Submitted:
21 March 2025
Posted:
25 March 2025
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Choi M, Kipps T, Kurzrock R. ATM Mutations in Cancer: Therapeutic Implications. Mol Cancer Ther. 2016 Aug;15(8):1781-91. [CrossRef]
- Hall MJ, Bernhisel R, Hughes E, Larson K, Rosenthal ET, Singh NA, et al. Germline Pathogenic Variants in the Ataxia Telangiectasia Mutated (ATM) Gene are Associated with High and Moderate Risks for Multiple Cancers. Cancer Prev Res (Phila). 2021 Apr;14(4):433-440. [CrossRef]
- Lu C, Xie M, Wendl MC, Wang J, McLellan MD, Leiserson MD, et al. Patterns and functional implications of rare germline variants across 12 cancer types. Nat Commun. 2015 Dec 22;6:10086. [CrossRef]
- Sorscher S, LoPiccolo J, Heald B, Chen E, Bristow SL, Michalski ST, et al. Rate of Pathogenic Germline Variants in Patients With Lung Cancer. JCO Precis Oncol. 2023 Sep;7:e2300190. [CrossRef]
- Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature. 2014 Jul 31;511(7511):543-50. [CrossRef]
- Cancer Genome Atlas Research Network. Comprehensive genomic characterization of squamous cell lung cancers. Nature. 2012 Sep 27;489(7417):519-25. [CrossRef]
- Thu KL, Yoon JY. ATM-the gene at the moment in non-small cell lung cancer. Transl Lung Cancer Res. 2024 Mar 29;13(3):699-705. [CrossRef]
- Ricciuti B, Elkrief A, Alessi J, Wang X, Li Y, Gupta H, et al. Clinicopathologic, Genomic, and Immunophenotypic Landscape of ATM Mutations in Non-Small Cell Lung Cancer. Clin Cancer Res 2023;29:2540-50. [CrossRef]
- Vokes NI, Galan Cobo A, Fernandez-Chas M, Molkentine D, Treviño S 3rd, Druker V, et al. ATM Mutations Associate with Distinct Co-Mutational Patterns and Therapeutic Vulnerabilities in NSCLC. Clin Cancer Res 2023;29:4958-72. [CrossRef]
- Schneider J, Illig T, Rosenberger A, Bickeböller H, Wichmann HE. Detection of ATM gene mutations in young lung cancer patients: a population-based control study. Arch Med Res. 2008 Feb;39(2):226-31. [CrossRef]
- Kim JH, Kim H, Lee KJ, Choe KH, Ryu JS, Yoon HI, et al. Genetic polymorphisms of ataxia telangiectasia mutation affect lung cancer risk. Hum Mol Gen 2006;15:1181e1186. [CrossRef]
- Jialin L, Jiliang H, Lifen J, Wei Z, Zhijian C, Shijie C, et al. Variation of ATM protein expression in response to irradiation of lymphocytes in lung cancer patients and controls. Toxicology 2006;224: 138e146. [CrossRef]
- Weber AM, Drobnitzky N, Devery AM, Bokobza SM, Adams RA, Maughan TS, Ryan AJ. Phenotypic consequences of somatic mutations in the ataxia-telangiectasia mutated gene in non-small cell lung cancer. Oncotarget. 2016; 7:60807–60822. [CrossRef]
- Villaruz LC, Jones H, Dacic S, Abberbock S, Kurland BF, Stabile LP, et al. ATM protein is deficient in over 40% of lung adenocarcinomas. Oncotarget. 2016; 7:57714–57725. [CrossRef]
- Petersen LF, Klimowicz AC, Otsuka S, Elegbede AA, Petrillo SK, Williamson T, et al. Loss of tumour-specific ATM protein expression is an independent prognostic factor in early resected NSCLC. Oncotarget. 2017 Jun 13;8(24):38326-38336. [CrossRef]
- Provencio M, Calvo V, Romero A, Spicer JD, Cruz-Bermúdez A. Treatment sequencing in resectable lung cancer: the good and the bad of adjuvant versus neoadjuvant therapy. Am Soc Clin Oncol Educ B 2022;42:711–28. [CrossRef]
- Felip E, Altorki N, Zhou C, Csóśzi T, Vynnychenko I, Goloborodko O, et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB–IIIA non–small cell lung cancer (IMpower010): a randomized, multicenter, open-label, phase III trial. Lancet 2021;398:1344–57. [CrossRef]
- Forde PM, Spicer J, Lu S, Provencio M, Mitsudomi T, Awad MM, et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med 2022;386:1973–85. [CrossRef]
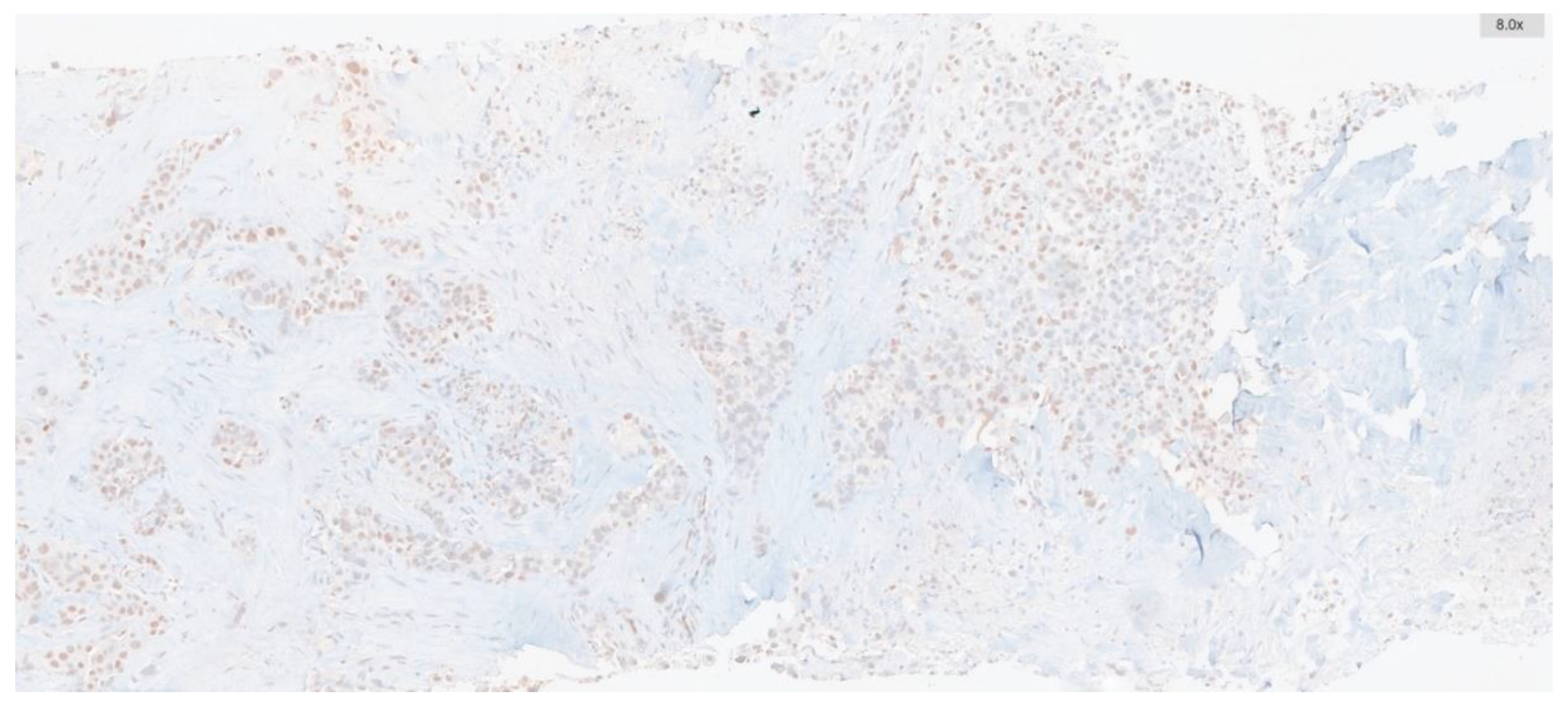
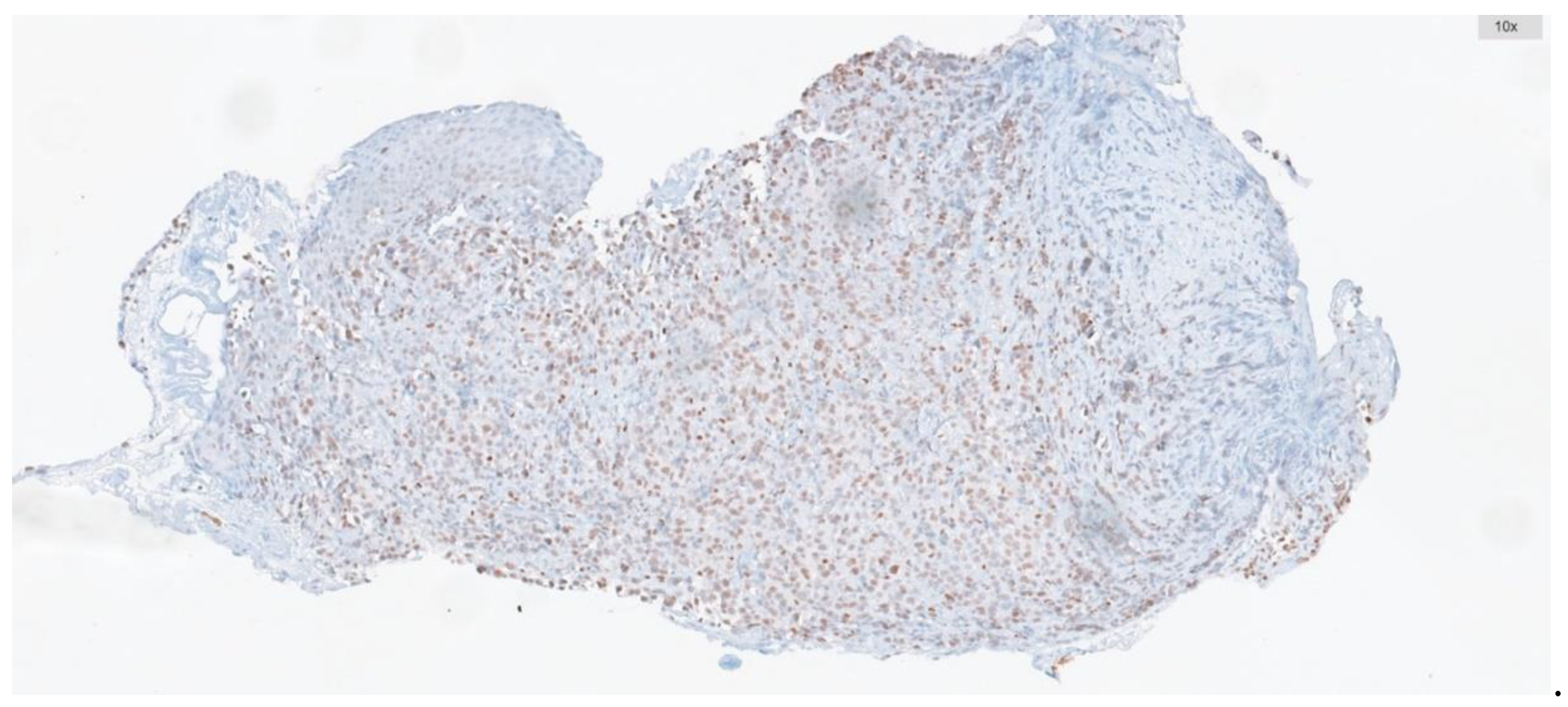
| Clinicopathological features | ATM Low expression n (%) |
ATM High expression n (%) |
p value |
|---|---|---|---|
| Age, years | 0.49 | ||
| <60 | 15 (40.5) | 3 (25.0) | |
| >60 | 22 (59.5) | 9 (75.0) | |
| Gender | 0.04 | ||
| Female | 1 (2.7) | 3 (25.0) | |
| Male | 36 (97.3) | 9 (75.0) | |
| Smoking History | 0.34 | ||
| Never | 3 (8.1) | 2 (16.7) | |
| Current | 20 (54.1) | 8 (66.7) | |
| Former | 14 (37.8) | 2 (16.7) | |
| Histopathological type | 0.16 | ||
| Adenocarcinoma | 30 (81.1) | 7 (58.3) | |
| Squamous cell carcinoma | 6 (16.2) | 5 (41.7) | |
| Nos | 1 (2.7) | 0 (0.0) | |
| Initial metastatic | 0.23 | ||
| Yes | 31 (83.8) | 8 (66.7) | |
| No | 6 (16.2) | 4 (33.3) | |
| Curative Surgery | 0.62 | ||
| Yes | 4 (10.8) | 2 (16.7) | |
| No | 33 (89.2) | 10 (83.3) | |
| Curative CRT | 0.25 | ||
| Yes | 2 (5.6) | 2 (16.7) | |
| No | 34 (94.4) | 10 (83.3) | |
| T Stage | 0.39 | ||
| T1 | 13 (35.1) | 2 (16.7) | |
| T2 | 11 (29.7) | 3 (25.0) | |
| T3 | 6 (16.2) | 2 (16.7) | |
| T4 | 7 (18.9) | 5 (41.7) | |
| Liver Metastases | 0.66 | ||
| Present | 6 (16.2) | 3 (25.0) | |
| Absent | 31 (83.8) | 9 (75.0) | |
| Brain metastases | 0.73 | ||
| Present | 12 (32.4) | 3 (25.0) | |
| Absent | 25 (67.6) | 9 (75.0) | |
| Bone metastases | 1.0 | ||
| Present | 20 (54.1) | 6 (50.0) | |
| Absent | 17 (45.9) | 6 (50.0) | |
| PD-L1 Status (TPS) | 0.90 | ||
| <%1 | 3 (8.1) | 1 (8.3) | |
| %1-50 | 15 (40.5) | 4 (33.3) | |
| >%50 | 19 (51.4) | 7 (58.3) | |
| First line treatment | 1.0 | ||
| Pembrolizumab monotherapy | 7 (18.9) | 2 (16.7) | |
| Pembrolizumab plus platin doublet | 30 (81.1) | 10 (83.3) |
| Variable | Median PFS (months) | Univariate p value | HR (%95 CI) | Multivariate p value |
|---|---|---|---|---|
| Age, years | 0.23 | |||
| <60 | 31.2 | |||
| >60 | 10.5 | |||
| Gender | 0.03 | 0.60 (0.17-2.12) |
0.43 | |
| Female | 6.1 | |||
| Male | 31.2 | |||
| Initial metastatic | 0.44 | |||
| Yes | 13.1 | |||
| No | 19.3 | |||
| Curative Surgery | 0.19 | |||
| Yes | 51.0 | |||
| No | 11.4 | |||
| T Stage | 0.44 | |||
| T1 | 51.0 | |||
| T2 | 14.8 | |||
| T3 | 8.1 | |||
| T4 | 5.4 | |||
| Liver Metastases | 0.26 | |||
| Present | 10.5 | |||
| Absent | 19.1 | |||
| Bone metastases | 0.11 | |||
| Present | 11.4 | |||
| Absent | 51.0 | |||
| PD-L1 Status (TPS) | 0.66 | |||
| <%1 | 8.13 | |||
| %1-50 | 13.1 | |||
| >%50 | 19.1 | |||
| First line treatment | 0.15 | 1.88 (0.62-5.66) |
0.26 | |
| Pembrolizumab monotherapy | 51.0 | |||
| Pembrolizumab plus platin doublet | 13.1 | |||
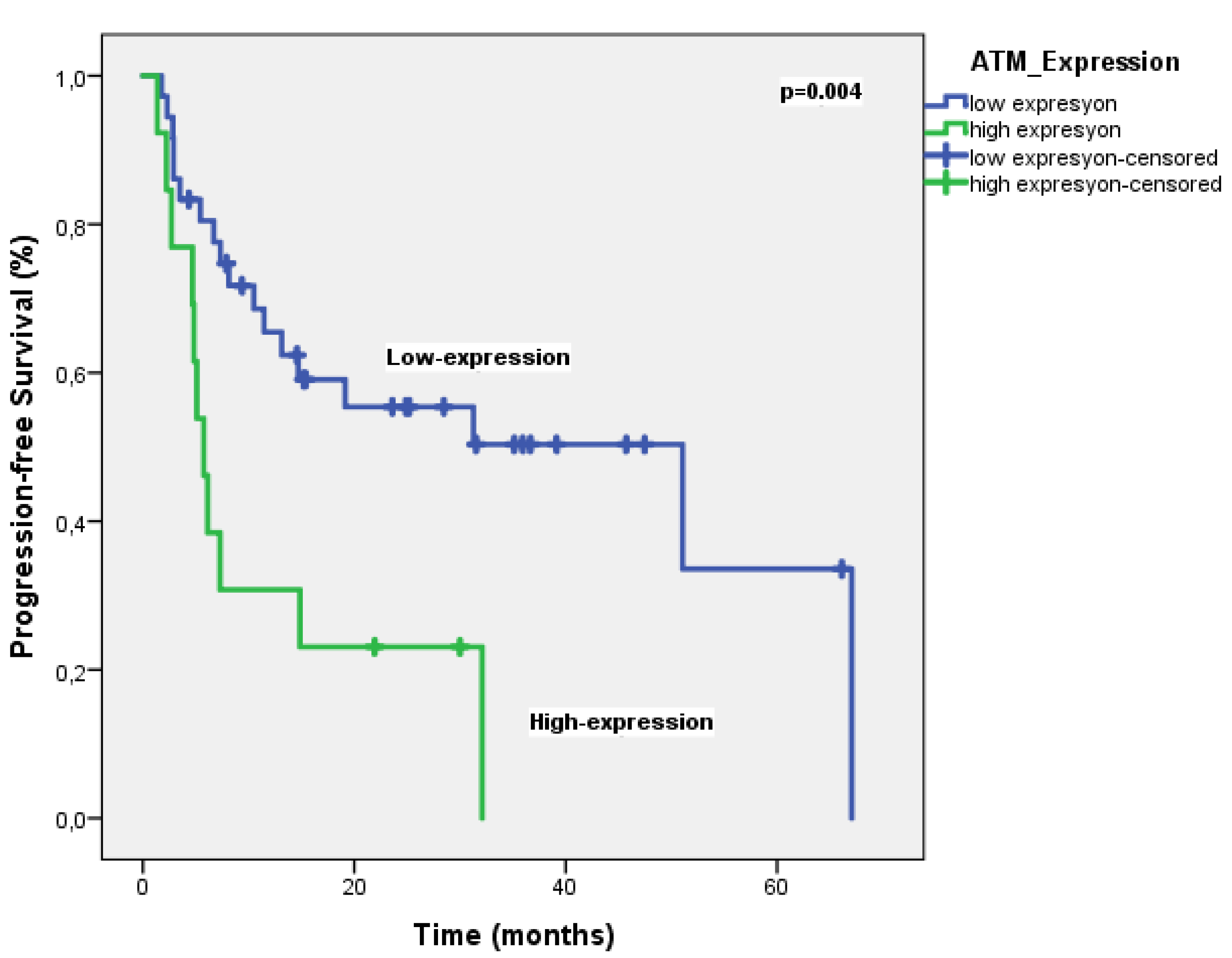
| ATM Score | 0.004 | 2.49 (1.07-5.76) |
0.033 | |
| Low expression | 51.0 | |||
| High expression | 5.7 | |||
| Site of metastasis | 0.38 | 0.91 (0.63-1.32) |
0.64 | |
| Liver | 7.3 | |||
| Brain | 32.1 | |||
| Bone | 11.4 |
| Variable | Median OS (months) | Univariate p value | HR (%95 CI) | Multivariate p value |
|---|---|---|---|---|
| Age, years | 0.29 | |||
| <60 | 35.8 | |||
| >60 | 19.1 | |||
| Gender | 0.16 | 0.75 (0.19-2.94) |
0.68 | |
| Female | 8.1 | |||
| Male | 35. | |||
| Initial metastatic | 0.42 | |||
| Yes | 19.9 | |||
| No | 51.0 | |||
| Curative Surgery | 0.30 | |||
| Yes | 51.0 | |||
| No | 19.9 | |||
| T Stage | 0.13 | |||
| T1 | 51.0 | |||
| T2 | 67.0 | |||
| T3 | 19.1 | |||
| T4 | 7.9 | |||
| Liver Metastases | 0.81 | |||
| Present | 35.8 | |||
| Absent | 24.5 | |||
| Bone metastases | 0.55 | |||
| Present | 19.9 | |||
| Absent | 51.0 | |||
| PD-L1 Status (TPS)* | 0.59 | |||
| <%1 | 9.36 | |||
| %1-50 | 35.8 | |||
| >%50 | 19.1 | |||
| First line treatment | 0.30 | 1.61 (0.52-4.95) |
0.40 | |
| Pembrolizumab monotherapy | 51.0 | |||
| Pembrolizumab plus platin doublet | 24.5 | |||
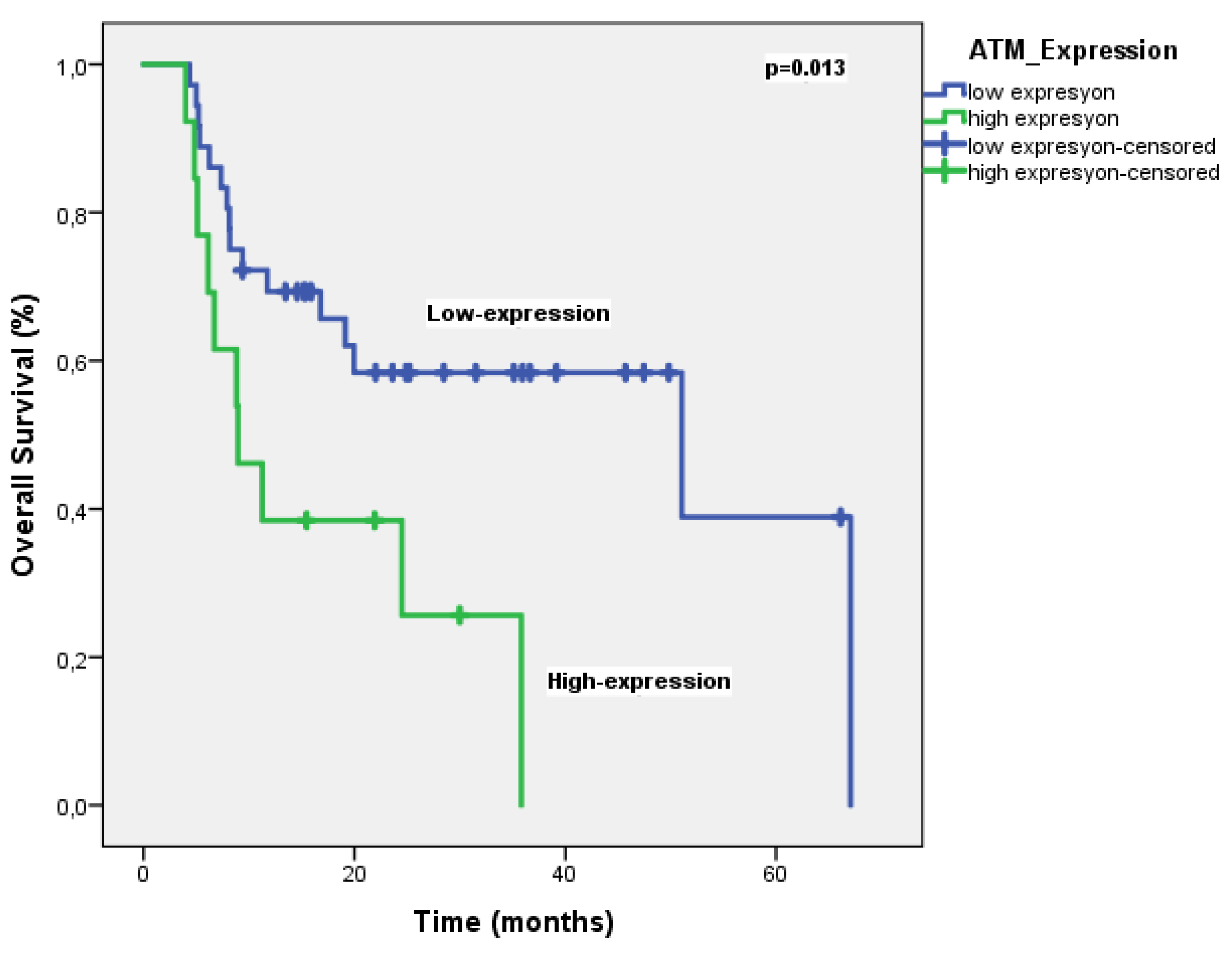
| ATM Score | 0.013 | 2.48 (1.03-5.94) |
0.041 | |
| Low expression | 51.0 | |||
| High expression | 8.9 | |||
| Site of metastasis | 0.90 | 1.09 (0.72-1.63) |
0.67 | |
| Liver | NR* | |||
| Brain | 35.83 | |||
| Bone | 19.96 |
| Factors | Coefficient ß | Wald X2 | p | OR | 95% CI |
|---|---|---|---|---|---|
| ATM Score (low vs high) | -2.80 | 7.42 | 0.006 | 0.06 | 0.008-0.45 |
| Gender | -0.14 | 0.003 | 0.95 | 0.86 | 0.007-109 |
| 1st line treatment | -1.74 | 2.12 | 0.14 | 0.17 | 0.017-1.81 |
| Liver metastasis | 3.28 | 5.20 | 0.023 | 26.65 | 1.58-447 |
| Brain metastasis | 1.47 | 1.97 | 0.16 | 4.38 | 0.55-34.5 |
| Bone metastasis | 2.39 | 4.67 | 0.031 | 10.99 | 1.25-96.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




