1. Introduction
Dengue is an acute viral disease caused by one of the four dengue virus serotypes (DENV-1, DENV-2, DENV-3, and DENV-4), transmitted to humans through the bite of mosquitoes of the genus
Aedes, primarily
Aedes aegypti [
1]. Factors such as unplanned urbanization, human migration, and climate change have facilitated the spread of both the vector and the virus [
2]. Annually, it is estimated that 390 million infections occur across 129 countries, with approximately four billion people at risk [
3]. The regions most affected by dengue include Latin America and the Caribbean, Southeast Asia, and parts of tropical Africa, where
Aedes aegypti is endemic. Brazil, India, and Indonesia report the highest global case numbers, with recurrent outbreaks in countries such as Colombia, Mexico, the Philippines, and Thailand [
4]. Between 75% and 80% of those infected are asymptomatic, suggesting significant underreporting of cases. During epidemic peaks, nearly one million individuals require hospitalization, and severe forms of the disease cause approximately 25,000 deaths annually, with case fatality rates ranging from 1% to 5% [
4]. In response to the exponential increase in cases over the past two decades, the World Health Organization (WHO) declared dengue a Level 3 emergency in 2023 [
5].
Dengue exhibits a wide range of clinical manifestations and can rapidly progress from mild febrile illness to severe and life-threatening forms [
6]. While most infections are self-limiting and present as dengue fever, a significant proportion can advance to severe complications, such as dengue hemorrhagic fever (DHF) or dengue shock syndrome (DSS) [
6]. These severe forms are often associated with multi-organ failure and high mortality rates [
7]. The unpredictable and dynamic nature of the disease, coupled with its growing global incidence, underscores the urgent need to strengthen surveillance, prevention, and clinical management strategies, particularly in highly affected regions [
7].
Despite significant research, the immunopathological mechanisms underlying the development of DHF remain incompletely understood. It has been proposed that DHF may result from dysregulated lymphocytic and humoral responses, associated with phenomena such as immunologic imprinting and antibody-dependent enhancement (ADE) [
8]. These mechanisms may explain both the clinical severity and the increased vascular permeability characteristic of DHF in patients experiencing secondary infections [
8]. Additionally, the cytokine storm triggered by macrophages, dendritic cells, and mast cells appears to be a key driver of plasma leakage [
8]. Previous studies have shown that the virus does not directly cause endothelial damage; instead, cytokines such as Tumor necrosis factor-alpha (TNF-α), and interleukins (IL-1β, and IL-6) contribute to thrombocytopenia and immune hyperactivation. These cytokines, together with non-structural protein 1 (NS1) immune complexes, induce endothelial dysfunction and promote vascular permeability, suggesting that the inflammatory microenvironment and paracrine processes play central roles in severe dengue pathophysiology [
9].
A recently highlighted paracrine mediator in dengue is extracellular vesicles (EVs), which have emerged as critical players in dengue pathogenesis, contributing to immune modulation, endothelial damage, and viral transmission [
10]. EVs are small membrane-bound structures released by eukaryotic and prokaryotic cells in response to stimuli such as cellular stress and apoptosis [
1]. These vesicles play crucial roles in intercellular communication, molecular transport, and immune system modulation, and they can be detected in body fluids, underscoring their potential as biomarkers [
1].
In this context, this review aim to synthesize and analyze the latest research on the role of EVs in dengue pathogenesis. Specifically, it will address their ability to elucidate the physiological mechanisms underlying vascular permeability, plasma leakage, and immune evasion. Additionally, their potential impact on the development of clinically relevant biomarkers such as microRNAs contained within EVs will be explored, emphasizing on their use for patient monitoring and clinical classification through advanced bioinformatics tools. Finally, potential therapeutic strategies based on current knowledge of EVs will be discussed, highlighting their significance in managing severe dengue.
2. Materials and Methods
2.1. Search Strategy
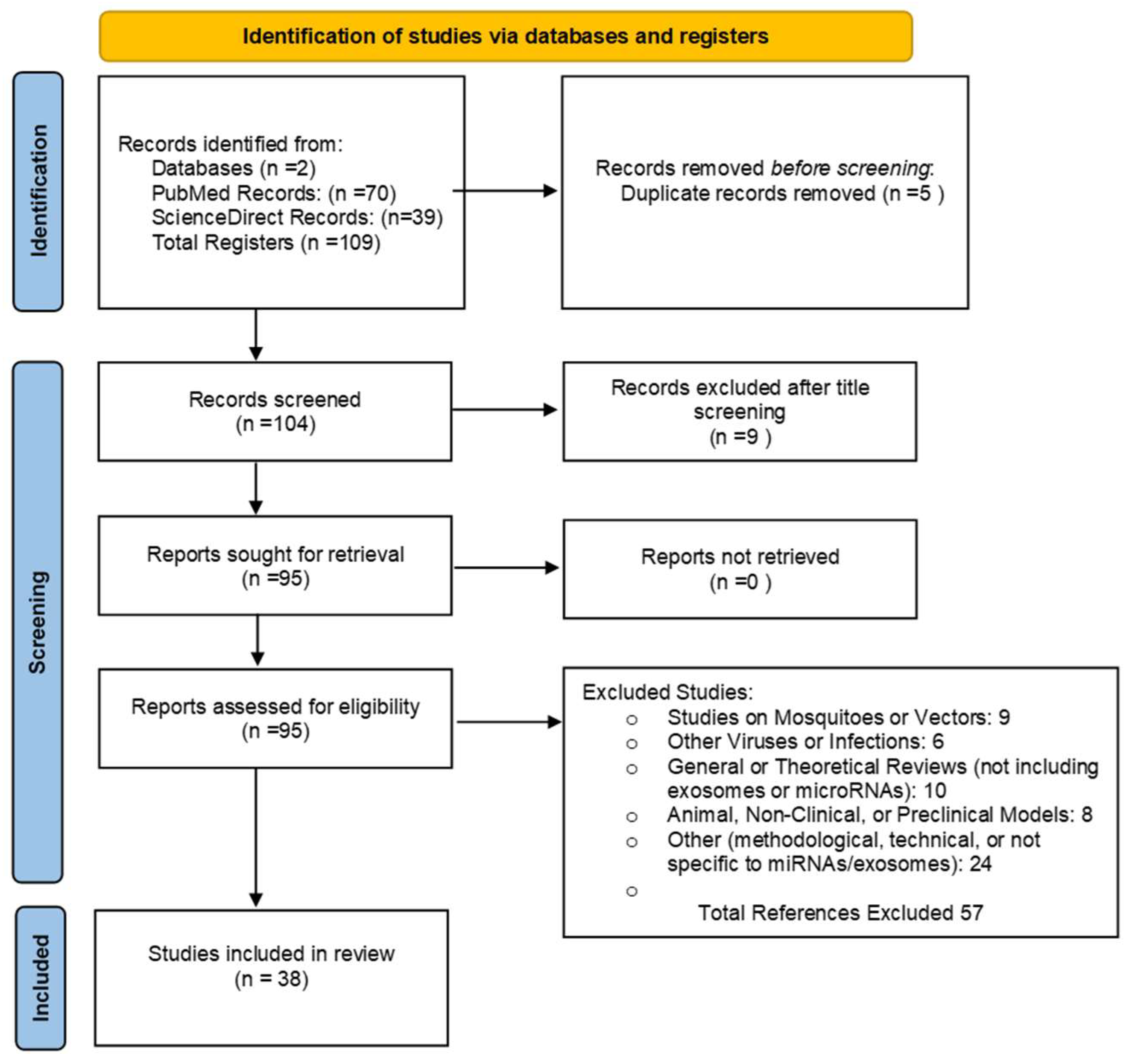
This study employs a narrative review to analyze recent research on the role of extracellular vesicles (EVs) in the pathogenesis of severe dengue. The review aims to address the following research question: What is the role of extracellular vesicles (EVs) in dengue pathogenesis, and how can they contribute to understanding vascular permeability, plasma leakage, immune evasion, and the development of clinically relevant biomarkers and therapeutic strategies. To robustly address this question using relevant and up to date evidence, we conducted a comprehensive literature search in two databases, ScienceDirect and PubMed, following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) framework [
11]. The search strategy was based on the following equation: ('Extracellular Vesicles' OR 'Exosomes' OR 'Microvesicles' OR 'microRNAs') AND ('Dengue' OR 'Severe Dengue'). To enhance the sensitivity and specificity of the search, we incorporated Medical Subject Headings (MeSH) terms. The detailed methodology is illustrated in
Figure 1. Additionally, to update data related to the epidemiology and general characteristics of the viral infection, the following search terms were used: ('Dengue' OR 'Severe Dengue' OR 'Dengue Hemorrhagic Fever') AND ('Epidemiology').
2.2. Eligibility Criteria
The inclusion criteria for this review focused on peer reviewed journal articles, review articles, research papers, and book chapters published in English, Portuguese, or Spanish, within the last 10 years (2014 to 2024), that involved human studies or preclinical research. Articles were excluded if they primarily addressed mosquitoes or vectors without discussing extracellular vesicles (EVs) or microRNAs (miRNAs) in the context of dengue, investigated viruses or infections unrelated to dengue, were general or theoretical reviews that did not specifically cover exosomes, EVs, or miRNAs in relation to dengue pathogenesis, involved animal models or non clinical studies without direct relevance to human clinical data on dengue, or were methodological or technical papers not focused on miRNAs, exosomes, or EVs, lacking clinical applicability to dengue. To ensure the quality of the paper, duplicates were meticulously reviewed, and article abstracts were included to verify the relevance and academic integrity of the literature
2.3. Bioinformatic Analysis of miRNAs in Dengue
Bioinformatics analyses were conducted to investigate the relationship between circulating microRNAs (miRNAs) within EVs and dengue virus (DENV) infection or disease severity. A reanalysis of differentially expressed miRNAs (DEmiRs) was performed using the RNA sequencing dataset GSE150623, obtained from the Gene Expression Omnibus (GEO). This dataset represents the circulating microtranscriptome of dengue patients. The analysis was carried out using the limma voom [
12] pipeline in R Studio [
13], enabling the identification of miRNAs potentially associated with disease progression and severity. DEmiRs cutoffs were set as |LogFC| > 2 and adjusted p-value < 0.05. Enrichment analysis of target genes of DemiRs was performed through Diana miRPath v4 [
14].
3. Clinical Presentation and Structural Overview of the Dengue Virus
The dengue virus is an enveloped 50-nm virus belonging to the Flavivirus genus and the Flaviviridae family [
15]. Its structure is icosahedral, and its genome consists of a single-stranded positive-sense RNA. This genome encodes a single open reading frame (ORF), which is translated into a polyprotein [
15]. The polyprotein is subsequently processed into three structural proteins (capsid [C], envelope [E], and prM/M) and seven nonstructural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5). These proteins play critical roles in viral assembly, replication, and immune evasion within the host [
15].
To date, four clinically relevant dengue virus serotypes have been identified: DENV-1, DENV-2, DENV-3, and DENV-4 [
16]. These serotypes are antigenically related, sharing 60–75% amino acid similarity. This limited cross-protective immunity contrasts with other viral infections. Within each serotype, viruses can be further divided into genotypes, differing genetically by up to 6% [
16]. These genetic variations can significantly influence virulence, disease severity, and the epidemiological dynamics of outbreaks [
15,
16].
Dengue pathogenesis progresses through three distinct phases: febrile, critical, and recovery, which define the clinical course of the disease. According to the 2009 WHO classification, dengue is categorized into dengue without warning signs, dengue with warning signs, and severe dengue. The febrile phase begins after an incubation period of 2 to 7 days, characterized by sudden fever exceeding 38°C, accompanied by symptoms such as headache, retro-orbital pain, myalgia, nausea, and rash [
17]. During this stage, viremia is high and detectable via quantitative polymerase chain reaction (qPCR), peaking within the first 4–5 days [
17]. Common complications include dehydration and febrile seizures in children due to high fever. In most cases, the infection is mild and self-limiting [
17].
The critical phase, occurring in some patients, particularly during heterotypic reinfections, lasts 24–48 hours and coincides with declining viremia and fever. This stage carries a higher risk of complications due to an exaggerated inflammatory response causing endothelial damage and increased capillary permeability [
18]. This can lead to warning signs such as abdominal pain, persistent vomiting, bleeding, and hepatomegaly. Severe cases may progress to significant fluid leakage, shock, and organ dysfunction affecting the liver, kidneys, and central nervous system (CNS) [
18].
Finally, the recovery phase involves the reabsorption of fluids and clinical stabilization. However, some patients may experience post-infection symptoms, including fatigue, muscle pain, depression, and neurological complications, particularly in adults [
19].
4. Immune Response and the Potential Role of EVs in Dengue Severity
Dengue infection begins with the mosquito bite, introducing the virus into the dermis (
Figure 1A). There, Langerhans cells recognize the virus through pattern recognition receptors (PRRs) such as Toll-like receptors (TLRs), RIG-I, and MDA5 [
20]. This triggers the production of type I interferons (IFN)-α/β by macrophages, epithelial cells, and dendritic cells (DCs). The DCs migrate to lymph nodes, where they facilitate viral dissemination to monocytes and other DCs, amplifying viremia and targeting organs such as the liver, spleen, and vascular endothelium [
20].
The innate immune response, led by macrophages, DCs, and natural killer (NK) cells, produces pro-inflammatory cytokines such as IL-1, IL-6, IL-8, and TNF-α, responsible for early symptoms like fever and malaise [
21]. Concurrently, DCs present viral antigens to T and B lymphocytes, activating the adaptive immune response. CD8+ T lymphocytes eliminate infected cells, while CD4+ T cells adopt a Th1 phenotype, promoting viral clearance via IFN-γ production (Fig. 1B). B lymphocytes generate antibodies targeting structural and nonstructural viral proteins, including M, E, and NS1. This adaptive response clears viremia and establishes specific immune memory, mediated by protective IgG antibodies against reinfection by the same serotype [
21].
Severe manifestations often arise during heterotypic secondary infections. In these cases, preexisting IgG antibodies fail to efficiently neutralize the new serotype, facilitating viral entry into monocytes and macrophages via Fc receptors. This process, known as antibody-dependent enhancement (ADE), increases viral replication and inflammation, contributing to disease severity [
22]. ADE excessively activates innate immune cells, triggering the release of cytokines such as TNF-α, IL-1β, IL-6, and IFN-γ, along with vasoactive mediators like histamine and tryptase released by mast cells. These factors cause endothelial damage and heightened vascular permeability, resulting in plasma leakage characteristic of severe cases. CD8+ T lymphocytes activated during secondary infections further exacerbate inflammation, producing excessive pro-inflammatory cytokines due to their low affinity for the new serotype [
22]. However, ADE or immunologic imprinting can explain not all severe dengue cases. Up to 5% of patients may develop severe forms during primary infection, suggesting that other factors such as host genetics, age, and viral genotype also contribute to disease severity [
22].
Extracellular vesicles have garnered significant attention in the study of dengue pathogenesis, particularly for their multifaceted roles in disease progression [
10]. As dynamic carriers of biomolecules, EVs derived from infected patients and in vitro models have been implicated in exacerbating dengue severity, including plasma leakage and endothelial dysfunction. Their ability to mediate immune interactions positions them as pivotal agents not only in dengue but also in other viral infections such as hepatitis, HIV, and flaviviruses [
23].
In severe dengue, EVs contribute to pathophysiological processes by facilitating viral dissemination, disrupting vascular integrity, and modulating host immune responses. Investigating EVs provides valuable insights into the molecular mechanisms driving these outcomes and offers potential pathways for developing advanced diagnostic tools and therapeutic interventions tailored to mitigate the impacts of severe dengue [
10,
23].
5. Biogenesis of Extracellular Vesicles and Their Role as microRNA Carriers in Dengue Virus Infection
Extracellular vesicles (EVs) are nanometer-sized structures released by nearly all cell types and play a central role in intercellular communication. Encapsulated by lipid bilayers, these vesicles carry bioactive molecules, including RNA, proteins, and lipids, which can modulate physiological and pathological processes in recipient cells. The synthesis of EVs involves different molecular pathways, depending on the specific vesicle subtype [
24]. The diversity of these biogenesis mechanisms reflects the functional heterogeneity of EVs and their adaptability to cellular contexts [
25,
26]. The molecular machinery that regulates the biogenesis of these vesicles not only determines their formation but also influences the selection of content, membrane composition, and, ultimately, their biological effects on recipient cells [
27].
Apoptotic bodies, for example, are formed during programmed cell death. As apoptosis progresses, the activation of caspases triggers the cleavage of cytoskeletal components, resulting in cell fragmentation [
28]. Segments of the plasma membrane, forming apoptotic bodies (Fig 1C), enclose these fragments. Unlike other EVs, apoptotic bodies can encapsulate organelles, chromatin fragments, and other cellular debris, serving as a mechanism to remove dead cells and modulate immune responses [
29,
30]. On the other hand, microvesicle formation occurs through the budding of the plasma membrane, a process driven by cytoskeletal rearrangements. The biogenesis of these vesicles depends on changes in membrane lipid asymmetry, mediated by enzymes such as flippases, floppases, and scramblases [
31,
32]. The activation of these enzymes promotes the externalization of phosphatidylserine, contributing to membrane curvature and budding. Actomyosin contractions, regulated by Rho-associated kinase (ROCK) and calcium signaling, provide the mechanical force needed for membrane fission [
33,
34]. Microvesicles are often enriched with integrins, selectins, and metalloproteinases, which contribute to their functional specialization and targeting capability (Fig 1C).
Exosomes, distinct from apoptotic bodies and microvesicles, are formed within multivesicular bodies (MVBs) through either ESCRT-dependent or ESCRT-independent mechanisms [
33,
35]. Within MVBs, intraluminal vesicles (ILVs) form inside late endosomes. The ESCRT machinery, composed of four main complexes (ESCRT-0, -I, -II, and -III), coordinates this process [
35]. ESCRT-0 recruits ubiquitinated cargo to the endosomal membrane, while ESCRT-I and -II promote membrane deformation, and ESCRT-III carries out membrane cleavage. Accessory proteins such as ALIX (ALG-2-interacting protein) and TSG101 (tumor susceptibility gene 101) interact with cargo and ESCRT components to fine-tune the vesicle formation process (Fig 1C). Exosome biogenesis can also occur via ESCRT-independent pathways, in which lipids like ceramides and tetraspanins (e.g., CD9, CD63, and CD81) play critical roles in membrane curvature and vesicle formation [
35]. After formation, MVBs are transported along microtubules, guided by motor proteins like kinesins and dyneins, to the plasma membrane [
36]. Rab family proteins, especially Rab27a and Rab27b, regulate MVB anchoring and fusion with the plasma membrane, resulting in exosome release into the extracellular space [
36,
37].
In this context, EVs are crucial vehicles in intercellular communication, mediating the efficient transport of microRNAs (miRNAs) and other biomolecules, particularly during viral infections [
38,
39]. miRNAs carried by EVs play essential roles in modulating host responses and promoting viral pathogenesis, acting as post-transcriptional regulators of genes involved in viral replication, immune response, and inflammation [
40]. In the context of dengue virus (DENV) infection, studies show that EVs released by infected cells carry specific miRNAs that can significantly alter infection dynamics and virus-host interactions [
41].
During DENV infections, exosomes can carry miRNAs such as miR-21, miR-146a, and miR-155, which play crucial roles in modulating innate immunity [
42,
43]. For example, miR-21, often enriched in EVs during viral infections, negatively regulates genes involved in apoptosis control and viral replication, promoting cell survival and allowing continuous viral replication [
44]. miR-146a suppresses pro-inflammatory pathways, such as nuclear factor kappa B (NF-κB) signaling, reducing excessive immune responses and promoting viral persistence [
45]. miR-155, in turn, is associated with the amplification of the immune response, being a miRNA with potentially ambiguous effects depending on the stage of infection. In addition to host-derived miRNAs, EVs can also carry viral RNA. Studies have demonstrated that EVs released during DENV infections contain fragments of viral genomic RNA [
42,
45,
46]. These EVs can facilitate viral spread to adjacent cells, contributing to infection amplification as well as modulation of the immune microenvironment (Fig 1C).
Recent studies have confirmed the presence of specific miRNAs in circulating EVs during DENV infection. Proteomic and transcriptomic analysis revealed that exosomes derived from dendritic cells infected with DENV carry miRNAs such as miR-21-5p, miR-222-3p, and miR-126-5p, which play important roles in regulating immune and inflammatory responses [
47,
48]. For example, miR-126-5p is involved in maintaining vascular integrity, and its dysfunction mediated by DENV contributes to the increased vascular permeability observed in severe dengue [
49,
50]. Additionally, miR-222-3p has been associated with the suppression of antiviral responses by negatively regulating genes such as STAT3 and SOCS3, critical components of interferon signaling [
50].
Exosomes released by infected cells have also been described as modulators of macrophage activation, promoting a pro-inflammatory phenotype that exacerbates the immune response and contributes to dengue pathogenesis. The transport of specific miRNAs to immune cells via EVs illustrates the ability of these vesicles to reshape the immune microenvironment, promoting both viral dissemination and exacerbated inflammation.
6. Overview of EVs' Role in Dengue Infection: Transmission Dynamics
During DENV infection, infected cells release EVs enriched with viral genetic material, including complete genomic RNA and structural and non-structural proteins like NS1 and E [
41]. These vesicles can act as vectors to infect neighboring cells, as the viral content within the EVs is protected from extracellular enzymatic degradation. Recent studies show that EVs can promote the entry of viral RNA into uninfected cells, where the viral replication cycle is initiated, contributing to the amplification of viral load in the host [
47,
51]. This process demonstrates that EVs are not merely a byproduct of the infection but active participants in viral dissemination.
Additionally, EVs can influence viral tropism. For example, exosomes released by infected hepatocytes may contain specific membrane proteins that facilitate fusion with target cells, such as monocytes and endothelial cells [
38,
52]. This specificity in targeting can enhance infection efficiency in critical tissues, such as the liver and blood vessels, exacerbating disease progression and contributing to the systemic spread of the virus [
53,
54].
Besides transporting viral components, EVs derived from infected cells play a central role in modulating immune responses. They can carry immunomodulatory molecules that suppress or redirect host responses to favor viral replication. For example, viral proteins carried in EVs, like NS1, can inhibit the IFN pathway in target cells, weakening the innate antiviral response [
55,
56]. This mechanism allows DENV to evade early detection by the immune system, facilitating its replication and spread. Furthermore, EVs containing specific microRNAs can directly influence cellular processes in the host [
57]. The transport of miRNAs such as miR-146a and miR-21, widely described in viral infections, has been associated with the suppression of inflammatory pathways and regulation of the immune environment [
58]. These miRNAs modulate the expression of genes crucial to the immune response, creating a cellular microenvironment conducive to viral replication and spread to other cells [
38,
59].
Extracellular vesicles are also implicated in regulating viral load and clinical progression of DENV infection. Studies show that elevated levels of EVs circulating in association with dengue virus (DENV) infection correlate with higher viral loads and greater disease severity [
38]. This association can be attributed to the role of EVs in local infection amplification, transferring viral material directly to neighboring cells and modulating immune responses in ways that avoid efficient viral clearance [
38,
47].
Moreover, EVs derived from infected cells can alter the function of immune cells such as macrophages and dendritic cells, promoting the secretion of pro-inflammatory cytokines like IL-6 and TNF-α [
60]. This increase in cytokine production can trigger systemic inflammatory responses, contributing to plasma leakage syndrome and other severe symptoms of dengue, such as hemorrhagic fever and shock.
7. Extracellular Vesicles as Mediators of Viral Spread and Immune Evasion in Dengue Virus Infection.
Recent evidence suggests that extracellular vesicles (EVs) present in dengue patients, as well as those derived from virus-infected innate immune cells, have the ability to modulate the immune response, induce inflammation, and, in some cases, transport the viral genome. These EVs may influence dengue pathogenesis by facilitating viral spread and altering immune system function.
On the other hand, it has been demonstrated that EVs released by virus-activated platelets contain IL-1β and TNF-α, which together contribute to inflammation and play a central role in endothelial activation and plasma leakage during severe dengue [
61]. Along the same lines, platelets from dengue patients have been shown to release EVs that stimulate the expression of inflammatory markers such as CRP, SAA, sVCAM-1, and sICAM-1 in endothelial cells, thereby promoting leukocyte adhesion, activation, and endothelial inflammation [
62]. Moreover, the microRNA miR-96-5p, identified in patient-derived exosomes, has been found to trigger the IL-1β inflammatory pathway by downregulating HSP70, a natural inhibitor of the NLRP3 inflammasome [
63]. These findings further support the role of microRNAs as key mediators of inflammation in dengue.
According to Kumari et al., EVs derived from patients with severe dengue express PD-L1 on their surface. This protein interacts with CD4+ lymphocytes, inhibiting their proliferation and increasing the expression of the PD-1 coreceptor [
64]. Furthermore, these EVs carry pro-inflammatory cytokines such as IFN-γ, TNF-α, IL-2, IL-6, IL-17A, IL-13, IL-5, and IL-4, which appear to contribute both to inflammation and the anergy observed in the study [
64]. Similarly, another study demonstrated that dendritic cells (DCs) infected with the dengue virus can secrete EVs containing mRNA of inflammatory mediators such as CXCR4, MIF, IL-17A, IL-8, and IL-6, which could be translated and exacerbate inflammation [
47].
Additionally, Mishra et al. showed that EVs derived from dengue-infected monocytes can transfer miR-148a, modulating USP33 and ATF3 levels in human microglial cells [
65]. This process may contribute to the expression of TNF-α, NF-κB, and IFN-β, factors associated with neuroinflammation—a complication observed in severe dengue cases that remains poorly understood from a pathophysiological perspective [
66]. These findings suggest that EVs play a significant role in dengue immunopathogenesis, acting as vehicles that transport molecules capable of modulating the immune response and potentially exacerbating disease-associated complications.
Although further evidence is needed, a potential immune evasion mechanism employed by the dengue virus involves the use of EVs to transport viral RNA or proteins that promote viral replication in target cells [
23,
47]. In a study led by Martins and collaborators, EVs derived from dengue-infected dendritic cells were shown to infect C6/36 cells in vitro, suggesting that these EVs may carry the complete viral genome [
47]. This mechanism could allow the virus to evade detection by antibodies and immune cells such as CD8+ lymphocytes and NK cells, which would typically recognize and eliminate the virus [
22].
Moreover, another study demonstrated that EVs derived from Aedes aegypti cells infected with dengue contain proteins such as AAEL017301 (elongation factor-1 alpha) and AAEL002675 (a protein of unknown function) that appear to increase susceptibility to infection in target cells [
66]. These findings highlight the critical role of EVs in dengue pathogenesis, emphasizing their potential as modulators of immune responses, facilitators of viral dissemination, and contributors to disease severity. Understanding the mechanisms underlying these processes could pave the way for novel diagnostic and therapeutic strategies.
8. The Role of Extracellular Vesicles in Endothelial Damage and Vascular Hyperpermeability in Dengue
The integrity of the endothelial barrier plays a critical role in vascular homeostasis and is severely compromised during dengue infections, particularly in severe cases such as dengue hemorrhagic fever and dengue shock syndrome [
67,
68,
69]. In this context, EVs are emerging as important mediators in processes that lead to endothelial dysfunction, directly contributing to vascular hyperpermeability and plasma leakage [
70,
71]. Detailed studies of these mechanisms have shed light on how EVs act as vehicles for bioactive molecules, including viral proteins, cytokines, and microRNAs, which alter the behavior of endothelial cells and exacerbate the clinical symptoms of the disease [
72,
73].
During dengue virus (DENV) infection, infected cells release EVs enriched with viral proteins, such as NS1 and E, that can directly interact with endothelial cells [
41]. NS1, in particular, is widely recognized for inducing the degradation of cell junction proteins, such as VE-cadherin and claudins, which are essential for maintaining vascular barrier integrity [
41,
72]. When carried by EVs, NS1 can reach distant endothelial cells, promoting systemic damage to the vascular network. This effect is not limited to localized areas of infection but extends to peripheral tissues, exacerbating plasma leakage and contributing to severe manifestations of dengue, such as edema and shock [
41,
73].
Additionally, exposure of endothelial cells to EVs derived from DENV-infected cells triggers metabolic and cytoskeletal changes in these cells [
38]. These changes, mediated by proteins and miRNAs transported by EVs, alter actin dynamics and impair the formation of stable cell contacts, further amplifying vascular permeability [
74]. EVs also carry molecules that activate inflammatory signaling pathways in endothelial cells, such as the TLR and tumor necrosis factor (TNF) pathways. For example, EVs containing viral RNA can interact with TLRs on the surface of endothelial cells, triggering a signaling cascade that results in the release of pro-inflammatory cytokines, such as IL-6 and TNF-α [
74]. These cytokines, in turn, increase the expression of adhesion molecules, such as ICAM-1 and VCAM-1, facilitating leukocyte recruitment and perpetuating inflammation [
75]. This continuous inflammatory cycle further aggravates endothelial dysfunction and promotes fluid leakage.
As previously discussed, EVs influence the activity of pathways associated with oxidative stress, such as the generation of reactive oxygen species (ROS) in endothelial cells [
76]. This oxidative stress damages cellular components and amplifies endothelial barrier disruption, contributing to the extensive damage observed in severe DENV infections [
77].
DENV infection in endothelial cells leads to ROS production through several pathways, including the activation of NADPH oxidase (NOX) and mitochondrial dysfunction. Inhibition of ROS production associated with NOX decreases viral replication, cell death, and secretion of inflammatory cytokines such as IL-6, IL-8, and CCL5 [
78].
Additionally, oxidative stress induced by DENV activates signaling pathways that lead to the degradation of the transcription factor Nrf2, which is crucial for the antioxidant response [
79]. The degradation of Nrf2 compromises the cell’s ability to regulate oxidative stress, exacerbating inflammation and apoptosis [
80].
Autophagy activation, associated with endoplasmic reticulum stress, also contributes to ROS production during DENV infection. This process can have dual effects, facilitating viral replication and protecting against cell death, but also exacerbating oxidative stress [
79].
9. Role of Extracellular Vesicle-Derived microRNAs in Dengue Severity
EVs are crucial mediators of intercellular communication and play an important role in modulating host-pathogen interactions. In the context of dengue infection, specific miRNAs derived from EVs have been implicated in influencing disease outcomes and response[
63,
65].
miRNAs are small non-coding RNA molecules, typically 20–22 nucleotides in length, that play a crucial role in regulating gene expression at the posttranscriptional level. Each miRNA can bind to multiple mRNA sequences with varying degrees of complementarity, enabling the simultaneous regulation of hundreds of genes in response to environmental changes.
Plasma EVs are derived from adaptive and innate immune cells as well as non-immune blood cells such as platelets [
81]. DENV infection leads to increased release of EVs from severe dengue patients platelets and monocytes [
64,
65], while also being able to change the load of such vesicles [
64].
In search for specific biomarkers candidates of severity of the pathological outcome, many studies aimed at miRNAs as potential biomarkers. Studies often reach different miRNAs candidates as they are usually performed on specific populations, geographically or age-restricted. Still, correlations of Differentially Expressed miRNAs (DEmiRs) found in DENV-infection and disease severity are reported in the literature for circulating miRNAs [
82,
83], thus indicating the possibility of miRNAs to function as disease biomarkers.
In this context, miRNAs derived from EVs are crucial in modulating immune responses, inflammation, and vascular dysfunction, particularly in the context of severe dengue. By targeting specific mRNAs, miRNAs regulate key pathways involved in these processes, influencing disease progression and severity.
A study showed that DENV infected cells increase the EVs release and the released levels of miR-148a, which suppresses USP33 and stabilizes the ATF3 protein levels, reporting a crucial role of the miR-148a/USP33/ATF3 axis on pro-inflammatory response and DENV replication maintained by EVs [
65]. miR-1204 and miR-491-5p are also reported to influence DENV replication response through p53 regulation of apoptosis [
84].
In the context of vascular dysfunction, miRNAs regulate endothelial cell integrity and permeability, two critical factors in dengue pathogenesis. For example, the regulation of EZH2 expression through miR-150 is suggested to interfere in plasma leakage during DENV infection [
85]. miRNAs are known to target junctional proteins and signaling molecules that maintain endothelial barrier function [
86,
87]. Their dysregulation can lead to increased vascular permeability, plasma leakage, and hemorrhagic manifestations characteristic of severe dengue, as is the case for the DENV-associated endothelial dysfunction related to miR-126-5p [
49,
50], although the link between miRNAs and thrombocytopenia in DENV is still unknown [
82]. Overall, miRNAs derived from EVs represent key molecular players that bridge immune dysregulation, inflammation, and vascular compromise in severe dengue.
10. Clinical Applications of EVs in Dengue Diagnosis
Recent research has highlighted the potential of EVs as key biomarkers for the identification and monitoring of severe dengue. Various studies have demonstrated that circulating EVs play a crucial role in the pathological processes associated with this disease [
10]. Both their concentration and the presence of specific surface antigens or miRNAs have been identified as valuable tools for diagnosing severe forms of dengue [
10].
For instance, Punyadee et al. (2015) reported significantly elevated levels of EVs in patients with DHF compared to those with non-severe dengue or healthy individuals. Moreover, they found that the quantity of these vesicles directly correlated with disease severity [
88]. Similarly, Kumari et al. (2023) identified an increase in platelet-derived EVs in patients with severe dengue, underscoring the immunoregulatory role of these vesicles [
64].
These findings are consistent with studies in other viral pathologies and even in cancer patients, where an increase in circulating EV concentration has also been observed [
89]. In dengue, circulating EVs may also transport viral components, such as the E, C, and NS1 proteins, potentially contributing to viral dissemination [
23].
Additionally, EVs derived from patients with severe dengue have shown the ability to carry the immune checkpoint PD-L1, highlighting their immunoregulatory role and potential to induce immune anergy [
64]. This phenomenon parallels observations in patients with severe COVID-19, where PD-L1 has been detected in both EVs and circulating monocytes [
90], suggesting shared features in immune modulation between the two pathologies.
Recent studies have also identified the presence of specific microRNAs in EVs found in the serum of patients with severe dengue [
10]. These microRNAs, which will be discussed in detail later, show promising potential as tools to differentiate between severe dengue, dengue with warning signs, and dengue without warning signs.
11. Isolation and Detection of EVs from Dengue Patients
EVs from dengue patients must first be isolated to enable subsequent quantification, phenotypic characterization, and molecular content analysis. These vesicles can be obtained from serum or plasma samples collected with anticoagulants such as EDTA or citrate. Samples may be processed immediately or stored under ultracold conditions for later analysis [
91].
Commonly used EV isolation methods include ultracentrifugation, immunoaffinity capture, precipitation, and size-exclusion chromatography [
92]. The choice of method should be guided by the specific objectives of the study and take into account factors such as EV yield, purity, and the intended downstream applications, including functional assays [
92]. Once isolated, EVs can be characterized using various analytical techniques.
Nanoparticle Tracking Analysis (NTA) is widely employed to assess the size distribution and concentration of EVs. When coupled with fluorescently labeled antibodies, NTA also enables the detection of surface antigens, making it a useful tool for initial phenotypic profiling, particularly in complex biological matrices. However, NTA faces several limitations. One major constraint is the limited number of fluorescence channels, which hinders the simultaneous detection of multiple markers and limits its ability to perform deep immunophenotyping comparable to high-resolution flow or mass cytometry. Additionally, fluorescent NTA may lack the sensitivity required to detect EV subpopulations expressing low-abundance markers. The technique also does not provide detailed information on vesicular cargo and cannot reliably distinguish EVs from similarly sized particles, such as lipoproteins or protein aggregates, without prior rigorous purification [
93].
Transmission Electron Microscopy (TEM), on the other hand, remains a gold-standard method for confirming EV morphology. It also enables the localization of exosomal markers through immunogold labeling using antibodies against proteins such as CD9, CD63, and CD81 [
3,
94]. Despite its high resolution and specificity, TEM is expensive, time-consuming, and requires specialized expertise and instrumentation.
In recent years, flow cytometry platforms have advanced considerably. Instruments like the CytoFLEX Nano allow for the sensitive detection of submicron particles as small as 40 nm. This technology enables detailed immunophenotyping of EVs by detecting both surface and intracellular antigens with fluorochrome-conjugated antibodies against classical tetraspanins (CD9, CD63, CD81) and cell-origin markers such as CD41a (platelet-derived EVs) and CD3 (T cell-derived EVs) [
7,
8,
94]. Furthermore, the use of permeabilizing agents combined with antibodies allows for the detection of intracellular molecules. The incorporation of NIST-traceable size standards, along with the FCMPASS software, has enabled the standardization of light scatter measurements using Mie theory-based calibration curves. This allows scatter data to be reported in comparable physical units (e.g., nanometers), thereby improving EV characterization and enabling more consistent assessment of EV size and concentration, particularly in instruments capable of event counting [
95].
Western blotting is also commonly used to identify specific proteins in EV preparations and represents a relatively low-cost and accessible method to validate the presence of EV-associated markers. According to the International Society for Extracellular Vesicles (ISEV), proper characterization should include the detection of at least one transmembrane protein (e.g., CD9 or CD63) and one cytosolic protein (e.g., TSG101 or ALIX), as well as negative markers such as albumin or uromodulin to assess purity [
10]. This technique is widely compatible with standard laboratory infrastructure, making it practical in many research settings.
Nevertheless, Western blotting has several limitations. It is semi-quantitative and has limited sensitivity, making it less effective for detecting low-abundance proteins or small EV subpopulations. It also provides no information on particle size, concentration, or heterogeneity, and cannot confirm whether the detected proteins are associated with intact EVs or are free-floating contaminants. Furthermore, it often requires relatively large protein inputs, which can be challenging when working with limited sample volumes or low EV yields [
10].
Simultaneously, nucleic acid analysis of EVs is routinely performed using traditional methods like quantitative PCR (qPCR) and more advanced platforms such as next-generation sequencing (NGS). These techniques allow for the detection of microRNAs, other non-coding RNAs, and DNA contained within EVs. A wide range of commercially available kits has been developed for the extraction and purification of EV-derived microRNAs, enhancing both the efficiency and reproducibility of downstream analyses [
92].
To ensure reproducible and high-quality results, it is essential to implement rigorous quality control, appropriate pre-analytical handling of samples, and continuous validation of methods. Moreover, adherence to standardized protocols aligned with international guidelines—such as the Minimal Information for Studies of Extracellular Vesicles (MISEV)—is strongly recommended [
92]. This will support the robust implementation of EV-based diagnostics for dengue in clinical settings and future research applications.
12. EVs as Potential Biomarkers for Dengue Severity: Role of NS1
EVs play a crucial role in the pathophysiology of dengue, particularly in endothelial dysfunction and vascular hyperpermeability, processes that are vital for disease progression [
96,
97]. These vesicles not only amplify the damage directly by promoting viral spread but also serve as molecular markers of the infection's severity. In severe cases of dengue, such as hemorrhagic fever and dengue shock syndrome, the profile of circulating EVs changes, reflecting an increase in viral load, the presence of viral proteins like NS1, and modulation of the host's immune response [
41,
73]. The presence of NS1 in circulating EVs is a key marker for diagnosing and monitoring dengue infection, but additional molecular components of EVs, such as viral RNA, miRNAs, and host proteins involved in immune modulation, could also serve as valuable biomarkers.
Analyzing the specific molecular content of these EVs could provide new biomarkers for predicting the transition from mild to severe dengue and monitoring therapeutic effectiveness. Identifying biomarkers based on circulating EVs offers a powerful tool for early detection of dengue progression and for tailoring treatment strategies. The role of EVs in viral material transport, their influence on inflammatory pathways, and their contribution to viral load highlight their importance in the pathogenesis of dengue. Therapeutic strategies that block the formation or release of EVs, or interfere with their interaction with endothelial cells, could prove promising for mitigating the severe effects of DENV infection and improving clinical outcomes.
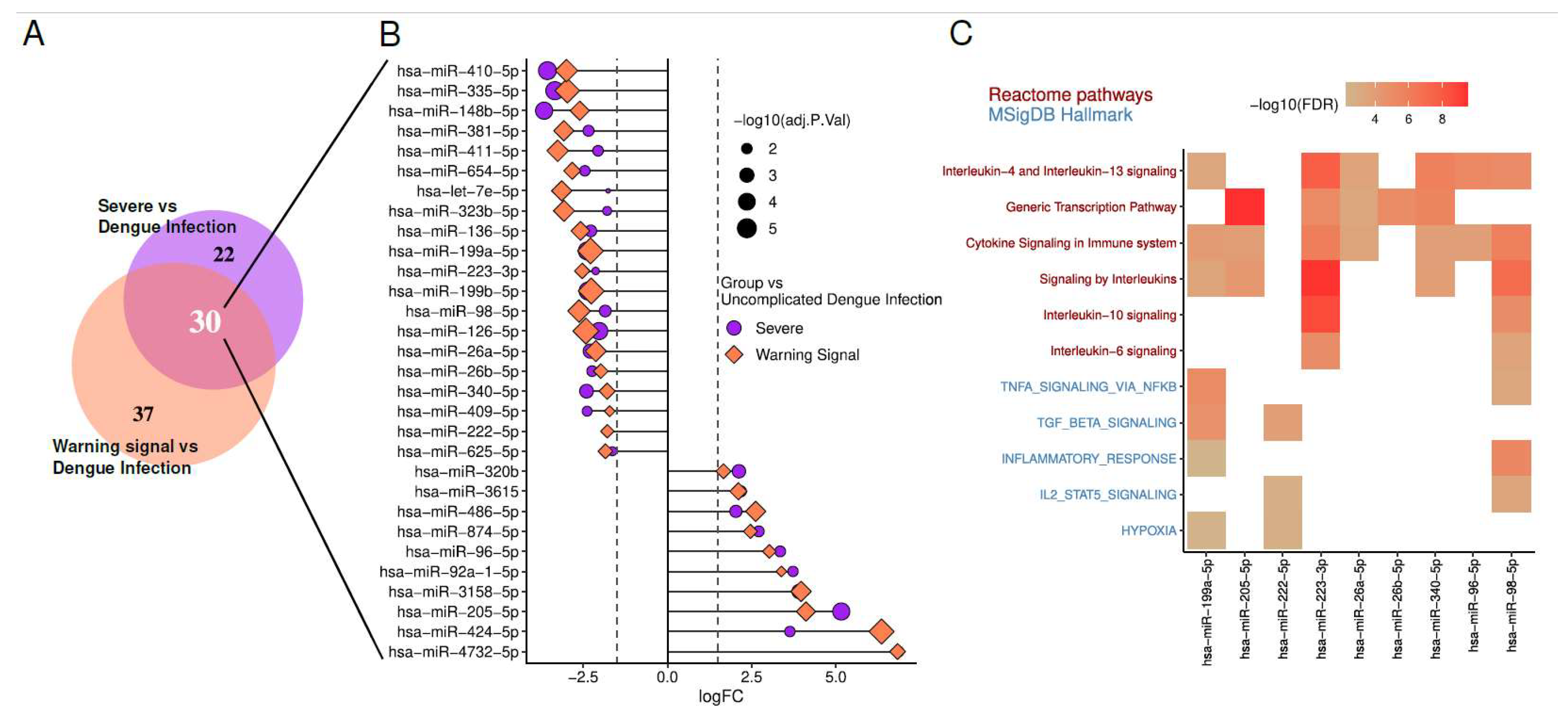
13. Differentially Expressed miRNAs in Dengue Patients and Potential Signaling Pathways Involved
To investigate the relationship between miRNAs and DENV infection or severity, we reanalyzed DEmiRs from an RNA sequencing dataset of the circulating microtranscriptome of dengue patients (GSE150623) [
98], using the limma-voom pipeline in R.
We focused on DEmiRs with |LogFC| ≥ 2 and adjusted p-value < 0.05. In both Severe vs Dengue Infection (DI) and Warning Sign vs DI comparisons. Among them, 20 miRNAs are downregulated, indicating a decrease in their expression levels. In contrast, 10 miRNAs are upregulated, showing increased expression levels. Enrichment analysis of predicted targets for these DEmiRs was performed through Diana Tools miRPath v4 [
14]. Enrichment terms associated with DENV manifestations are illustrated in the
Figure 2.
Regarding hypoxia, it has been demonstrated that the secretion and composition of exosomes are closely influenced by oxygen pressure in the microenvironment. As reviewed by Yaghoubi et al (2020), several studies suggest that cells respond to hypoxia by altering the molecular profile of exosomes [
99]. In vitro analyses further revealed that monocytes under hypoxic conditions required higher concentrations of antibodies for DENV neutralization [
100]. For Stat5, members of the JAK-STAT signaling pathway play a critical role in the interferon-induced inflammatory response, driving the expression of antiviral and pro-inflammatory proteins during DENV infection [
101]. Interestingly, the inhibition of Janus Kinase (JAK), which prevents interferon signaling, has shown therapeutic effects in experimental model of arenavirus hemorrhagic fever, suggesting potential applicability to DENV infections [
102].
Concerning TNFα, blocking TNFα alone reduced plasma leakage, while co-treatment with sunitinib significantly improved the survival of DENV-infected mice without affecting viremia [
103]. DENV entry via ADE has been shown to suppress the host cell's antiviral response by inhibiting IFN-α production while inducing IL-10 expression, thereby facilitating increased viral replication. This aligns with clinical observations of higher viremia, elevated IL-10 levels, and reduced IFN levels in patients with severe dengue [
104]. Additionally, interleukins 4, 6, 10, and 13 were found to be significantly elevated in EVs derived from patients with severe dengue [
64].
These findings underscore the importance of miRNAs in modulating critical biological processes during DENV infection, including immune responses, cytokine regulation, and cellular signaling pathways. The observed changes in miRNA expression, coupled with their enrichment in pathways linked to hypoxia, JAK-STAT signaling, and pro-inflammatory cytokines, suggest that miRNAs may serve as potential biomarkers for disease severity and therapeutic targets.
Figure 2.
Impact of dengue virus infection - Role of the NS1 Protein, Immune Signaling, and Extracellular Vesicles: (A) Dengue transmission occurs through the bite of the Aedes aegypti mosquito, the vector of the dengue virus, a positive-sense single-stranded RNA virus belonging to the genus Flavivirus. The virus encodes the non-structural protein 1 (NS1), a multifunctional glycoprotein essential for viral replication and pathogenesis, contributing to endothelial dysfunction and immune modulation. (B) Following viral infection, a signaling cascade is activated, involving the recruitment of immune cells and increased vascular permeability. This response induces cytoskeletal alterations, oxidative stress, and inflammatory signaling mediated by the activation of Toll-like receptors (TLRs). These receptors ultimately lead to the activation of the inflammatory pathway regulated by nuclear factor kappa B (NF-κB), promoting the immune response and the transcription of related genes such as interferons (INF), tumor necrosis factor (TNF), and interleukins (IL-6 and IL-1β). (C) Immunologically active cells may undergo apoptosis, releasing apoptotic bodies, or activate paracrine signaling pathways mediated by extracellular vesicles, including exosomes. These exosomes transport RNA, microRNAs, and the viral NS1 protein itself, playing an immunomodulatory role through receptors such as MHC-I, MHC-II, and PDL-1. These mechanisms contribute to the propagation of inflammatory signaling and the modulation of the immune response during dengue virus infection.
Figure 2.
Impact of dengue virus infection - Role of the NS1 Protein, Immune Signaling, and Extracellular Vesicles: (A) Dengue transmission occurs through the bite of the Aedes aegypti mosquito, the vector of the dengue virus, a positive-sense single-stranded RNA virus belonging to the genus Flavivirus. The virus encodes the non-structural protein 1 (NS1), a multifunctional glycoprotein essential for viral replication and pathogenesis, contributing to endothelial dysfunction and immune modulation. (B) Following viral infection, a signaling cascade is activated, involving the recruitment of immune cells and increased vascular permeability. This response induces cytoskeletal alterations, oxidative stress, and inflammatory signaling mediated by the activation of Toll-like receptors (TLRs). These receptors ultimately lead to the activation of the inflammatory pathway regulated by nuclear factor kappa B (NF-κB), promoting the immune response and the transcription of related genes such as interferons (INF), tumor necrosis factor (TNF), and interleukins (IL-6 and IL-1β). (C) Immunologically active cells may undergo apoptosis, releasing apoptotic bodies, or activate paracrine signaling pathways mediated by extracellular vesicles, including exosomes. These exosomes transport RNA, microRNAs, and the viral NS1 protein itself, playing an immunomodulatory role through receptors such as MHC-I, MHC-II, and PDL-1. These mechanisms contribute to the propagation of inflammatory signaling and the modulation of the immune response during dengue virus infection.
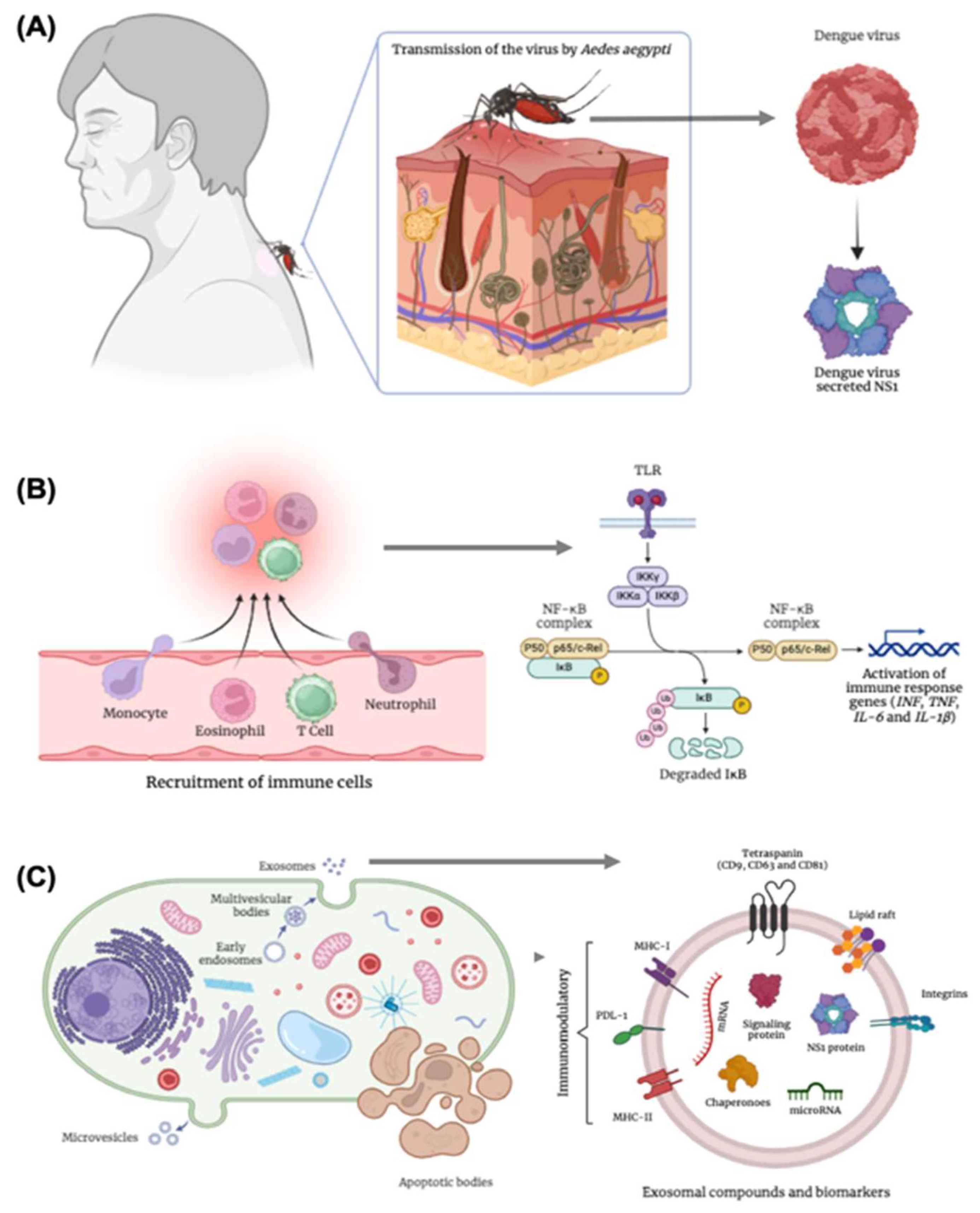
Figure 3.
Analysis of circulating miRNAs in dengue infection (GSE150623) reveal 30 miRNAs differentially expressed (|LogFC| > 2 and Adjusted p-value < 0.05) mutually in severe and warning signal cases compared to dengue infection without signs.
Figure 3.
Analysis of circulating miRNAs in dengue infection (GSE150623) reveal 30 miRNAs differentially expressed (|LogFC| > 2 and Adjusted p-value < 0.05) mutually in severe and warning signal cases compared to dengue infection without signs.
Potential therapeutic strategies based on current knowledge of EVs
Extracellular vesicles, including exosomes, microvesicles, and apoptotic bodies, have gained significant attention for their therapeutic potential. These vesicles naturally facilitate intercellular communication by transferring bioactive molecules such as proteins, RNAs, and lipids. Their ability to influence various biological processes like immune modulation, tissue regeneration, and disease progression has led to the exploration of several therapeutic strategies.
One promising approach involves the use of extracellular vesicles (EVs) as drug delivery vehicles. Their ability to encapsulate therapeutic cargo and protect it from degradation, along with their inherent biocompatibility and capacity to cross biological barriers, makes them ideal candidates for targeted drug delivery. EVs can be engineered to carry chemotherapeutic agents, gene-editing tools, or immune-modulating molecules, demonstrating significant potential in the treatment of cancer, genetic disorders, autoimmune diseases, and infectious diseases.
Extracellular vesicles have emerged as versatile tools in therapeutic development due to their ability to modulate immune responses, promote tissue regeneration, and serve as non-invasive biomarkers. For instance, EVs derived from regulatory T cells or mesenchymal stem cells can suppress inflammation and enhance immune tolerance, making them valuable in the treatment of autoimmune diseases. Conversely, tumor-derived EVs can be engineered to enhance anti-tumor immune responses. In regenerative medicine, EVs facilitate tissue repair by delivering growth factors, cytokines, and mRNAs that stimulate cell proliferation and modulate inflammation particularly mesenchymal stem cell-derived EVs, which have been studied for applications in cardiovascular disease, stroke, and bone healing. Additionally, the molecular cargo of EVs reflects the physiological state of their cells of origin, making them promising candidates as biomarkers for early diagnosis and disease monitoring in conditions such as cancer. Their surface can also be modified with ligands (e.g., antibodies or peptides) to enable precise therapeutic targeting, thereby minimizing off-target effects and improving treatment efficacy.
In the context of dengue virus infection, EVs mainly exosome offer novel therapeutic opportunities. EVs derived from dendritic cells have been shown to activate CD4+ T cells and modulate Th1 or Th2 responses depending on their size. Antigenic stimulation enhances the incorporation of MHC class II molecules into these vesicles, and their uptake by recipient dendritic cells further amplifies T cell activation and immune modulation. Building on these findings, the targeted loading of dengue virus antigens into exosomes may represent a promising strategy to stimulate both CD4+ and CD8+ T cell responses, thereby strengthening the adaptive immune response. This approach could improve antigen presentation and guide T cell functional polarization, offering potential applications in vaccine development or immunotherapeutic interventions for dengue and other viral infections.
Moreover, EVs can be engineered to deliver antisense oligonucleotides that inhibit microRNAs essential for viral replication. A precedent for this strategy is provided by Santaris Pharma, which developed an anti-miR compound targeting miR-122 a microRNA critical for hepatitis C virus replication. This compound demonstrated safety and antiviral efficacy in phase II clinical trials. Adapting such approaches to dengue could enable the development of innovative antiviral therapies using antisense oligonucleotides, provided that their safety, pharmacodynamics, and pharmacokinetics are thoroughly validated through dose-escalation studies.
14. Concluding Remarks
An expanding body of evidence underscores the critical role of extracellular vesicles (EVs) in the pathogenesis and progression of dengue virus (DENV) infection. EVs actively participate in viral dissemination, immune modulation, and endothelial dysfunction key features of dengue pathophysiology, especially in severe cases such as dengue hemorrhagic fever and dengue shock syndrome.
In dengue patients, circulating EVs contribute to immune dysregulation by transporting immunosuppressive molecules, including checkpoint inhibitors such as PD-L1 and anti-inflammatory cytokines like IL-6 and IL-10. These components suppress antiviral immune responses and facilitate viral persistence. Furthermore, EVs carry microRNAs (miRNAs) such as miR-146a and miR-21, which are known to reprogram host cell signaling pathways, promoting immune evasion while exacerbating inflammatory cascades. In severe disease, EV-associated miRNAs and viral proteins, particularly NS1 compromise endothelial integrity through Toll-like receptor (TLR) activation and reactive oxygen species (ROS) signaling, leading to vascular hyperpermeability and plasma leakage.
Beyond their pathogenic role, EVs hold great promise as diagnostic and prognostic biomarkers. Elevated EV concentrations and their molecular cargo, including PD-L1, miR-21, and miR-126-5p, have been correlated with higher viral loads and poor clinical outcomes. These findings support their potential as tools for early detection and risk stratification in dengue.
EVs also represent a promising platform for therapeutic development. Their biocompatibility, capacity for targeted delivery, and ability to cross biological barriers make them suitable for delivering antiviral agents, immunomodulatory molecules, or antisense oligonucleotides designed to suppress viral replication. For instance, targeted delivery of dengue virus antigens via exosomes may enhance CD4+ and CD8+ T cell responses, improving adaptive immunity. Likewise, miRNA-based interventions such as antisense strategies modeled after those used in hepatitis C virus could be adapted for dengue therapy, pending comprehensive pharmacological validation.
Ongoing technological advancements further enhance the feasibility of EV-based applications. The development of microfluidic devices, biosensors, and point-of-care diagnostic platforms is paving the way for rapid, cost-effective detection of EV-associated biomarkers, particularly in resource-limited settings. Simultaneously, progress in standardizing EV isolation and miRNA profiling will strengthen their clinical utility.
Author Contributions
All authors contributed equally to the conceptualization, methodology, search, analysis, and writing of the article. Supervision, review, and editing were carried out by JSHA and CJCF. Funding acquisition: JSHA. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the Ministry of Science and Technology of Colombia under Call 890 of 2020 (Code: 82613) and additionally funded by the Central Unit of Valle del Cauca (Colombia) through Internal Call No. 17.
Data Availability Statement
Bioinformatics analyses performed in this article are available upon request. Inquiries should be directed to jshenao@uceva.edu.co.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Murugesan, A.; Manoharan, M. Dengue Virus. In Emerging and Reemerging Viral Pathogens; Elsevier, 2020; pp. 281–359 ISBN 978-0-12-819400-3.
- Ebi, K.L.; Nealon, J. Dengue in a Changing Climate. Environ Res 2016, 151, 115–123. [CrossRef]
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The Global Distribution and Burden of Dengue. Nature 2013, 496, 504–507. [CrossRef]
- Sarker, R.; Roknuzzaman, A.S.M.; Haque, M.A.; Islam, M.R.; Kabir, E.R. Upsurge of Dengue Outbreaks in Several WHO Regions: Public Awareness, Vector Control Activities, and International Collaborations Are Key to Prevent Spread. Health Sci Rep 2024, 7, e2034. [CrossRef]
- Dengue Multi-Country Grade 3 Outbreak 2024 - PAHO/WHO | Pan American Health Organization Available online: https://www.paho.org/en/topics/dengue/dengue-multi-country-grade-3-outbreak (accessed on 31 December 2024).
- Kularatne, S.A.; Dalugama, C. Dengue Infection: Global Importance, Immunopathology and Management. Clin Med (Lond) 2022, 22, 9–13. [CrossRef]
- Singh, R.K.; Tiwari, A.; Satone, P.D.; Priya, T.; Meshram, R.J. Updates in the Management of Dengue Shock Syndrome: A Comprehensive Review. Cureus 2023, 15, e46713. [CrossRef]
- Bhatt, P.; Sabeena, S.P.; Varma, M.; Arunkumar, G. Current Understanding of the Pathogenesis of Dengue Virus Infection. Curr Microbiol 2021, 78, 17–32. [CrossRef]
- Malavige, G.N.; Ogg, G.S. Pathogenesis of Vascular Leak in Dengue Virus Infection. Immunology 2017, 151, 261–269. [CrossRef]
- Mishra, R.; Lata, S.; Ali, A.; Banerjea, A.C. Dengue Haemorrhagic Fever: A Job Done via Exosomes? Emerg Microbes Infect 2019, 8, 1626–1635. [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma Powers Differential Expression Analyses for RNA-Sequencing and Microarray Studies. Nucleic Acids Research 2015, 43, e47–e47. [CrossRef]
- R Core Team R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2023;.
- Tastsoglou, S.; Skoufos, G.; Miliotis, M.; Karagkouni, D.; Koutsoukos, I.; Karavangeli, A.; Kardaras, F.S.; Hatzigeorgiou, A.G. DIANA-miRPath v4.0: Expanding Target-Based miRNA Functional Analysis in Cell-Type and Tissue Contexts. Nucleic Acids Research 2023, 51, W154–W159. [CrossRef]
- Dwivedi, V.D.; Tripathi, I.P.; Tripathi, R.C.; Bharadwaj, S.; Mishra, S.K. Genomics, Proteomics and Evolution of Dengue Virus. Brief Funct Genomics 2017, 16, 217–227. [CrossRef]
- Guzman, M.G.; Gubler, D.J.; Izquierdo, A.; Martinez, E.; Halstead, S.B. Dengue Infection. Nat Rev Dis Primers 2016, 2, 1–25. [CrossRef]
- Beltrán-Silva, S.L.; Chacón-Hernández, S.S.; Moreno-Palacios, E.; Pereyra-Molina, J.Á. Clinical and Differential Diagnosis: Dengue, Chikungunya and Zika. Revista Médica del Hospital General de México 2018, 81, 146–153. [CrossRef]
- Tejo, A.M.; Hamasaki, D.T.; Menezes, L.M.; Ho, Y.-L. Severe Dengue in the Intensive Care Unit. J Intensive Med 2024, 4, 16–33. [CrossRef]
- Tam, D.T.H.; Clapham, H.; Giger, E.; Kieu, N.T.T.; Nam, N.T.; Hong, D.T.T.; Nuoi, B.T.; Cam, N.T.H.; Quyen, N.T.H.; Turner, H.C.; et al. Burden of Postinfectious Symptoms after Acute Dengue, Vietnam. Emerg Infect Dis 2023, 29, 160–163. [CrossRef]
- Kok, B.H.; Lim, H.T.; Lim, C.P.; Lai, N.S.; Leow, C.Y.; Leow, C.H. Dengue Virus Infection - a Review of Pathogenesis, Vaccines, Diagnosis and Therapy. Virus Res 2023, 324, 199018. [CrossRef]
- St. John, A.L.; Rathore, A.P.S. Adaptive Immune Responses to Primary and Secondary Dengue Virus Infections. Nat Rev Immunol 2019, 19, 218–230. [CrossRef]
- Lee, M.F.; Voon, G.Z.; Lim, H.X.; Chua, M.L.; Poh, C.L. Innate and Adaptive Immune Evasion by Dengue Virus. Front. Cell. Infect. Microbiol. 2022, 12. [CrossRef]
- Latanova, A.; Karpov, V.; Starodubova, E. Extracellular Vesicles in Flaviviridae Pathogenesis: Their Roles in Viral Transmission, Immune Evasion, and Inflammation. Int J Mol Sci 2024, 25, 2144. [CrossRef]
- Liu, Y.-J.; Wang, C. A Review of the Regulatory Mechanisms of Extracellular Vesicles-Mediated Intercellular Communication. Cell Communication and Signaling 2023, 21, 77. [CrossRef]
- Aloi, N.; Drago, G.; Ruggieri, S.; Cibella, F.; Colombo, P.; Longo, V. Extracellular Vesicles and Immunity: At the Crossroads of Cell Communication. Int J Mol Sci 2024, 25, 1205. [CrossRef]
- Berumen Sánchez, G.; Bunn, K.E.; Pua, H.H.; Rafat, M. Extracellular Vesicles: Mediators of Intercellular Communication in Tissue Injury and Disease. Cell Communication and Signaling 2021, 19, 104. [CrossRef]
- Tetta, C.; Ghigo, E.; Silengo, L.; Deregibus, M.C.; Camussi, G. Extracellular Vesicles as an Emerging Mechanism of Cell-to-Cell Communication. Endocrine 2013, 44, 11–19. [CrossRef]
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol Pathol 2007, 35, 495–516. [CrossRef]
- Wu, S.; Yang, T.; Ma, M.; Fan, L.; Ren, L.; Liu, G.; Wang, Y.; Cheng, B.; Xia, J.; Hao, Z. Extracellular Vesicles Meet Mitochondria: Potential Roles in Regenerative Medicine. Pharmacological Research 2024, 206, 107307. [CrossRef]
- Yu, L.; Zhu, G.; Zhang, Z.; Yu, Y.; Zeng, L.; Xu, Z.; Weng, J.; Xia, J.; Li, J.; Pathak, J.L. Apoptotic Bodies: Bioactive Treasure Left behind by the Dying Cells with Robust Diagnostic and Therapeutic Application Potentials. Journal of Nanobiotechnology 2023, 21, 218. [CrossRef]
- Nagata, S.; Sakuragi, T.; Segawa, K. Flippase and Scramblase for Phosphatidylserine Exposure. Curr Opin Immunol 2020, 62, 31–38. [CrossRef]
- Clancy, J.W.; Schmidtmann, M.; D’Souza-Schorey, C. The Ins and Outs of Microvesicles. FASEB Bioadv 2021, 3, 399–406. [CrossRef]
- Ramachandran, C.; Patil, R.V.; Combrink, K.; Sharif, N.A.; Srinivas, S.P. Rho-Rho Kinase Pathway in the Actomyosin Contraction and Cell-Matrix Adhesion in Immortalized Human Trabecular Meshwork Cells. Mol Vis 2011, 17, 1877–1890.
- Sakurada, S.; Takuwa, N.; Sugimoto, N.; Wang, Y.; Seto, M.; Sasaki, Y.; Takuwa, Y. Ca2+-Dependent Activation of Rho and Rho Kinase in Membrane Depolarization-Induced and Receptor Stimulation-Induced Vascular Smooth Muscle Contraction. Circ Res 2003, 93, 548–556. [CrossRef]
- Lee, Y.J.; Shin, K.J.; Jang, H.-J.; Ryu, J.-S.; Lee, C.Y.; Yoon, J.H.; Seo, J.K.; Park, S.; Lee, S.; Je, A.R.; et al. GPR143 Controls ESCRT-Dependent Exosome Biogenesis and Promotes Cancer Metastasis. Developmental Cell 2023, 58, 320-334.e8. [CrossRef]
- Ostrowski, M.; Carmo, N.B.; Krumeich, S.; Fanget, I.; Raposo, G.; Savina, A.; Moita, C.F.; Schauer, K.; Hume, A.N.; Freitas, R.P.; et al. Rab27a and Rab27b Control Different Steps of the Exosome Secretion Pathway. Nat Cell Biol 2010, 12, 19–30; sup pp 1-13. [CrossRef]
- Villarroya-Beltri, C.; Baixauli, F.; Gutiérrez-Vázquez, C.; Sánchez-Madrid, F.; Mittelbrunn, M. Sorting It out: Regulation of Exosome Loading. Semin Cancer Biol 2014, 28, 3–13. [CrossRef]
- Kumar, M.A.; Baba, S.K.; Sadida, H.Q.; Marzooqi, S.A.; Jerobin, J.; Altemani, F.H.; Algehainy, N.; Alanazi, M.A.; Abou-Samra, A.-B.; Kumar, R.; et al. Extracellular Vesicles as Tools and Targets in Therapy for Diseases. Sig Transduct Target Ther 2024, 9, 1–41. [CrossRef]
- Yoshikawa, F.S.Y.; Teixeira, F.M.E.; Sato, M.N.; Oliveira, L.M. da S. Delivery of microRNAs by Extracellular Vesicles in Viral Infections: Could the News Be Packaged? Cells 2019, 8, 611. [CrossRef]
- Singh, S.; Chen, C.C.; Kim, S.; Singh, A.; Singh, G. Role of Extracellular Vesicle microRNAs and RNA Binding Proteins on Glioblastoma Dynamics and Therapeutics Development. Extracellular Vesicle 2024, 4, 100049. [CrossRef]
- Safadi, D.E.; Lebeau, G.; Lagrave, A.; Mélade, J.; Grondin, L.; Rosanaly, S.; Begue, F.; Hoareau, M.; Veeren, B.; Roche, M.; et al. Extracellular Vesicles Are Conveyors of the NS1 Toxin during Dengue Virus and Zika Virus Infection. Viruses 2023, 15, 364. [CrossRef]
- Su, Y.; Lin, T.; Liu, C.; Cheng, C.; Han, X.; Jiang, X. microRNAs, the Link Between Dengue Virus and the Host Genome. Front Microbiol 2021, 12, 714409. [CrossRef]
- Wong, R.R.; Abd-Aziz, N.; Affendi, S.; Poh, C.L. Role of microRNAs in Antiviral Responses to Dengue Infection. Journal of Biomedical Science 2020, 27, 4. [CrossRef]
- Kanokudom, S.; Vilaivan, T.; Wikan, N.; Thepparit, C.; Smith, D.R.; Assavalapsakul, W. miR-21 Promotes Dengue Virus Serotype 2 Replication in HepG2 Cells. Antiviral Res 2017, 142, 169–177. [CrossRef]
- Fu, Y.; Zhang, L.; Zhang, F.; Tang, T.; Zhou, Q.; Feng, C.; Jin, Y.; Wu, Z. Exosome-Mediated miR-146a Transfer Suppresses Type I Interferon Response and Facilitates EV71 Infection. PLoS Pathog 2017, 13, e1006611. [CrossRef]
- Mao, L.; Chen, Y.; Gu, J.; Zhao, Y.; Chen, Q. Roles and Mechanisms of Exosomal microRNAs in Viral Infections. Arch Virol 2023, 168, 121. [CrossRef]
- Martins, S. de T.; Kuczera, D.; Lötvall, J.; Bordignon, J.; Alves, L.R. Characterization of Dendritic Cell-Derived Extracellular Vesicles During Dengue Virus Infection. Front Microbiol 2018, 9, 1792. [CrossRef]
- Sorop, A.; Iacob, R.; Iacob, S.; Constantinescu, D.; Chitoiu, L.; Fertig, T.E.; Dinischiotu, A.; Chivu-Economescu, M.; Bacalbasa, N.; Savu, L.; et al. Plasma Small Extracellular Vesicles Derived miR-21-5p and miR-92a-3p as Potential Biomarkers for Hepatocellular Carcinoma Screening. Front Genet 2020, 11, 712. [CrossRef]
- Sriprapun, M.; Rattanamahaphoom, J.; Sriburin, P.; Chatchen, S.; Limkittikul, K.; Sirivichayakul, C. The Expression of Circulating Hsa-miR-126-3p in Dengue-Infected Thai Pediatric Patients. Pathog Glob Health 2023, 117, 76–84. [CrossRef]
- Aloia, A.L.; Abraham, A.M.; Bonder, C.S.; Pitson, S.M.; Carr, J.M. Dengue Virus-Induced Inflammation of the Endothelium and the Potential Roles of Sphingosine Kinase-1 and MicroRNAs. Mediators Inflamm 2015, 2015, 509306. [CrossRef]
- Cloherty, A.P.M.; Rader, A.G.; Patel, K.S.; Eisden, T.-J.T.H.D.; van Piggelen, S.; Schreurs, R.R.C.E.; Ribeiro, C.M.S. Dengue Virus Exploits Autophagy Vesicles and Secretory Pathways to Promote Transmission by Human Dendritic Cells. Front Immunol 2024, 15, 1260439. [CrossRef]
- Wu, X.; Niu, J.; Shi, Y. Exosomes Target HBV-Host Interactions to Remodel the Hepatic Immune Microenvironment. Journal of Nanobiotechnology 2024, 22, 315. [CrossRef]
- Chapter 3 - Pathogenesis of Viral Infections and Diseases. In Fenner’s Veterinary Virology (Fourth Edition); MacLachlan, N.J., Dubovi, E.J., Eds.; Academic Press: San Diego, 2011; pp. 43–74 ISBN 978-0-12-375158-4.
- Klasse, P.J. The Molecular Basis of Viral Infection; Academic Press, 2015;.
- Tisoncik, J.R.; Billharz, R.; Burmakina, S.; Belisle, S.E.; Proll, S.C.; Korth, M.J.; García-Sastre, A.; Katze, M.G. The NS1 Protein of Influenza A Virus Suppresses Interferon-Regulated Activation of Antigen-Presentation and Immune-Proteasome Pathways. J Gen Virol 2011, 92, 2093–2104. [CrossRef]
- Caobi, A.; Nair, M.; Raymond, A.D. Extracellular Vesicles in the Pathogenesis of Viral Infections in Humans. Viruses 2020, 12, 1200. [CrossRef]
- Fernández-Messina, L.; Gutiérrez-Vázquez, C.; Rivas-García, E.; Sánchez-Madrid, F.; de la Fuente, H. Immunomodulatory Role of microRNAs Transferred by Extracellular Vesicles. Biol Cell 2015, 107, 61–77. [CrossRef]
- Torri, A.; Carpi, D.; Bulgheroni, E.; Crosti, M.-C.; Moro, M.; Gruarin, P.; Rossi, R.L.; Rossetti, G.; Di Vizio, D.; Hoxha, M.; et al. Extracellular MicroRNA Signature of Human Helper T Cell Subsets in Health and Autoimmunity. J Biol Chem 2017, 292, 2903–2915. [CrossRef]
- Nail, H.M.; Chiu, C.-C.; Leung, C.-H.; Ahmed, M.M.M.; Wang, H.-M.D. Exosomal miRNA-Mediated Intercellular Communications and Immunomodulatory Effects in Tumor Microenvironments. Journal of Biomedical Science 2023, 30, 69. [CrossRef]
- Wang, S.; Sun, J.; Dastgheyb, R.M.; Li, Z. Tumor-Derived Extracellular Vesicles Modulate Innate Immune Responses to Affect Tumor Progression. Front Immunol 2022, 13, 1045624. [CrossRef]
- Perez-Toledo, M.; Beristain-Covarrubias, N. A New Player in the Game: Platelet-Derived Extracellular Vesicles in Dengue Hemorrhagic Fever. Platelets 2020, 31, 412–414. [CrossRef]
- Vedpathak, S.; Sharma, A.; Palkar, S.; Bhatt, V.R.; Patil, V.C.; Kakrani, A.L.; Mishra, A.; Bhosle, D.; Arankalle, V.A.; Shrivastava, S. Platelet Derived Exosomes Disrupt Endothelial Cell Monolayer Integrity and Enhance Vascular Inflammation in Dengue Patients. Front. Immunol. 2024, 14. [CrossRef]
- Pradhan, A.; Aneja, A.; Ghosh, S.; Devvanshi, H.; C., D.; Sahu, R.; Ross, C.; Kshetrapal, P.; Maitra, A.; Das, S. Association of Exosomal miR-96-5p and miR-146a-5p with the Disease Severity in Dengue Virus Infection. Journal of Medical Virology 2023, 95, e28614. [CrossRef]
- Kumari, S.; Bandyopadhyay, B.; Singh, A.; Aggarwal, S.; Yadav, A.K.; Vikram, N.K.; Guchhait, P.; Banerjee, A. Extracellular Vesicles Recovered from Plasma of Severe Dengue Patients Induce CD4+ T Cell Suppression through PD-L1/PD-1 Interaction. mBio 2023, 14, e01823-23. [CrossRef]
- Mishra, R.; Lahon, A.; Banerjea, A.C. Dengue Virus Degrades USP33–ATF3 Axis via Extracellular Vesicles to Activate Human Microglial Cells. The Journal of Immunology 2020, 205, 1787–1798. [CrossRef]
- Gold, A.S.; Feitosa-Suntheimer, F.; Araujo, R.V.; Hekman, R.M.; Asad, S.; Londono-Renteria, B.; Emili, A.; Colpitts, T.M. Dengue Virus Infection of Aedes Aegypti Alters Extracellular Vesicle Protein Cargo to Enhance Virus Transmission. International Journal of Molecular Sciences 2020, 21, 6609. [CrossRef]
- Liu, P.; Woda, M.; Ennis, F.A.; Libraty, D.H. Dengue Virus Infection Differentially Regulates Endothelial Barrier Function over Time through Type I Interferon Effects. J Infect Dis 2009, 200, 10.1086/599795. [CrossRef]
- Yacoub, S.; Wertheim, H.; Simmons, C.P.; Screaton, G.; Wills, B. Cardiovascular Manifestations of the Emerging Dengue Pandemic. Nat Rev Cardiol 2014, 11, 335–345. [CrossRef]
- Srikiatkhachorn, A.; Kelley, J.F. Endothelial Cells in Dengue Hemorrhagic Fever. Antiviral Res 2014, 109, 160–170. [CrossRef]
- Chatterjee, V.; Yang, X.; Ma, Y.; Wu, M.H.; Yuan, S.Y. Extracellular Vesicles: New Players in Regulating Vascular Barrier Function. Am J Physiol Heart Circ Physiol 2020, 319, H1181–H1196. [CrossRef]
- Wang, X.; He, B. Insight into Endothelial Cell-Derived Extracellular Vesicles in Cardiovascular Disease: Molecular Mechanisms and Clinical Implications. Pharmacol Res 2024, 207, 107309. [CrossRef]
- Guo, N.; Wang, Y.; Wen, Z.; Fan, X. Promising Nanotherapeutics of Stem Cell Extracellular Vesicles in Liver Regeneration. Regen Ther 2024, 26, 1037–1047. [CrossRef]
- Javdani-Mallak, A.; Salahshoori, I. Environmental Pollutants and Exosomes: A New Paradigm in Environmental Health and Disease. Sci Total Environ 2024, 925, 171774. [CrossRef]
- Li, Y.; Tan, J.; Miao, Y.; Zhang, Q. MicroRNA in Extracellular Vesicles Regulates Inflammation through Macrophages under Hypoxia. Cell Death Discov 2021, 7, 285. [CrossRef]
- Signorelli, S.S.; Mazzarino, M.C.; Di Pino, L.; Malaponte, G.; Porto, C.; Pennisi, G.; Marchese, G.; Costa, M.P.; Digrandi, D.; Celotta, G.; et al. High Circulating Levels of Cytokines (IL-6 and TNFalpha), Adhesion Molecules (VCAM-1 and ICAM-1) and Selectins in Patients with Peripheral Arterial Disease at Rest and after a Treadmill Test. Vasc Med 2003, 8, 15–19. [CrossRef]
- Chiaradia, E.; Tancini, B.; Emiliani, C.; Delo, F.; Pellegrino, R.M.; Tognoloni, A.; Urbanelli, L.; Buratta, S. Extracellular Vesicles under Oxidative Stress Conditions: Biological Properties and Physiological Roles. Cells 2021, 10, 1763. [CrossRef]
- Tang, F.; Zhao, X.-L.; Xu, L.-Y.; Zhang, J.-N.; Ao, H.; Peng, C. Endothelial Dysfunction: Pathophysiology and Therapeutic Targets for Sepsis-Induced Multiple Organ Dysfunction Syndrome. Biomed Pharmacother 2024, 178, 117180. [CrossRef]
- Meuren, L.M.; Prestes, E.B.; Papa, M.P.; de Carvalho, L.R.P.; Mustafá, Y.M.; da Costa, L.S.; Da Poian, A.T.; Bozza, M.T.; Arruda, L.B. Infection of Endothelial Cells by Dengue Virus Induces ROS Production by Different Sources Affecting Virus Replication, Cellular Activation, Death and Vascular Permeability. Front Immunol 2022, 13, 810376. [CrossRef]
- Ferrari, M.; Zevini, A.; Palermo, E.; Muscolini, M.; Alexandridi, M.; Etna, M.P.; Coccia, E.M.; Fernandez-Sesma, A.; Coyne, C.; Zhang, D.D.; et al. Dengue Virus Targets Nrf2 for NS2B3-Mediated Degradation Leading to Enhanced Oxidative Stress and Viral Replication. J Virol 2020, 94, e01551-20. [CrossRef]
- Park, J.-S.; Rustamov, N.; Roh, Y.-S. The Roles of NFR2-Regulated Oxidative Stress and Mitochondrial Quality Control in Chronic Liver Diseases. Antioxidants (Basel) 2023, 12, 1928. [CrossRef]
- Auber, M.; Svenningsen, P. An Estimate of Extracellular Vesicle Secretion Rates of Human Blood Cells. J Extracell Biol 2022, 1, e46. [CrossRef]
- Limothai, U.; Jantarangsi, N.; Suphavejkornkij, N.; Tachaboon, S.; Dinhuzen, J.; Chaisuriyong, W.; Trongkamolchai, S.; Wanpaisitkul, M.; Chulapornsiri, C.; Tiawilai, A.; et al. Discovery and Validation of Circulating miRNAs for the Clinical Prognosis of Severe Dengue. PLoS Negl Trop Dis 2022, 16, e0010836. [CrossRef]
- Ouyang, X.; Jiang, X.; Gu, D.; Zhang, Y.; Kong, S.K.; Jiang, C.; Xie, W. Dysregulated Serum MiRNA Profile and Promising Biomarkers in Dengue-Infected Patients. Int J Med Sci 2016, 13, 195–205. [CrossRef]
- Tambyah, P.A.; Ching, C.S.; Sepramaniam, S.; Ali, J.M.; Armugam, A.; Jeyaseelan, K. microRNA Expression in Blood of Dengue Patients. Ann Clin Biochem 2016, 53, 466–476. [CrossRef]
- Hapugaswatta, H.; Amarasena, P.; Premaratna, R.; Seneviratne, K.N.; Jayathilaka, N. Differential Expression of microRNA, miR-150 and Enhancer of Zeste Homolog 2 (EZH2) in Peripheral Blood Cells as Early Prognostic Markers of Severe Forms of Dengue. J Biomed Sci 2020, 27, 25. [CrossRef]
- Fernández-Hernando, C.; Suárez, Y. MicroRNAs in Endothelial Cell Homeostasis and Vascular Disease. Curr Opin Hematol 2018, 25, 227–236. [CrossRef]
- Pena-Philippides, J.C.; Gardiner, A.S.; Caballero-Garrido, E.; Pan, R.; Zhu, Y.; Roitbak, T. Inhibition of MicroRNA-155 Supports Endothelial Tight Junction Integrity Following Oxygen-Glucose Deprivation. Journal of the American Heart Association 2018, 7, e009244. [CrossRef]
- Punyadee, N.; Mairiang, D.; Thiemmeca, S.; Komoltri, C.; Pan-Ngum, W.; Chomanee, N.; Charngkaew, K.; Tangthawornchaikul, N.; Limpitikul, W.; Vasanawathana, S.; et al. Microparticles Provide a Novel Biomarker to Predict Severe Clinical Outcomes of Dengue Virus Infection. J Virol 2015, 89, 1587–1607. [CrossRef]
- König, L.; Kasimir-Bauer, S.; Bittner, A.-K.; Hoffmann, O.; Wagner, B.; Santos Manvailer, L.F.; Kimmig, R.; Horn, P.A.; Rebmann, V. Elevated Levels of Extracellular Vesicles Are Associated with Therapy Failure and Disease Progression in Breast Cancer Patients Undergoing Neoadjuvant Chemotherapy. OncoImmunology 2018, 7, e1376153. [CrossRef]
- Henao-Agudelo, J.S.; Ayala, S.; Badiel, M.; Zea-Vera, A.F.; Matta Cortes, L. Classical Monocytes-Low Expressing HLA-DR Is Associated with Higher Mortality Rate in SARS-CoV-2+ Young Patients with Severe Pneumonia. Heliyon 2024, 10, e24099. [CrossRef]
- Lucien, F.; Gustafson, D.; Lenassi, M.; Li, B.; Teske, J.J.; Boilard, E.; von Hohenberg, K.C.; Falcón-Perez, J.M.; Gualerzi, A.; Reale, A.; et al. MIBlood-EV: Minimal Information to Enhance the Quality and Reproducibility of Blood Extracellular Vesicle Research. J Extracell Vesicles 2023, 12, e12385. [CrossRef]
- Welsh, J.A.; Goberdhan, D.C.I.; O’Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal Information for Studies of Extracellular Vesicles (MISEV2023): From Basic to Advanced Approaches. J Extracell Vesicles 2024, 13, e12404. [CrossRef]
- Kowkabany, G.; Bao, Y. Nanoparticle Tracking Analysis: An Effective Tool to Characterize Extracellular Vesicles. Molecules 2024, 29, 4672. [CrossRef]
- Asish, P.R.; Dasgupta, S.; Rachel, G.; Bagepally, B.S.; Girish Kumar, C.P. Global Prevalence of Asymptomatic Dengue Infections - a Systematic Review and Meta-Analysis. Int J Infect Dis 2023, 134, 292–298. [CrossRef]
- Welsh, J.A.; Horak, P.; Wilkinson, J.S.; Ford, V.J.; Jones, J.C.; Smith, D.; Holloway, J.A.; Englyst, N.A. FCMPASS Software Aids Extracellular Vesicle Light Scatter Standardization. Cytometry A 2020, 97, 569–581. [CrossRef]
- Chen, H.-R.; Chuang, Y.-C.; Lin, Y.-S.; Liu, H.-S.; Liu, C.-C.; Perng, G.-C.; Yeh, T.-M. Dengue Virus Nonstructural Protein 1 Induces Vascular Leakage through Macrophage Migration Inhibitory Factor and Autophagy. PLoS Negl Trop Dis 2016, 10, e0004828. [CrossRef]
- Metz, S.W.; Thomas, A.; White, L.; Stoops, M.; Corten, M.; Hannemann, H.; de Silva, A.M. Dengue Virus-like Particles Mimic the Antigenic Properties of the Infectious Dengue Virus Envelope. Virology Journal 2018, 15, 60. [CrossRef]
- Saini, J.; Bandyopadhyay, B.; Pandey, A.D.; Ramachandran, V.G.; Das, S.; Sood, V.; Banerjee, A.; Vrati, S. High-Throughput RNA Sequencing Analysis of Plasma Samples Reveals Circulating microRNA Signatures with Biomarker Potential in Dengue Disease Progression. mSystems 2020, 5, e00724-20. [CrossRef]
- Yaghoubi, S.; Najminejad, H.; Dabaghian, M.; Karimi, M.H.; Abdollahpour-Alitappeh, M.; Rad, F.; Mahi-Birjand, M.; Mohammadi, S.; Mohseni, F.; Sobhani Lari, M.; et al. How Hypoxia Regulate Exosomes in Ischemic Diseases and Cancer Microenvironment? IUBMB Life 2020, 72, 1286–1305. [CrossRef]
- Gan, E.S.; Cheong, W.F.; Chan, K.R.; Ong, E.Z.; Chai, X.; Tan, H.C.; Ghosh, S.; Wenk, M.R.; Ooi, E.E. Hypoxia Enhances Antibody-dependent Dengue Virus Infection. The EMBO Journal 2017, 36, 1348–1363. [CrossRef]
- Roy, S.K.; Bhattacharjee, S. Dengue Virus: Epidemiology, Biology, and Disease Aetiology. Can. J. Microbiol. 2021, 67, 687–702. [CrossRef]
- Sahin, M.; Remy, M.M.; Merkler, D.; Pinschewer, D.D. The Janus Kinase Inhibitor Ruxolitinib Prevents Terminal Shock in a Mouse Model of Arenavirus Hemorrhagic Fever. Microorganisms 2021, 9, 564. [CrossRef]
- Branche, E.; Tang, W.W.; Viramontes, K.M.; Young, M.P.; Sheets, N.; Joo, Y.; Nguyen, A.-V.T.; Shresta, S. Synergism Between the Tyrosine Kinase Inhibitor Sunitinib and Anti-TNF Antibody Protects Against Lethal Dengue Infection. Antiviral Res 2018, 158, 1–7. [CrossRef]
- Srikiatkhachorn, A.; Mathew, A.; Rothman, A.L. Immune-Mediated Cytokine Storm and Its Role in Severe Dengue. Semin Immunopathol 2017, 39, 563–574. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).