Submitted:
12 April 2025
Posted:
14 April 2025
You are already at the latest version
Abstract
Keywords:

1. Introduction
| Inset: “Goldilocks” refers to the classic fairy tale Goldilocks and the Three Bears, first modernized by Joseph Cundall in 1850. It tells the story of a young blonde girl who stumbles upon a cottage in the forest. Inside, she samples the bears’ porridge—one is too hot, one is too cold, and one is just right. She tries their chairs—one is too big, one too small, and one is just right. Finally, she tests their beds—one is too hard, one is too soft, and one is just right, where she falls asleep. The tale underscores the importance of balance. |
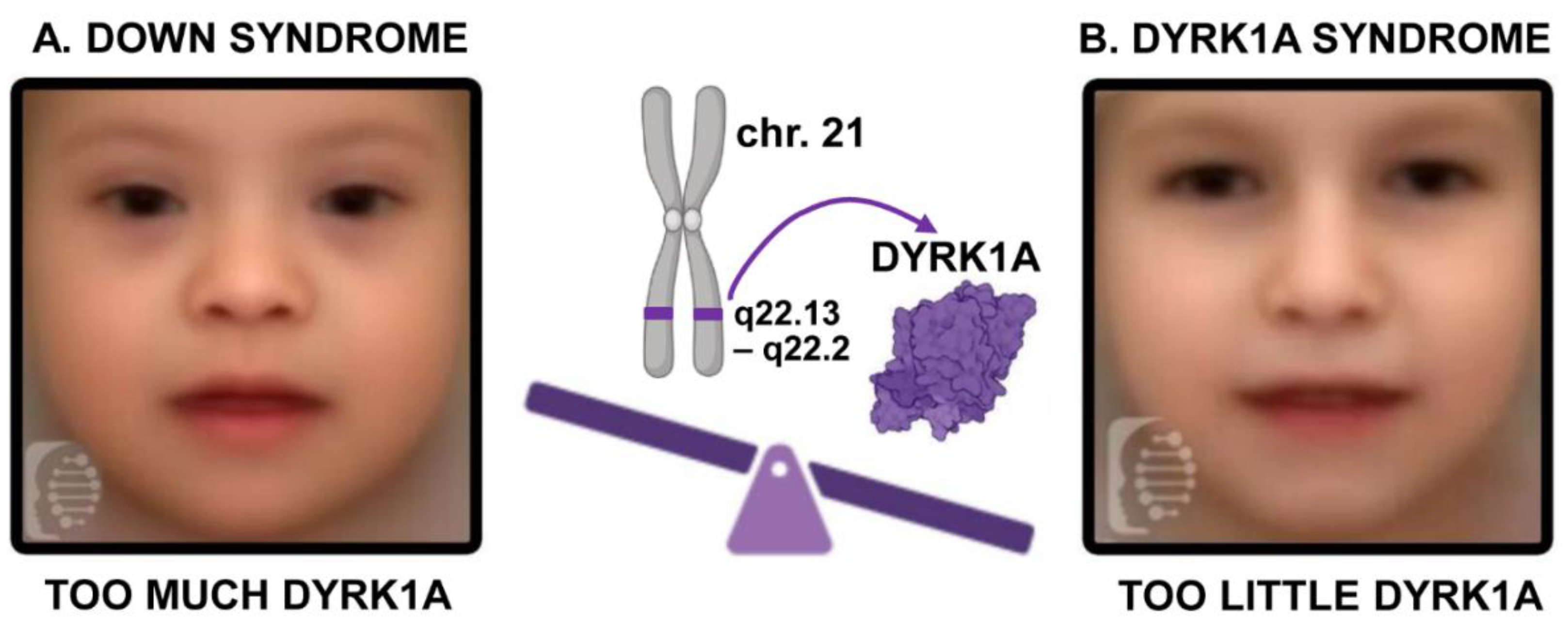
2. Too Little DYRK1A
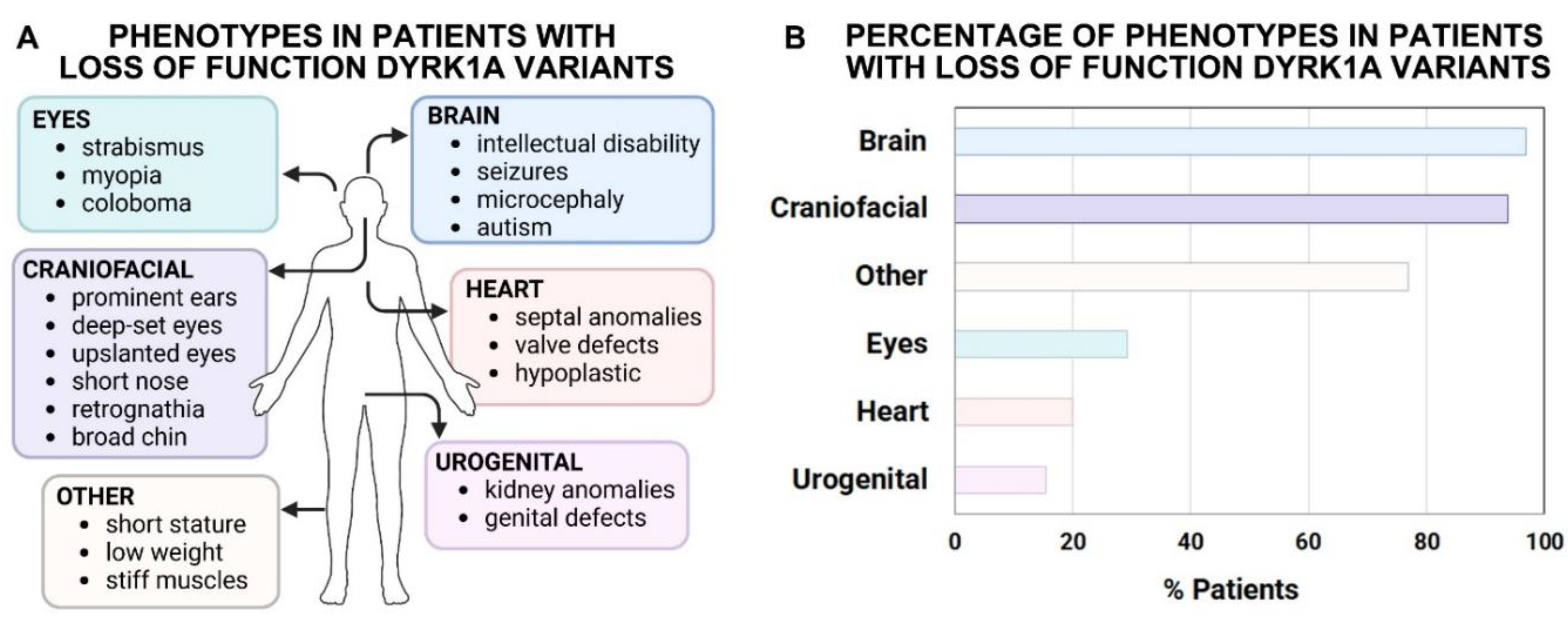
2.1. DYRK1A Syndrome and Loss of Function Variants in Humans
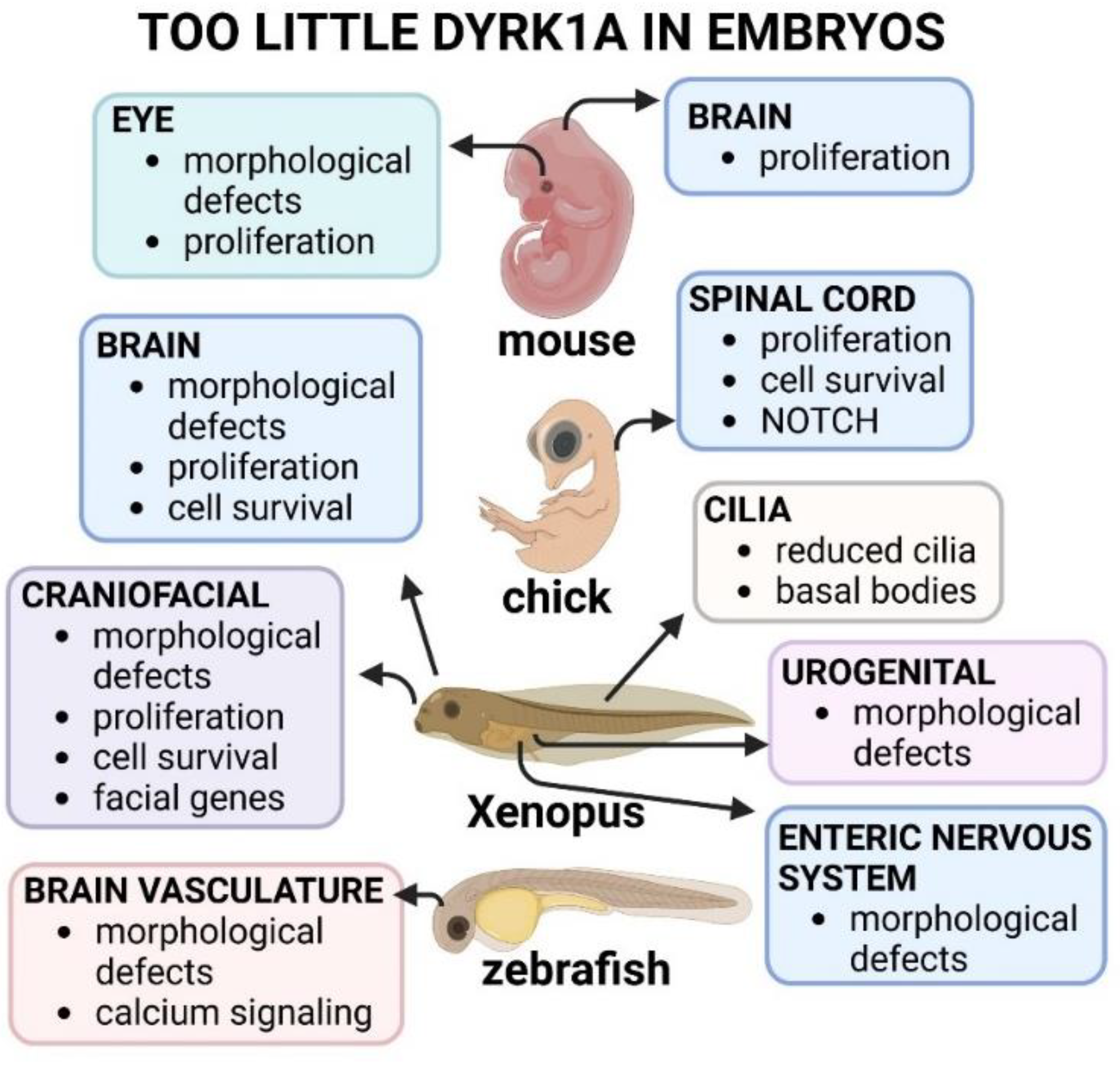
2.2. Too Little Dyrk1a Causes Developmental Anomalies in Vertebrate Animal Models
2.2.1. Too Little Dyrk1a Perturbs Central Nervous System Development
2.2.2. Too Little Dyrk1a Causes Craniofacial Differences
2.2.3. Too Little Dyrk1a Causes Defects in the Cerebral Vasculature
2.2.4. Too Little Dyrk1a Causes Kidney Anomalies
2.2.6. Too Little Dyrk1a Perturbs Ciliogenesis
2.2.7. Too Little Dyrk1a Alters Eye Development
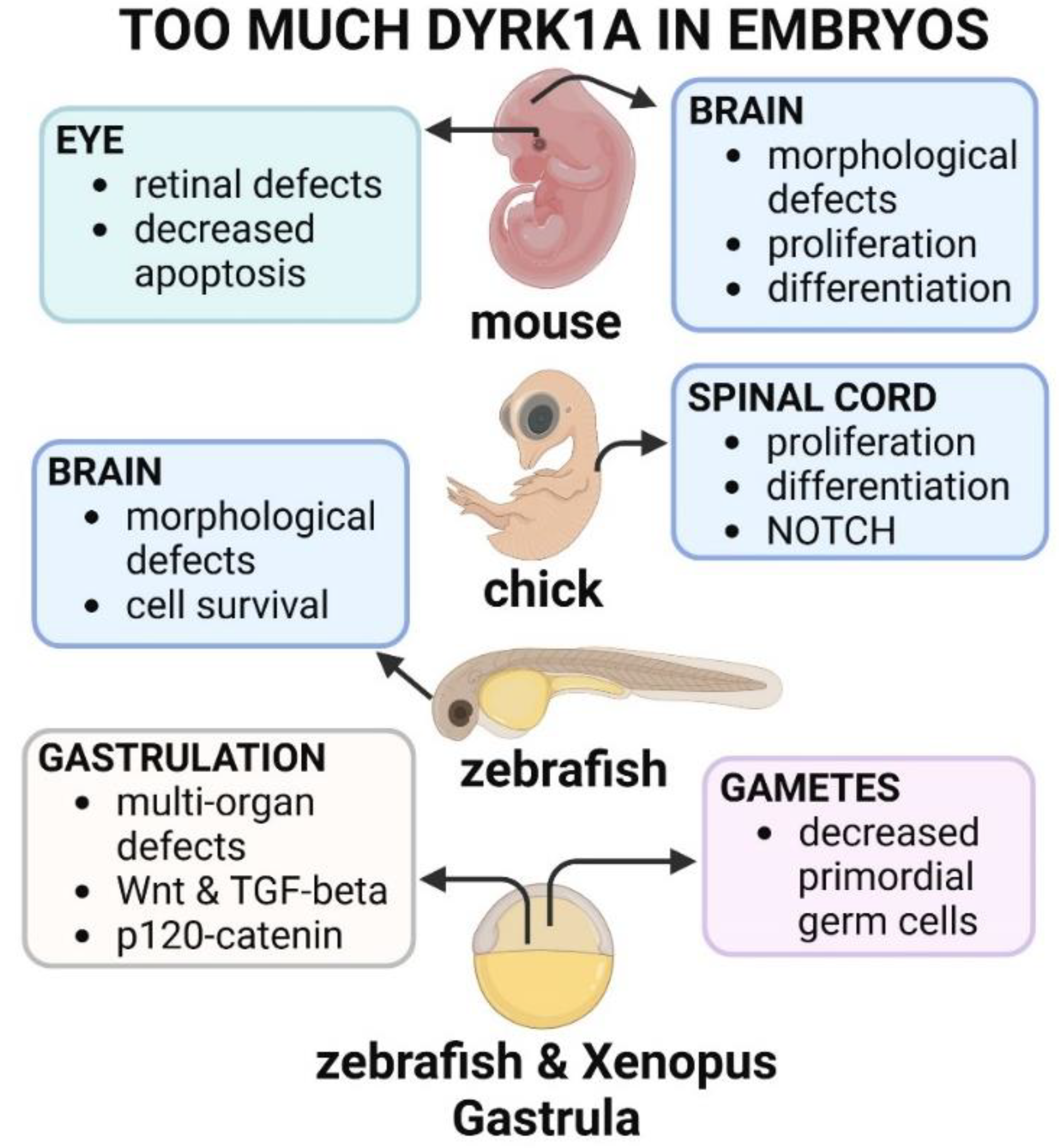
3. Too Much DYRK1A
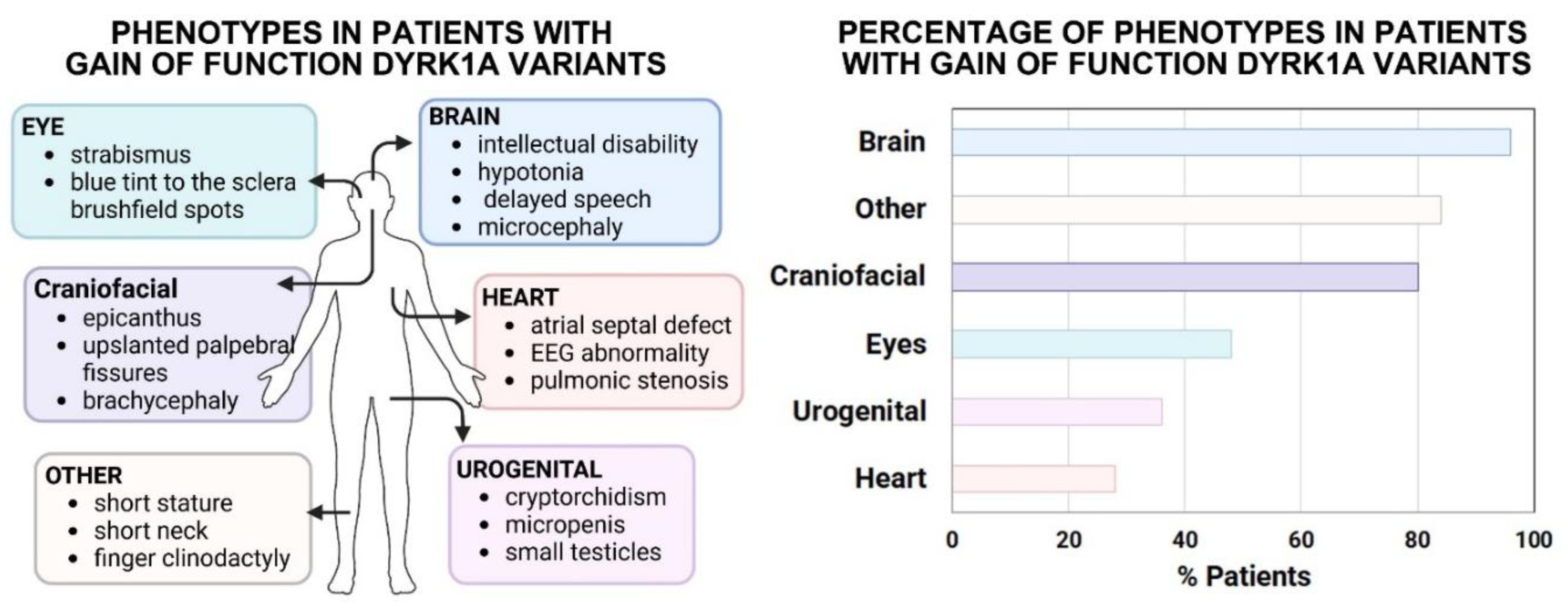
3.1. DYRK1A Gain of Function Copy Number Variants in Humans
3.2. Too Much Dyrk1a in Vertebrate Animal Models Causes Developmental Defects
3.2.1. Too Much Dyrk1a Disrupts Gastrulation
3.2.2. Too Much Dyrk1a Disrupts Central Nervous System Development
3.2.3. Too Much Dyrk1a Disrupts Germ Cell Development
3.2.4. Too Much Dyrk1a Causes Eye Malformations
3.3. Down Syndrome
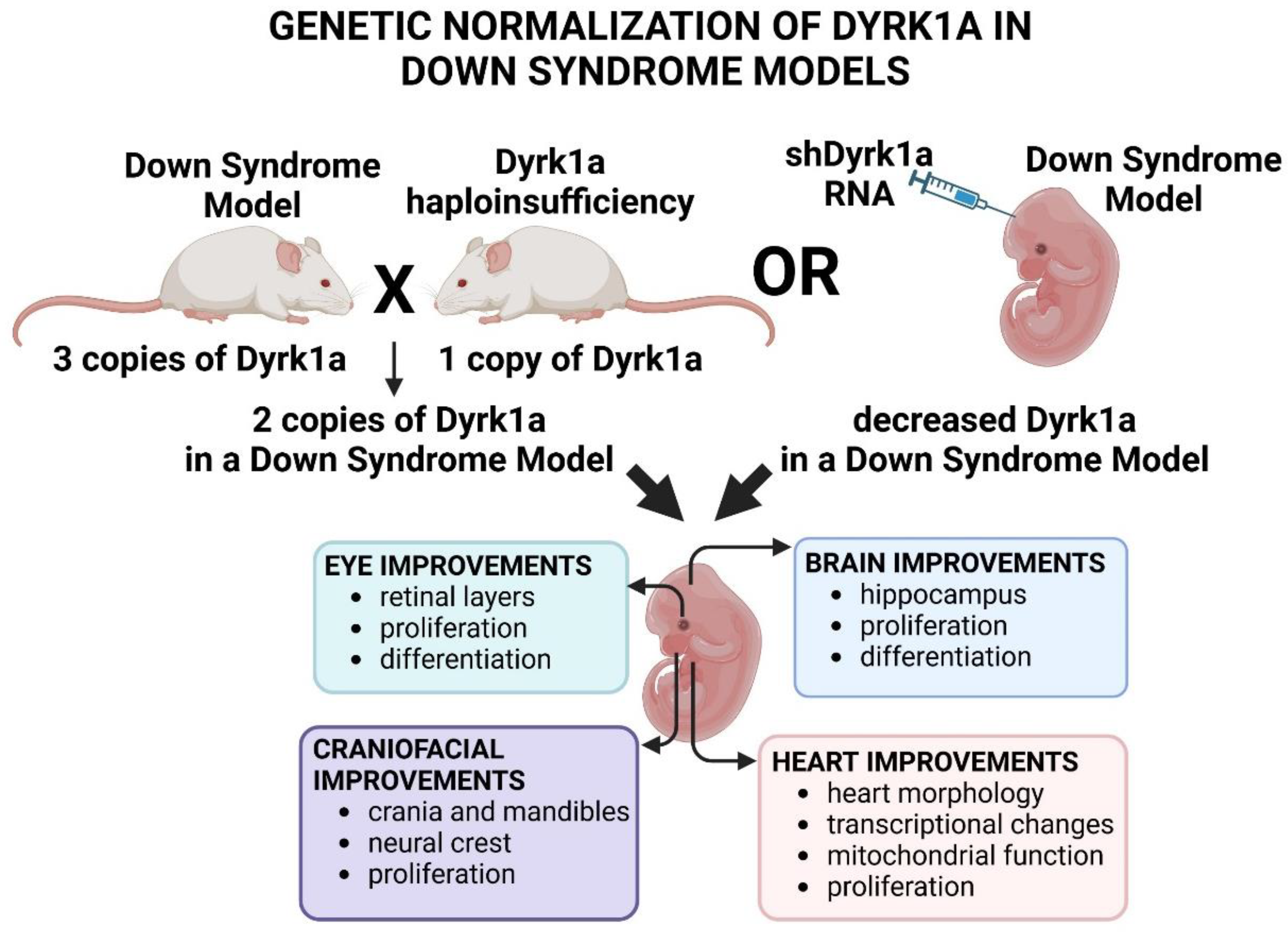
3.4. Normalizing DYRK1A Dosage in Animal Models of Down Syndrome
3.4.1. Genetically Normalizing Dyrk1a Can Improve Developmental Anomalies in Down Syndrome Mouse Models
3.4.1.1. Brain Development
3.4.1.2. Craniofacial Development
3.4.1.3. Heart Development
3.4.1.4. Eye Development
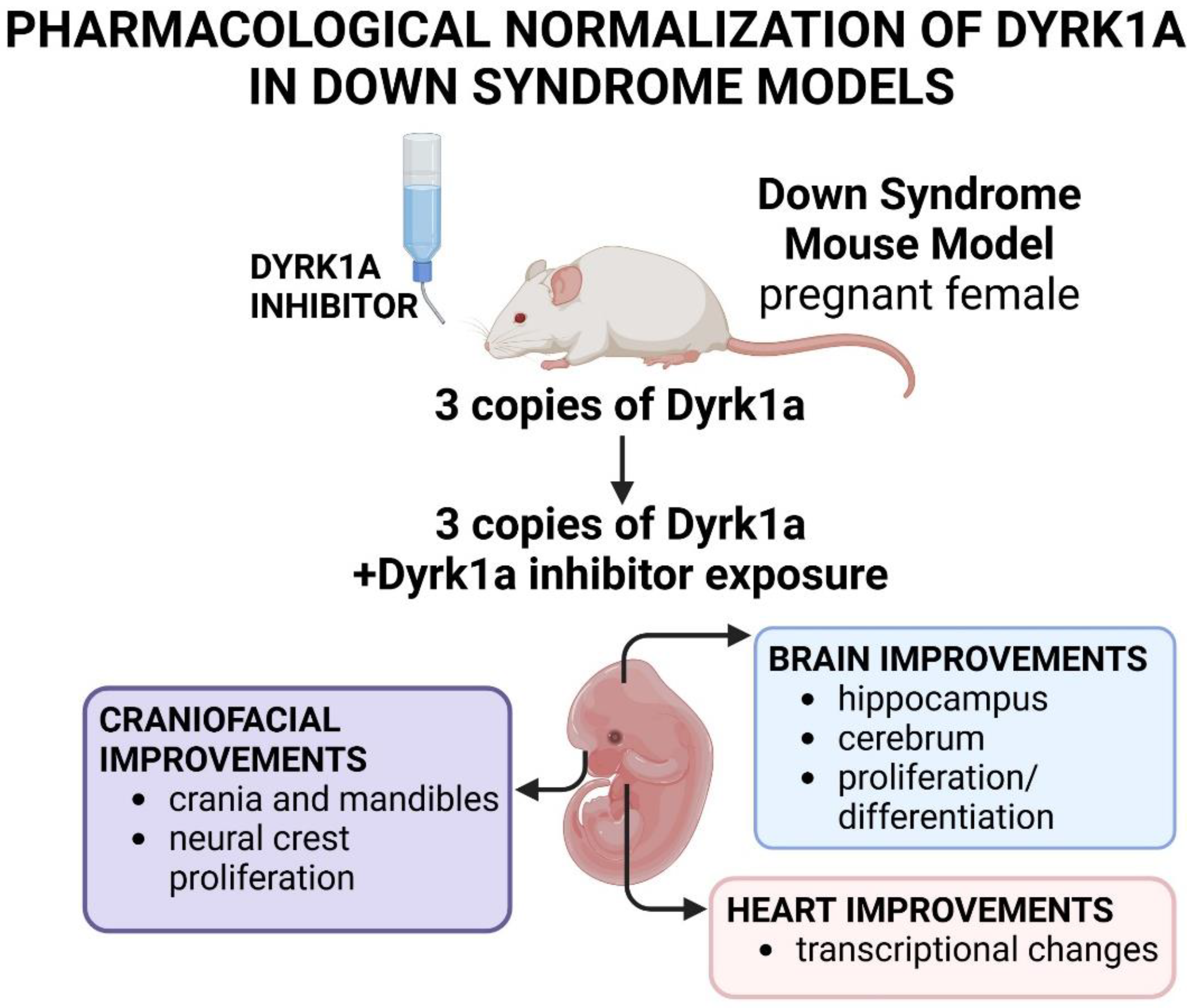
3.4.2. Normalizing Dyrk1a Pharmacologically Can Improve Developmental Anomalies in Down Syndrome Mouse Models
3.4.2.1. Brain Development
3.4.2.2. Craniofacial Development
3.4.2.3. Heart Development
4. Conclusions and Future Directions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abbassi, R.; Johns, T.G.; Kassiou, M.; Munoz, L. DYRK1A in neurodegeneration and cancer: Molecular basis and clinical implications. Pharmacol Ther, 2015, 151, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Abramyan, J. Hedgehog Signaling and Embryonic Craniofacial Disorders. J Dev Biol 2019, 7. [Google Scholar] [CrossRef]
- Adayev, T.; Wegiel, J.; Hwang, Y.W. Harmine is an ATP-competitive inhibitor for dual-specificity tyrosine phosphorylation-regulated kinase 1A (Dyrk1A). Arch Biochem Biophys, 2011, 507, 212–218. [Google Scholar] [CrossRef]
- Ahn, K.J.; Jeong, H.K.; Choi, H.S.; Ryoo, S.R.; Kim, Y.J.; Goo, J.S.; Choi, S.Y.; Han, J.S.; Ha, I.; Song, W.J. DYRK1A BAC transgenic mice show altered synaptic plasticity with learning and memory defects. Neurobiol Dis, 2006, 22, 463–472. [Google Scholar] [CrossRef]
- Allareddy, V.; Ching, N.; Macklin, E.A.; Voelz, L.; Weintraub, G.; Davidson, E.; Prock, L.A.; Rosen, D.; Brunn, R.; Skotko, B.G. Craniofacial features as assessed by lateral cephalometric measurements in children with Down syndrome. Progress in orthodontics, 2016, 17, 35. [Google Scholar] [CrossRef]
- Altafaj, X.; Dierssen, M.; Baamonde, C.; Martí, E.; Visa, J.; Guimerà, J.; Oset, M.; González, J.R.; Flórez, J.; Fillat, C.; Estivill, X. Neurodevelopmental delay, motor abnormalities and cognitive deficits in transgenic mice overexpressing Dyrk1A (minibrain), a murine model of Down's syndrome. Human molecular genetics, 2001, 10, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Altafaj, X.; Martín, E.D.; Ortiz-Abalia, J.; Valderrama, A.; Lao-Peregrín, C.; Dierssen, M.; Fillat, C. Normalization of Dyrk1A expression by AAV2/1-shDyrk1A attenuates hippocampal-dependent defects in the Ts65Dn mouse model of Down syndrome. Neurobiol Dis, 2013, 52, 117–127. [Google Scholar] [CrossRef]
- Amack, J.D. Structures and functions of cilia during vertebrate embryo development. Mol Reprod Dev, 2022, 89, 579–596. [Google Scholar] [CrossRef] [PubMed]
- Ananthapadmanabhan, V.; Shows, K.H.; Dickinson, A.J.; Litovchick, L. Insights from the protein interaction Universe of the multifunctional "Goldilocks" kinase DYRK1A. Front Cell Dev Biol, 2023, 11, 1277537. [Google Scholar] [CrossRef]
- Antonarakis, S.E. Down syndrome and the complexity of genome dosage imbalance. Nature reviews. Genetics, 2017, 18, 147–163. [Google Scholar] [CrossRef]
- Antonarakis, S.E.; Lyle, R.; Dermitzakis, E.T.; Reymond, A.; Deutsch, S. Chromosome 21 and down syndrome: From genomics to pathophysiology. Nature reviews. Genetics, 2004, 5, 725–738. [Google Scholar] [CrossRef]
- Antonarakis, S.E.; Skotko, B.G.; Rafii, M.S.; Strydom, A.; Pape, S.E.; Bianchi, D.W.; Sherman, S.L.; Reeves, R.H. Down syndrome. Nat Rev Dis Primers, 2020, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- Anvarian, Z.; Mykytyn, K.; Mukhopadhyay, S.; Pedersen, L.B.; Christensen, S.T. Cellular signalling by primary cilia in development, organ function and disease. Nat Rev Nephrol, 2019, 15, 199–219. [Google Scholar] [CrossRef]
- Asim, A.; Kumar, A.; Muthuswamy, S.; Jain, S.; Agarwal, S. "Down syndrome: An insight of the disease". Journal of biomedical science, 2015, 22, 41. [Google Scholar] [CrossRef] [PubMed]
- Atas-Ozcan, H.; Brault, V.; Duchon, A.; Herault, Y. Dyrk1a from Gene Function in Development and Physiology to Dosage Correction across Life Span in Down Syndrome. Genes (Basel) 2021, 12. [Google Scholar] [CrossRef]
- Bain, J.; McLauchlan, H.; Elliott, M.; Cohen, P. The specificities of protein kinase inhibitors: An update. The Biochemical journal, 2003, 371, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Barallobre, M.J.; Perier, C.; Bové, J.; Laguna, A.; Delabar, J.M.; Vila, M.; Arbonés, M.L. DYRK1A promotes dopaminergic neuron survival in the developing brain and in a mouse model of Parkinson's disease. Cell Death Dis, 2014, 5, e1289. [Google Scholar] [CrossRef]
- Becker, W.; Sippl, W. Activation, regulation, and inhibition of DYRK1A. FEBS J, 2011, 278, 246–256. [Google Scholar] [CrossRef]
- Becker, W.; Soppa, U.; Tejedor, F.J. DYRK1A: A potential drug target for multiple Down syndrome neuropathologies. CNS & neurological disorders drug targets, 2014, 13, 26–33. [Google Scholar]
- Biehlmaier, O.; Neuhauss, S.C.; Kohler, K. Onset and time course of apoptosis in the developing zebrafish retina. Cell Tissue Res, 2001, 306, 199–207. [Google Scholar] [CrossRef]
- Blackburn, A.T.M.; Bekheirnia, N.; Uma, V.C.; Corkins, M.E.; Xu, Y.; Rosenfeld, J.A.; Bainbridge, M.N.; Yang, Y.; Liu, P.; Madan-Khetarpal, S.; Delgado, M.R.; Hudgins, L.; Krantz, I.; Rodriguez-Buritica, D.; Wheeler, P.G.; Al-Gazali, L.; Mohamed Saeed Mohamed Al Shamsi, A.; Gomez-Ospina, N.; Chao, H.T.; Mirzaa, G.M.; Scheuerle, A.E.; Kukolich, M.K.; Scaglia, F.; Eng, C.; Willsey, H.R.; Braun, M.C.; Lamb, D.J.; Miller, R.K.; Bekheirnia, M.R. DYRK1A-related intellectual disability: A syndrome associated with congenital anomalies of the kidney and urinary tract. Genet Med, 2019, 21, 2755–2764. [Google Scholar] [CrossRef] [PubMed]
- Bouwmeester, T. The Spemann-Mangold organizer: The control of fate specification and morphogenetic rearrangements during gastrulation in Xenopus. Int J Dev Biol, 2001, 45, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Branchi, I.; Bichler, Z.; Minghetti, L.; Delabar, J.M.; Malchiodi-Albedi, F.; Gonzalez, M.C.; Chettouh, Z.; Nicolini, A.; Chabert, C.; Smith, D.J.; Rubin, E.M.; Migliore-Samour, D.; Alleva, E. Transgenic mouse in vivo library of human Down syndrome critical region 1: Association between DYRK1A overexpression, brain development abnormalities, and cell cycle protein alteration. J Neuropathol Exp Neurol, 2004, 63, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Brooks, E.R.; Wallingford, J.B. Multiciliated cells. Curr Biol, 2014, 24, R973–R982. [Google Scholar] [CrossRef]
- Buchberger, A.; Schepergerdes, L.; Flaßhoff, M.; Kunick, C.; Köster, R.W. A novel inhibitor rescues cerebellar defects in a zebrafish model of Down syndrome-associated kinase Dyrk1A overexpression. The Journal of biological chemistry 2021, 297, 100853. [Google Scholar] [CrossRef]
- Bull, M.J. Down Syndrome. N Engl J Med, 2020, 382, 2344–2352. [Google Scholar] [CrossRef] [PubMed]
- Carron, C.; Shi, D.L. Specification of anteroposterior axis by combinatorial signaling during Xenopus development. Wiley Interdiscip Rev Dev Biol, 2016, 5, 150–168. [Google Scholar] [CrossRef]
- Chen, Y.; Xiao, Y.; Zhang, Y.; Wang, R.; Wang, F.; Gao, H.; Liu, Y.; Zhang, R.; Sun, H.; Zhou, Z.; Wang, S.; Chen, K.; Sun, Y.; Tu, M.; Li, J.; Luo, Q.; Wu, Y.; Zhu, L.; Huang, Y.; Sun, X.; Guo, G.; Zhang, D. Single-cell landscape analysis reveals systematic senescence in mammalian Down syndrome. Clin Transl Med, 2023, 13, e1310. [Google Scholar] [CrossRef]
- Cho, H.J.; Lee, J.G.; Kim, J.H.; Kim, S.Y.; Huh, Y.H.; Kim, H.J.; Lee, K.S.; Yu, K.; Lee, J.S. Vascular defects of DYRK1A knockouts are ameliorated by modulating calcium signaling in zebrafish. Dis Model Mech 2019, 12. [Google Scholar] [CrossRef]
- Cooper, G.M.; Coe, B.P.; Girirajan, S.; Rosenfeld, J.A.; Vu, T.H.; Baker, C.; Williams, C.; Stalker, H.; Hamid, R.; Hannig, V.; Abdel-Hamid, H.; Bader, P.; McCracken, E.; Niyazov, D.; Leppig, K.; Thiese, H.; Hummel, M.; Alexander, N.; Gorski, J.; Kussmann, J.; Shashi, V.; Johnson, K.; Rehder, C.; Ballif, B.C.; Shaffer, L.G.; Eichler, E.E. A copy number variation morbidity map of developmental delay. Nature genetics, 2011, 43, 838–846. [Google Scholar] [CrossRef]
- Courraud, J.; Chater-Diehl, E.; Durand, B.; Vincent, M.; Del Mar Muniz Moreno, M.; Boujelbene, I.; Drouot, N.; Genschik, L.; Schaefer, E.; Nizon, M.; Gerard, B.; Abramowicz, M.; Cogné, B.; Bronicki, L.; Burglen, L.; Barth, M.; Charles, P.; Colin, E.; Coubes, C.; David, A.; Delobel, B.; Demurger, F.; Passemard, S.; Denommé, A.S.; Faivre, L.; Feger, C.; Fradin, M.; Francannet, C.; Genevieve, D.; Goldenberg, A.; Guerrot, A.M.; Isidor, B.; Johannesen, K.M.; Keren, B.; Kibæk, M.; Kuentz, P.; Mathieu-Dramard, M.; Demeer, B.; Metreau, J.; Steensbjerre Møller, R.; Moutton, S.; Pasquier, L.; Pilekær Sørensen, K.; Perrin, L.; Renaud, M.; Saugier, P.; Rio, M.; Svane, J.; Thevenon, J.; Tran Mau Them, F.; Tronhjem, C.E.; Vitobello, A.; Layet, V.; Auvin, S.; Khachnaoui, K.; Birling, M.C.; Drunat, S.; Bayat, A.; Dubourg, C.; El Chehadeh, S.; Fagerberg, C.; Mignot, C.; Guipponi, M.; Bienvenu, T.; Herault, Y.; Thompson, J.; Willems, M.; Mandel, J.L.; Weksberg, R.; Piton, A. Integrative approach to interpret DYRK1A variants, leading to a frequent neurodevelopmental disorder. Genet Med, 2021, 23, 2150–2159. [Google Scholar] [CrossRef] [PubMed]
- Dard, R.; Moreau, M.; Parizot, E.; Ghieh, F.; Brehier, L.; Kassis, N.; Serazin, V.; Lamaziere, A.; Racine, C.; di Clemente, N.; Vialard, F.; Janel, N. DYRK1A Overexpression in Mice Downregulates the Gonadotropic Axis and Disturbs Early Stages of Spermatogenesis. Genes (Basel) 2021, 12. [Google Scholar] [CrossRef]
- De Felici, M. Regulation of primordial germ cell development in the mouse. Int J Dev Biol, 2000, 44, 575–580. [Google Scholar] [PubMed]
- de la Torre, R.; Dierssen, M. Therapeutic approaches in the improvement of cognitive performance in Down syndrome: Past, present, and future. Prog Brain Res, 2012, 197, 1–14. [Google Scholar] [PubMed]
- De Robertis, E.M. Spemann's organizer and the self-regulation of embryonic fields. Mech Dev, 2009, 126, 925–941. [Google Scholar] [CrossRef]
- Deboever, E.; Fistrovich, A.; Hulme, C.; Dunckley, T. The Omnipresence of DYRK1A in Human Diseases. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Dierssen, M.; de Lagrán, M.M. DYRK1A (dual-specificity tyrosine-phosphorylated and -regulated kinase 1A): A gene with dosage effect during development and neurogenesis. ScientificWorldJournal, 2006, 6, 1911–1922. [Google Scholar] [CrossRef]
- Duchon, A.; Herault, Y. DYRK1A, a Dosage-Sensitive Gene Involved in Neurodevelopmental Disorders, Is a Target for Drug Development in Down Syndrome. Front Behav Neurosci, 2016, 10, 104. [Google Scholar] [CrossRef]
- Exner, C.R.T.; Willsey, H.R. Xenopus leads the way: Frogs as a pioneering model to understand the human brain. Genesis, 2021, 59, e23405. [Google Scholar] [CrossRef]
- Farah, M.H.; Easter, S.S. ; Jr. Cell birth and death in the mouse retinal ganglion cell layer. J Comp Neurol, 2005, 489, 120–134. [Google Scholar]
- Farkas, L.G.; Katic, M.J.; Forrest, C.R. Surface anatomy of the face in Down's syndrome: Anthropometric proportion indices in the craniofacial regions. J Craniofac Surg, 2001, 12, 519–524, discussion 525–516. [Google Scholar] [CrossRef]
- Feki, A.; Hibaoui, Y. DYRK1A Protein, A Promising Therapeutic Target to Improve Cognitive Deficits in Down Syndrome. Brain Sci 2018, 8. [Google Scholar] [CrossRef]
- Fernandez-Martinez, P.; Zahonero, C.; Sanchez-Gomez, P. DYRK1A: The double-edged kinase as a protagonist in cell growth and tumorigenesis. Mol Cell Oncol, 2015, 2, e970048. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Martínez, P.; Zahonero, C.; Sánchez-Gómez, P. DYRK1A: The double-edged kinase as a protagonist in cell growth and tumorigenesis. Mol Cell Oncol, 2015, 2, e970048. [Google Scholar] [CrossRef] [PubMed]
- Firth, H.V.; Richards, S.M.; Bevan, A.P.; Clayton, S.; Corpas, M.; Rajan, D.; Van Vooren, S.; Moreau, Y.; Pettett, R.M.; Carter, N.P. DECIPHER: Database of Chromosomal Imbalance and Phenotype in Humans Using Ensembl Resources. American journal of human genetics, 2009, 84, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Fotaki, V.; Dierssen, M.; Alcántara, S.; Martínez, S.; Martí, E.; Casas, C.; Visa, J.; Soriano, E.; Estivill, X.; Arbonés, M.L. Dyrk1A haploinsufficiency affects viability and causes developmental delay and abnormal brain morphology in mice. Molecular and cellular biology, 2002, 22, 6636–6647. [Google Scholar] [CrossRef]
- García-Cerro, S.; Martínez, P.; Vidal, V.; Corrales, A.; Flórez, J.; Vidal, R.; Rueda, N.; Arbonés, M.L.; Martínez-Cué, C. Overexpression of Dyrk1A is implicated in several cognitive, electrophysiological and neuromorphological alterations found in a mouse model of Down syndrome. PLoS ONE, 2014, 9, e106572. [Google Scholar] [CrossRef]
- Goodlett, C.R.; Stringer, M.; LaCombe, J.; Patel, R.; Wallace, J.M.; Roper, R.J. Evaluation of the therapeutic potential of Epigallocatechin-3-gallate (EGCG) via oral gavage in young adult Down syndrome mice. Scientific reports, 2020, 10, 10426. [Google Scholar] [CrossRef]
- Guedj, F.; Pereira, P.L.; Najas, S.; Barallobre, M.J.; Chabert, C.; Souchet, B.; Sebrie, C.; Verney, C.; Herault, Y.; Arbones, M.; Delabar, J.M. DYRK1A: A master regulatory protein controlling brain growth. Neurobiol Dis, 2012, 46, 190–203. [Google Scholar] [CrossRef]
- Gurovich, Y.; Hanani, Y.; Bar, O.; Nadav, G.; Fleischer, N.; Gelbman, D.; Basel-Salmon, L.; Krawitz, P.M.; Kamphausen, S.B.; Zenker, M.; Bird, L.M.; Gripp, K.W. Identifying facial phenotypes of genetic disorders using deep learning. Nat Med, 2019, 25, 60–64. [Google Scholar] [CrossRef]
- Hämmerle, B.; Elizalde, C.; Tejedor, F.J. The spatio-temporal and subcellular expression of the candidate Down syndrome gene Mnb/Dyrk1A in the developing mouse brain suggests distinct sequential roles in neuronal development. Eur J Neurosci, 2008, 27, 1061–1074. [Google Scholar] [CrossRef] [PubMed]
- Hammerle, B.; Ulin, E.; Guimera, J.; Becker, W.; Guillemot, F.; Tejedor, F.J. Transient expression of Mnb/Dyrk1a couples cell cycle exit and differentiation of neuronal precursors by inducing p27KIP1 expression and suppressing NOTCH signaling. Development, 2011, 138, 2543–2554. [Google Scholar] [CrossRef] [PubMed]
- Hämmerle, B.; Vera-Samper, E.; Speicher, S.; Arencibia, R.; Martínez, S.; Tejedor, F.J. Mnb/Dyrk1A is transiently expressed and asymmetrically segregated in neural progenitor cells at the transition to neurogenic divisions. Dev Biol, 2002, 246, 259–273. [Google Scholar] [CrossRef]
- Hawley, L.E.; Stringer, M.; Deal, A.J.; Folz, A.; Goodlett, C.R.; Roper, R.J. Sex-specific developmental alterations in DYRK1A expression in the brain of a Down syndrome mouse model. Neurobiol Dis, 2024, 190, 106359. [Google Scholar] [CrossRef]
- Haworth, K.E.; Wilson, J.M.; Grevellec, A.; Cobourne, M.T.; Healy, C.; Helms, J.A.; Sharpe, P.T.; Tucker, A.S. Sonic hedgehog in the pharyngeal endoderm controls arch pattern via regulation of Fgf8 in head ectoderm. Dev Biol, 2007, 303, 244–258. [Google Scholar] [CrossRef] [PubMed]
- Herault, Y.; Delabar, J.M.; Fisher, E.M.C.; Tybulewicz, V.L.J.; Yu, E.; Brault, V. Rodent models in Down syndrome research: Impact and future opportunities. Dis Model Mech, 2017, 10, 1165–1186. [Google Scholar] [CrossRef]
- Hikasa, H.; Sokol, S.Y. Wnt signaling in vertebrate axis specification. Cold Spring Harb Perspect Biol, 2013, 5, a007955. [Google Scholar] [CrossRef]
- Hildebrandt, F.; Benzing, T.; Katsanis, N. Ciliopathies. N Engl J Med, 2011, 364, 1533–1543. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.Y.; Park, J.I.; Lee, M.; Munoz, W.A.; Miller, R.K.; Ji, H.; Gu, D.; Ezan, J.; Sokol, S.Y.; McCrea, P.D. Down's-syndrome-related kinase Dyrk1A modulates the p120-catenin-Kaiso trajectory of the Wnt signaling pathway. J Cell Sci, 2012, 125, 561–569. [Google Scholar] [CrossRef]
- Jamal, R.; LaCombe, J.; Patel, R.; Blackwell, M.; Thomas, J.R.; Sloan, K.; Wallace, J.M.; Roper, R.J. Increased dosage and treatment time of Epigallocatechin-3-gallate (EGCG) negatively affects skeletal parameters in normal mice and Down syndrome mouse models. PLoS ONE, 2022, 17, e0264254. [Google Scholar] [CrossRef]
- Ji, J.; Lee, H.; Argiropoulos, B.; Dorrani, N.; Mann, J.; Martinez-Agosto, J.A.; Gomez-Ospina, N.; Gallant, N.; Bernstein, J.A.; Hudgins, L.; Slattery, L.; Isidor, B.; Le Caignec, C.; David, A.; Obersztyn, E.; Wiśniowiecka-Kowalnik, B.; Fox, M.; Deignan, J.L.; Vilain, E.; Hendricks, E.; Horton Harr, M.; Noon, S.E.; Jackson, J.R.; Wilkens, A.; Mirzaa, G.; Salamon, N.; Abramson, J.; Zackai, E.H.; Krantz, I.; Innes, A.M.; Nelson, S.F.; Grody, W.W.; Quintero-Rivera, F. DYRK1A haploinsufficiency causes a new recognizable syndrome with microcephaly, intellectual disability, speech impairment, and distinct facies. Eur J Hum Genet, 2015, 23, 1473–1481. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, C.; Yu, T.; Zhang, L.; Meng, K.; Xing, Z.; Belichenko, P.V.; Kleschevnikov, A.M.; Pao, A.; Peresie, J.; Wie, S.; Mobley, W.C.; Yu, Y.E. Genetic dissection of the Down syndrome critical region. Human molecular genetics, 2015, 24, 6540–6551. [Google Scholar] [CrossRef]
- Johnson, A.F.; Nguyen, H.T.; Veitia, R.A. Causes and effects of haploinsufficiency. Biol Rev Camb Philos Soc, 2019, 94, 1774–1785. [Google Scholar] [CrossRef] [PubMed]
- Johnson, H.K.; Wahl, S.E.; Sesay, F.; Litovchick, L.; Dickinson, A.J. Dyrk1a is required for craniofacial development in Xenopus laevis. Dev Biol, 2024, 511, 63–75. [Google Scholar] [CrossRef]
- Kaczorowska, N.; Kaczorowski, K.; Laskowska, J.; Mikulewicz, M. Down syndrome as a cause of abnormalities in the craniofacial region: A systematic literature review. Adv Clin Exp Med, 2019, 28, 1587–1592. [Google Scholar] [CrossRef] [PubMed]
- Kay, L.J.; Smulders-Srinivasan, T.K.; Soundararajan, M. Understanding the Multifaceted Role of Human Down Syndrome Kinase DYRK1A. Adv Protein Chem Struct Biol, 2016, 105, 127–171. [Google Scholar] [PubMed]
- Kim, O.H.; Cho, H.J.; Han, E.; Hong, T.I.; Ariyasiri, K.; Choi, J.H.; Hwang, K.S.; Jeong, Y.M.; Yang, S.Y.; Yu, K.; Park, D.S.; Oh, H.W.; Davis, E.E.; Schwartz, C.E.; Lee, J.S.; Kim, H.G.; Kim, C.H. Zebrafish knockout of Down syndrome gene, DYRK1A, shows social impairments relevant to autism. Molecular autism, 2017, 8, 50. [Google Scholar] [CrossRef]
- Knight, R.D.; Schilling, T.F. Cranial neural crest and development of the head skeleton. Adv Exp Med Biol, 2006, 589, 120–133. [Google Scholar]
- Kofron, M.; Demel, T.; Xanthos, J.; Lohr, J.; Sun, B.; Sive, H.; Osada, S.; Wright, C.; Wylie, C.; Heasman, J. Mesoderm induction in Xenopus is a zygotic event regulated by maternal VegT via TGFbeta growth factors. Development, 1999, 126, 5759–5770. [Google Scholar] [CrossRef]
- Kumar, V.; Park, S.; Lee, U.; Kim, J. The Organizer and Its Signaling in Embryonic Development. J Dev Biol 2021, 9. [Google Scholar] [CrossRef]
- LaCombe, J.M.; Sloan, K.; Thomas, J.R.; Blackwell, M.P.; Crawford, I.; Bishop, F.; Wallace, J.M.; Roper, R.J. Sex-specific trisomic Dyrk1a-related skeletal phenotypes during development in a Down syndrome model. Dis Model Mech 2024, 17. [Google Scholar] [CrossRef]
- Laguna, A.; Aranda, S.; Barallobre, M.J.; Barhoum, R.; Fernández, E.; Fotaki, V.; Delabar, J.M.; de la Luna, S.; de la Villa, P.; Arbonés, M.L. The protein kinase DYRK1A regulates caspase-9-mediated apoptosis during retina development. Developmental cell, 2008, 15, 841–853. [Google Scholar] [CrossRef]
- Laguna, A.; Barallobre, M.J.; Marchena, M.; Mateus, C.; Ramírez, E.; Martínez-Cue, C.; Delabar, J.M.; Castelo-Branco, M.; de la Villa, P.; Arbonés, M.L. Triplication of DYRK1A causes retinal structural and functional alterations in Down syndrome. Human molecular genetics, 2013, 22, 2775–2784. [Google Scholar] [CrossRef]
- Lana-Elola, E.; Aoidi, R.; Llorian, M.; Gibbins, D.; Buechsenschuetz, C.; Bussi, C.; Flynn, H.; Gilmore, T.; Watson-Scales, S.; Haugsten Hansen, M.; Hayward, D.; Song, O.R.; Brault, V.; Herault, Y.; Deau, E.; Meijer, L.; Snijders, A.P.; Gutierrez, M.G.; Fisher, E.M.C.; Tybulewicz, V.L.J. Increased dosage of DYRK1A leads to congenital heart defects in a mouse model of Down syndrome. Sci Transl Med, 2024, 16, eadd6883. [Google Scholar] [CrossRef] [PubMed]
- Lana-Elola, E.; Watson-Scales, S.; Slender, A.; Gibbins, D.; Martineau, A.; Douglas, C.; Mohun, T.; Fisher, E.M.; Tybulewicz, V.L. Genetic dissection of Down syndrome-associated congenital heart defects using a new mouse mapping panel. eLife 2016, 5. [Google Scholar] [CrossRef]
- Lee, M.; Nagashima, K.; Yoon, J.; Sun, J.; Wang, Z.; Carpenter, C.; Lee, H.K.; Hwang, Y.S.; Westlake, C.J.; Daar, I.O. CEP97 phosphorylation by Dyrk1a is critical for centriole separation during multiciliogenesis. J Cell Biol 2022, 221. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Ha, J.; Kim, H.J.; Kim, Y.S.; Chang, E.J.; Song, W.J.; Kim, H.H. Negative feedback Inhibition of NFATc1 by DYRK1A regulates bone homeostasis. The Journal of biological chemistry, 2009, 284, 33343–33351. [Google Scholar] [CrossRef] [PubMed]
- Litovchick, L.; Florens, L.; Swanson, S.K.; Washburn, M.P.; DeCaprio, J.A. DYRK1A protein kinase promotes quiescence and senescence through DREAM complex assembly. Genes & development, 2011, 25, 801–813. [Google Scholar]
- Liu, Y.; Lin, Z.; Liu, M.; Wang, H.; Sun, H. Overexpression of DYRK1A, a Down Syndrome Candidate gene, Impairs Primordial Germ Cells Maintenance and Migration in zebrafish. Scientific reports, 2017, 7, 15313. [Google Scholar] [CrossRef]
- Liu, Y.; Lin, Z.; Peng, Y.; Jiang, Y.; Zhang, X.; Zhu, H.; Zhang, L.; Chen, J.; Shu, X.; Luo, M.; Xie, D.; Chen, Y.; Liao, H.; Liu, M.; Zhang, X.; Liu, S.; Wang, H.; Zhou, B.; Sun, H. Embryonic organizer formation disorder leads to multiorgan dysplasia in Down syndrome. Cell Death Dis, 2022, 13, 1054. [Google Scholar] [CrossRef]
- Locatelli, C.; Onnivello, S.; Gori, C.; Ramacieri, G.; Pulina, F.; Marcolin, C.; Vianello, R.; Vione, B.; Caracausi, M.; Pelleri, M.C.; Vitale, L.; Pirazzoli, G.L.; Cocchi, G.; Corvaglia, L.; Strippoli, P.; Antonaros, F.; Piovesan, A.; Lanfranchi, S. A reassessment of Jackson's checklist and identification of two Down syndrome sub-phenotypes. Scientific reports, 2022, 12, 3104. [Google Scholar] [CrossRef] [PubMed]
- Macho, V.; Coelho, A.; Areias, C.; Macedo, P.; Andrade, D. Craniofacial features and specific oral characteristics of Down syndrome children. Oral Health Dent Manag, 2014, 13, 408–411. [Google Scholar]
- Mahajan, K.; Mahajan, N.P. PI3K-independent AKT activation in cancers: A treasure trove for novel therapeutics. J Cell Physiol, 2012, 227, 3178–3184. [Google Scholar] [CrossRef]
- McCluskey, K.E.; Stovell, K.M.; Law, K.; Kostyanovskaya, E.; Schmidt, J.D.; Exner, C.R.T.; Dea, J.; Brimble, E.; State, M.W.; Willsey, A.J.; Willsey, H.R. Autism gene variants disrupt enteric neuron migration and cause gastrointestinal dysmotility. Nat Commun, 2025, 16, 2238. [Google Scholar] [CrossRef] [PubMed]
- McElyea, S.D.; Starbuck, J.M.; Tumbleson-Brink, D.M.; Harrington, E.; Blazek, J.D.; Ghoneima, A.; Kula, K.; Roper, R.J. Influence of prenatal EGCG treatment and Dyrk1a dosage reduction on craniofacial features associated with Down syndrome. Human molecular genetics, 2016, 25, 4856–4869. [Google Scholar]
- Meharena, H.S.; Marco, A.; Dileep, V.; Lockshin, E.R.; Akatsu, G.Y.; Mullahoo, J.; Watson, L.A.; Ko, T.; Guerin, L.N.; Abdurrob, F.; Rengarajan, S.; Papanastasiou, M.; Jaffe, J.D.; Tsai, L.H. Down-syndrome-induced senescence disrupts the nuclear architecture of neural progenitors. Cell Stem Cell, 2022, 29, 116–130.e117. [Google Scholar] [CrossRef] [PubMed]
- Mill, P.; Christensen, S.T.; Pedersen, L.B. Primary cilia as dynamic and diverse signalling hubs in development and disease. Nature reviews. Genetics, 2023, 24, 421–441. [Google Scholar] [CrossRef]
- Miyata, Y.; Nishida, E. Identification of FAM53C as a cytosolic-anchoring inhibitory binding protein of the kinase DYRK1A. Life Sci Alliance 2023, 6. [Google Scholar] [CrossRef]
- Moore, C.S.; Roper, R.J. The power of comparative and developmental studies for mouse models of Down syndrome. Mamm Genome, 2007, 18, 431–443. [Google Scholar] [CrossRef]
- Moore, R.; Alexandre, P. Delta-Notch Signaling: The Long and The Short of a Neuron's Influence on Progenitor Fates. J Dev Biol 2020, 8. [Google Scholar] [CrossRef]
- Murphy, A.J.; Wilton, S.D.; Aung-Htut, M.T.; McIntosh, C.S. Down syndrome and DYRK1A overexpression: Relationships and future therapeutic directions. Front Mol Neurosci, 2024, 17, 1391564. [Google Scholar] [CrossRef] [PubMed]
- Naert, G.; Ferré, V.; Meunier, J.; Keller, E.; Malmström, S.; Givalois, L.; Carreaux, F.; Bazureau, J.P.; Maurice, T. Leucettine L41, a DYRK1A-preferential DYRKs/CLKs inhibitor, prevents memory impairments and neurotoxicity induced by oligomeric Aβ25-35 peptide administration in mice. Eur Neuropsychopharmacol, 2015, 25, 2170–2182. [Google Scholar] [CrossRef]
- Nakano-Kobayashi, A.; Awaya, T.; Kii, I.; Sumida, Y.; Okuno, Y.; Yoshida, S.; Sumida, T.; Inoue, H.; Hosoya, T.; Hagiwara, M. Prenatal neurogenesis induction therapy normalizes brain structure and function in Down syndrome mice. Proc Natl Acad Sci U S A, 2017, 114, 10268–10273. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.L.; Duchon, A.; Manousopoulou, A.; Loaëc, N.; Villiers, B.; Pani, G.; Karatas, M.; Mechling, A.E.; Harsan, L.A.; Limanton, E.; Bazureau, J.P.; Carreaux, F.; Garbis, S.D.; Meijer, L.; Herault, Y. Correction of cognitive deficits in mouse models of Down syndrome by a pharmacological inhibitor of DYRK1A. Dis Model Mech 2018, 11. [Google Scholar] [CrossRef]
- Obara, K.; Abe, E.; Toyoshima, I. Whole-Exome Sequencing Identified a Novel DYRK1A Variant in a Patient With Intellectual Developmental Disorder, Autosomal Dominant 7. Cureus, 2023, 15, e33379. [Google Scholar] [CrossRef]
- Ogawa, Y.; Nonaka, Y.; Goto, T.; Ohnishi, E.; Hiramatsu, T.; Kii, I.; Yoshida, M.; Ikura, T.; Onogi, H.; Shibuya, H.; Hosoya, T.; Ito, N.; Hagiwara, M. Development of a novel selective inhibitor of the Down syndrome-related kinase Dyrk1A. Nat Commun, 2010, 1, 86. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Abalia, J.; Sahún, I.; Altafaj, X.; Andreu, N.; Estivill, X.; Dierssen, M.; Fillat, C. Targeting Dyrk1A with AAVshRNA attenuates motor alterations in TgDyrk1A, a mouse model of Down syndrome. American journal of human genetics, 2008, 83, 479–488. [Google Scholar] [CrossRef]
- Otte, E.D.; Roper, R.J. Skeletal health in DYRK1A syndrome. Front Neurosci, 2024, 18, 1462893. [Google Scholar] [CrossRef]
- Park, J.; Song, W.J.; Chung, K.C. Function and regulation of Dyrk1A: Towards understanding Down syndrome. Cell Mol Life Sci, 2009, 66, 3235–3240. [Google Scholar] [CrossRef]
- Peng, X.D.; Xu, P.Z.; Chen, M.L.; Hahn-Windgassen, A.; Skeen, J.; Jacobs, J.; Sundararajan, D.; Chen, W.S.; Crawford, S.E.; Coleman, K.G.; Hay, N. Dwarfism, impaired skin development, skeletal muscle atrophy, delayed bone development, and impeded adipogenesis in mice lacking Akt1 and Akt2. Genes & development, 2003, 17, 1352–1365. [Google Scholar]
- Rafii, M.S.; Kleschevnikov, A.M.; Sawa, M.; Mobley, W.C. Down syndrome. Handbook of clinical neurology, 2019, 167, 321–336. [Google Scholar] [PubMed]
- Raveau, M.; Shimohata, A.; Amano, K.; Miyamoto, H.; Yamakawa, K. DYRK1A-haploinsufficiency in mice causes autistic-like features and febrile seizures. Neurobiol Dis, 2018, 110, 180–191. [Google Scholar] [CrossRef]
- Raz, E. Primordial germ-cell development: The zebrafish perspective. Nature reviews. Genetics, 2003, 4, 690–700. [Google Scholar] [CrossRef]
- Redhead, Y.; Gibbins, D.; Lana-Elola, E.; Watson-Scales, S.; Dobson, L.; Krause, M.; Liu, K.J.; Fisher, E.M.C.; Green, J.B.A.; Tybulewicz, V.L.J. Craniofacial dysmorphology in Down syndrome is caused by increased dosage of Dyrk1a and at least three other genes. Development 2023, 150. [Google Scholar] [CrossRef]
- Reymond, A.; Marigo, V.; Yaylaoglu, M.B.; Leoni, A.; Ucla, C.; Scamuffa, N.; Caccioppoli, C.; Dermitzakis, E.T.; Lyle, R.; Banfi, S.; Eichele, G.; Antonarakis, S.E.; Ballabio, A. Human chromosome 21 gene expression atlas in the mouse. Nature, 2002, 420, 582–586. [Google Scholar] [CrossRef]
- Rice, A.M.; McLysaght, A. Dosage-sensitive genes in evolution and disease. BMC Biol 2017, 15, 78. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.M.; McLysaght, A. Dosage sensitivity is a major determinant of human copy number variant pathogenicity. Nat Commun 2017, 8, 14366. [Google Scholar] [CrossRef] [PubMed]
- Roese-Koerner, B.; Stappert, L.; Brüstle, O. Notch/Hes signaling and miR-9 engage in complex feedback interactions controlling neural progenitor cell proliferation and differentiation. Neurogenesis (Austin), 2017, 4, e1313647. [Google Scholar] [CrossRef]
- Roper, R.J.; Reeves, R.H. Understanding the basis for Down syndrome phenotypes. PLoS genetics, 2006, 2, e50. [Google Scholar] [CrossRef]
- Roper, R.J.; VanHorn, J.F.; Cain, C.C.; Reeves, R.H. A neural crest deficit in Down syndrome mice is associated with deficient mitotic response to Sonic hedgehog. Mechanisms of development, 2009, 126, 212–219. [Google Scholar] [CrossRef]
- Santagati, F.; Rijli, F.M. Cranial neural crest and the building of the vertebrate head. Nat Rev Neurosci, 2003, 4, 806–818. [Google Scholar] [CrossRef]
- Santos-Durán, G.N.; Barreiro-Iglesias, A. Roles of dual specificity tyrosine-phosphorylation-regulated kinase 2 in nervous system development and disease. Front Neurosci, 2022, 16, 994256. [Google Scholar] [CrossRef] [PubMed]
- Sebrié, C.; Chabert, C.; Ledru, A.; Guedj, F.; Po, C.; Smith, D.J.; Rubin, E.; Rivals, I.; Beloeil, J.C.; Gillet, B.; Delabar, J.M. Increased dosage of DYRK1A and brain volumetric alterations in a YAC model of partial trisomy 21. Anat Rec (Hoboken), 2008, 291, 254–262. [Google Scholar] [CrossRef]
- Sloan, K.; Thomas, J.; Blackwell, M.; Voisard, D.; Lana-Elola, E.; Watson-Scales, S.; Roper, D.L.; Wallace, J.M.; Fisher, E.M.C.; Tybulewicz, V.L.J.; Roper, R.J. Genetic dissection of triplicated chromosome 21 orthologs yields varying skeletal traits in Down syndrome model mice. Dis Model Mech 2023, 16. [Google Scholar] [CrossRef]
- Souchet, B.; Duchon, A.; Gu, Y.; Dairou, J.; Chevalier, C.; Daubigney, F.; Nalesso, V.; Créau, N.; Yu, Y.; Janel, N.; Herault, Y.; Delabar, J.M. Prenatal treatment with EGCG enriched green tea extract rescues GAD67 related developmental and cognitive defects in Down syndrome mouse models. Scientific reports, 2019, 9, 3914. [Google Scholar] [CrossRef]
- Starbuck, J.M.; Llambrich, S.; Gonzàlez, R.; Albaigès, J.; Sarlé, A.; Wouters, J.; González, A.; Sevillano, X.; Sharpe, J.; De La Torre, R.; Dierssen, M.; Vande Velde, G.; Martínez-Abadías, N. Green tea extracts containing epigallocatechin-3-gallate modulate facial development in Down syndrome. Scientific reports, 2021, 11, 4715. [Google Scholar] [CrossRef] [PubMed]
- Stringer, M.; Abeysekera, I.; Thomas, J.; LaCombe, J.; Stancombe, K.; Stewart, R.J.; Dria, K.J.; Wallace, J.M.; Goodlett, C.R.; Roper, R.J. Epigallocatechin-3-gallate (EGCG) consumption in the Ts65Dn model of Down syndrome fails to improve behavioral deficits and is detrimental to skeletal phenotypes. Physiol Behav 2017, 177, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Stringer, M.; Goodlett, C.R.; Roper, R.J. Targeting trisomic treatments: Optimizing Dyrk1a inhibition to improve Down syndrome deficits. Molecular genetics & genomic medicine 2017, 5, 451–465. [Google Scholar]
- Tran, K.T.; Le, V.S.; Bui, H.T.P.; Do, D.H.; Ly, H.T.T.; Nguyen, H.T.; Dao, L.T.M.; Nguyen, T.H.; Vu, D.M.; Ha, L.T.; Le, H.T.T.; Mukhopadhyay, A.; Nguyen, L.T. Genetic landscape of autism spectrum disorder in Vietnamese children. Scientific reports, 2020, 10, 5034. [Google Scholar] [CrossRef]
- Tripi, G.; Roux, S.; Matranga, D.; Maniscalco, L.; Glorioso, P.; Bonnet-Brilhault, F.; Roccella, M. Cranio-Facial Characteristics in Children with Autism Spectrum Disorders (ASD). J Clin Med 2019, 8. [Google Scholar] [CrossRef]
- van Bon BWM, C.B.; de Vries, BBA; et al. ; 2015 Dec 17 [Updated 2021 Mar 18]. DYRK1A Syndrome.; Adam, M.P., Mirzaa, G.M., Pagon, R.A., et al., Eds.; GeneReviews® [Internet]; University of Washington: Seattle, WA, USA, Seattle; 1993-2023. [Google Scholar]
- Vicente, A.; Bravo-González, L.A.; López-Romero, A.; Muñoz, C.S.; Sánchez-Meca, J. Craniofacial morphology in down syndrome: A systematic review and meta-analysis. Scientific reports, 2020, 10, 19895. [Google Scholar] [CrossRef] [PubMed]
- Vis, J.C.; Duffels, M.G.; Winter, M.M.; Weijerman, M.E.; Cobben, J.M.; Huisman, S.A.; Mulder, B.J. Down syndrome: A cardiovascular perspective. J Intellect Disabil Res, 2009, 53, 419–425. [Google Scholar] [CrossRef]
- Wegiel, J.; Gong, C.X.; Hwang, Y.W. The role of DYRK1A in neurodegenerative diseases. Febs j, 2011, 278, 236–245. [Google Scholar] [CrossRef]
- Werner, M.E.; Mitchell, B.J. Understanding ciliated epithelia: The power of Xenopus. Genesis, 2012, 50, 176–185. [Google Scholar] [CrossRef]
- Widowati, E.W.; Ernst, S.; Hausmann, R.; Müller-Newen, G.; Becker, W. Functional characterization of DYRK1A missense variants associated with a syndromic form of intellectual deficiency and autism. Biol Open 2018, 7. [Google Scholar] [CrossRef]
- Willsey, H.R.; Xu, Y.; Everitt, A.; Dea, J.; Exner, C.R.T.; Willsey, A.J.; State, M.W.; Harland, R.M. The neurodevelopmental disorder risk gene DYRK1A is required for ciliogenesis and control of brain size in Xenopus embryos. Development 2020, 147. [Google Scholar] [CrossRef]
- Wiseman, F.K.; Alford, K.A.; Tybulewicz, V.L.; Fisher, E.M. Down syndrome--recent progress and future prospects. Human molecular genetics, 2009, 18, R75–R83. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Iyyanar, P.P.R.; Lan, Y.; Jiang, R. Sonic hedgehog signaling in craniofacial development. Differentiation, 2023, 133, 60–76. [Google Scholar] [CrossRef] [PubMed]
- Yabut, O.; Domogauer, J.; D'Arcangelo, G. Dyrk1A overexpression inhibits proliferation and induces premature neuronal differentiation of neural progenitor cells. J Neurosci, 2010, 30, 4004–4014. [Google Scholar] [CrossRef]
- Yang, Y.; Fan, X.; Liu, Y.; Ye, D.; Liu, C.; Yang, H.; Su, Z.; Zhang, Y.; Liu, Y. Function and inhibition of DYRK1A: Emerging roles of treating multiple human diseases. Biochem Pharmacol, 2023, 212, 115521. [Google Scholar] [CrossRef]
- Yoshida, S.; Yoshida, K. New insights into the roles for DYRK family in mammalian development and congenital diseases. Genes Dis, 2023, 10, 758–770. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tian, Z.; Ye, S.; Mu, Q.; Wang, X.; Ren, S.; Hou, X.; Yu, W.; Guo, J. Changes in bone mineral density in Down syndrome individuals: A systematic review and meta-analysis. Osteoporos Int, 2022, 33, 27–37. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



