Submitted:
21 March 2025
Posted:
24 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
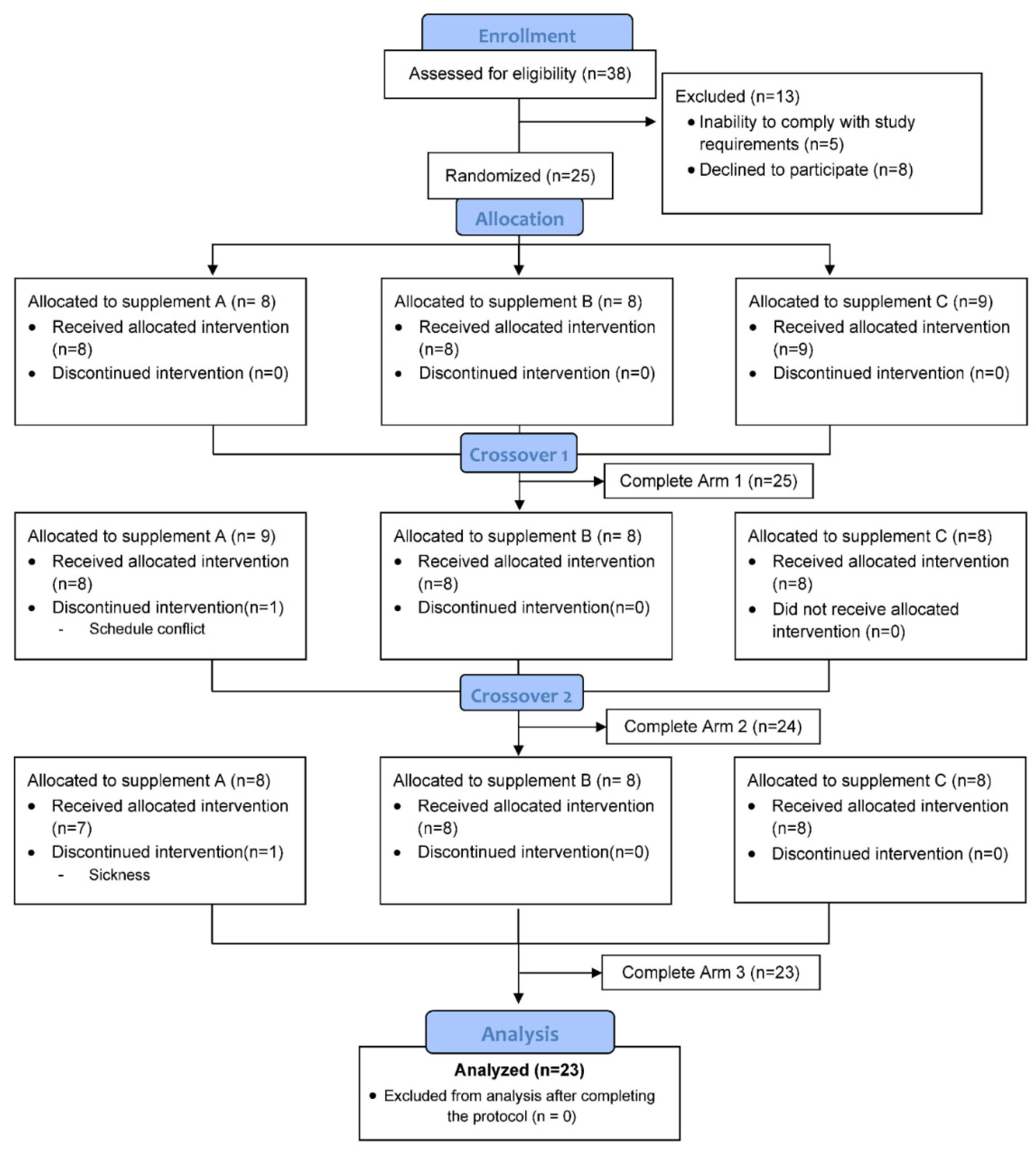
2.1. Study Participants
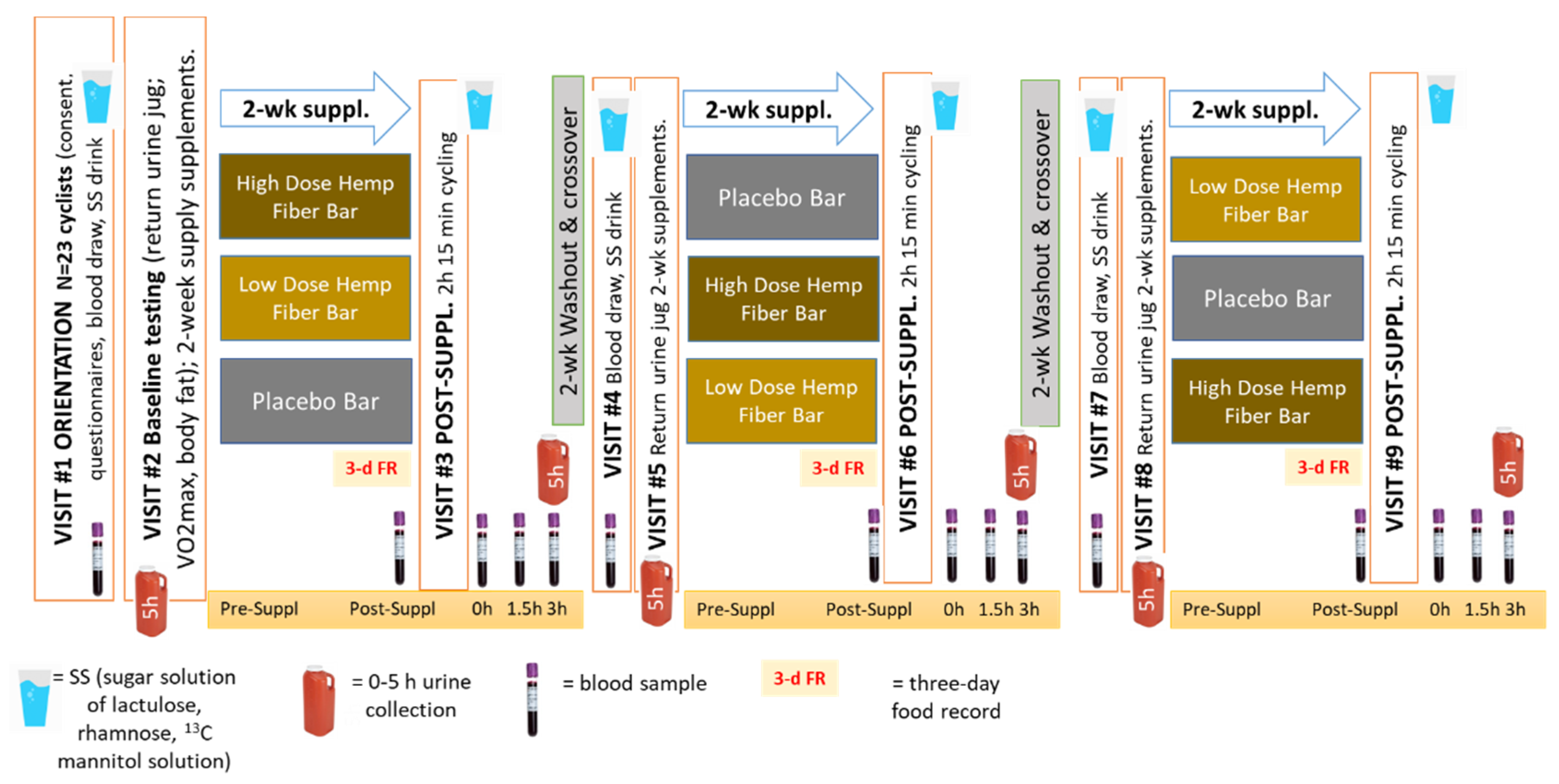
2.2. Study Design
- 7:00 am: Participants turned in the 3-d food record. A 30 ml blood sample was collected. Participants provided DOMS and POMS ratings and completed a 2-week retrospective symptom survey (with ratings of gastrointestinal symptoms, mental health, respiratory illness, sleep quality, pain symptoms, and overall wellbeing).
- 7:10 am: Participants ingested one supplement bar with one cup of water.
- 7:30 am: After a warm-up, participants cycled for 2.25h at approximately 70% VO2max on their own bicycles fitted to Saris H3 direct drive smart trainers (Madison, WI, USA) with monitoring by the Zwift online training platform (Long Beach, CA, USA) and the Cosmed CPET metabolic cart (Rome, Italy). Heart rate, cycling speed, cadence, distance, and power were measured and recorded continuously during the 2.25 h bout. Metabolic parameters such as breathing rate, ventilation, and oxygen intake were measured after 15 min and then every 30 min during the cycling session. To ensure performance consistency between trials, performance data from the first trial was used to ensure a similar power and metabolic output during the second and third trials. Participants consumed 3 ml/kg water every 15 min. No other beverage or food containing energy or nutrients were allowed during the 2.25h cycling sessions.
- 3h post-exercise period: Participants ingested the 450 mL SS within the first minute of getting off the bicycle, and urine was collected for the next five hours. Blood samples were collected immediately after completing the cycling session, and then 1.5h and 3.0h post-exercise. Participants were allowed to shower and change their clothes. The DOMS and POMS questionnaires were administered each time blood samples were collected. No food or beverage other than water (7 ml/kg) was ingested during the first 1.5h post-exercise. After the 1.5h post-exercise blood draw, participants ingested a fortified nutrient beverage (Boost, Nestlé S.A., Vevey, Switzerland). Another blood sample was collected 3-h post-exercise. Afterwards, participants were allowed to stay in the lab to complete the 5h urine collection or leave the lab and return later in the day to turn in the 5h urine container.
2.3. Sample Analysis
2.3.1. Urine Sugar Analysis
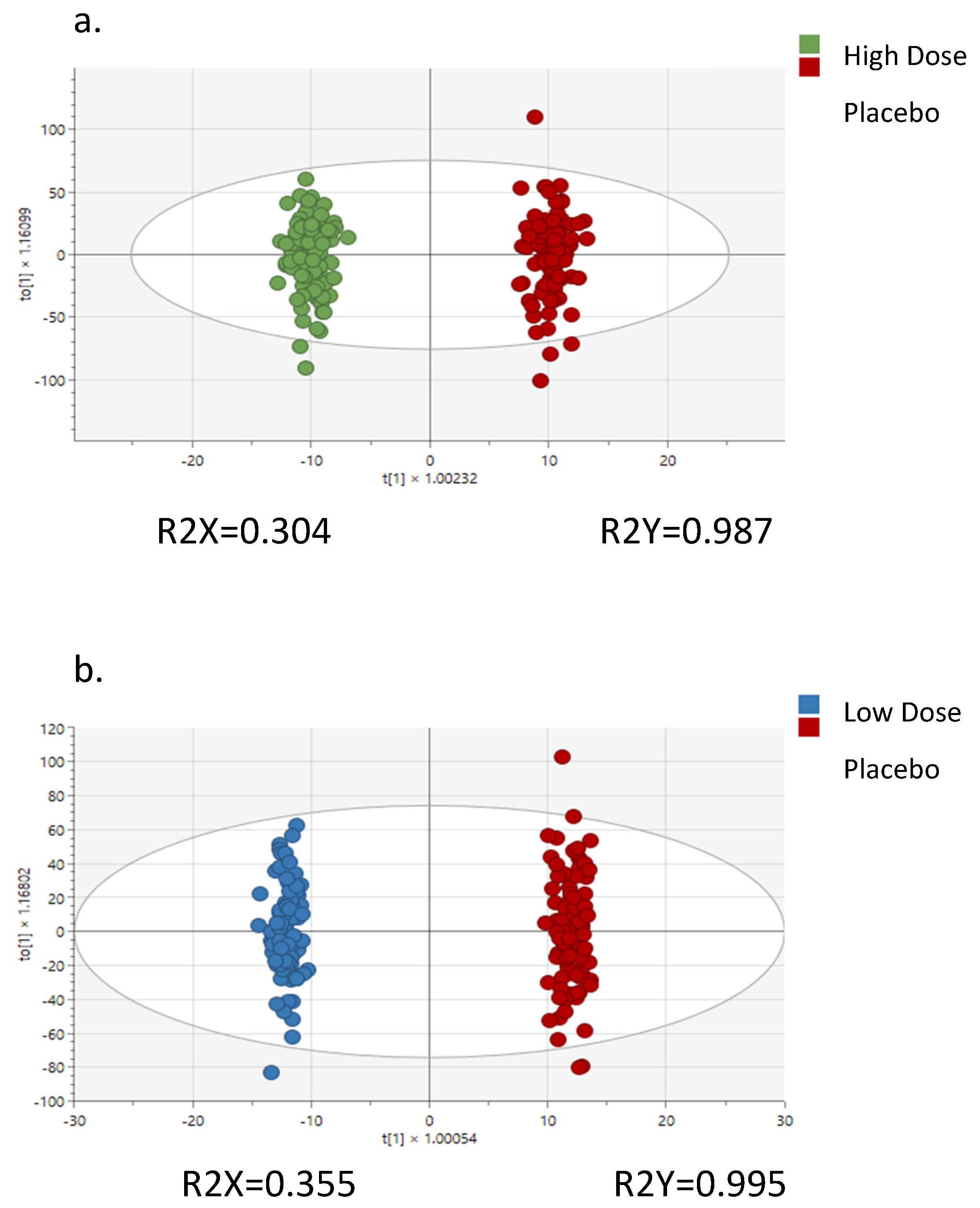
2.3.2. Plasma Untargeted Metabolomics Analysis and Statistical Procedures
2.4. Additional Statistical Procedures
3. Results
4. Discussion
Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ribeiro FM, Petriz B, Marques G, Kamilla LH, Franco OL. Is there an exercise-intensity threshold capable of avoiding the leaky gut? Front Nutr. 2021;8:627289. [CrossRef]
- Keirns BH, Koemel NA, Sciarrillo CM, Anderson KL, Emerson SR. Exercise and intestinal permeability: another form of exercise-induced hormesis? Am J Physiol Gastrointest Liver Physiol. 2020;319:G512-G518. [CrossRef]
- Chantler S, Griffiths A, Matu J, Davison G, Holliday A, Jones B. A systematic review: Role of dietary supplements on markers of exercise-associated gut damage and permeability. PLoS One. 2022;17:e0266379. [CrossRef]
- Dziewiecka H, Buttar HS, Kasperska A, Ostapiuk-Karolczuk J, Domagalska M, Cichoń J, Skarpańska-Stejnborn A. A systematic review of the influence of bovine colostrum supplementation on leaky gut syndrome in athletes: Diagnostic biomarkers and future directions. Nutrients. 2022;14:2512. [CrossRef]
- Tataka Y, Haramura M, Hamada Y, Ono M, Toyoda S, Yamada T, et al. Effects of oral cystine and glutamine on exercise-induced changes in gastrointestinal permeability and damage markers in young men. Eur J Nutr. 2022;61:2331-2339. [CrossRef]
- Nieman DC, Gillitt ND, Chen GY, Zhang Q, Sha W, Kay CD, et al. Blueberry and/or banana consumption mitigate arachidonic, cytochrome p450 oxylipin generation during recovery from 75-Km cycling: A randomized trial. Front Nutr. 2020;7:121. [CrossRef]
- Nieman DC, Kay CD, Rathore AS, Grace MH, Strauch RC, Stephan EH, et al. Increased plasma levels of gut-derived phenolics linked to walking and running following two weeks of flavonoid supplementation. Nutrients. 2018;10:1718. [CrossRef]
- Nieman DC, Gillitt ND, Knab AM, Shanely RA, Pappan KL, Jin F, Lila MA. Influence of a polyphenol-enriched protein powder on exercise-induced inflammation and oxidative stress in athletes: a randomized trial using a metabolomics approach. PLoS One. 2013;8:e72215. [CrossRef]
- Flores Martinez KE, Bloszies CS, Bolino MJ, Henrick BM, Frese SA. Hemp hull fiber and two constituent compounds, N-trans-caffeoyltyramine and N-trans-feruloyltyramine, shape the human gut microbiome in vitro. Food Chem X. 2024;23:101611. [CrossRef]
- van Klinken BJ, Stewart ML, Kalgaonkar S, Chae L. Health-promoting opportunities of hemp hull: The potential of bioactive compounds. J Diet Suppl. 2024;21:543-557. [CrossRef]
- Bolster D, Chae L, van Klinken JW, Kalgaonkar S. Impact of selected novel plant bioactives on improvement of impaired gut barrier function using human primary cell intestinal epithelium. J Food Bioact. 2022;20:11–16. [CrossRef]
- Lee SH, Veeriah V, Levine F. A potent HNF4α agonist reveals that HNF4α controls genes important in inflammatory bowel disease and Paneth cells. PLoS One. 2022;17:e0266066. [CrossRef]
- Smith LL, Brunetz MH, Chenier TC, McCammon MR, Houmard JA, Franklin ME, et al. The effects of static and ballistic stretching on delayed onset muscle soreness and creatine kinase. Res Q Exerc Sport. 1993;64:103–107. [CrossRef]
- Curran SL, Andrykowski MA, Studts J L. Short form of the Profile of Mood States (POMS-SF): Psychometric information. Psyc Assess. 1995;7:80–83. doi.org/10.1037/1040-3590.7.1.80.
- Khoshbin K, Khanna L, Maselli D, Atieh J, Breen-Lyles M, Arndt K, et al. Development and validation of test for “leaky gut” small intestinal and colonic permeability using sugars in healthy adults. Gastroenterology. 2021;161:463-475.e13. [CrossRef]
- Larkey NE, Fatica EM, Singh RJ. Detection of 13C-mannitol and other saccharides using tandem mass spectrometry for evaluation of intestinal permeability or leaky gut. Methods Mol Biol. 2022;2546:285-294. [CrossRef]
- Pathmasiri W, Rushing BR, McRitchie S, Choudhari M, Du X, Smirnov A, et al. Untargeted metabolomics reveal signatures of a healthy lifestyle. Sci Rep. 2024;14:13630. [CrossRef]
- Li YY, Ghanbari R, Pathmasiri W, McRitchie S, Poustchi H, Shayanrad A, et al. Untargeted metabolomics: biochemical perturbations in golestan cohort study opium users inform intervention strategies. Front Nutr. 2020;7:584585. [CrossRef]
- Ghanbari R, Li Y, Pathmasiri W, McRitchie S, Etemadi A, Pollock JD, et al. Metabolomics reveals biomarkers of opioid use disorder. Transl Psychiatry. 2021;11:103. [CrossRef]
- Lynch DH, Rushing BR, Pathmasiri W, McRitchie S, Batchek DJ, Petersen CL, et al. Baseline serum biomarkers predict response to a weight loss intervention in older adults with obesity: A pilot study. Metabolites. 2023;13:853. [CrossRef]
- Sun J, Xia Y. Pretreating and normalizing metabolomics data for statistical analysis. Genes Dis. 2023;11:100979. [CrossRef]
- Välikangas T, Suomi T, Elo LL. A systematic evaluation of normalization methods in quantitative label-free proteomics. Brief Bioinform. 2018;19:1-11. [CrossRef]
- Smirnov A, Liao Y, Fahy E, Subramaniam S, Du X. ADAP-KDB: A spectral knowledgebase for tracking and prioritizing unknown GC-MS spectra in the NIH’s Metabolomics Data Repository. Anal Chem. 2021;93:12213-12220. [CrossRef]
- Lynch DH, Rushing BR, Pathmasiri W, McRitchie S, Batchek DJ, Petersen CL, et al. Baseline serum biomarkers predict response to a weight loss intervention in older adults with obesity: a pilot study. Metabolites. 2023;13:853. [CrossRef]
- Chen X, Zhu X, Wu G, Wang X, Zhang Y, Jiang N. Structure-based identification of HNF4α agonists: Rosmarinic acid as a promising candidate for NAFLD treatment. Comput Struct Biotechnol J. 2024;27:171-183. [CrossRef]
- Deehan EC, Mocanu V, Madsen KL. Effects of dietary fibre on metabolic health and obesity. Nat Rev Gastroenterol Hepatol. 2024;21:301-318. [CrossRef]
- Mocanu V, Madsen KL. Dietary fibre and metabolic health: A clinical primer. Clin Transl Med. 2024;14(10):e70018. [CrossRef]
- Wang Z, Peters BA, Yu B, Grove ML, Wang T, Xue X, Thyagarajan B, Daviglus ML, Boerwinkle E, Hu G, Mossavar-Rahmani Y, Isasi CR, Knight R, Burk RD, Kaplan RC, Qi Q. Gut microbiota and blood metabolites related to fiber intake and type 2 diabetes. Circ Res. 2024;134(7):842-854. [CrossRef]
- Myhrstad MCW, Tunsjø H, Charnock C, Telle-Hansen VH. Dietary fiber, gut microbiota, and metabolic regulation-current status in human randomized trials. Nutrients. 2020;12(3):859. [CrossRef]
- Mahalak KK, Liu L, Bobokalonov J, Narrowe AB, Firrman J, Bittinger K, Hu W, Jones SM, Moustafa AM. Supplementation with soluble or insoluble rice-bran fibers increases short-chain fatty acid producing bacteria in the gut microbiota in vitro. Front Nutr. 2024 May 10;11:1304045. [CrossRef]
- Vinelli V, Biscotti P, Martini D, Del Bo’ C, Marino M, Meroño T, Nikoloudaki O, Calabrese FM, Turroni S, Taverniti V, Unión Caballero A, Andrés-Lacueva C, Porrini M, Gobbetti M, De Angelis M, Brigidi P, Pinart M, Nimptsch K, Guglielmetti S, Riso P. Effects of dietary fibers on short-chain fatty acids and gut microbiota composition in healthy adults: a systematic review. Nutrients. 2022 Jun 21;14(13):2559. [CrossRef]
- Fernández MA, García MD, Sáenz MT. Antibacterial activity of the phenolic acids fractions of Scrophularia frutescens and Scrophularia sambucifolia. J Ethnopharmacol. 1996;53(1):11-4. [CrossRef]
- Meng D, Sommella E, Salviati E, Campiglia P, Ganguli K, Djebali K, Zhu W, Walker WA. Indole-3-lactic acid, a metabolite of tryptophan, secreted by Bifidobacterium longum subspecies infantis is anti-inflammatory in the immature intestine. Pediatr Res. 2020 Aug;88(2):209-217. Epub 2020 Jan 16. [CrossRef] [PubMed] [PubMed Central]
- Alonso-Esteban JI, Pinela J, Ćirić A, Calhelha RC, Soković M, Ferreira ICFR, Barros L, Torija-Isasa E, Sánchez-Mata MC. Chemical composition and biological activities of whole and dehulled hemp (Cannabis sativa L.) seeds. Food Chem. 2022;374:131754. [CrossRef]
- van Wijck K, Lenaerts K, van Loon LJ, Peters WH, Buurman WA, Dejong CH. Exercise-induced splanchnic hypoperfusion results in gut dysfunction in healthy men. PLoS One. 2011;6:e22366. [CrossRef]
- Groschwitz KR, Hogan SP. Intestinal barrier function: molecular regulation and disease pathogenesis. J Allergy Clin Immunol. 2009;124:3-20; quiz 21-2. [CrossRef]
- Shu LZ, Ding YD, Xue QM, Cai W, Deng H. Direct and indirect effects of pathogenic bacteria on the integrity of intestinal barrier. Therap Adv Gastroenterol. 2023;16:17562848231176427. [CrossRef]
- Peters HP, Bos M, Seebregts L, Akkermans LM, van Berge Henegouwen GP, Bol E, Mosterd WL, de Vries WR. Gastrointestinal symptoms in long-distance runners, cyclists, and triathletes: prevalence, medication, and etiology. Am J Gastroenterol. 1999;94:1570-81. [CrossRef]
- Nieman DC, Henson DA, Dumke CL, Oley K, McAnulty SR, Davis JM, Murphy EA, Utter AC, Lind RH, McAnulty LS, Morrow JD. Ibuprofen use, endotoxemia, inflammation, and plasma cytokines during ultramarathon competition. Brain Behav Immun. 2006;20:578-84. [CrossRef]
- Chelakkot C, Ghim J, Ryu SH. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp Mol Med. 2018;50(8):1-9. [CrossRef]
- Quaresma MVLDS, Mancin L, Paoli A, Mota JF. The interplay between gut microbiome and physical exercise in athletes. Curr Opin Clin Nutr Metab Care. 2024;27(5):428-433. [CrossRef]
- Nolte S, Krüger K, Lenz C, Zentgraf K. Optimizing the Gut Microbiota for Individualized Performance Development in Elite Athletes. Biology (Basel). 2023;12:1491. [CrossRef]
- Han M, Yang K, Yang P, Zhong C, Chen C, Wang S, Lu Q, Ning K. Stratification of athletes’ gut microbiota: the multifaceted hubs associated with dietary factors, physical characteristics and performance. Gut Microbes. 2020;12:1-18. [CrossRef]
- Plamada D, Vodnar DC. Polyphenols-gut microbiota interrelationship: a transition to a new generation of prebiotics. Nutrients. 2021;14:137. [CrossRef]
| Ingredient name | High dose Weight (g) |
Low dose Weight (g) |
Placebo Weight (g) |
|---|---|---|---|
| Slurry Binder | 27.45 | 27.45 | 0 |
| Hemp Hull Powder | 10.00 | 2.50 | 0 |
| Colorant | 0.05 | 0.10 | 0.30 |
| Sunflower oil | 1.00 | 1.00 | 1.00 |
| Plain Rice Crisps | 11.50 | 11.50 | 11.50 |
| Milk powder blend | 0 | 7.45 | 9.75 |
| Tapioca and rice flour binder | 0 | 0 | 27.45 |
| Total weight (g) | 50.00 | 50.00 | 50.00 |
| Kilocalories | 182 | 192 | 189 |
| % carbohydrate | 84 | 77 | 75 |
| % fat | 10 | 16 | 18 |
| % protein | 5 | 7 | 7 |
| Sex | Mean±SE | |
|---|---|---|
| Age (yrs) | M | 45.5±2.2 |
| F | 46.5±4.1 | |
| Weight (kg) | M | 79.2±2.1* |
| F | 61.7±2.9 | |
| Height (cm) | M | 181±1.4* |
| F | 165±0.6 | |
| BMI (kg/m2) | M | 24.2±0.5 |
| F | 22.6±1.1 | |
| Body fat (%) | M | 19.4±1.5* |
| F | 28.0±2.0 | |
| V02max (ml.kg.-1min-1) | M | 43.5±1.7* |
| F | 34.5±3.0 | |
| Max watts | M | 265±10.9* |
| F | 167±19.0 | |
| Max heart rate (beats/min) | M | 172±2.7 |
| F | 171±5.5 | |
| Max ventilation (L/min) | M | 128±6.0* |
| F | 79.0±7.5 | |
| Max respiratory rate (breaths/min) | M | 47.1±2.0 |
| F | 40.0±1.5 |
| Performance Measurement | Supplement | Mean±SE |
|---|---|---|
| Cycling power (watts, % maximum) |
High dose hemp | 138±9.3 (57.2±1.8% max) |
| Low dose hemp | 138±8.6 (57.6±1.4% max) | |
| Placebo | 137±8.6 (57.1±1.3% max) | |
| Heart rate (beats/min, % maximum) |
High dose hemp | 134±3.4 (78.4±1.5% max) |
| Low dose hemp | 133±3.1 (77.5±1.4% max) | |
| Placebo | 133±3.6 (77.5±1.8% max) | |
| Oxygen consumption (VO2) (ml.kg.-1min-1, % maximum) | High dose hemp | 30.1±1.1 (73.8±1.5% max) |
| Low dose hemp | 29.9±1.2 (73.1±1.5% max) | |
| Placebo | 29.5±1.2 (72.2±1.5% max) | |
| Distance cycled (km) |
High dose hemp | 66.1±2.1 |
| Low dose hemp | 66.2±1.6 | |
| Placebo | 65.8±1.8 | |
| Speed (km/h) |
High dose hemp | 29.0±0.9 |
| Low dose hemp | 29.3±0.7 | |
| Placebo | 28.6±0.8 |
| High-Dose Hemp | Low-Dose Hemp | Placebo | Time & interaction effects, p-values |
||||
|---|---|---|---|---|---|---|---|
| Pre-Suppl | Post-Exerc | Pre-Suppl | Post-Exerc | Pre-Suppl | Post-Exerc | ||
| L:C13M | 0.518±0.051 | 0.780±0.230 | 0.450±0.028 | 0.613±0.077 | 0.498±0.039 | 0.490±0.028 | 0.108; 0.195 |
| L:C12M | 0.031±0.003 | 0.053±0.002 | 0.027±0.002 | 0.037±0.004 | 0.033±0.003 | 0.031±0.002 | 0.070; 0.172 |
| VIP | Metabolites | Description |
|---|---|---|
| 2.3 | Uridine | A pyrimidine nucleoside involved with many biological processes including RNA, glycogen, and biomembrane synthesis. |
| 2.0 | Linoleic acid | An essential polyunsaturated fatty acid (18:2ω6). |
| 1.9 | Uric acid | Chemical created when purines are metabolized. Uric acid is a significant antioxidant in the human body. |
| 1.9 | Valyl-Serine | Dipeptide formed from L-valine and L-serine residues. Incomplete breakdown product of protein digestion or protein catabolism |
| 1.9 | O-Cresol | A derivative of phenol and an isomer of p-cresol and m-cresol. Phenol is primarily used to synthesize plastics and related materials. |
| 1.9 | 3-hydroxy-4-methoxybenzoic acid | A plant metabolite (isovanillic acid) with antibacterial properties. |
| 1.8 | Stearidonic acid | A plant-based omega-3 fatty acid (18:3 n-3) that increases the levels of long-chain omega-3 PUFAs such as EPA. |
| 1.8 | Glycerophosphocholine | A choline derivative involved in multiple brain functions. |
| 1.8 | Creatinine | An endogenous product of muscle metabolism. |
| 1.8 | 2-Aminoheptanoate | An alpha amino acid. May enhance the effect of ketones as fuel to the Krebs cycle in the brain. |
| 1.8 | Quinaldic acid | A kynurenine metabolite. |
| 1.7 | Phenylacetylglutamine | A novel metabolite derived from gut microbial metabolism of dietary proteins, specifically phenylalanine, which may be linked to risks of adverse cardiovascular events. |
| 1.7 | Indolelactic acid | Formed primarily from gut bacterial metabolism of tryptophan. Functions as an anti-inflammatory molecule. |
| 1.5 | Serotonin | Made from tryptophan, an essential amino acid. A chemical messenger that affects mood, sleep, digestion. |
| 1.5 | N-Butyrylglycine | An acyl glycine that is a minor metabolite of fatty acids. |
| 1.5 | Cytidine | A pyrimidine nucleoside that serves as a precursor for uridine and is involved in RNA synthesis. |
| 1.5 | S-Allylcysteine | An organosulfur compound that exhibits antioxidant, anti-inflammatory, and redox modulatory activities |
| 1.5 | Adipoyl-L-carnitine | An acylcarnitine. |
| 1.5 | 12,13-DiHOME | An inflammatory oxylipin. |
| 1.5 | N-Acetylmethionine | A derivative of methionine. |
| 1.5 | Calcifediol | The precursor for calcitriol, the active form of vitamin D. |
| 1.4 | 5-Hydroxytryptophan | Metabolite of tryptophan and the immediate precursor of the neurotransmitter serotonin |
| 1.4 | 4-Methoxycinnamic acid | A methyl derivative of ferulic acid that has been found in hemp fibers. |
| 1.4 | Proline | An amino acid important in protein synthesis, nutrition metabolism, wound healing and immunity, and antioxidative reactions. |
| 1.4 | 5-Aminolevulinic acid | An amino acid that is the first compound in the porphyrin synthesis pathway leading to heme. |
| 1.4 | Procyanidin B1 | A flavonoid group of condensed flavan-3-ols that can be found in many plants. |
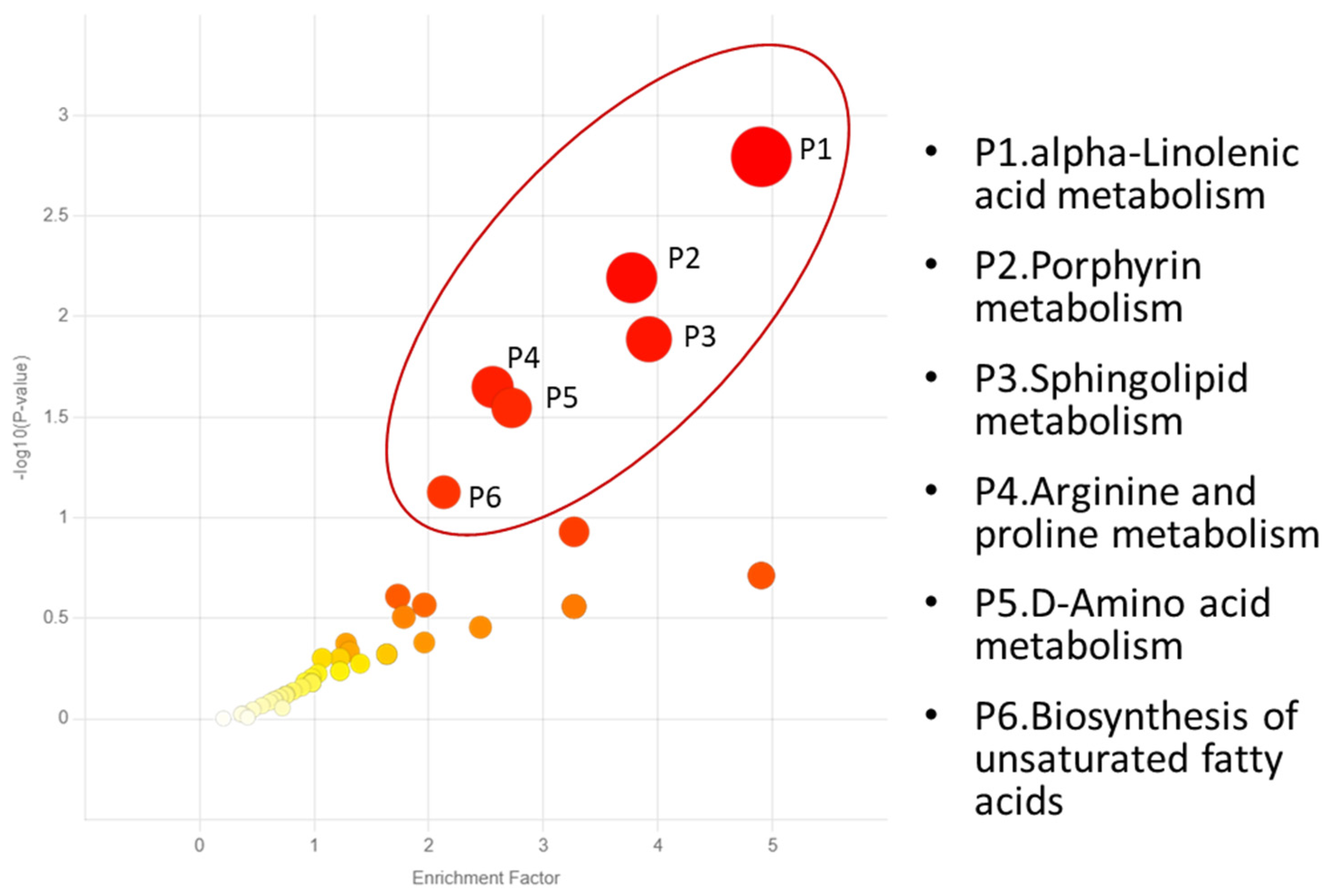
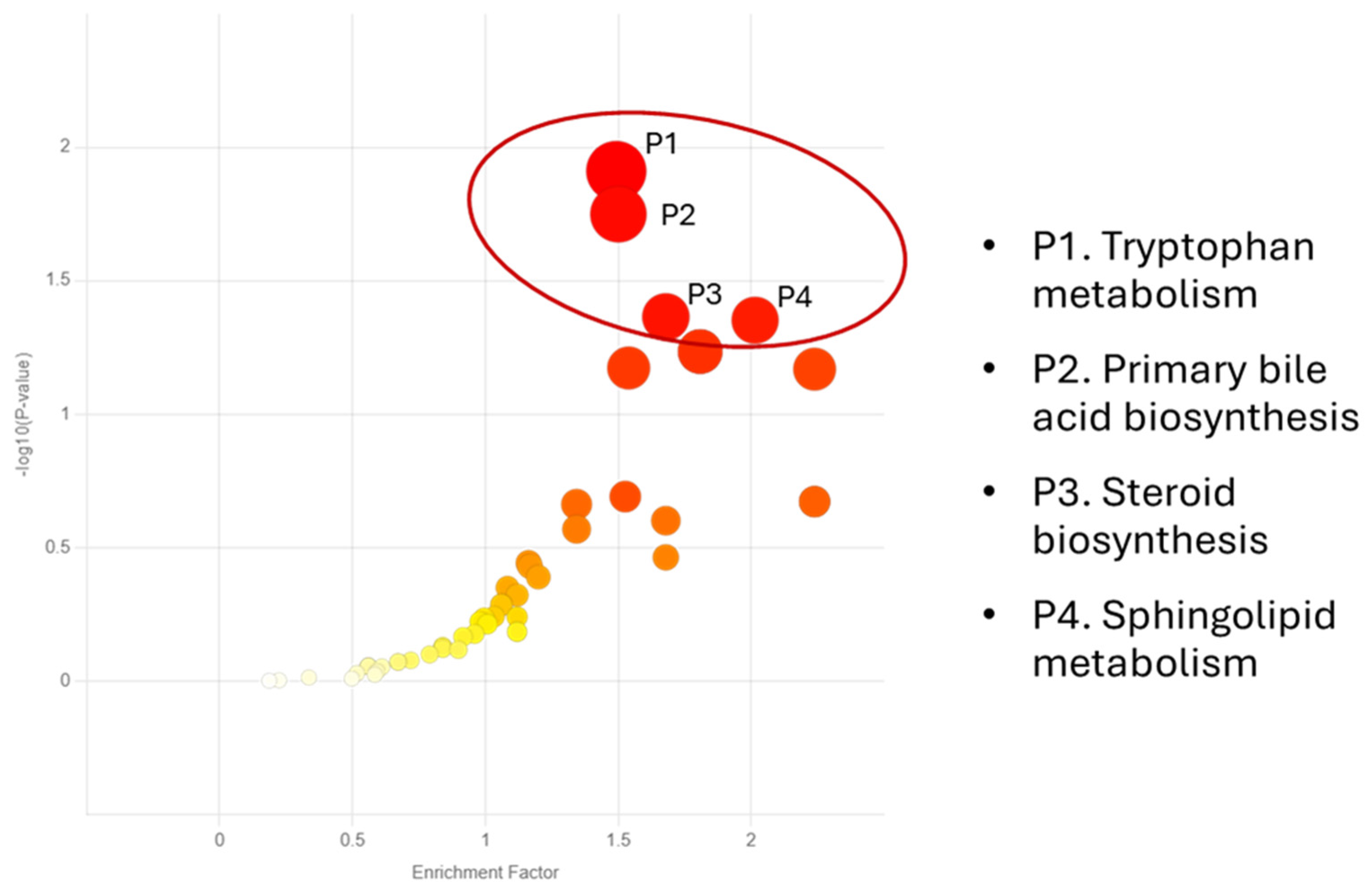
| PATHWAY | KEGG ID and METABOLITES |
|---|---|
| P1. Alpha-linolenic acid metabolism | C06427 Alpha-linolenic acid C16300 Stearidonic acid |
| P2. Porphyrin metabolism | C00931 Porphobilinogen C00430 5-Aminolevulinic acid C00486 Bilirubin |
| P3. Sphingolipid metabolism | C06124 Sphingosine 1-phosphate C00836 Sphinganine C00319 Sphingosine |
| P4. Arginine and proline metabolism | C00555 4-Aminobutyraldehyde C05147 Trans-3-hydroxy-L-proline C00763 D-Proline C01157 4-Hydroxyproline C01165 L-Glutamic gamma-semialdehyde C00077 Ornithine C03564 1-Pyrroline-2-carboxylic acid C00022 Pyruvic acid C03912 1-Pyrroline-5-carboxylic acid |
| P5. D-Amino acid metabolism | C00819 D-Glutamine C00515 D-Ornithine C00763 D-Proline C03440 cis-4-Hydroxy-D-proline C01110 5-Amino-2-oxopentanoic acid C03564 1-Pyrroline-2-carboxylic acid |
|
P6. Biosynthesis of unsaturated fatty acids |
C00712 Oleic acid C01595 Linoleic acid C00219 Arachidonic acid C06426 gamma-Linolenic acid C06428 Eicosapentaenoic acid C06427 alpha-Linolenic acid |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




