Submitted:
11 April 2025
Posted:
14 April 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Methodology: A Thematic Narrative Review
- energy availability and substrate partitioning,
- adaptive stress responses across biological levels,
- chronic stress or allostatic load,
- aging mechanisms and chronic disease progression.
- Open coding identified recurrent concepts such as metabolic trade-offs, substrate reallocation, and hormesis.
- Axial coding mapped relationships among energy dynamics, stress adaptation, and physiological outcomes.
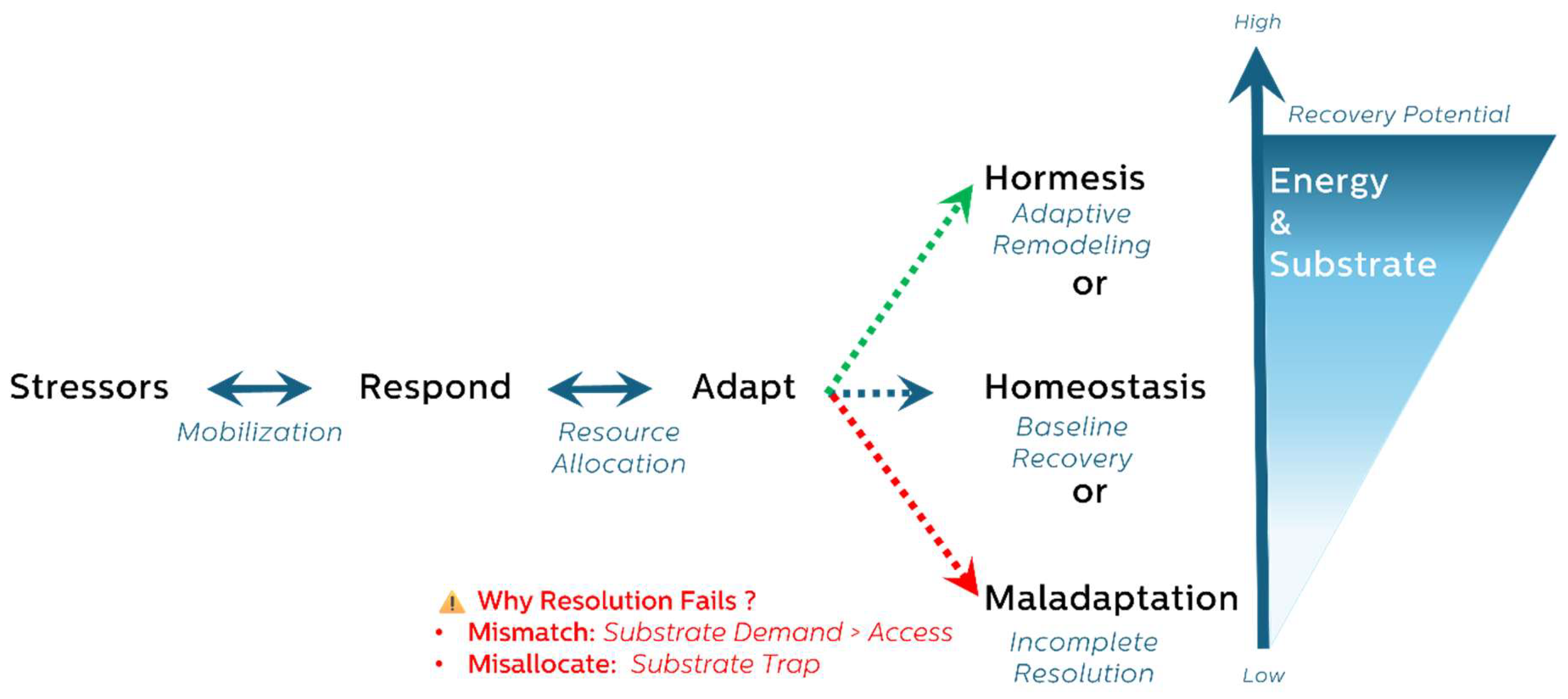
- Narrative integration constructed a coherent, system-level framework that reflects a consistent adaptive trajectory: Respond → Adapt → Recover, culminating in either homeostasis, hormesis, or maladaptation.
3. The Energetic Trajectory of Stress Adaptation: From Response to Resolution
- Restored homeostasis
- Adaptive overcompensation (hormesis)
- Maladaptation and decline (exhaustion)
- Phase 1: Respond – Emergency activation and energy mobilization.
- Phase 2: Adapt – Metabolic prioritization and reprogramming.
- Phase 3: Resolve – Transition toward recovery or dysfunction.
4. The Energetic Architecture of Adaptation: Substrate Reallocation in Systemic Stress Responses
- Neuroendocrine System activates the hypothalamic-pituitary-adrenal (HPA) axis and sympathetic-adrenal-medullary (SAM) system, rapidly mobilizing glucose and suppressing growth and reproduction (Tsigos & Chrousos, 2002).
- Immune System initiates acute inflammation via pattern recognition receptors (PRRs), releasing pro-inflammatory cytokines (e.g., TNF-α, IL-6) and shifting immune metabolism toward glycolysis (Alack et al., 2019; Straub, 2017).
- Muscle tissue supplies gluconeogenic substrates by breaking down amino acids via proteolytic pathways (Cahill, 2006; Wolfe, 2006).
- Cellular ISR halts general protein synthesis via eIF2α phosphorylation while promoting selective translation of stress-resilient genes (Pakos-Zebrucka et al., 2016).
- Mitochondria shift metabolic output and activate antioxidant signaling and the mitochondrial unfolded protein response (UPRmt), increase ATP production, and activate antioxidant signaling (Picard & Shirihai, 2022).
- Neuroendocrine System– Cortisol orchestrates systemic prioritization, supporting cerebral glucose supply while suppressing insulin, growth, and reproduction (McEwen & Wingfield, 2003).
- Immune cells undergo metabolic polarization: pro-inflammatory cells rely on glycolysis, while regulatory or reparative cells depend on oxidative phosphorylation (Olenchock et al., 2017; Willmann & Moita, 2024) (Geric et al., 2019; Olenchock et al., 2017). Chronic stress can trap cells in inflammatory states.
- Skeletal muscle, a major metabolic sink, attempts to transition from catabolism to repair. This shift requires amino acid availability and immune–muscle coordination, both of which are impaired under conditions of anabolic resistance. Anabolic resistance is not only a consequence but also a signal of unresolved adaptation, a state in which substrates and signaling are insufficient to restore muscle regeneration (Paulussen et al., 2021).
- Cellular ISR, when energetically supported, transitions from acute translation suppression to remodeling via autophagy, stress granule formation, and selective translation of repair-promoting factors (Gambardella et al., 2020). This metabolic reprioritization also drives epigenetic remodeling that accelerates cellular aging, especially under persistent stress (Gambardella et al., 2020).
- Mitochondria undergo remodeling, including mitophagy, fission/fusion dynamics, and shifts in substrate utilization to meet tissue-specific energy demands (Lockhart et al., 2020).
| System | Stress Priority Function | Energy Source Preference | Resolution Potential | Vulnerabilities Under Deficit |
|---|---|---|---|---|
| Neuroendocrine | Glucose mobilization, survival triage | Gluconeogenesis, lipolysis | Moderate (via cortisol tapering) | HPA overactivation, insulin resistance |
| Immune | Inflammation, defense | Glycolysis (pro-inflammatory), OXPHOS (repair) | High if balance restored | Chronic inflammation, immune senescence |
| Muscle | Amino acid reservoir, repair coordination | Glycogen, fatty acids, structural protein | High if nutrients available | Anabolic resistance, sarcopenia |
| Cellular ISR | Proteostasis, autophagy | Internal recycling, selective translation | Moderate tohigh | Persistent translation block, apoptosis |
| Mitochondria | Energy production, redox signaling | OXPHOS, glycolysis, fatty acids | High if fission/fusion restored | ROS overload, mitokine dysfunction |
- Neuroendocrine systems reduce HPA axis activity and re-establish circadian rhythm and metabolic homeostasis. Flattened or delayed cortisol recovery signals impaired resolution.
- Immune systems transition from inflammation to repair, with M1 macrophages converting to M2 phenotypes and resolution pathways (e.g., resolvins, lipoxins) facilitating tissue remodeling (Olenchock et al., 2017). Micronutrient sufficiency—particularly zinc, selenium, and iron—is critical to this process.
- Skeletal muscle resumes protein synthesis and regeneration, but only if inflammation resolves and energy/nutrient levels support mTORC1 and satellite cell activation. Without adequate support, fibrosis or sarcopenia may ensue (Paulussen et al., 2021).
- Cellular ISR mechanisms, such as GADD34-mediated dephosphorylation of eIF2α, permit selective restoration of protein synthesis. This reactivation depends on sufficient ATP, proteostasis, and redox control (Gambardella et al., 2020).
- Mitochondria stabilize through restored fission/fusion dynamics and mitophagy, allowing redox homeostasis and efficient energy production. Transient mitokine signaling subsides as systemic demands normalize (Picard & Shirihai, 2022).
- Homeostasis – Return to pre-stress function and balance.
- Hormesis – Functional overcompensation and increased resilience.
- Maladaptation – Incomplete resolution, leading to persistent dysfunction.
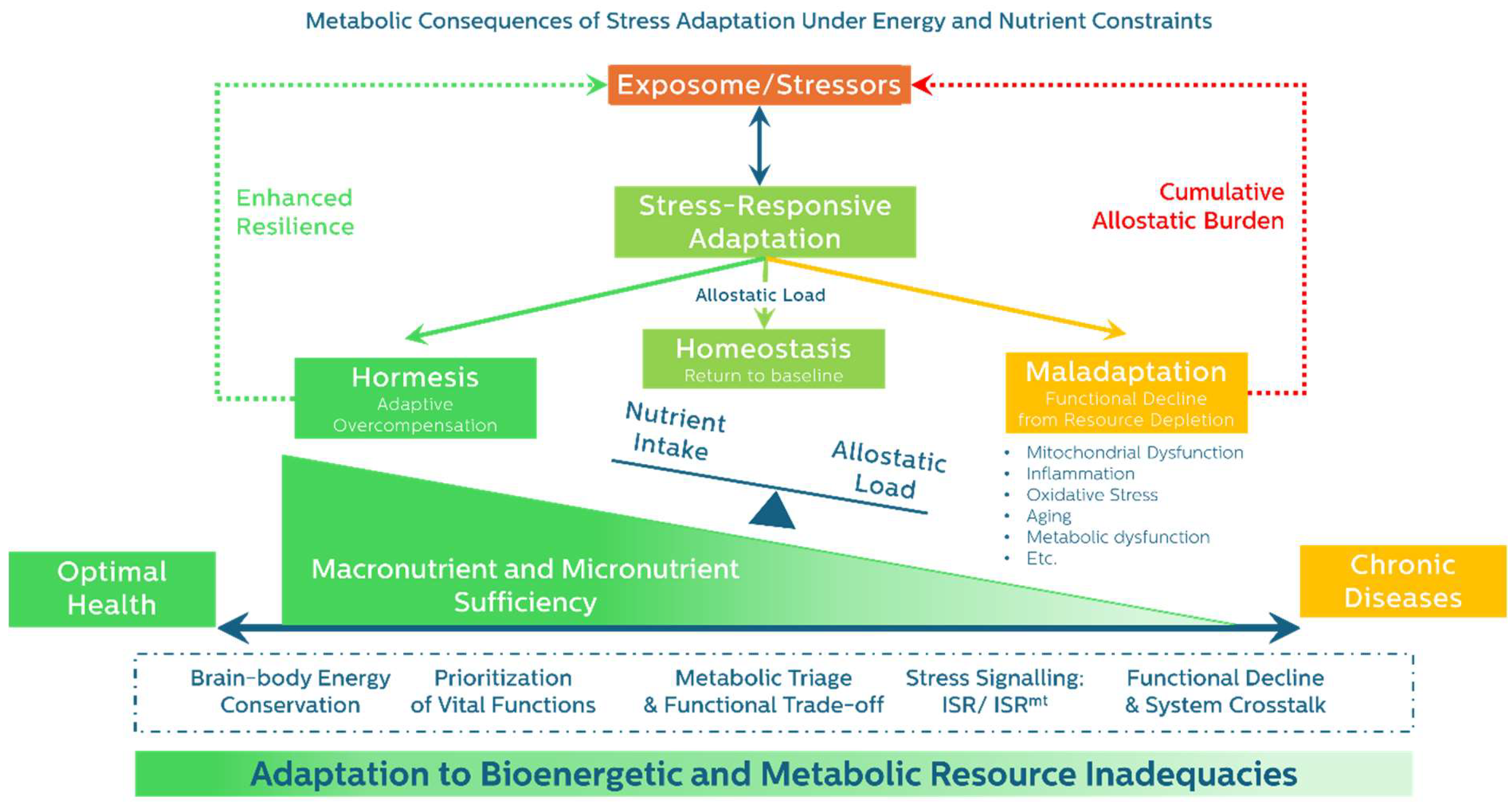
5. Resolution and Its Consequences: Divergent Outcomes Shaped by Energy and Resource Allocation.
5.1. Homeostasis — Energetic Recovery and Structural Recalibration
- Cortisol and inflammatory cytokines decline, parasympathetic tone is restored, and insulin sensitivity improves (Bobba-Alves et al., 2022).
- Immune resolution involves clearance of apoptotic cells, matrix remodeling, and macrophage transition from M1 to M2 phenotypes—processes that rely on mitochondrial OxPhos, redox regulation, and micronutrients like zinc, iron, and selenium (Alack et al., 2019; Laurent et al., 2017; Olenchock et al., 2017).
- In skeletal muscle, recovery depends on satellite cell activation and nutrient-sensitive pathways such as mTORC1, supported by leucine, vitamin D, and redox cofactors (Beaudart et al., 2017; Careccia et al., 2023; Paulussen et al., 2021).
- Cellular ISR resolves through GADD34-mediated dephosphorylation of eIF2α, enabling proteostasis and selective translation restoration (Gambardella et al., 2020; Novoa et al., 2001).
- Mitochondrial recovery via mitophagy and biogenesis restores ATP production and oxidative balance; transient ROS bursts activate adaptive pathways via NRF2 and FOXO, while sustained oxidative stress impairs recovery (Picard & Shirihai, 2022).
5.2. Hormesis — Energetic Overcompensation and Adaptive Remodeling
- Trained immunity: Monocytes, macrophages, and NK cells undergo glycolytic and epigenetic reprogramming via mTOR–HIF-1α signaling, increasing responsiveness and tolerance (Netea et al., 2016; Ochando et al., 2023; Vuscan et al., 2024).
- Immune resolution and tolerance: Regulatory T cells and M2 macrophages mediate inflammation resolution and tissue repair via mitochondrial metabolism (Vuscan et al., 2024).
- Exercise-induced muscle remodeling: IL-13–producing ILC2s, IL-33–expressing stromal cells, and macrophage–Treg signaling coordinate mitochondrial biogenesis and type 2 immunity in recovery (Langston & Mathis, 2024; Metallo & Vander Heiden, 2013).
- Mild ISR activation: Transient eIF2α phosphorylation enhances redox balance, proteostasis, and metabolic flexibility via ATF4/CHOP signaling (Costa-Mattioli & Walter, 2020; Sparkenbaugh et al., 2011).
- Mitohormesis: Low-level ROS from mitochondrial stress induces biogenesis, antioxidant upregulation, and mitokine release (e.g., FGF21, MOTS-c) for systemic coordination (Lockhart et al., 2020; Ristow & Schmeisser, 2014).
5.3. Maladaptation — Energetic Collapse and Structural Degeneration
- Neuroendocrine: Sustained cortisol, insulin resistance, hippocampal atrophy, and central fatigue due to prolonged stress signaling (Chrousos, 2009; Meeusen et al., 2006; Shaulson et al., 2024).
- Immune: Inflammaging and immunosenescence from persistent IL-6, TNF-α, SASP signaling, and impaired clearance of senescent cells(Franceschi et al., 2018; Fulop et al., 2018; Wang et al., 2024).
- Skeletal muscle: Anabolic resistance, mitochondrial dysfunction, and catabolism lead to sarcopenia and frailty, compounded by aging, nutrient deficits, and inflammation(Cruz-Jentoft et al., 2023; Walrand et al., 2021).
- Cellular ISR: Chronic eIF2α phosphorylation impairs translation, promotes apoptosis, and drives redox imbalance and mitochondrial damage (Hetz & Papa, 2018; Wek, 2018).
- Mitochondria: PGAM5-driven mitochondrial fragmentation, ROS generation, and mtDNA-triggered inflammasome activation fuel a cycle of mitophagy failure, pyroptosis, and degeneration (Qi et al., 2025; Youle & van der Bliek, 2012; Yuk et al., 2020)
| Feature |
 Homeostasis Homeostasis |
 Hormesis Hormesis |
 Maladaptation Maladaptation |
|---|---|---|---|
| Energy Availability | Restored baseline levels | Sufficient with transient surplus | Depleted or misallocated |
| Functional Outcome | Re-equilibration | Enhanced resilience or capacity | Persistent dysfunction |
| Immune Response | Inflammation resolves | Trained immunity and regulatory tolerance | Chronic inflammation, immune exhaustion |
| Muscle Remodeling | Repair of damaged fibers | Functional hypertrophy, mitochondrial gains | Catabolism, fibrosis, sarcopenia |
| ISR Recovery | Reinstated proteostasis | Increased stress resilience, adaptive memory | Persistent translation block, apoptosis |
| Mitochondrial Dynamics | Normalized bioenergetics | Improved redox balance, adaptive signaling | ROS overload, mitophagy failure |
| Recovery Dependency | Energy repletion, stress withdrawal | Surplus energy, time, micronutrient support | Insufficient recovery, chronic demand |
6. Energy and Substrate Insufficiency in High-Demand States
- Disease-Related Malnutrition (DRM) occurs in patients with acute or chronic disease where inflammation increases resting energy expenditure and protein breakdown, often despite ongoing feeding (Cederholm & Bosaeus, 2024; Muscaritoli et al., 2023).
- Chronic Energy Deficiency (CED) reflects persistent nutritional insufficiency during states of heightened need—such as pregnancy—where energy and nutrient reallocation can compromise maternal and fetal health even when BMI appears normal (Prisabela et al., 2023; Taylor-Baer & Herman, 2018).
- Relative Energy Deficiency in Sport (REDs) results from chronic low energy availability in athletes, leading to impaired endocrine, immune, bone, and cognitive function, often with normal or low-normal body weight (Cabre et al., 2022; Mountjoy et al., 2018).
| Feature | DRM | CED | REDs |
|---|---|---|---|
| Primary Affected Populations | Hospitalized or chronically ill patients | Pregnant women, children, low-resource settings | Endurance athletes, dancers, military recruits |
| Onset | Acute or chronic illness | Gradual under chronic stress (e.g., pregnancy) | Subacute with high training load |
| Triggers | Inflammation, disease burden | Increased physiological need, low intake | Prolonged mismatch between training intensity and caloric intake |
| Nutritional Markers | Often abnormal (e.g., prealbumin ↓) | Subclinical changes; may appear normal in standard labs | May have normal BMI, altered hormones |
| Clinical Presentation | Weight loss, immune dysfunction, poor healing | Maternal fatigue, micronutrient depletion, fetal risk | Performance decline, bone loss, endocrine suppression |
| Response to Nutrition | Requires nutritional support alongside anti-inflammatory therapy | Improves with energy/nutrient repletion | Requires coordinated refeeding and training balance |
- No overt intake deficit – energy intake may appear normal, and BMI may be stable or elevated.
- Triggered by cumulative exposome burden – including inflammation, toxin exposure, psychosocial stress, circadian disruption, or chronic low-grade infections (Pizzorno, 2020; Vermeulen et al., 2020).
- Manifests through the metabolic trade-offs pattern – such as impaired muscle recovery, fatigue, immunosuppression, or anabolic resistance, often before clinical thresholds of dysfunction are met.
7. Metabolic Substrate Misallocation, Aging, and the Modern Disease Burden
8. Recognizing ERM as the Cost of Adaptation in Clinical Practice
- Chronic fatigue despite adequate sleep and nutrition
- Poor exercise recovery or delayed wound healing
- Frequent mild infections or persistent low-grade inflammation
- Difficulty maintaining or gaining lean mass despite adequate intake and physical activity
- Biomarker patterns suggesting inflammation-driven nutrient diversion
- External triggers: air pollution, persistent organic pollutants (POPs), heavy metals (e.g., lead, arsenic, mercury), microplastics, endocrine-disrupting chemicals, and circadian rhythm disruption
- Internal triggers: chronic inflammation, dysbiosis, latent infections, psychosocial stress, and trauma
- Mild ERM: Subtle reductions in physical performance, cognitive clarity, or recovery from exertion; typically reversible with adequate substrate support and removal of stressors
- Moderate ERM: Emergence of functional trade-offs, including low-grade inflammation, anabolic resistance, downregulated housekeeping and transport proteins, or early hormonal imbalances
- Severe ERM: Entrenched catabolism, sarcopenia, immune dysfunction, and metabolic rigidity, often accompanied by structural decline and reduced adaptability
- Persistent elevation of CRP alongside declining prealbumin or transferrin
- Declining phase angle and lean body mass, even with preserved or rising body weight
- Elevated intracellular enzymes (e.g., ALT, AST, CPK) without clear organ-specific pathology
- Persistent hypercholesterolemia or hyperglycemia despite appropriate dietary and lifestyle interventions
- “What phase of adaptation is this patient in?”
- “What exposures, stressors, or nutritional deficits are sustaining this trade-off—and what interventions could restore metabolic balance?”
- Targeted Dietary support: Emphasize high-quality protein and healthy fats, with controlled and context-specific carbohydrate intake
- Micronutrient repletion: Address subclinical deficiencies in protein and their critical cofactors for metabolic and immune functions such as zinc, selenium, magnesium, and iron
- Exposome reduction: Minimize environmental and dietary stressors through clean air and water, toxin avoidance, and anti-inflammatory, nutrient-dense foods
- Circadian and metabolic tempo optimization: align light exposure, sleep-wake cycles, and feeding-fasting windows to support hormonal and metabolic coherence
- Lifestyle-based resilience building: Encourage regular physical activity, stress reduction techniques, restorative sleep, and social connectedness
- Functional monitoring tools: utilize technologies such as BIA and AI-powered wearables to track recovery and adaptation capacity in real time
9. Conclusion: The Metabolic Cost of Resilience
List of Abbreviations
| Abbreviation | Full Term | |
| ACE | Adverse Childhood Experiences | |
| ACTH | Adrenocorticotropic Hormone | |
| ALT | Alanine Transaminase | |
| AMPK | AMP-Activated Protein Kinase | |
| APP | Acute Phase Proteins | |
| AST | Aspartate Transaminase | |
| ATP | Adenosine Triphosphate | |
| BIA | Bioelectrical Impedance Analysis | |
| BMI | Body Mass Index | |
| CED | Chronic Energy Deficiency | |
| CPK | Creatine Phosphokinase | |
| CRP | C-Reactive Protein | |
| DHEA | Dehydroepiandrosterone | |
| DRM | Disease-Related Malnutrition | |
| eIF2α | Eukaryotic Initiation Factor 2 Alpha | |
| ERM | Exposure-Related Malnutrition | |
| FGF21 | Fibroblast Growth Factor 21 | |
| GAS | General Adaptation Syndrome | |
| GDF15 | Growth Differentiation Factor 15 | |
| HIF-1α | Hypoxia-Inducible Factor 1-Alpha | |
| HPA axis | Hypothalamic–Pituitary–Adrenal Axis | |
| IL | Interleukin (e.g., IL-6, IL-13, IL-33) | |
| ISR | Integrated Stress Response | |
| ISRmt | Mitochondrial Integrated Stress Response | |
| LDH | Lactate Dehydrogenase | |
| M1/M2 | Macrophage Polarization States (Pro-inflammatory / Anti-inflammatory) | |
| mTORC1 | Mechanistic Target of Rapamycin Complex 1 | |
| mtDNA | Mitochondrial DNA | |
| NK cells | Natural Killer Cells | |
| NCDs | Non-Communicable Diseases | |
| NRF2 | Nuclear Factor Erythroid 2–Related Factor 2 | |
| OxPhos | Oxidative Phosphorylation | |
| POPs | Persistent Organic Pollutants | |
| PRR | Pattern Recognition Receptor | |
| REDs | Relative Energy Deficiency in Sport | |
| ROS | Reactive Oxygen Species | |
| SAM system | Sympathetic–Adrenal–Medullary System | |
| SASP | Senescence-Associated Secretory Phenotype | |
| Treg | Regulatory T Cells | |
| TNF-α | Tumor Necrosis Factor Alpha | |
| UPRmt | Mitochondrial Unfolded Protein Response | |
Funding
Conflicts of interest/Competing interests
Written Consent for publication
Availability of data and material
Code availability
Authors' contributions
Acknowledgments
Declaration of generative AI and AI-assisted technologies in the writing process
References
- Adams, S. H.; Anthony, J. C.; Carvajal, R.; Chae, L.; Khoo, C. S. H.; Latulippe, M. E.; Yan, W. Perspective: Guiding Principles for the Implementation of Personalized Nutrition Approaches That Benefit Health and Function. Adv Nutr 2020, 11(1), 25–34. [Google Scholar] [CrossRef] [PubMed]
- Alack, K.; Pilat, C.; Krüger K*Shared, a. Current Knowledge and New Challenges in Exercise Immunology. Deutsche Zeitschrift für Sportmedizin 2019, 70(No. 10), 250–260. [Google Scholar] [CrossRef]
- Ames, B. N. Low micronutrient intake may accelerate the degenerative diseases of aging through allocation of scarce micronutrients by triage. Proc Natl Acad Sci U S A 2006, 103(47), 17589–17594. [Google Scholar] [CrossRef] [PubMed]
- Aujla, R. S.; Zubair, M.; Patel, R. Creatine Phosphokinase. In StatPearls; StatPearls Publishing, 2025. [Google Scholar]
- Baldwin, J. R.; Caspi, A.; Meehan, A. J.; Ambler, A.; Arseneault, L.; Fisher, H. L.; Danese, A. Population vs Individual Prediction of Poor Health From Results of Adverse Childhood Experiences Screening. JAMA Pediatrics 2021, 175(4), 385–393. [Google Scholar] [CrossRef]
- Bar-Ziv, R.; Bolas, T.; Dillin, A. Systemic effects of mitochondrial stress. EMBO reports 2020, 21(6), e50094. [Google Scholar] [CrossRef] [PubMed]
- Beaudart, C.; Dawson, A.; Shaw, S. C.; Harvey, N. C.; Kanis, J. A.; Binkley, N.; Dennison, E. M. Nutrition and physical activity in the prevention and treatment of sarcopenia: systematic review. Osteoporos Int 2017, 28(6), 1817–1833. [Google Scholar] [CrossRef]
- Bobba-Alves, N.; Juster, R.-P.; Picard, M. The energetic cost of allostasis and allostatic load. Psychoneuroendocrinology 2022, 146, 105951. [Google Scholar] [CrossRef]
- Bobba-Alves, N.; Sturm, G.; Lin, J.; Ware, S. A.; Karan, K. R.; Monzel, A. S.; Picard, M. Cellular allostatic load is linked to increased energy expenditure and accelerated biological aging. Psychoneuroendocrinology 2023, 155, 106322. [Google Scholar] [CrossRef]
- Branco, M. G.; Mateus, C.; Capelas, M. L.; Pimenta, N.; Santos, T.; Mäkitie, A.; Ravasco, P. Bioelectrical Impedance Analysis (BIA) for the Assessment of Body Composition in Oncology: A Scoping Review. Nutrients 2023, 15(22), 4792. Available online: https://www.mdpi.com/2072-6643/15/22/4792. [CrossRef]
- Bresnahan, K. A.; Tanumihardjo, S. A. Undernutrition, the acute phase response to infection, and its effects on micronutrient status indicators. Adv Nutr 2014, 5(6), 702–711. [Google Scholar] [CrossRef]
- Cabre, H. E.; Moore, S. R.; Smith-Ryan, A. E.; Hackney, A. C. Relative Energy Deficiency in Sport (RED-S): Scientific, Clinical, and Practical Implications for the Female Athlete. Dtsch Z Sportmed 2022, 73(7), 225–234. [Google Scholar] [CrossRef] [PubMed]
- Cahill, G. F., Jr. Fuel metabolism in starvation. Annu Rev Nutr 2006, 26, 1–22. [Google Scholar] [CrossRef]
- Calabrese, E. J.; Agathokleous, E. Building Biological Shields via Hormesis. Trends Pharmacol Sci 2019, 40(1), 8–10. [Google Scholar] [CrossRef]
- Careccia, G.; Mangiavini, L.; Cirillo, F. Regulation of Satellite Cells Functions during Skeletal Muscle Regeneration: A Critical Step in Physiological and Pathological Conditions. Int J Mol Sci 2023, 25(1). [Google Scholar] [CrossRef] [PubMed]
- Carling, R. S.; Witek, K.; Emmett, E. C.; Gallagher, C.; Moat, S. J. Urine organic acid metabolomic profiling by gas chromatography mass spectrometry: Assessment of solvent extract evaporation parameters on the recovery of key diagnostic metabolites. Clinica Chimica Acta 2025, 565, 120015. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Bosaeus, I. Malnutrition in Adults. New England Journal of Medicine 2024, 391(2), 155–165. [Google Scholar] [CrossRef]
- Chen, W.; Kahn, C. R. Insulin. Trends in Endocrinology & Metabolism 2024. [Google Scholar] [CrossRef]
- Chrousos, G. P. Stress and disorders of the stress system. Nat Rev Endocrinol 2009, 5(7), 374–381. [Google Scholar] [CrossRef]
- Costa-Mattioli, M.; Walter, P. The integrated stress response: From mechanism to disease. Science 2020, 368(6489), eaat5314. [Google Scholar] [CrossRef]
- Crane, P. A.; Wilkinson, G.; Teare, H. Healthspan versus lifespan: new medicines to close the gap. Nature Aging 2022, 2(11), 984–988. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A. J.; Gonzalez, M. C.; Prado, C. M. Sarcopenia ≠ low muscle mass. European Geriatric Medicine 2023, 14(2), 225–228. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R. P.; Finlay, D. K. Glucose, glycolysis and lymphocyte responses. Mol Immunol 2015, 68 2 Pt C, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Evans, D. C.; Corkins, M. R.; Malone, A.; Miller, S.; Mogensen, K. M.; Guenter, P.; Committee, A. M. The Use of Visceral Proteins as Nutrition Markers: An ASPEN Position Paper. Nutr Clin Pract 2021, 36(1), 22–28. [Google Scholar] [CrossRef]
- Fang, Y.; Li, Z.; Yang, L.; Li, W.; Wang, Y.; Kong, Z.; Zeng, L. Emerging roles of lactate in acute and chronic inflammation. Cell Communication and Signaling 2024, 22(1), 276. [Google Scholar] [CrossRef]
- Foy, B. H.; Petherbridge, R.; Roth, M. T.; Zhang, C.; De Souza, D. C.; Mow, C.; Higgins, J. M. Haematological setpoints are a stable and patient-specific deep phenotype. Nature 2024. [Google Scholar] [CrossRef]
- Franceschi, C.; Garagnani, P.; Parini, P.; Giuliani, C.; Santoro, A. Inflammaging: a new immune–metabolic viewpoint for age-related diseases. Nature Reviews Endocrinology 2018, 14(10), 576–590. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M. I.; Sørensen, T. I. A.; Taubes, G.; Lund, J.; Ludwig, D. S. Trapped fat: Obesity pathogenesis as an intrinsic disorder in metabolic fuel partitioning. Obesity Reviews 2024. n/a(n/a). [Google Scholar] [CrossRef]
- Fulop, T.; Larbi, A.; Dupuis, G.; Le Page, A.; Frost, E. H.; Cohen, A. A.; Franceschi, C. Immunosenescence and Inflamm-Aging As Two Sides of the Same Coin: Friends or Foes? [Review]. Front Immunol 2018, 8. [Google Scholar] [CrossRef]
- Gambardella, G.; Staiano, L.; Moretti, M. N.; De Cegli, R.; Fagnocchi, L.; Di Tullio, G.; di Bernardo, D. GADD34 is a modulator of autophagy during starvation. Science Advances 2020, 6(39), eabb0205. [Google Scholar] [CrossRef]
- Geric, I.; Schoors, S.; Claes, C.; Gressens, P.; Verderio, C.; Verfaillie, C. M.; Baes, M. Metabolic Reprogramming during Microglia Activation. Immunometabolism 2019, 1(1), e190002, Article e190002. [Google Scholar] [CrossRef]
- Gulhar, R.; Ashraf, M. A.; Jialal, I. Physiology, Acute Phase Reactants. In StatPearls; StatPearls Publishing, 2024. [Google Scholar]
- Herman, J. P. Neural control of chronic stress adaptation. Front Behav Neurosci 2013, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C.; Papa, F. R. The Unfolded Protein Response and Cell Fate Control. Molecular Cell 2018, 69(2), 169–181. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R. J.; Sánchez-Lozada, L. G.; Lanaspa, M. A. The fructose survival hypothesis as a mechanism for unifying the various obesity hypotheses. Obesity 2024, 32(1), 12–22. [Google Scholar] [CrossRef]
- Kalinkovich, A.; Livshits, G. Sarcopenic obesity or obese sarcopenia: A cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of the pathogenesis. Ageing Res Rev 2017, 35, 200–221. [Google Scholar] [CrossRef] [PubMed]
- Langston, P. K.; Mathis, D. Immunological regulation of skeletal muscle adaptation to exercise. Cell Metab 2024. [Google Scholar] [CrossRef]
- Laurent, P.; Jolivel, V.; Manicki, P.; Chiu, L.; Contin-Bordes, C.; Truchetet, M.-E.; Pradeu, T. Immune-Mediated Repair: A Matter of Plasticity [Mini Review]. Front Immunol 2017, 8. [Google Scholar] [CrossRef]
- Lee, S. Y.; Lee, Y. J.; Yang, J. H.; Kim, C. M.; Choi, W. S. The Association between Phase Angle of Bioelectrical Impedance Analysis and Survival Time in Advanced Cancer Patients: Preliminary Study. Korean J Fam Med 2014, 35(5), 251–256. [Google Scholar] [CrossRef]
- Lockhart, S. M.; Saudek, V.; O'Rahilly, S. GDF15: A Hormone Conveying Somatic Distress to the Brain. Endocr Rev 2020, 41(4). [Google Scholar] [CrossRef]
- Ludwig, D. S. Carbohydrate-insulin model: does the conventional view of obesity reverse cause and effect? Philosophical Transactions of the Royal Society B: Biological Sciences 2023, 378(1888), 20220211. [Google Scholar] [CrossRef]
- McEwen, B. S. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev 2007, 87(3), 873–904. [Google Scholar] [CrossRef]
- McEwen, B. S.; Wingfield, J. C. The concept of allostasis in biology and biomedicine. Hormones and Behavior 2003, 43(1), 2–15. [Google Scholar] [CrossRef] [PubMed]
- Meeusen, R.; Watson, P.; Hasegawa, H.; Roelands, B.; Piacentini, M. F. Central fatigue: the serotonin hypothesis and beyond. Sports Med 2006, 36(10), 881–909. [Google Scholar] [CrossRef]
- Metallo, Christian M.; Vander Heiden, Matthew G. Understanding Metabolic Regulation and Its Influence on Cell Physiology. Molecular Cell 2013, 49(3), 388–398. [Google Scholar] [CrossRef] [PubMed]
- Monzel, A. S.; Levin, M.; Picard, M. The energetics of cellular life transitions. Life Metabolism 2023, 3(3). [Google Scholar] [CrossRef]
- Mountjoy, M.; Sundgot-Borgen, J. K.; Burke, L. M.; Ackerman, K. E.; Blauwet, C.; Constantini, N.; Budgett, R. IOC consensus statement on relative energy deficiency in sport (RED-S): 2018 update. Br J Sports Med 2018, 52(11), 687–697. [Google Scholar] [CrossRef] [PubMed]
- Muscaritoli, M.; Imbimbo, G.; Jager-Wittenaar, H.; Cederholm, T.; Rothenberg, E.; di Girolamo, F. G.; Molfino, A. Disease-related malnutrition with inflammation and cachexia. Clin Nutr 2023, 42(8), 1475–1479. [Google Scholar] [CrossRef]
- Nakajima, K.; Yuno, M.; Tanaka, K.; Nakamura, T. High Aspartate Aminotransferase/Alanine Aminotransferase Ratio May Be Associated with All-Cause Mortality in the Elderly: A Retrospective Cohort Study Using Artificial Intelligence and Conventional Analysis. Healthcare (Basel) 2022, 10(4). [Google Scholar] [CrossRef]
- Netea, M. G.; Joosten, L. A.; Latz, E.; Mills, K. H.; Natoli, G.; Stunnenberg, H. G.; Xavier, R. J. Trained immunity: A program of innate immune memory in health and disease. Science 2016, 352(6284), aaf1098. [Google Scholar] [CrossRef]
- Novoa, I.; Zeng, H.; Harding, H. P.; Ron, D. Feedback inhibition of the unfolded protein response by GADD34-mediated dephosphorylation of eIF2alpha. J Cell Biol 2001, 153(5), 1011–1022. [Google Scholar] [CrossRef]
- Ochando, J.; Mulder, W. J. M.; Madsen, J. C.; Netea, M. G.; Duivenvoorden, R. Trained immunity—basic concepts and contributions to immunopathology. Nature Reviews Nephrology 2023, 19(1), 23–37. [Google Scholar] [CrossRef]
- Güngör, Olcay; Kıreker Köylü, Oya; Dalkıran, Tahir; Kırık, Serkan; Tepe, Elif; Cevizli, Derya; Dilber, C. Evaluation of blood neuron specific enolase and S-100 beta protein levels in acute mercury toxicity. Trace Elements and Electrolytes 2018, 35, 131–136. [Google Scholar] [CrossRef]
- Olenchock, B. A.; Rathmell, J. C.; Vander Heiden, M. G. Biochemical Underpinnings of Immune Cell Metabolic Phenotypes. Immunity 2017, 46(5), 703–713. [Google Scholar] [CrossRef] [PubMed]
- Pakos-Zebrucka, K.; Koryga, I.; Mnich, K.; Ljujic, M.; Samali, A.; Gorman, A. M. The integrated stress response. EMBO reports 2016, 17(10), 1374–1395. [Google Scholar] [CrossRef]
- Paulussen, K. J. M.; McKenna, C. F.; Beals, J. W.; Wilund, K. R.; Salvador, A. F.; Burd, N. A. Anabolic Resistance of Muscle Protein Turnover Comes in Various Shapes and Sizes. Front Nutr 2021, 8, 615849. [Google Scholar] [CrossRef]
- Peeri, N. C.; Chai, W.; Cooney, R. V.; Tao, M. H. Association of serum levels of antioxidant micronutrients with mortality in US adults: National Health and Nutrition Examination Survey 1999-2002. Public Health Nutr 2021, 24(15), 4859–4868. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; Shirihai, O. S. Mitochondrial signal transduction. Cell Metab 2022, 34(11), 1620–1653. [Google Scholar] [CrossRef] [PubMed]
- Pizzorno, J. Thoughts on a Unified Theory of Disease. Integrative medicine (Encinitas, Calif.) 2020, 19(6), 8–17. [Google Scholar]
- Prisabela, M.; Nadhiroh, S. R.; Isaura, E. R. Characteristics of Pregnant Woman with Chronic Energy Deficiency in Puskesmas Gesang, Lumajang on 2020: Descriptive Analysis. Media Gizi Kesmas 2023, 12(2), 643–648. [Google Scholar] [CrossRef]
- Qi, Y.; Rajbanshi, B.; Hao, R.; Dang, Y.; Xu, C.; Lu, W.; Zhang, X. The dual role of PGAM5 in inflammation. Exp Mol Med 2025, 57(2), 298–311. [Google Scholar] [CrossRef]
- Ristow, M.; Schmeisser, K. Mitohormesis: Promoting Health and Lifespan by Increased Levels of Reactive Oxygen Species (ROS). Dose-Response 2014, 12(2), dose-response.13-035.Ristow. [Google Scholar] [CrossRef]
- Ryan, M.; Ryznar, R. The Molecular Basis of Resilience: A Narrative Review [Review]. Frontiers in Psychiatry 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Sapolsky, R. M. Why zebras don’t get ulcers: The acclaimed guide to stress, stress-related diseases, and coping, 3rd ed.; Henry Holt, 2004. [Google Scholar]
- Seiler, A.; Fagundes, C. P.; Christian, L. M. Choukèr, A., Ed.; The Impact of Everyday Stressors on the Immune System and Health. In Stress Challenges and Immunity in Space: From Mechanisms to Monitoring and Preventive Strategies; Springer International Publishing, 2020; pp. 71–92. [Google Scholar] [CrossRef]
- Selye, H. Stress and the General Adaptation Syndrome. British Medical Journal 1950, 1(4667), 1383–1392. Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2038162/. [CrossRef]
- Sganga, G.; Siegel, J. H.; Brown, G.; Coleman, B.; Wiles, C. E., 3rd; Belzberg, H.; Placko, R. Reprioritization of hepatic plasma protein release in trauma and sepsis. Arch Surg 1985, 120(2), 187–199. [Google Scholar] [CrossRef]
- Shaulson, E. D.; Cohen, A. A.; Picard, M. The brain–body energy conservation model of aging. Nature Aging 2024, 4(10), 1354–1371. [Google Scholar] [CrossRef] [PubMed]
- Sparkenbaugh, E. M.; Saini, Y.; Greenwood, K. K.; LaPres, J. J.; Luyendyk, J. P.; Copple, B. L.; Roth, R. A. The Role of Hypoxia-Inducible Factor-1α in Acetaminophen Hepatotoxicity. J Pharmacol Exp Ther 2011, 338(2), 492–502. [Google Scholar] [CrossRef] [PubMed]
- Speakman, J. R.; Hall, K. D. Carbohydrates, insulin, and obesity. Science 2021, 372(6542), 577–578. [Google Scholar] [CrossRef]
- Straub, R. H. The brain and immune system prompt energy shortage in chronic inflammation and ageing [Perspective]. Nat Rev Rheumatol 2017, 13, 743–751. [Google Scholar] [CrossRef]
- Taylor-Baer, M.; Herman, D. Halfon, N., Forrest, C. B., Lerner, R. M., Faustman, E. M., Eds.; From Epidemiology to Epigenetics: Evidence for the Importance of Nutrition to Optimal Health Development Across the Life Course. In Handbook of Life Course Health Development; Springer International Publishing, 2018; pp. 431–462. [Google Scholar] [CrossRef]
- Tsigos, C.; Chrousos, G. P. Hypothalamic–pituitary–adrenal axis, neuroendocrine factors and stress. Journal of Psychosomatic Research 2002, 53(4), 865–871. [Google Scholar] [CrossRef]
- Vermeulen, R.; Schymanski, E. L.; Barabási, A. L.; Miller, G. W. The exposome and health: Where chemistry meets biology. Science 2020, 367(6476), 392–396. [Google Scholar] [CrossRef]
- Vuscan, P.; Kischkel, B.; Joosten, L. A. B.; Netea, M. G. Trained immunity: General and emerging concepts. Immunological Reviews 2024, 323(1), 164–185. [Google Scholar] [CrossRef]
- Walrand, S.; Guillet, C.; Boirie, Y. Nutrition, Protein Turnover and Muscle Mass. In Sarcopenia; 2021; pp. 45–62. [Google Scholar] [CrossRef]
- Wang, L.; Hong, W.; Zhu, H.; He, Q.; Yang, B.; Wang, J.; Weng, Q. Macrophage senescence in health and diseases. Acta Pharm Sin B 2024, 14(4), 1508–1524. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, G. The mitochondrial integrated stress response: A novel approach to anti-aging and pro-longevity. Ageing Res Rev 2025, 103, 102603. [Google Scholar] [CrossRef] [PubMed]
- Warde, K. M.; Smith, L. J.; Basham, K. J. Age-related Changes in the Adrenal Cortex: Insights and Implications. Journal of the Endocrine Society 2023, 7(9), bvad097. [Google Scholar] [CrossRef]
- Wek, R. C. Role of eIF2α Kinases in Translational Control and Adaptation to Cellular Stress. Cold Spring Harb Perspect Biol 2018, 10(7). [Google Scholar] [CrossRef]
- West, J. Effects of a cognitive behavioural intervention on athletes' stress, recovery and performance; The University of Queensland, 2008. Available online: https://espace.library.uq.edu.au/view/UQ:151292.
- WHO. Constitution of the World Health Organization; World Health Organization, 1948. Available online: https://www.who.int/about/governance/constitution.
- WHO. Noncommunicable diseases; WHO, 2025. Available online: https://www.who.int/health-topics/noncommunicable-diseases#tab=tab_1.
- Willmann, K.; Moita, L. F. Physiologic disruption and metabolic reprogramming in infection and sepsis. Cell Metab 2024. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R. R. The underappreciated role of muscle in health and disease. Am J Clin Nutr 2006, 84(3), 475–482. [Google Scholar] [CrossRef]
- Yiallouris, A.; Tsioutis, C.; Agapidaki, E.; Zafeiri, M.; Agouridis, A. P.; Ntourakis, D.; Johnson, E. O. Adrenal Aging and Its Implications on Stress Responsiveness in Humans [Review]. Frontiers in Endocrinology 2019, 10(54). [Google Scholar] [CrossRef]
- Youle, R. J.; van der Bliek, A. M. Mitochondrial Fission, Fusion, and Stress. Science 2012, 337(6098), 1062–1065. [Google Scholar] [CrossRef]
- Yuk, J.-M.; Silwal, P.; Jo, E.-K. Inflammasome and Mitophagy Connection in Health and Disease. Int J Mol Sci 2020, 21(13), 4714. Available online: https://www.mdpi.com/1422-0067/21/13/4714. [CrossRef]
- Zera, A. J.; Harshman, L. G. The Physiology of Life History Trade-Offs in Animals. Annual Review of Ecology, Evolution, and Systematics 2001, 32(2001), 95–126. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




