Submitted:
03 April 2025
Posted:
07 April 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Understanding the Purpose of a Research Proposal
Choosing a Research Topic
Formulating a Research Question
Importance of a Specific, Measurable, and Answerable Research Question
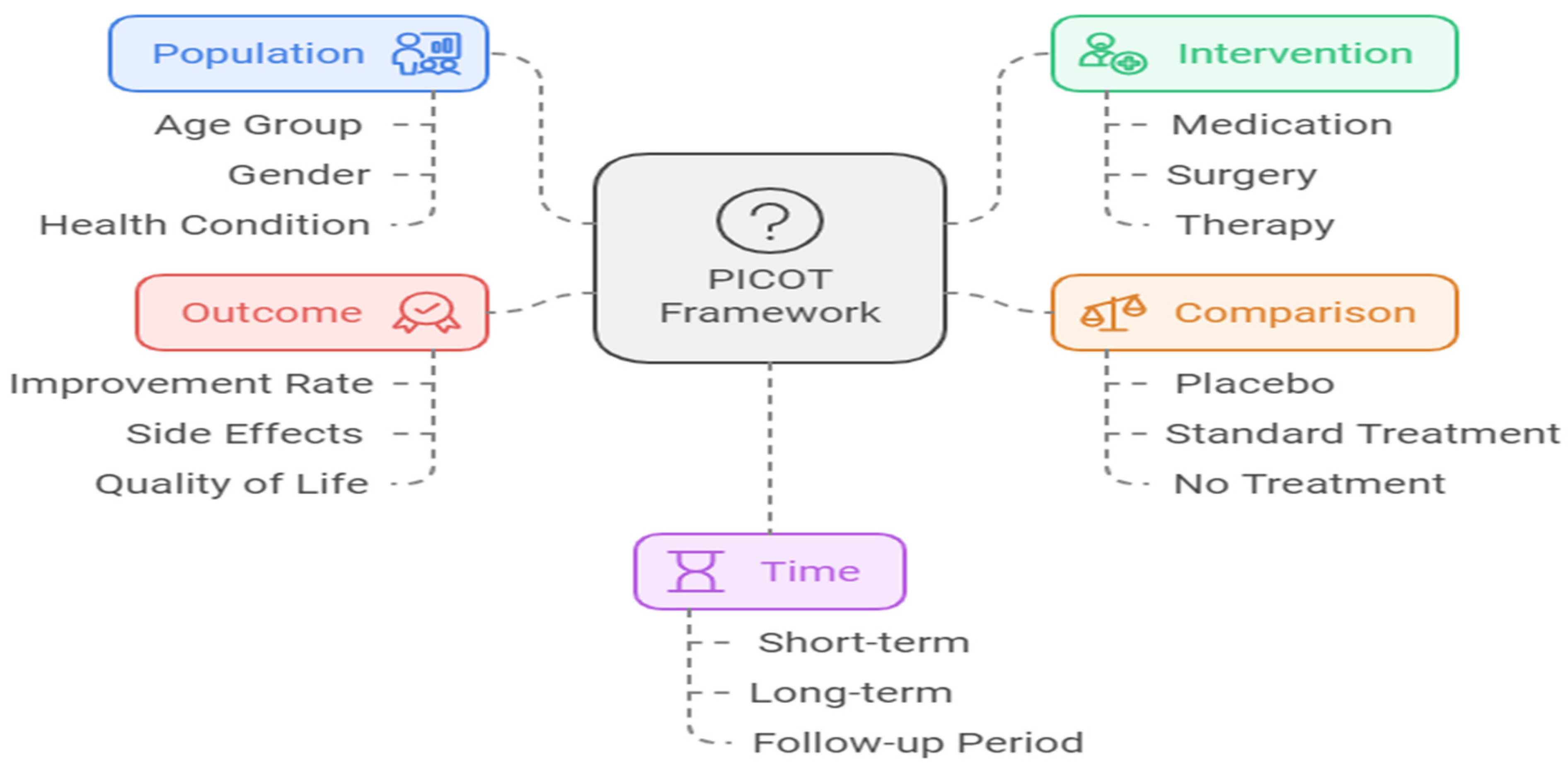
The PICOT Framework for Structuring Research Questions
Conducting a Literature Review
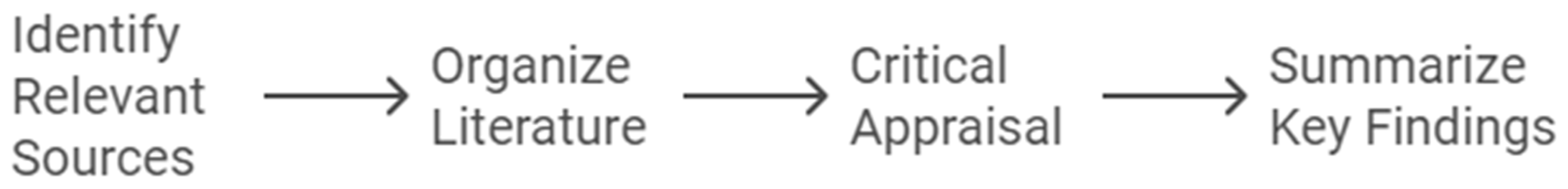
How to Perform a Comprehensive Literature Review
Identifying Gaps in Existing Knowledge and Demonstrating Originality
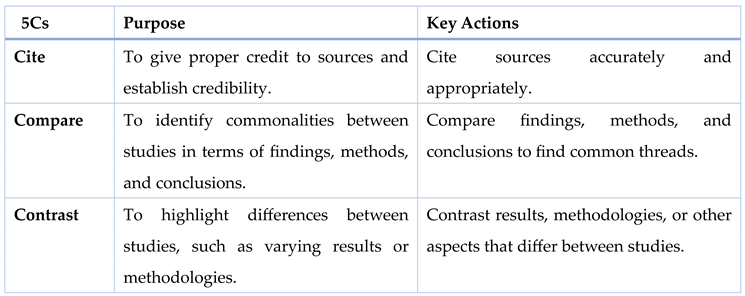
Using the 5Cs for Reviewing Literature
 |
 |
Research Design and Methodology
Overview of Research Designs in Nursing
| Research Design | Purpose | Methods/Examples | Key Considerations | Ethical Implications |
| Qualitative | Explores meaning, experiences, and perceptions in healthcare contexts. | Interviews, focus groups, observational studies (e.g., studying nursing burnout). | Time-intensive but less expensive; suitable for exploring lived experiences. | Ensure participant confidentiality and informed consent for personal interviews. |
| Quantitative | Measures effectiveness of interventions, patient outcomes, or tests hypotheses. | Surveys, clinical trials, statistical analysis (e.g., measuring recovery times with new medication). | Resource-intensive; requires participants, equipment, and statistical tools. | Careful management of RCTs to minimize risks for vulnerable populations. |
| Mixed Methods | Combines qualitative and quantitative approaches to investigate complex healthcare issues. | Triangulates numerical data and perspectives (e.g., assessing telehealth efficacy through admissions and satisfaction). | Demands proficiency in both methodologies can involve higher costs and extended timeframes. | Balance ethical concerns from both qualitative and quantitative aspects. |
Steps in Designing the Methodology
- Defining the Population
- 2.
- Determining Sample Size
- 3.
- Selecting Data Collection Methods
- 4.
- Choosing Data Analysis Techniques
- 7.
- Ethical Considerations
Ethical Issues in Nursing Research
Institutional Review Board (IRB) Approvals
- Risk-benefit ratio: The IRB ensures that the potential benefits of the research outweigh any potential risks to participants. Studies that pose significant risks without sufficient benefit are unlikely to be approved.
- Participant safety: The IRB evaluates whether adequate measures have been put in place to protect participants from harm. For instance, in clinical trials, monitoring protocols and emergency care provisions may need to be included.
- Confidentiality protections: The IRB checks whether researchers have adequate systems in place to protect the privacy and confidentiality of participants.
Informed Consent Processes
- Voluntary Participation: Participation in the study must be completely voluntary, without any form of coercion or undue influence. Participants must have the freedom to withdraw from the study at any time without penalty (Millum & Bromwich, 2021).
- Comprehensive Information: Researchers must provide participants with all necessary information about the study, including its purpose, procedures, risks, and benefits. This information must be presented in a clear and understandable manner, avoiding technical jargon that participants might not understand (Duggappa et al., 2016).
- Competence and Understanding: Participants must be competent to give consent, meaning they must have the mental capacity to understand the information provided to them. This is especially important when conducting research with vulnerable populations, such as children or individuals with cognitive impairments. In such cases, consent may also need to be obtained from legal guardians (Millum & Bromwich, 2021).
- Documentation of Consent: In most studies, informed consent is documented through a signed consent form. This form serves as legal proof that participants have been fully informed and have agreed to participate. However, in certain cases, such as studies involving online surveys, implied consent may be used, provided the study presents minimal risk to participants (Millum & Bromwich, 2021).
How to Articulate the Potential Outcomes of Research
- Define Specific Outcomes: When presenting expected outcomes, it is important to be specific. General statements such as "improving patient care" may not be sufficient. Instead, detail what specific aspects of patient care you aim to improve, such as reducing hospital readmissions, improving medication adherence, or enhancing nursing workflows (Duggappa et al., 2016).For example: In a study examining the impact of telehealth interventions on chronic disease management, a potential outcome might be a measurable reduction in hospital admissions among patients using telehealth services compared to those receiving standard care.
- Link Outcomes to Evidence-Based Practice: In nursing research, expected outcomes should contribute to the body of evidence-based practice (EBP). Outcomes that align with EBP aim to improve nursing interventions, enhance clinical decision-making, or support better health outcomes for patients. For instance, expected outcomes might show that a new intervention reduces the time nurses spend on administrative tasks, allowing more time for direct patient care (Kivunja, 2016; Ford & Melnyk, 2019).
- Short-Term vs. Long-Term Outcomes: Consider both short-term and long-term outcomes. Short-term outcomes could include immediate improvements in clinical procedures or patient satisfaction, while long-term outcomes might involve sustained reductions in healthcare costs or improved patient quality of life over several years (Duggappa et al., 2016). For example: In a study on nurse burnout, a short-term outcome might be a reduction in stress levels among nurses who participate in mindfulness training. A long-term outcome might be lower turnover rates and higher job satisfaction among the same group over a period of years.
Emphasizing the Significance of Research
- Improving Nursing Practice: Emphasizing how your research will improve nursing practice is essential in demonstrating its significance. Explain how the study addresses gaps in current practice, introduces new methods or interventions, or provides evidence to support changes in nursing protocols or policies (Barker et al., 2016). For example: If your study focuses on the implementation of simulation training in nursing education, you could highlight its potential to better prepare nurses for real-world clinical situations, thus improving overall patient care.
- Enhancing Patient Care and Outcomes: One of the primary goals of nursing research is to improve patient outcomes. Demonstrating the potential for your research to enhance patient safety, treatment efficacy, or quality of care will reinforce its significance. For example, a study on chronic disease management might emphasize the importance of early intervention and patient education in reducing hospital readmissions and improving patient quality of life (Duggappa et al., 2016). For example: A study exploring the use of evidence-based guidelines for wound care could emphasize how it may reduce infection rates and shorten healing times for patients, thereby improving their recovery experience.
- Addressing Gaps in Nursing Knowledge: To further illustrate the significance of your research, it is important to identify and address gaps in the current body of nursing knowledge. If your study fills a gap that has not been thoroughly investigated, it can lead to new insights and advancements in nursing. Highlight how your research contributes to expanding the evidence base for certain interventions or practices, providing healthcare professionals with new tools or approaches (Wong et al., 2021). For example: Researching the effectiveness of telehealth interventions in rural areas may fill a gap in studies predominantly focused on urban settings, thereby contributing to a more equitable healthcare approach.
- Supporting Healthcare Policy and Decision-Making: In addition to improving nursing practice and patient outcomes, some research studies may influence healthcare policy and decision-making. If your study has the potential to inform policies at an institutional, regional, or national level, it can further demonstrate its significance (Luijken et al., 2022). For example: A study demonstrating cost-effectiveness in telehealth interventions for managing chronic diseases might influence healthcare policies promoting wider adoption of telehealth services, especially in resource-limited settings.
- Global Health Impact: For research with broader implications, consider the global significance of your findings. Nursing research often transcends national borders, especially when addressing universal healthcare challenges such as infection control, chronic disease management, or nursing shortages (Bono et al., 2014; Wong et al., 2021). For example: A study examining infection control practices in hospitals could have global relevance by helping to standardize best practices for preventing hospital-acquired infections worldwide.
How to Develop a Realistic Timeline for Conducting Research
-
Break the Project into Phases: Divide the research process into manageable phases. Common phases include:
- ○
- Preparation Phase: Literature review, IRB approval, recruitment planning.
- ○
- Data Collection Phase: Recruitment, data collection (e.g., surveys, interviews, clinical measurements).
- ○
- Data Analysis Phase: Data cleaning, coding, and analysis using appropriate methods.
- ○
- Reporting and Dissemination Phase: Writing up results, preparing presentations or publications (Duggappa et al., 2016).
- Set Realistic Deadlines: Assign deadlines for each phase based on the complexity of the tasks and the resources available. Be mindful of potential delays, such as participant recruitment challenges or unforeseen issues in data collection, and account for these in the timeline (Barker et al., 2016). For example: For a six-month study on telehealth interventions, the preparation phase might take 1-2 months, followed by 2 months for data collection, 1 month for analysis, and 1 month for reporting.
- Incorporate Buffer Time: To account for unexpected delays, include buffer time in each phase. For example, participant recruitment may take longer than expected, or there may be delays in receiving ethical approvals. Including buffer periods ensures that the project remains on track (Barker et al., 2016).
- Gantt Charts or Timelines: Use tools like Gantt charts to visually represent the timeline. Gantt charts are useful for mapping out each task or phase, showing when tasks will be completed and where they overlap. They help researchers, stakeholders, and funders easily understand the progression of the study (Barker et al., 2016).
Budgeting for Research Expenses
-
Staffing Costs:
- Research Personnel: Include costs for any research assistants, data analysts, or coordinators who will assist with the study. These roles may be necessary for data collection, managing participants, or conducting statistical analysis (Barker et al., 2016).
- Principal Investigator Time: If the principal investigator (PI) dedicates a significant portion of their time to the study, this should be accounted for in the budget. Some grants allow for part of the PI’s salary to be covered by the research budget.
-
Equipment and Materials:
- Research Equipment: If the study involves medical equipment, laboratory tools, or software (such as statistical analysis programs), these costs should be included. Ensure that each piece of equipment is justified in the budget based on its necessity for research (Duggappa et al., 2016).
- Supplies: Include costs for any consumables required for the study, such as office supplies, printing materials for surveys, or recording devices for interviews.
-
Participant Costs:
- Incentives: Some studies may provide financial compensation or incentives to participants for their time and involvement. Be sure to include these costs if applicable. Ethical considerations also require that participants are compensated fairly, particularly if the study requires substantial commitment of time.
- Travel and Accommodation: If participants are required to travel to a research facility, include travel reimbursement and, if necessary, accommodation expenses. This is particularly relevant for studies involving patients who may need to visit clinics for testing or follow-up appointments (Barker et al., 2016).
-
Data Management and Software:
- Software Licensing: If specific software (e.g., SPSS for statistical analysis or NVivo for qualitative analysis) is required, include the cost of licensing in the budget. Some projects may also need to pay for cloud storage or data management services to store sensitive patient data securely.
- Data Analysis Costs: If external statisticians or data analysts are needed, their fees should be clearly itemized in the budget.
-
Miscellaneous Costs:
- Ethical Review Fees: Some institutions charge for processing IRB applications. If applicable, include these fees in the budget.
- Publication and Dissemination: If the results of the study are to be published in journals or presented at conferences, budget for submission fees, travel, and accommodation. Dissemination is a crucial part of research, as it helps to share findings with the wider healthcare community (Duggappa et al., 2016).
| Category | Description | Estimated Cost |
| Personnel | Research Assistant (20 hrs/week for 6 months) | $15,000 |
| Equipment | Telehealth platform licenses (10 units) | $3,000 |
| Participant Costs | Incentives for 50 participants ($50 per participant) | $2,500 |
| Travel Costs | Participant travel reimbursements (approx. $50 per trip) | $1,000 |
| Software | SPSS license (1-year subscription) | $1,200 |
| Miscellaneous | IRB review fee, printing, and supplies | $800 |
| Dissemination | Conference presentation (travel and accommodation) | $2,000 |
| Total Estimated Budget | $25,500 |
Importance of Revising the Proposal for Clarity, Consistency, and Completeness
- Clarity: A clear and well-organized proposal is easier for reviewers to understand. If the language is too complex or convoluted, the core message of the proposal may be lost, leading to misunderstandings about the research's purpose, methods, or significance. Revising for clarity involves simplifying complex ideas, eliminating jargon, and ensuring that the research objectives and methodology are clearly articulated (Kivunja, 2016). For example: When describing your research methodology, avoid overly technical language that could confuse readers. Instead, use straightforward descriptions of the research design, tools, and data collection methods.
- Consistency: Ensure that the proposal is internally consistent, meaning that all sections align with one another. The research question, objectives, and methodology should all work together without contradictions. For instance, if the research question suggests a qualitative approach, but the methodology focuses on quantitative methods, this inconsistency can weaken the proposal (Barker et al., 2016). For example: If your research question is focused on understanding nurses' perceptions of workplace stress, ensure that your methodology emphasizes qualitative methods such as interviews or focus groups, rather than quantitative surveys alone.
- Completeness: A well-rounded research proposal should leave no critical sections incomplete or vague. This includes providing adequate detail in the methodology, literature review, budget, and timeline. Missing or incomplete information may cause reviewers to question the feasibility or thoroughness of the study (Barker et al., 2016). For example: Ensure that your budget covers all necessary expenses, such as personnel, equipment, and participant costs, and that your timeline includes all phases of the research process from start to finish.
Seeking Feedback from Mentors, Peers, or Faculty Before Submission
- Mentor Feedback: Your academic or professional mentor is a valuable resource when reviewing your research proposal. Mentors have typically guided other research projects and can offer expert advice on both the content and presentation of your proposal. They can provide constructive feedback on whether the research question is appropriate, if the methodology is sound, and whether the proposal demonstrates the significance of the research (Williamson & Whittaker, 2020). For example: A mentor might suggest refining your research question to make it more focused or suggest additional literature that could strengthen your literature review.
- Peer Review: Peers can offer fresh perspectives, especially if they are familiar with your field of research. While they may not have the same level of expertise as mentors, peers can provide helpful insights into the clarity and readability of the proposal. They may catch grammatical errors, suggest alternative ways to present complex ideas, or point out inconsistencies that you might have missed (Duggappa et al., 2016). For example: A peer reviewer might highlight sections that are unclear to someone less familiar with the topic, prompting you to rephrase or restructure those parts for better understanding.
- Faculty Review: Faculty members, especially those with expertise in your specific area of research, can offer invaluable feedback on the technical and methodological aspects of your proposal. They can identify potential challenges in your research design, provide insights into funding opportunities, and offer suggestions for improving your argument's strength and coherence (Barker et al., 2016). For example: A faculty reviewer might suggest alternative data collection techniques that are more feasible or ethical, given the study's constraints, or recommend additional sources for your literature review.
- Incorporating Feedback: After receiving feedback from mentors, peers, and faculty, incorporate their suggestions into your revisions. Be open to criticism and willing to refine the proposal to address any concerns or weaknesses raised during the review process. Remember, feedback is a tool for improvement and applying it effectively can greatly increase the likelihood of your proposal’s success (Kivunja, 2016).
- Final Review: Once all feedback has been incorporated and the proposal has been revised for clarity, consistency, and completeness, conduct a final review to ensure it meets all the necessary criteria. Double-check that all sections are cohesive, and that the proposal follows the required formatting and submission guidelines. Consider running a spell check and grammar review and verify that all references are correctly cited (Kivunja, 2016).
Conclusion
Final Recommendations for Success
Formatting of funding sources
Declaration of Generative AI and AI-assisted Technologies in the Writing Process
Declaration of Interest
References
- Abbade, L. P. F., Wang, M., Sriganesh, K., Jin, Y., Mbuagbaw, L., & Thabane, L. (2017). The framing of research questions using the PICOT format in randomized controlled trials of venous ulcer disease is suboptimal: A systematic survey. Wound Repair and Regeneration, 25(5), 892–900. [CrossRef]
- Allen, J. (2019). The productive graduate student writer: How to manage your time, process, and energy to write your research proposal, thesis, and dissertation and get published. Sterling, Virginia: Stylus.
- Arienti, C., Lazzarini, S. G., Patrini, M., Puljak, L., Pollock, A., & Negrini, S. (2021). The structure of research questions in randomized controlled trials in the rehabilitation field. American Journal of Physical Medicine & Rehabilitation, 100(1), 29–33. [CrossRef]
- Barker, L., Rattihalli, R. R., & Field, D. (2016). How to write a good research grant proposal. Paediatrics and Child Health, 26(3), 105–109. [CrossRef]
- Barrow, J. M., Khandhar, P. B., & Brannan, G. D. (2022). Research ethics. National Library of Medicine; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK459281/.
- Bono, S. D., Heling, G., & Borg, M. A. (2014). Organizational culture and its implications for prevention and control in healthcare institutions. Journal of Hospital Infection, 86(1), 1–6. [CrossRef]
- Callahan, J. L. (2014). Writing literature reviews. Human Resource Development Review, 13(3), 271–275. [CrossRef]
- Creswell, J. W., & Creswell, J. D. (2023). Research design: Qualitative, quantitative, and mixed methods approaches (5th ed.). SAGE Publications.
- Creswell, J. W., & Poth, C. N. (2018). Qualitative inquiry & research design: Choosing among five approaches (4th ed.). SAGE Publications.
- Duggappa, D. R., Sudheesh, K., & Nethra, S. (2016). How to write a research proposal? Indian Journal of Anaesthesia, 60(9), 631. [CrossRef]
- Ford, L. G., & Melnyk, B. M. (2019). The Underappreciated and Misunderstood PICOT Question: a Critical Step in the EBP Process. Worldviews on Evidence-Based Nursing, 16(6), 422–423. [CrossRef]
- Gerrish, K., & Lathlean, J. (2015). The research process in nursing (7th ed.). John Wiley & Sons, Ltd.
- Hosseini, M.-S., Jahanshahlou, F., Akbarzadeh, M.-A., Zarei, M., & Vaez-Gharamaleki, Y. (2023). Formulating research questions for evidence-based studies. Journal of Medicine, Surgery, and Public Health, 2(2), 100046. [CrossRef]
- Kivunja, C. (2016). How to write an effective research proposal for higher degree research in higher education: Lessons from practice. International Journal of Higher Education, 5(2). [CrossRef]
- Luijken, K., Dekkers, O. M., Rosendaal, F. R., & Groenwold, R. H. H. (2022). Exploratory analyses in aetiologic research and considerations for assessment of credibility: Mini-review of literature. BMJ, 377, e070113. [CrossRef]
- Millum, J., & Bromwich, D. (2021). Informed consent: What must be disclosed and what must be understood? The American Journal of Bioethics, 21(5), 1–19. [CrossRef]
- Rios, L. P., Ye, C., & Thabane, L. (2010). Association between framing of the research question using the PICOT format and reporting quality of randomized controlled trials. BMC Medical Research Methodology, 10(1). [CrossRef]
- Salmona, M., & Kaczynski, D. (2024). Qualitative data analysis strategies. In D. Kaczynski, M. Salmona, & T. Smith (Eds.), How to Conduct Qualitative Research in Finance (pp. 80–96). Edward Elgar Publishing. https://www.elgaronline.com/edcollchap/book/9781803927008/chapter6.xml.
- Schneider, Z., & Fuller, J. (2018). Writing Research Proposals in the Health Sciences (1st ed.). London: SAGE Publications, Limited. [CrossRef]
- Sheppard, V. (2020). Research methods for the social sciences: An introduction. In pressbooks.bccampus.ca. Pressbooks. https://pressbooks.bccampus.ca/jibcresearchmethods/.
- Tappen, R. M. (2023). Advanced nursing research: From theory to practice (3rd ed.). Jones & Bartlett Learning.
- Williamson, G., & Whittaker, A. (2020). Succeeding in literature reviews and research project plans for nursing students. (4th ed.). London ; Thousand Oaks, California : Learning Matters, an Imprint of SAGE Publications Ltd. (Original work published 2011).
- Wong, E. C., Maher, A. R., Motala, A., Ross, R., Akinniranye, O., Larkin, J., & Hempel, S. (2021). Methods for identifying health research gaps, needs, and priorities: A scoping review. Journal of General Internal Medicine, 37(1). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




