Submitted:
01 April 2025
Posted:
02 April 2025
You are already at the latest version
Abstract
Keywords:
Introduction
2. Materials and Methods
2.1. Test Material
2.2. Dermatological Clinical Efficacy Study
2.3. Statistics
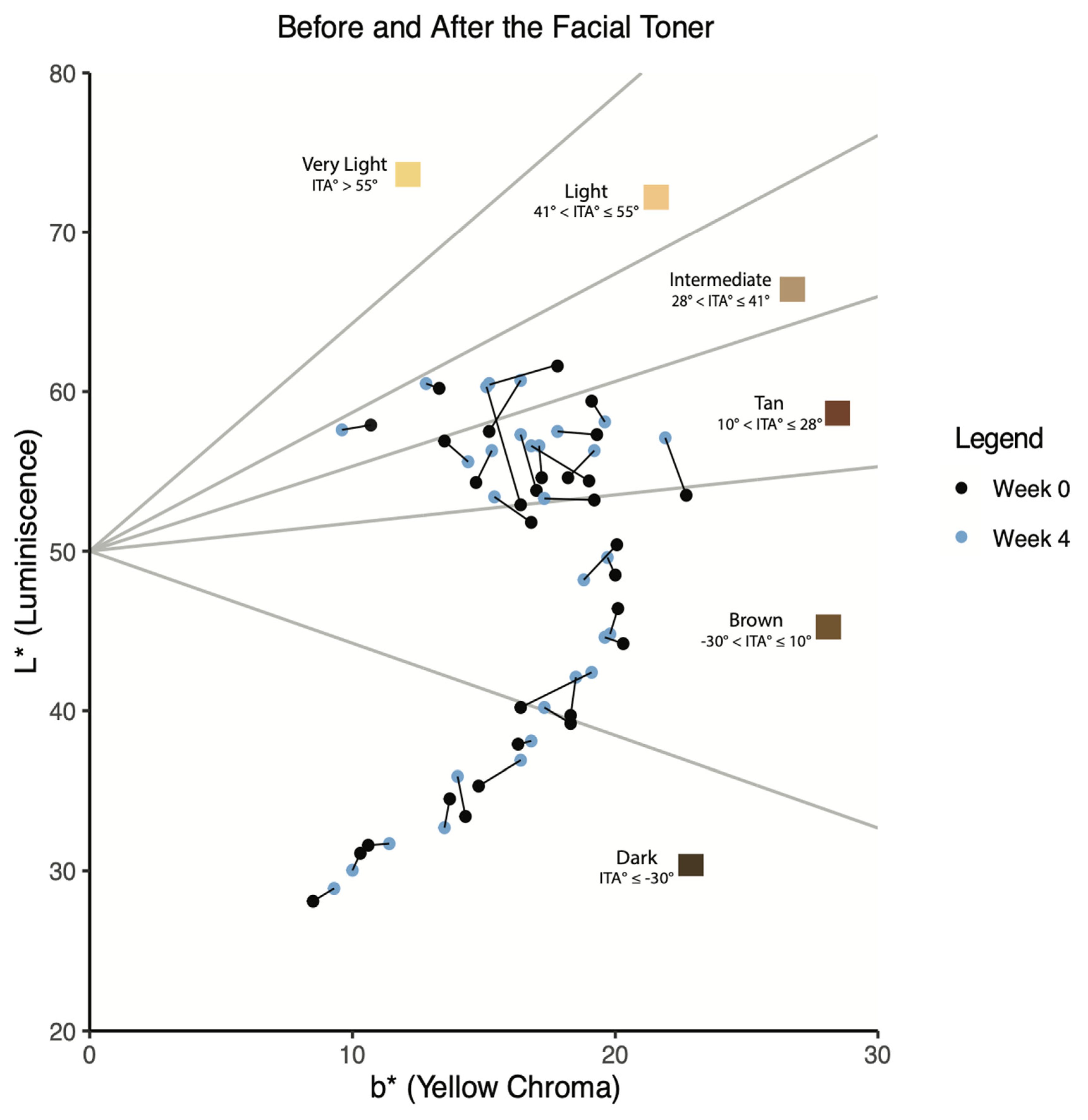
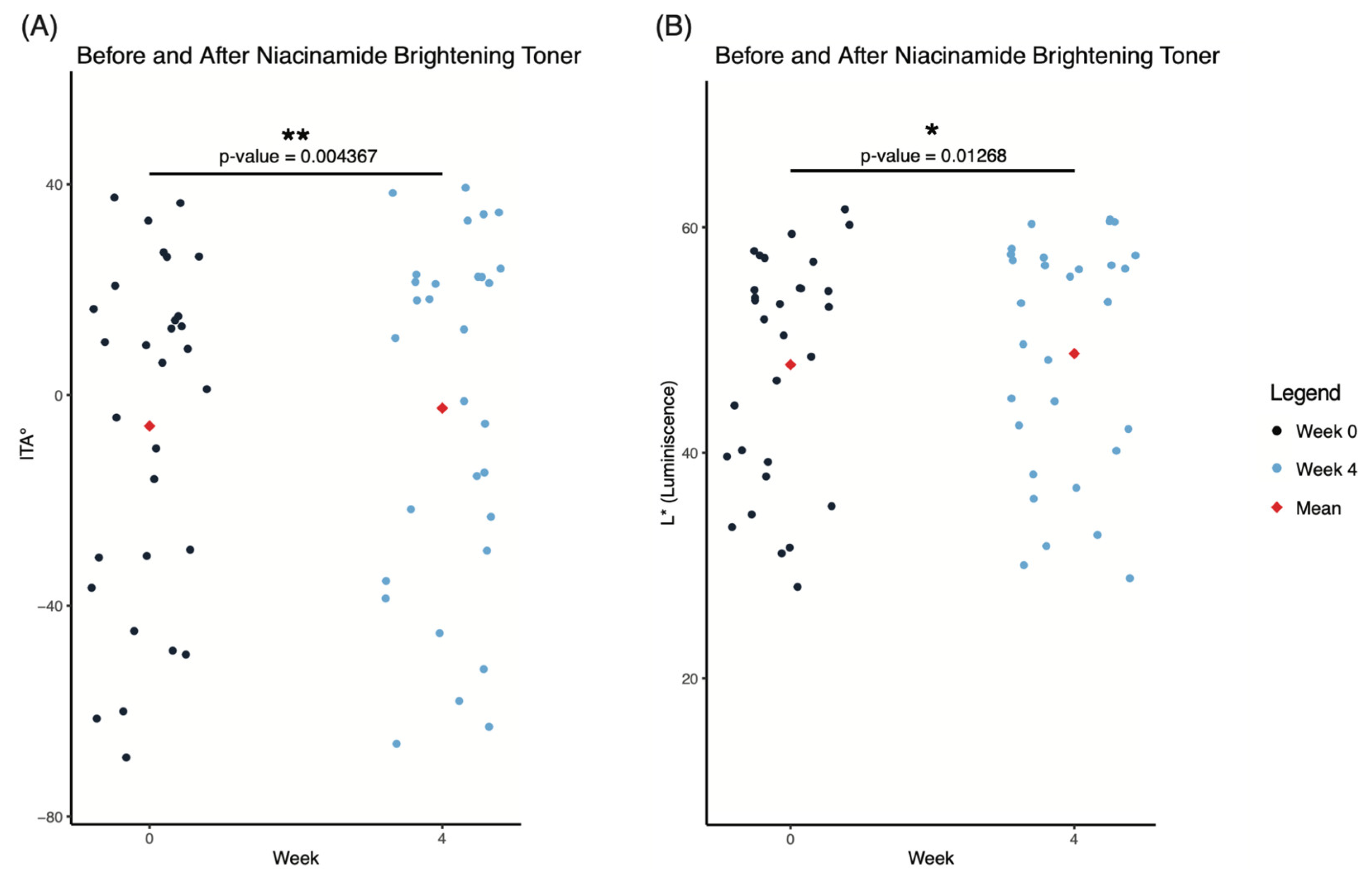
Results
Discussion
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflict of Interest
References
- Kligman, A. M.; Koblenzer, C. Demographics and Psychological Implications for the Aging Population. Dermatol. Clin. 1997, 15, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Samson, N.; Fink, B.; Matts, P. J. Visible Skin Condition and Perception of Human Facial Appearance. Int. J. Cosmet. Sci. 2010, 32, 167–184. [Google Scholar] [CrossRef] [PubMed]
- Derogatis, L. R.; Spencer, P. M. Administration and Procedures: BSI Manual I. Baltim. Clin. Psychom. Res. 1982, 28–51. [Google Scholar]
- Gupta, M. A.; Schork, N. J.; Ellis, C. N. Psychosocial Correlates of the Treatment of Photodamaged Skin with Topical Retinoic Acid: A Prospective Controlled Study. J. Am. Acad. Dermatol. 1994, 30, 969–972. [Google Scholar] [CrossRef] [PubMed]
- Baumann, L. Skin Ageing and Its Treatment. J. Pathol. 2007, 211, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, Y.; Hearing, V. J. Physiological Factors That Regulate Skin Pigmentation. BioFactors 2009, 35, 193–199. [Google Scholar] [CrossRef] [PubMed]
- França, K.; Keri, J. Psychosocial Impact of Acne and Postinflammatory Hyperpigmentation. An. Bras. Dermatol. 2017, 92, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Nautiyal, A.; Wairkar, S. Management of Hyperpigmentation: Current Treatments and Emerging Therapies. Pigment Cell Melanoma Res. 2021, 34, 1000–1014. [Google Scholar] [CrossRef] [PubMed]
- Woolery-Lloyd, H.; Kammer, J. N. Treatment of Hyperpigmentation. Semin. Cutan. Med. Surg. 2011, 30, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Boo, Y. C. Arbutin as a Skin Depigmenting Agent with Antimelanogenic and Antioxidant Properties. Antioxidants 2021, 10, 1129. [Google Scholar] [CrossRef] [PubMed]
- Scientific Committee on Consumer Safety (SCCS). Opinion on the Safety of Alpha- (CAS No. 84380-018, EC No. 617-561-8) and Beta-Arbutin (CAS No. 497-76- 7, EC No. 207-8503) in Cosmetic Products, Preliminary Version of 15–16 March 2022, Final Version of 31 January 2023, SCCS/1642/22. 2023. Available online: https://health.ec.europa.eu/publications/safety-alpha-arbutin-and-beta-arbutin-cosmetic-products_en.
- Inoue, Y.; Hasegawa, S.; Yamada, T.; Date, Y.; Mizutani, H.; Nakata, S.; Matsunaga, K.; Akamatsu, H. Analysis of the Effects of Hydroquinone and Arbutin on the Differentiation of Melanocytes. Biol. Pharm. Bull. 2013, 36, 1722–1730. [Google Scholar] [CrossRef] [PubMed]
- Boo, Y. C. Mechanistic Basis and Clinical Evidence for the Applications of Nicotinamide (Niacinamide) to Control Skin Aging and Pigmentation. Antioxidants 2021, 10, 1315. [Google Scholar] [CrossRef] [PubMed]
- Bissett, D. L.; Miyamoto, K.; Sun, P.; Li, J.; Berge, C. A. Topical Niacinamide Reduces Yellowing, Wrinkling, Red Blotchiness, and Hyperpigmented Spots in Aging Facial Skin. Int. J. Cosmet. Sci. 2004, 26, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Del Bino, S.; Bernerd, F. Variations in Skin Colour and the Biological Consequences of Ultraviolet Radiation Exposure. Br. J. Dermatol. 2013, 169, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Tanaka, T.; Akimoto, M. Utilization of Individual Typology Angle (ITA) and Hue Angle in the Measurement of Skin Color on Images. Bioimaging Society 2020. [CrossRef]
- Morag, M.; Nawrot, J.; Siatkowski, I.; Adamski, Z.; Fedorowicz, T.; Dawid-Pac, R.; Urbanska, M.; Nowak, G. A Double-blind, Placebo-controlled Randomized Trial of Serratulae Quinquefoliae Folium, a New Source of β -arbutin, in Selected Skin Hyperpigmentations. J. Cosmet. Dermatol. 2015, 14, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Bissett, D. L.; Oblong, J. E.; Berge, C. A. Niacinamide: A B Vitamin That Improves Aging Facial Skin Appearance. Dermatol. Surg. 2005, 31, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Krishnapriya, K. S.; Pangelinan, G.; King, M. C.; Bowyer, K. W. Analysis of Manual and Automated Skin Tone Assignments. In 2022 IEEE/CVF Winter Conference on Applications of Computer Vision Workshops (WACVW); IEEE: Waikoloa, HI, USA, 2022; pp. 429–438. [Google Scholar] [CrossRef]
- Ly, B. C. K.; Dyer, E. B.; Feig, J. L.; Chien, A. L.; Del Bino, S. Research Techniques Made Simple: Cutaneous Colorimetry: A Reliable Technique for Objective Skin Color Measurement. J. Invest. Dermatol. 2020, 140, 3–12e1. [Google Scholar] [CrossRef] [PubMed]
- Lee, E. J.; Ryu, J. H.; Baek, J. H.; Boo, Y. C. Skin Color Analysis of Various Body Parts (Forearm, Upper Arm, Elbow, Knee, and Shin) and Changes with Age in 53 Korean Women, Considering Intrinsic and Extrinsic Factors. J. Clin. Med. 2024, 13, 2500. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Positive Anchor (0) | Negative Anchor (9) |
|---|---|---|
| Pore Appearance | Small, not easily visible | Large, easily visible |
| Skin Texture/Smoothness | Smooth, even surface appearance | Rough, uneven surface appearance |
| Radiance/Luminosity | Radiant, luminous, glowing appearance | Dull/matte and/or sallow, lackluster |
| Factor | n | Percentage (%) | |
|---|---|---|---|
| Sex | Female | 23 | 74.19 |
| Male | 8 | 25.81 | |
| Race & Ethnicity | Asian | 1 | 3.23 |
| Black | 13 | 41.94 | |
| Caucasian | 15 | 48.39 | |
| Hispanic or Latino | 2 | 6.45 | |
| Fitzpatrick Skin Type | I | 0 | 0.00 |
| II | 9 | 29.03 | |
| III | 7 | 22.58 | |
| IV | 5 | 16.13 | |
| V | 9 | 29.03 | |
| VI | 1 | 3.23 | |
| Age | Mean | 45 | |
| Min | 18 | ||
| Max | 68 | ||
| n = 31 | |||
| Parameter | Time Point | Mean ± SD | p-value | Mean Percent Change From Baseline | Percent of Volunteers with Improvement |
|---|---|---|---|---|---|
| Pore Appearance | Baseline | 5.35 ± 1.25 | |||
| Week 4 | 4.65 ± 1.38 | 0.000* | -13.25% | 58.06% | |
| Skin Texture Smoothness | Baseline | 5.68 ± 0.83 | |||
| Week 4 | 4.97 ± 0.75 | 0.000* | -12.50% | 64.52% | |
| Radiance/Luminosity | Baseline | 6.00 ± 0.77 | |||
| Week 4 | 4.94 ± 0.77 | 0.000* | -17.74% | 87.10% | |
| * Statistically Significant Improvement (p < 0.0001) n = 31 Decrease = Improvement | |||||
| Self-perception statements: | Overall Agreement (%)* |
|---|---|
| After using the product... | After 4 weeks |
| 1. My skin felt smoother after using the Toner. | 96.8 |
| 2. My skin looked more radiant after using the Toner. | 93.6 |
| 3. My skin looked brighter after using the Toner. | 87.1 |
| 4. My skin looked more youthful after using the Toner. | 87.1 |
| 5. My skin felt softer after using the Toner. | 87.1 |
| 6. I noticed a more refined skin texture after using the Toner. | 87.1 |
| 7. I would recommend this Toner to a friend. | 87.1 |
| 8. I would purchase this Toner. | 87.1 |
| 9. The pores on my face looked less visible after using the Toner. | 83.9 |
| 10. My skin looked less dull after using the Toner. | 80.7 |
| 11. My skin looked more even after using the Toner. | 80.7 |
| 12. The dark spots on my face looked lighter after using the Toner. | 80.7 |
| 13. My skin felt less oily after using the Toner. | 77.4 |
| 14. My skin felt tighter after using the Toner. | 77.4 |
| 15. I noticed less blemishes on my skin after using the Toner. | 77.4 |
| 16. I noticed less discoloration on my skin after using the Toner. | 74.2 |
| * Calculated from a self-perception questionnaire using a 4-point Likert scale at week 4. Overall Agreement represents volunteers that Agree or Strongly Agree with the statements. Results ≥50% are favorable responses. n = 31 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




