Submitted:
21 March 2025
Posted:
24 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
-
Treatment of minerals with sulfur-containing reagents:
- Minerals are treated with sulfidizing agents such as sodium sulfide (Na₂S), sodium or ammonium polysulfides, hydrogen sulfide (H₂S), or other compounds that can interact with the oxidized surfaces.
-
Sulfidization reaction:
- The reagent interacts with the surface of oxidized minerals, forming a sulfide film on the surface. For example:PbO + Na₂S → PbS↓ + Na₂OPbCO₃ + Na₂S → PbS↓ + Na₂CO₃
- As a result, the oxide and carbonate compounds of lead are converted into lead sulfide.
-
Fixation of the sulfide film:
- The sulfide film formed on the surface of the minerals improves their hydrophobicity, which facilitates the attachment of flotation reagents and air bubbles during flotation.
-
Flotation:
- The modified sulfide minerals are extracted by flotation using xanthates or other collector reagents that effectively interact with the sulfide surface.
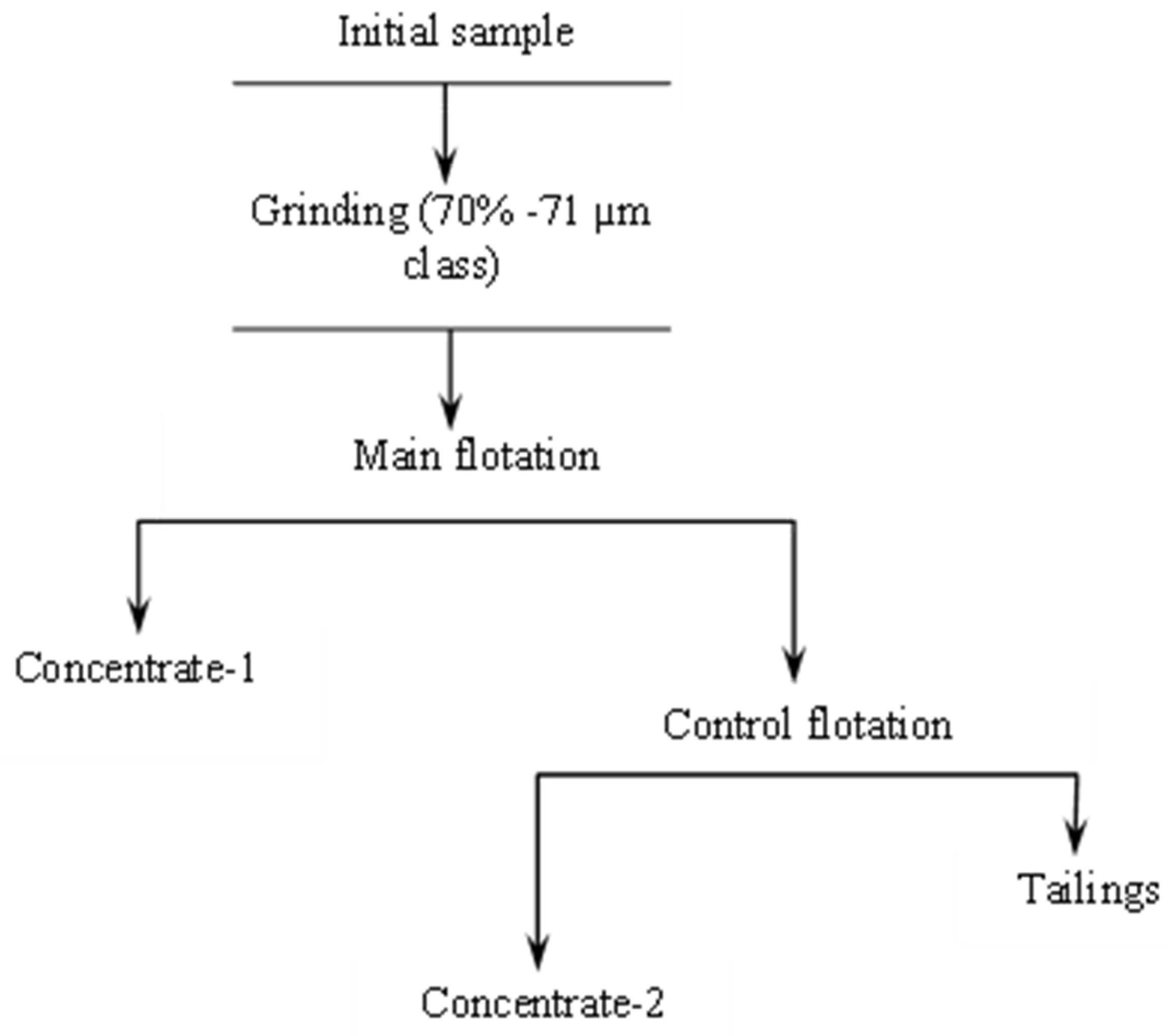
2. Materials and Methods
3. Results and Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- https://dknews.kz/ru/ekonomika/105991-v-kazahstane-ezhegodno-proizvoditsya-300-tysyach-tonn.
- Merkibayev, Y.S. Processing of poor hard-to-process complex lead-zinc ores and enrichment intermediate products // Dissertation for the degree of Doctor of Philosophy (PhD), Almaty, 2024, p.187. Available online: https://official.satbayev.university/ru/protection.
- Nayak, A., Jena, M.S., Mandre, N.R. Beneficiation of lead–zinc ores—a review // Miner. Process. Extr. Metall. Rev., 43 (5) (2022), pp. 564-583. [CrossRef]
- Yuangan Chen, Yongsheng Suna, Yuexin Hana. Efficient flotation separation of lead–zinc oxide ores using mineral sulfidation reconstruction technology: A review // Green and Smart Mining Engineering 1 (2024), pp.175–189. Available online: https://www.sciencedirect.com/.
- Turysbekov D., Semushkina L., Narbekovа S., Mukhamedilova A. On the use of environmentally safe sulfidizator in the beneficiation of ores // Proceedings of the Universities of Kyrgyzstan, № 8, 2019, p. 24-29. Available online: https://elibrary.ru.
- Hongliang Zhang, Heng Yu, Wei Sun, Shangyong Lin, Chenyang Zhang. Beneficiation of silver and silver-bearing lead–zinc ores: A review // Minerals Engineering Volume 208, March 2024.
- Chepushtanova T.A., Motovilov I.Yu, Merkibayev Y.S., Polyakov K.V., Gostu S. Flotation studies of the middling product of lead-zinc ores with preliminary sulfidizing roasting of oxidized lead and zinc compounds // KIMS. Kazakhstan №4 (323), 2022, p. 78-83. Available online: https://kims-imio.kz/wp-content/uploads/2024/04/2022-4-10.pdf.
- Motovilov I.Yu., Barmenshinova M.B., Telkov Sh.A., Omar R.S. Study of the chemical composition and assessment of the gravity beneficiation of oxidized polymetallic ores of the RK deposit // Mining Journal of Kazakhstan, No. 9, 2024. pp. 51-58. Available online: https://minmag.kz/wp-content/uploads/2024/10/2409_51-58.pdf.
- Majid Ejtemaei, Mahdi Gharabaghi, Mehdi Irannajad. A review of zinc oxide mineral beneficiation using flotation method // Advances in Colloid and Interface Science Volume 206, April 2014, pp. 68-78.
- Wonder Chimonyo, Kirsten Corin, Jenny Wiese and Cyril O’Connor. Redox potential control during flotation of sulphide minerals // Centre for Minerals Research, Department of Chemical Engineering, University of Cape Town.—November, 2015. Available online: https://www.researchgate.net/publication/283510716_Redox_potential_control_during_flotation_of_sulphide_minerals.
- Oskembekov I.M., Bekturganov N.S., Katkeeva G.L., Burkitseterkyzy G., Gizatullina D.R. Use of the sulphidation process in the processing of oxidised copper ores // KIMS, 2017, рр.16-22. Available online: https://kims-imio.com.
- Dospaev M.M., Figurinene I.V., Gabdullin S.T. Combined electrochemical sulphidisation of difficult to enrich oxidised copper ores // Obogashchenie Rud, 2024, pp. 19-24.
- Xojimuratova X. B., Abdusamieva L. N. Method for Processing Sulphide-Oxidized Copper Ores with Copper and Silver Extraction //EUROPEAN JOURNAL OF INNOVATION IN NONFORMAL EDUCATION.—2024.—Т. 4.—№. 3.—р. 510-513. https://api.scienceweb.uz/storage/publication_files/9820/26493/666bdfded87a5___510_513+Method+for+Processing+Sulphide_Oxidized+Copper+Ores+wit.pdf.
- Li, J.L., Liu, S.Y., Liu, D.W., Liu, R.Z., Liu, Z.C., Jia, X.D., Chang, T.C., Sulfidization mechanism in the flotation of cerussite: a heterogeneous solid-liquid reaction that yields PbCO3/PbS core–shell particles, Miner. Eng. 153 (2020).
- Yuan, W.Y., Li, J.H., Zhang, Q.W., Saito, F., Mechanochemical sulfidization of lead oxides by grinding with sulfur, Powder Technol. 230 (2012), p.63–66. Available online: https://www.researchgate.net/publication/262725367_Mechanochemical_Sulfidization_of_Lead_Oxides_by_Grinding_with_Sulfur.
- Anthropova, I.G., Merinov, А.A., Gulyashinov, Р.A., Damdinov, B.B.. Sulfidation of oxidized lead and zinc with pyrite-bearing lead-and-zinc ore // Physical and technical problems of mineral resources development. July 2023.
- Oskembekov, I.M., Burkitseterkyzy G., Akubaeva, M.A., Gizatullina D.R., Zhunussov E.M. Use of the sulphidation process in the processing of oxidised copper ores // KIMS, 2017, рр.16-22. Available online: https://kims-imio.com.
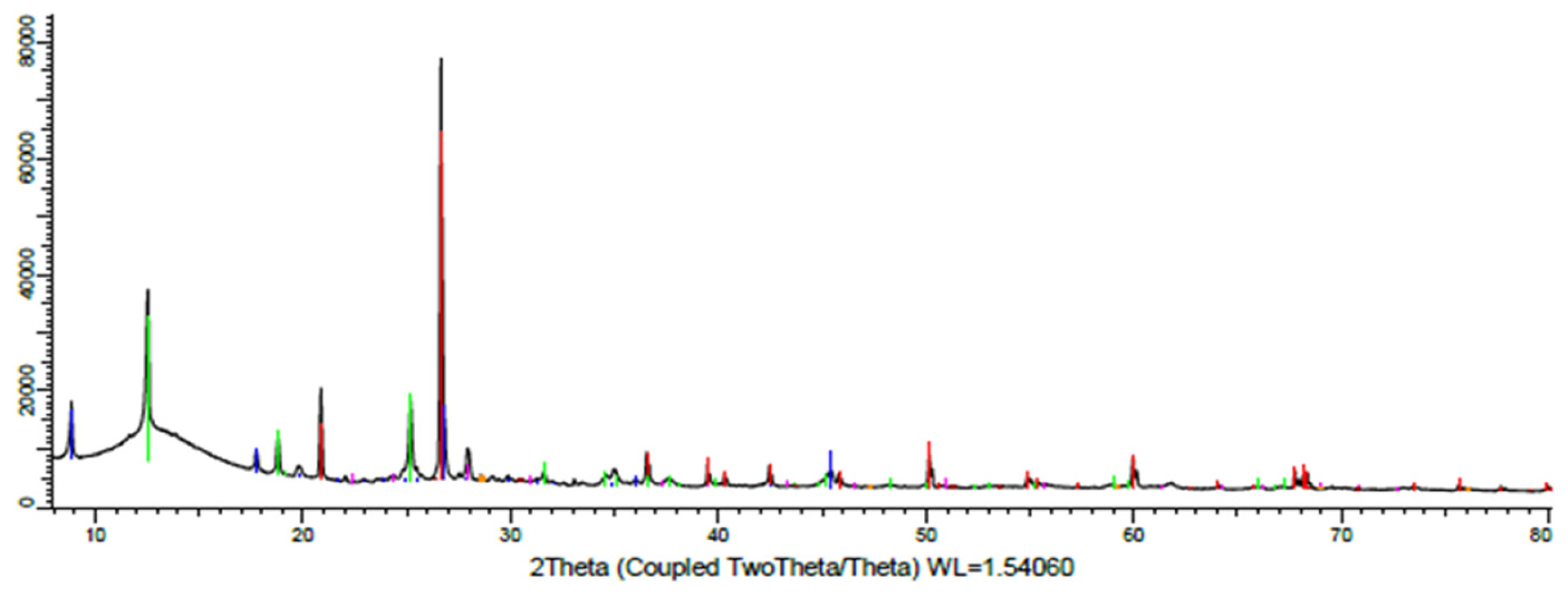


| Component | Cu | Pb | Zn | Fe | СаО | MgO | As |
| Mass Fraction, % | 0,02 | 0,90 | 0,70 | 6,42 | 1,33 | 3,35 | 0,023 |
| Component | SiO2 | Al2O3 | Au, g/ton* |
Au, g/ton |
Na | S total | K |
| Mass Fraction, % | 55,23 | 26,34 | 0,51 | 6,1 | 0,48 | 0,17 | 3,1 |
| The form of occurrence of the metal. | Content, % (absolute). | Distribution, % (relative). |
| Zn of oxidized minerals. | 0,59 | 84,72 |
| Zn of sulfide minerals (sphalerite). | 0,11 | 15,28 |
| Total. | 0,70 | 100,0 |
| Pb of oxidized minerals | 0,72 | 79,69 |
| Pb of sulfide minerals (galena) | 0,18 | 20,31 |
| Total | 0,90 | 100,0 |
| Main minerals and complexes. | Content, % |
| Galena | 0,16 |
| Oxidized and residual forms of lead. | 0,89 |
| Sphalerite | 0,12 |
| Oxidized and insoluble forms of zinc. | 1,07 |
| Copper minerals. | 0,04 |
| Pyrite | 0,05 |
| Goethite, hematite | 4,51 |
| Mica minerals. | 53,44 |
| Quartz | 30,95 |
| Others | 8,77 |
| Total | 100,0 |
| Consumption of Na2S (g/t) and redox potential (mV). | Product | Yield, % | Content, % | Extraction, % | ||
| Zn | Pb | Zn | Pb | |||
| Test 1 | ||||||
| 0 (100) | Concentrate-1 | 3,45 | 1,92 | 7,43 | 9,46 | 28,47 |
| Concentrate-2 | 0,98 | 2,34 | 8,28 | 3,28 | 9,02 | |
| ΣConcentrate | 4,43 | 2,01 | 7,62 | 12,74 | 37,48 | |
| Tailings | 95,57 | 0,64 | 0,59 | 87,26 | 62,52 | |
| Initial sample | 100,0 | 0,70 | 0,90 | 100,0 | 100,0 | |
| Test 2 | ||||||
| 500 (-120) | Concentrate-3 | 3,76 | 1,74 | 9,48 | 9,34 | 39,60 |
| Concentrate-4 | 1,09 | 2,08 | 5,81 | 3,24 | 7,03 | |
| Σ Concentrate | 4,85 | 1,82 | 8,65 | 12,58 | 46,63 | |
| Tailings | 95,15 | 0,64 | 0,51 | 87,42 | 53,37 | |
| Initial sample | 100,0 | 0,70 | 0,90 | 100,0 | 100,0 | |
| Test 3 | ||||||
| 600 (-160) | Concentrate-3 | 4,02 | 1,63 | 9,13 | 9,36 | 40,76 |
| Concentrate-4 | 1,19 | 1,95 | 5,21 | 3,31 | 6,89 | |
| ΣConcentrate | 5,21 | 1,70 | 8,23 | 12,67 | 47,65 | |
| Tailings | 94,79 | 0,65 | 0,50 | 87,33 | 52,35 | |
| Initial sample | 100,0 | 0,70 | 0,90 | 100,0 | 100,0 | |
| Test 4 | ||||||
| 700 (-200) | Concentrate-5 | 4,12 | 1,39 | 9,18 | 8,18 | 42,03 |
| Concentrate-6 | 1,28 | 1,87 | 5,65 | 3,42 | 8,03 | |
| ΣConcentrate | 5,40 | 1,50 | 8,34 | 11,60 | 50,07 | |
| Tailings | 94,60 | 0,65 | 0,48 | 88,40 | 49,93 | |
| Initial sample | 100,0 | 0,70 | 0,90 | 100,0 | 100,0 | |
| Test 5 | ||||||
| 800 (-260) | Concentrate-5 | 4,08 | 1,42 | 8,86 | 8,28 | 40,18 |
| Concentrate-6 | 1,30 | 1,75 | 5,44 | 3,24 | 7,86 | |
| ΣConcentrate | 5,38 | 1,50 | 8,03 | 11,52 | 48,04 | |
| Tailings | 94,62 | 0,66 | 0,49 | 88,48 | 51,96 | |
| Initial sample | 100,0 | 0,70 | 0,90 | 100,0 | 100,0 | |
| Test 6 | ||||||
| 900 (-310) | Concentrate-5 | 5,42 | 0,97 | 6,73 | 7,49 | 40,52 |
| Concentrate-6 | 1,06 | 1,97 | 5,15 | 2,98 | 6,06 | |
| ΣConcentrate | 6,48 | 1,13 | 6,47 | 10,47 | 46,58 | |
| Tailings | 93,52 | 0,67 | 0,51 | 89,53 | 53,42 | |
| Initial sample | 100,0 | 0,70 | 0,90 | 100,0 | 100,0 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




