Submitted:
29 March 2025
Posted:
02 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- Data Curation Process
- Peer-reviewed articles published within the last five decades.
- Studies demonstrating acute or chronic effects on circadian rhythms.
- Compounds documented as in vitro and in vivo studies.
- Data Extraction
- Ethical Considerations
- Database Implementation
- Data Sources of the Chronobiotics Database
- Data Curation Process and Implementation
- Data Validation
- Data Integration and Standardization
2.1. Database Organization (Primary and Secondary Data)
2.2. Database Architecture
2.2.2. Auxiliary Tables
- One-to-Many Relationships: The synonyms table is connected to Chronobiotic via the foreign key originalbiotic.
- Many-to-Many Relationships: The target, mechanism, and class tables are linked to Chronobiotic through intermediary tables, which are automatically generated by Django.
- Chronobiotic → synonyms: A single compound may have multiple synonyms.
- Chronobiotic → target: A single compound may interact with multiple targets, and a single target may be associated with multiple compounds.
- Chronobiotic → mechanism: A single compound may exhibit multiple mechanisms of action, and a single mechanism may be associated with multiple compounds.
- Chronobiotic → class: A single compound may belong to multiple classes, and a single class may encompass multiple compounds.
- DBMS: PostgreSQL, a robust and reliable relational database, provides high performance and supports complex queries (Simkovics, S. and Petersgasse, P., 1998).
- ORM: Django ORM is employed for database interactions at the Python code level. This eliminates the need for manual SQL query writing and facilitates efficient data management. (Holovaty. and Willison, 2003.)
- Indexes: Indexes have been created on frequently queried fields, such as gname, smiles, and targetsname, to optimize search performance.
- Migrations: Django's built-in migration system allows for seamless modifications to the database structure without data loss.
3. Ensuring Data Integrity and Security
- Foreign Keys: All inter-table relationships are implemented through foreign keys, ensuring data integrity.
- Unique Constraints: Unique fields (gname, smiles, molecula, iupacname) prevent record duplication.
- Role-Based Access Control: Database access is restricted at the user and role levels, ensuring data security.
- Encryption: Confidential data is stored in an encrypted format.
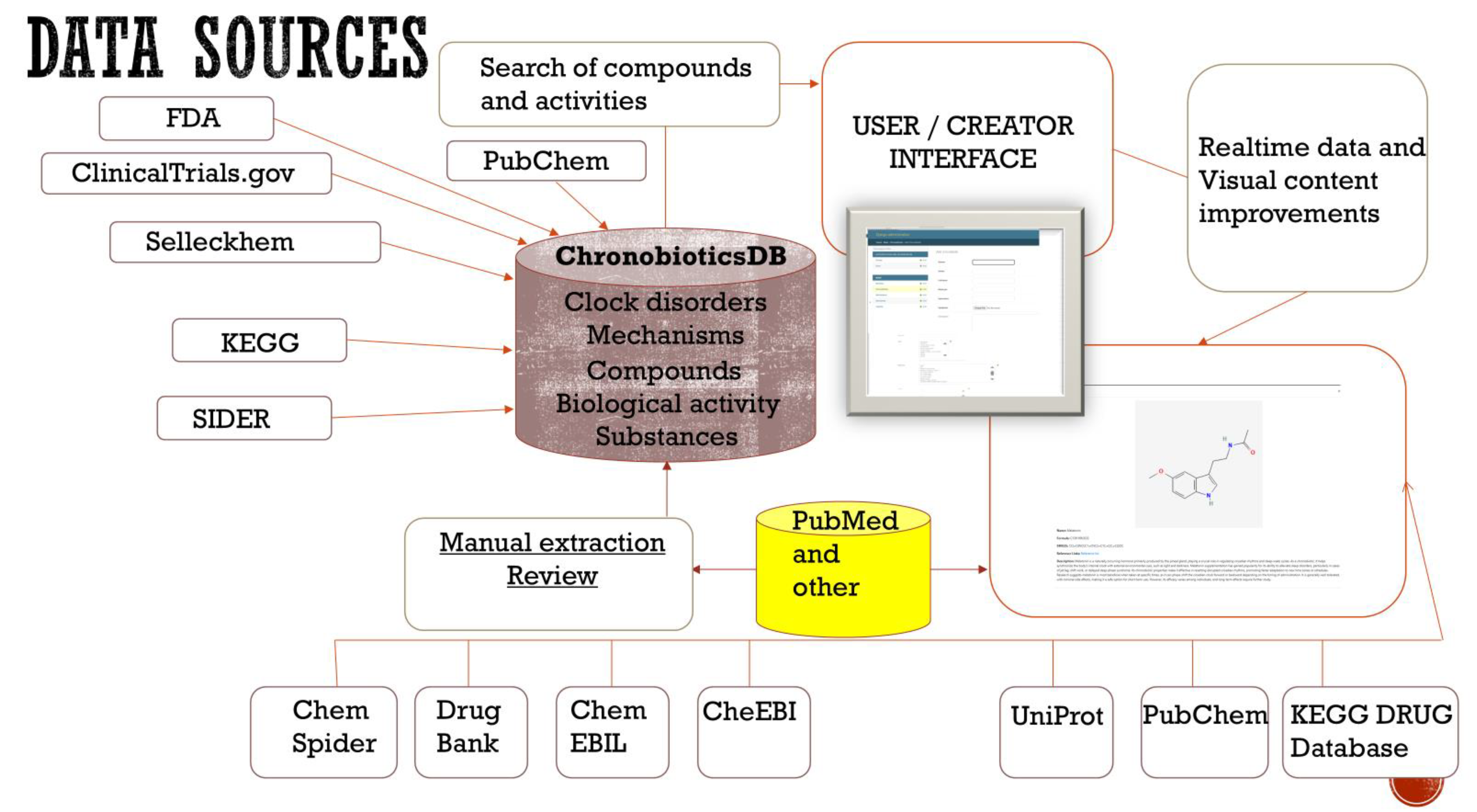
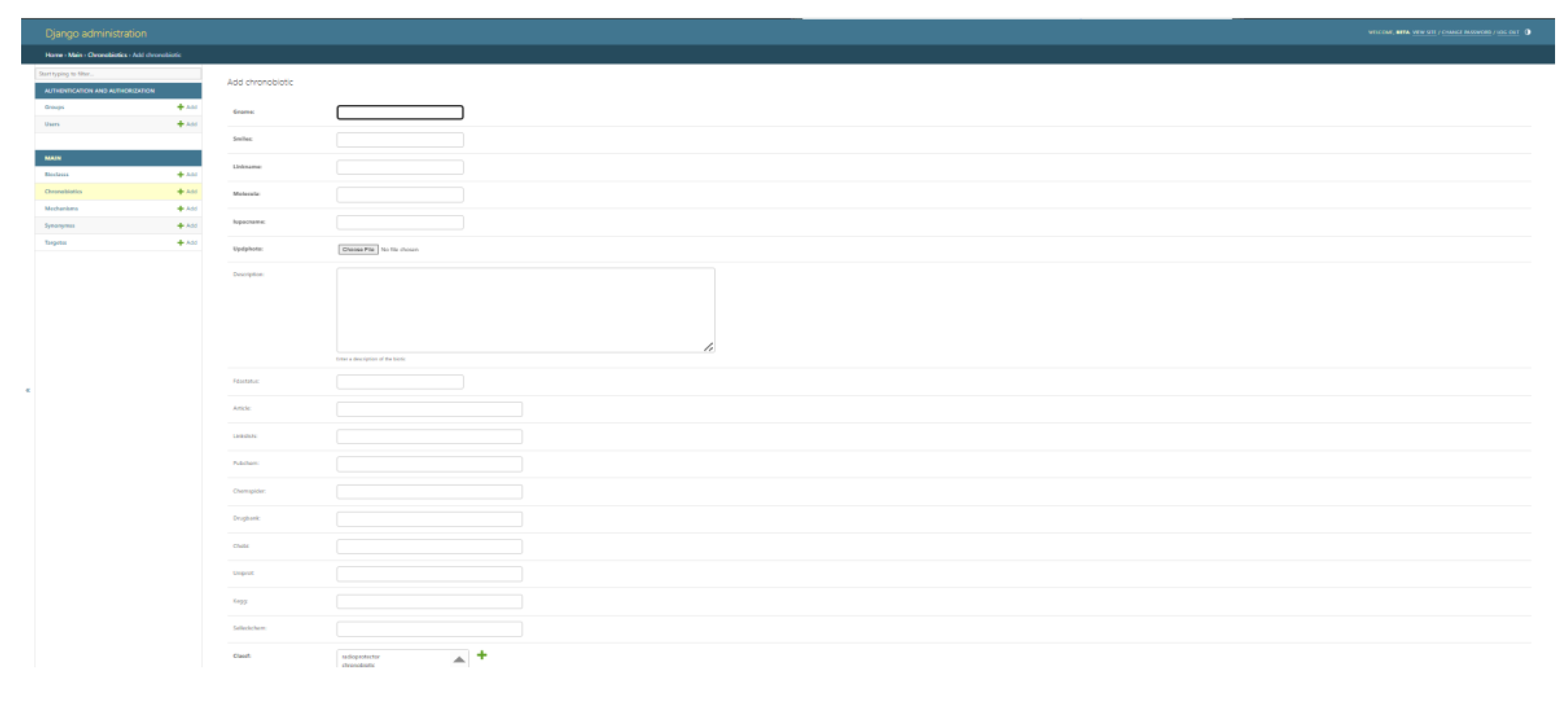
4. Results
- Database Web Interface
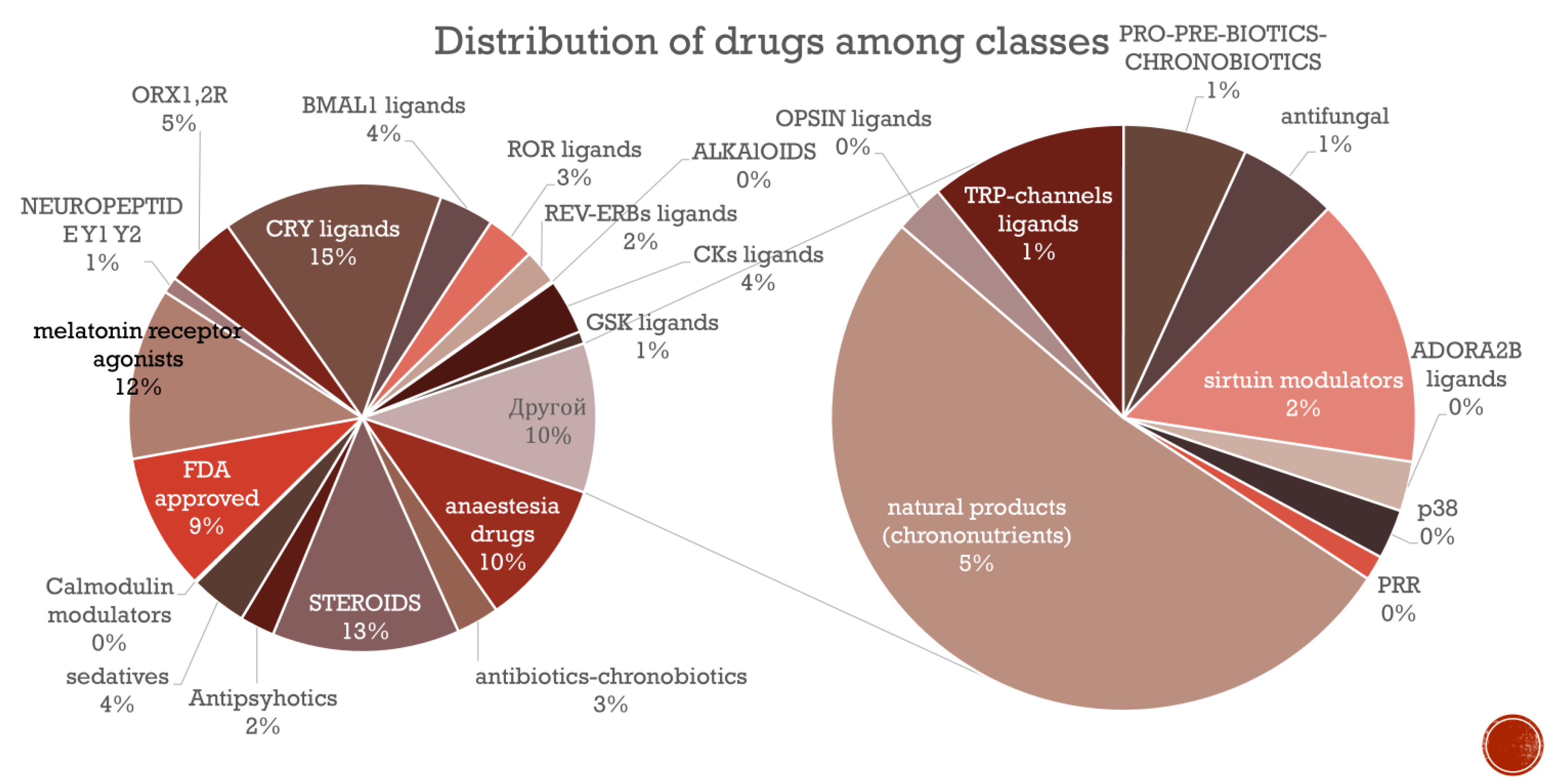
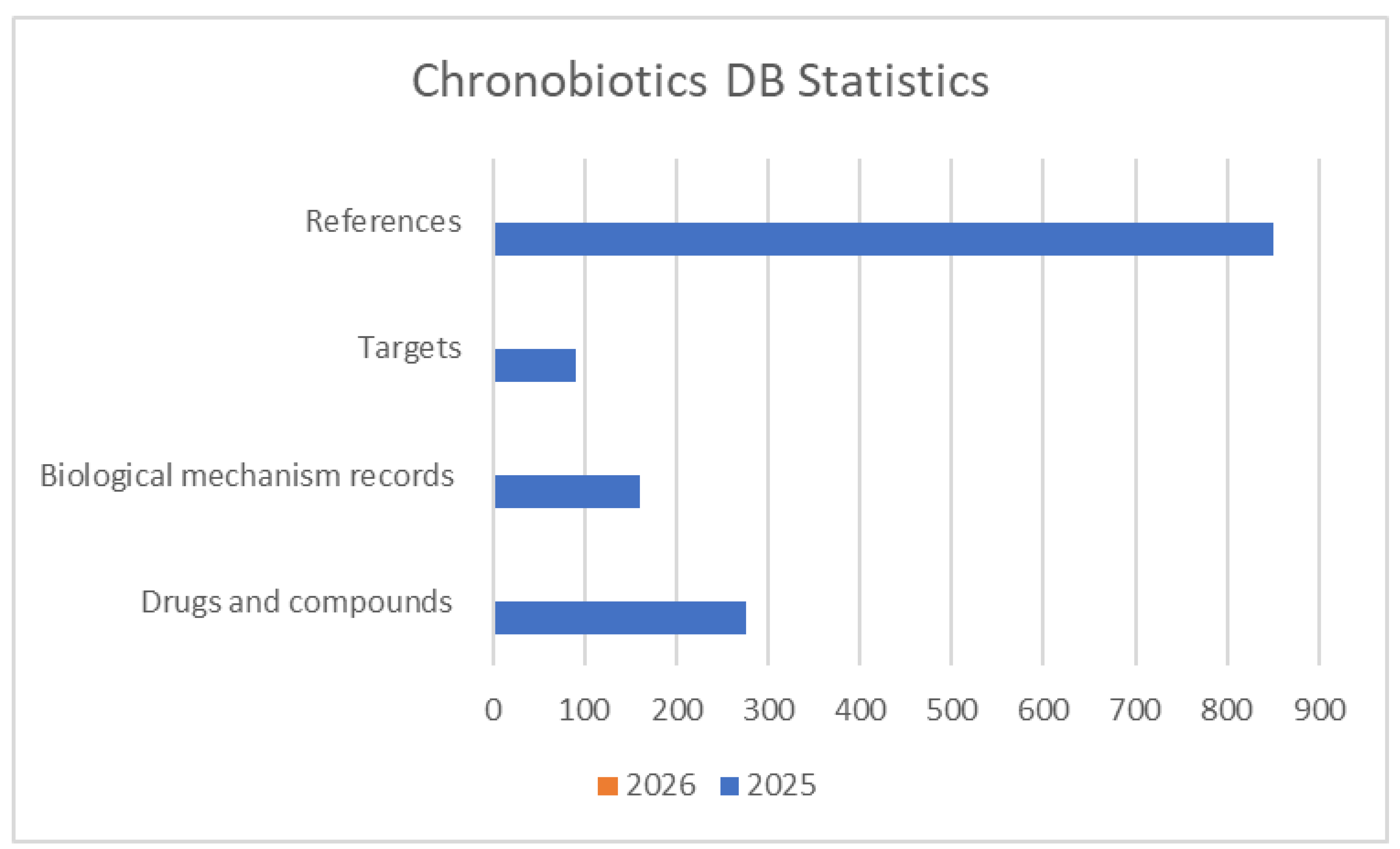
4.1. Database Statistics and Analysis
- Terpenes/Steroids
- Examples:
- CCCCCC1=CC(=C(C(=C1)O)C2C=C(CCC2C(=C)C)C)O
- CC12CCC3C(C1CCC2O)CCC4=CC(=C(C=C34)OC)O
- CC(=O)C1CCC2C1(CCC3C2CC=C4C3(CCC(C4)O)C)C
- Features: Multi-ring systems with hydroxyl groups and isoprene-like chains. Common in triterpenes, steroids, or sterols.
- Alkaloids
- Examples:
- CN1CCC23C4C(=O)CCC2(C1CC5=C3C(=C(C=C5)OC)O4)O
- CN1C2CCC1C(C(C2)OC(=O)C3=CC=CC=C3)C(=O)OC
- Features: Nitrogen-containing heterocycles (e.g., indole, piperidine) and fused ring systems.
- Sulfonamides/Sulfonates
- Examples:
- C1=CC=C(C=C1)S(=O)(=O)[O-]
- C1=CC(=C(C=C1)Cl)Cl)CON=C(...)Cl.[N+](=O)(O)[O-]
- Features: Sulfonyl groups (S(=O)₂) and aromatic rings. Common in antibiotics or diuretics.
- Aromatic Compounds (Phenols, Flavonoids)
- Examples:
- CC1=CC(=C(C(=C1C=CC(=CC=CC(=CC(=O)O)C)C)C)OC
- COC1=CC(=CC(=C1OC)OC)C2C3C(COC3=O)C(C4=CC5=C(C=C24)OCO5)O
- Features: Multiple hydroxyl/methoxy groups on benzene rings, common in polyphenols or flavonoids.
- Organofluorines/Organochlorines
- Examples:
- C(C(F)(F)F)(OC(F)F)F
- C1=CC(=C(C=C1Cl)Cl)CON=C(...)Cl
- Features: Trifluoromethyl (CF₃), chloro substituents. Found in pharmaceuticals (e.g., antidepressants).
- Quaternary Ammonium Compounds
- Examples:
- C[N+](C)(C)CCOC(=O)CCC(=O)OCC[N+](C)(C)C
- Features: Positively charged nitrogen with alkyl chains. Used as surfactants or disinfectants.
- Amides/Peptides
- Examples:
- CCC(=O)N(C1CCN(CC1)CCC2=CC=CC=C2)C3=CC=CC=C3
- CC(=O)N(C1=CC=CC=C1)C2(CCN(CC2)CCC3=CC=CS3)COC
- Features: NC=O groups, common in pharmaceuticals (e.g., β-lactams).
- Esters/Lipids
- Examples:
- CCCCCCCCCC(=O)O
- CCOC(=O)C1=CC=CC=C1C(=O)OCCCC
- Features: Ester linkages (COO), long alkyl chains. Found in fatty acid esters.
- Heterocycles (Pyridine, Thiophene)
- Examples:
- C1=CN(C(=O)N=C1N)CC(CO)OCP(=O)(O)O
- C1=CC(=C2C(=C1NCCNCCO)C(=O)C3=C(C=CC(=C3C2=O)O)O)NCCNCCO
- Features: Nitrogen/sulfur-containing rings (e.g., pyridine, thiophene).
- Nitro Compounds
- Examples:
- C1=CC(=CC=C1C=CC2=CC(=CC(=C2)O)O)O
- Features: NO₂ groups. Used in explosives or dyes.
- Phosphates/Nucleotides
- Examples:
- C1=CN(C(=O)N=C1N)CC(CO)OCP(=O)(O)O
- Features: Phosphate groups (PO₄³⁻). Found in nucleotides (e.g., ATP).
- Organometallics/Salts
- Examples:
- [O-2].[O-2].[O-2].[As+3].[As+3] (Arsenate salt)
- C(=O)(C(F)(F)F)O (Trifluoroacetate salt)
- Features: Metal ions or ionic groups (e.g., [Na+]).
- Macrocyclic Compounds
- Examples:
- CC12CCC(=O)C=C1C3CC3C4C2CCC5(C4C6CC6C57CCC(=O)O7)C
- Features: Large rings (12+ atoms). Seen in macrolide antibiotics.
- Disulfides
- Examples:
- C(C(=O)NC(...)CSSCC(...)
- Features: S-S bonds. Common in peptides (e.g., insulin).
- Alkyne Derivatives
- Examples:
- CCC#CC(C)C1(C(=O)NC(=O)N(C1=O)C)CC=C
- Features: Triple bonds (C#C). Used in click chemistry.
- Barbiturates
- Examples:
- CCC(=O)NC(=O)NC1=O
- Features: Pyrimidine-2,4,6-trione core. Sedative/hypnotic agents.
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bittrich, S., Segura, J., Duarte, J.M., Burley, S.K. and Rose, Y., 2024. RCSB protein Data Bank: exploring protein 3D similarities via comprehensive structural alignments. Bioinformatics, 40(6), p.btae370.
- Califf, R.M., Cutler, T.L., Marston, H.D. and Meeker-O’Connell, A., 2025. The importance of ClinicalTrials. gov in informing trial design, conduct, and results. Journal of Clinical and Translational Science, 9(1), p.e42.
- Cha, H.K., Chung, S., Lim, H.Y., Jung, J.W. and Son, G.H., 2019. Small molecule modulators of the circadian molecular clock with implications for neuropsychiatric diseases. Frontiers in molecular neuroscience, 11, p.496.
- Chen, Z., Yoo, S.H. and Takahashi, J.S., 2018. Development and therapeutic potential of small-molecule modulators of circadian systems. Annual review of pharmacology and toxicology, 58(1), pp.231-252.
- Holovaty, A. Holovaty, A. and Willison, S., 2003. Django web framework. Available online: http://www. djangoproject. Com.
- Imming, P., 2015. Medicinal chemistry: Definitions and objectives, drug activity phases, drug classification systems. In The Practice of Medicinal Chemistry (pp. 3-13). Academic Press.
- Kim, S. and Bolton, E.E., 2024. PubChem: A Large-Scale Public Chemical Database for Drug Discovery. Open Access Databases and Datasets for Drug Discovery, pp.39-66.
- Knox, C. Knox, C., Wilson, M., Klinger, C.M., Franklin, M., Oler, E., Wilson, A., Pon, A., Cox, J., Chin, N.E., Strawbridge, S.A. and Garcia-Patino, M., 2024. DrugBank 6.0: the DrugBank knowledgebase for 2024. Nucleic acids research, 52(D1), pp.D1265-D1275.
- Odell, S.G., Lazo, G.R., Woodhouse, M.R., Hane, D.L. and Sen, T.Z., 2017. The art of curation at a biological database: principles and application. Current Plant Biology, 11, pp.2-11.
- Pence, H.E. and Williams, A., 2010. ChemSpider: an online chemical information resource.
- Pizarro, A., Hayer, K., Lahens, N.F. and Hogenesch, J.B., 2012. CircaDB: a database of mammalian circadian gene expression profiles. Nucleic acids research, 41(D1), pp.D1009-D1013.
- Robles-Piedras, A.L., Bautista-Sánchez, U., Olvera-Hernández, E.G. and Chehue-Romero, A., 2024. Chronopharmacokinetics: A Brief Analysis of the Influence of Circadian Rhythm on the Absorption, Distribution, Metabolism, and Elimination of Drugs. Biomedical and Pharmacology Journal, 17(3), pp.2011-2017.
- Simkovics, S. and Petersgasse, P., 1998. Enhancement of the ANSI SQL Implementation of PostgreSQL.
- Solovev, I.A. and Golubev, D.A., 2024. Chronobiotics: classifications of existing circadian clock modulators, future perspectives. Biomeditsinskaya khimiya, 70(6), pp.381-393.
- Tamai, T.K., Nakane, Y., Ota, W., Kobayashi, A., Ishiguro, M., Kadofusa, N., Ikegami, K., Yagita, K., Shigeyoshi, Y., Sudo, M. and Nishiwaki-Ohkawa, T., 2018. Identification of circadian clock modulators from existing drugs. EMBO molecular medicine, 10(5), p.e8724.
- Zhu, X., Han, X., Li, Z., Zhou, X., Yoo, S.H., Chen, Z. and Ji, Z., 2025. CircaKB: a comprehensive knowledgebase of circadian genes across multiple species. Nucleic Acids Research, 53(D1), pp.D67-D78.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



