Submitted:
14 March 2025
Posted:
17 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
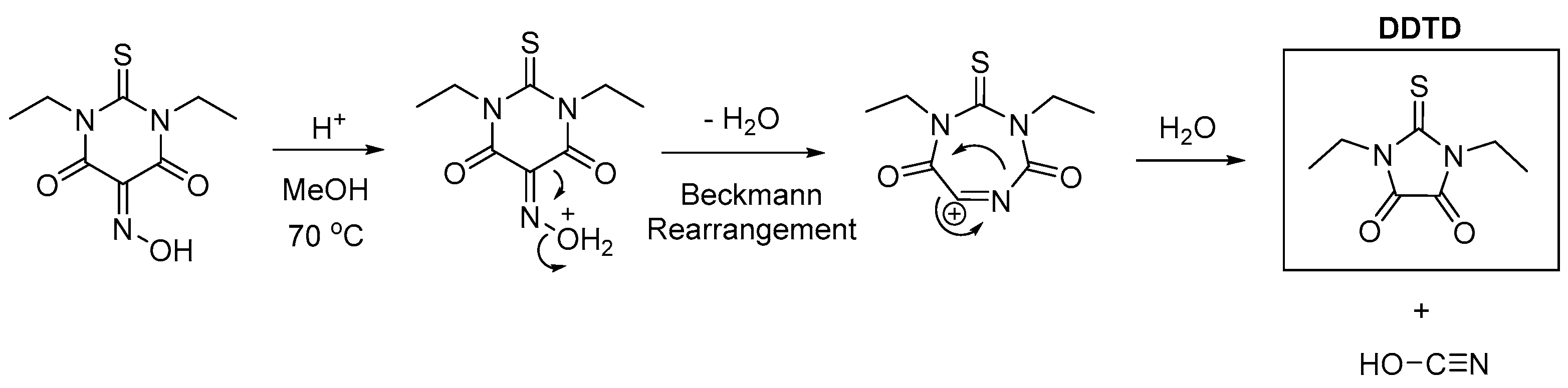
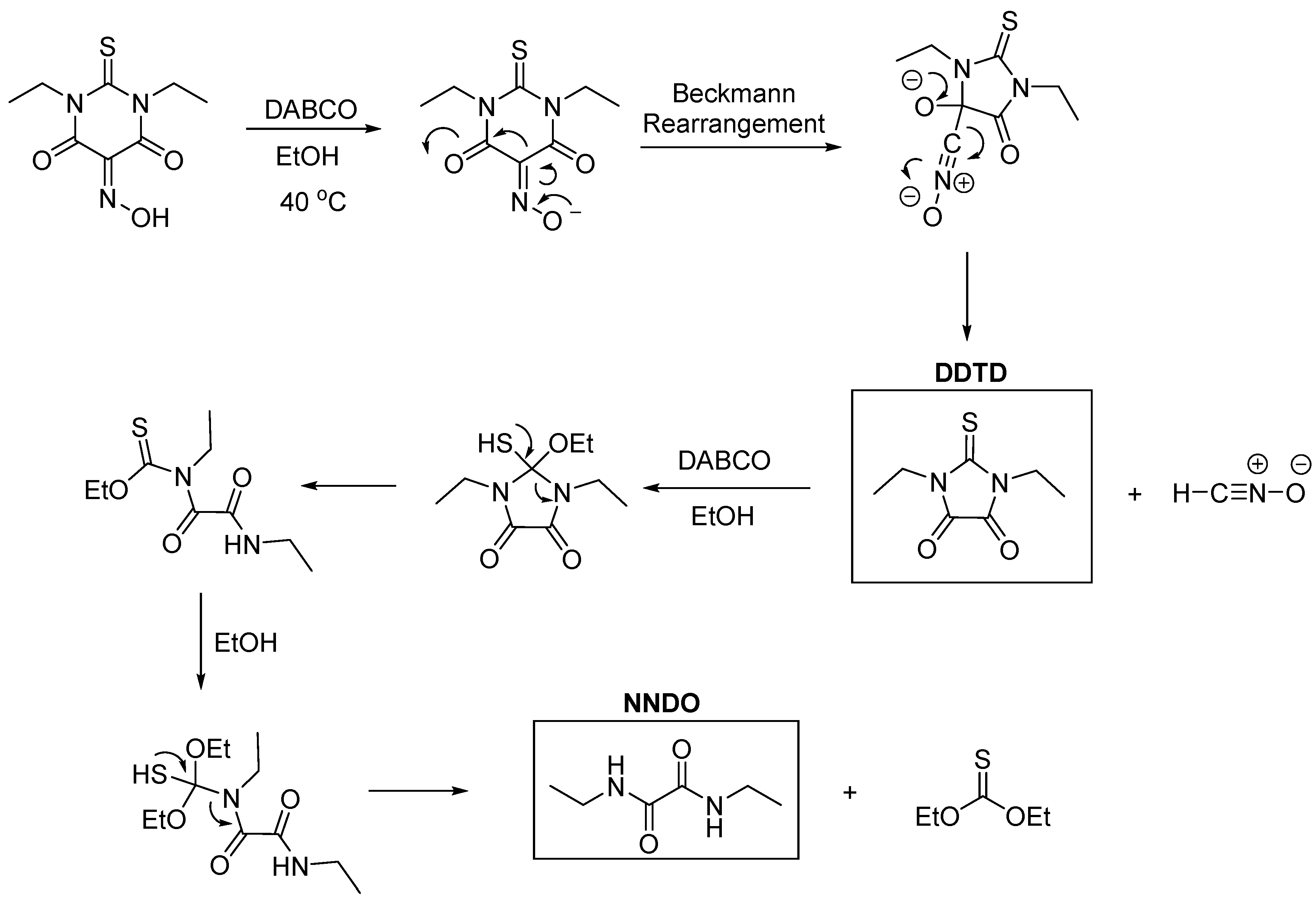
2.1. Synthesis and Crystallization
2.2. Crystallography Data
2.3. Theoretical Methods
3. Results and Discussion
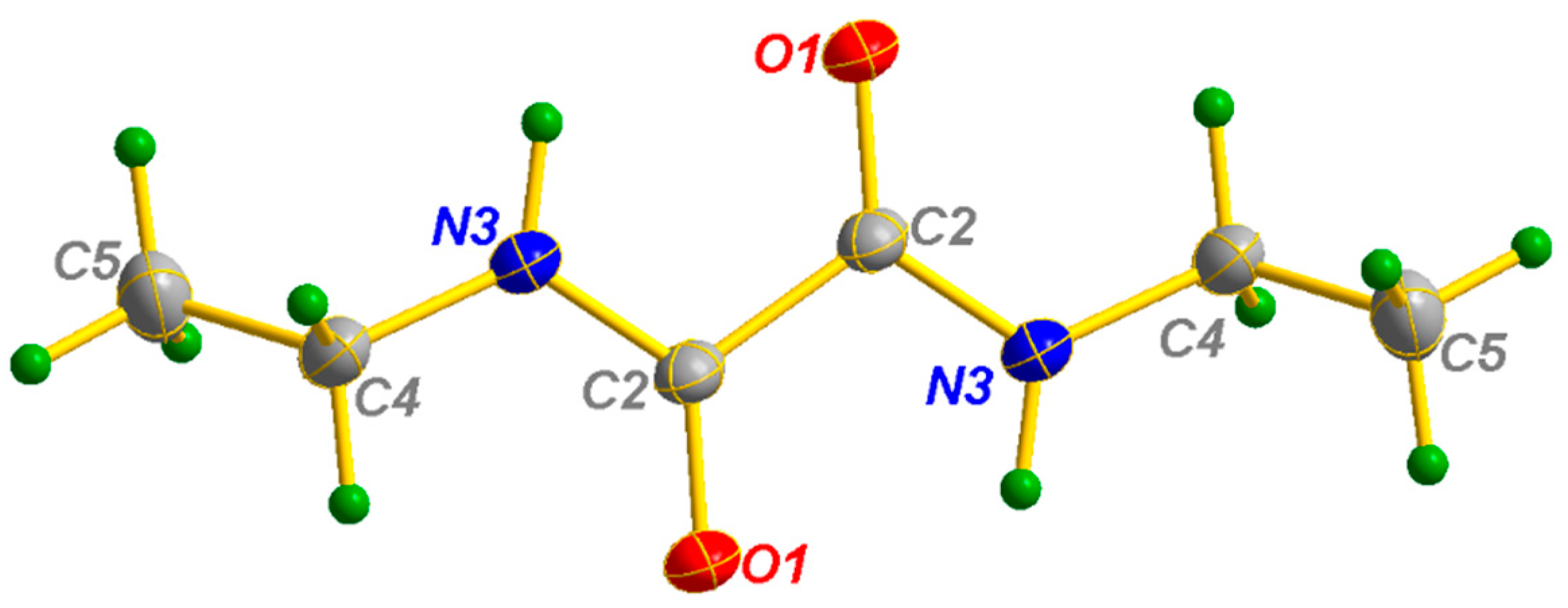
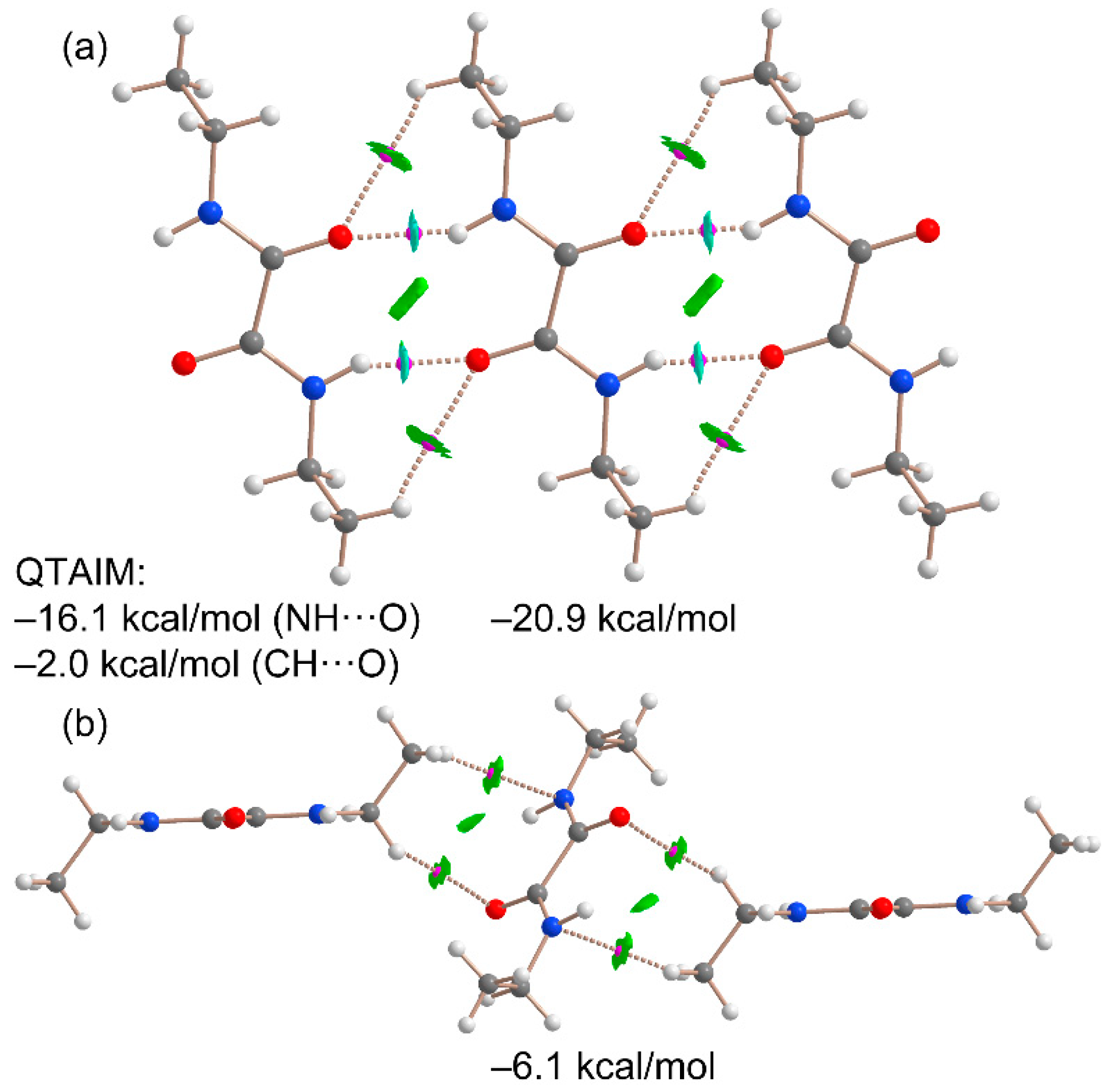
3.1. Structural Description
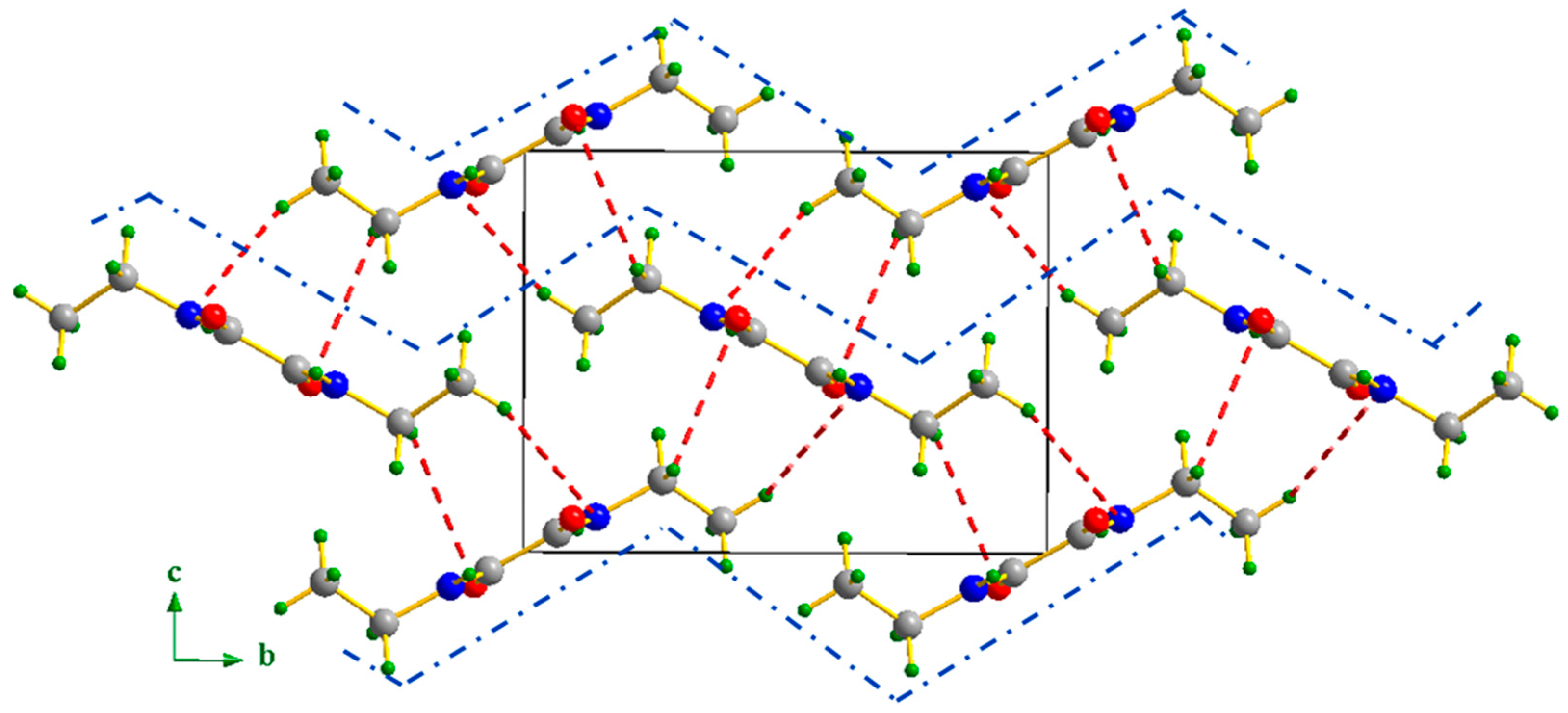
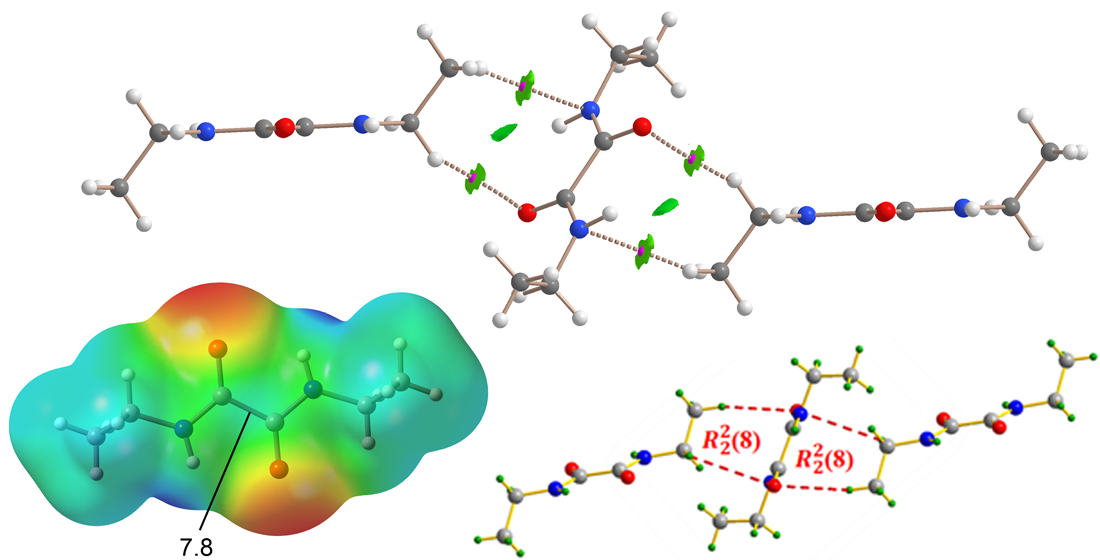
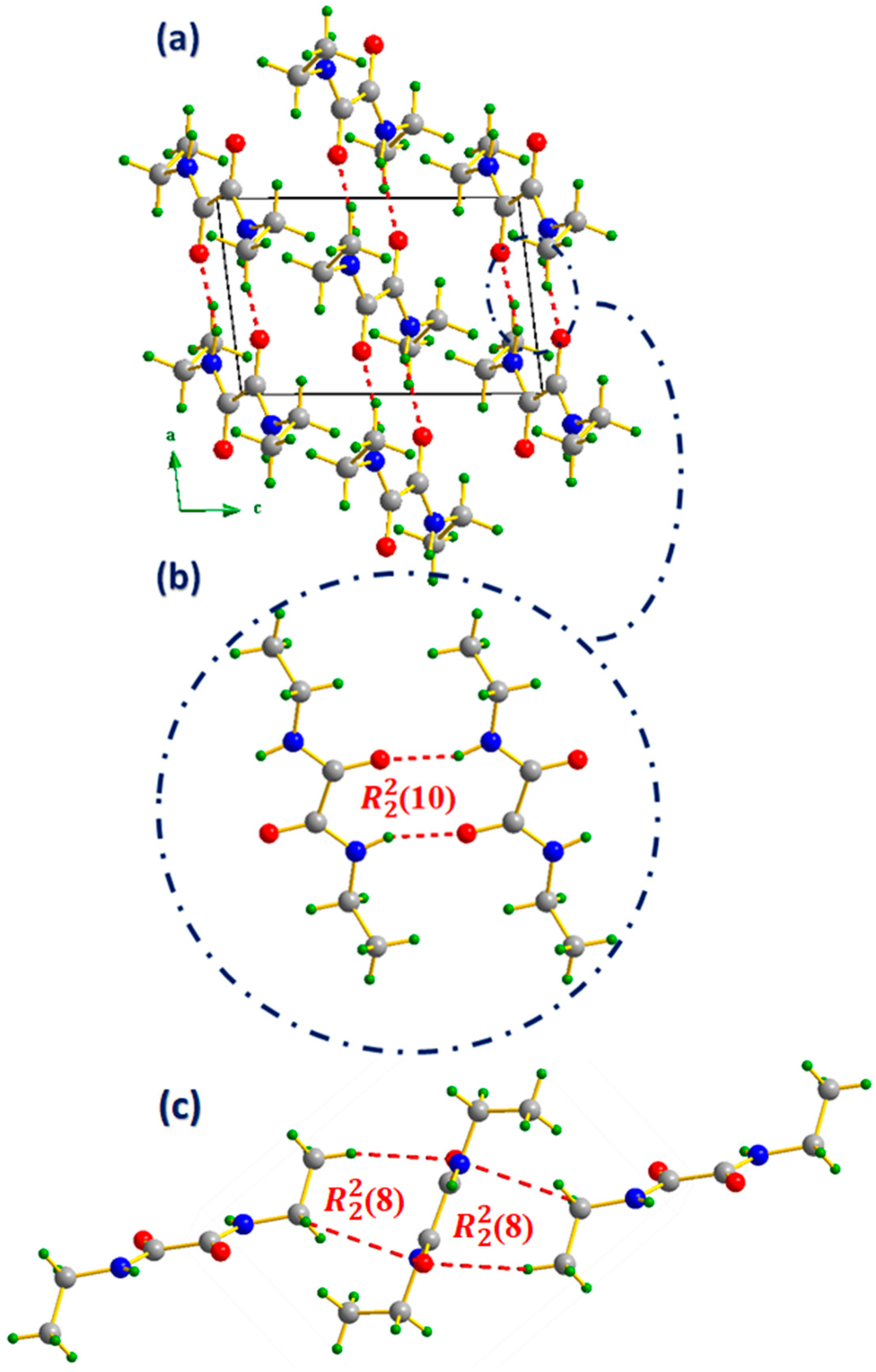
3.2. Supramolecular Features
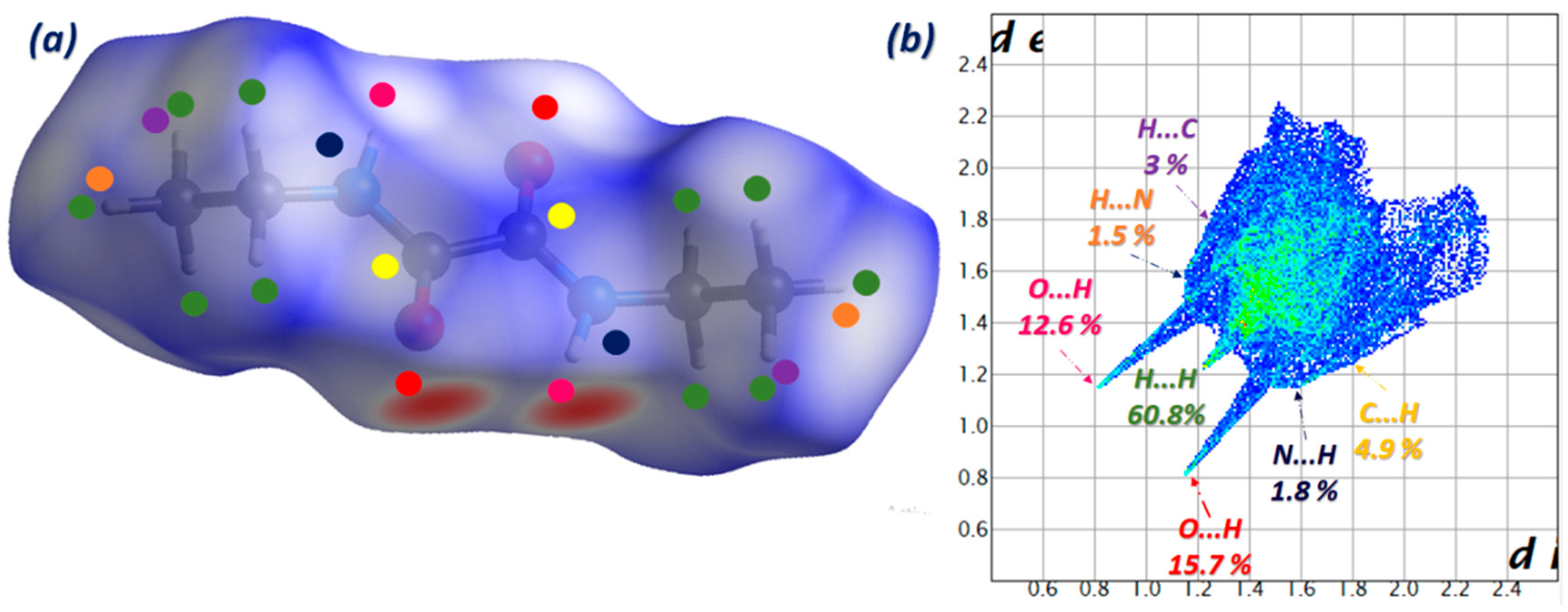
3.3. Hirshfeld Surface Analysis
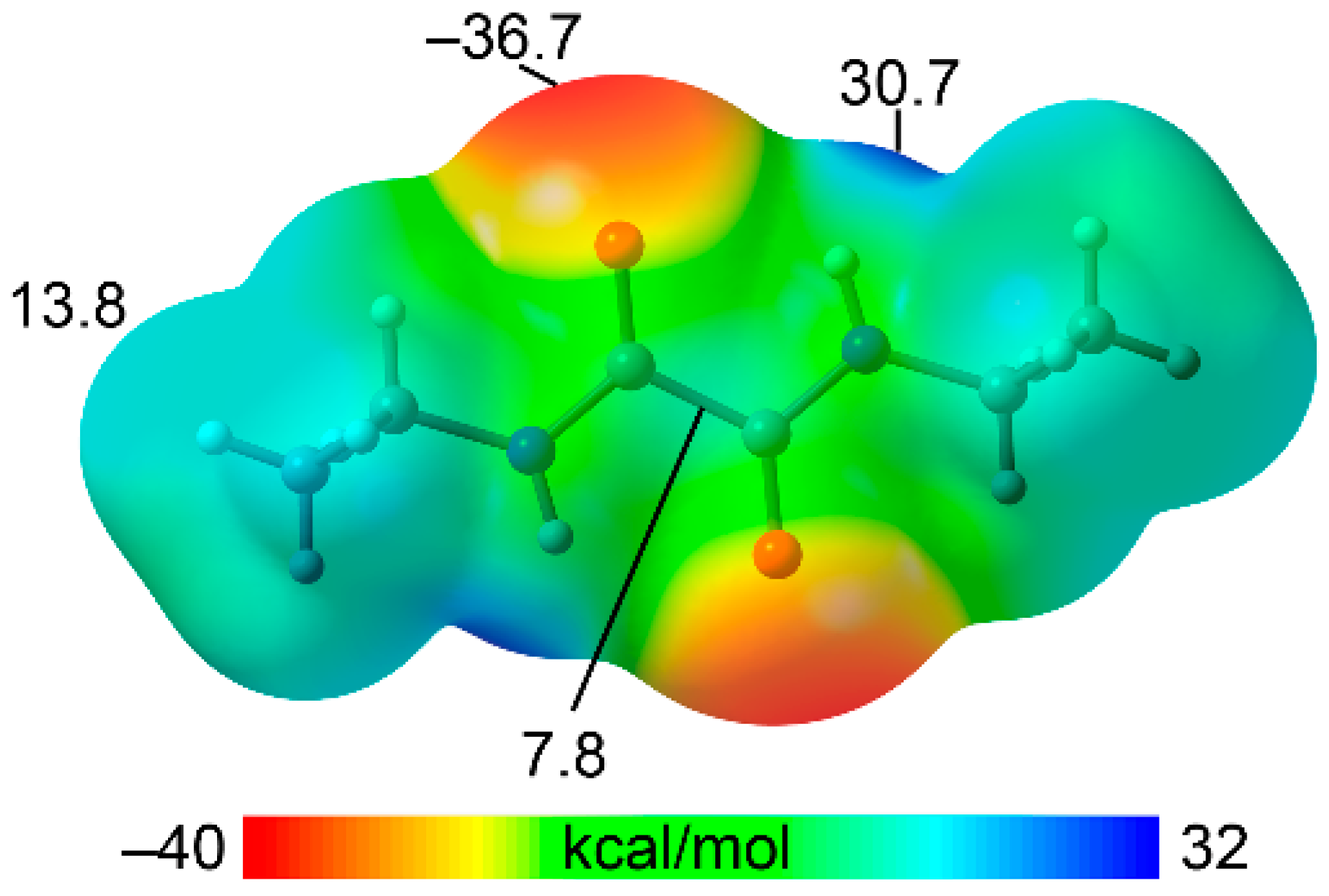
3.4. DFT Calculations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kolb, H.C.; Finn, M. G.; Sharpless, K. B. Click Chemistry: Diverse Chemical Function from a Few Good Reactions. Angew. Chem. Int. Ed., 2001, 40, 2004-2024.
- Paul, G.; Bisio, C.; Braschi, I.; Cossi, M.; Gatti, G.; Gianotti, E.; Marchese, L. Combined solid-state NMR, FT-IR and computational studies on layered and porous materials. Chem. Soc. Rev., 2018, 47, 5684–5739. [Google Scholar] [PubMed]
- Heller, S.T; Duncan, A.P.; Moy, C.L.; Kirk, S.R. The Value of Failure: A Student-Driven Course-Based Research Experience in an Undergraduate Organic Chemistry Lab Inspired by an Unexpected Result. J. Chem. Educ., 2020, 97, 3609–3616. [Google Scholar]
- Carver, K.M.; Snyder, R.C. Unexpected Polymorphism and Unique Particle Morphologies from Monodisperse Droplet Evaporation. Ind. Eng. Chem. Res., 2012, 51, 15720–15728. [Google Scholar]
- Abdullah, S.; Ganguly, S. An Overview of Imidazole and Its Analogues As Potent Anticancer Agents, Future Med. Chem., 2023, 15, 1621–1646. [Google Scholar]
- Chaudhury, D.; Banerjee, J.; Sharma, N.; Shrestha, N. Routes of synthesis and biological significances of Imidazole derivatives: Review. World J. Pharm. Pharm. Sci., 2015, 3(8), 1471-1746.
- Casas, M.T.; Armelin, E.; Alemán, C.; Puiggalí, J. On the Crystalline Structure of Even Polyoxalamides, Macromolecules. , 2002, 35, 8781–8787. [Google Scholar]
- Curtis, S.M.; Le, N.; Fowler, F.W.; Lauher, J.W. A Rational Approach to the Preparation of Polydipyridyldiacetylenes: An Exercise in Crystal Design. Cryst. Growth. Des., 2005, 5, 2313–2321. [Google Scholar]
- Hoffmann, M.; Rychlewska, U.; Warżajtis, B. The role of multiple parallel and antiparallel local dipoles for molecular structure and intermolecular interactions of oxalamides. CrystEngComm., 2005, 7, 260–265. [Google Scholar]
- Molina-Paredes, A.A.; Lara-Cerón, J.A.; Ibarra-Rodríguez, M.; Angel-Mosqueda, C.D.; Rasika Dias, H.V.; Jiménez-Pérez, V.M.; Muñoz-Flores, B.M. ; Supramolecular interactions in X-ray structures of oxalamides: Green synthesis and characterization. J. Mol. Struct., 2022, 1263, 133144. [Google Scholar]
- Dhanishta, P.; Mishra, S.K.; Suryaprakash, N. Intramolecular HB Interactions Evidenced in Dibenzoyl Oxalamide Derivatives: NMR, QTAIM, and NCI Studies, J. Phys. Chem. A., 2018, 122 199–208.
- Alemán, C.; Casanovas, J. Analysis of the oxalamide functionality as hydrogen bonding former: geometry, energetics, cooperative effects, NMR chemical characterization and implications in molecular engineering, J. Mol. Struct. THEOCHEM., 2004, 675, 9–17. [Google Scholar]
- Coe, S.; Kane, J.J.; Nguyen, T.L.; Toledo, L.M.; Wininger, E.; Fowler, F.W.; Lauher, J.W. Molecular Symmetry and the Design of Molecular Solids: The Oxalamide Functionality as a Persistent Hydrogen Bonding Unit, J. Am. Chem. Soc., 1997, 119, 86–93. [Google Scholar]
- Podda, E.; Dodd, E.; Arca, M.; Aragoni, M.C.; Lippolis, V.; Coles, S.J.; Pintus, A. N,N′-Dipropyloxamide, Molbank., 2024, M1753.
- Jad, Y.E.; Torre, B.G. Govender, T.; Kruger, H.G.; El-Faham, A.; Albericio, F. Oxyma-T, expanding the arsenal of coupling reagents, Tetrahedron Lett., 2016, 57, 3523-3525. 16.
- Kelley,S.P.; Smetana, V.; Nuss, J.S.; Dixon, D.A.; Vasiliu, M.; Mudring, A.; Rogers, R.D. Dehydration of UO2Cl2·3H2O and Nd(NO3)3·6H2O with a Soft Donor Ligand and Comparison of Their Interactions through X-ray Diffraction and Theoretical In-vestigation, Inorg. Chem., 2020, 59, 2861–2869.
- Heldt, W.Z. Beckmann Rearrangement. I. Syntheses of Oxime p-Toluenesulfonates and Beckmann Rearrangement in Acetic Acid, Methyl Alcohol and Chloroform, J. Am. Chem. Soc., 1958, 80, 5880–5885.
- Orlandin, A.; Guryanov, I.; Ferrazzano, L.; Biondi, B.; Biscaglia, F.; Storti, C.; Rancan, M.; Formaggio, F.; Ricci, A.; Cabri, W. Carbodiimide-Mediated Beckmann Rearrangement of Oxyma-B as a Side Reaction in Peptide Synthesis. Molecules, 2022, 27, 4235. [Google Scholar]
- Bruker 2023, Bruker AXS Inc., Madison, Wisconsin, USA.
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program, J. Appl. Crystallogr., 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta crystallogr., C Struct. Chem., 2015, 71 3-8.
- Spek, A.L. Single-crystal structure validation with the program PLATON, J. Appl. Cryst., 2003, 36, 7–11. [Google Scholar]
- Groom, C.R.; Bruno, I.J.; Lightfoota, M.P.; Ward, S.C. The Cambridge Structural Database, Acta Crystallogr. B Struct. Sci. Cryst. Eng. Mater., 2016, 72, 171–179. [Google Scholar]
- Desseyn, H.O.; Perlepes, S.P.; Clou, K.; Blaton, N.; Van der Veken, B.J.; Dommisse, R.; Hansen, P. E. Theoretical, Structural, Vibrational, NMR, and Thermal Evidence of the Inter- versus Intramolecular Hydrogen Bonding in Oxamides and Thiooxamide, J. Phys. Chem. A., 2004, 108, 5175–5182. [Google Scholar]
- Gaussian 16, Revision C.01, Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Petersson, G. A.; Nakatsuji, H.; Li, X.; Caricato, M.; Marenich, A. V.; Bloino, J.; Janesko, B. G.; Gomperts, R.; Mennucci, B.; Hratchian, H. P.; Ortiz, J. V.; Izmaylov, A. F.; Sonnenberg, J. L.; Williams-Young, D.; Ding, F.; Lipparini, F.; Egidi, F.; Goings, J.; Peng, B.; Petrone, A.; Henderson, T.; Ranasinghe, D.; Zakrzewski, V. G.; Gao, J.; Rega, N.; Zheng, G.; Liang, W.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Throssell, K.; Montgomery, J. A., Jr.; Peralta, J. E.; Ogliaro, F.; Bearpark, M. J.; Heyd, J. J.; Brothers, E. N.; Kudin, K. N.; Staroverov, V. N.; Keith, T. A.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A. P.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Millam, J. M.; Klene, M.; Adamo, C.; Cammi, R.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Farkas, O.; Foresman, J. B.; Fox, D. J. Gaussian, Inc., Wallingford CT, 2016.
- Adamo, C.; Barone, V. Toward Reliable Density Functional Methods Without Adjustable Parameters: The PBE0 Model. J. Chem. Phys., 1999, 110 (13), 6158–6170.
- Grimme, S.; Antony, J.; Ehrlich, S. Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132, 154104.
- Weigend. F. Accurate Coulomb-fitting basis sets for H to Rn, Phys. Chem. Chem. Phys., 2006, 8, 1057-1065.
- Boys, S. F.; Bernardi. F. The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors, Mol. Phys., 1970, 19, 553-566.
- Bader, R. F. W. A Bond Path: A Universal Indicator of Bonded Interactions, J. Phys. Chem. A, 1998, 102, 7314–7323. [Google Scholar]
- Keith, T. A. AIMAll (Version 13.05.06), TK Gristmill Software, Overland Park, KS, 2013.
- Contreras-García, J.; Johnson, E. R.; Keinan, S.; Chaudret, R.; Piquemal, J.P; Beratan, D. N.; Yang, W. NCIPLOT: A Program for Plotting Noncovalent Interaction Regions, J. Chem. Theory Comput., 2011, 7, 625–632. [Google Scholar]
- Johnson, E. R.; Keinan, S.; Mori-Sánchez, P.; Contreras-García, J.; Cohen A., J.; Yang, W. Revealing noncovalent interactions, J. Am. Chem. Soc., 2010, 132, 6498–6506. [Google Scholar]
- Bernstein, J. Polymorphism of L-glutamic acid: decoding the α-β phase relationship via graph-set analysis, Acta Cryst, B, 1991, 74, 1004–1010.
- Spackman, M.A.; Jayatilaka, D. Hirshfeld surface analysis, CrstEngComm, 2009, 11, 19-32.
- Spackman, M.A.; Mckinnon, J.J. Fingerprinting intermolecular interactions in molecular crystals, CrstEngComm, 2002, 4, 378–392.
| Crystal data | |
| Temperature | 100 K |
| Empirical formula | C6H12N2O2 |
| Formula weight (g mol-1) | 144.18 |
| Crystal size (mm) | 0.34 × 0.11 × 0.06 |
| Crystal system | monoclinic |
| Space group | P21/n |
| a (Å) | 5.0669(5) |
| b (Å) | 10.0285(10) |
| c (Å) | 7.7373(8) |
| 96.915(5) | |
| Z | 2 |
| V(Å3) | 390.30(7) |
| F (000) | 156.0 |
| Cu Kα (mm-1) | µ= 1.54178 |
| Reflections collected | 4207 |
| Independent reflections | 705 |
| Reflections with I > 2σ(I) | 660 |
| R int | 0.0489 |
| Absorption correction: | multi-scan Tmin= 0.480 , Tmax= 0.753 |
| Refined parameters | 51 |
| R[F2 ˃ 2s(F2)] | 0.0477 |
| wR(F2) | 0.1334 |
| Goodness-of-fit on F2 CCDC |
1.071 2420343 |
| D—H…A | D—H (Å) | H…A (Å) | D…A (Å) | D—H…A (°) |
|---|---|---|---|---|
| N3—H3···O1ii | 2.8752 (17) | 2.8752 (17) | 2.8752 (17) | 2.8752 (17) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




