Submitted:
26 March 2025
Posted:
27 March 2025
You are already at the latest version
Abstract
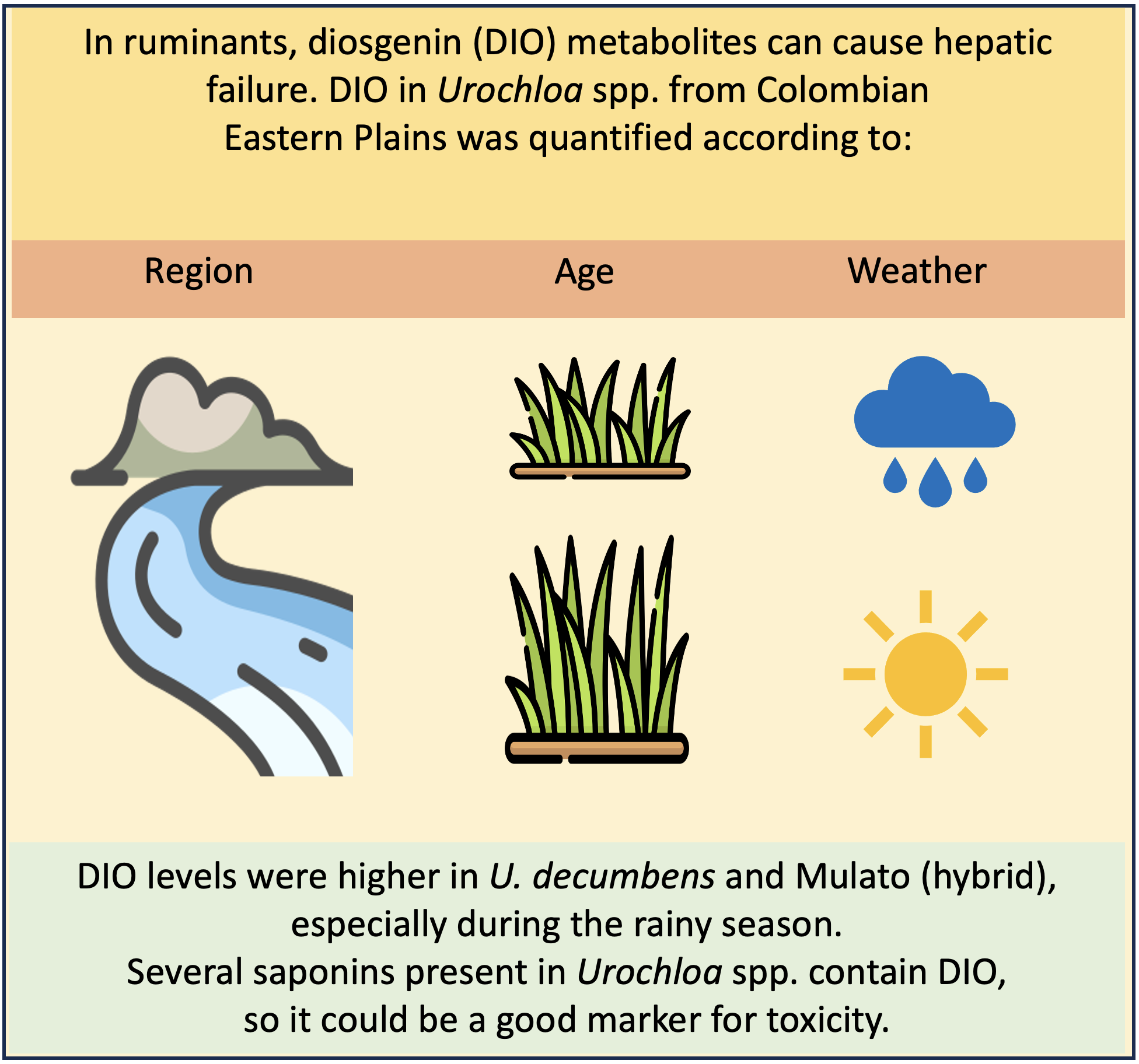
Keywords:
1. Introduction
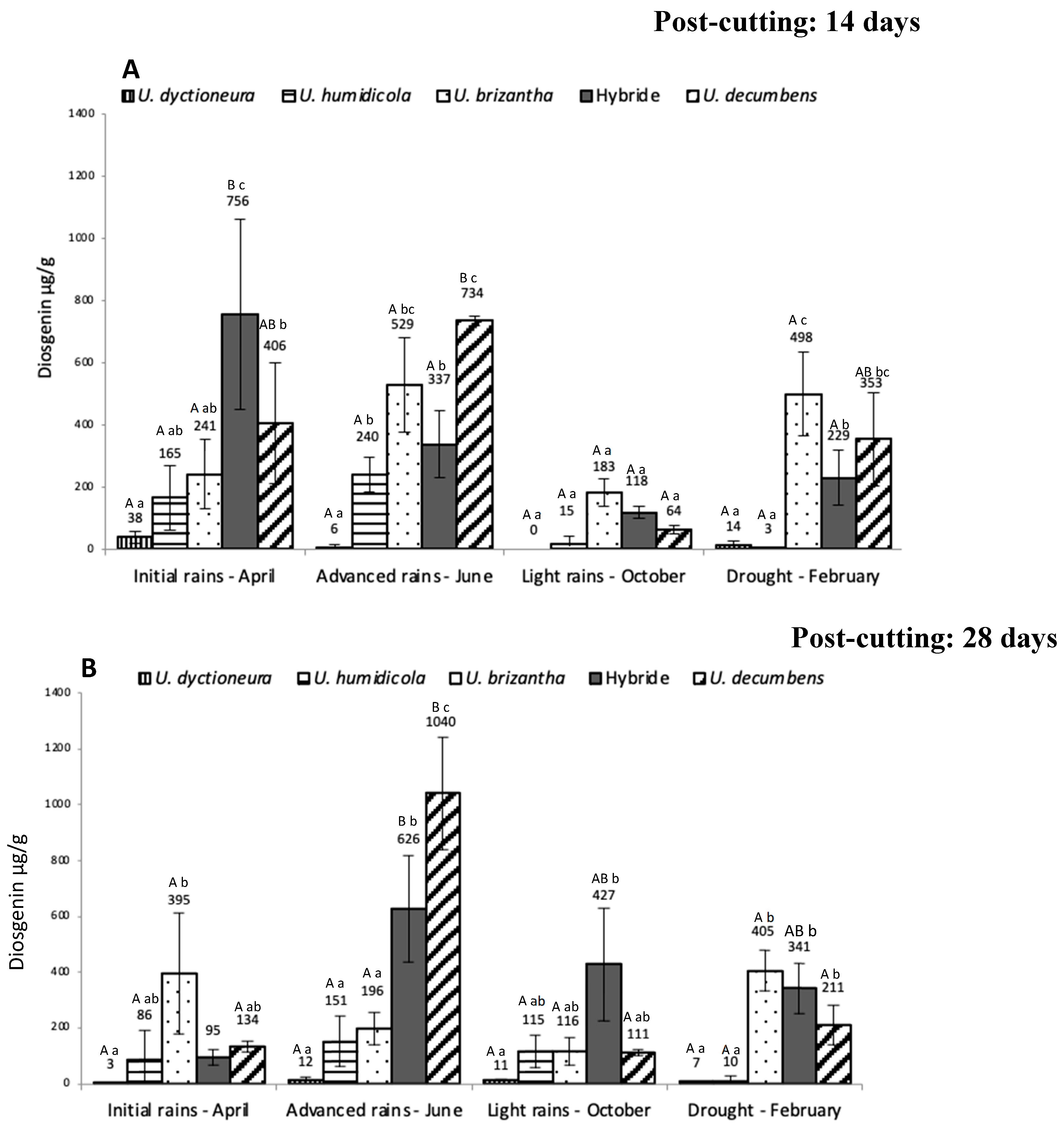
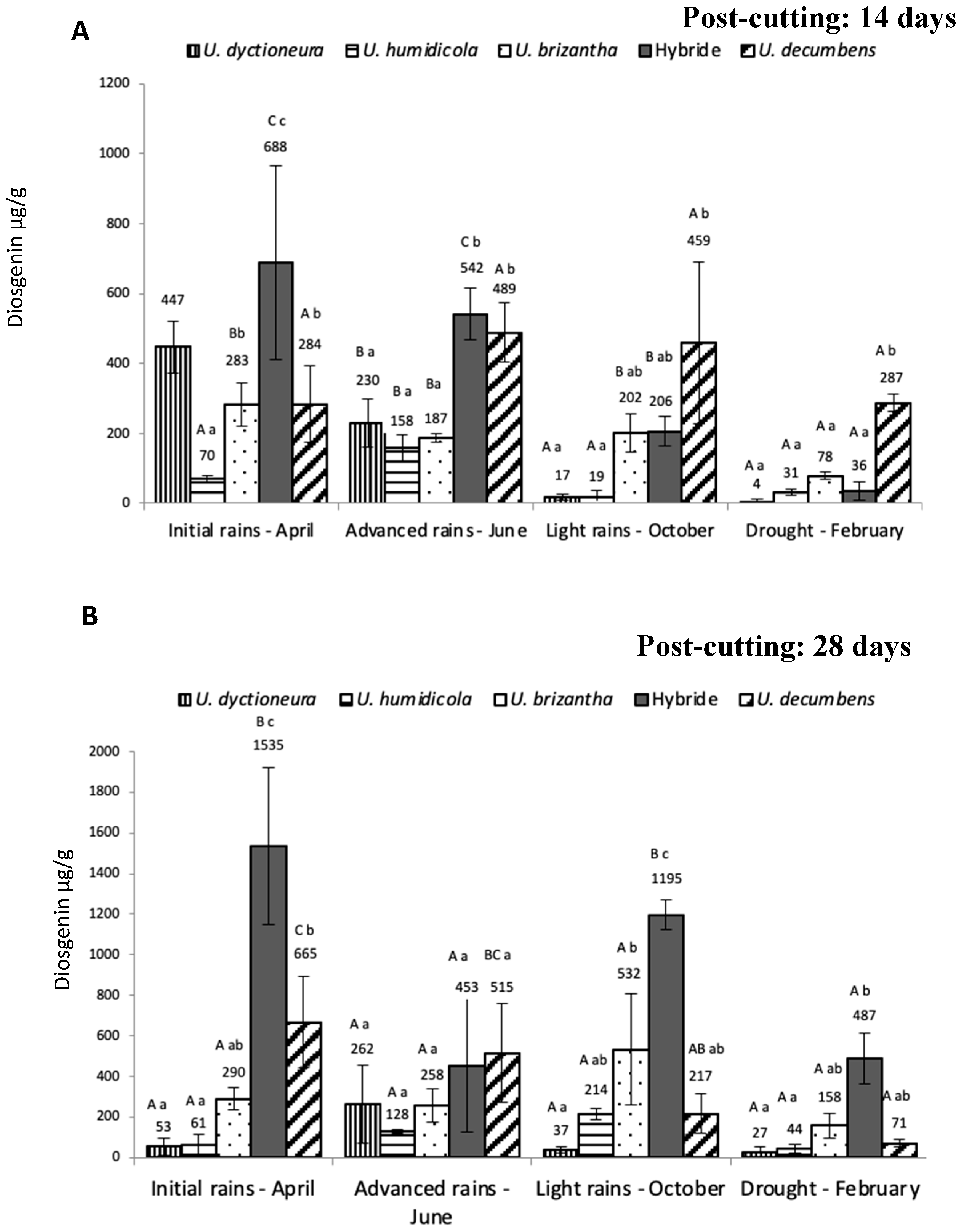
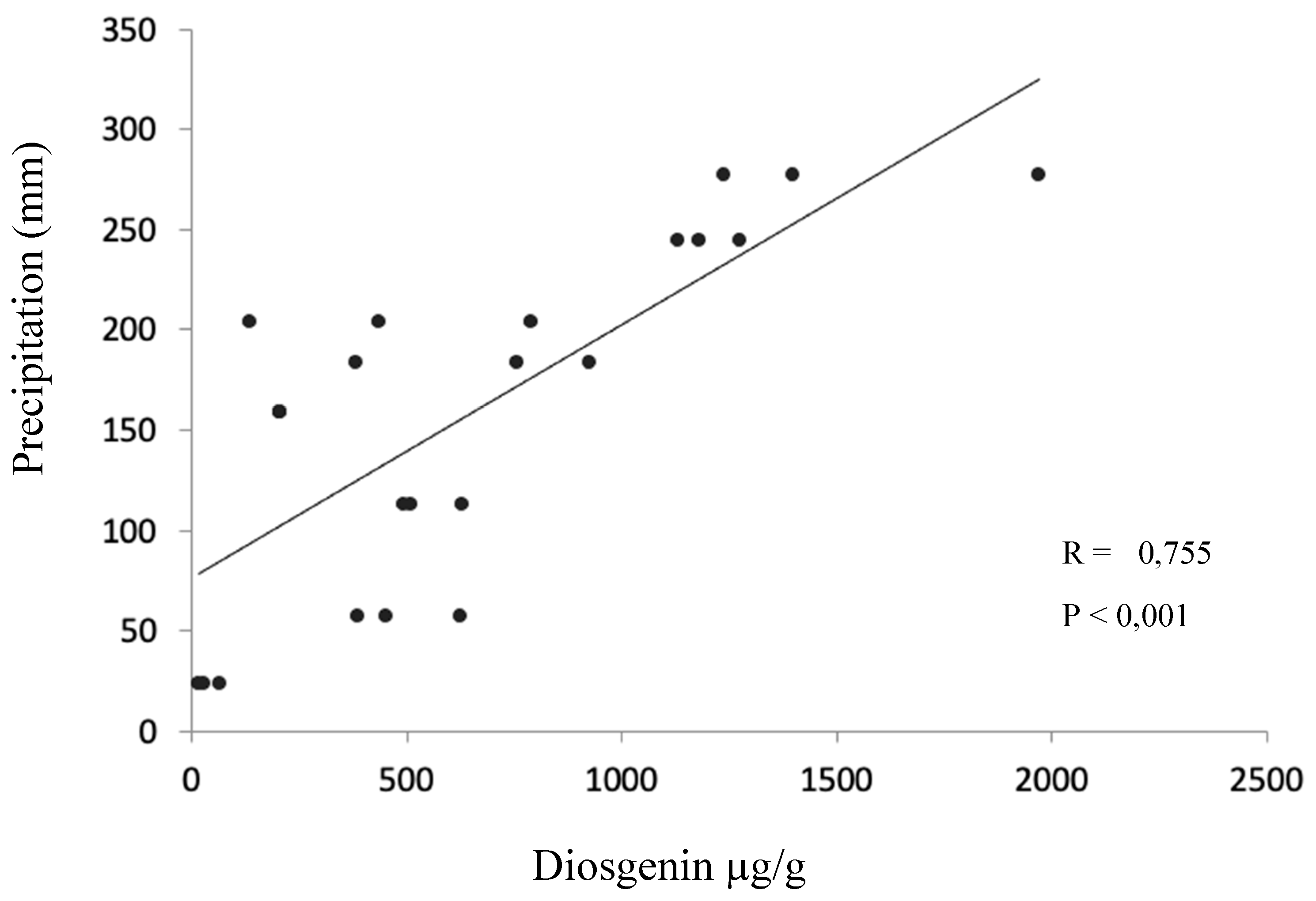
2. Results
3. Discussion
4. Materials and Methods
4.1. Region of Study
4.2. Sampling
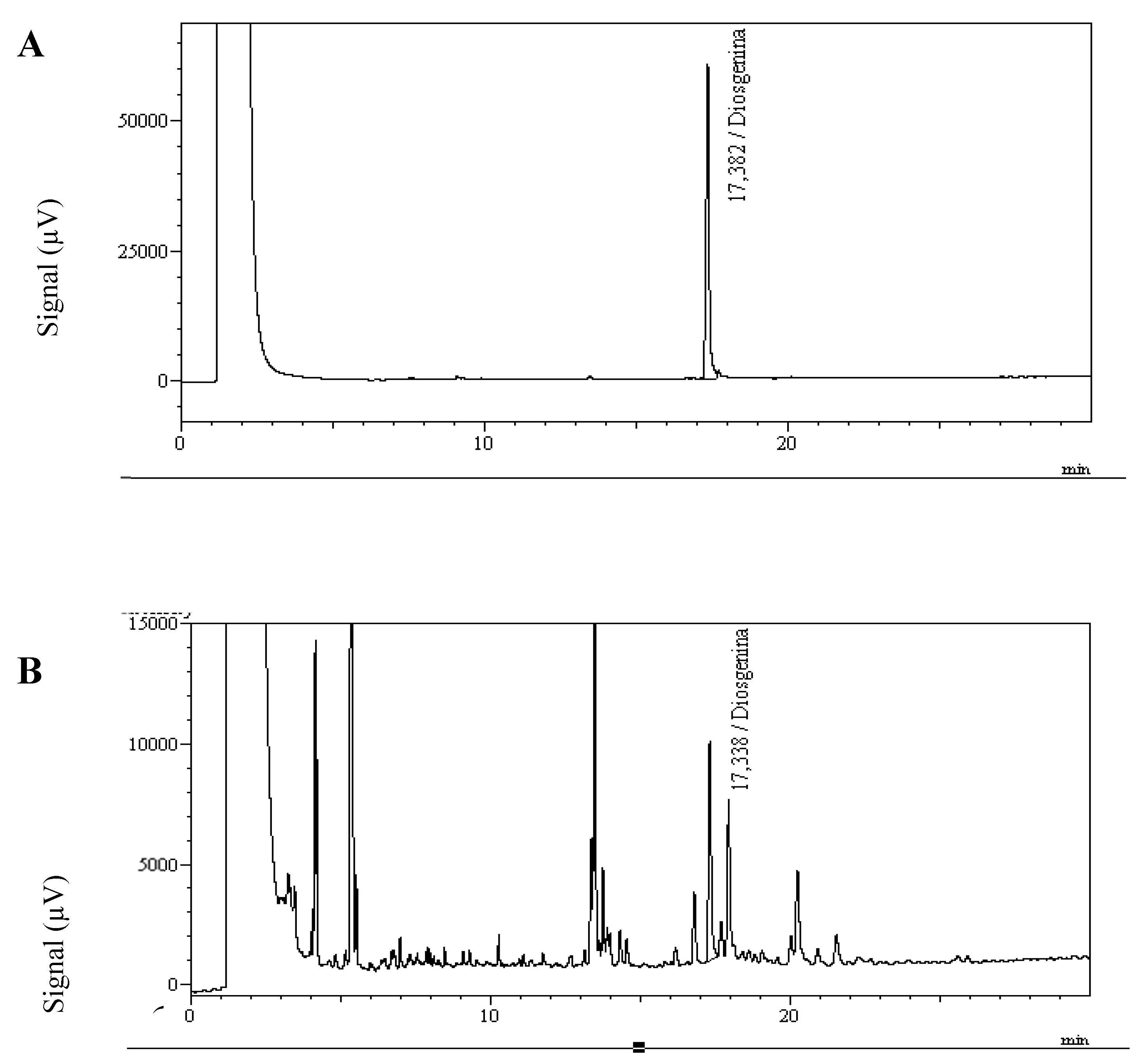
4.3. Determination and Quantitation of Diosgenin
4.4. Statistical Analyses
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- ICA. Censos Pecuarios Nacional 2024; ICA: Bogotá, Colombia. Available online: https://www.ica.gov.co/areas/pecuaria/servicios/epidemiologia-veterinaria/censos-2016/censo-2018 (accessed on 23 March 2025).
- Lozano, M. C.; Doncel, B.; Moreno, C. A. Manual de plantas tóxicas para bovinos. Región Llanos Orientales de Colombia: Meta y Casanare; Universidad Nacional de Colombia: Bogotá, Colombia, 2011. [Google Scholar]
- Díaz, G. J. Plantas tóxicas de importancia en salud y producción animal en Colombia; Universidad Nacional de Colombia: Bogotá, Colombia, 2010. [Google Scholar]
- Cruz, C.; Driemeier, D.; Pires, V.; Colodel, E.; Daketa, A. Isolation of steroidal sapogenins implicated in experimentally induced cholangiopathy of sheep grazing Brachiaria decumbens in Brazil. Vet. Hum. Toxicol. 2000, 42, 142–145. [Google Scholar]
- Cruz, C.; Driemeier, D.; Pires, V.; Schenkel, E. Experimentally induced cholangiohepatopathy by dosing sheep with fractionated extracts from Brachiaria decumbens. J. Vet. Diagn. Invest. 2001, 13, 170–172. [Google Scholar] [CrossRef]
- Brum, K.; Haraguchi, M.; Lemos, R.; Riet-Correa, F.; Fioravanti, M. Crystal-associated cholangiopathy in sheep grazing Brachiaria decumbens containing the saponin protodioscin. Pesq. Vet. Bras. 2007, 27, 39–42. [Google Scholar] [CrossRef]
- Miles, C.; Wilkins, A.; Munday, S.; Holland, P.; Smith, B.; Lancaster, M. Identification of the calcium salt of epismilagenin β-D-glucuronide in the bile crystals of sheep affected by Panicum dichotomiflorum and Panicum schinzii toxicoses. J. Agric. Food Chem. 1992, 40, 1606–1609. [Google Scholar] [CrossRef]
- Miles, C.; Wilkins, A.; Munday, S.; Flayoen, A.; Holland, P.; Smith, B. Identification of insoluble salts of the β-D-glucuronides of episarsasapogenin and epismilagenin in the bile of lambs with alveld and examination of Narthecium ossifragum, Tribulus terrestris, and Panicum miliaceum for sapogenins. J. Agric. Food Chem. 1993, 41, 914–917. [Google Scholar] [CrossRef]
- Mysterud, I.; Flåøyen, A.; Loader, J. I.; Wilkins, A. L. Sapogenin levels in Narthecium ossifragum plants and Ovis aries lamb feces during two alveld outbreaks in Møre og Romsdal, Norway, 2001. Vet. Res. Commun. 2007, 31, 895–908. [Google Scholar] [CrossRef]
- Riet-Correa, B.; Castro, M. B.; De Lemos, R. A.; Riet-Correa, G.; Mustafa, V.; Riet-Correa, F.; Lemos, R. Brachiaria spp. poisoning of ruminants in Brazil. Pesq. Vet. Bras. 2011, 31, 183–192. [Google Scholar] [CrossRef]
- Lima, F. G.; Haraguchi, M.; Pfister, J. A.; Guimaraes, V. Y.; Diogo, D. F. Weather and plant age affect the levels of steroidal saponin and Pithomyces chartarum spores in Brachiaria grass, main forage source for ruminants. IJPPR. 2012, 2, 45–53. [Google Scholar]
- Lozano, M. C.; Martinez, N. M.; Diaz, G. J. Content of the Saponin Protodioscin in Brachiaria spp. from the Eastern Plains of Colombia. Toxins 2017, 9, 220. [Google Scholar] [CrossRef] [PubMed]
- da Costa, M. C. M.; Ítavo, L. C. V.; Ítavo, C. C. B. F.; Dias, A. M.; Dos Santos Difante, G.; Buschinelli de Goes, R. H. T.; de Souza Leal, E.; Nonato, L. M.; Kozerski, N. D.; de Moraes, G. J.; Niwa, M. V. G.; Gurgel, A. L. C.; de Souza Arco, T. F. F. Natural Intoxication Caused by Protodioscin in Lambs Kept in Brachiaria Pastures. Trop. Anim. Health Prod. 2021, 53, 336. [Google Scholar] [CrossRef] [PubMed]
- Torres, P.; Diaz, G. J.; Cárdenas, E.; Lozano, M. C. Ethnobotanical study of plants poisonous to cattle in Eastern Colombia. IJPPR. 2002, 2, 14–19. [Google Scholar]
- Gracindo, C. V.; Louvandini, H.; Riet-Correa, F.; Barbosa-Ferreira, M.; Botelho De Castro, M. Performance of sheep grazing in pastures of Brachiaria decumbens, Brachiaria brizantha, Panicum maximum, and Andropogon gayanus with different protodioscin concentrations. Trop. Anim. Health Prod. 2014, 46, 733–737. [Google Scholar] [CrossRef]
- Santana, J. C. S.; Difante, G. D. S.; Rodrigues, J. G.; Pereira, M. G.; Fernandes, H. J.; Ítavo, C. C. B. F.; Longhini, V. Z.; Dias, A. M.; Ítavo, L. C. V. Mathematical Models for Predicting Protodioscin in Tropical Forage Grasses. Toxicon 2024, 240, 107628. [Google Scholar] [CrossRef]
- Meagher, L.; Wilkins, A.; Miles, C.; Fagliari, J. Hepatogenous photosensitization of ruminants by Brachiaria decumbens and Panicum dichotomiflorum in the absence of sporidesmin: lithogenic saponins may be responsible. Vet. Hum. Toxicol. 1996, 38, 271–274. [Google Scholar]
- Pires, V. S.; Taketa, A. T. C.; Gosmann, G.; Schenkel, E. P. Saponins and sapogenins from Brachiaria decumbens Stapf. J. Braz. Chem. Soc. 2002, 13, 135–139. [Google Scholar] [CrossRef]
- Lee, S.; Mitchell, R.; Gardner, D.; Tokarnia, C.; Riet-Correa, F. Measurement of steroidal saponins in Panicum and Brachiaria grasses in the USA and Brazil. In Riet-Correa, F.; Pfister, J., Schild, A. L., Wierenga, T. (Eds.), Poisoning by Plants, Eds.; CABI: Oxfordshire, UK, 2011; pp. 142–147. [Google Scholar]
- Oliveira, D. R.; Nepomuceno, D. D.; Castro, R. N.; Braz, R. F.; Carvalho, M. G. Special Metabolites Isolated from Urochloa humidicola (Poaceae). An. Acad. Bras. Cienc. 2017, 89, 789–797. [Google Scholar] [CrossRef]
- Low, S. Signal grass (Brachiaria decumbens) toxicity in grazing ruminants. Agriculture 2015, 5, 971–990. [Google Scholar] [CrossRef]
- Hussain, M.; Debnath, B.; Qasim, M.; Bamisile, B. S.; Islam, W.; Hameed, M. S.; Wang, L.; Qiu, D. Role of saponins in plant defense against specialist herbivores. Molecules 2019, 24, 2067. [Google Scholar] [CrossRef]
- Ciura, J.; Szeliga, M.; Tyrka, M. Optimization of in vitro culture conditions for accumulation of diosgenin by fenugreek. J. Med. Plants Stud. 2015, 3, 22–25. [Google Scholar]
- Kono, I. S.; Faccin, T. C.; Amorim de Lemos, G. A.; Di Santis, G. W.; Bacha, F. B.; Guerreiro, Y. A.; de Oliveira Gaspar, A.; Lee, S. T.; de Castro Guizelini, C.; Leal, C. B.; Amaral de Lemos, R. A. Outbreaks of Brachiaria ruziziensis and Brachiaria brizantha Intoxications in Brazilian Experienced Cattle. Toxicon 2022, 219, 106931. [Google Scholar] [CrossRef] [PubMed]
- Moore, G. Saponins in Sub-Tropical Grasses in WA; Government of Western Australia: Perth, Australia, 2014; p. 4. [Google Scholar]
- Chong Dubé, D.; Figueredo, J. M.; Percedo, M. I.; Domínguez, P.; Martínez-García, Y.; Alfonso, P.; Marrero-Faz, E. Toxicosis por pasto Mulato (Brachiaria ruziziensis - Brachiaria brizantha) en cabras de la provincia Artemisa. Rev. Salud Anim. 2016, 38, 19–24. [Google Scholar]
- Marin, R. E.; Gimeno, E. J.; Riet-Correa, F.; Uzal, F. A. Hepatic and Renal Lesions in Sheep Intoxicated with Urochloa Hybrid Mulato II in Argentina. J. Vet. Diagn. Invest. 2024, 36, 262–268. [Google Scholar] [CrossRef]
- Chen, Y.; Dong, Y.; Chi, Y.; He, Q.; Wu, H.; Ren, Y. Eco-friendly microbial production of diosgenin from saponins in Dioscorea zingiberensis tubers in the presence of Aspergillus awamori. Steroids 2018, 136, 40–46. [Google Scholar] [CrossRef]
- Zheng, T.; Yu, L.; Zhu, Y.; Zhao, B. Evaluation of different pretreatments on microbial transformation of saponins in Dioscorea zingiberensis for diosgenin production. Biotechnol. Biotechnol. Equip. 2014, 28, 740–746. [Google Scholar] [CrossRef]
- Dong, J.; Lei, C.; Lu, D.; Wang, Y. Direct biotransformation of dioscin into diosgenin in rhizome of Dioscorea zingiberensis by Penicillium dioscin. Indian J. Microbiol. 2015, 55, 200–206. [Google Scholar] [CrossRef]
- Feng, S.; Pan, L.; Li, Q.; Zhang, Y.; Mou, F.; Liu, Z.; Zhang, Y.; Duan, L.; Qin, B.; Hu, Z. The Isolation, Identification and Immobilization Method of Three Novel Enzymes with Diosgenin-Producing Activity Derived from an Aspergillus flavus. Int. J. Mol. Sci. 2023, 24, 17611. [Google Scholar] [CrossRef]
- Seki, H.; Tamura, K.; Muranaka, T. P450s and UGTs: Key players in the structural diversity of triterpenoid saponin. Plant Cell Physiol. 2015, 56, 1436–1471. [Google Scholar] [CrossRef] [PubMed]
- Haralampidis, K.; Trojanowska, M.; Osbourn, A. E. Biosynthesis of triterpenoid saponins in plants. In Scheper, T. (Ed.), History and Trends in Bioprocessing and Biotransformation; Advances in Biochemical Engineering/Biotechnology; Springer: Berlin, Germany, 2002. [Google Scholar]
- Pulido, J. I.; Romero, M.; Rivero, S. T.; Duarte, O. A.; Gómez, P. J.; Vanegas, E.; Jaime, W. E.; Parra, J. L.; Pérez, R. A.; Cipagauta, M.; Gómez, J. E.; Velásquez, J. E.; García, J.; Gutiérrez, A.; Narváez, L. Atlas de los Sistemas de Producción Bovina. Módulo Orinoquía y Amazonía; CORPOICA: Bogotá, Colombia, 2002. [Google Scholar]
- Taylor, W. G.; Elder, J. L.; Chang, P. R.; Richards, K. W. Microdetermination of diosgenin from fenugreek (Trigonella foenum-graecum) seeds. J. Agric. Food Chem. 2000, 48, 5206–5210. [Google Scholar] [CrossRef] [PubMed]
- EPA. 40 CFR Appendix B to Part 136—Definition and Procedure for the Determination of the Method Detection Limit-Revision 1.11. EPA Code of Federal Regulations; Government Printing Office: USA, 2011. [Google Scholar]
- Martínez, R.; Martínez, N. N.; Martínez, M. V. Diseño de Experimentos en Ciencias Agropecuarias y Biológicas con SAS, SPSS, R Y STATISTIX; Fondo Nacional Universitario: Bogotá, Colombia, 2011. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



