Submitted:
01 April 2025
Posted:
01 April 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
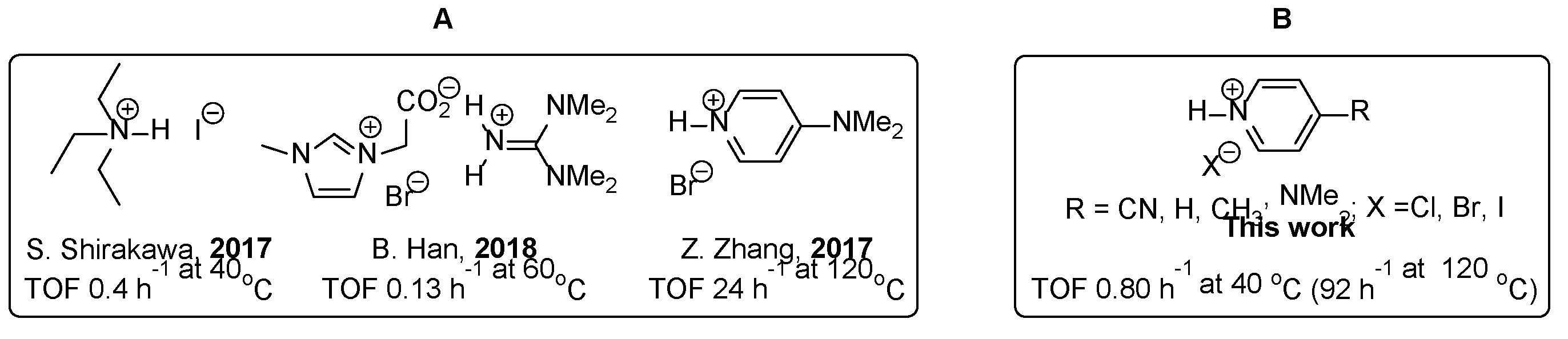
2. Results and Discussion
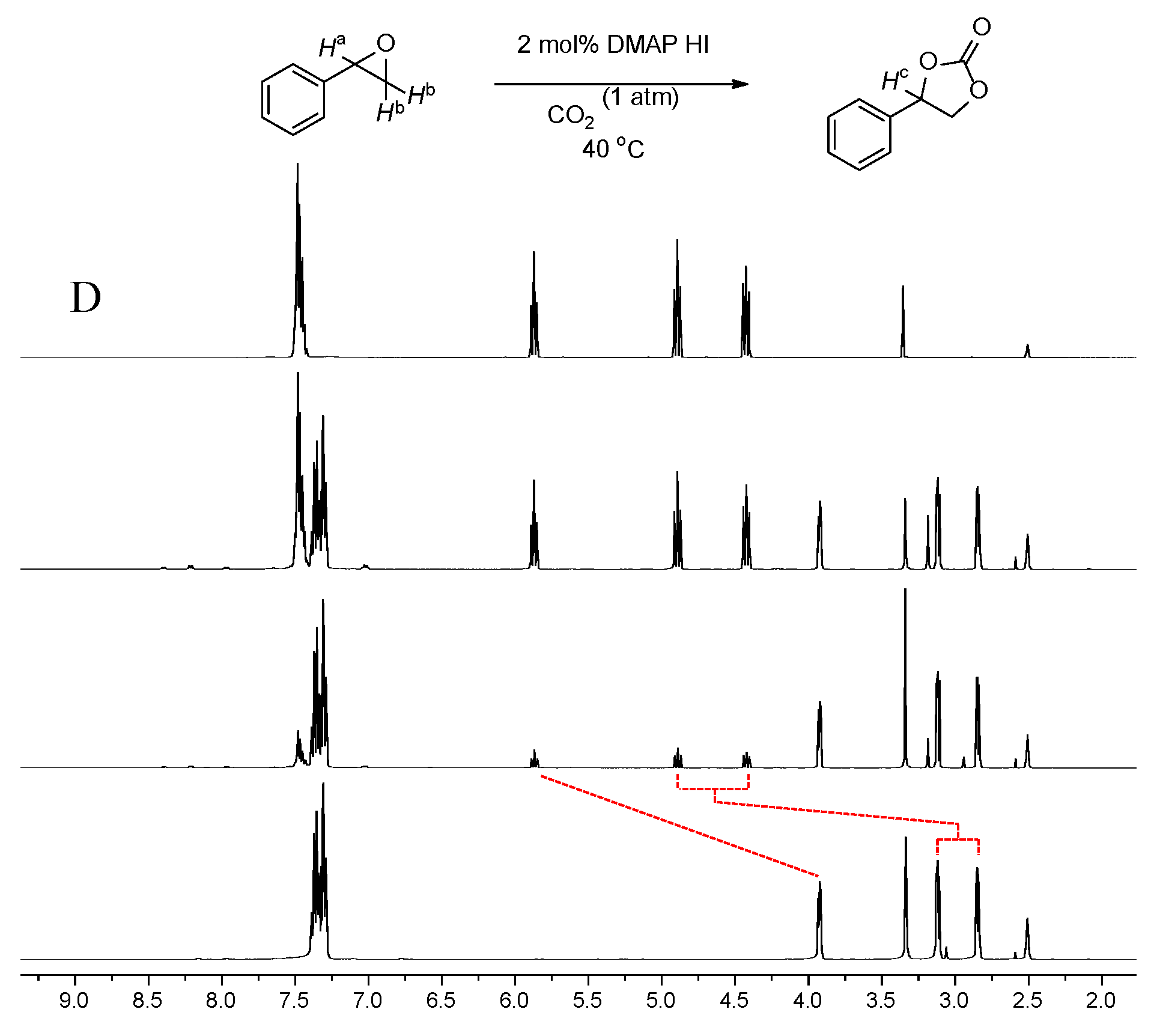
2.1. Model Reaction
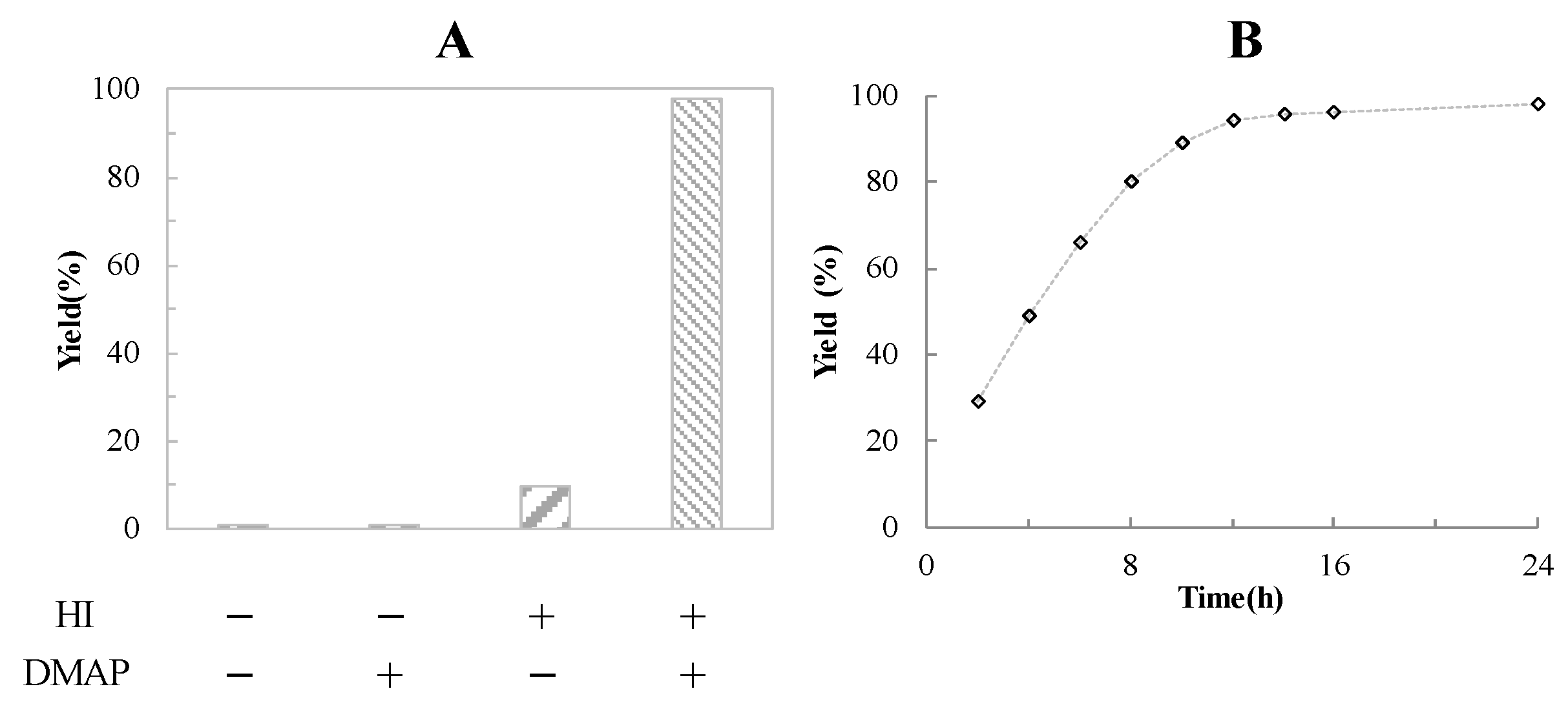
2.2. Counterion Effect
2.3. Analysis of the Catalyst System (DMAP-HI)
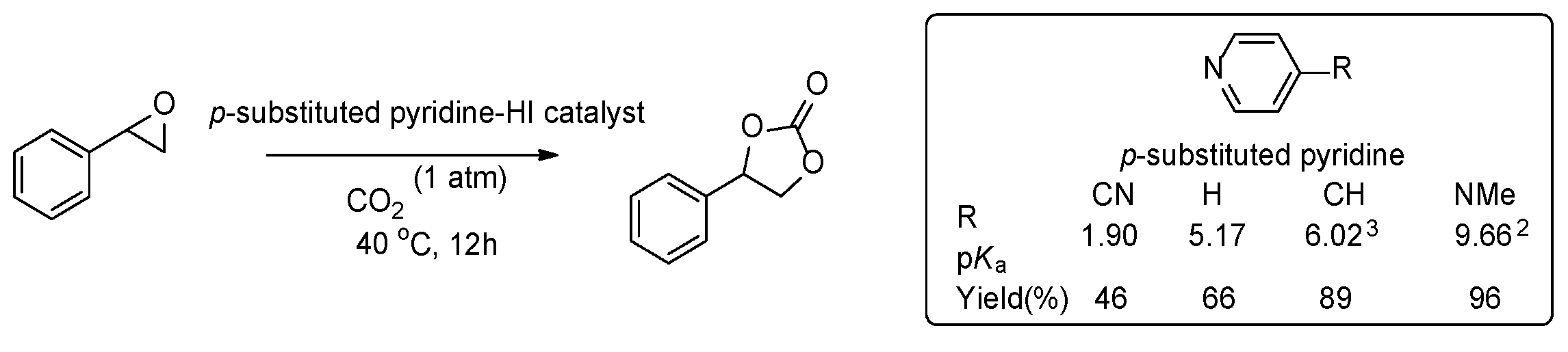
2.4. Structure and Activity Relationship of Catalyst: Electronic Effect
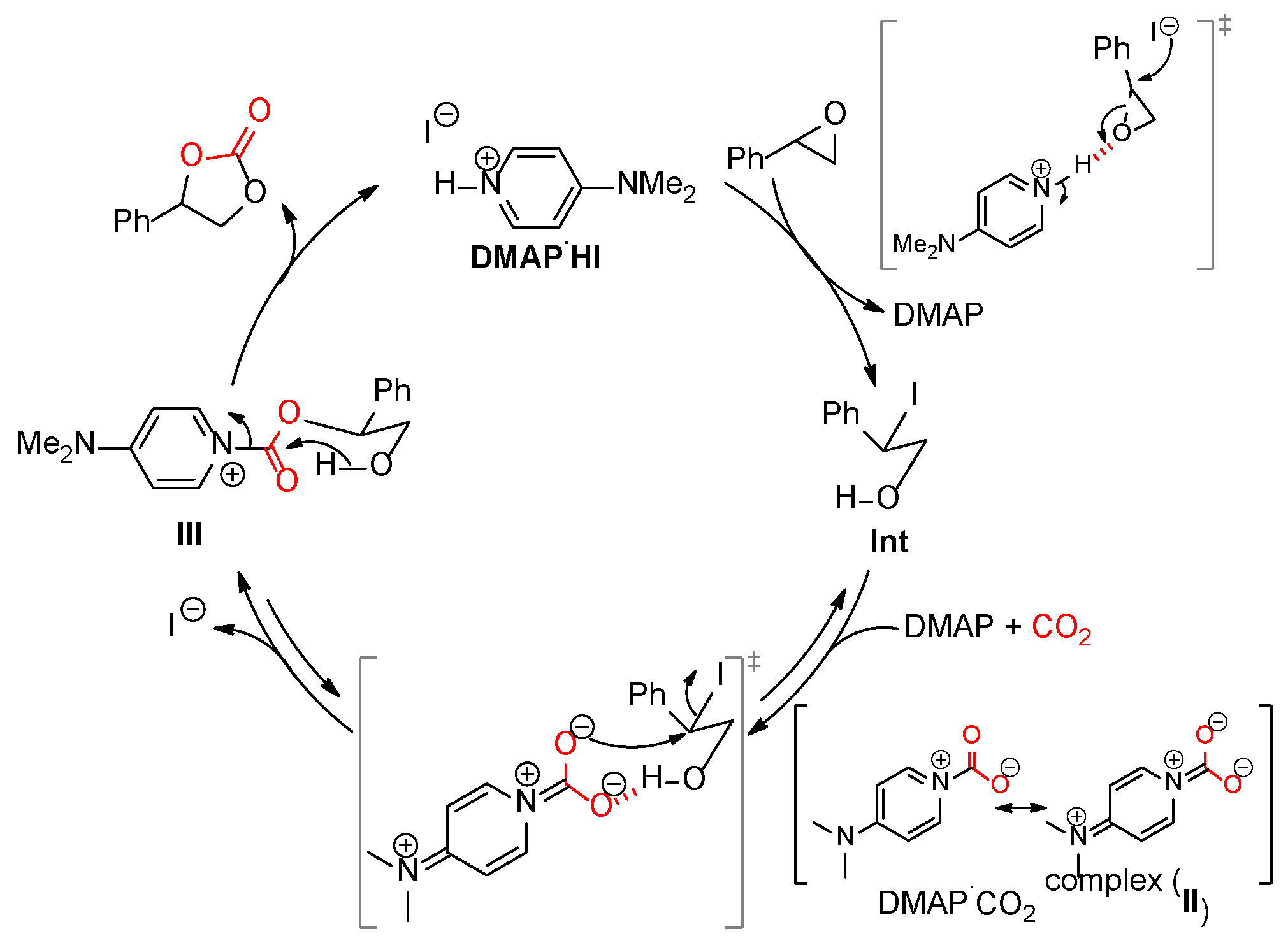
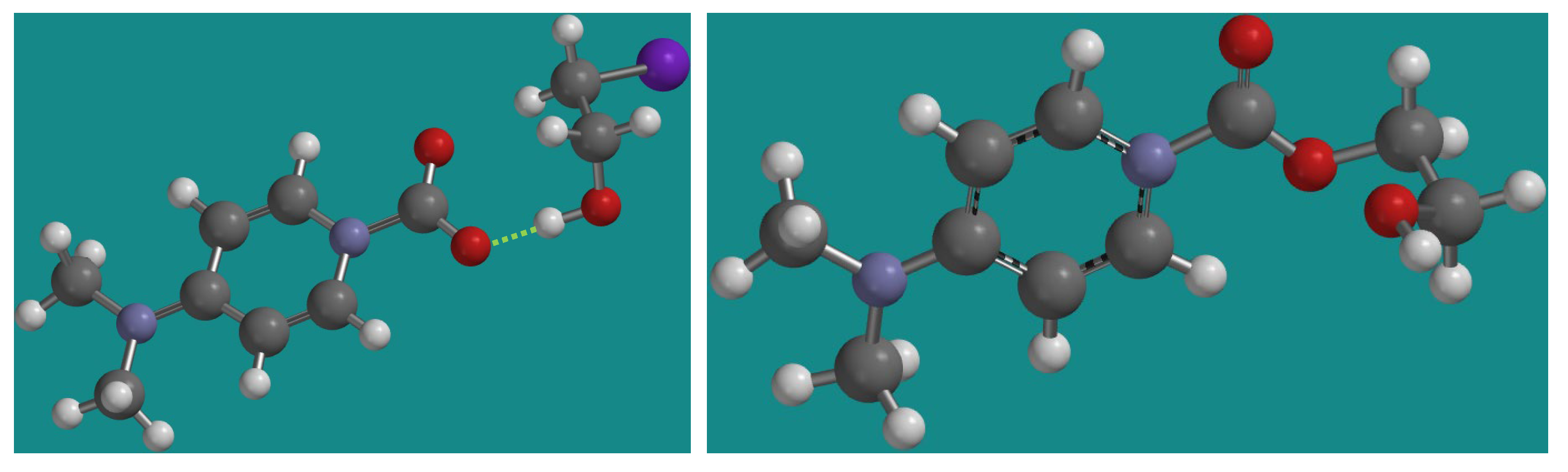
2.5. Proposed Mechanism
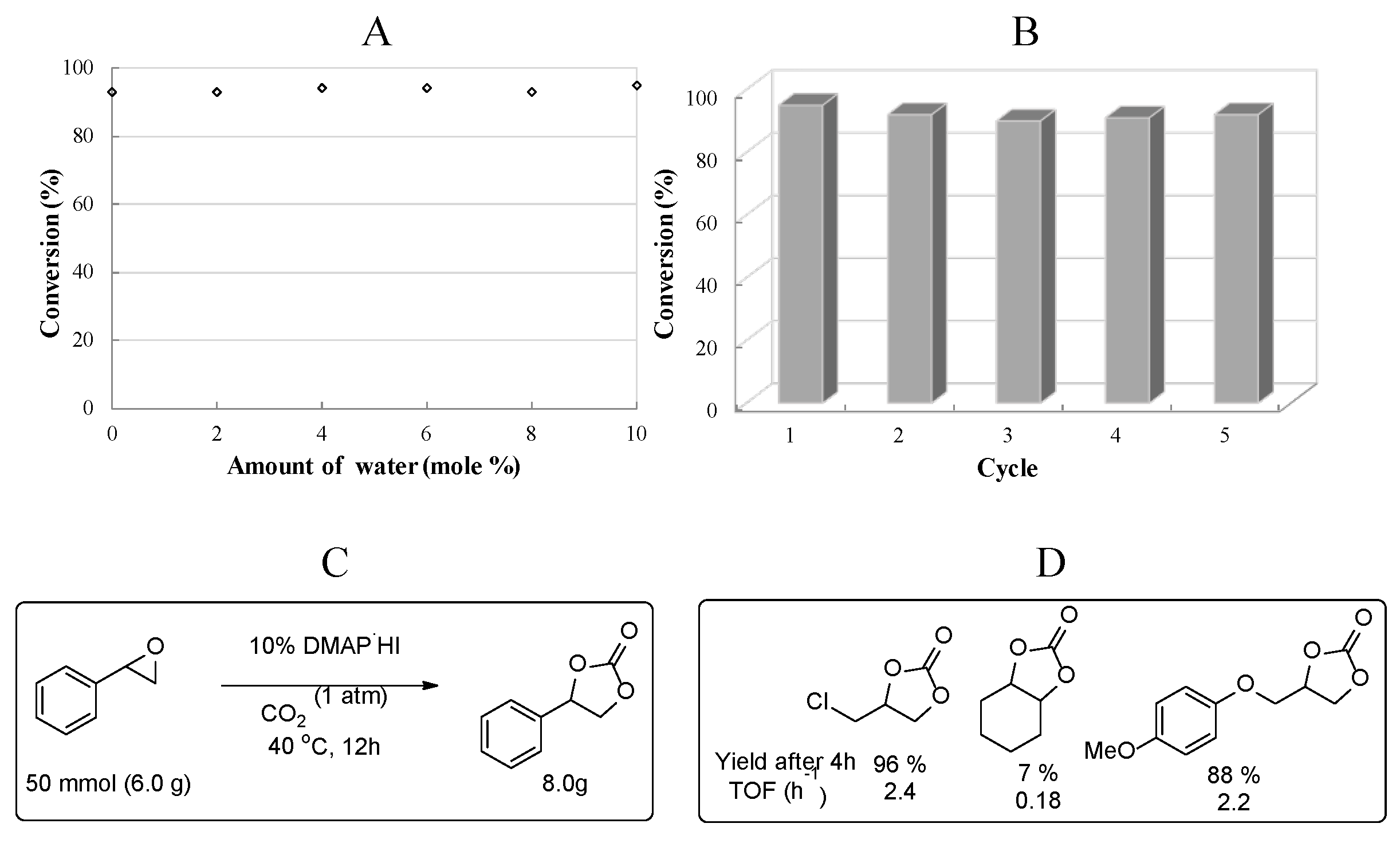
2.6. Practical Applicability
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CCU | Carbon dioxide capture and utilization |
| DMAP | Dimethylaminopyridine |
| NMR | Nuclear magnetic resonance |
References
- IPCC Fourth Assessment Report: Climate Change 2007, Cambridge University Press, Cambridge, 2007.
- Karl, T. R.; Trenberth, K. E. Modern Global Change. Science, 2003, 302, 1719–1723. [Google Scholar] [CrossRef] [PubMed]
- Lu, A. H.; Dai, S. (Eds.), Porous Materials for Carbon Dioxide Capture, Springer-Verlag, Berlin, 2014.
- Sumida, K.; Rogow, D. L.; Mason, J. A.; McDonald, T. M.; Bloch, E. D.; Herm, Z. R.; Bae, T.-H.; Long, J. R. Carbon dioxide capture in metal-organic frameworks. Chem. Rev., 2012, 112, 724–781. [Google Scholar] [CrossRef] [PubMed]
- Walton, K. S.; Millward, A. R.; Dubbeldam, D.; Frost, H.; Low, J. J.; Yaghi, O. M.; Snurr, R. Q. Understanding inflections and steps in carbon dioxide adsorption isotherms in metal-organic frameworks. J. Am. Chem. Soc., 2008, 130, 406–407. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.; Min, K.; Kim, C.; Ko, Y. S.; Jeon, J. W.; Seo, H.; Park, Y.-K.; Choi, M. Epoxide-functionalization of polyethyleneimine for synthesis of stable carbon dioxide adsorbent in temperature swing adsorption. Nat. Commun., 2016, 7, 12640. [Google Scholar] [CrossRef] [PubMed]
- McDonald, T. M.; Long, J. R.; et al. Cooperative insertion of CO2 in diamine-appended metal-organic frameworks. Nature, 2015, 519, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Goeppert, A.; Czaun, M.; May, R. B.; Prakash, G. K. S.; Olah, G. A.; Narayanan, S. R. Carbon dioxide capture from the air using a polyamine based regenerable solid adsorbent. J. Am. Chem. Soc. 2011, 133, 20164–20167. [Google Scholar] [CrossRef] [PubMed]
- Kim, S. H.; Kim, K. H.; Hong, S. H. arbon Dioxide Capture and Use: Organic Synthesis Using Carbon Dioxide from Exhaust Gas. Angew. Chem. Int. Ed., 2014, 53, 771–774. [Google Scholar] [CrossRef] [PubMed]
- Bruneau, C.; Dixneuf, P. H. Catalytic incorporation of CO2 into organic substrates: Synthesis of unsaturated carbamates, carbonates and ureas. J. Mol. Catal., 1992, 74, 97–107. [Google Scholar] [CrossRef]
- Liu, Q.; Wa, L.; Jackstell, R.; Beller, M. Using carbon dioxide as a building block in organic synthesis. Nat. Commun., 2015, 6, 5953. [Google Scholar] [CrossRef] [PubMed]
- Sujith, S.; Min, J. K.; Seong, J. E.; Na, S. J.; Lee, B. Y. A highly active and recyclable catalytic system for CO2/propylene oxide copolymerization. Angew. Chem. Int. Ed., 2008, 47, 7306–7309. [Google Scholar]
- North, M.; Pasquale, R. Mechanism of cyclic carbonate synthesis from epoxides and CO2. Angew. Chem. Int. Ed. 2009, 48, 2946–2948. [Google Scholar] [CrossRef] [PubMed]
- Decortes, A.; Kleij, A. W. Ambient Fixation of Carbon Dioxide using a Zn(II)-salphen Catalyst. ChemCatChem 2011, 3, 831–834. [Google Scholar] [CrossRef]
- Kumatabara, Y.; Okada, M.; Shirakawa, S. Triethylamine Hydroiodide as a Simple Yet Effective Bifunctional Catalyst for CO2 Fixation Reactions with Epoxides under Mild Conditions. ACS Sustainable Chem. Eng., 2017, 5, 7295–7301. [Google Scholar] [CrossRef]
- Hu, J.; Ma, J.; Liu, H.; Qian, Q.; Xie, C.; Han, B. Dual-ionic liquid system: an efficient catalyst for chemical fixation of CO2 to cyclic carbonates under mild conditions. Green Chem., 2018, 20, 2990–2994. [Google Scholar] [CrossRef]
- Zhang, Z.; Fan, F.; Xing, H.; Yang, Q.; Bao, Z.; Ren, Q. Efficient Synthesis of Cyclic Carbonates from Atmospheric CO2 Using a Positive Charge Delocalized Ionic Liquid Catalyst. ACS Sustainable Chem. Eng., 2017, 5, 2841–2846. [Google Scholar] [CrossRef]
- Clayden, J.; Greeves, N.; Warren, S. Organic Chemistry, 2nd ed.; Oxford: New York, USA, 2012; pp. 198–200. [Google Scholar]
- Cheng, W.; Su, Q.; Wang, J.; Sun, J.; Ng, F.T.T. Ionic Liquids: The Synergistic Catalytic Effect in the Synthesis of Cyclic Carbonates. Catalysts 2013, 3, 878–901. [Google Scholar] [CrossRef]
- Ma, J.; Song, J. L.; Liu, H. Z.; Liu, J. L.; Zhang, Z. F.; Jiang, T.; Fan, H. L.; Han, B. X. One-pot conversion of CO2 and glycerol to value-added products using propylene oxide as the coupling agent. Green Chem., 2012, 14, 1743–1748. [Google Scholar] [CrossRef]
- Jencks W. P.; Regenstein, J. Handbook of Biochemistry and Molecular Biology, 4th Ed. 2010.
- Bordwell, F. G. Equilibrium acidities in dimethyl sulfoxide solution. Acc. Chem. Res., 1988, 21, 456–463. [Google Scholar] [CrossRef]
- The role of pyridine as a base was excluded because we observed the reaction rate of 2,5-dimethylpyridien (lutidine, pKa 6.77) was much slower than those of 2-methyl (2-picoline, pKa 5.97) or 4-methylpyrdine (4-picoline, pKa 6.02). Lutidine (3.5%), 2-picoline (35%), and 4-picoline (89%).
- Clayden, J.; Greeves, N.; Warren, S. Organic Chemistry, 2nd ed.; Oxford: New York, USA, 2012; pp. 810–814. [Google Scholar]
- Clayden, J.; Greeves, N.; Warren, S. Organic Chemistry, 2nd ed.; Oxford: New York, USA, 2012; pp. 869–870. [Google Scholar]
| Entry | Catalyst (mol%) | Yield (%) | TOF (h-1) |
| 1 | – | 0 | – |
| 2 3 4 5 6 7 8 9 10 11 12 |
DMAP (10) HI (10) p-CNPy⸱HI (10) Py (10) ⸱HI p-CH3Py⸱HI (10) DMAP⸱HI (10) DMAP⸱HI (4) DMAP⸱HI (2) DMAP⸱HI (1) DMAP⸱HI (10) DMAP⸱HI (10) |
0 9.9 (8.3)b 46 66 89 96 57 32 17 61 c 46 d |
– 0.08 0.38 0.55 0.74 0.80 1.2 1.3 1.4 0.51 0.38 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




