Submitted:
20 March 2025
Posted:
21 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction to Liposomal and Nanocarrier Drug Delivery
1.1. Evolution of Liposomal Drug Delivery and Initial Breakthroughs
1.2. Emergence of Nanocarrier Systems and Their Impact on Therapeutics
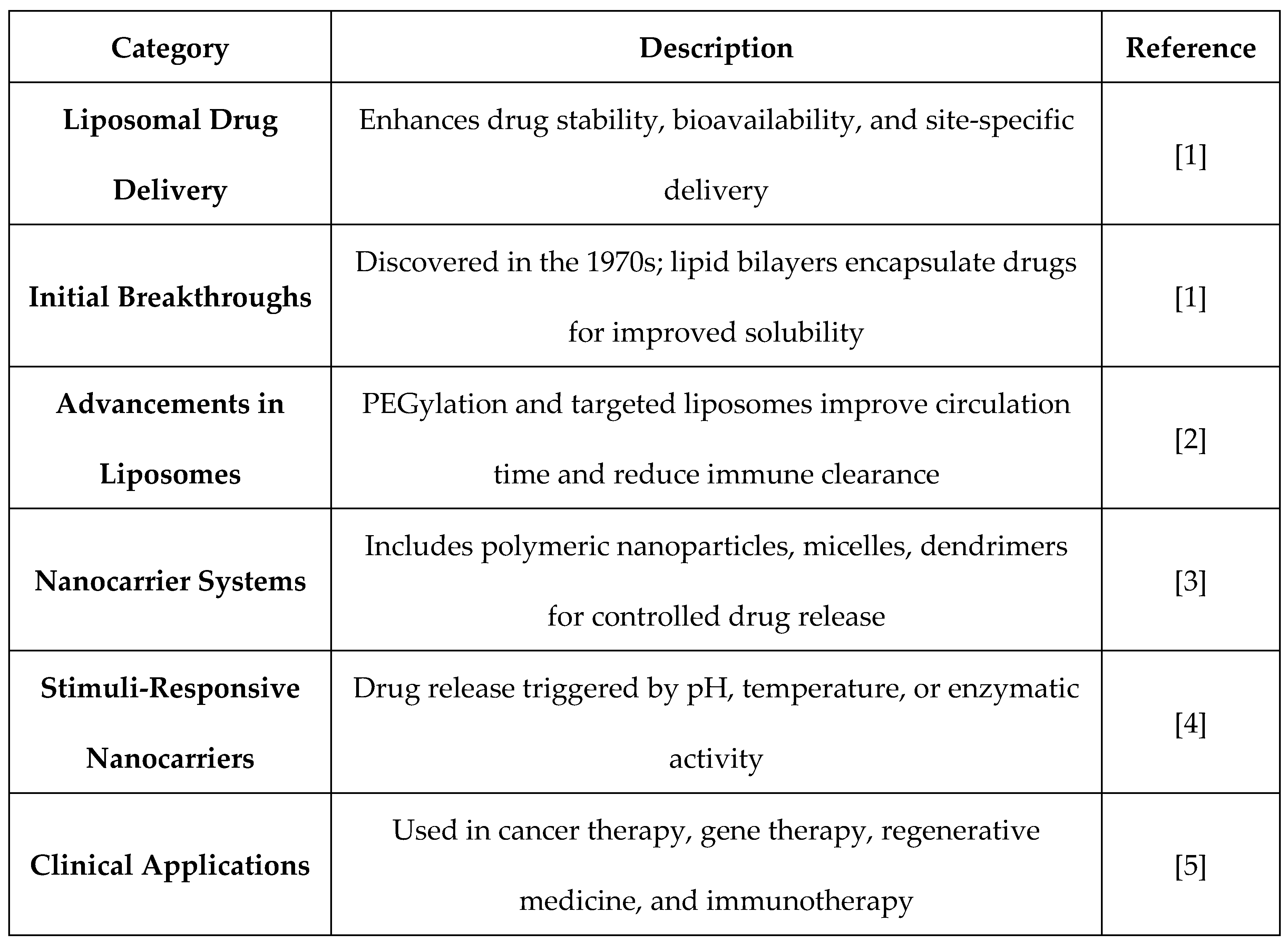
2. Liposomal Drug Delivery Mechanisms and Structural Composition
2.1. Liposome Formation, Classification, and Functionalization
2.2. Mechanisms of Drug Encapsulation and Release Kinetics
3. Advancements in Liposomal Formulations for Targeted Therapy
3.1. Stealth Liposomes and Their Role in Prolonged Circulation
3.2. Liposome-Based Cancer Therapy and FDA-Approved Formulations
4. Comparative Analysis of Liposomes with Other Nanocarriers
4.1. Micellar Nanocarriers and Solid Lipid Nanoparticles (SLNs)
4.2. Polymeric Nanoparticles and Hybrid Vesicular Systems
5. Challenges, Limitations, and Future Prospects in Liposomal Drug Delivery
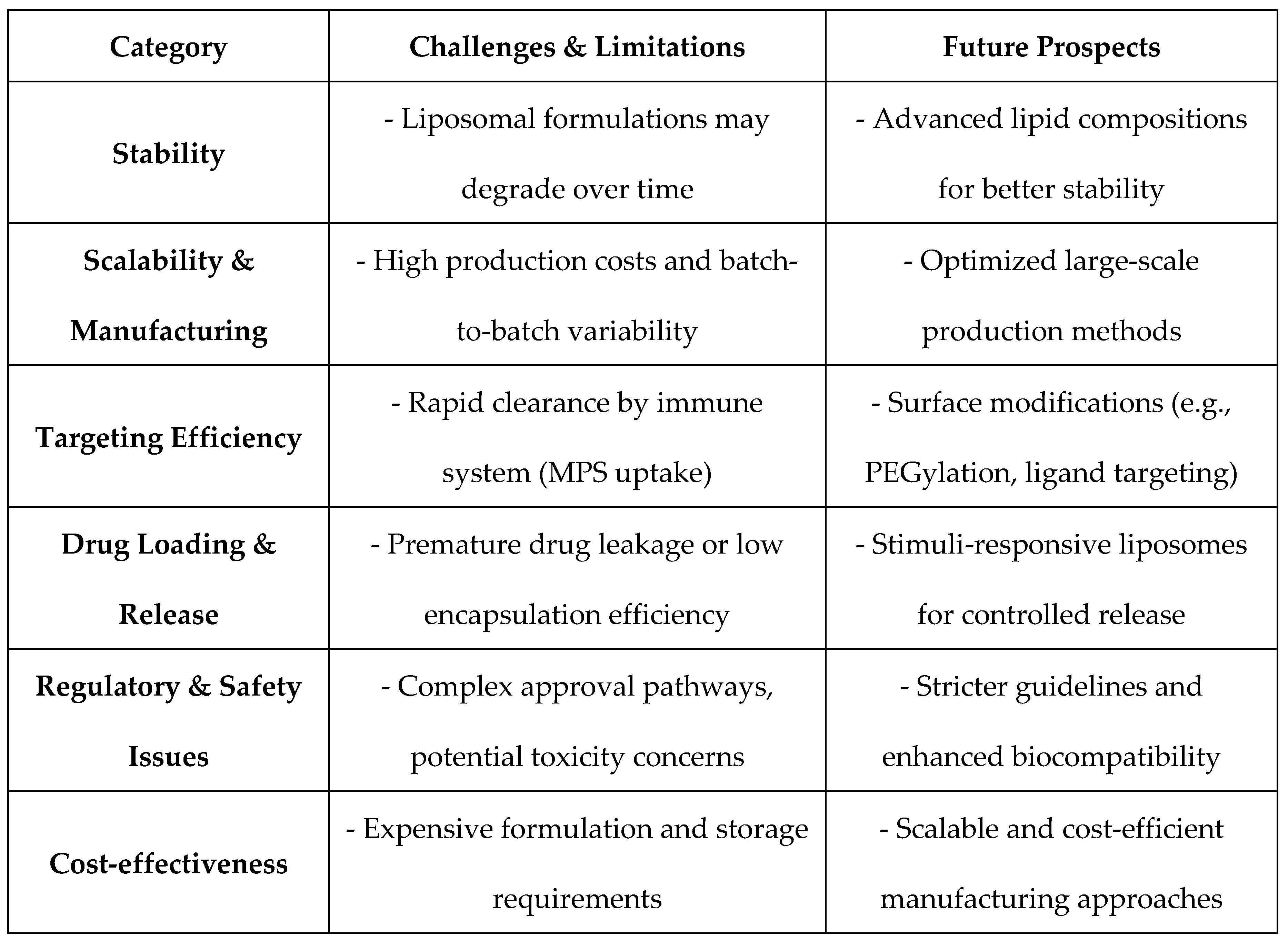
5.1. Current Limitations in Stability, Scalability, and Targeting Efficiency
5.2. Emerging Trends in Nanomedicine for Next-Gen Therapeutics
Conclusions
References
- Gregoriadis, G. (1976). The carrier potential of liposomes in biology and medicine. New England Journal of Medicine, 295(14), 765-770. [CrossRef]
- Allen, T. M., & Cullis, P. R. (2013). Liposomal drug delivery systems: From concept to clinical applications. Advanced Drug Delivery Reviews, 65(1), 36-48. [CrossRef]
- Peer, D., Karp, J. M., Hong, S., Farokhzad, O. C., Margalit, R., & Langer, R. (2007). Nanocarriers as an emerging platform for cancer therapy. Nature Nanotechnology, 2(12), 751-760. [CrossRef]
- Moghimi, S. M., Hunter, A. C., & Murray, J. C. (2005). Nanomedicine: Current status and future prospects. The FASEB Journal, 19(3), 311-330.
- Farokhzad, O. C., & Langer, R. (2009). Impact of nanotechnology on drug delivery. ACS Nano, 3(1), 16-20. [CrossRef]
- Akbarzadeh, A., Rezaei-Sadabady, R., Davaran, S., Joo, S. W., Zarghami, N., Hanifehpour, Y., Samiei, M., Kouhi, M., & Nejati-Koshki, K. (2013). Liposome: Classification, preparation, and applications. Nanoscale Research Letters, 8(1), 102.
- Sharma, A., & Sharma, U. S. (1997). Liposomes in drug delivery: Progress and limitations. International Journal of Pharmaceutics, 154(2), 123-140.
- Garg, T., & Goyal, A. K. (2014). Liposomes: Targeted and controlled delivery system. Drug Delivery Letters, 4(1), 62-71. [CrossRef]
- Bozzuto, G., & Molinari, A. (2015). Liposomes as nanomedical devices. International Journal of Nanomedicine, 10, 975-999.
- Paliwal, R., Paliwal, S. R., Mishra, N., & Mehta, A. (2011). Engineered polymeric nanoparticles: Current status and future directions. Expert Opinion on Drug Delivery, 8(4), 469-482.
- Immordino, M. L., Dosio, F., & Cattel, L. (2006). Stealth liposomes: Review of the basic science, rationale, and clinical applications, existing and potential. International Journal of Nanomedicine, 1(3), 297-315.
- Torchilin, V. P. (2005). Recent advances with liposomes as pharmaceutical carriers. Nature Reviews Drug Discovery, 4(2), 145-160. [CrossRef]
- Barenholz, Y. (2012). Doxil®—the first FDA-approved nano-drug: Lessons learned. Journal of Controlled Release, 160(2), 117-134.
- Bulbake, U., Doppalapudi, S., Kommineni, N., & Khan, W. (2017). Liposomal formulations in clinical use: An updated review. Pharmaceutics, 9(2), 12. [CrossRef]
- Silverman, J. A., & Elmer, G. W. (1990). Liposomal amphotericin B (AmBisome) for the treatment of systemic fungal infections. Antimicrobial Agents and Chemotherapy, 34(7), 1281-1283.
- Torchilin, V. P. (2006). Micellar nanocarriers: Pharmaceutical perspectives. Pharmacology & Therapeutics, 120(2), 119-127. [CrossRef]
- Müller, R. H., Mäder, K., & Gohla, S. (2000). Solid lipid nanoparticles (SLN) for controlled drug delivery – A review of the state of the art. European Journal of Pharmaceutics and Biopharmaceutics, 50(1), 161-177.
- Sercombe, L., Veerati, T., Moheimani, F., Wu, S. Y., Sood, A. K., & Hua, S. (2015). Advances and challenges of liposome assisted drug delivery. Frontiers in Pharmacology, 6, 286. [CrossRef]
- Sengar, A., Tile, S. A., Sen, A., Malunjkar, S. P., Bhagat, D. T., & Thete, A. K. (2024). Effervescent tablets explored: Dosage form benefits, formulation strategies, and methodological insights. World Journal of Pharmaceutical Research, 13(18), 1424-1435.
- Sengar, A., Saha, S., Sharma, L., Hemlata, Saindane, P. S., & Sagar, S. D. (2024). Fundamentals of proniosomes: Structure & composition, and core principles. World Journal of Pharmaceutical Research, 13(21), 1063-1071.
- Sengar, A. (2024). Liposomes and beyond: Pioneering vesicular systems for drug delivery. Preprints. [CrossRef]
- Sengar, A. (2024). Precision in practice: Nanotechnology and targeted therapies for personalized care. International Journal of Advanced Nano Computing and Analytics, 3(2), 56-67. [CrossRef]
- Sengar, A. (2025). Liposomal drug delivery systems: An intro as a primer for advanced therapeutics. Preprints. [CrossRef]
- Bozzuto, G., & Molinari, A. (2015). Liposomes as nanomedical devices. International Journal of Nanomedicine, 10, 975-999.
- Barenholz, Y. (2012). Doxil®—the first FDA-approved nano-drug: Lessons learned. Journal of Controlled Release, 160(2), 117-134.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




