Submitted:
25 March 2025
Posted:
26 March 2025
You are already at the latest version
Abstract
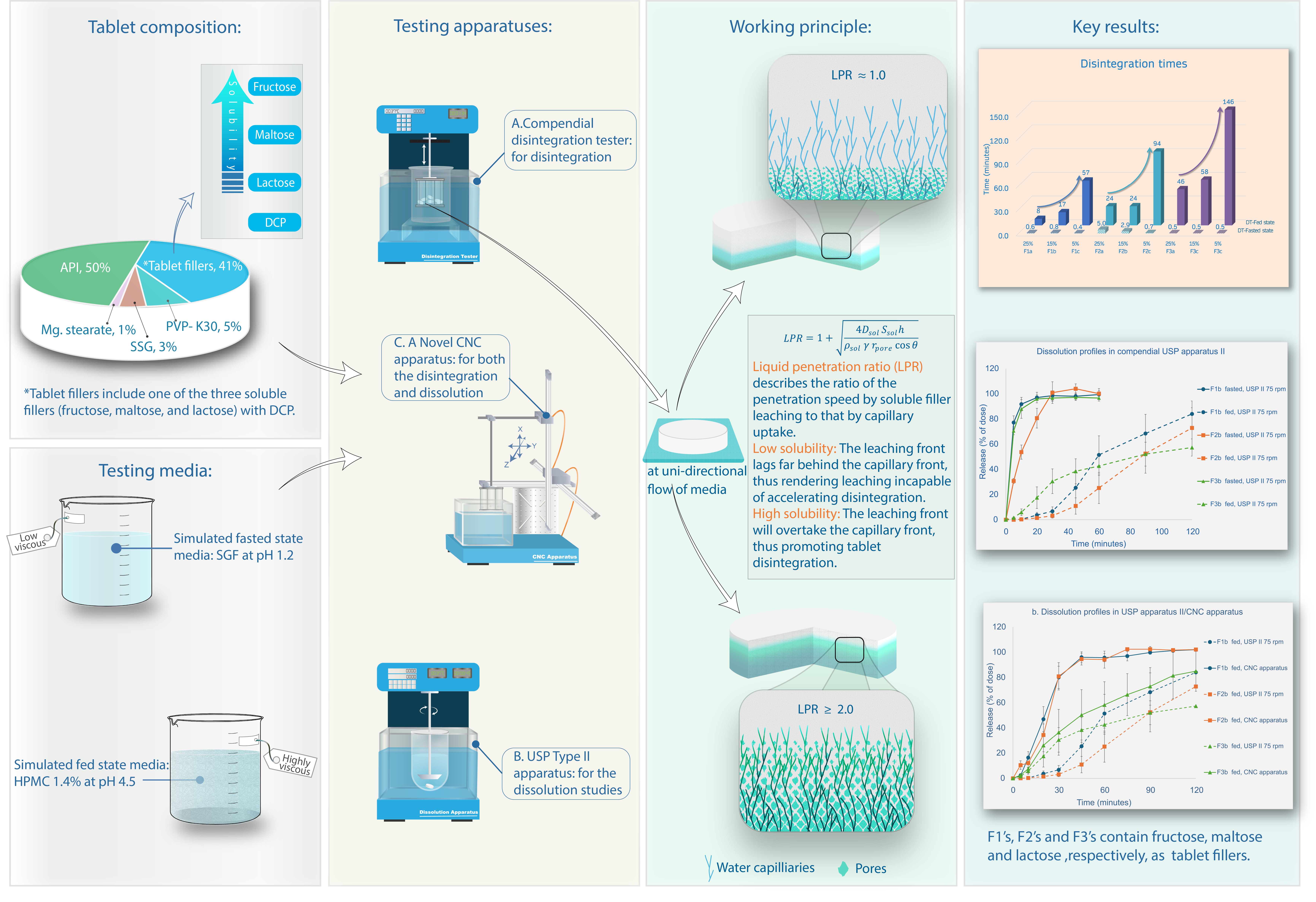
Keywords:
1. Introduction
2. Materials and Methods
Materials
| Formulation Code | Percentage content of soluble fillers and DCP, total of 30 % | |||||
|---|---|---|---|---|---|---|
| Fructose | Maltose | Lactose | Valine | Acetaminophen | DCP | |
| F1a | 25 | -- | -- | -- | -- | 5 |
| F1b | 15 | -- | -- | -- | -- | 15 |
| F1c | 5 | -- | -- | -- | -- | 25 |
| F2a | -- | 25 | -- | -- | -- | 5 |
| F2b | -- | 15 | -- | -- | -- | 15 |
| F2c | -- | 5 | -- | -- | -- | 25 |
| F3a | -- | -- | 25 | -- | -- | 5 |
| F3b | -- | -- | 15 | -- | -- | 15 |
| F3c | -- | -- | 5 | -- | -- | 25 |
| F4 | -- | -- | -- | 25 | -- | 5 |
| F5 | -- | -- | -- | -- | 25 | 5 |
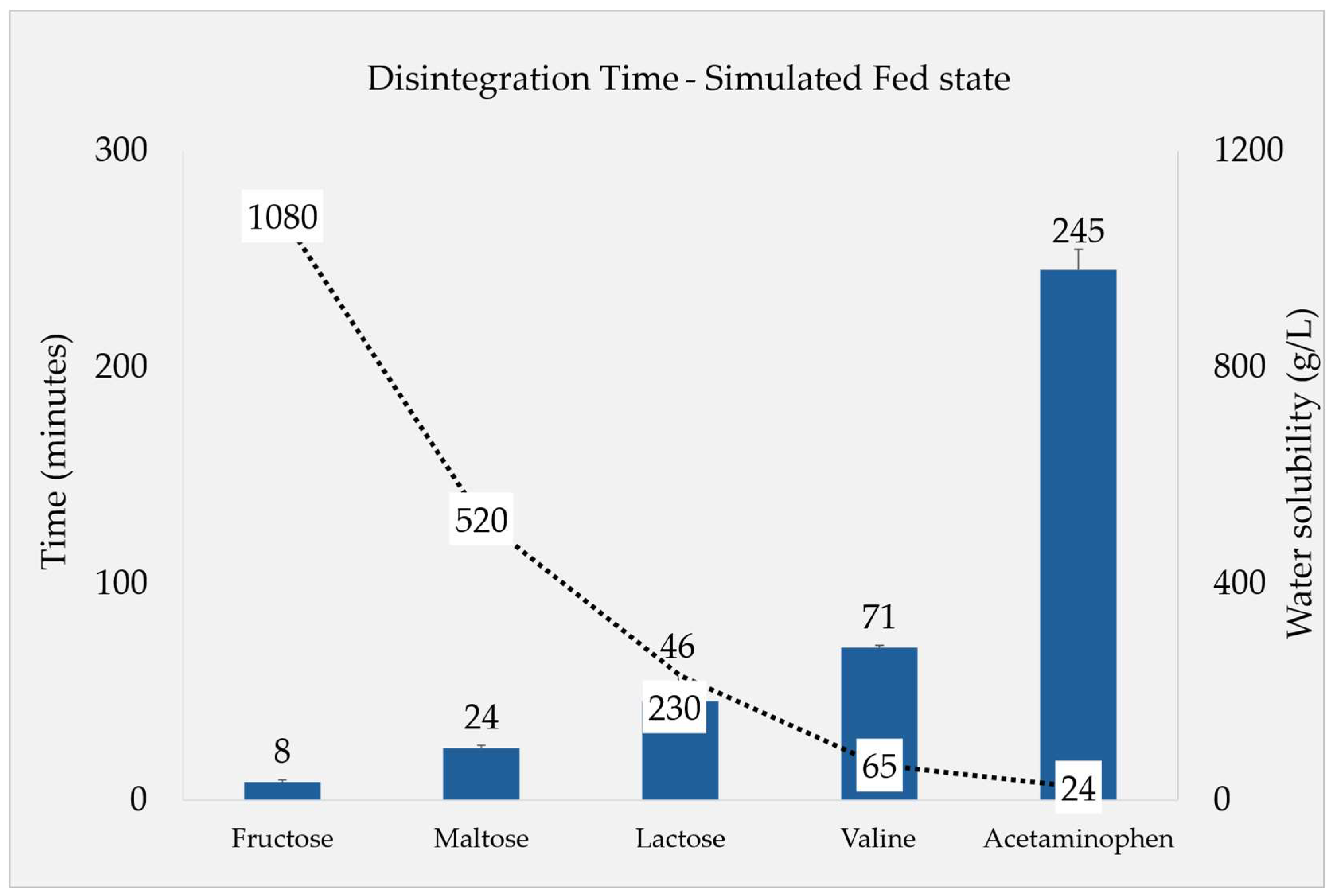
| Water solubility g/L |
1080 [16] |
520 [17] |
230 [18] |
65 [19] |
24 [20] |
Considered insoluble |
Tablet Formulation
Testing Media Composition
Tablet Hardness Measurements
Disintegration Test
Compendial Disintegration Test Under Simulated Fasted State Conditions
CNC Disintegration Test Under Simulated Fed State Conditions
Dissolution Studies
Dissolution Testing in Compendial Device Under Simulated Fasted and Fed State Conditions
Dissolution Testing in CNC Device Under Simulated Fed State Conditions
Analytics
| Parameter | Value |
|---|---|
| Absorption wavelength | 242 nm |
| Linear range | 1 - 14 µg/mL |
| Calibration curve R2 Slope Intercept |
1.000 0.0634 -0.0001 |
| Limit of detection | 0.09 µg/mL |
| Limit of quantification | 0.26 µg/mL |
| Intraday precision (triplicate) 4.0 µg/mL 8.0 µg/mL 12.0 µg/mL |
± 0.68 % relative standard deviation (RSD) ± 0.23 % RSD ± 0.53 % RSD |
| Interday precision (8.0 µg/mL at three consecutive days) |
± 0.34 % RSD |
Porosimetry
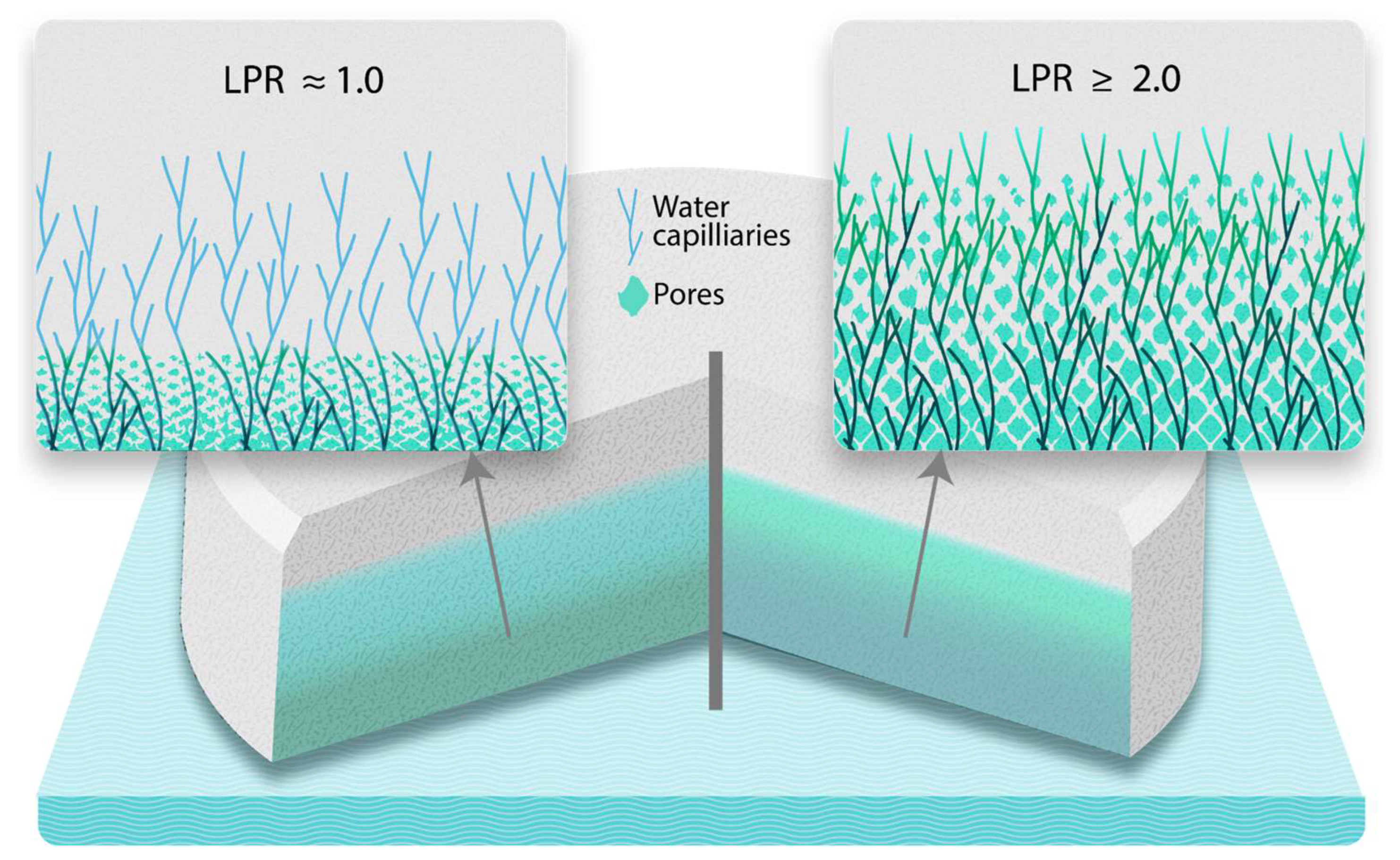
Working Principle – Liquid Penetration Ratio (LPR)
3. Results
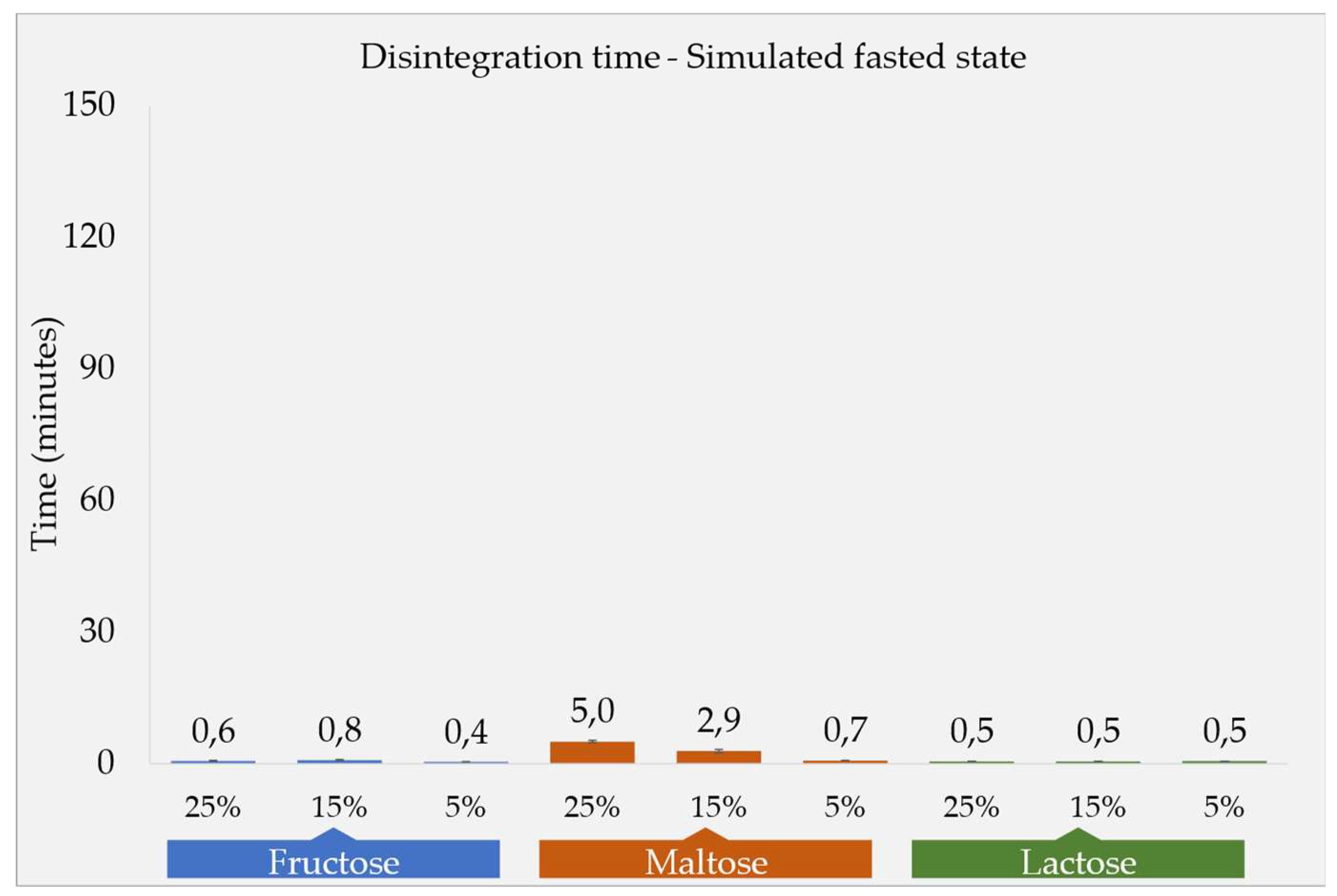
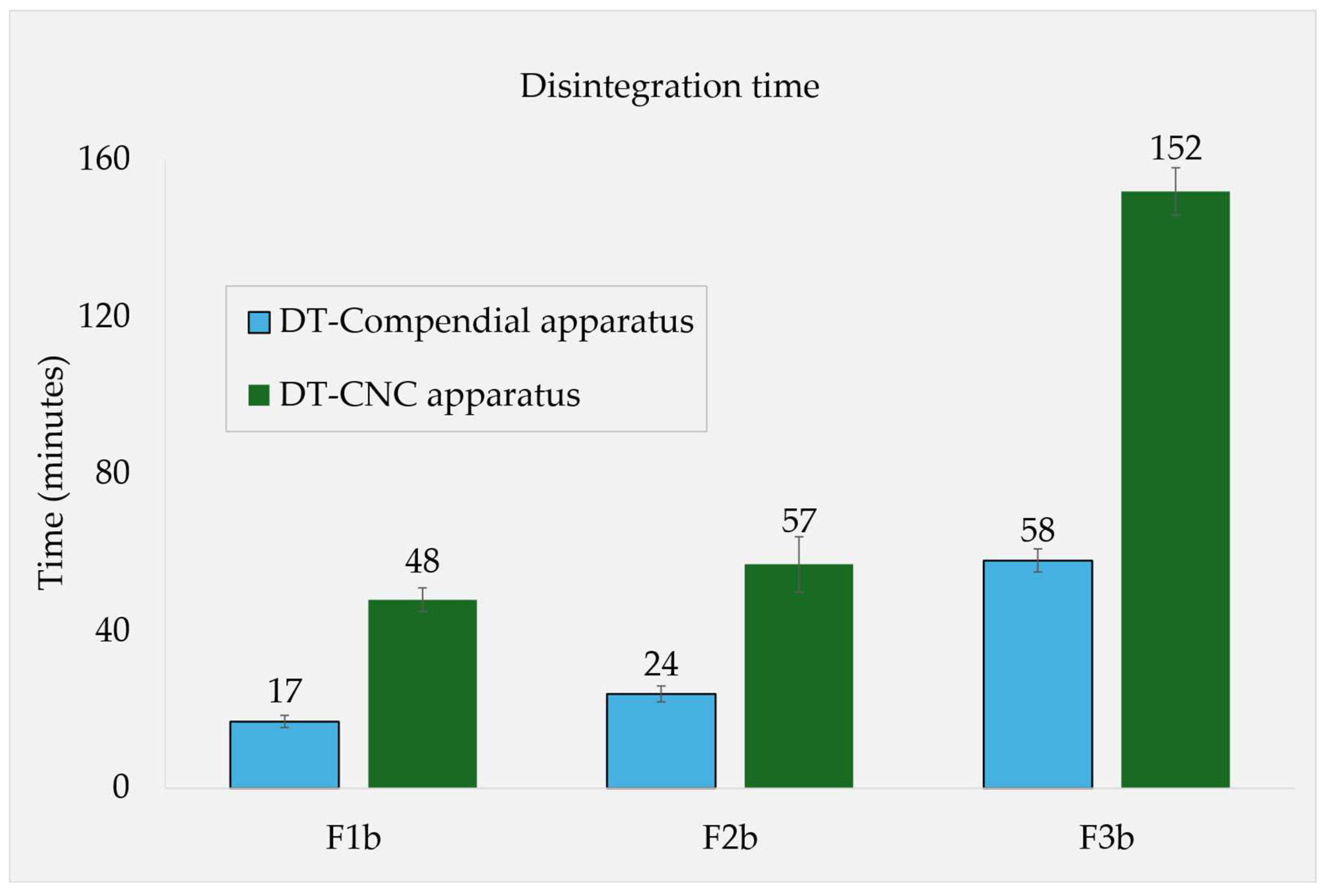
Disintegration Times and Tablet Properties
| Formulation Code | DT-fasted state compendial app., Mean ± SD (n=6) (minutes) |
DT-fed state compendial app., Mean ± SD (n=6) (minutes) |
DT-fed state CNC app., Mean ± SD (n=3) (minutes) |
Tablet weight, Mean ± SD (n=3) (mg) |
Tablet hardness, Mean ± SD (n=3) (N) |
|---|---|---|---|---|---|
| F1a | 37 ± 07 | 08 ± 01 | -* | 650 ± 05 | 64 ± 04 |
| F1b | 47 ± 05 | 17 ± 02 | 48 ± 03 | 651 ± 05 | 76 ± 02 |
| F1c | 26 ± 02 | 57 ± 04 | -* | 648 ± 03 | 92 ± 05 |
| F2a | 301 ± 19 | 24 ± 01 | -* | 653 ± 03 | 113 ± 02 |
| F2b | 172 ± 23 | 24 ± 02 | 57 ± 07 | 649 ± 05 | 87 ± 05 |
| F2c | 42 ± 03 | 94 ± 02 | -* | 649 ± 02 | 78 ± 02 |
| F3a | 31 ± 02 | 46 ± 01 | -* | 653 ± 01 | 91 ± 06 |
| F3b | 32 ± 02 | 58 ± 03 | 152 ± 06 | 650 ± 02 | 88 ± 02 |
| F3c | 32 ± 02 | 146 ± 03 | -* | 650 ± 02 | 91 ± 04 |
| F4 | 88 ± 04 | 71 ± 01 | -* | 649 ± 01 | 76.10 ± 2.3 |
| F5 | 19 ± 2 | 245 ± 09 | -* | 649 ± 01 | 40.73 ± 2.5 |
Fasted State Conditions
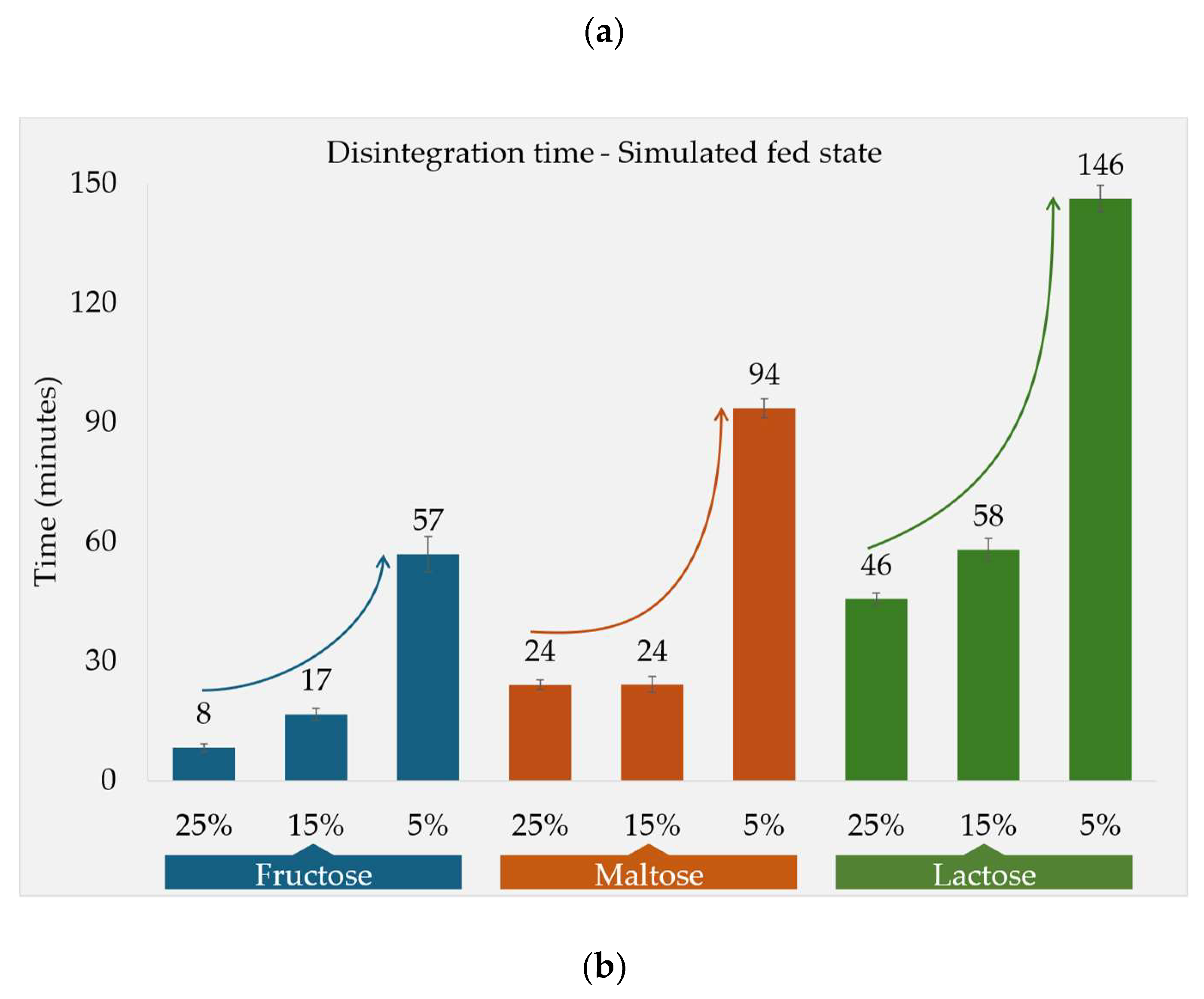
Fed State Conditions
Porosity Determination and Physical Characterization
| Formulation | F1b | F2b | F3b |
|---|---|---|---|
| Bulk density (g/cm³) at 0 Mpa | 1.2697 | 1.2630 | 1.2919 |
| Bulk density (g/cm³) at 400 Mpa | 1.4390 | 1.3934 | 1.4069 |
| Average pore diameter (nm) | 33.34 | 41.06 | 35.32 |
| Porosity (%) | 11.67 | 9.36 | 8.17 |
Dissolution Studies
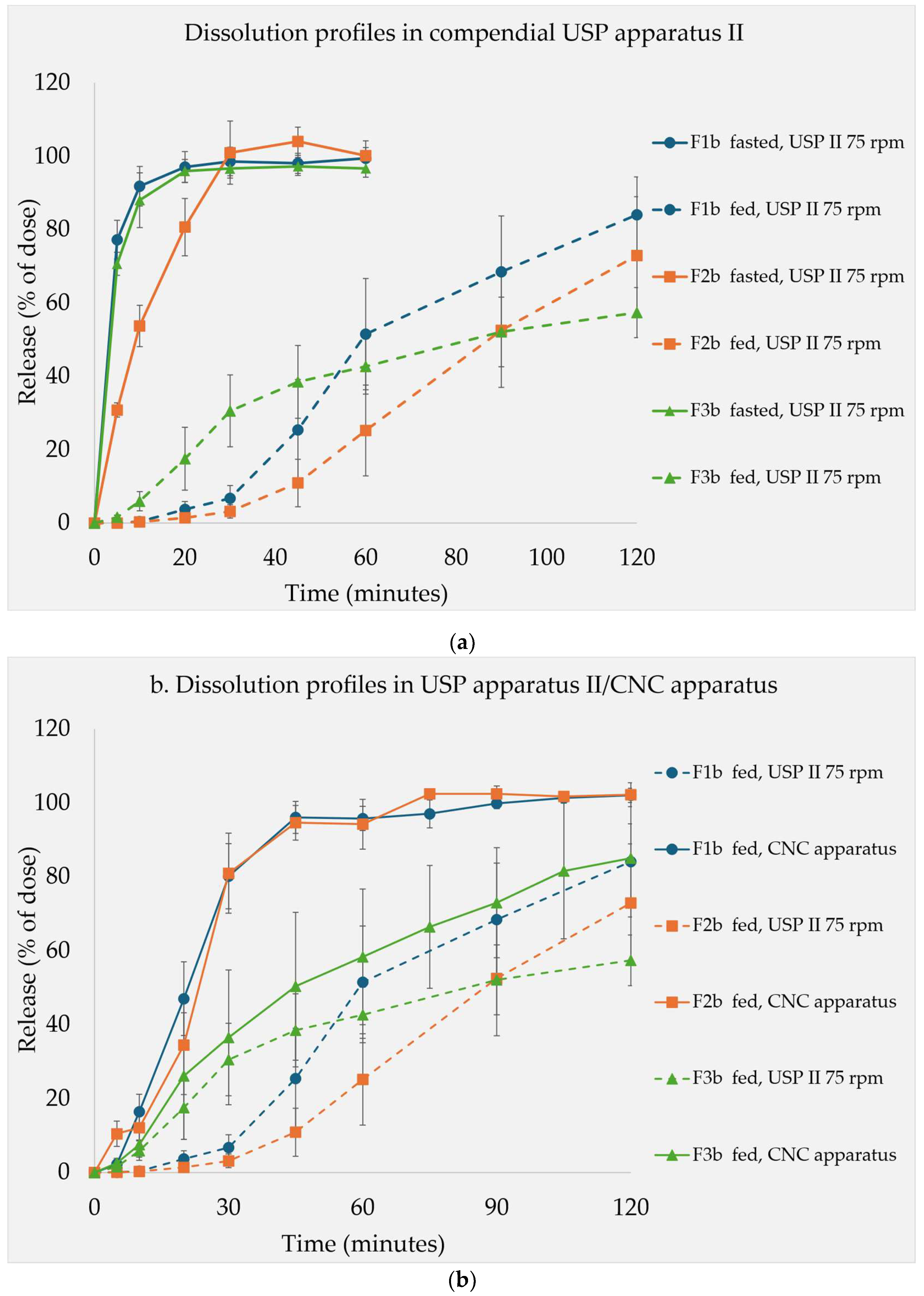
4. Discussion
Disintegration and Dissolution Under Simulated Fasted State
Tablet Disintegration and LPR
Dissolution Tests Under Fed State
Physical Characterization
Filler Quantity
5. Conclusions
Disclaimer
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| API | Active pharmaceutical ingredient |
| CNC | Computed numerical control |
| DCP | Dicalcium Phosphate |
| DT | Disintegration time |
| HCl | Hydrochloric acid |
| HPMC | Hydroxypropyl methylcellulose |
| ICH | International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use |
| IR | Immediate release |
| LPR | Liquid penetration ratio |
| MCC | Microcrystalline cellulose |
| MPa | Mega Pascals |
| NaCl | Sodium Chloride |
| NaOH | Sodium hydroxide |
| PVP | Polyvinyl pyrrolidone |
| RSD | Relative standard deviation |
| SD | Standard deviation |
| SSG | sodium starch glycolate |
| US FDA | US food and drug administration |
| USP | United States Pharmacopeia |
References
- Ghosh, S. Oral Solid Dosage Pharmaceutical Formulation Market Outlook: Oral Solid Dosage Pharmaceutical Formulation Market Forecast by Tablets, Capsules, and Other Dosage Forms From 2024 To 2034. Surging Demand for Innovative Oral Dosage Formulations Driven by Shift Towards Personalized Medicine and Customized Treatments. FMI Insights into Biodegradable Polymer Sustainability REP-GB-2782, 2017. Available online: https://www.futuremarketinsights.com/reports/oral-solid-dosage-pharmaceutical-formulation-market# (accessed on 12 April 2024).
- Kotsybar, J.; Hakeem, S.; Zhang, L.; Jiang, W. Global harmonization of immediate-release solid oral drug product bioequivalence recommendations and the impact on generic drug development, 2023. Clinical and translational science, 16 (12).
- Horkovics-Kovats, S. Disintegration Rate and Properties of Active Pharmaceutical Ingredient Particles as Determined from the Dissolution Time Profile of a Pharmaceutical Formulation: An Inverse Problem. J. Pharm. Sci. 2014, 103, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Berardi, A.; Bisharat, L.; Quodbach, J.; Abdel Rahim, S.; Perinelli, D.R.; Cespi, M. Advancing the understanding of the tablet disintegration phenomenon – An update on recent studies. Int. J. Pharm. 2021, 598, 120390. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Shi, C.; Fang, Y.; Zhao, H.; Mu, Y.; Zhao, L.; Shen, L. A comprehensive understanding of disintegrants and disintegration quantification techniques: From the perspective of tablet microstructure. Journal of Drug Delivery Science and Technology 2023, 88, 104891. [Google Scholar] [CrossRef]
- Radwan, A.; Wagner, M.; Amidon, G.L.; Langguth, P. Bio-predictive tablet disintegration: effect of water diffusivity, fluid flow, food composition and test conditions. Eur. J. Pharm. Sci. 2014, 57, 273–279. [Google Scholar] [CrossRef]
- Al-Gousous, J.; Langguth, P. Oral Solid Dosage Form Disintegration Testing - The Forgotten Test. Journal of Pharmaceutical Sciences 2015, 104, 2664–2675. [Google Scholar] [CrossRef]
- Abdallah, D.B.; Charoo, N.A.; Elgorashi, A.S. Comparative binding and disintegrating property of Echinochloa colona starch (difra starch) against maize, sorghum, and cassava starch. Pharm. Biol. 2014, 52, 935–943. [Google Scholar] [CrossRef]
- Quodbach, J.; Kleinebudde, P. A critical review on tablet disintegration. Pharm. Dev. Technol. 2016, 21, 763–774. [Google Scholar] [CrossRef]
- Dvořák, J.; Tomas, J.; Lizoňová, D.; Schöngut, M.; Dammer, O.; Pekárek, T.; Beránek, J.; Štěpánek, F. Investigation of tablet disintegration pathways by the combined use of magnetic resonance imaging, texture analysis and static light scattering. International Journal of Pharmaceutics 2020, 587, 119719. [Google Scholar] [CrossRef]
- Radwan, A.; Amidon, G.L.; Langguth, P. Mechanistic investigation of food effect on disintegration and dissolution of BCS class III compound solid formulations: the importance of viscosity. Biopharm. Drug Dispos. 2012, 33, 403–416. [Google Scholar] [CrossRef]
- Kindgen, S.; Rach, R.; Nawroth, T.; Abrahamsson, B.; Langguth, P. A Novel Disintegration Tester for Solid Dosage Forms Enabling Adjustable Hydrodynamics. Journal of Pharmaceutical Sciences 2016, 105, 2402–2409. [Google Scholar] [CrossRef]
- Ruiz-Picazo, A.; Colón-Useche, S.; Gonzalez-Alvarez, M.; Gonzalez-Alvarez, I.; Bermejo, M.; Langguth, P. Effect of thickener on disintegration, dissolution and permeability of common drug products for elderly patients. Eur. J. Pharm. Biopharm. 2020, 153, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Washburn, E.W. The Dynamics of Capillary Flow.
- Zaheer, K.; Langguth, P. Formulation strategy towards minimizing viscosity mediated negative food effect on disintegration and dissolution of immediate release tablets. Drug Dev. Ind. Pharm. 2018, 44, 444–451. [Google Scholar] [CrossRef]
- Crestani, C.E.; Bernardo, A.; Costa, C.B.B.; Giulietti, M. Fructose Solubility in Mixed (Ethanol + Water) Solvent: Experimental Data and Comparison among Different Thermodynamic Models. J. Chem. Eng. Data, 58 (11).
- Gong, X.; Wang, C.; Zhang, L.; Qu, H. Solubility of Xylose, Mannose, Maltose Monohydrate, and Trehalose Dihydrate in Ethanol–Water Solutions. J. Chem. Eng. Data, 57 (11).
- Fox, P.F.; Uniacke-Lowe, T.; McSweeney, P.L.H.; O’Mahony, J.A. Dairy Chemistry and Biochemistry; Springer International Publishing: Cham, 2015; ISBN 978-3-319-14892-2. [Google Scholar]
- Zhang, C.; Liu, B.; Wang, X.; Wang, H. Measurement and Correlation of the Solubilities of l -Valine in Water, Ethanol, N, N -Dimethylformamide, Acetone, and Isopropyl Alcohol between (293.15 and 343.15) K. J. Chem. Eng. Data 2014, 59, 2704–2708. [Google Scholar] [CrossRef]
- Kalantzi, L.; Reppas, C.; Dressman, J.B.; Amidon, G.L.; Junginger, H.E.; Midha, K.K.; Shah, V.P.; Stavchansky, S.A.; Barends, D.M. Biowaiver monographs for immediate release solid oral dosage forms: acetaminophen (paracetamol). Journal of Pharmaceutical Sciences 2006, 95, 4–14. [Google Scholar] [CrossRef]
- Higuchi, M.; Nishida, S.; Yoshihashi, Y.; Tarada, K.; Sugano, K. Prediction of coning phenomena for irregular particles in paddle dissolution test. Eur. J. Pharm. Sci. 2015, 76, 213–216. [Google Scholar] [CrossRef]
- Al-Sharabi, M.; Markl, D.; Mudley, T.; Bawuah, P.; Karttunen, A.-P.; Ridgway, C.; Gane, P.; Ketolainen, J.; Peiponen, K.-E.; Rades, T.; et al. Simultaneous investigation of the liquid transport and swelling performance during tablet disintegration. International Journal of Pharmaceutics 2020, 584, 119380. [Google Scholar] [CrossRef]
- Mizumoto, T.; Masuda, Y.; Yamamoto, T.; Yonemochi, E.; Terada, K. Formulation design of a novel fast-disintegrating tablet. International Journal of Pharmaceutics 2005, 306, 83–90. [Google Scholar] [CrossRef]
- Desai, P.M.; Liew, C.V.; Heng, P.W.S. Assessment of disintegration of rapidly disintegrating tablets by a visiometric liquid jet-mediated disintegration apparatus. International Journal of Pharmaceutics 2013, 442, 65–73. [Google Scholar] [CrossRef]
- Handbook of pharmaceutical excipients; Rowe, R.C.; Sheskey, P.J.; Quinn, M.E., Eds., 6. ed.; APhA (PhP) Pharmaceutical Press: London, 2009.
- Lourdin, D.; Colonna, P.; Ring, S.G. Volumetric behaviour of maltose-water, maltose-glycerol and starch-sorbitol-water systems mixtures in relation to structural relaxation. Carbohydr. Res. 2003, 338, 2883–2887. [Google Scholar] [CrossRef]
- Parojcić, J.; Vasiljević, D.; Ibrić, S.; Djurić, Z. Tablet disintegration and drug dissolution in viscous media: paracetamol IR tablets. International Journal of Pharmaceutics 2008, 355, 93–99. [Google Scholar] [CrossRef]
- Vrbanac, H.; Trontelj, J.; Kalčič, Š.; Legen, I. Mechanistic study of model drug release from HPMC matrices in fed gastric media. Journal of Drug Delivery Science and Technology 2020, 60, 102034. [Google Scholar] [CrossRef]
- Berardi, A.; Bauhuber, S.; Sawafta, O.; Warnke, G. Alginates as tablet disintegrants: Understanding disintegration mechanisms and defining ranges of applications. International Journal of Pharmaceutics 2021, 601, 120512. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




