Submitted:
12 April 2025
Posted:
14 April 2025
Read the latest preprint version here
Abstract
Keywords:
Introduction
Materials and Methods
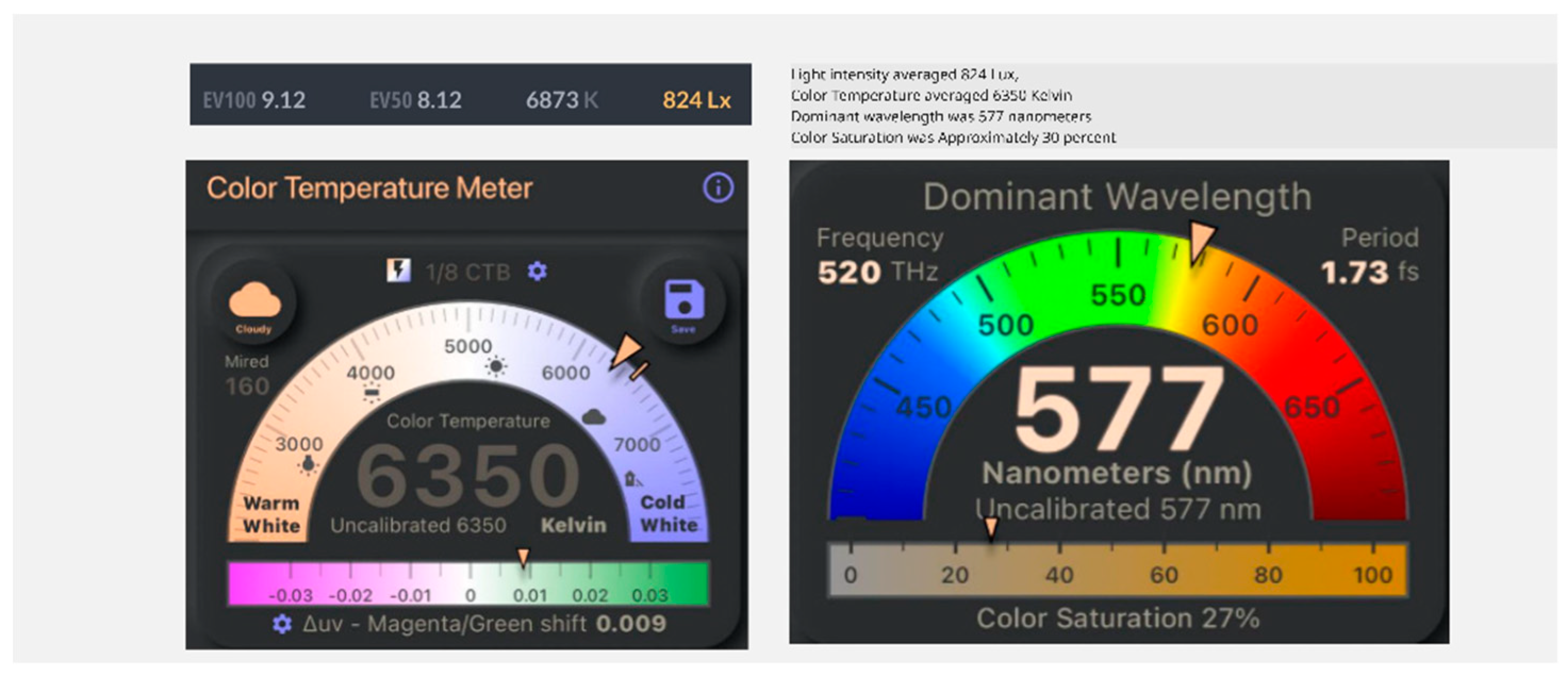
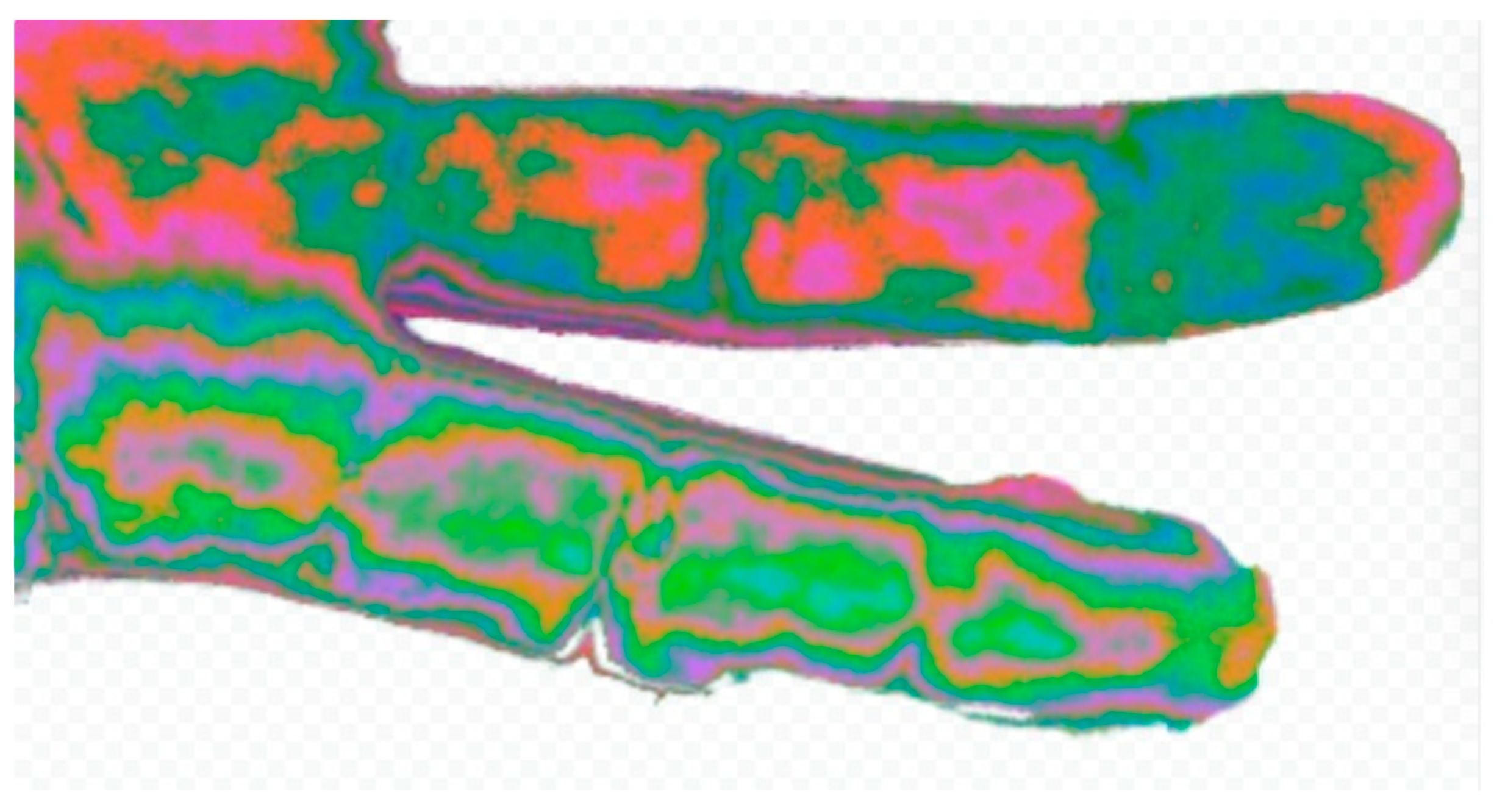
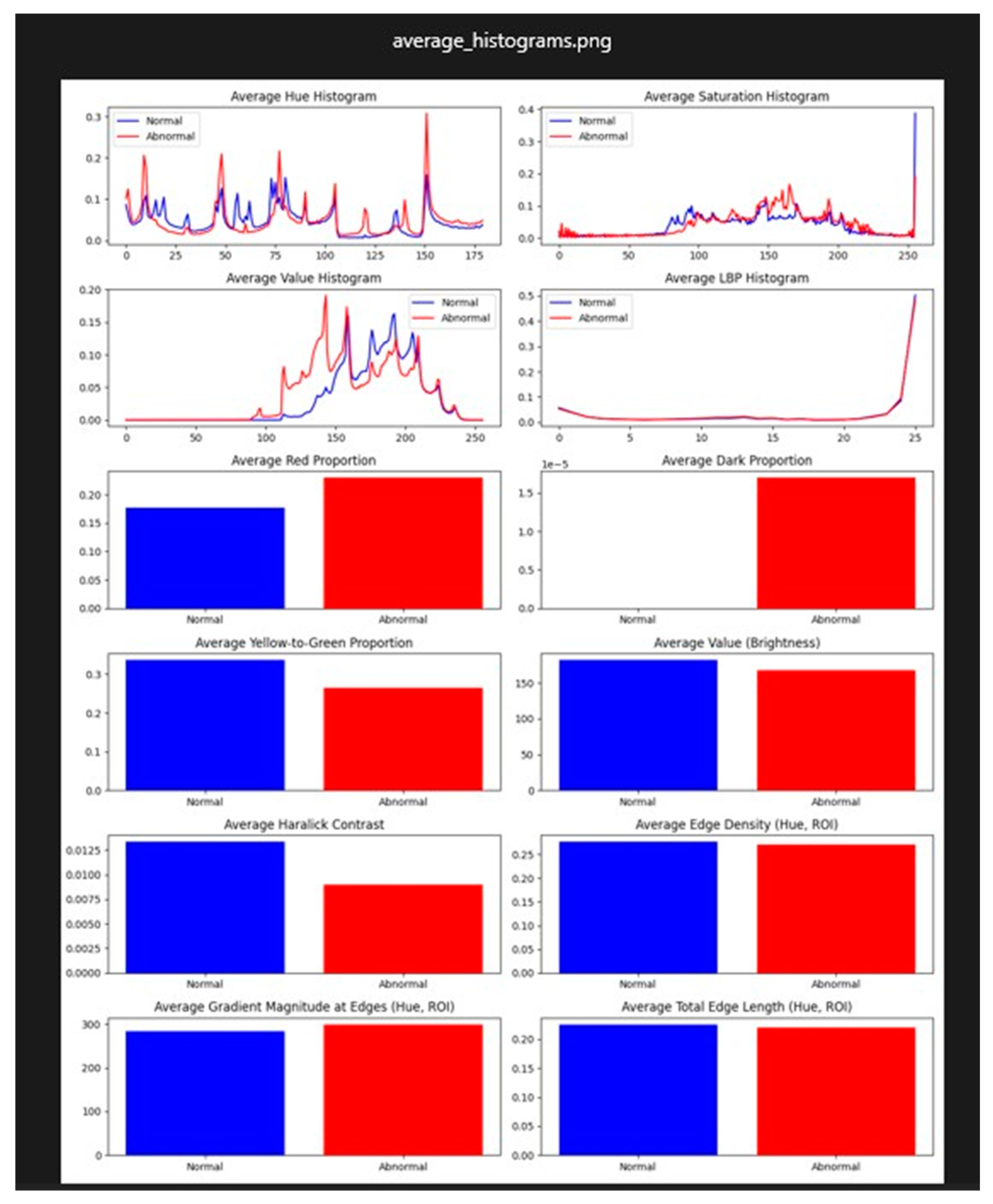
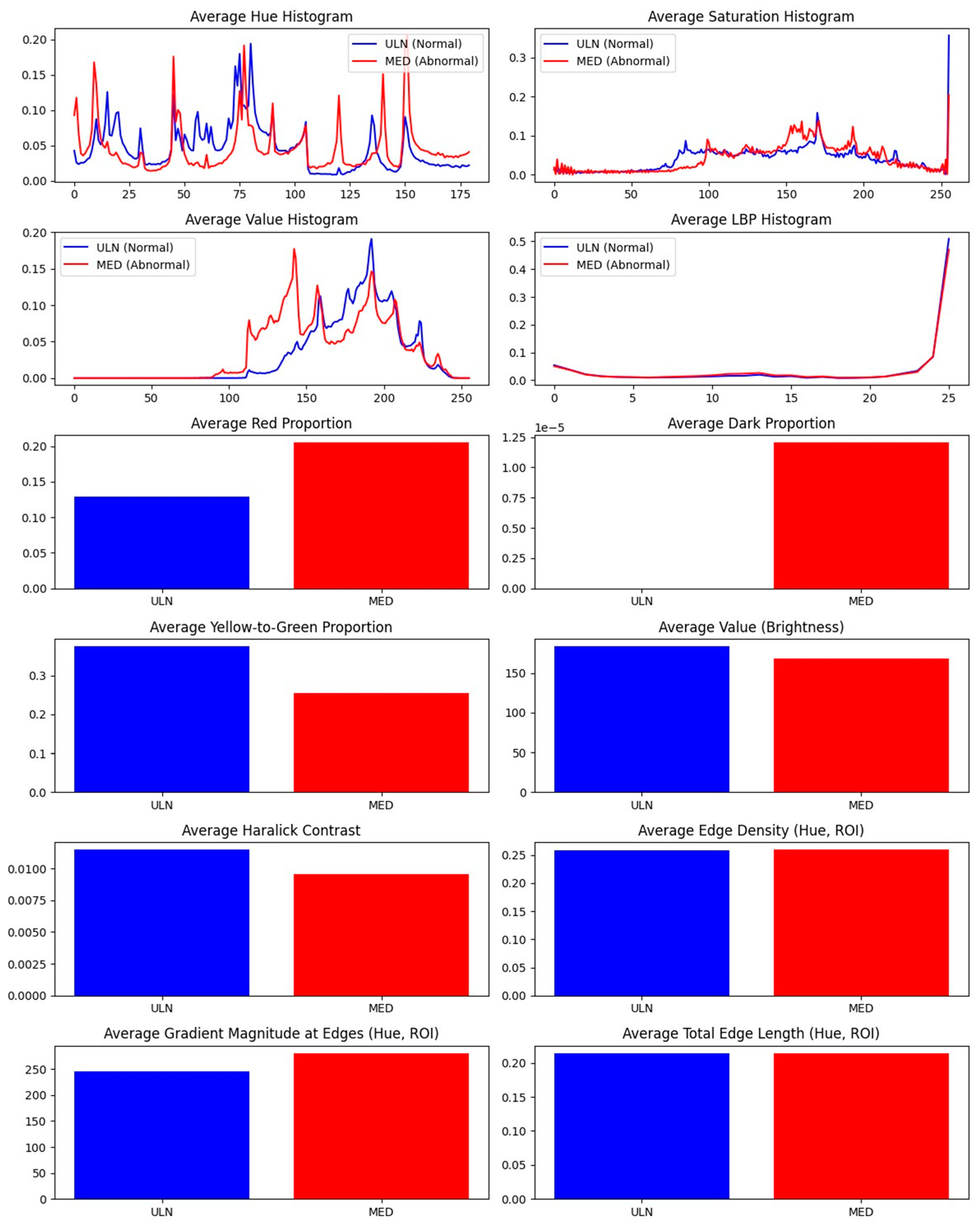
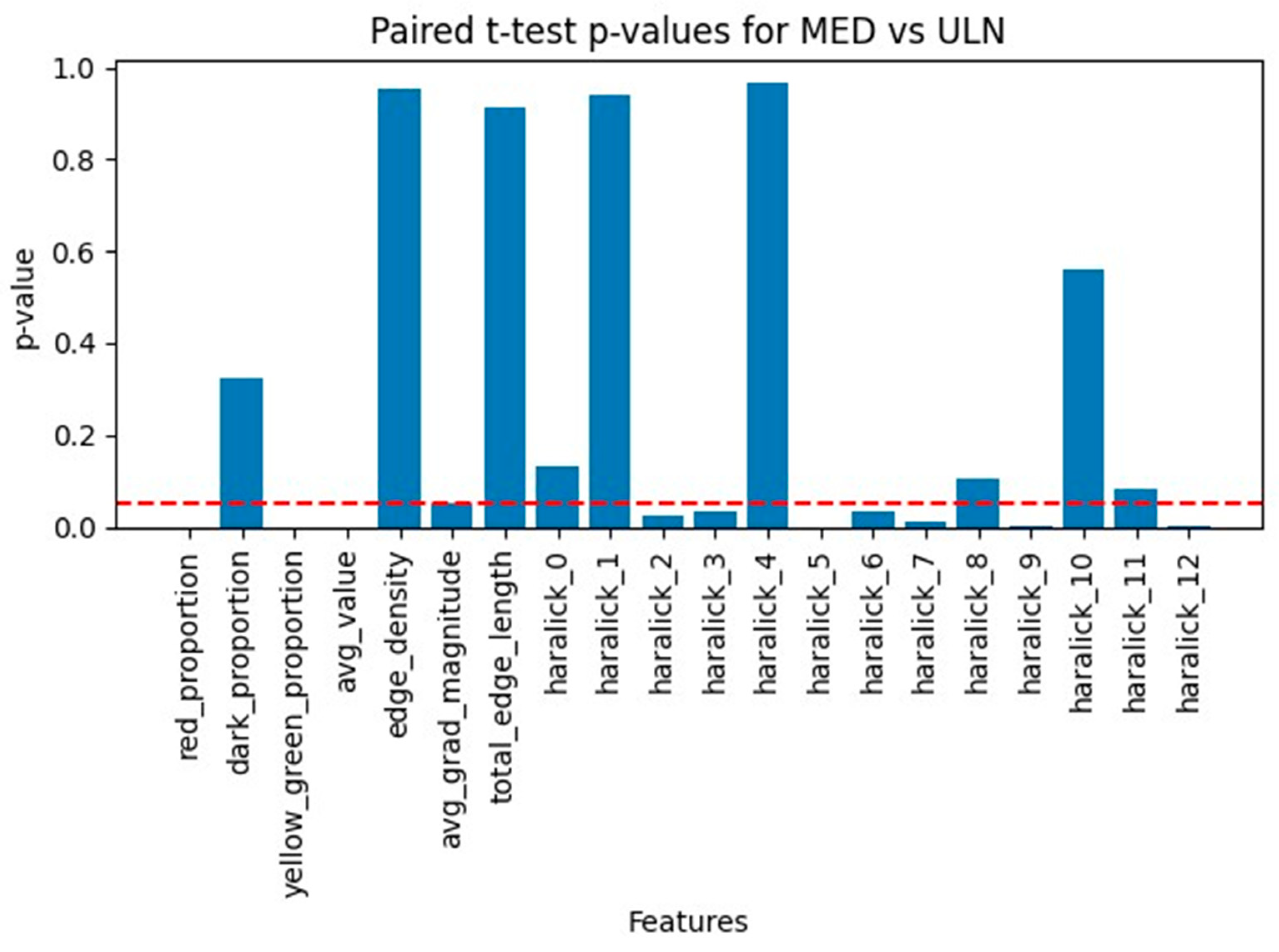
Results
Limitations and Recommendations
Discussion
References
- Kim, J.; Kim, M.W.; Kim, J.M. Enhanced diagnosis and severity assessment of carpal tunnel syndrome using combined shear wave elastography and cross-sectional area analysis: A prospective case-control study. PLoS One 2025, 20, e0320011. [Google Scholar] [CrossRef]
- Klauser, A.S.; Halpern, E.J.; De Zordo, T.; Feuchtner, G.M.; Arora, R.; Gruber, J.; Martinoli, C.; Loscher, W.N. Carpal tunnel syndrome assessment with US: value of additional cross-sectional area measurements of the median nerve in patients versus healthy volunteers. Radiology 2009, 250, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Yamaguchi, I. Magnetocardiograms in clinical medicine: unique information on cardiac ischemia, arrhythmias, and fetal diagnosis. Intern Med 2005, 44, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Bu, Y.; Prince, J.; Mojtahed, H.; Kimball, D.; Shah, V.; Coleman, T.; Sarkar, M.; Rao, R.; Huang, M.; Schwindt, P.; et al. Peripheral Nerve Magnetoneurography With Optically Pumped Magnetometers. Front Physiol 2022, 13, 798376. [Google Scholar] [CrossRef]
- Oldfield, H. New Energy Vision (NEV) for Windows PC. (accessed on. Available online: https://www.electrocrystal.com/nev_pc.html.
- Yang, A.; Cavanaugh, P.; Beredjiklian, P.K.; Matzon, J.L.; Seigerman, D.; Jones, C.M. Correlation of Carpal Tunnel Syndrome 6 Score and Physical Exam Maneuvers With Electrodiagnostic Test Severity in Carpal Tunnel Syndrome: A Blinded Prospective Cohort Study. J Hand Surg Am 2023, 48, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Zaki, H.A.; Shaban, E.; Salem, W.; Bilal, F.; Fayed, M.; Hendy, M.; Abdelrahim, M.G.; Masood, M.; Mohamed Khair, Y.; Shallik, N.A. A Comparative Analysis Between Ultrasound and Electromyographic and Nerve Conduction Studies in Diagnosing Carpal Tunnel Syndrome (CTS): A Systematic Review and Meta-Analysis. Cureus 2022, 14, e30476. [Google Scholar] [CrossRef]
- Dinescu, V.C.; Bica, M.; Vasile, R.C.; Gresita, A.; Catalin, B.; Rotaru-Zavaleanu, A.D.; Vreju, F.A.; Sas, L.; Bunescu, M. Limitations of the Boston Carpal Tunnel Questionnaire in Assessing Severity in a Homogeneous Occupational Cohort. Life (Basel) 2025, 15. [Google Scholar] [CrossRef]
- Merkel, D.; Lueders, C.; Schneider, C.; Yousefzada, M.; Ruppert, J.; Weimer, A.; Herzog, M.; Lorenz, L.A.; Vieth, T.; Buggenhagen, H.; et al. Prospective Comparison of Nine Different Handheld Ultrasound (HHUS) Devices by Ultrasound Experts with Regard to B-Scan Quality, Device Handling and Software in Abdominal Sonography. Diagnostics (Basel) 2024, 14. [Google Scholar] [CrossRef]
- Diagnosing and assessing CTS. (accessed on. Available online: https://www.carpal-tunnel.net/diagnosing.
- Wilson, K.E.; Tat, J.; Keir, P.J. Effects of Wrist Posture and Fingertip Force on Median Nerve Blood Flow Velocity. Biomed Res Int 2017, 2017, 7156489. [Google Scholar] [CrossRef]
- Shapiro, L.G.; Haralick, R.M. Relational matching. Appl Opt 1987, 26, 1845–1851. [Google Scholar] [CrossRef]
- Albuquerque, A.; Almeida, E.; Queiroz, F. A Comparative Study of Texture Analysis Methods on the Classification Problem of DPLDs in HRCT scans. Annu Int Conf IEEE Eng Med Biol Soc 2024, 2024, 1–4. [Google Scholar] [CrossRef]
- Ojala, T.; Punkki, J. Estimating the Workability of Concrete with a Stereovision Camera during Mixing. Sensors (Basel) 2024, 24. [Google Scholar] [CrossRef] [PubMed]
- Loke, K.S. A novel approach to texture recognition combining deep learning orthogonal convolution with regional input features. PeerJ Comput Sci 2024, 10, e1927. [Google Scholar] [CrossRef] [PubMed]
- Varoquaux, G.; Cheplygina, V. Machine learning for medical imaging: methodological failures and recommendations for the future. NPJ Digit Med 2022, 5, 48. [Google Scholar] [CrossRef]
- Mooshage, C.M.; Tsilingiris, D.; Schimpfle, L.; Fleming, T.; Herzig, S.; Szendroedi, J.; Heiland, S.; Bendszus, M.; Kopf, S.; Kurz, F.; et al. Intradermal Advanced Glycation End-products Relate to Reduced Sciatic Nerve Structural Integrity in Type 2 Diabetes. Clin Neuroradiol 2025. [Google Scholar] [CrossRef] [PubMed]
- Chikubu, H.; Inage, K.; Orita, S.; Shiga, Y.; Inoue, M.; Okuyama, K.; Tokeshi, S.; Shimizu, K.; Suzuki-Narita, M.; Ohtori, S. A Study on Subjective Symptoms and Plantar Temperature Imbalance in Lumbar Spinal Stenosis: A Preliminary Study. Cureus 2025, 17, e79388. [Google Scholar] [CrossRef]
- Prasad, A.; Mihacova, E.; Manoharan, R.R.; Pospisil, P. Application of ultra-weak photon emission imaging in plant stress assessment. J Plant Res 2025, 138, 389–400. [Google Scholar] [CrossRef]
- Casey, H.; DiBerardino, I.; Bonzanni, M.; Rouleau, N.; Murugan, N.J. Exploring ultraweak photon emissions as optical markers of brain activity. iScience 2025, 28, 112019. [Google Scholar] [CrossRef]
- Prasad, A.; Duchova, H.; Manoharan, R.R.; Rathi, D.; Pospisil, P. Imaging and Characterization of Oxidative Protein Modifications in Skin. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Poplova, M.; Prasad, A.; Van Wijk, E.; Pospisil, P.; Cifra, M. Biological Auto(chemi)luminescence Imaging of Oxidative Processes in Human Skin. Anal Chem 2023, 95, 14853–14860. [Google Scholar] [CrossRef]
- Shi, W.; Chen, L. An effective method for detecting the wheat freshness by integrating biophotonics and machine learning algorithm. Sci Rep 2024, 14, 32145. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, M.K.; Sefati, N.; Esmaeilpour, T.; Salari, V.; Oblak, D.; Simon, C. The impact of ketamine and thiopental anesthesia on ultraweak photon emission and oxidative-nitrosative stress in rat brains. Front Syst Neurosci 2025, 19, 1502589. [Google Scholar] [CrossRef] [PubMed]
- Herron, M.L.; Kar, S.; Beard, D.; Binfield, P. Sensory dysfunction in the great toe in hallux valgus. J Bone Joint Surg Br 2004, 86, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, B.F.R.; Faloppa, F.; Tamaoki, M.J.S.; Belloti, J.C. Effectiveness of ultrasonography and nerve conduction studies in the diagnosing of carpal tunnel syndrome: clinical trial on accuracy. BMC Musculoskelet Disord 2018, 19, 115. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, W.; Wang, T.; Qin, K.; Teng, J.; Qi, H. Comparison of ultrasound and magnetic resonance imaging of the median nerve's recurrent motor branch and the value of its diameter in diagnosing carpal tunnel syndrome. Quant Imaging Med Surg 2025, 15, 383–394. [Google Scholar] [CrossRef]
- Kefah, K.R., D. Multispectral Imaging for CTS Diagnosis. In Proceedings of the Israeli Orthopedic Association Tel Aviv; 2025. [Google Scholar]
- Geetha Devi, A.; Borra, S.P.R.; Rajesh Kumar, P. A new multimodal medical image fusion framework using Convolution Neural Networks. J Med Eng Technol 2025, 1–8. [Google Scholar] [CrossRef]
- Mazaheri, S.; Poorolajal, J.; Mazaheri, A. Sensitivity and specificity of electrodiagnostic parameters in diagnosing carpal tunnel syndrome. Bone Jt Open 2024, 5, 898–903. [Google Scholar] [CrossRef]
- Shi, X.; Yu, T.; Yuan, Y.; Wang, D.; Cui, J.; Bai, L.; Zheng, F.; Dai, X.; Zhou, Z. Multimodal Deep Learning for Grading Carpal Tunnel Syndrome: A Multicenter Study in China. Acad Radiol 2025. [Google Scholar] [CrossRef]
- Mercan, M.; Yayla, V. Utility of Median Nerve Electrophysiological Parameters in Differentiating Immune-Mediated Demyelination From Compressive Median Neuropathy at the Wrist. Muscle Nerve 2025. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




