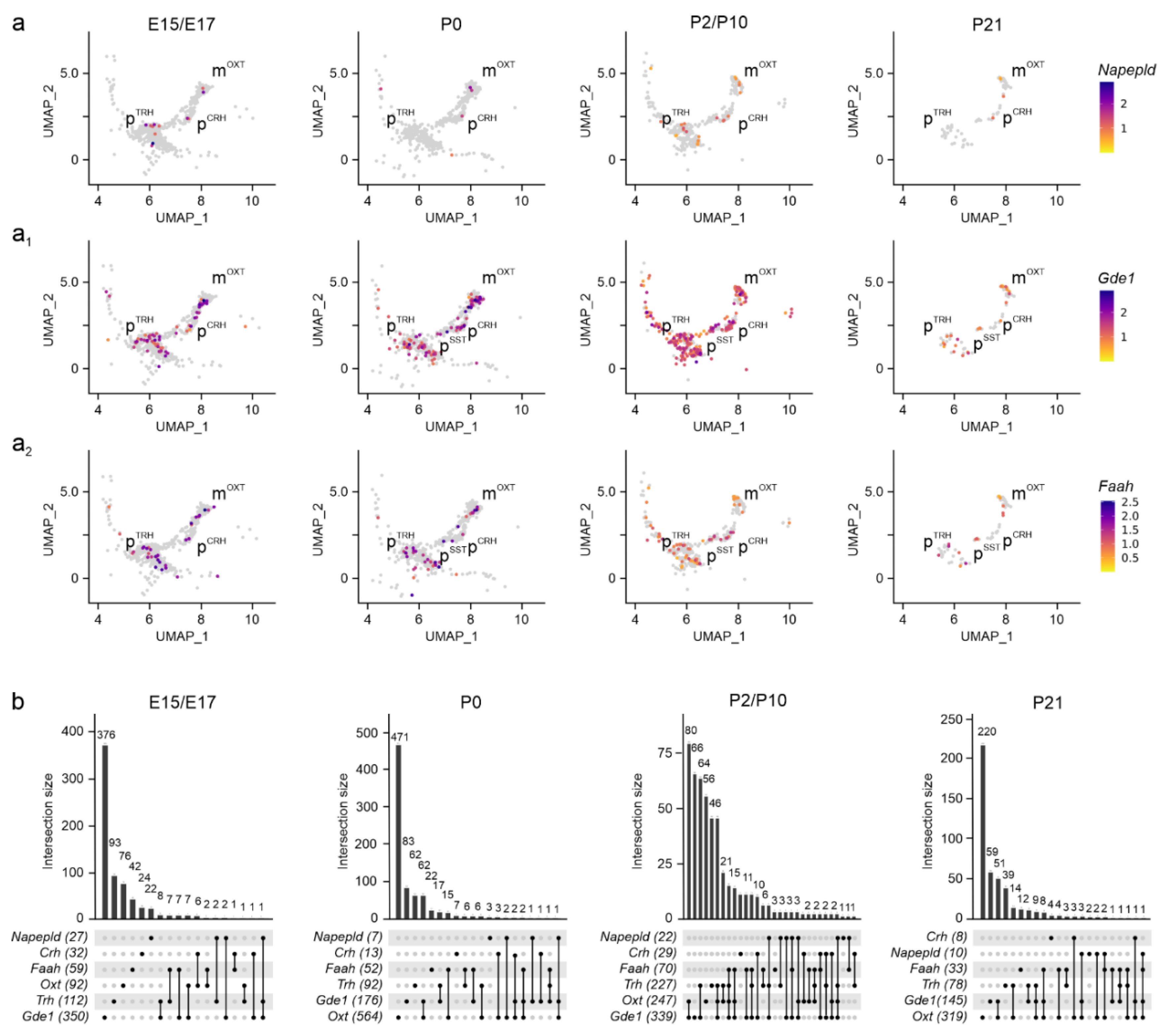
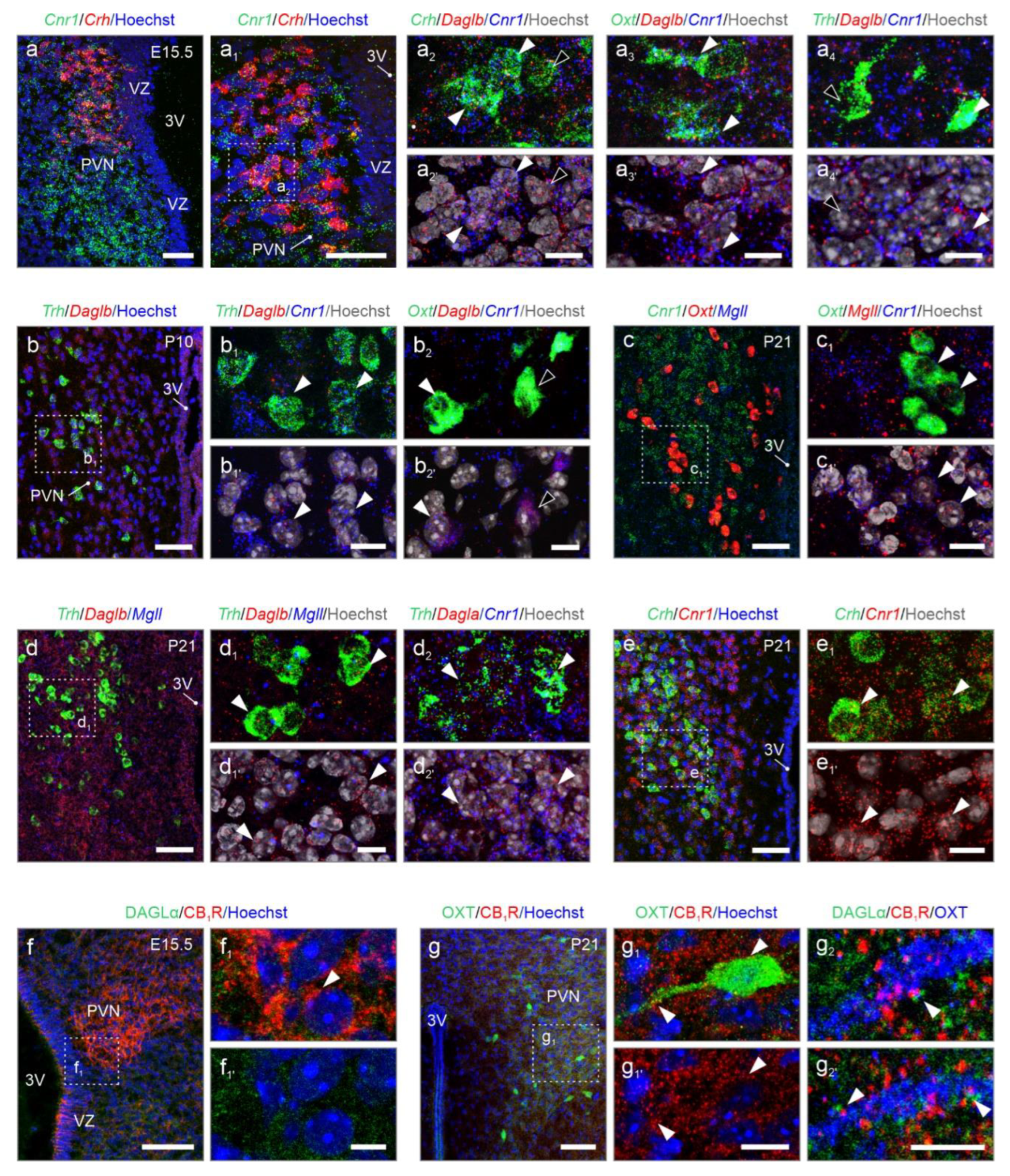
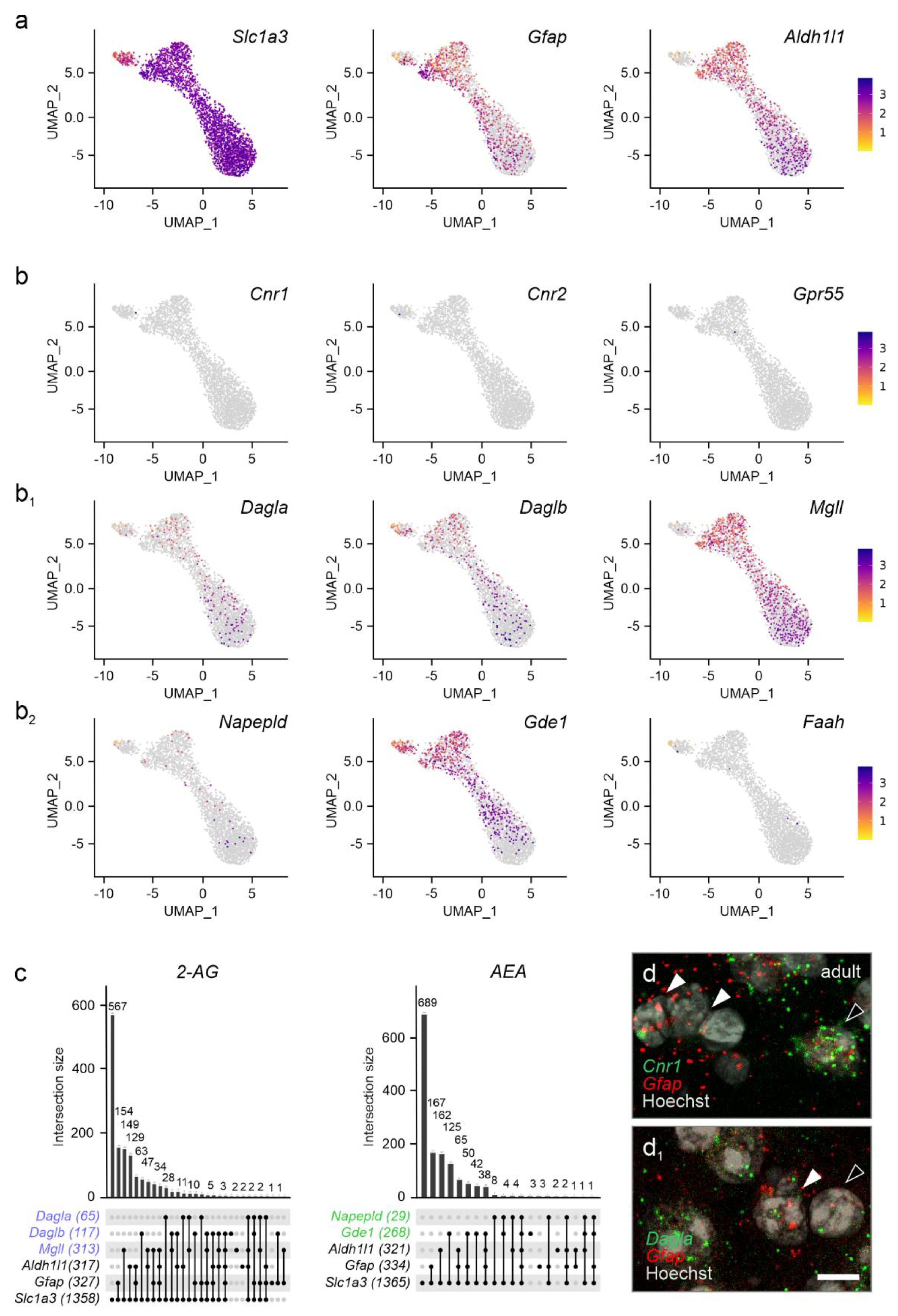
2. Results
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Code availability
Data availability
Competing interests statement
References
- Fong, H., Zheng, J. & Kurrasch, D. The structural and functional complexity of the integrative hypothalamus. Science 382, 388–394 (2023). [CrossRef]
- Qin, C., Li, J. & Tang, K. The Paraventricular Nucleus of the Hypothalamus: Development, Function, and Human Diseases. Endocrinology 159, 3458–3472 (2018). [CrossRef]
- Ben-Shlomo, A. & Melmed, S. Pituitary Somatostatin Receptor Signaling. Trends Endocrinol Metab 21, 123–133 (2010). [CrossRef]
- Wang, J. et al. Role and mechanism of PVN-sympathetic-adipose circuit in depression and insulin resistance induced by chronic stress. EMBO Rep 24, e57176 (2023). [CrossRef]
- Li, Y.-J. et al. Paraventricular nucleus-central amygdala oxytocinergic projection modulates pain-related anxiety-like behaviors in mice. CNS Neurosci Ther 29, 3493–3506 (2023). [CrossRef]
- Fenselau, H. et al. A rapidly acting glutamatergic ARC→PVH satiety circuit postsynaptically regulated by α-MSH. Nat Neurosci 20, 42–51 (2017). [CrossRef]
- Krashes, M. J., Shah, B. P., Koda, S. & Lowell, B. B. Rapid versus Delayed Stimulation of Feeding by the Endogenously Released AgRP Neuron Mediators GABA, NPY, and AgRP. Cell Metabolism 18, 588–595 (2013). [CrossRef]
- Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001). [CrossRef]
- Caron, A. et al. POMC neurons expressing leptin receptors coordinate metabolic responses to fasting via suppression of leptin levels. eLife 7, e33710. [CrossRef]
- Kondoh, K. et al. A specific area of olfactory cortex involved in stress hormone responses to predator odours. Nature 532, 103–106 (2016). [CrossRef]
- Kaouane, N., Ada, S., Hausleitner, M. & Haubensak, W. Dorsal Bed Nucleus of the Stria Terminalis-Subcortical Output Circuits Encode Positive Bias in Pavlovian Fear and Reward. Front Neural Circuits 15, 772512 (2021). [CrossRef]
- Decavel, C. & Van Den Pol, A. N. GABA: A dominant neurotransmitter in the hypothalamus. Journal of Comparative Neurology 302, 1019–1037 (1990). [CrossRef]
- Mendonça, M. M. et al. Involvement of GABAergic and Adrenergic Neurotransmissions on Paraventricular Nucleus of Hypothalamus in the Control of Cardiac Function. Front. Physiol. 9, (2018). [CrossRef]
- Li, Y.-F., Jackson, K. L., Stern, J. E., Rabeler, B. & Patel, K. P. Interaction between glutamate and GABA systems in the integration of sympathetic outflow by the paraventricular nucleus of the hypothalamus. American Journal of Physiology-Heart and Circulatory Physiology 291, H2847–H2856 (2006). [CrossRef]
- Romanov, R. A. et al. Molecular design of hypothalamus development. Nature 1–7 (2020). [CrossRef]
- Matsuda, L. A., Lolait, S. J., Brownstein, M. J., Young, A. C. & Bonner, T. I. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 346, 561–564 (1990). [CrossRef]
- Mechoulam, R. et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem. Pharmacol. 50, 83–90 (1995). [CrossRef]
- Stella, N., Schweitzer, P. & Piomelli, D. A second endogenous cannabinoid that modulates long-term potentiation. Nature 388, 773–778 (1997). [CrossRef]
- Kano, M., Ohno-Shosaku, T., Hashimotodani, Y., Uchigashima, M. & Watanabe, M. Endocannabinoid-mediated control of synaptic transmission. Physiol. Rev. 89, 309–380 (2009). [CrossRef]
- Cardinal, P. et al. Cannabinoid type 1 (CB1) receptors on Sim1-expressing neurons regulate energy expenditure in male mice. Endocrinology 156, 411–418 (2015). [CrossRef]
- Mazier, W. et al. mTORC1 and CB1 receptor signaling regulate excitatory glutamatergic inputs onto the hypothalamic paraventricular nucleus in response to energy availability. Mol Metab 28, 151–159 (2019). [CrossRef]
- Cruz-Martínez, A. M. et al. CB1 receptors in the paraventricular nucleus of the hypothalamus modulate the release of 5-HT and GABA to stimulate food intake in rats. Eur Neuropsychopharmacol 28, 1247–1259 (2018). [CrossRef]
- Petrie, G. N. et al. Disruption of tonic endocannabinoid signalling triggers cellular, behavioural and neuroendocrine responses consistent with a stress response. Br J Pharmacol 180, 3146–3159 (2023). [CrossRef]
- Gorzalka, B. B. & Hill, M. N. Integration of endocannabinoid signaling into the neural network regulating stress-induced activation of the hypothalamic-pituitary-adrenal axis. Curr Top Behav Neurosci 1, 289–306 (2009). [CrossRef]
- Tanimura, A. et al. The endocannabinoid 2-arachidonoylglycerol produced by diacylglycerol lipase alpha mediates retrograde suppression of synaptic transmission. Neuron 65, 320–327 (2010). [CrossRef]
- Navarrete, M. & Araque, A. Endocannabinoids Mediate Neuron-Astrocyte Communication. Neuron 57, 883–893 (2008). [CrossRef]
- Navarrete, M. & Araque, A. Endocannabinoids Potentiate Synaptic Transmission through Stimulation of Astrocytes. Neuron 68, 113–126 (2010). [CrossRef]
- Ivell, R. & Richter, D. Structure and comparison of the oxytocin and vasopressin genes from rat. Proceedings of the National Academy of Sciences 81, 2006–2010 (1984). [CrossRef]
- Kim, D. W. et al. The cellular and molecular landscape of hypothalamic patterning and differentiation from embryonic to late postnatal development. Nat Commun 11, 4360 (2020). [CrossRef]
- Munro, S., Thomas, K. L. & Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 365, 61–65 (1993). [CrossRef]
- Lauckner, J. E. et al. GPR55 is a cannabinoid receptor that increases intracellular calcium and inhibits M current. Proc. Natl. Acad. Sci. U.S.A. 105, 2699–2704 (2008). [CrossRef]
- Grabon, W. et al. CB2 expression in mouse brain: from mapping to regulation in microglia under inflammatory conditions. Journal of Neuroinflammation 21, 206 (2024). [CrossRef]
- Cherif, H. et al. Role of GPR55 during Axon Growth and Target Innervation. eNeuro 2, (2015). [CrossRef]
- Harkany, T., Mackie, K. & Doherty, P. Wiring and firing neuronal networks: endocannabinoids take center stage. Curr Opin Neurobiol 18, 338–345 (2008). [CrossRef]
- Bisogno, T. et al. Cloning of the first sn1-DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain. J. Cell Biol. 163, 463–468 (2003). [CrossRef]
- Dinh, T. P. et al. Brain monoglyceride lipase participating in endocannabinoid inactivation. Proc Natl Acad Sci U S A 99, 10819–10824 (2002). [CrossRef]
- Oudin, M. J., Hobbs, C. & Doherty, P. DAGL-dependent endocannabinoid signalling: roles in axonal pathfinding, synaptic plasticity and adult neurogenesis. Eur. J. Neurosci. 34, 1634–1646 (2011). [CrossRef]
- Mulder, J. et al. Endocannabinoid signaling controls pyramidal cell specification and long-range axon patterning. Proc. Natl. Acad. Sci. U.S.A. 105, 8760–8765 (2008). [CrossRef]
- Keimpema, E. et al. Diacylglycerol lipase alpha manipulation reveals developmental roles for intercellular endocannabinoid signaling. Scientific reports 3, 2093 (2013). [CrossRef]
- Okamoto, Y., Morishita, J., Tsuboi, K., Tonai, T. & Ueda, N. Molecular Characterization of a Phospholipase D Generating Anandamide and Its Congeners*. Journal of Biological Chemistry 279, 5298–5305 (2004). [CrossRef]
- Leishman, E., Mackie, K., Luquet, S. & Bradshaw, H. B. Lipidomics profile of a NAPE-PLD KO mouse provides evidence of a broader role of this enzyme in lipid metabolism in the brain. Biochim Biophys Acta 1861, 491–500 (2016). [CrossRef]
- Simon, G. M. & Cravatt, B. F. Anandamide Biosynthesis Catalyzed by the Phosphodiesterase GDE1 and Detection of Glycerophospho-N-acyl Ethanolamine Precursors in Mouse Brain*. Journal of Biological Chemistry 283, 9341–9349 (2008). [CrossRef]
- Deutsch, D. G. & Chin, S. A. Enzymatic synthesis and degradation of anandamide, a cannabinoid receptor agonist. Biochem Pharmacol 46, 791–796 (1993). [CrossRef]
- Hill, M. N. & Tasker, J. G. Endocannabinoid Signaling, Glucocorticoid-Mediated Negative Feedback and Regulation of the HPA Axis. Neuroscience 204, 5–16 (2012). [CrossRef]
- Coiret, G. et al. Neuron to astrocyte communication via cannabinoid receptors is necessary for sustained epileptiform activity in rat hippocampus. PLoS One 7, e37320 (2012). [CrossRef]
- Eraso-Pichot, A. et al. Endocannabinoid signaling in astrocytes. Glia 71, 44–59 (2023). [CrossRef]
- Hebert-Chatelain, E. et al. A cannabinoid link between mitochondria and memory. Nature 539, 555–559 (2016). [CrossRef]
- Akdemir, E. S., Huang, A. Y.-S. & Deneen, B. Astrocytogenesis: where, when, and how. F1000Res 9, F1000 Faculty Rev-233 (2020). [CrossRef]
- Rothstein, J. D. et al. Localization of neuronal and glial glutamate transporters. Neuron 13, 713–725 (1994). [CrossRef]
- Jacque, C. M. et al. Determination of glial fibrillary acidic protein (GFAP) in human brain tumors. Journal of the Neurological Sciences 35, 147–155 (1978). [CrossRef]
- Cahoy, J. D. et al. A Transcriptome Database for Astrocytes, Neurons, and Oligodendrocytes: A New Resource for Understanding Brain Development and Function. J. Neurosci. 28, 264–278 (2008). [CrossRef]
- Bosier, B. et al. Astroglial CB1 cannabinoid receptors regulate leptin signaling in mouse brain astrocytes. Molecular Metabolism 2, 393–404 (2013). [CrossRef]
- Jimenez-Blasco, D. et al. Glucose metabolism links astroglial mitochondria to cannabinoid effects. Nature 583, 603–608 (2020). [CrossRef]
- Dudek, K. A. et al. Astrocytic cannabinoid receptor 1 promotes resilience by dampening stress-induced blood-brain barrier alterations. Nat Neurosci 28, 766–782 (2025). [CrossRef]
- Moro Chao, D. H. et al. Hypothalamic astrocytes control systemic glucose metabolism and energy balance. Cell Metab 34, 1532-1547.e6 (2022). [CrossRef]
- Begbie, J., Doherty, P. & Graham, A. Cannabinoid receptor, CB1, expression follows neuronal differentiation in the early chick embryo. J Anat 205, 213–218 (2004). [CrossRef]
- Gao, Y. et al. Loss of Retrograde Endocannabinoid Signaling and Reduced Adult Neurogenesis in Diacylglycerol Lipase Knock-out Mice. J. Neurosci. 30, 2017–2024 (2010). [CrossRef]
- Zaia, C. T. B. V. et al. Vasoactive intestinal peptide promotes hypophagia and metabolic changes: Role of paraventricular hypothalamic nucleus and nitric oxide. Brain Research Bulletin 189, 102–110 (2022). [CrossRef]
- Abtahi, S., Mirza, A., Howell, E. & Currie, P. J. Ghrelin enhances food intake and carbohydrate oxidation in a nitric oxide dependent manner. General and Comparative Endocrinology 250, 9–14 (2017). [CrossRef]
- Zhang, K., Mayhan, W. G. & Patel, K. P. Nitric oxide within the paraventricular nucleus mediates changes in renal sympathetic nerve activity. Am J Physiol 273, R864-872 (1997). [CrossRef]
- McBryde, F. D., Liu, B. H., Roloff, E. V., Kasparov, S. & Paton, J. F. R. Hypothalamic paraventricular nucleus neuronal nitric oxide synthase activity is a major determinant of renal sympathetic discharge in conscious Wistar rats. Experimental Physiology 103, 419–428 (2018). [CrossRef]
- Yamaguchi, N., Okada, S., Usui, D. & Yokotani, K. Nitric oxide synthase isozymes in spinally projecting PVN neurons are involved in CRF-induced sympathetic activation. Autonomic Neuroscience 148, 83–89 (2009). [CrossRef]
- Savinainen, J. R., Saario, S. M. & Laitinen, J. T. The serine hydrolases MAGL, ABHD6 and ABHD12 as guardians of 2-arachidonoylglycerol signalling through cannabinoid receptors. Acta Physiol (Oxf) 204, 267–276 (2012). [CrossRef]
- Marrs, W. R. et al. The serine hydrolase ABHD6 controls the accumulation and efficacy of 2-AG at cannabinoid receptors. Nat Neurosci 13, 951–957 (2010). [CrossRef]
- Blankman, J. L., Simon, G. M. & Cravatt, B. F. A Comprehensive Profile of Brain Enzymes that Hydrolyze the Endocannabinoid 2-Arachidonoylglycerol. Chemistry & Biology 14, 1347–1356 (2007). [CrossRef]
- Maccarrone, M. Metabolism of the Endocannabinoid Anandamide: Open Questions after 25 Years. Front. Mol. Neurosci. 10, (2017). [CrossRef]
- Simard, M. et al. Biosynthesis and metabolism of endocannabinoids and their congeners from the monoacylglycerol and N-acyl-ethanolamine families. Biochemical Pharmacology 205, 115261 (2022). [CrossRef]
- Simon, G. M. & Cravatt, B. F. Characterization of mice lacking candidate N-acyl ethanolamine biosynthetic enzymes provides evidence for multiple pathways that contribute to endocannabinoid production in vivo. Mol. BioSyst. 6, 1411–1418 (2010). [CrossRef]
- Buczynski, M. W. & Parsons, L. H. Quantification of brain endocannabinoid levels: methods, interpretations and pitfalls. Br J Pharmacol 160, 423–442 (2010). [CrossRef]
- Ohno-Shosaku, T. & Kano, M. Endocannabinoid-mediated retrograde modulation of synaptic transmission. Current Opinion in Neurobiology 29, 1–8 (2014). [CrossRef]
- Savinainen, J. R., Järvinen, T., Laine, K. & Laitinen, J. T. Despite substantial degradation, 2-arachidonoylglycerol is a potent full efficacy agonist mediating CB1 receptor-dependent G-protein activation in rat cerebellar membranes. British Journal of Pharmacology 134, 664–672 (2001). [CrossRef]
- Chapman, C. D. et al. Paraventricular nucleus anandamide signaling alters eating and substrate oxidation. Neuroreport 23, 425–429 (2012). [CrossRef]
- Abruzzese, F., Greco, M., Perlino, E., Doonan, S. & Marra, E. Lack of correlation between mRNA expression and enzymatic activity of the aspartate aminotransferase isoenzymes in various tissues of the rat. FEBS Lett 366, 170–172 (1995). [CrossRef]
- Walter, L., Dinh, T. & Stella, N. ATP induces a rapid and pronounced increase in 2-arachidonoylglycerol production by astrocytes, a response limited by monoacylglycerol lipase. J Neurosci 24, 8068–8074 (2004). [CrossRef]
- Virshup, I., Rybakov, S., Theis, F. J., Angerer, P. & Wolf, F. A. anndata: Access and store annotated datamatrices. JOSS 9, 4371 (2024). [CrossRef]
- Blischak, J., Carbonetto, P. & Stephens, M. Creating and Sharing Reproducible Research Code the Workflowr Way. F1000research (2019). [CrossRef]
- Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell (2021). [CrossRef]
- McInnes, L., Healy, J., Saul, N. & Großberger, L. UMAP: Uniform Manifold Approximation and Projection. JOSS 3, 861 (2018). [CrossRef]
- Kobak, D. & Linderman, G. C. Initialization is critical for preserving global data structure in both t-SNE and UMAP. Nat Biotechnol 39, 156–157 (2021). [CrossRef]
- Zupančič, M. et al. Concerted transcriptional regulation of the morphogenesis of hypothalamic neurons by ONECUT3. Nat Commun 15, 8631 (2024). [CrossRef]
- Simmons, D. M. & Swanson, L. W. Comparison of the spatial distribution of seven types of neuroendocrine neurons in the rat paraventricular nucleus: toward a global 3D model. J Comp Neurol 516, 423–441 (2009). [CrossRef]
- Romanov, R. A. & Harkany, T. Neuronal heterogeneity in the paraventricular nucleus of the hypothalamus as revealed by single-cell RNA-seq. Current Opinion in Endocrine and Metabolic Research 25, 100366 (2022). [CrossRef]
- Keimpema, E., Di Marzo, V. & Harkany, T. Biological basis of cannabinoid medicines. Science 374, 1449–1450 (2021). [CrossRef]
- Maccarrone, M., Guzmán, M., Mackie, K., Doherty, P. & Harkany, T. Programming of neural cells by (endo)cannabinoids: from physiological rules to emerging therapies. Nat Rev Neurosci 15, 786–801 (2014). [CrossRef]
- Conway, J. R., Lex, A. & Gehlenborg, N. UpSetR: an R package for the visualization of intersecting sets and their properties. Bioinformatics 33, 2938–2940 (2017). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



