Submitted:
21 April 2025
Posted:
22 April 2025
You are already at the latest version
Abstract
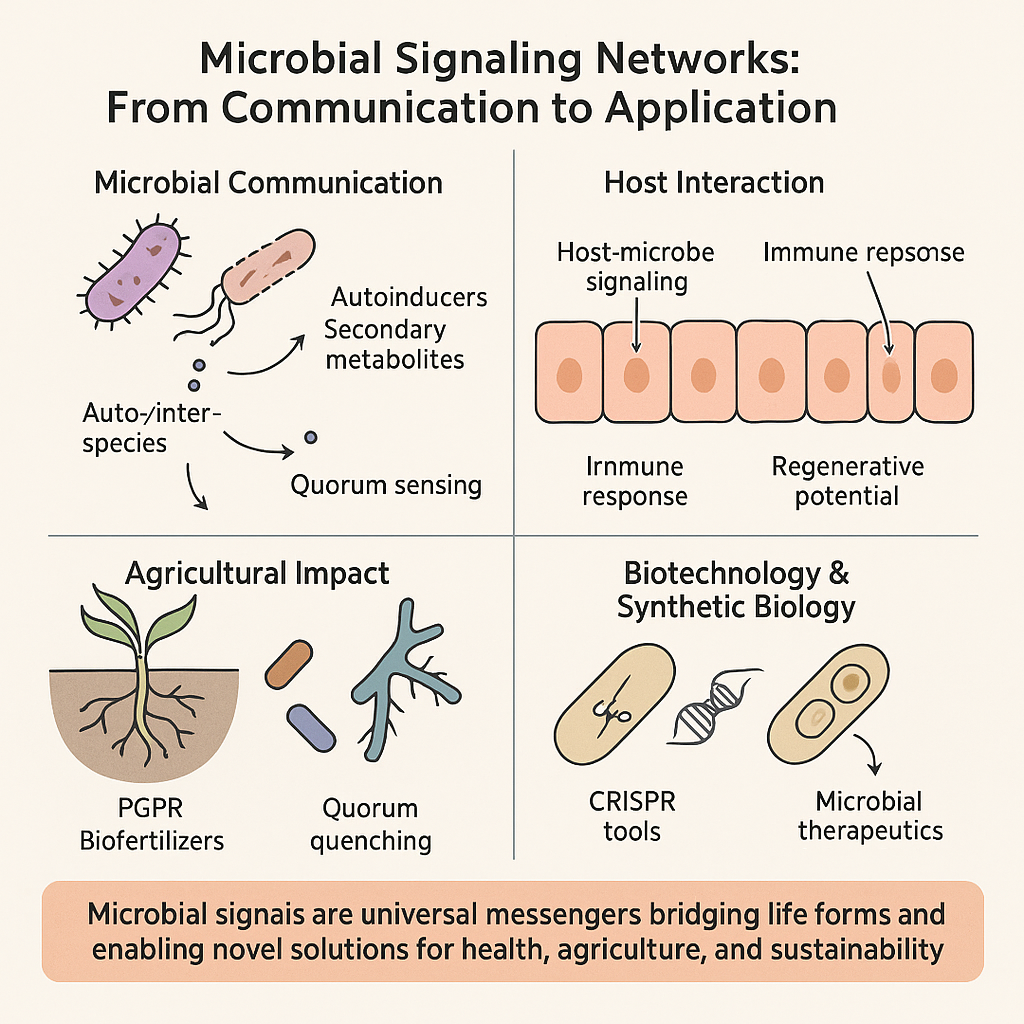
Keywords:
1. Introduction
2. Quorum Sensing and Autoinducers
2.1. Overview of Quorum Sensing
2.2. Diversity and Function of Autoinducers
2.3. Evolutionary Perspectives on Cell-Cell Signaling
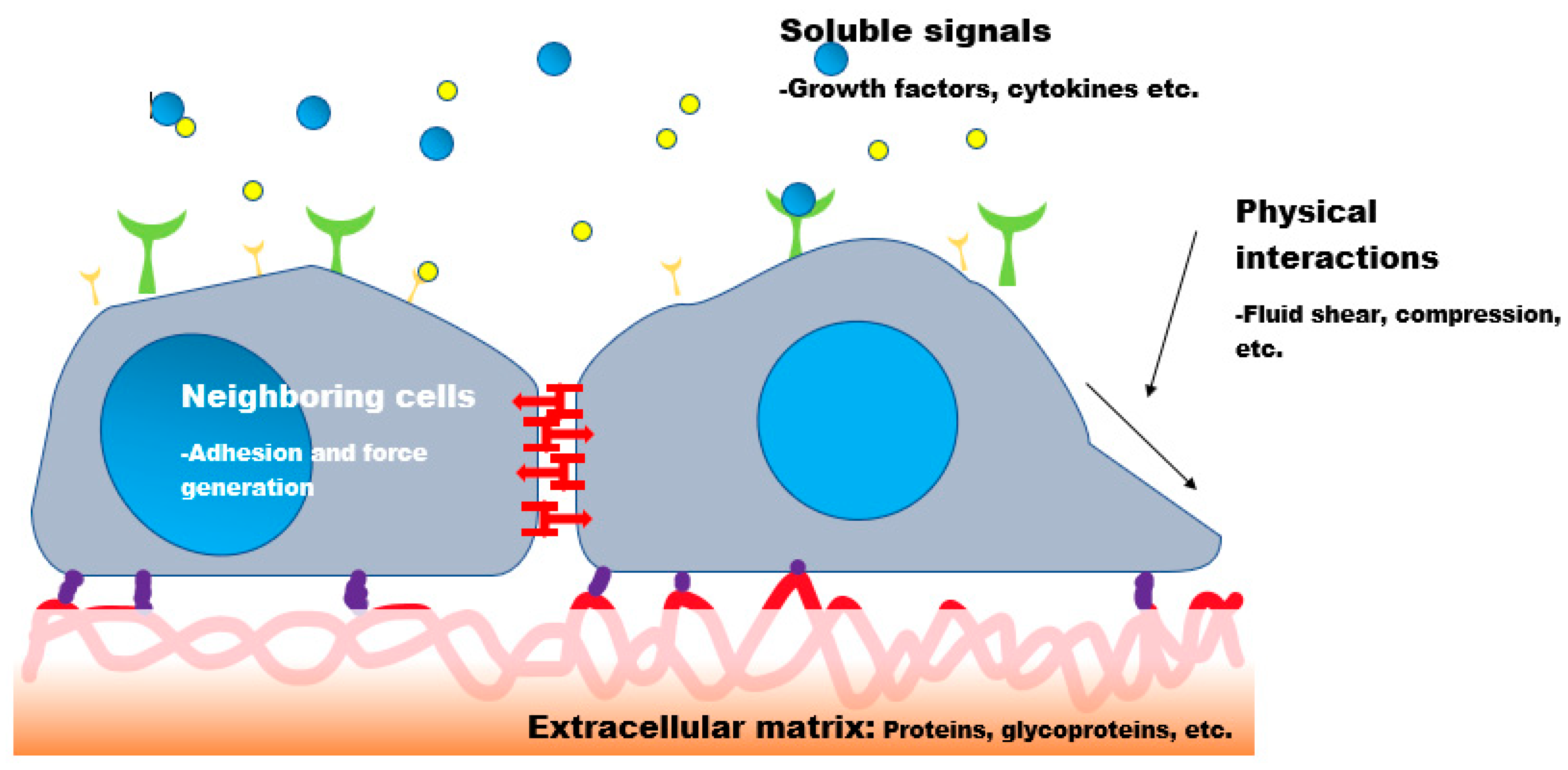
3. The Cellular Micro-environment and Host Modulation
3.1. Definition and Components of the Micro-Environment
3.2. Receptor-Mediated Signal Recognition
3.3. Cell-Micro-environment Interactions
3.4. Soluble Signaling Molecules and Autocrine/Paracrine Loops
3.5. Role of Extracellular Matrix (ECM)
4. Microbial Signaling in Agriculture
4.1. Plant-Microbe Interactions and Rhizosphere Communication
4.2. Role of Endophytes and Plant Growth Promoting Rhizobacteria (PGPR)
4.3. Quorum Sensing in Biocontrol and Biofertilizer Development
4.4. Inter-Kingdom Signaling Between Plants and Microbes
5. Synthetic and Genetic Engineering Applications
5.1. Engineered Microbial Products for Agriculture and Medicine
5.2. Signal Interference and Quorum Sensing Inhibition
5.3. CRISPR and Synthetic Biology for Signal Modulation
6. Inter-Kingdom Communication
6.1. Microbial Response to Host Hormones
6.2. Host Immune Sensing and Microbial Adaptation
6.3. Stress-Induced Microbial Virulence
7. Conclusions
Authors Contributions
Code availability
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgment
Conflict of Interest
References
- Abebe, G.M. Oral biofilm and its impact on oral health, psychological and social interaction. Int. J. Oral Dent. Health 2021, 7, 127–137. [Google Scholar]
- AlMusawi, S.; Ahmed, M.; Nateri, A.S. Understanding cell-cell communication and signaling in the colorectal cancer microenvironment. Clin. Transl. Med. 2021, 11, e308. [Google Scholar] [CrossRef] [PubMed]
- Balan, B.; Dhaulaniya, A.S.; Varma, D.A.; Sodhi, K.K.; Kumar, M.; Tiwari, M.; Singh, D.K. Microbial biofilm ecology, in silico study of quorum sensing receptor-ligand interactions and biofilm mediated bioremediation. Arch. Microbiol. 2020, 203, 13–30. [Google Scholar] [CrossRef] [PubMed]
- Brindhadevi, K.; LewisOscar, F.; Mylonakis, E.; Shanmugam, S.; Verma, T.N.; Pugazhendhi, A. Biofilm and Quorum sensing mediated pathogenicity in Pseudomonas aeruginosa. Process. Biochem. 2020, 96, 49–57. [Google Scholar] [CrossRef]
- Camilli, A.; Bassler, B.L. Bacterial Small-Molecule Signaling Pathways. Science 2006, 311, 1113–1116. [Google Scholar] [CrossRef]
- Chamkhi, I.; El Omari, N.; Benali, T.; Bouyahya, A. Quorum sensing and plant-bacteria interaction: role of quorum sensing in the rhizobacterial community colonization in the rhizosphere. In Quorum Sensing: Microbial Rules of Life; ACS Publications: 2020; pp. 139–153.
- Combarnous, Y.; Nguyen, T.M.D. Cell communications among microorganisms, plants, and animals: origin, evolution, and interplays. International Journal of Molecular Sciences 2020, 21, 8052. [Google Scholar] [CrossRef]
- Cui, F.; Zhou, Z.; Zhou, H.S. Molecularly Imprinted Polymers and Surface Imprinted Polymers Based Electrochemical Biosensor for Infectious Diseases. Sensors 2020, 20, 996. [Google Scholar] [CrossRef]
- Dalton, C.J.; Lemmon, C.A. Fibronectin: Molecular Structure, Fibrillar Structure and Mechanochemical Signaling. Cells 2021, 10, 2443. [Google Scholar] [CrossRef]
- Davares, A.K.L.; Arsene, M.M.J.; Viktorovna, P.I.; Vyacheslavovna, Y.N.; Vladimirovna, Z.A.; Aleksandrovna, V.E.; Nikolayevich, S.A.; Nadezhda, S.; Anatolievna, G.O.; Nikolaevna, S.I.; et al. Quorum-Sensing Inhibitors from Probiotics as a Strategy to Combat Bacterial Cell-to-Cell Communication Involved in Food Spoilage and Food Safety. Fermentation 2022, 8, 711. [Google Scholar] [CrossRef]
- Davies, D.G.; Parsek, M.R.; Pearson, J.P.; Iglewski, B.H.; Costerton, J.W.; Greenberg, E.P. The Involvement of Cell-to-Cell Signals in the Development of a Bacterial Biofilm. Science 1998, 280, 295–298. [Google Scholar] [CrossRef]
- DeMali, K.A.; Sun, X.; Bui, G.A. Force Transmission at Cell–Cell and Cell–Matrix Adhesions. Biochemistry 2014, 53, 7706–7717. [Google Scholar] [CrossRef] [PubMed]
- Duddy, O.P.; Bassler, B.L. Quorum sensing across bacterial and viral domains. PLOS Pathog. 2021, 17, e1009074. [Google Scholar] [CrossRef] [PubMed]
- Dufort, C.C.; Paszek, M.J.; Weaver, V.M. Balancing forces: Architectural control of mechanotransduction. Nat. Rev. Mol. Cell Biol. 2011, 12, 308–319. [Google Scholar] [CrossRef]
- Elvevoll, E.O.; James, D.; Toppe, J.; Gamarro, E.G.; Jensen, I.-J. Food Safety Risks Posed by Heavy Metals and Persistent Organic Pollutants (POPs) related to Consumption of Sea Cucumbers. Foods 2022, 11, 3992. [Google Scholar] [CrossRef]
- Gross, S.M.; Dane, M.A.; Smith, R.L.; Devlin, K.L.; McLean, I.C.; Derrick, D.S.; Mills, C.E.; Subramanian, K.; London, A.B.; Torre, D.; et al. A multi-omic analysis of MCF10A cells provides a resource for integrative assessment of ligand-mediated molecular and phenotypic responses. Commun. Biol. 2022, 5, 1066. [Google Scholar] [CrossRef] [PubMed]
- Groult, B.; Bredin, P.; Lazar, C.S. Ecological processes differ in community assembly of Archaea, Bacteria and Eukaryotes in a biogeographical survey of groundwater habitats in the Quebec region (Canada). Environ. Microbiol. 2022, 24, 5898–5910. [Google Scholar] [CrossRef]
- Hellmund, K.S.; Koksch, B. Self-Assembling Peptides as Extracellular Matrix Mimics to Influence Stem Cell's Fate. Front. Chem. 2019, 7, 172. [Google Scholar] [CrossRef]
- Hu, H.; Liu, Y.; Luo, F.; Zhang, X.; Wang, Y.; Ye, Z.; Chen, J.; Li, T. Stable and rapid partial nitrification achieved by boron stimulating autoinducer-2 mediated quorum sensing at room & low temperature. Chemosphere 2022, 304, 135327. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, L.; Wan, D.; Zhou, L.; Zheng, S.; Lin, S.; Qiao, Y. Extracellular matrix and its therapeutic potential for cancer treatment. Signal Transduct. Target. Ther. 2021, 6, 153. [Google Scholar] [CrossRef]
- Huang, S.; Liu, X.; Yang, W.; Ma, L.; Li, H.; Liu, R.; Qiu, J.; Li, Y. Insights into Adaptive Mechanisms of Extreme Acidophiles Based on Quorum Sensing/Quenching-Related Proteins. mSystems 2022, 7, e0149121. [Google Scholar] [CrossRef]
- Hughes, D.T.; Sperandio, V. Inter-kingdom signalling: communication between bacteria and their hosts. Nat. Rev. Microbiol. 2008, 6, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Hwang, O.-J. , & Back, K. (2022). Functional Characterization of Arylalkylamine N-Acetyltransferase, a Pivotal Gene in Antioxidant Melatonin Biosynthesis from Chlamydomonas reinhardtii. Antioxidants, 11(8), 1531.
- Im, G.-I. Biomaterials in orthopaedics: the past and future with immune modulation. Biomater. Res. 2020, 24, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Jin, T.; Patel, S.J.; Van Lehn, R.C. Molecular simulations of lipid membrane partitioning and translocation by bacterial quorum sensing modulators. PLOS ONE 2021, 16, e0246187. [Google Scholar] [CrossRef]
- Keller, K.E.; Peters, D.M. Pathogenesis of glaucoma: Extracellular matrix dysfunction in the trabecular meshwork-A review. Clin. Exp. Ophthalmol. 2022, 50, 163–182. [Google Scholar] [CrossRef] [PubMed]
- Kong, F. (2022). Identification and Characterization of Arabidopsis Toxicos en Levadura 12: A Gene Involved in Chitin-Elicitor-Triggered Immunity and Salt Tolerance. The University of Alabama.
- Legant, W.R.; Chen, C.S.; Vogel, V. Force-induced fibronectin assembly and matrix remodeling in a 3D microtissue model of tissue morphogenesis. Integr. Biol. 2012, 4, 1164–1174. [Google Scholar] [CrossRef]
- Lemmon, C.A.; Chen, C.S.; Romer, L.H. Cell Traction Forces Direct Fibronectin Matrix Assembly. Biophys. J. 2009, 96, 729–738. [Google Scholar] [CrossRef]
- Lu, P.; Takai, K.; Weaver, V.M.; Werb, Z. Extracellular Matrix Degradation and Remodeling in Development and Disease. Cold Spring Harb. Perspect. Biol. 2011, 3, a005058. [Google Scholar] [CrossRef]
- Miller, A. E. , Hu, P., & Barker, T. H. (2020). Feeling things out: bidirectional signaling of the cell–ECM interface, implications in the mechanobiology of cell spreading, migration, proliferation, and differentiation. Advanced Healthcare Materials, 9(8), 1901445.
- Nealson, K.H.; Platt, T.; Hastings, J.W. Cellular Control of the Synthesis and Activity of the Bacterial Luminescent System. J. Bacteriol. 1970, 104, 313–22. [Google Scholar] [CrossRef]
- Pan, J.; Zhou, J.; Tang, X.; Guo, Y.; Zhao, Y.; Liu, S. Bacterial Communication Coordinated Behaviors of Whole Communities to Cope with Environmental Changes. Environ. Sci. Technol. 2023, 57, 4253–4265. [Google Scholar] [CrossRef]
- Patra, D.; Mandal, S. Non-rhizobia are the alternative sustainable solution for growth and development of the nonlegume plants. Biotechnol. Genet. Eng. Rev. 2022, 39, 435–464. [Google Scholar] [CrossRef]
- Patrad, E.; Khalighfard, S.; Amiriani, T.; Khori, V.; Alizadeh, A.M. Molecular mechanisms underlying the action of carcinogens in gastric cancer with a glimpse into targeted therapy. Cell. Oncol. 2022, 45, 1073–1117. [Google Scholar] [CrossRef] [PubMed]
- Rosset, S.L.; Oakley, C.A.; Ferrier-Pagès, C.; Suggett, D.J.; Weis, V.M.; Davy, S.K. The Molecular Language of the Cnidarian–Dinoflagellate Symbiosis. Trends Microbiol. 2021, 29, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.; Chane, A.; Jung, M.; Lee, Y. Recent Advances in Understanding the Roles of Pectin as an Active Participant in Plant Signaling Networks. Plants 2021, 10, 1712. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Nakahata, M.; Linke, P.; Kaufmann, S. Stimuli-responsive hydrogels as a model of the dynamic cellular microenvironment. Polym. J. 2020, 52, 861–870. [Google Scholar] [CrossRef]
- Tang, M.; Liao, S.; Qu, J.; Liu, Y.; Han, S.; Cai, Z.; Fan, Y.; Yang, L.; Li, S.; Li, L. Evaluating Bacterial Pathogenesis Using a Model of Human Airway Organoids Infected with Pseudomonas aeruginosa Biofilms. Microbiol. Spectr. 2022, 10, e0240822. [Google Scholar] [CrossRef]
- Tang, Y.; Chen, H.; Lin, Z.; Zhang, L.; Upadhyay, A.; Liao, C.; Merkler, D.J.; Han, Q. Evolutionary genomics analysis reveals gene expansion and functional diversity of arylalkylamine N-acetyltransferases in the Culicinae subfamily of mosquitoes. Insect Sci. 2022, 30, 569–581. [Google Scholar] [CrossRef]
- Thomas, G.J.; Hart, I.R.; Speight, P.M.; Marshall, J.F. Binding of TGF-β1 latency-associated peptide (LAP) to αvβ6 integrin modulates behaviour of squamous carcinoma cells. Br. J. Cancer 2002, 87, 859–867. [Google Scholar] [CrossRef]
- Tyson, J.; Bundy, K.; Roach, C.; Douglas, H.; Ventura, V.; Segars, M.F.; Schwartz, O.; Simpson, C.L. Mechanisms of the Osteogenic Switch of Smooth Muscle Cells in Vascular Calcification: WNT Signaling, BMPs, Mechanotransduction, and EndMT. Bioengineering 2020, 7, 88. [Google Scholar] [CrossRef]
- Virdi, J.K.; Pethe, P. Biomaterials Regulate Mechanosensors YAP/TAZ in Stem Cell Growth and Differentiation. Tissue Eng. Regen. Med. 2021, 18, 199–215. [Google Scholar] [CrossRef]
- Wan, X.; Liu, Z.; Li, L. Manipulation of Stem Cells Fates: The Master and Multifaceted Roles of Biophysical Cues of Biomaterials. Adv. Funct. Mater. 2021, 31, 2010626. [Google Scholar] [CrossRef]
- Wang, S.; Payne, G.F.; Bentley, W.E. Quorum Sensing Communication: Molecularly Connecting Cells, Their Neighbors, and Even Devices. Annu. Rev. Chem. Biomol. Eng. 2020, 11, 447–468. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, W.; Han, Y.; Huang, X.; Wang, C.; Ma, K.; Kong, M.; Jiang, N.; Pan, J. Microbial diversity of archaeological ruins of Liangzhu City and its correlation with environmental factors. Int. Biodeterior. Biodegradation 2022, 175. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, B.; Grenier, D.; Yi, L. Regulatory Mechanisms of the LuxS/AI-2 System and Bacterial Resistance. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-J.; Li, X.; Wang, J.-H.; Qi, S.-S.; Dai, Z.-C.; Du, D.-L. Effect of nitrogen-fixing bacteria on resource investment of the root system in an invasive clonal plant under low nutritional environment. Flora 2022, 297. [Google Scholar] [CrossRef]
- Wiesmann, C.L.; Wang, N.R.; Zhang, Y.; Liu, Z.; Haney, C.H. Origins of symbiosis: shared mechanisms underlying microbial pathogenesis, commensalism and mutualism of plants and animals. FEMS Microbiol. Rev. 2022, 47. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S. Cell Wall Signaling in Plant Development and Defense. Annu. Rev. Plant Biol. 2022, 73, 323–353. [Google Scholar] [CrossRef]
- Wright, P.P.; Ramachandra, S.S. Quorum Sensing and Quorum Quenching with a Focus on Cariogenic and Periodontopathic Oral Biofilms. Microorganisms 2022, 10, 1783. [Google Scholar] [CrossRef]
- Wu, L.; Luo, Y. Bacterial Quorum-Sensing Systems and Their Role in Intestinal Bacteria-Host Crosstalk. Front. Microbiol. 2021, 12. [Google Scholar] [CrossRef]
- Xiao, L.; Sun, Y.; Liao, L.; Su, X. Response of mesenchymal stem cells to surface topography of scaffolds and the underlying mechanisms. J. Mater. Chem. B 2023, 11, 2550–2567. [Google Scholar] [CrossRef]
- Xing, H.; Lee, H.; Luo, L.; Kyriakides, T.R. Extracellular matrix-derived biomaterials in engineering cell function. Biotechnol. Adv. 2020, 42, 107421–107421. [Google Scholar] [CrossRef]
- Yajima, A. Recent advances in the chemistry and chemical biology of quorum-sensing pheromones and microbial hormones. Studies in Natural Products Chemistry 2016, 47, 331–355. [Google Scholar]
- Zhou, L.; Zhang, Y.; Ge, Y.; Zhu, X.; Pan, J. Regulatory Mechanisms and Promising Applications of Quorum Sensing-Inhibiting Agents in Control of Bacterial Biofilm Formation. Front. Microbiol. 2020, 11, 589640. [Google Scholar] [CrossRef] [PubMed]
| Name | Produced by | Triggered enzyme | Reference |
| Acylated homoserine lactones (AHLs) | Gram-negative bacteria | AHL synthases | (Marketon et al., 2002) |
| Peptides | Gram-positive bacteria | ATP binding cassette transporters | (Zeng & Charkowski, 2021) |
| Furanosyl borate diester (FBD) | Both (gram-positive and gram-negative bacteria) | In addition, on an AHL synthases | (Won et al., 2020) |
| Organisms | QS molecules produced | Function of QS molecules |
| Arthrobacter agilis | Dimethyl-hexadecylamine |
|
| B. licheniformis | ComX pheromone |
|
| B. subtilis subsp. Subtilis C9 | Acetyl-buanediol |
|
| P. aeruginosa | Rhamnolipids |
|
| P. fluorescens | 2,4-Diacetyl-phloroglucinol |
|
| Pseudomonas spp. | Dimethyl disulphide |
|
| Sinorhizobium meliloti | N-(tetrahydro-2-oxo-3-furanyl)-octanamide (C8-HL) |
|
| QS Mimicry Molecule | Plants | Affected Microbes | Function |
| Rosmarinic acid | Rosmarinus officinalis, Salivia officinalis, Thymus vulgaris, Melissa officinalis, Symphytum officinale, Aegiphila mollis, Ocimum basilicum | P. aeruginosa |
|
| Eugenol | Anethum graveolens, Syzygium aromaticum | Chromobacterium violaceum, P. aeruginosa |
|
| Curcumin | Curcuma longa | P. aeruginosa |
|
| Naringenin | Citrus sp., Ficus carica, Solanum lycopersicum | P. aeruginosa, C. violaceum |
|
| Type | Marketing Name | Active Ingredients | Target Pathogen, Diseases or Weeds | Mode of Action |
| Bio fungicide | AQ10 Biofungicide | Spores of a naturally occurring Ampelomyces Quisqualis strain AQ10 |
|
|
| Trichodex | T. harzianum T39 |
|
|
|
| Rootshield WP | T. harzianum strain T-22 |
|
|
|
| Binab T |
|
|
|
|
| Primastop | Gliocladium catenulatum strain J1446 |
|
|
|
| Contans WG | Coniothyrium minitans, strain CON/M/91-08 |
|
|
|
| Biosave | Pseudomonas syringae strain ESC-11 |
|
|
|
| Bio herbicide | Biochon | Chondrostereum purpureum |
|
|
| Dr. Biosedge | Puccinia canaliculate |
|
|
|
| Solvinix | Tobacco mild green mosaic tobamovirus (TMGMV) |
|
|
|
| Sarritor | Sclerotinia minor IMI 344141 |
|
|
|
| Organo-sol |
|
|
|
| Signal | Prokaryotic receptor | Prokaryotic function | Eukaryotic receptor | Eukaryotic function | |
| Prokaryotic | Unknown | Peptidoglycan modifications | Unknown | Unknown | |
| Al-3 | QseC | Type III secretion system (T3SS) activation, motility, toxin expression, iron uptake and virulence. | Unknown | Unknown | |
| Acyl homoserine lactones | LuxR, TraR, LasR and others | Virulence, T3SS regulation, biofilm formation, motility, antibiotic production and others | Unknown | Immunomodulation, intracellular calcium signaling and apoptosis | |
| Eukaryotic | Adrenaline and nor-adrenaline | QseC | T3SS activation, motility, toxin expression, iron uptake, virulence, growth and QS | Adrenergic receptors | Cyclic AMP levels, phospholipase C activation, stress, cell proliferation, enzyme production and ion channels |
| Peptide (epidermal growth factor (EGF)) | Unknown | Unknown | EFC receptor | Cell proliferation, growth and development | |
| Dynorphin | Unknown | QS and virulence | Υ dynorphin opiate receptor | Stress responses | |
| Steroid hormones | Unknown | Unknown | Nuclear receptors | Reproduction and regulated metabolism |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




