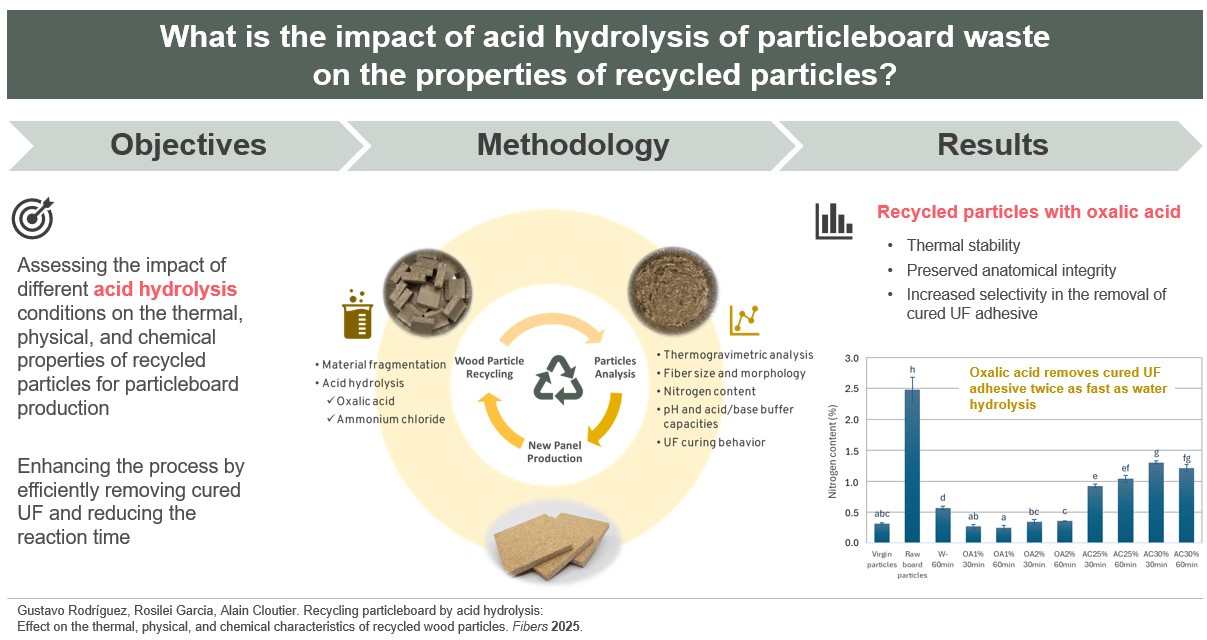
3.1. Thermal Stability
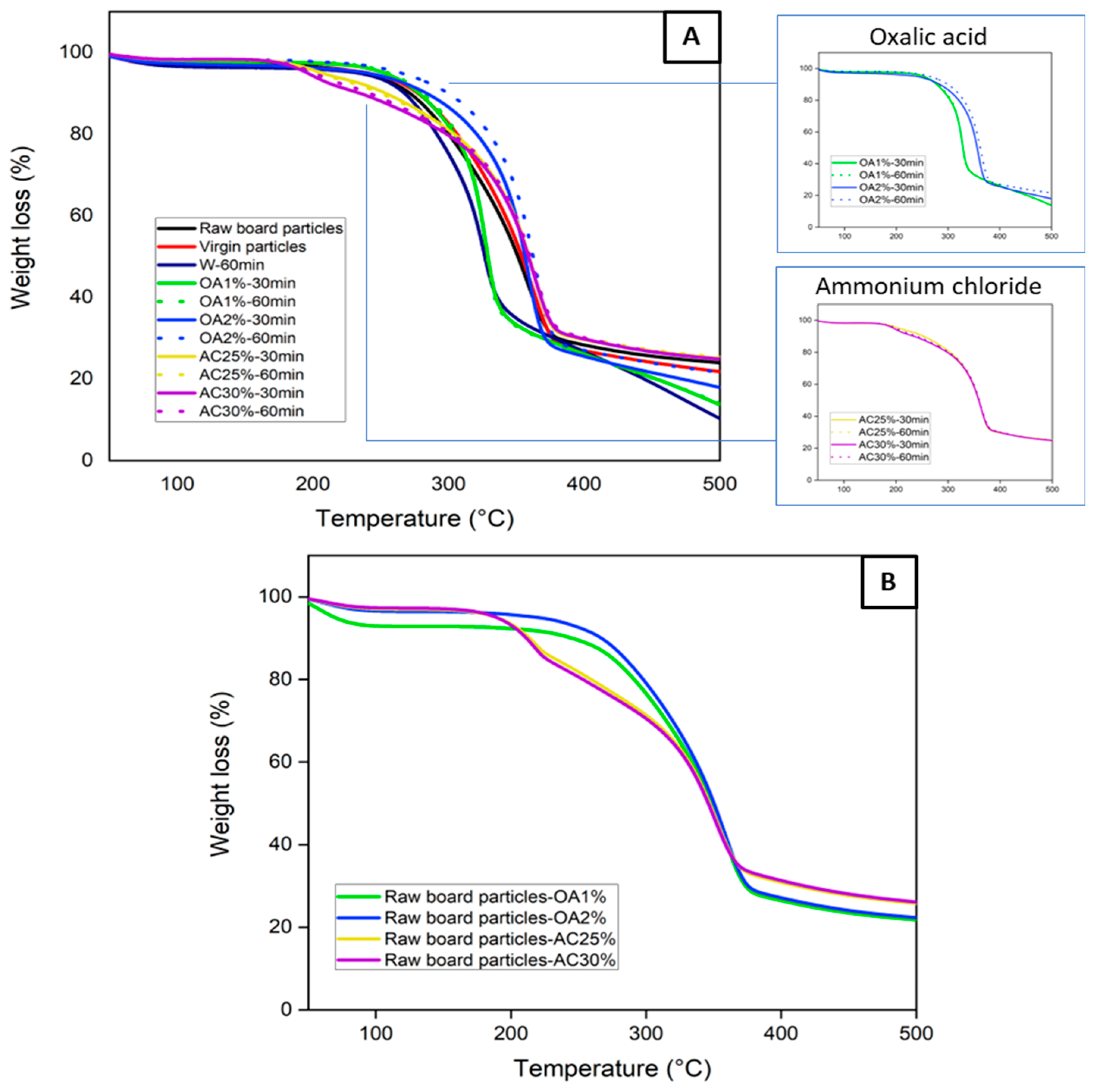
Figure 2A shows the thermal stability of the virgin particles, the raw board particles, and the recycled particles under different hydrolysis conditions. Thermogravimetric curves provide information on the particle degradation stage as a function of temperatures. Virgin, raw board particles, and recycled particles treated with water and oxalic acid exhibit a single well-defined degradation stage. Degradation begins at approximately 120 °C in all cases, with a maximum degradation temperature of about 330 °C for W-60min, OA1%-60min, and OA1%-30min. For virgin particles, raw board particles, OA2%-60min, and OA2%-30min, the maximum degradation temperature was close to 365 °C. At these temperatures, the degradation of cellulose present in the sample structure is likely to have occurred [
15]. In wood fibers, hemicelluloses typically begin to degrade at temperatures ranging from 180 to 350 °C [
16]. Cellulose, on the other hand, degrades between 200 and 450 °C [
17]. The pyrolysis process notably slowed between 400 and 500 °C, resulting in minimal mass loss. Within this temperature range, lignin degradation occurs [
18].
During the panel manufacturing process, it is crucial to carefully select the pressing conditions to prevent excessive degradation of the wood’s chemical components, which could negatively impact the panel's mechanical properties. On the other hand, the recycled particles obtained using ammonium chloride exhibited two degradation stages. The first stage of degradation began below 200 °C, with a weight loss primarily attributed to the decomposition of ammonium chloride [
19]. The most significant weight loss occurred between 300 and 380 °C, which is associated with the thermal decomposition of cellulose present in the particles. This degradation pattern can also be observed in
Figure 2B, where raw board particle samples were tested in the presence of the different acid solutions. The samples containing ammonium chloride solution (yellow and purple lines) exhibited two degradation stages, which align with the TGA curves of the recycled particles (
Figure 2A). This indicates that in AC25%-30min, AC25%-60min, AC30%-30min, and AC30%-60min, ammonium chloride residues remain within the particles, despite multiple washings after the acid hydrolysis treatment. Also, the TGA curves of the raw board particle samples in the presence of oxalic acid (
Figure 2B) exhibit a similar degradation behavior to that of the particles recycled with this acid (
Figure 2A). In general, all recycled particles exhibit good thermal stability, similar to that observed in the TGA curve of virgin particles used for particleboard manufacturing. The highest degradation rates occur at temperatures above 200 °C. As these temperatures are generally higher than those used in the panel manufacturing process, particles recycled from all hydrolysis conditions can be used in the manufacture of new panels.
3.2. Granulometric and Morphological Characterization
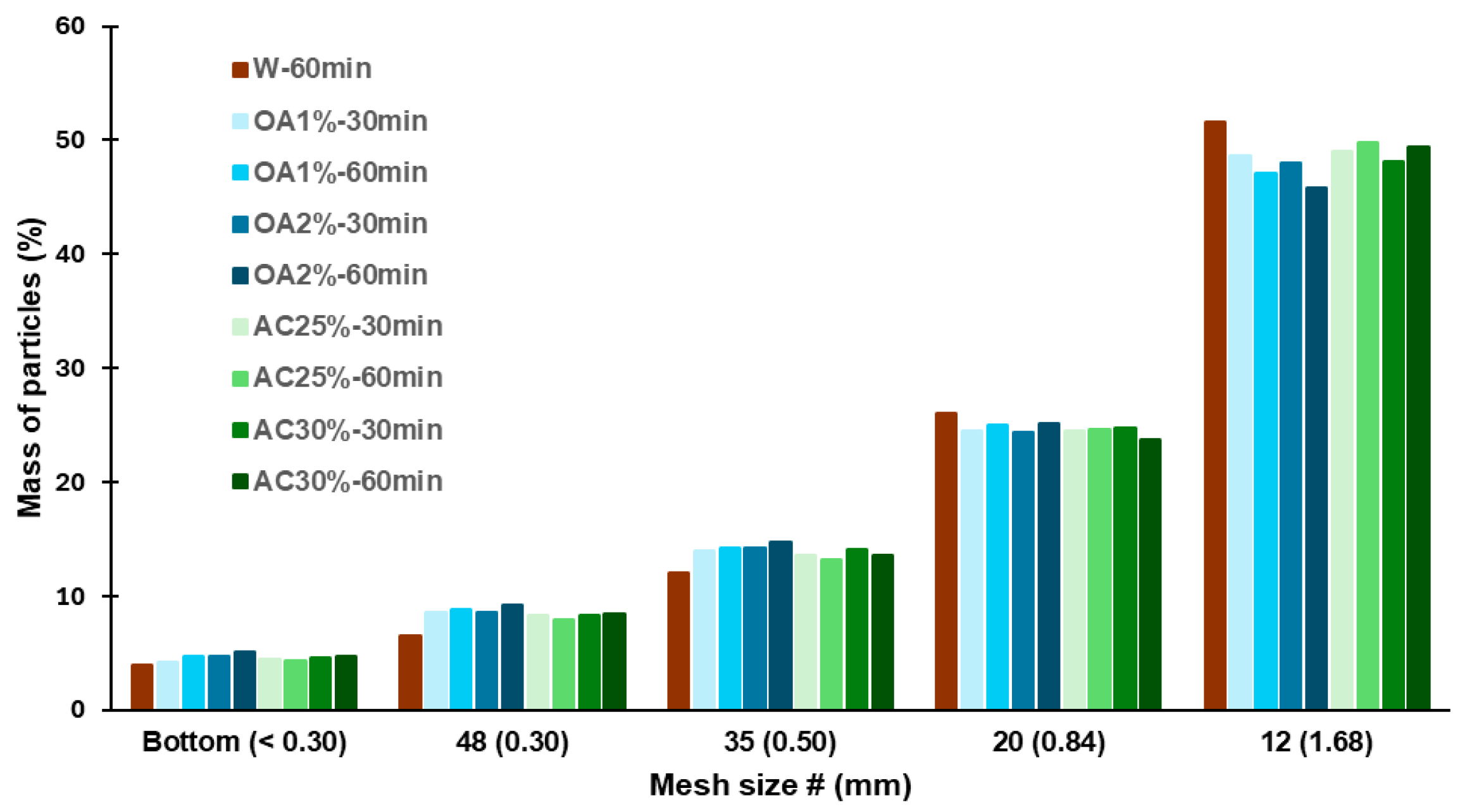
The size distribution of the recycled particles is shown in
Figure 3. For all hydrolysis conditions, the highest percentage of particles was retained by the 12-mesh sieve, corresponding to particles ≥1.68 mm. The retention percentage ranged from 46% to 52%, with AC25%-60min and AC30%-60min exhibiting the highest proportion among the acid hydrolysis-treated recycled particles (49.8% and 49.4% respectively). The values observed for particles recycled using only water were higher, at 51.6%. For the 20-mesh sieve, the retention percentage ranged from 24% to 26% across all cases, corresponding to particle sizes between 0.84 mm and 1.68 mm. Between 12% and 15% of the particles were retained by the 35-mesh sieve, with all acid treatments exhibiting a slightly higher percentage of particles in this category than W-60min. A similar trend was observed for the 48-mesh sieve, where the retention percentage ranged from 6% to 9% in all acid treatments.
Particles smaller than or equal to 0.30 mm were collected at the bottom of the sieve, and their content never exceeded 5% in any case. All the treatments evaluated showed a low percentage of fine particles, which is ideal if the aim is to reuse these particles in the production of new panels. Fine particles have a high specific surface area. Therefore, an increased proportion of fines would necessitate the use of more adhesive during manufacturing to attain the desired properties of the panels. This is not optimal from a cost perspective. Additionally, the results showed that using acid solutions in the hydrolysis process does not significantly alter the dimensions of the recycled particles compared to those obtained using water alone. The SEM images of the recycled particles from acid hydrolysis treatments are shown in
Figure 4.
The anatomical structure of the particles obtained through acid hydrolysis remains unaffected by the acidic solutions used; the fibers stay well-defined across all treatments. The images indicate that recycled particles are composed of a mixture of softwood and hardwood species. The acid hydrolysis treatment was highly selective toward the adhesive, effectively preserving the integrity of the prosenchymatous elements. This is evident in the perforation plates of hardwoods, which can be observed in
Figure 4A; bordered pits in the softwood tracheides shown in
Figure 4C–E; as well as in cell walls. Another noteworthy observation is the absence of delamination in the longitudinal tracheids in softwoods and fibers in hardwoods. This serves as a strong indicator that the acid was not sufficiently strong to dissolve the middle lamella. This conclusion is supported by the fact that no signs of separation were observed in the cell bundles. Likewise, a clearly defined vessel composed of various vessel elements can also be distinguished in
Figure 4F, as well as the presence of bordered pits in
Figure 4G. In
Figure 4H, a tangential section of a softwood particle reveals well-defined uniseriate rays. The presence of these structures in the samples indicates a high-quality anatomical integrity of the recycled particles, suggesting their potential as a viable alternative for producing new particleboards. Furthermore, no adhesive residues or foreign elements were detected in the recycled particles. The TGA analysis revealed the presence of ammonium chloride in the recovered particles (AC25%-30min; AC25%-60min; AC30%-30min; and AC30%-60min). However, no visible crystals or precipitates were observed in the SEM images (
Figure 4E-H). This suggests that the ammonium chloride may be retained within the cell lumens and void spaces of the particles.
3.3. Nitrogen Content
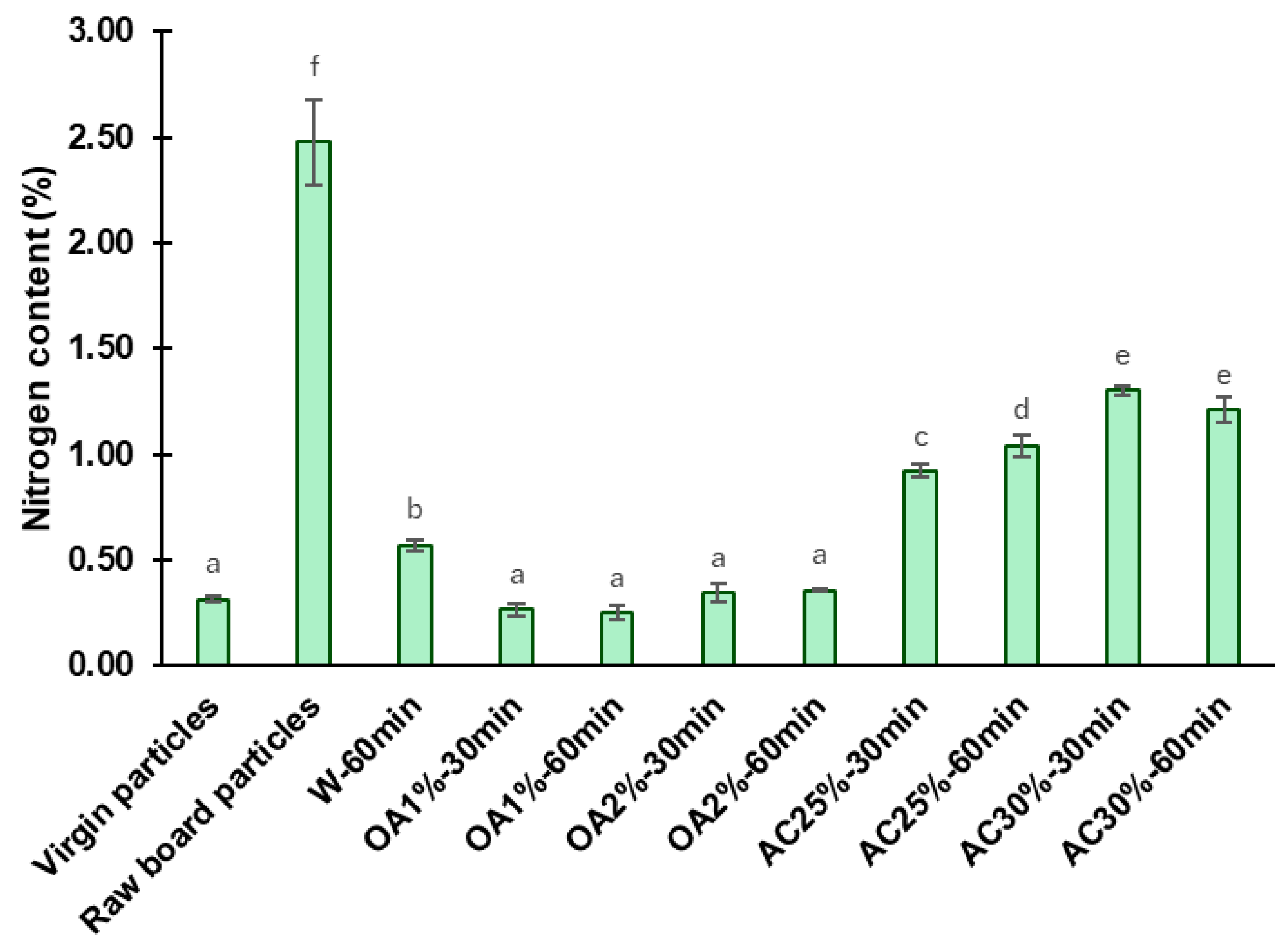
The nitrogen content found in the virgin particles, the raw board particles, and the recycled particles is shown in
Figure 5. The nitrogen content of raw board particles was determined as a control sample, yielding a value of 2.48%. This amount contrasts with and exceeds that obtained in virgin particles, which was 0.31%. Hagel and Saake [
20] reported similar nitrogen contents (0.3%) in neet wood particles from poplar and spruce species.
The high nitrogen content in the raw board particles compared to virgin particles is due to the presence of cured UF adhesive and the ammonium chloride catalyst, both used in particleboard manufacturing [
9]. Consequently, a decrease in nitrogen content following hydrolysis is associated with the removal of the cured adhesive from the recycled particles. After the acidic hydrolysis, the nitrogen content values of recycled particles were significantly lower than those of the raw board particles. Hydrolysis treatments using oxalic acid proved to be more effective than those using ammonium chloride. For oxalic acid treatment, the nitrogen content of recycled particles was reduced by 85% to 90%, depending on concentration and reaction time. The OA1%-60min treatment exhibited the lowest nitrogen content in absolute terms (0.25%), representing a 90% reduction in nitrogen content compared to raw board particles. Among the ammonium chloride treatments, AC25%-30min yielded the best results in terms of removing cured UF, with a nitrogen content of 0.92%, representing a 63% reduction compared to raw board fibers. As previously described, ammonium chloride particles remained in the recycled particles, which may have contributed to slightly higher nitrogen content values compared to the oxalic acid treatments. The W-60min treatment exhibited a nitrogen content of 0.57%, representing a 77% reduction compared to the raw board particles. The W-60min remains a better treatment than ammonium chloride under the conditions tested in our study. Also, the W-60min performed better than the treatment reported by Fu et al. [
9] under comparable conditions, specifically at a temperature of 120 °C and a reaction time of 60 min. In their study, the nitrogen content in the recycled particles was found to be 0.95% (a 58.7% reduction compared to the control samples). The factorial ANOVA reveals a strong interaction between reaction time, acid type, and its concentration, which significantly affects the nitrogen content (
Table 2). The effect of time depends on the type of acid and its concentration. When using a lower concentration, the nitrogen content in the ammonium chloride treatments increased with longer reaction times, whereas at higher concentrations, it remained constant. In contrast, when using oxalic acid, neither time nor concentration has an impact on the nitrogen content. In addition, statistical analysis showed that particles recycled with oxalic acid had the same nitrogen content as virgin particles and a lower nitrogen content than those recycled at W-60min, indicating a higher efficiency of oxalic acid hydrolysis (
Figure 5).
3.4. pH and Acid/Base Buffer Capacity
Table 3 presents the pH levels of virgin particles, raw board particles, and recycled particles. The pH of the virgin particles was measured at 5.25, while the raw board particles had a pH of 5.37. The pH levels of all particles obtained through acid hydrolysis were lower than these values. Using oxalic acid, the highest recorded pH was in OA1%-60min, with a value of 4.14, which represents a 23% reduction compared to the raw board particles. In contrast, OA2%-30min showed the most significant reduction in pH, approximately 39%. On the other hand, among the recycled particles treated with ammonium chloride, AC30%-60min showed the highest pH reduction, with a 44% decrease, resulting in a pH of 3.00. The pH levels of particles treated with ammonium chloride were lower than those obtained with oxalic acid. This could be attributed to the presence of small quantities of residual ammonium chloride in the recycled material.
The pH level of particles is a crucial property for the manufacture of new particleboard, as it can influence gel time, which in turn impacts pressing time and the final properties of the panels. Although the polymerization of UF adhesive used in particleboard production is most effective under acidic conditions, excessively low pH levels can adversely affect the fabrication of new panels due to the rapid polymerization of the adhesive. Unlike the acid treatments, the pH of the W-60min recycled particles was higher (5.89) than that of the virgin particles and the raw board particles. Using similar conditions, Fu et al. [
9] obtained a pH of 6.57 in recycled particles using water alone. The statistical analysis revealed that reaction time has a differential effect on pH, depending on the type of acid solution and its concentration (
Table 2). When using oxalic acid, the pH increases over time, whereas with ammonium chloride, it tends to decrease (
Table 3). In this case, notable differences in the slope of the pH level obtained with oxalic acid and ammonium chloride under both concentration conditions suggest an interaction between treatment and time. On the other hand, there are significant differences in pH levels obtained for acid hydrolysis treatments compared with W-60min, the virgin particles, and the raw board particles.
The buffer capacity of a solution is a measure of its ability to neutralize strong acids (acid buffer capacity) or strong bases (base buffer capacity) while maintaining a stable pH [
21]. It indicates the amount of a strong acid (such as H₂SO₄) or a strong base (such as NaOH) that can be added to the solution before a significant increase or decrease in pH occurs.
Table 4 presents the acid/base buffer capacity of the different recycled particles. Particles obtained through acid hydrolysis show a lower acid buffer capacity than virgin and raw board particles. This indicates that these particles will be less effective in resisting pH changes in the presence of strong acids, leading to a faster pH decrease compared to virgin and raw board particles. This effect is more pronounced in treatments with ammonium chloride, reaching its maximum in the case of AC30%-60min. The W-60min treatment exhibits the same acid buffer capacity as virgin particles and is highly similar to that of raw board particles. Since all the acid buffer capacity values in the AC30%-60min treatment were 0, the model assumptions were not met, and an ANOVA could not be performed on the factorial design to evaluate the influence of the factors. When comparing the acid hydrolysis treatments, significant differences in acid buffer capacity were found among the virgin particles, the raw board particles, and the W-60min treatment. According to the Tukey test, the treatments with ammonium chloride are statistically similar. Differences are observed between the oxalic acid treatments at 1% and 2%, but no significant differences exist between treatments with the same concentration. Finally, virgin and raw board particles do not differ significantly from each other. The acid buffer capacity is an important factor to consider when reusing recycled particles to produce new panels. Wood particles with a high acid buffer capacity can significantly affect the curing process of the UF adhesive commonly used in particleboard manufacturing [
22].
On the other hand, the base buffer capacity exhibits a different behavior. The base buffer capacities of virgin particles and raw board particles are similar. However, the base buffer capacity of the particles recycled by acid hydrolysis increased, with a more pronounced effect with ammonium chloride regardless of acid concentration or reaction time. In particular, the AC30%-60min treatment exhibited an almost eightfold increase compared to raw board particles. The base buffer capacity of the particles recycled with oxalic acid also increased, but to a lesser extent. This increased capacity makes these particles more stable against disturbances, such as the addition of strong bases. Conversely, in particles obtained through hydrolysis with water (W-60min), the base buffer capacity significantly decreased compared to virgin and raw board particles. The factorial ANOVA results indicate that the effect of reaction time on the base buffer capacity depends on the type of acid solution used (
Table 2). When using ammonium chloride, the base buffer capacity decreases at lower acid concentration and increases at a higher concentration. In contrast, with oxalic acid, the base buffer capacity remains constant as both reaction time and acid concentration increase.
3.5. Influence of the Recycled Particles on the UF Curing Behavior and Gel Time
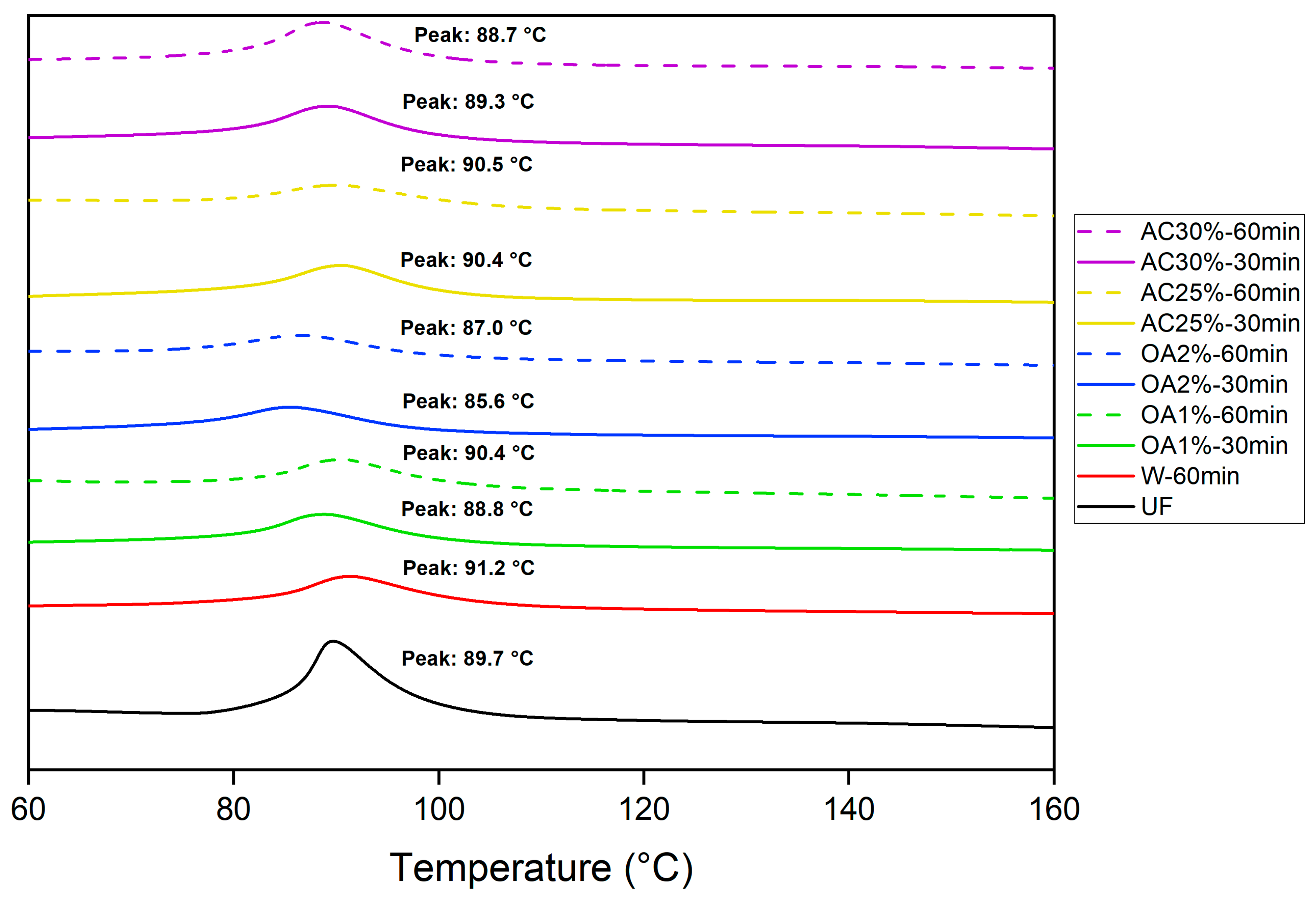
The polymerization reactions of UF adhesives are typically exothermic. The adhesive cures through a polycondensation process, during which water and methanol are released through an exothermic reaction, as characterized by DSC analysis.
Figure 6 shows the peak temperature for pure UF adhesive and mixed with each type of recycled particles obtained by hydrolysis with water alone and in acid solutions. This peak temperature corresponds to the point at which the reaction reaches its maximum intensity [
23] and, in our case, represents the temperature at which the UF adhesive polymerizes in the presence of recycled particles. The peak temperature of the pure UF adhesive obtained in our study was 89.7 °C. Particles treated with oxalic acid showed a slight decrease in peak temperature during the curing reaction, except for OA1%-60min. A more pronounced effect was observed in treatments using a 2% oxalic acid concentration compared to pure UF adhesive. However, in the OA1%-60min treatment, the peak temperature was slightly higher than that of the pure UF adhesive. Conversely, the presence of particles treated with 25% ammonium chloride resulted in a higher peak temperature compared to pure UF adhesive. In the cases of AC30%-30min and AC30%-60min, the peak temperatures were marginally lower than that of pure UF adhesive.
Therefore, no clear pattern emerges regarding the influence of recycled particles on the peak temperature. However, their presence subtly affects the peak temperature of the UF curing reaction. If the recycled particles from our study are used to produce new panels, the UF adhesive curing reaction would be suitable, as indicated by the DSC results.
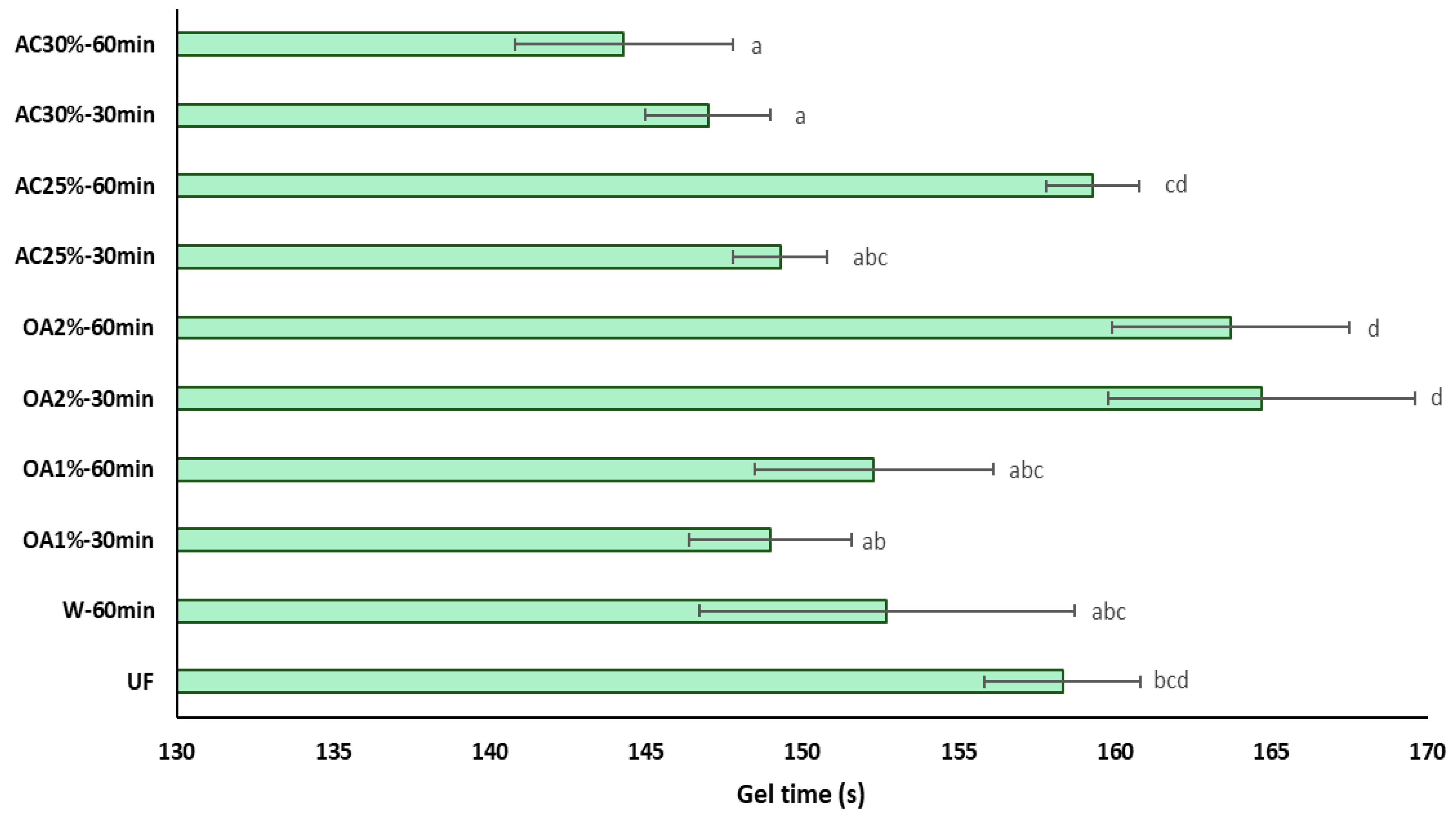
The influence of recycled particles on the gel time of UF adhesive is shown in
Figure 7. The gel time of UF adhesive in the presence of recycled particles treated with oxalic acid does not exhibit significant differences compared to pure UF adhesive. This is a positive indicator for reusing these particles in producing new panels, as it would not require additional pressing time or a higher amount of catalyst to accelerate the polymerization reaction of the adhesive. Additionally, the OA1% treatments differ from OA2% treatments regardless of reaction time. On the other hand, the influence of particles obtained with 30% ammonium chloride is primarily reflected in the reduction of the gel time of the UF adhesive. In treatments AC25%-30min and AC25%-60min, the gel time is equal to that of pure UF adhesive. These results may be attributed to the lower acidity of the recycled particles compared to the virgin particles. This result aligns with the findings of Xing et al. [
24], who suggested that UF adhesive mixed with lower pH particles requires less curing time. Additionally, it has been reported that an increase in the acid buffer capacity of the wood particles is associated with a longer gel time [
25]. In this case, the acid buffer capacity of the recycled particles is low. Therefore, it can be inferred that the gel time of the UF adhesive in the presence of these particles would be shorter than that of the UF adhesive alone.
The ANOVA revealed a significant interaction between the type of acid used and its concentration (
Table 2). As oxalic acid concentration increases, the gel time also increases. In contrast, when the ammonium chloride concentration increases, the gel time decreases. Furthermore, there is a significant interaction between the concentration used and the hydrolysis reaction time. At lower concentrations, the gel time is longer, reaching 60 minutes. However, at higher concentrations, the gel time remains constant regardless of the hydrolysis reaction time. When comparing the acid hydrolysis treatments with W-60min and pure UF adhesive, significant differences were observed between some treatments.