Submitted:
08 April 2025
Posted:
09 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
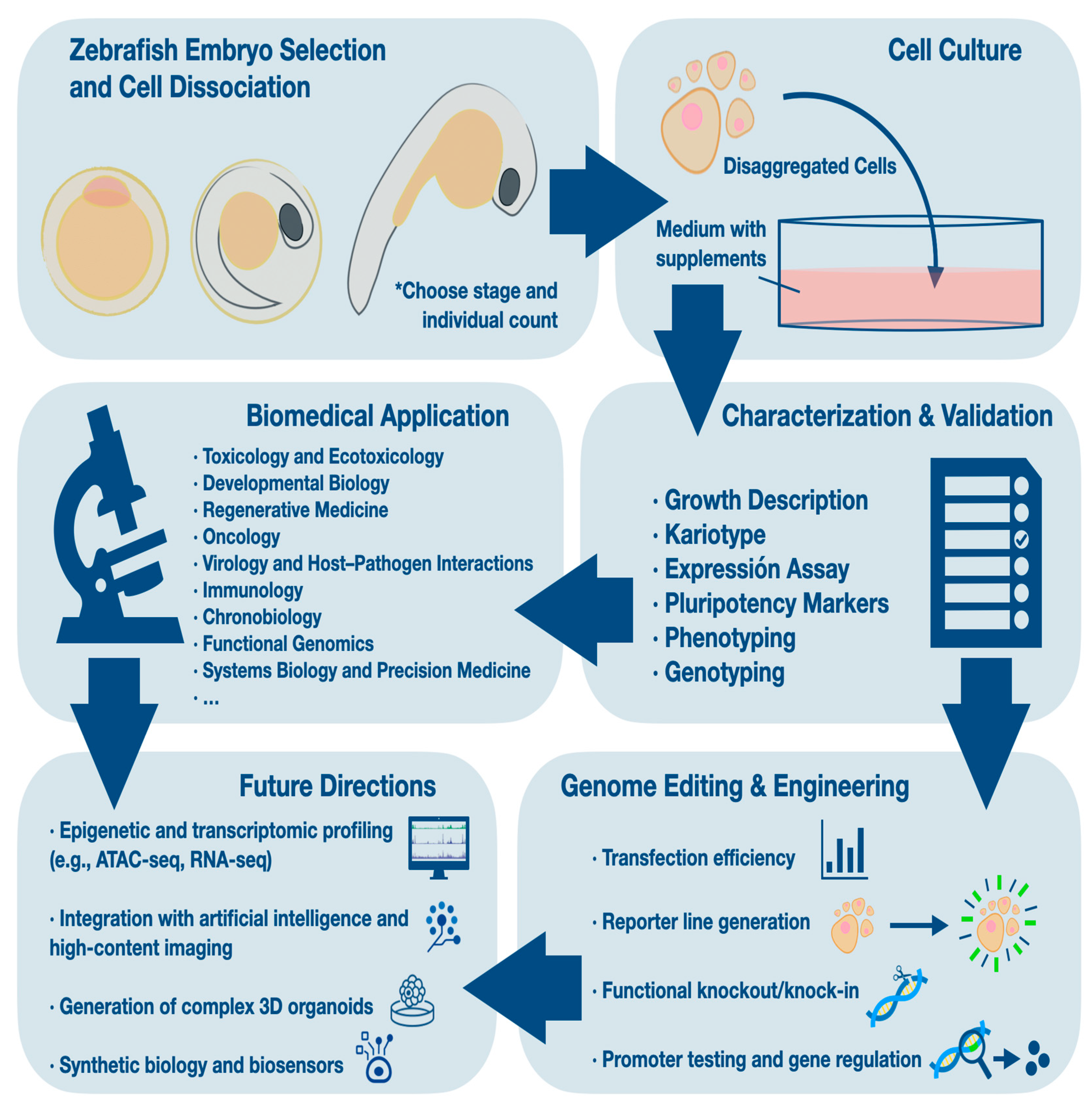
2. Overview of Zebrafish Embryo Cell Culture
3. Protocols for Establishing Cell Lines
4. Pluripotency in Zebrafish Cell Lines
5. Transfection Capabilities and Strategies
6. Applications of Zebrafish Cell Lines
7. Generation of Genotype-Defined Zebrafish Cell Lines from Individual Embryos
8. Efficiency and Challenges
9. Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| 3Rs | Replacement, Reduction, and Refinement |
| AP | Alkaline Phosphatase |
| ALP | Alkaline Phosphatase |
| ATAC-seq | Assay for Transposase-Accessible Chromatin using sequencing |
| ATCC | American Type Culture Collection |
| bFGF | Basic Fibroblast Growth Factor |
| CD24 | Cluster of Differentiation 24 |
| CMV | Cytomegalovirus (viral promoter) |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
| CRISPRa/i | CRISPR activation/interference |
| ctgfa | Connective Tissue Growth Factor a |
| CYP1A1 | Cytochrome P450 1A1 |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DMEM/F12 | DMEM supplemented with Ham’s F12 Nutrient Mix |
| DRCF | Danio rerio Caudal Fin cell line |
| ECACC | European Collection of Authenticated Cell Cultures |
| ESC | Embryonic Stem Cell |
| FBS | Fetal Bovine Serum |
| fli1 | Friend Leukemia Virus Integration 1 |
| G418 | Geneticin (aminoglycoside antibiotic) |
| gdf3 | Growth Differentiation Factor 3 |
| GFP | Green Fluorescent Protein |
| HEPES | 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (buffering agent) |
| hESC | Human Embryonic Stem Cell |
| hpf | Hours Post-Fertilization |
| kdr | Kinase Insert Domain Receptor (VEGFR2) |
| klf4 | Kruppel-like Factor 4 |
| KO | Knockout |
| L-15 | Leibovitz’s L-15 Medium |
| lin28 | LIN28 Homolog A |
| MPC | 2-methacryloxyloxyethyl phosphorylcholine |
| nanog | Homeobox protein Nanog |
| oct4 | Octamer-binding transcription factor 4 (pou5f1) |
| PAC2 | Zebrafish embryonic fibroblast cell line derived from 24 hpf embryos |
| PCR | Polymerase Chain Reaction |
| pEGFP-N1 | Plasmid expressing Enhanced Green Fluorescent Protein under CMV promoter |
| PGC | Primordial Germ Cell |
| pou5f1 | POU domain class 5 transcription factor 1 |
| ronin | Required for Nuclear Factor Activation in Stem Cells |
| RSV | Rous Sarcoma Virus |
| RTS34st | Rainbow Trout Spleen Stromal Cell Line |
| sall4 | Spalt Like Transcription Factor 4 |
| SDF-1b | Stromal Cell-Derived Factor 1 beta |
| sox2 | SRY-box Transcription Factor 2 |
| SSEA | Stage-Specific Embryonic Antigen |
| STR | Short Tandem Repeat |
| TCDD | 2,3,7,8-Tetrachlorodibenzo-p-dioxin |
| tcf3 | Transcription Factor 3 |
| tdgf1 | Teratocarcinoma-Derived Growth Factor 1 |
| Tol2 | Transposon originally from Oryzias latipes |
| TRA-1-60/81 | Tumor-Related Antigens |
| vasa | DEAD-box Helicase 4 (germ cell marker) |
| zPDX | Zebrafish Patient-Derived Xenograft |
| Z428 | Zebrafish embryonic stem-like cell line |
| ZBE3 | Zebrafish Blastula-derived Embryonic cell line with high transfection efficiency |
| ZEB2J | Zebrafish Embryonic Blastula-derived cell line adapted from co-culture |
| ZEM-2 ZEM-2A ZEM2S |
Zebrafish Embryonic cell lines with varying serum/media conditions |
| ZES1 | Zebrafish embryonic stem-like cell line |
| ZF4 | Zebrafish Fibroblast cell line |
| ZFL | Zebrafish Liver cell line |
References
- Bradford, C.S.; Sun, L.; Collodi, P.; Barnes, D.W. Cell Cultures from Zebrafish Embryos and Adult Tissues. J. Tissue Cult. Methods 1994, 16, 99–107. [Google Scholar] [CrossRef]
- Collodi, P.; Kame, Y.; Ernst, T.; Miranda, C.; Buhler, D.R.; Barnes, D.W. Culture of Cells from Zebrafish (Brachydanio Rerio) Embryo and Adult Tissues. Cell Biol. Toxicol. 1992, 8, 43–61. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The Zebrafish Reference Genome Sequence and Its Relationship to the Human Genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef]
- Ye, X.; Lin, J.; Chen, Q.; Lv, J.; Liu, C.; Wang, Y.; Wang, S.; Wen, X.; Lin, F. An Efficient Vector-Based CRISPR/Cas9 System in Zebrafish Cell Line. Mar. Biotechnol. 2024, 26, 588–598. [Google Scholar] [CrossRef]
- Driever, W.; Rangini, Z. Characterization of a Cell Line Derived from Zebrafish (Brachydanio Rerio) Embryos. Vitro Cell. Dev. Biol. - Anim. 1993, 29, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, C.; Zhou, Y.L.; Collodi, P. Derivation and Characterization of a Zebrafish Liver Cell Line. Cell Biol. Toxicol. 1994, 10, 167–176. [Google Scholar] [CrossRef]
- Ghosh, C.; Liu, Y.; Ma, C.; Collodi, P. Cell Cultures Derived from Early Zebrafish Embryos Differentiate in Vitro into Neurons and Astrocytes.
- Fan, L.; Moon, J.; Wong, T.-T.; Crodian, J.; Collodi, P. Zebrafish Primordial Germ Cell Cultures Derived from Vasa::RFP Transgenic Embryos. Stem Cells Dev. 2008, 17, 585–598. [Google Scholar] [CrossRef]
- Ciarlo, C.A.; Zon, L.I. Embryonic Cell Culture in Zebrafish. In Methods in Cell Biology; Elsevier, 2016; Vol. 133, pp. 1–10 ISBN 978-0-12-803475-0.
- Ho, S.Y.; Goh, C.W.P.; Gan, J.Y.; Lee, Y.S.; Lam, M.K.K.; Hong, N.; Hong, Y.; Chan, W.K.; Shu-Chien, A.C. Derivation and Long-Term Culture of an Embryonic Stem Cell-Like Line from Zebrafish Blastomeres Under Feeder-Free Condition. Zebrafish 2014, 11, 407–420. [Google Scholar] [CrossRef]
- Hong, N.; Schartl, M.; Hong, Y. Derivation of Stable Zebrafish ES-like Cells in Feeder-Free Culture. Cell Tissue Res. 2014, 357, 623–632. [Google Scholar] [CrossRef]
- Badakov, R.; Jaźwińska, A. Efficient Transfection of Primary Zebrafish Fibroblasts by Nucleofection. Cytotechnology 2006, 51, 105–110. [Google Scholar] [CrossRef]
- Geyer, N.; Kaminsky, S.; Confino, S.; Livne, Z.B.-M.; Gothilf, Y.; Foulkes, N.S.; Vallone, D. Establishment of Cell Lines from Individual Zebrafish Embryos. Lab. Anim. 2023, 57, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Mizgirev, I.; Revskoy, S. Generation of Clonal Zebrafish Lines and Transplantable Hepatic Tumors. Nat. Protoc. 2010, 5, 383–394. [Google Scholar] [CrossRef]
- De Souza, I.R.; Micali Canavez, A.D.P.; Schuck, D.C.; Costa Gagosian, V.S.; De Souza, I.R.; De Albuquerque Vita, N.; Da Silva Trindade, E.; Cestari, M.M.; Lorencini, M.; Leme, D.M. A 3D Culture Method of Spheroids of Embryonic and Liver Zebrafish Cell Lines. J. Vis. Exp. 2023, 64859. [Google Scholar] [CrossRef] [PubMed]
- Kamei, Y.; Collodi, P.; Ernst, T.; Barnes, D.W. CULTURE OF CELLS FROM ADULT ZEBRAFISH (BRACHYDANIO RERIO) AND EMBRYO. In Animal Cell Technology; Elsevier, 1992; pp. 17–19 ISBN 978-0-7506-0421-5.
- Meena, L.L.; Goswami, M.; Chaudhari, A.; Nagpure, N.S.; Gireesh-Babu, P.; Dubey, A.; Das, D.K. Development and Characterization of a New DRCF Cell Line from Indian Wild Strain Zebrafish Danio Rerio (Hamilton 1822). Fish Physiol. Biochem. 2020, 46, 1337–1347. [Google Scholar] [CrossRef]
- Vallone, D.; Santoriello, C.; Gondi, S.B.; Foulkes, N.S. Basic Protocols for Zebrafish Cell Lines.
- Jin, Y.L.; Chen, L.M.; Le, Y.; Li, Y.L.; Hong, Y.H.; Jia, K.T.; Yi, M.S. Establishment of a Cell Line with High Transfection Efficiency from Zebrafish Danio Rerio Embryos and Its Susceptibility to Fish Viruses. J. Fish Biol. 2017, 91, 1018–1031. [Google Scholar] [CrossRef] [PubMed]
- Choorapoikayil, S.; Overvoorde, J.; Den Hertog, J. Deriving Cell Lines from Zebrafish Embryos and Tumors. Zebrafish 2013, 10, 316–325. [Google Scholar] [CrossRef]
- Xing, J.G.; Lee, L.E.J.; Fan, L.; Collodi, P.; Holt, S.E.; Bols, N.C. Initiation of a Zebrafish Blastula Cell Line on Rainbow Trout Stromal Cells and Subsequent Development Under Feeder-Free Conditions into a Cell Line, ZEB2J. Zebrafish 2008, 5, 49–63. [Google Scholar] [CrossRef]
- Ma, C.; Fan, L.; Ganassin, R.; Bols, N.; Collodi, P. Production of Zebrafish Germ-Line Chimeras from Embryo Cell Cultures. Proc. Natl. Acad. Sci. 2001, 98, 2461–2466. [Google Scholar] [CrossRef]
- Fan, L.; Crodian, J.; Collodi, P. Production of Zebrafish Germline Chimeras by Using Cultured Embryonic Stem (ES) Cells. In Methods in Cell Biology; Elsevier, 2004; Vol. 77, pp. 113–119 ISBN 978-0-12-564172-2.
- Senghaas, N.; Köster, R.W. Culturing and Transfecting Zebrafish PAC2 Fibroblast Cells. Cold Spring Harb. Protoc. 2009, 2009, pdb.prot5235. [Google Scholar] [CrossRef]
- He, S.; Salas-Vidal, E.; Rueb, S.; Krens, S.F.G.; Meijer, A.H.; Snaar-Jagalska, B.E.; Spaink, H.P. Genetic and Transcriptome Characterization of Model Zebrafish Cell Lines. Zebrafish 2006, 3, 441–453. [Google Scholar] [CrossRef]
- Culp, P.; Nüsslein-Volhard, C.; Hopkins, N. High-Frequency Germ-Line Transmission of Plasmid DNA Sequences Injected into Fertilized Zebrafish Eggs. Proc. Natl. Acad. Sci. 1991, 88, 7953–7957. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, C.; Collodi, P. Culture of Cells from Zebrafish (Brachydanio Rerio) Blastula-Stage Embryos. Cytotechnology 1994, 14, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xu, W.; Xiang, S.; Tao, L.; Fu, W.; Liu, J.; Liu, W.; Xiao, Y.; Peng, L. Defining the Pluripotent Marker Genes for Identification of Teleost Fish Cell Pluripotency During Reprogramming. Front. Genet. 2022, 13, 819682. [Google Scholar] [CrossRef]
- Robles, V.; Martí, M.; Belmonte, J.C.I. Study of Pluripotency Markers in Zebrafish Embryos and Transient Embryonic Stem Cell Cultures. Zebrafish 2011, 8, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.-M.; Gerbal-Chaloin, S.; Milhavet, O.; Qiang, B.; Becker, F.; Assou, S.; Lemaître, J.-M.; Hamamah, S.; De Vos, J. Brief Report: Benchmarking Human Pluripotent Stem Cell Markers During Differentiation Into the Three Germ Layers Unveils a Striking Heterogeneity: All Markers Are Not Equal. Stem Cells 2011, 29, 1469–1474. [Google Scholar] [CrossRef]
- Xiao, Y.; Gao, M.; Gao, L.; Zhao, Y.; Hong, Q.; Li, Z.; Yao, J.; Cheng, H.; Zhou, R. Directed Differentiation of Zebrafish Pluripotent Embryonic Cells to Functional Cardiomyocytes. Stem Cell Rep. 2016, 7, 370–382. [Google Scholar] [CrossRef]
- De Los Angeles, A.; Ferrari, F.; Xi, R.; Fujiwara, Y.; Benvenisty, N.; Deng, H.; Hochedlinger, K.; Jaenisch, R.; Lee, S.; Leitch, H.G.; et al. Hallmarks of Pluripotency. Nature 2015, 525, 469–478. [Google Scholar] [CrossRef]
- Mihailovic, S.; Wolff, S.C.; Kedziora, K.M.; Smiddy, N.M.; Redick, M.A.; Wang, Y.; Lin, G.K.; Zikry, T.M.; Simon, J.; Ptacek, T.; et al. Single-Cell Dynamics of Core Pluripotency Factors in Human Pluripotent Stem Cells 2022.
- Bertero, A.; Madrigal, P.; Galli, A.; Hubner, N.C.; Moreno, I.; Burks, D.; Brown, S.; Pedersen, R.A.; Gaffney, D.; Mendjan, S.; et al. Activin/Nodal Signaling and NANOG Orchestrate Human Embryonic Stem Cell Fate Decisions by Controlling the H3K4me3 Chromatin Mark. Genes Dev. 2015, 29, 702–717. [Google Scholar] [CrossRef]
- Hong, Y.; Schartl, M. Isolation and Differentiation of Medaka Embryonic Stem Cells. In Embryonic Stem Cell Protocols; Humana Press: New Jersey, 2006; Volume 329, pp. 3–16. ISBN 978-1-59745-037-9. [Google Scholar]
- Wang, D.; Manali, D.; Wang, T.; Bhat, N.; Hong, N.; Li, Z.; Wang, L.; Yan, Y.; Liu, R.; Hong, Y. Identification of Pluripotency Genes in the Fish Medaka. Int. J. Biol. Sci. 2011, 7, 440–451. [Google Scholar] [CrossRef]
- Chen, S. -L.; Ye, H. -Q.; Sha, Z. -X.; Hong, Y. Derivation of a Pluripotent Embryonic Cell Line from Red Sea Bream Blastulas. J. Fish Biol. 2003, 63, 795–805. [Google Scholar] [CrossRef]
- Sánchez-Sánchez, A.V.; Camp, E.; Mullor, J.L. Fishing Pluripotency Mechanisms In Vivo. Int. J. Biol. Sci. 2011, 7, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Putri, R.R.; Ayisi, C.L. Transfection Of Difficult-To-Transfect Zebrafish (Danio Rerio) ZF4 Cells Using Chemical Transfection and Nucleofection Method. Juv. Ilm. Kelaut. Dan Perikan. 2023, 4, 104–108. [Google Scholar] [CrossRef]
- Kikuta, H.; Kawakami, K. Transient and Stable Transgenesis Using Tol2 Transposon Vectors. In Zebrafish; Lieschke, G.J., Oates, A.C., Kawakami, K., Eds.; Methods in Molecular Biology; Humana Press: Totowa, NJ, 2009; Volume 546, pp. 69–84. ISBN 978-1-60327-976-5. [Google Scholar]
- Lungu-Mitea, S.; Lundqvist, J. Potentials and Pitfalls of Transient in Vitro Reporter Bioassays: Interference by Vector Geometry and Cytotoxicity in Recombinant Zebrafish Cell Lines. Arch. Toxicol. 2020, 94, 2769–2784. [Google Scholar] [CrossRef]
- Di Renzo, M.F.; Corso, S. Patient-Derived Cancer Models. Cancers 2020, 12, 3779. [Google Scholar] [CrossRef] [PubMed]
- Park, C.G.; Ryu, C.S.; Sung, B.; Manz, A.; Kong, H.; Kim, Y.J. Transcriptomic and Physiological Analysis of Endocrine Disrupting Chemicals Impacts on 3D Zebrafish Liver Cell Culture System. Aquat. Toxicol. 2022, 245, 106105. [Google Scholar] [CrossRef]
- Yang, H.; Luan, Y.; Liu, T.; Lee, H.J.; Fang, L.; Wang, Y.; Wang, X.; Zhang, B.; Jin, Q.; Ang, K.C.; et al. A Map of Cis-Regulatory Elements and 3D Genome Structures in Zebrafish. Nature 2020, 588, 337–343. [Google Scholar] [CrossRef]
- Petratou, K.; Stehling, M.; Müller, F.; Schulte-Merker, S. Integration of ATAC and RNA-Sequencing Identifies Chromatin and Transcriptomic Signatures in Classical and Non-Classical Zebrafish Osteoblasts and Indicates Mechanisms of Entpd5a Regulation 2025.
- Quillien, A.; Abdalla, M.; Yu, J.; Ou, J.; Zhu, L.J.; Lawson, N.D. Robust Identification of Developmentally Active Endothelial Enhancers in Zebrafish Using FANS-Assisted ATAC-Seq. Cell Rep. 2017, 20, 709–720. [Google Scholar] [CrossRef]
| Cell Line | Source | Embryos Used | Culture Medium | Karyotype | Transfection | Application | Ref |
|---|---|---|---|---|---|---|---|
| DRCF | Adult fin-derived | Adult tissue | L-15 + 20% FBS | — | Standard reagents | Transfection studies; fibroblastic morphology | [17] |
| PAC2 | 24 hpf embryos | ~50 embryos per culture | L-15 + 15% FBS | — | FuGENE HD, Nanofectin; luciferase stable | Circadian & CRISPR studies; light-entrainable reporter | [18,24,25,26] |
| PAC-2 luc reporter | Transfected PAC-2 | 24 hpf; number not specified | L-15 + 15% FBS + G-418 (stable clones) | — | Luciferase (stable >20 d) | Circadian studies; light-inducible gene expression | [18] |
| PTEN-KO | Single ptenb−/− embryo | 1 | DMEM + 10% FBS | — | Standard methods | Tumor and migration modeling | [20] |
| PTEN-mutant tumor | ptena+/− ptenb−/− tumor (adult) | Adult tissue | L-15 + supplements; room temp | — | — | Endothelial markers; tumor and vascular studies | [20] |
| Z428 | Blastula-stage embryos | 200 | DMEM + 10% FBS + bFGF (feeder-free) | Diploid (48 chr) | High efficiency; stable GFP | Pluripotency; 3 germ layers | [11] |
| ZBE3 | Blastula-stage embryos | Not specified | DMEM + 10% FBS | — | High efficiency | Virus-host interactions; immune studies | [19] |
| ZEB2J | Blastula-stage; from ZEB2 | Not specified | DMEM/F12 + 15% FBS (initially with RTS34st) | Heteroploid | — | ES-like; adherent; Pou-2 expression | [21] |
| ZEF1/ZEF2 | 5–10 somite embryos | Not specified | L-15 + 10% FBS + antibiotics | — | Nucleofection (up to 43% GFP+) | Fibroblast cultures; high nucleofection | [12] |
| ZEM-2 | Blastula-stage embryos | 10 embryos/well (batch) | Trout extract + FBS (complex medium) | Aneuploid (modal = 73) | CMV, SV40, RSV active | Neural & early development studies | [2,7] |
| ZEM-2A | Derived from ZEM-2 | Derived from ZEM-2 | 5% FBS (simplified medium) | Aneuploid (modal = 77) | CMV, SV40, RSV active | Transfection studies; simplified conditions | [27] |
| ZEM2S | Selected from ZEM-2 | Derived from ZEM-2 | DMEM/L-15 (no trout extract) | — | — | Cytotoxicity; ecotoxicology | [15,25,26] |
| ZES1 | Blastula-stage embryos | 200 | DMEM + 10% FBS + bFGF (feeder-free) | Diploid; stable | FuGENE HD; stable GFP | Pluripotency; multi-lineage differentiation | [10] |
| ZF4 | 24 hpf embryos | 50 per batch | DMEM/F12 + 10% FBS | Hyperploid (~120) | Efficient; supports CMV, RSV, SV40 | Gene regulation; wound healing; cancer research | [5,25] |
| ZFL | Adult liver | Derived from adult liver | L-15 + 15% FBS + HEPES | — | CYP1A1 inducible (TCDD) | Cytotoxicity; ecotoxicology | [15] |
| Feature | Zebrafish ES-like Lines | Human ESCs | Ref |
|---|---|---|---|
| Key transcription factors | pou5f1,nanog,sox2,lin28 | OCT4, SOX2, NANOG | [10,11] |
| Enzymatic marker | Alkaline phosphatase (AP) | Alkaline phosphatase (AP) | [10,30] |
| Surface markers | SSEA1 | SSEA3, SSEA4, TRA-1-60, TRA-1-81, CD24 | [29] |
| Unique markers (species-specific) | tdgf1, gdf3 | — | [28] |
| Functional pluripotency | Embryoid body formation; contribution to germ layers | Embryoid bodies; teratoma formation; chimera | [22,23] |
| Directed differentiation | Neuronal, hepatic, cardiac lineages (e.g., Z428) | Neuronal, cardiac, endodermal, etc. | [7,31] |
| Pluripotency state | Primed-like | Naïve and primed | [29,32] |
| Sox2 function | Neural specification | Pluripotency maintenance | [33] |
| Epigenetic regulation | Less characterized; prone to drift | Defined enhancer usage (oct4, nanog loci) | [34] |
| Method | Cell Line | Cargo | Efficiency Type | Efficiency (%) | Ref |
|---|---|---|---|---|---|
| Nucleofection | ZF4 | pmaxGFP plasmid | Transfection | 50–75 | [39] |
| Nucleofection | PAC2 | pmaxGFP plasmid | Transfection | 40–50 | [25] |
| Lipofection | ZF4 | pmaxGFP plasmid | Transfection | 3–5 | [39] |
| Lipofection | PAC2 | GFP plasmid | Transfection | Moderate (qual.) | [24] |
| Lipofection | PAC2 | Tol2 construct + transposase | Transfection | Stable integration (qual.) | [40,41] |
| Lipofection | DRCF | pEGFP-N1 | Transfection | Not reported (GFP positive) | [17] |
| Lentivirus | PAC2 | Cas9 + sgRNA vector | Editing | Not reported (functional knockout confirmed) | [4] |
| FuGENE 6 | ZF4 | GFP plasmid | Transfection | 15–20 | [25] |
| FuGENE 6 | PAC2 | GFP plasmid | Transfection | ~5 | [25] |
| Quality Control Parameter | Method | Frequency |
|---|---|---|
| Mycoplasma contamination | PCR-based or luminescent assay | Monthly |
| Chromosomal stability | Karyotype analysis (Giemsa or DAPI) | Every 10 passages |
| Genetic identity | STR or gene-specific PCR profiling | At establishment and yearly |
| Phenotypic consistency | Morphology, doubling time | Each passage |
| Viral contamination (if relevant) | PCR for viral sequences (e.g., SnRV) | As needed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




