Submitted:
27 April 2025
Posted:
28 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
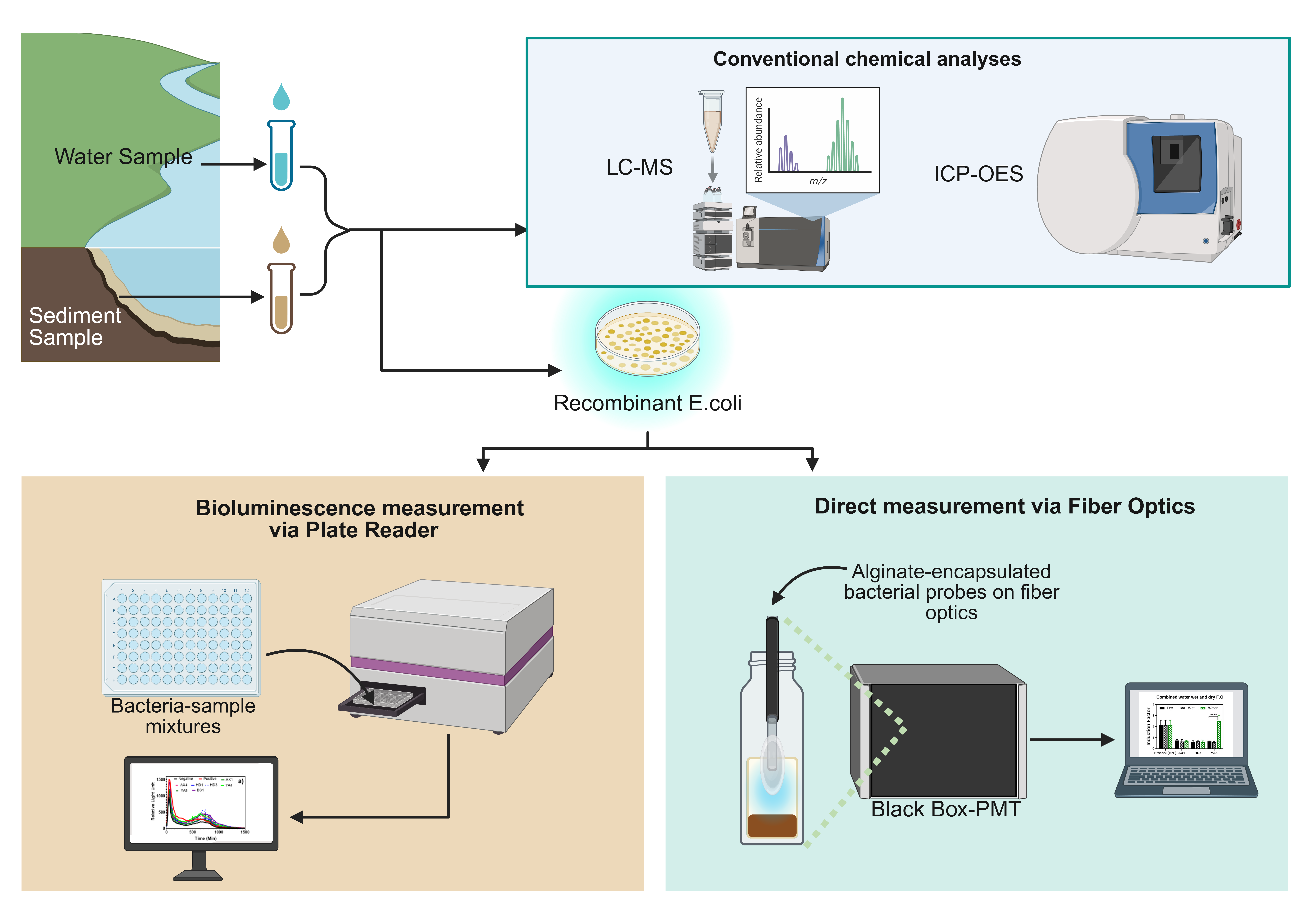
2. Materials and Methods
2.1. Materials
2.2. Sample Collection
2.3. Bacterial Toxicity Measurement
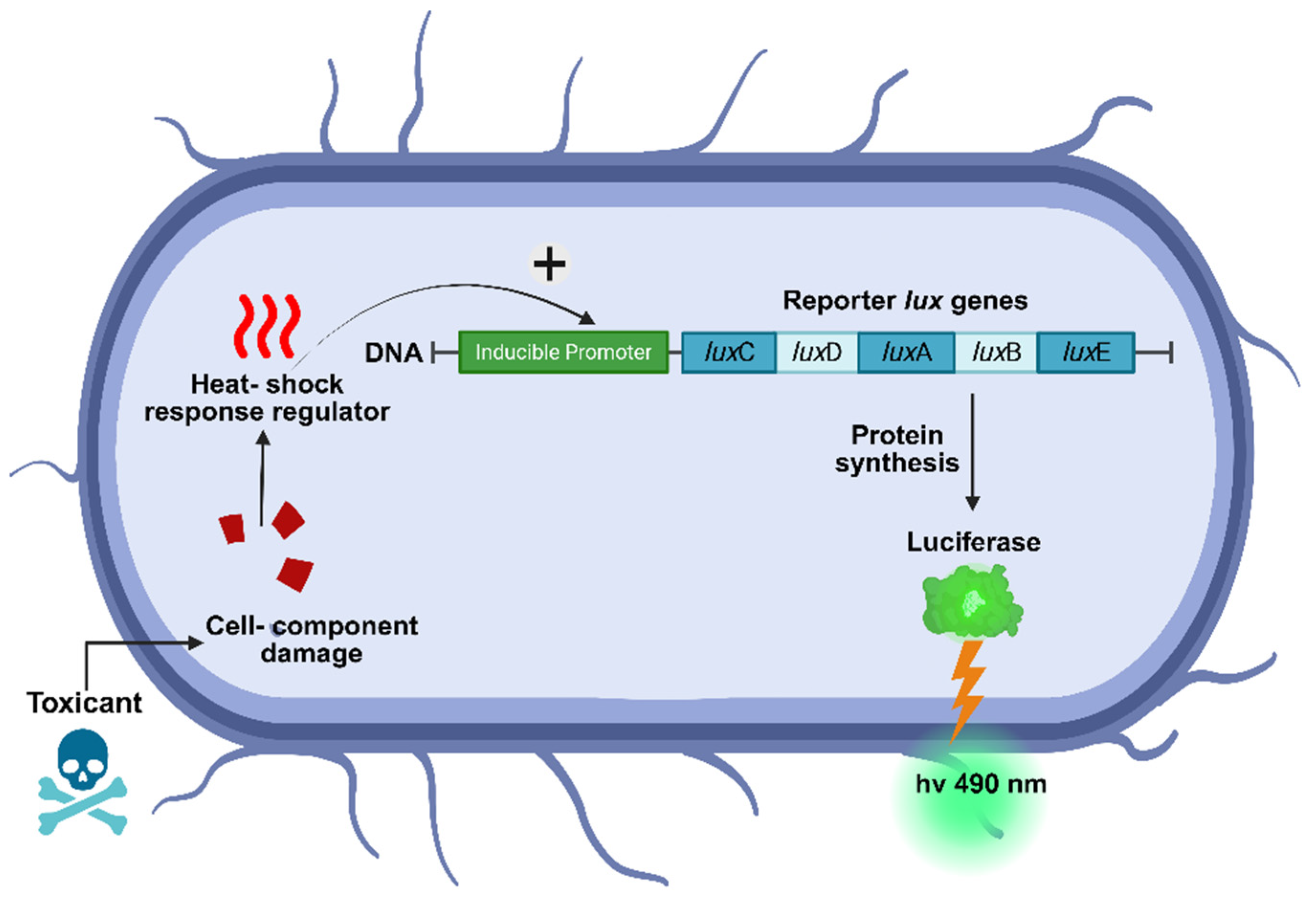
2.3.1. Strain Description
2.3.2. Culturing Conditions
2.3.3. Preparation of Bacterial Suspensions for Immobilization
2.3.4. Sample Collection and Preparation for Analysis
2.3.5. Bioluminescence Measurement Using Multimode Plate Reader
2.3.6. Immobilization of the Bioluminescent Bacteria onto the Optical Fiber
2.3.7. Fiber Optic Instrument Set-Up
2.3.8. Fiber Optic Bioluminescent Toxicity Acquisition
2.4. Conventional Chemical Analyses
2.4.1. Elemental Analysis by ICP-OES
2.4.2. LC-MS Analysis
2.4. Statistical Analysis
3. Results
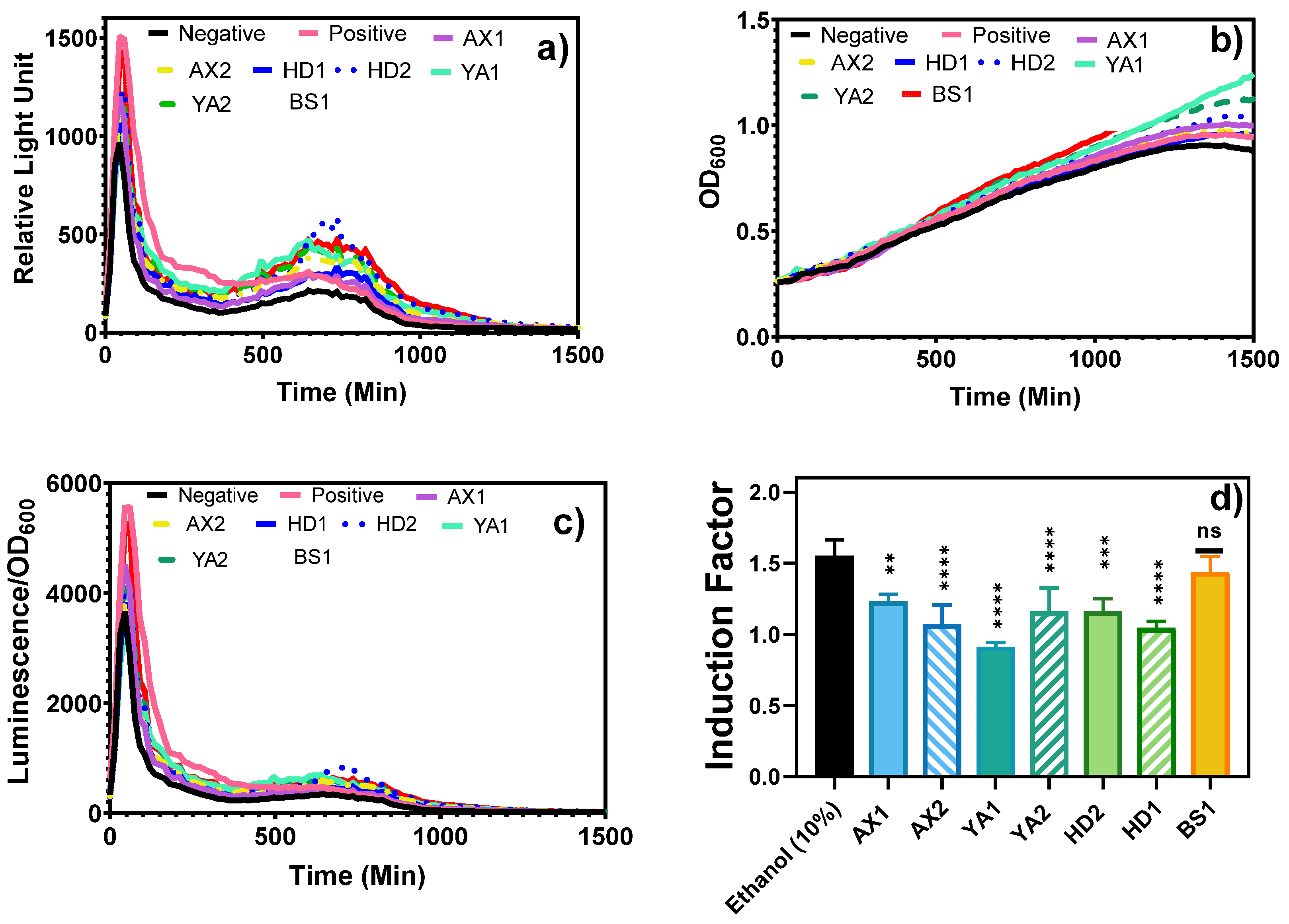
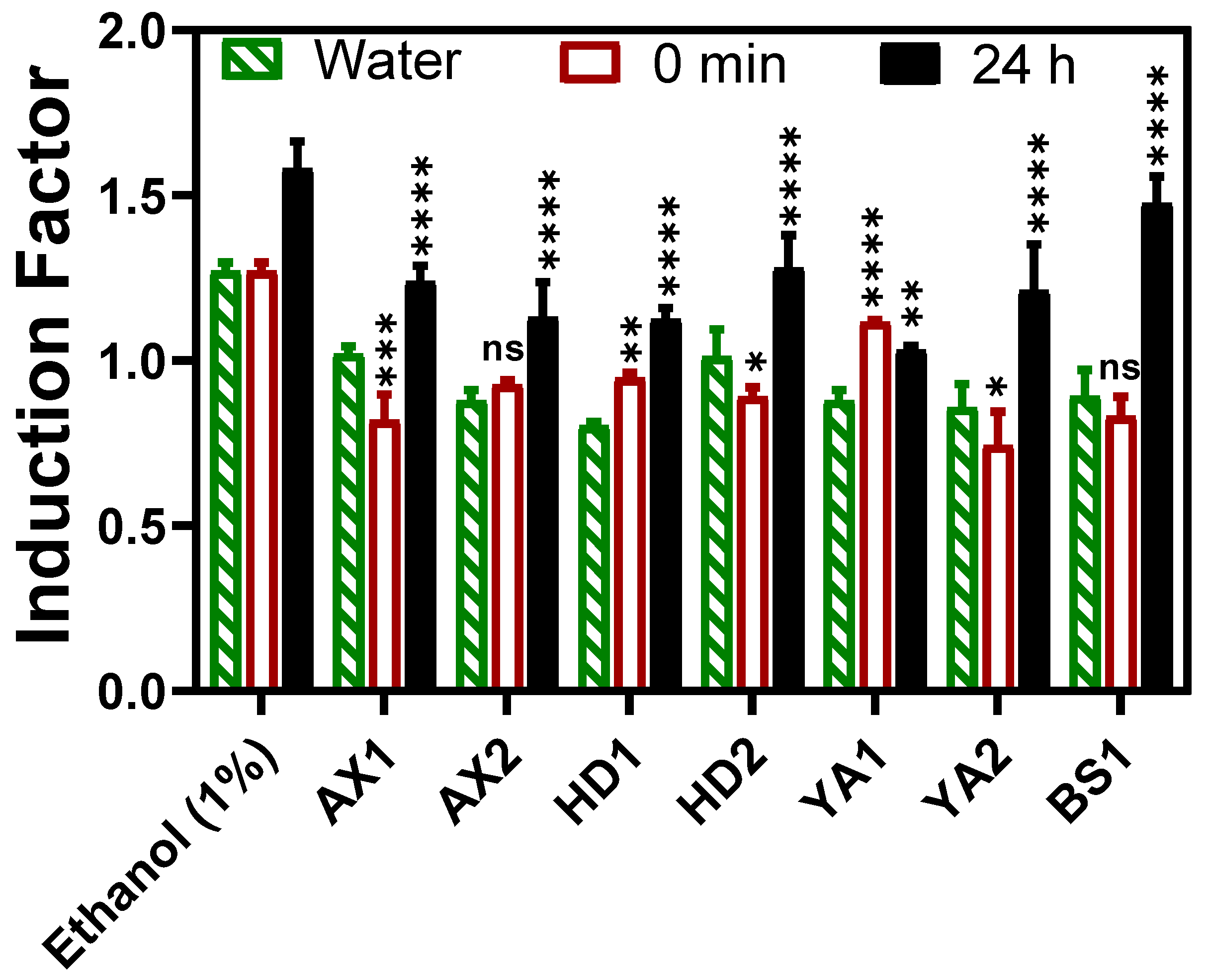
3.1. Bioluminescent Toxicity Response
3.1.1. The Fiber Optic-Based Bacterial Toxicity Measurement
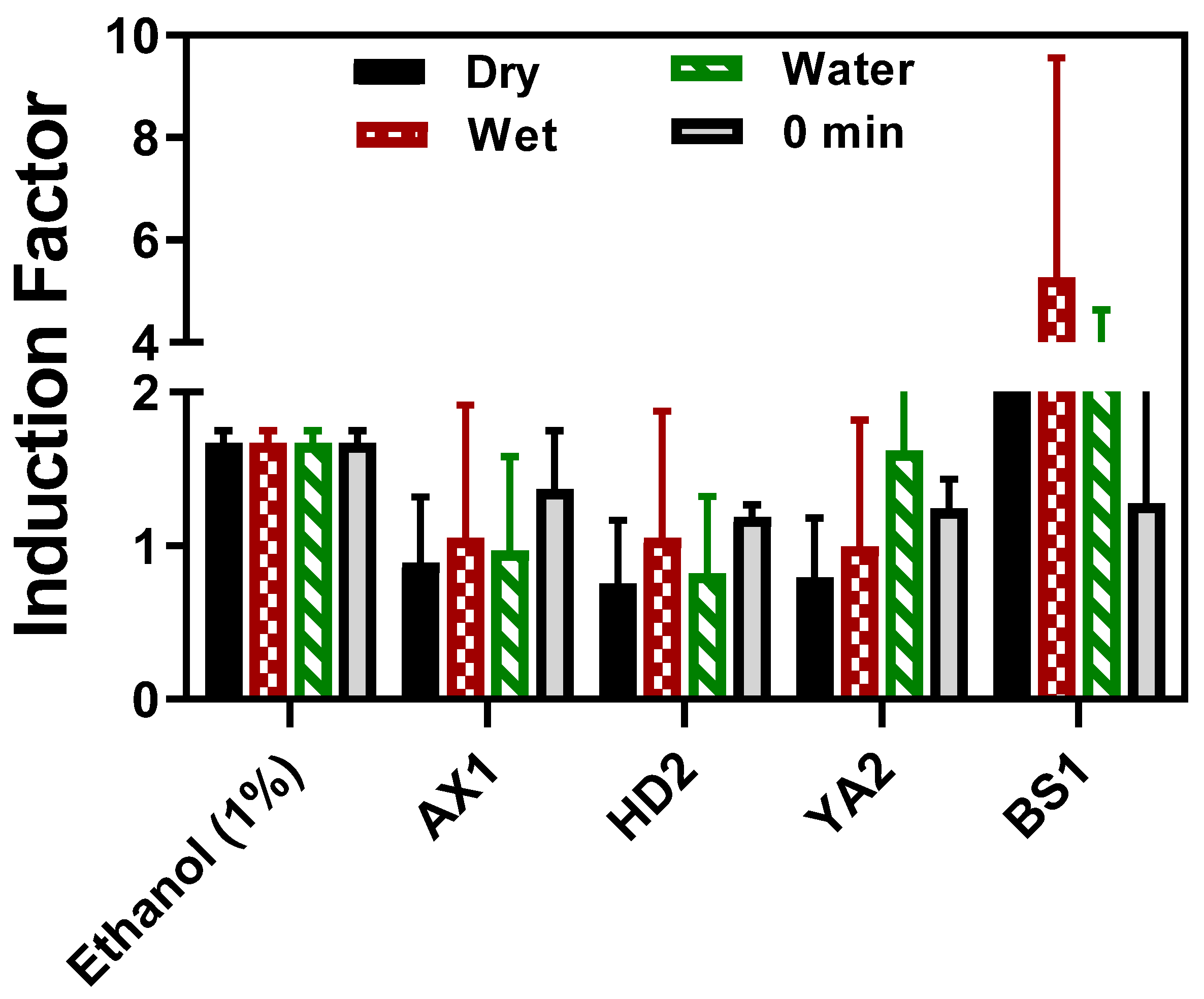
3.1.2. The Laboratory-Based Toxicity Response
3.2. Conventional Analysis
3.2.1. ICP Analysis
| Element | HD1 | AX2 | YA1 | YA2 |
|---|---|---|---|---|
| Ag | - | - | - | - |
| Al | - | - | - | - |
| B | - | + | - | - |
| Ba | - | - | - | - |
| Bi | - | - | - | - |
| Ca | + | - | + | - |
| Cd | - | - | - | - |
| Co | - | - | - | - |
| Cr | - | - | - | - |
| Cu | - | - | - | - |
| Fe | - | - | - | - |
| Ga | - | - | - | - |
| K | + | + | + | + |
| Li | - | - | - | - |
| Mg | + | + | + | - |
| Mn | - | - | - | - |
| Na | + | + | + | + |
| Ni | - | - | - | - |
| Pb | - | - | - | - |
| Sr | - | - | - | - |
| Zn | - | - | - | - |
| S | + | + | + | + |
| P | - | - | - | - |
3.2.2. LC-MS Analysis

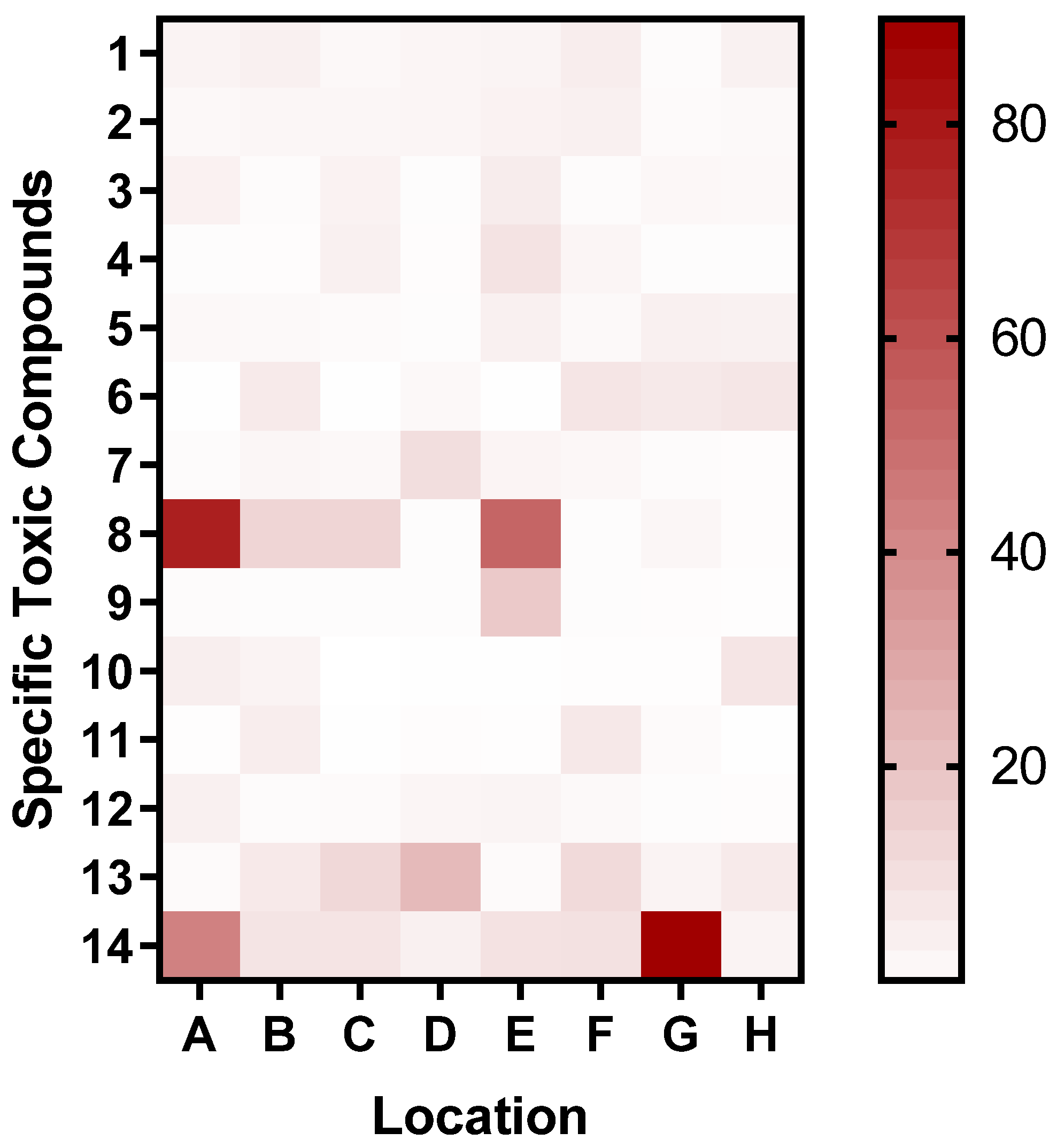
| Number | Name | References |
|---|---|---|
| 1 | Bis(2-ethylhexyl) amine | [32] |
| 2 | Chlorphentermine | [33,34,35] |
| 3 | Cyclohexylamine | [36,37,38,39] |
| 4 | Dichloromethane | [40,41,42] |
| 5 | Dicyclohexylamine | [43,44,45] |
| 6 | Diethanolamine | [46,47,48,49] |
| 7 | Ethephon | [50,51,52] |
| 8 | MDMA | [53,54,55] |
| 9 | N,N-Dimethylacetamide | [56,57,58] |
| 10 | N-ethylmaleimide | [59,60,61] |
| 11 | o-Toluidine | [62,63,64,65] |
| 12 | Phentermine | [66,67,68,69] |
| 13 | Phenylethyl alcohol | [70,71,72] |
| 14 | Triethanolamine | [46,48,73,74,75] |
| Element | Compounds in water | Proportion in water (%) | Compounds in sediment | Proportion in sediment (%) |
|---|---|---|---|---|
| Sulfur | 299 | 40.13 | 154 | 14.41 |
| Chlorine | 279 | 37.45 | 35 | 3.27 |
| Bromine | 110 | 14.77 | 7 | 0.65 |
| Fluorine | 3 | 0.4 | 6 | 0.56 |
| Iodine | 2 | 0.27 | 1 | 0.09 |
| Selenium | 1 | 0.13 | 1 | 0.09 |
| Indium | 1 | 0.13 | 0 | 0 |
| Phosphorus | 422 | 56.64 | 198 | 18.52 |
| Nitrogen | 497 | 66.71 | 862 | 80.64 |
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Baat, M.L.; Wieringa, N.; Droge, S.T.; van Hall, B.G.; van der Meer, F.; Kraak, M.H.J.E.s.; technology. Smarter sediment screening: effect-based quality assessment, chemical profiling, and risk identification. 2019, 53, 14479-14488.
- Burton Jr, G.A.J.E.t.; chemistry. Assessing sediment toxicity: past, present, and future. 2013, 32, 1438-1440.
- Massei, R.; Hollert, H.; Krauss, M.; Von Tümpling, W.; Weidauer, C.; Haglund, P.; Küster, E.; Gallampois, C.; Tysklind, M.; Brack, W.J.E.S.E. Toxicity and neurotoxicity profiling of contaminated sediments from Gulf of Bothnia (Sweden): a multi-endpoint assay with Zebrafish embryos. 2019, 31, 1-12. [CrossRef]
- Borja, A.; Valencia, V.; Franco, J.; Muxika, I.; Bald, J.; Belzunce, M.; Solaun, O.J.M.P.B. The water framework directive: water alone, or in association with sediment and biota, in determining quality standards? 2004, 49, 8-11. [CrossRef]
- Den Besten, P.J.; De Deckere, E.; Babut, M.P.; Power, B.; DelValls, T.A.; Zago, C.; Oen, A.M.; Heise, S.J.J.o.s.; sediments. Biological effects-based sediment quality in ecological risk assessment for European waters. 2003, 3, 144-162. [CrossRef]
- Ivask, A.; Green, T.; Polyak, B.; Mor, A.; Kahru, A.; Virta, M.; Marks, R. Fibre-optic bacterial biosensors and their application for the analysis of bioavailable Hg and As in soils and sediments from Aznalcollar mining area in Spain. Biosensors and Bioelectronics 2007, 22, 1396-1402. [CrossRef]
- Eltzov, E.; Marks, R.S. Whole-cell aquatic biosensors. Analytical and Bioanalytical Chemistry 2011, 400, 895-913. [CrossRef]
- Anderson, B.S.; Hunt, J.W.; Phillips, B.M.; Fairey, R.; Roberts, C.A.; Oakden, J.M.; Puckett, H.M.; Stephenson, M.; Tjeerdema, R.S.; Long, E.R.J.E.T.; et al. Sediment quality in Los Angeles Harbor, USA: A triad assessment. 2001, 20, 359-370. [CrossRef]
- Jonker, M.T.; Diepens, N.J.J.E.T.; Chemistry. Improving sediment toxicity testing for very hydrophobic chemicals: Part 1—Spiking, equilibrating, and exposure quantification. 2024, 43, 1717-1727.
- Daunert, S.; Barrett, G.; Feliciano, J.S.; Shetty, R.S.; Shrestha, S.; Smith-Spencer, W.J.C.r. Genetically engineered whole-cell sensing systems: coupling biological recognition with reporter genes. 2000, 100, 2705-2738. [CrossRef]
- Trif, C.; Vunduk, J.; Parcharoen, Y.; Bualuang, A.; Marks, R.S.J.B. Bioluminescent Whole-Cell Bioreporter Bacterial Panel for Sustainable Screening and Discovery of Bioactive Compounds Derived from Mushrooms. 2024, 14, 558. [CrossRef]
- Eltzov, E.; Marks, R.S.J.I.i.; magazine, m. Parameters to consider in the construction of fiber-optic biosensors as alternative bioanalytical tools. 2009, 12, 10-16.
- Polyak, B.; Bassis, E.; Novodvorets, A.; Belkin, S.; Marks, R.S. Bioluminescent whole cell optical fiber sensor to genotoxicants: system optimization. Sensors and Actuators B: Chemical 2001, 74, 18-26. [CrossRef]
- Trif, C.; Vunduk, J.; Parcharoen, Y.; Bualuang, A.; Marks, R.S. Bioluminescent Whole-Cell Bioreporter Bacterial Panel for Sustainable Screening and Discovery of Bioactive Compounds Derived from Mushrooms. Biosensors 2024, 14, 558. [CrossRef]
- Lior, E.; Axelrod, T.; Eltzov, E.; Kushmaro, A.; Marks, R.S. Lachish river event monitored for toxicity using bioluminescent reporter organisms. The EuroBiotech Journal 2018, 2, 47-58. [CrossRef]
- Meighen, E.A.J.T.F.j. Bacterial bioluminescence: organization, regulation, and application of the lux genes. 1993, 7, 1016-1022.
- Kronfeld, J.; Navrot, J. Aspects of trace metal contamination in the coastal rivers of Israel. Water, Air, and Soil Pollution 1975, 4, 127-134. [CrossRef]
- Gitsis, A. Ha-yaʿad ha-mafṭīʾ shel ha-kharishim: Naḥal Alexander. N12 (mako) 2025/04/16 2025.
- Asaf, L.; Negaoker, N.; Tal, A.; Laronne, J.; Khateeb, N.A. Transboundary stream restoration in Israel and the Palestinian Authority. In Integrated water resources management and security in the Middle East; Springer: 2007; pp. 285-295.
- Van Dyk, T.K.; Majarian, W.R.; Konstantinov, K.B.; Young, R.M.; Dhurjati, P.S.; LaRossa, R.A. Rapid and sensitive pollutant detection by induction of heat shock gene-bioluminescence gene fusions. Applied and environmental microbiology 1994, 60, 1414-1420. [CrossRef]
- Meighen, E.A. Bacterial bioluminescence: organization, regulation, and application of the lux genes. The FASEB Journal 1993, 7, 1016-1022. [CrossRef]
- Elsemore, D.A. Insertion of promoter region::luxCDABE fusions into the Escherichia coli chromosome. Methods in molecular biology (Clifton, N.J.) 1998, 102, 97-104. [CrossRef]
- Arsène, F.; Tomoyasu, T.; Bukau, B. The heat shock response of Escherichia coli. International journal of food microbiology 2000, 55, 3-9. [CrossRef]
- Bazhenov, S.; Novoyatlova, U.; Scheglova, E.; Prazdnova, E.; Mazanko, M.; Kessenikh, A.; Kononchuk, O.; Gnuchikh, E.; Liu, Y.; Al Ebrahim, R.J.B.; et al. Bacterial lux-biosensors: Constructing, applications, and prospects. 2023, 13, 100323. [CrossRef]
- Eltzov, E.; Prilutsky, D.; Kushmaro, A.; Marks, R.; Geddes, C. Metal-enhanced bioluminescence: An approach for monitoring biological luminescent processes. Applied Physics Letters 2009, 94, 083901-083901. [CrossRef]
- Polyak, B.; Bassis, E.; Novodvorets, A.; Belkin, S.; Marks, R.S. Optical fiber bioluminescent whole-cell microbial biosensors to genotoxicants. Water Science and Technology 2000, 42, 305-311. [CrossRef]
- Paul, A.A.; Kadosh, Y.S.; Kushmaro, A.; Marks, R.S.J.B. Microbead-Encapsulated Luminescent Bioreporter Screening of P. aeruginosa via Its Secreted Quorum-Sensing Molecules. 2024, 14, 383.
- Matsunaga, T.; Sudo, H.; Takemasa, H.; Wachi, Y.; Nakamura, N. Sulfated extracellular polysaccharide production by the halophilic cyanobacterium Aphanocapsa halophytia immobilized on light-diffusing optical fibers. Applied Microbiology and Biotechnology 1996, 45, 24-27. [CrossRef]
- Carletti, M.; Sforza, E.; Batushansky, A.; Boussiba, S.; Bertucco, A.; Khozin-Goldberg, I.; Zorin, B.J.B.T. Enhanced cyanophycin accumulation in diazotrophic cyanobacterium through random mutagenesis and tailored selection under varying phosphorus availability. 2025, 419, 132018. [CrossRef]
- Team, R.D.C.J. R: A language and environment for statistical computing. 2010.
- Kolde, R.J.I.o.H.t.O.M.C.o.D.; Appearance. Package “pheatmap”: Pretty heatmaps. 2018.
- Material Safety Data Sheet: Di-(2-ethylhexyl)amine; Parchem, 415 Huguenot Street, New Rochelle, NY 10801: New Rochelle, NY, 2009/07/14 2009.
- Caldwell, J. Amphetamines and related stimulants: some introductory remarks. In Amphetamines and Related Stimulants; CRC Press: 2019; pp. 1-12.
- Mitchell, S.C.; Waring, R.H.; Smith, R.L. Curiosities in drug metabolism. Xenobiotica 2014, 44, 666-676. [CrossRef]
- Xia, Z.; Ying, G.; Hansson, A.L.; Karlsson, H.; Xie, Y.; Bergstrand, A.; DePierre, J.W.; Nässberger, L. Antidepressant-induced lipidosis with special reference to tricyclic compounds. Progress in neurobiology 2000, 60, 501-512. [CrossRef]
- Brust, K. Toxicity of aliphatic amines on the embryos of zebrafish Danio rerio-experimental studies and QSAR. 2001.
- Koppel, N.; Maini Rekdal, V.; Balskus, E.P. Chemical transformation of xenobiotics by the human gut microbiota. Science 2017, 356, eaag2770. [CrossRef]
- Chattopadhyay, S.; Raychaudhuri, U.; Chakraborty, R. Artificial sweeteners–a review. Journal of food science and technology 2014, 51, 611-621.
- Yurttas, L.; Kaya, B.; Yilmaz Cankilic, M.; Levent, S. New Cyclohexylamine-dithiocarbamate Derivatives as Potential Anti-microbial Agents. Letters in Drug Design & Discovery 2017, 14, 1308-1315. [CrossRef]
- Schlosser, P.M.; Bale, A.S.; Gibbons, C.F.; Wilkins, A.; Cooper, G.S. Human health effects of dichloromethane: key findings and scientific issues. Environmental health perspectives 2015, 123, 114-119. [CrossRef]
- Obici, S.; Otobone, F.J.; da Silva Sela, V.R.; Ishida, K.; da Silva, J.C.; Nakamura, C.V.; Cortez, D.A.G.; Audi, E.A. Preliminary toxicity study of dichloromethane extract of Kielmeyera coriacea stems in mice and rats. Journal of Ethnopharmacology 2008, 115, 131-139. [CrossRef]
- Dekant, W.; Jean, P.; Arts, J. Evaluation of the carcinogenicity of dichloromethane in rats, mice, hamsters and humans. Regulatory Toxicology and Pharmacology 2021, 120, 104858. [CrossRef]
- Westphal, G.A.; Müller, M.M.; Herting, C.; Bünger, J.; Hallier, E. Genotoxic effects of N-nitrosodicyclohexylamine in isolated human lymphocytes. Archives of toxicology 2001, 75, 118-122. [CrossRef]
- Stevanović, J.; Stanimirović, Z.; Pejin, I.I.; Lazarević, M. Monitoring of mitotic index and frequency of micronuclei in evaluation of genotoxic potential of fumagillin (dicyclohexylamine) in vivo. Acta Veterinaria-Beograd 2006, 56, 437-448.
- Casey, J.P. Amines, cycloaliphatic. Kirk-Othmer Encyclopedia of Chemical Technology 2000.
- Libralato, G.; Ghirardini, A.V.; Avezzu, F. Seawater ecotoxicity of monoethanolamine, diethanolamine and triethanolamine. Journal of hazardous materials 2010, 176, 535-539. [CrossRef]
- Abdl-Razzaq, H.T.; Al-Azzawi, M.N.A. Toxicology Study the Acute effects of Diethanolamine in Mice Blood and Liver (Oral study). Journal of International Environmental Application and Science 2017, 12, 14-23.
- Bakalova, S.; Mincheva, V.; Doycheva, A.; Groudeva, V.; Dimkov, R. Microbial toxicity of ethanolamines. Biotechnology & Biotechnological Equipment 2008, 22, 716-720.
- Fiume, M.M.; Heldreth, B.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks Jr, J.G.; Shank, R.C.; Slaga, T.J. Safety assessment of diethanolamine and its salts as used in cosmetics. International journal of toxicology 2017, 36, 89S-110S.
- Trakulsrichai, S.; Chuayaupakarn, K.; Tansuwannarat, P.; Rittilert, P.; Tongpoo, A.; Sriapha, C.; Wananukul, W. Ethephon Poisoning: Clinical Characteristics and Outcomes. Toxics 2025, 13, 115. [CrossRef]
- Authority, E.F.S.; Alvarez, F.; Arena, M.; Auteri, D.; Binaglia, M.; Castoldi, A.F.; Chiusolo, A.; Colagiorgi, A.; Colas, M.; Crivellente, F. Peer review of the pesticide risk assessment of the active substance ethephon. EFSA Journal 2023, 21, e07742. [CrossRef]
- Bhadoria, P.; Nagar, M.; Bharihoke, V.; Bhadoria, A.S. Ethephon, an organophosphorous, a fruit and vegetable ripener: has potential hepatotoxic effects? Journal of family medicine and primary care 2018, 7, 179–183.
- Hall, A.; Henry, J. Acute toxic effects of ‘Ecstasy’(MDMA) and related compounds: overview of pathophysiology and clinical management. BJA: British Journal of Anaesthesia 2006, 96, 678–685. [CrossRef]
- Greene, S.L.; Dargan, P.I.; O’connor, N.; Jones, A.L.; Kerins, M. Multiple toxicity from 3, 4-methylenedioxymethamphetamine (“ecstasy”). The American journal of emergency medicine 2003, 21, 121–124.
- Quinton, M.S.; Yamamoto, B.K. Causes and consequences of methamphetamine and MDMA toxicity. The AAPS journal 2006, 8, E337-E337. [CrossRef]
- Okuda, H.; Takeuchi, T.; Senoh, H.; Arito, H.; Nagano, K.; Yamamoto, S.; Matsushima, T. Developmental toxicity induced by inhalation exposure of pregnant rats to N, N-dimethylacetamide. Journal of occupational health 2006, 48, 154–160.
- Menegola, E.; Broccia, M.; Prati, M.; Giavini, E. In vitro embryotoxicity study of n, n-dimethylacetamide and its main metabolite N-monomethylacetamide. Toxicology in vitro 1999, 13, 409–415. [CrossRef]
- Nomiyama, T.; Tsukahara, T.; Hasegawa, K. Health risks of N, N-dimethylacetamide (DMAC) in humans. Journal of Occupational Health 2025, uiaf010.
- Chen, Y.; Go, Y.-M.; Pohl, J.; Reed, M.; Cai, J.; Jones, D.P. Increased mitochondrial thioredoxin 2 potentiates N-ethylmaleimide-induced cytotoxicity. Chemical research in toxicology 2008, 21, 1205–1210. [CrossRef]
- Bowes, T.J.; Gupta, R.S. Induction of mitochondrial fusion by cysteine-alkylators ethacrynic acid and N-ethylmaleimide. Journal of cellular physiology 2005, 202, 796–804. [CrossRef]
- Gu, M.B.; Mitchell, R.J.; Kim, B.C.J.B. Whole-cell-based biosensors for environmental biomonitoring and application. 2004, 269-305.
- Nos, C. o-Toluidine and o-Toluidine. Report on Carcinogens: Carcinogen Profiles 2002, 10, 242.
- Toyoda, T.; Kobayashi, T.; Miyoshi, N.; Matsushita, K.; Akane, H.; Morikawa, T.; Ogawa, K. Toxicological effects of two metabolites derived from o-toluidine and o-anisidine after 28-day oral administration to rats. The Journal of Toxicological Sciences 2022, 47, 457–466. [CrossRef]
- Ochsmann, E. o-Toluidine–Evaluation of a BAR. Assessment Values in Biological Material–Translation of the German version from 2010.
- English, J.C.; Bhat, V.S.; Ball, G.L.; McLellan, C.J. Establishing a total allowable concentration of o-toluidine in drinking water incorporating early lifestage exposure and susceptibility. Regulatory Toxicology and Pharmacology 2012, 64, 269–284. [CrossRef]
- Adegunsoye, A.; Akhter, R.; Rachshtut, M.; Green, E. 856: PULMONARY INFARCTION: AN UNUSUAL COMPLICATION OF PHENTERMINE/PHENDIMETRAZINE FOR WEIGHT LOSS. Critical Care Medicine 2011, 39.
- Yen, M.; Ewald, M.B. Toxicity of Weight Loss Agents. Journal of Medical Toxicology 2012, 8, 145–152. [CrossRef]
- Lim, S.; Rogers, L.K.; Tessler, O.; Mundinger, G.S.; Rogers, C.; Lau, F.H. Phentermine: a systematic review for plastic and reconstructive surgeons. Annals of plastic surgery 2018, 81, 503–507. [CrossRef]
- Steidl, K.E.; Darko, W.; Probst, L.A.; Noviasky, J.A.; Nasser, S. Rhabdomyolysis associated with phentermine. American Journal of Health-System Pharmacy 2010, 67, 1929–1932.
- Yu-han, L.; Jing-jing, M.; Shou-xiang, W.; Lu-qing, P.; Yu-fei, L.; Jiang-yue, W. Acute toxicity and species sensitivity distribution of p-chloroaniline, 1, 2-dichloroethane, butyl benzyl phthalate and 1-phenylethyl alcohol on two marine bivalves. Chinese Journal of MARINE ENVIRONMENTAL SCIENCE 2020, 39, 732–737.
- Politano, V.T.; Diener, R.M.; Christian, M.S.; Hoberman, A.M.; Palmer, A.; Ritacco, G.; Adams, T.B.; Api, A.M. Oral and dermal developmental toxicity studies of phenylethyl alcohol in rats. International journal of toxicology 2013, 32, 32–38. [CrossRef]
- Scognamiglio, J.; Jones, L.; Letizia, C.; Api, A. Fragrance material review on phenylethyl alcohol. Food and chemical toxicology 2012, 50, S224-S239.
- Gamer, A.O.; Rossbacher, R.; Kaufmann, W.; van Ravenzwaay, B. The Inhalation toxicity of di- and triethanolamine upon repeated exposure. Food and Chemical Toxicology 2008, 46, 2173–2183. [CrossRef]
- Service, P.H.; Services, H.; Program, N.T. NTP toxicology and carcinogenesis studies of triethanolamine (Cas No. 102-71-6) in B6C3F1 mice (dermal studies). National Toxicology Program technical report series 2004, 5-163.
- Stott, W.; Radtke, B.; Linscombe, V.; Mar, M.-H.; Zeisel, S. Evaluation of the potential of triethanolamine to alter hepatic choline levels in female B6C3F1 mice. Toxicological Sciences 2004, 79, 242–247. [CrossRef]
- Leppanen, M.T.; Sourisseau, S.; Burgess, R.M.; Simpson, S.L.; Sibley, P.; Jonker, M.T.J.E.T.; Chemistry. Sediment toxicity tests: A critical review of their use in environmental regulations. 2024, 43, 1697–1716. [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




