Submitted:
27 March 2025
Posted:
28 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
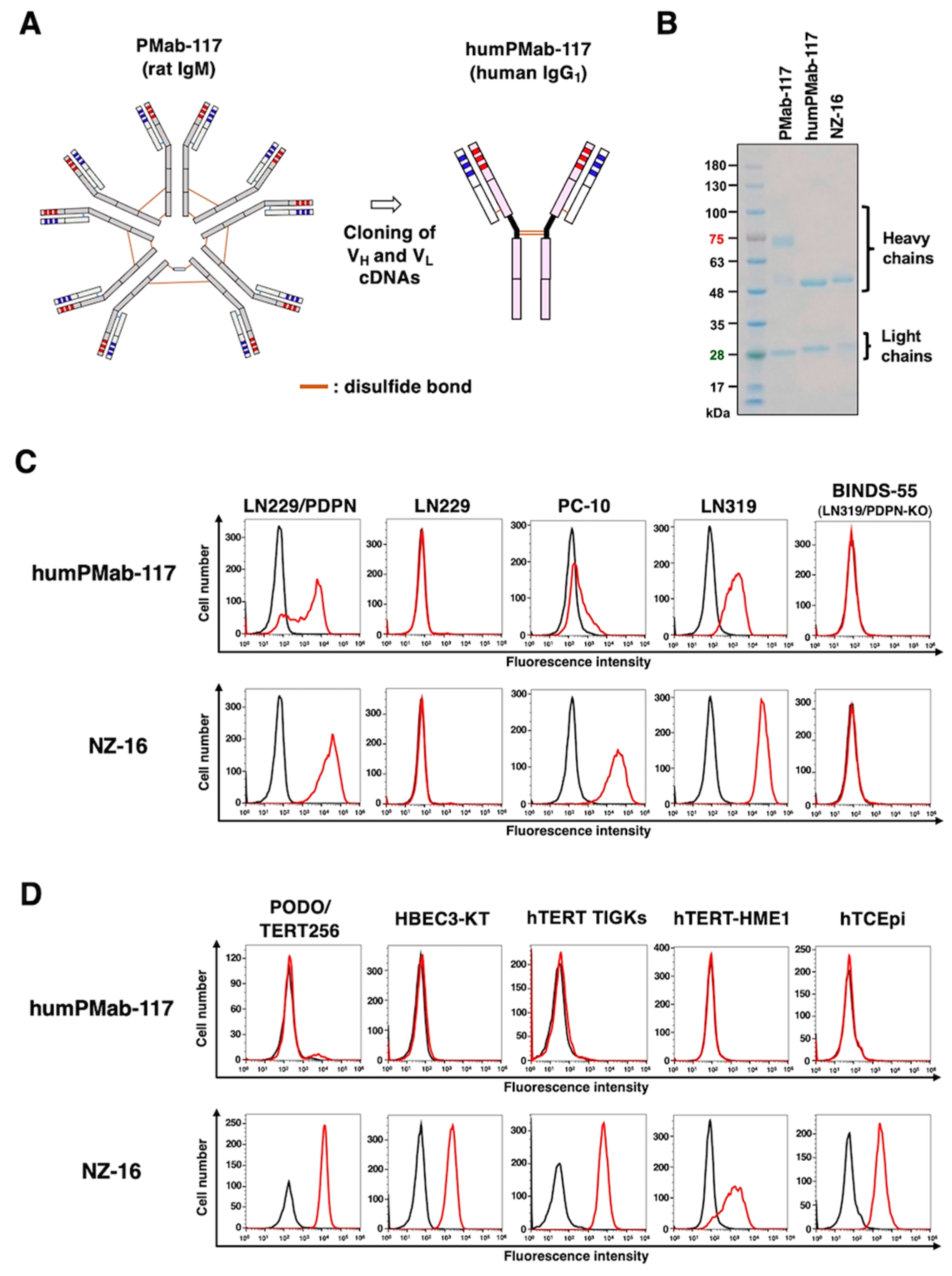
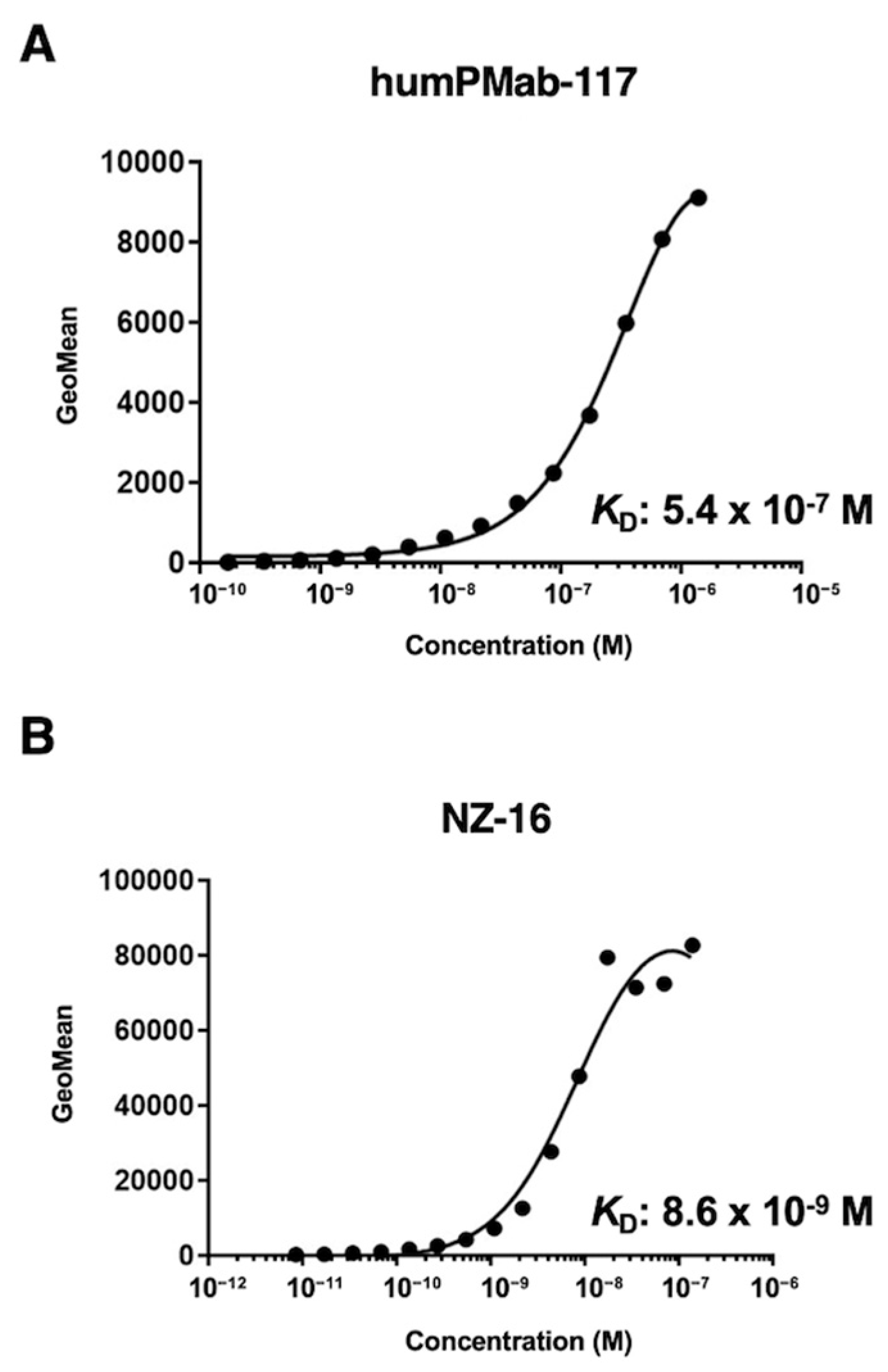
2.1. Production of Humanized Anti-PDPN mAb, humPMab-117
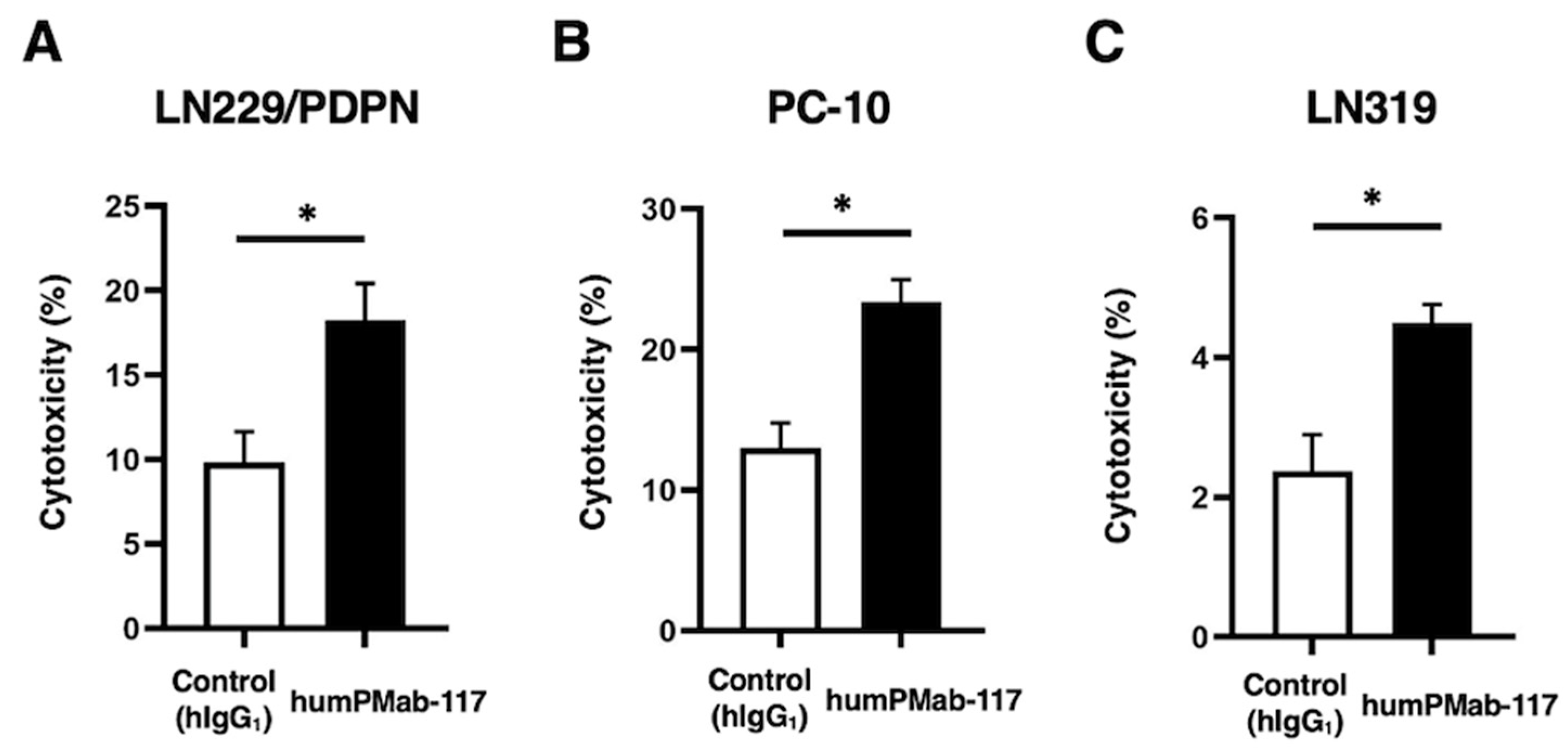
2.2. ADCC by humPMab-117 Against PDPN-Positive Cells
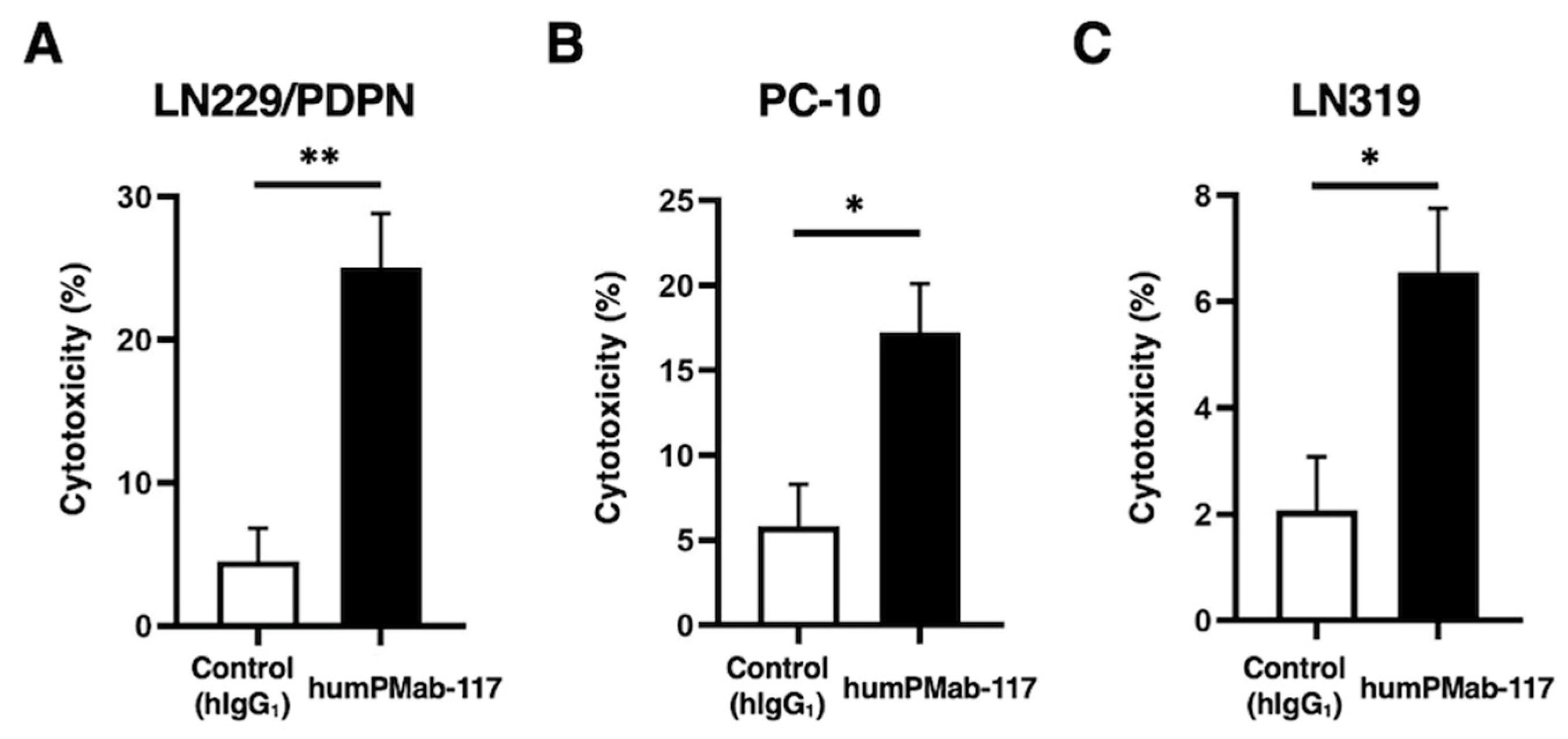
2.3. CDC by humPMab-117 Against PDPN-Positive Cells
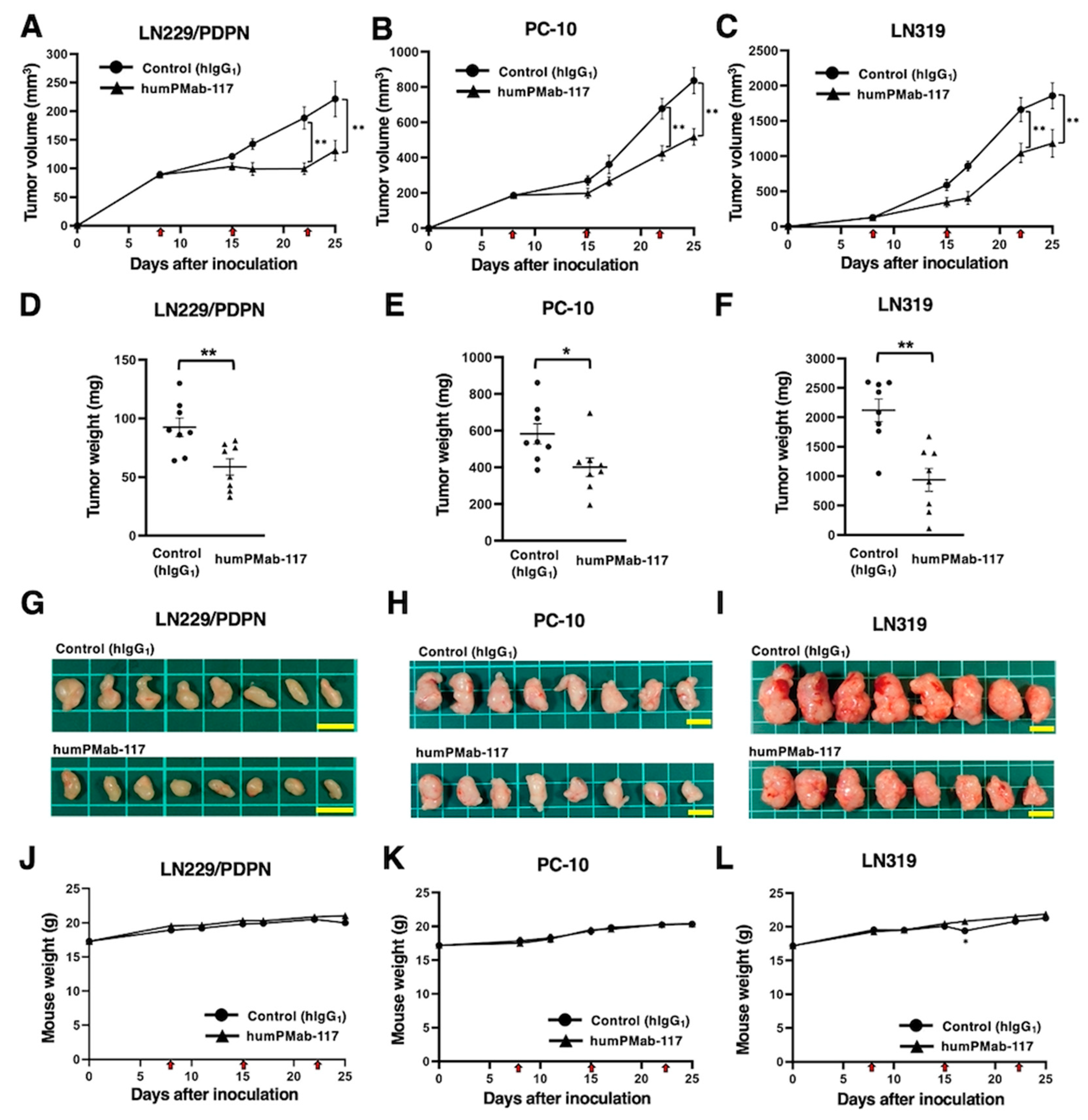
2.4. Antitumor Effects of humPMab-117 Against PDPN-Positive Cells in Mouse Xenograft Models
3. Discussion
4. Materials and Methods
4.1. Cell Lines
4.2. Animals
4.3. Antibodies
4.4. Flow Cytometry
4.5. ADCC
4.6. CDC
4.7. Antitumor Activities of humPMab-117 in Xenografts of Human Tumors
4.8. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Paul, S.; Konig, M.F.; Pardoll, D.M.; et al. Cancer therapy with antibodies. Nat Rev Cancer 2024;24(6): 399-426.
- Kaneko, M.K.; Suzuki, H.; Kato, Y. Establishment of a Novel Cancer-Specific Anti-HER2 Monoclonal Antibody H(2)Mab-250/H(2)CasMab-2 for Breast Cancers. Monoclon Antib Immunodiagn Immunother 2024;43(2): 35-43.
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. Antitumor Activities of a Humanized Cancer-Specific Anti-HER2 Monoclonal Antibody, humH(2)Mab-250 in Human Breast Cancer Xenografts. Int J Mol Sci 2025;26(3). [CrossRef]
- Suzuki, H.; Ohishi, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Anti-HER2 Cancer-Specific mAb, H(2)Mab-250-hG(1), Possesses Higher Complement-Dependent Cytotoxicity than Trastuzumab. Int J Mol Sci 2024;25(15). [CrossRef]
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. A Cancer-Specific Monoclonal Antibody against HER2 Exerts Antitumor Activities in Human Breast Cancer Xenograft Models. Int J Mol Sci 2024;25(3). [CrossRef]
- Hosking, M.; Shirinbak, S.; Omilusik, K.; et al. 268 Development of FT825/ONO-8250: an off-the-shelf CAR-T cell with preferential HER2 targeting and engineered to enable multi-antigen targeting, improve trafficking, and overcome immunosuppression. Journal for ImmunoTherapy of Cancer 2023;11(Suppl 1): A307-A307.
- Suzuki, H.; Kaneko, M.K.; Kato, Y. Roles of Podoplanin in Malignant Progression of Tumor. Cells 2022;11(3). [CrossRef]
- Quintanilla, M.; Montero-Montero, L.; Renart, J.; Martín-Villar, E. Podoplanin in Inflammation and Cancer. Int J Mol Sci 2019;20(3).
- Kato, Y.; Fujita, N.; Kunita, A.; et al. Molecular identification of Aggrus/T1alpha as a platelet aggregation-inducing factor expressed in colorectal tumors. J Biol Chem 2003;278(51): 51599-51605. [CrossRef]
- Kaneko, M.K.; Kato, Y.; Kameyama, A.; et al. Functional glycosylation of human podoplanin: glycan structure of platelet aggregation-inducing factor. FEBS Lett 2007;581(2): 331-336. [CrossRef]
- Sekiguchi, T.; Takemoto, A.; Takagi, S.; et al. Targeting a novel domain in podoplanin for inhibiting platelet-mediated tumor metastasis. Oncotarget 2016;7(4): 3934-3946. [CrossRef]
- Pandya, P.; Orgaz, J.L.; Sanz-Moreno, V. Modes of invasion during tumour dissemination. Mol Oncol 2017;11(1): 5-27. [CrossRef]
- Lambert, A.W.; Weinberg, R.A. Linking EMT programmes to normal and neoplastic epithelial stem cells. Nat Rev Cancer 2021;21(5): 325-338. [CrossRef]
- Astarita, J.L.; Acton, S.E.; Turley, S.J. Podoplanin: emerging functions in development, the immune system, and cancer. Front Immunol 2012;3: 283. [CrossRef]
- Wicki, A.; Lehembre, F.; Wick, N.; et al. Tumor invasion in the absence of epithelial-mesenchymal transition: podoplanin-mediated remodeling of the actin cytoskeleton. Cancer Cell 2006;9(4): 261-272. [CrossRef]
- de Winde, C.M.; George, S.L.; Crosas-Molist, E.; et al. Podoplanin drives dedifferentiation and amoeboid invasion of melanoma. iScience 2021;24(9): 102976. [CrossRef]
- Martín-Villar, E.; Megías, D.; Castel, S.; et al. Podoplanin binds ERM proteins to activate RhoA and promote epithelial-mesenchymal transition. J Cell Sci 2006;119(Pt 21): 4541-4553. [CrossRef]
- Pecora, A.; Laprise, J.; Dahmene, M.; Laurin, M. Skin Cancers and the Contribution of Rho GTPase Signaling Networks to Their Progression. Cancers (Basel) 2021;13(17). [CrossRef]
- Zhang, Z.; Liu, M.; Zheng, Y. Role of Rho GTPases in stem cell regulation. Biochem Soc Trans 2021. [CrossRef]
- Li, Y.Y.; Zhou, C.X.; Gao, Y. Podoplanin promotes the invasion of oral squamous cell carcinoma in coordination with MT1-MMP and Rho GTPases. Am J Cancer Res 2015;5(2): 514-529.
- Martín-Villar, E.; Fernández-Muñoz, B.; Parsons, M.; et al. Podoplanin associates with CD44 to promote directional cell migration. Mol Biol Cell 2010;21(24): 4387-4399. [CrossRef]
- Friedman, G.; Levi-Galibov, O.; David, E.; et al. Cancer-associated fibroblast compositions change with breast cancer progression linking the ratio of S100A4(+) and PDPN(+) CAFs to clinical outcome. Nat Cancer 2020;1(7): 692-708. [CrossRef]
- Hirayama, K.; Kono, H.; Nakata, Y.; et al. Expression of podoplanin in stromal fibroblasts plays a pivotal role in the prognosis of patients with pancreatic cancer. Surg Today 2018;48(1): 110-118. [CrossRef]
- Liu, X.; Cao, Y.; Lv, K.; et al. Tumor-infiltrating podoplanin(+) cells in gastric cancer: clinical outcomes and association with immune contexture. Oncoimmunology 2020;9(1): 1845038. [CrossRef]
- Wang, H.; Hu, C.; Song, X.; et al. Expression of Podoplanin in Sinonasal Squamous Cell Carcinoma and Its Clinical Significance. Am J Rhinol Allergy 2020;34(6): 800-809. [CrossRef]
- Ramirez, M.I.; Millien, G.; Hinds, A.; et al. T1alpha, a lung type I cell differentiation gene, is required for normal lung cell proliferation and alveolus formation at birth. Dev Biol 2003;256(1): 61-72. [CrossRef]
- Koop, K.; Eikmans, M.; Wehland, M.; et al. Selective loss of podoplanin protein expression accompanies proteinuria and precedes alterations in podocyte morphology in a spontaneous proteinuric rat model. Am J Pathol 2008;173(2): 315-326. [CrossRef]
- Ijpelaar, D.H.; Schulz, A.; Koop, K.; et al. Glomerular hypertrophy precedes albuminuria and segmental loss of podoplanin in podocytes in Munich-Wistar-Frömter rats. Am J Physiol Renal Physiol 2008;294(4): F758-767. [CrossRef]
- Kato, Y.; Kaneko, M.K. A cancer-specific monoclonal antibody recognizes the aberrantly glycosylated podoplanin. Sci Rep 2014;4: 5924. [CrossRef]
- Yamada, S.; Ogasawara, S.; Kaneko, M.K.; Kato, Y. LpMab-23: A Cancer-Specific Monoclonal Antibody Against Human Podoplanin. Monoclon Antib Immunodiagn Immunother 2017;36(2): 72-76. [CrossRef]
- Kaneko, M.K.; Nakamura, T.; Kunita, A.; et al. ChLpMab-23: Cancer-Specific Human-Mouse Chimeric Anti-Podoplanin Antibody Exhibits Antitumor Activity via Antibody-Dependent Cellular Cytotoxicity. Monoclon Antib Immunodiagn Immunother 2017;36(3): 104-112. [CrossRef]
- Kaneko, M.K.; Yamada, S.; Nakamura, T.; et al. Antitumor activity of chLpMab-2, a human-mouse chimeric cancer-specific antihuman podoplanin antibody, via antibody-dependent cellular cytotoxicity. Cancer Med 2017;6(4): 768-777.
- Tanaka, T.; Suzuki, H.; Ohishi, T.; Kaneko, M.K.; Kato, Y. A Cancer-Specific Anti-Podoplanin Monoclonal Antibody, PMab-117-mG(2a) Exerts Antitumor Activities in Human Tumor Xenograft Models. Cells 2024;13(22).
- Sudo, H.; Tsuji, A.B.; Sugyo, A.; et al. Preclinical Evaluation of Podoplanin-Targeted Alpha-Radioimmunotherapy with the Novel Antibody NZ-16 for Malignant Mesothelioma. Cells 2021;10(10). [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kunita, A.; et al. Molecular analysis of the pathophysiological binding of the platelet aggregation-inducing factor podoplanin to the C-type lectin-like receptor CLEC-2. Cancer Sci 2008;99(1): 54-61. [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kuno, A.; et al. Inhibition of tumor cell-induced platelet aggregation using a novel anti-podoplanin antibody reacting with its platelet-aggregation-stimulating domain. Biochem Biophys Res Commun 2006;349(4): 1301-1307. [CrossRef]
- Takemoto, A.; Takagi, S.; Ukaji, T.; et al. Targeting Podoplanin for the Treatment of Osteosarcoma. Clin Cancer Res 2022;28(12): 2633-2645. [CrossRef]
- Suzuki, H.; Ohishi, T.; Kaneko, M.K.; Kato, Y. A Humanized and Defucosylated Antibody against Podoplanin (humLpMab-23-f) Exerts Antitumor Activities in Human Lung Cancer and Glioblastoma Xenograft Models. Cancers (Basel) 2023;15(20). [CrossRef]
- Zhou, Y.; Tao, L.; Qiu, J.; et al. Tumor biomarkers for diagnosis, prognosis and targeted therapy. Signal Transduct Target Ther 2024;9(1): 132.
- First-Ever CAR T-cell Therapy Approved in U.S. Cancer Discov 2017;7(10): Of1.
- Kakarla, S.; Gottschalk, S. CAR T cells for solid tumors: armed and ready to go? Cancer J 2014;20(2): 151-155.
- Feng, K.; Guo, Y.; Dai, H.; et al. Chimeric antigen receptor-modified T cells for the immunotherapy of patients with EGFR-expressing advanced relapsed/refractory non-small cell lung cancer. Sci China Life Sci 2016;59(5): 468-479. [CrossRef]
- Ahmed, N.; Brawley, V.; Hegde, M.; et al. HER2-Specific Chimeric Antigen Receptor-Modified Virus-Specific T Cells for Progressive Glioblastoma: A Phase 1 Dose-Escalation Trial. JAMA Oncol 2017;3(8): 1094-1101.
- Mei, Z.; Zhang, K.; Lam, A.K.; et al. MUC1 as a target for CAR-T therapy in head and neck squamous cell carinoma. Cancer Med 2020;9(2): 640-652. [CrossRef]
- Chalise, L.; Kato, A.; Ohno, M.; et al. Efficacy of cancer-specific anti-podoplanin CAR-T cells and oncolytic herpes virus G47Δ combination therapy against glioblastoma. Mol Ther Oncolytics 2022;26: 265-274. [CrossRef]
- Shiina, S.; Ohno, M.; Ohka, F.; et al. CAR T Cells Targeting Podoplanin Reduce Orthotopic Glioblastomas in Mouse Brains. Cancer Immunol Res 2016;4(3): 259-268. [CrossRef]
- Castellarin, M.; Sands, C.; Da, T.; et al. A rational mouse model to detect on-target, off-tumor CAR T cell toxicity. JCI Insight 2020;5(14). [CrossRef]
- Ghorashian, S.; Kramer, A.M.; Onuoha, S.; et al. Enhanced CAR T cell expansion and prolonged persistence in pediatric patients with ALL treated with a low-affinity CD19 CAR. Nat Med 2019;25(9): 1408-1414. [CrossRef]
- Arcangeli, S.; Rotiroti, M.C.; Bardelli, M.; et al. Balance of Anti-CD123 Chimeric Antigen Receptor Binding Affinity and Density for the Targeting of Acute Myeloid Leukemia. Mol Ther 2017;25(8): 1933-1945. [CrossRef]
- Olson, M.L.; Mause, E.R.V.; Radhakrishnan, S.V.; et al. Low-affinity CAR T cells exhibit reduced trogocytosis, preventing rapid antigen loss, and increasing CAR T cell expansion. Leukemia 2022;36(7): 1943-1946. [CrossRef]
- Caraballo Galva, L.D.; Jiang, X.; Hussein, M.S.; et al. Novel low-avidity glypican-3 specific CARTs resist exhaustion and mediate durable antitumor effects against HCC. Hepatology 2022;76(2): 330-344. [CrossRef]
- Hamieh, M.; Dobrin, A.; Cabriolu, A.; et al. CAR T cell trogocytosis and cooperative killing regulate tumour antigen escape. Nature 2019;568(7750): 112-116. [CrossRef]
- Suzuki, H.; Ohishi, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. A Cancer-Specific Monoclonal Antibody against Podocalyxin Exerted Antitumor Activities in Pancreatic Cancer Xenografts. Int J Mol Sci 2023;25(1). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



