Submitted:
14 March 2025
Posted:
14 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Experimental
2.1. Materials
2.2. Synthesis of INPCEs
2.2.1. Orthogonal Test
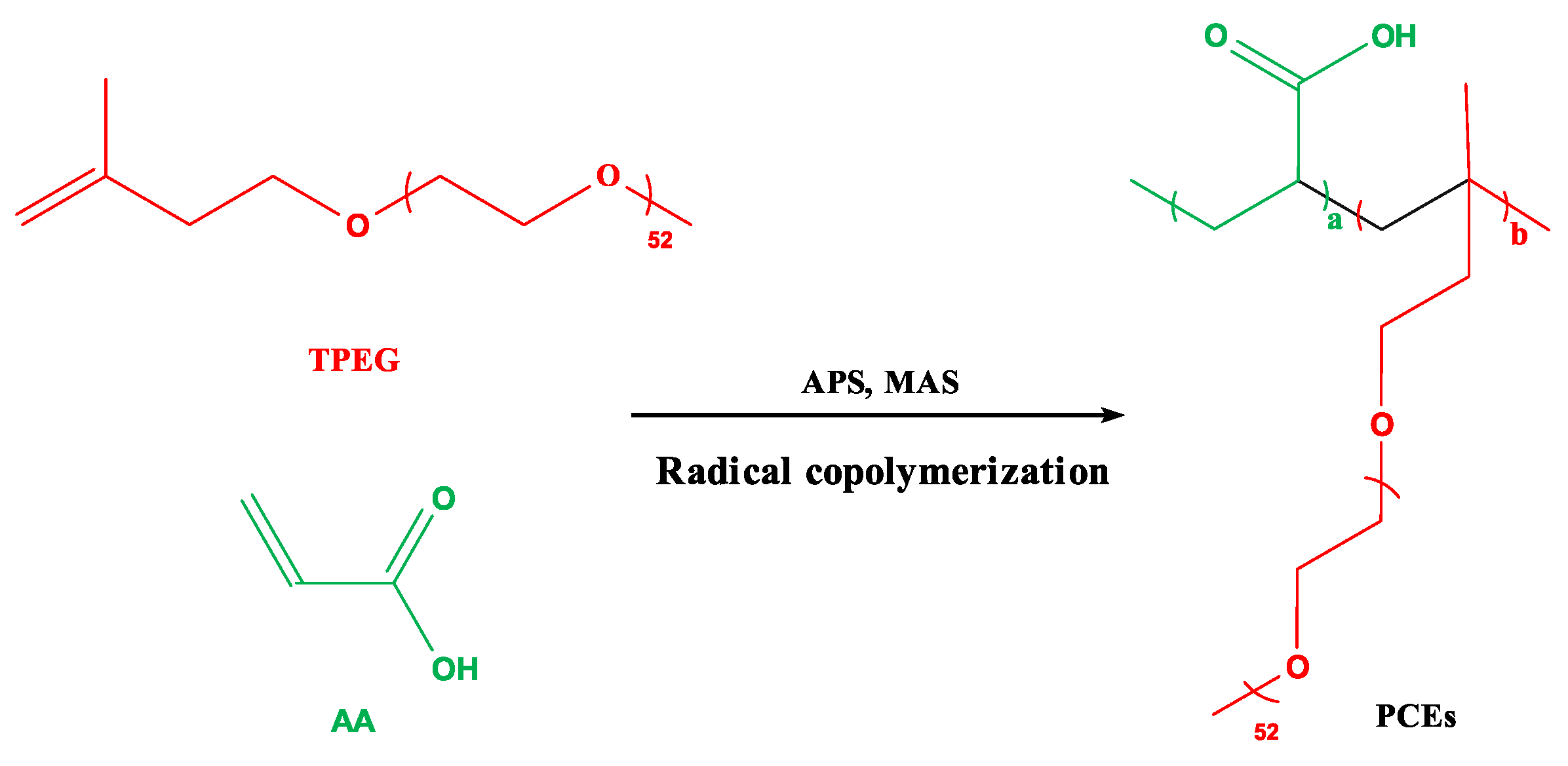
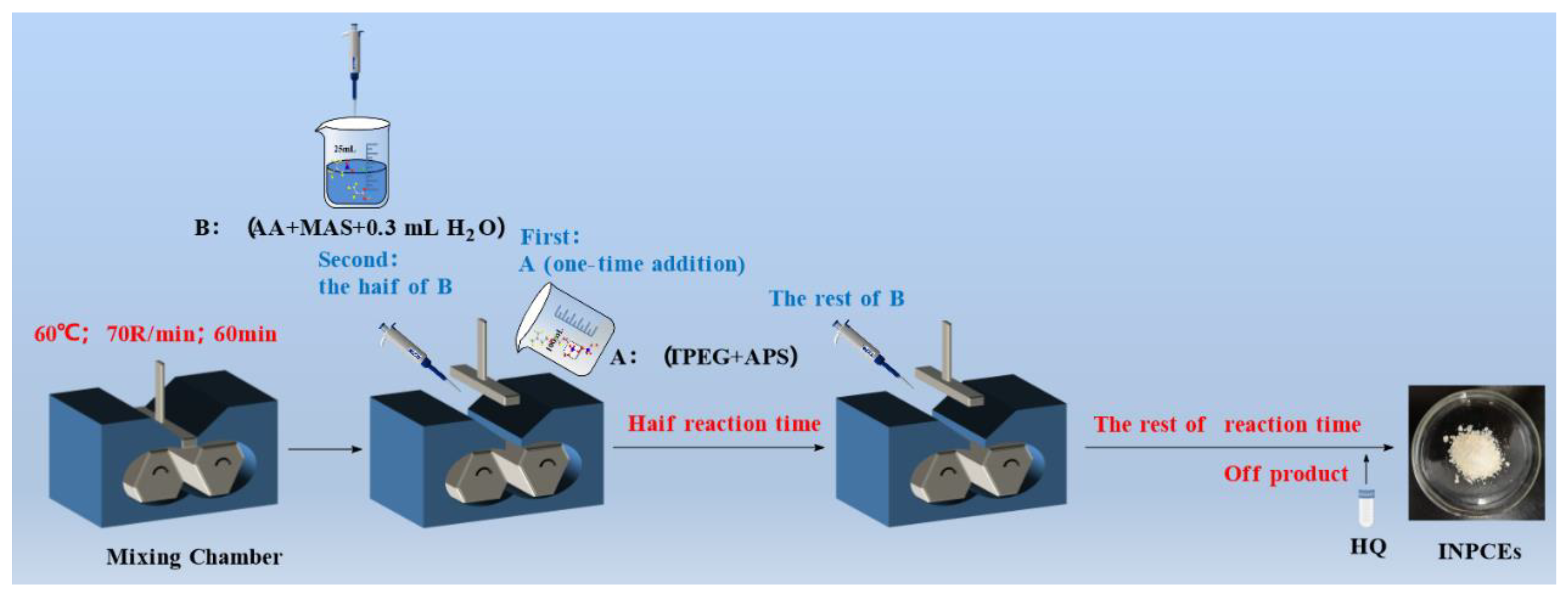
2.2.2. Synthesis of INPCEs
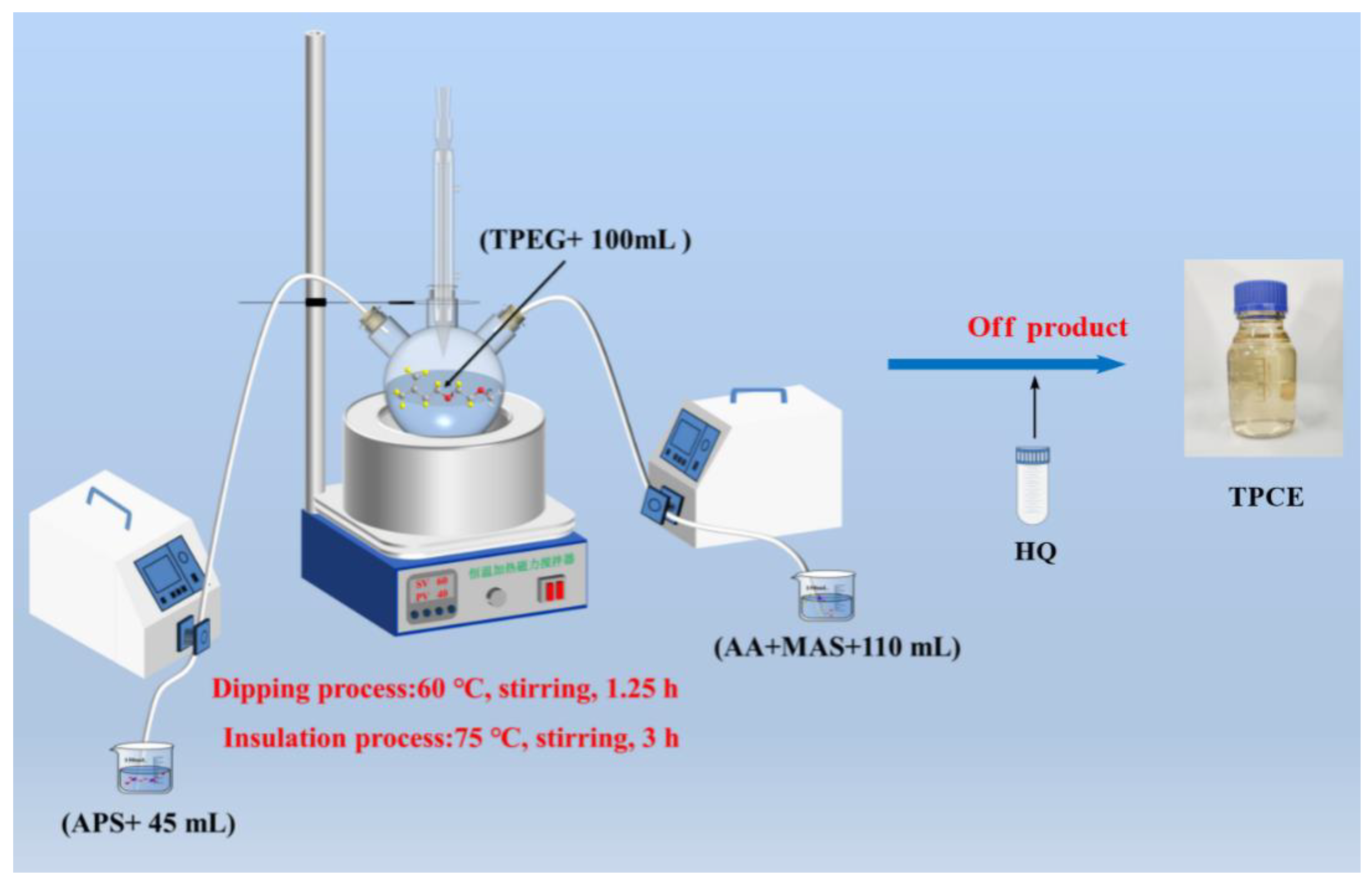
2.3. Synthesis of TPCE
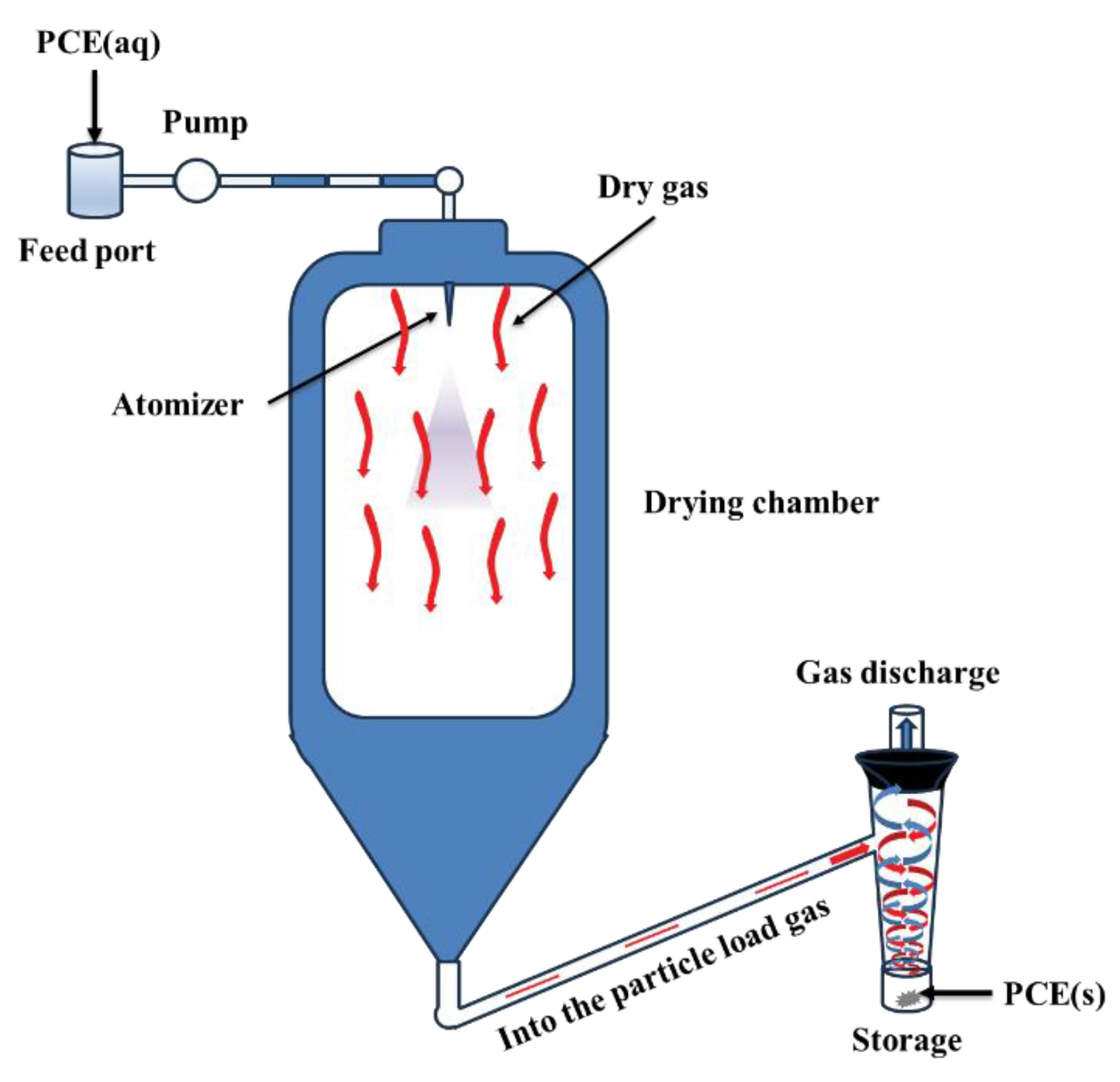
2.4. Purification of PCEs
2.5. Characterization of PCEs
2.5.1. FT-IR
2.5.2. GPC
2.6. Performance Testing of PCEs
2.6.1. Concentration
2.6.2. Initial Fluidity
2.6.3. Fluidity Loss
2.7. Energy Consumption
3. Results and Discussion
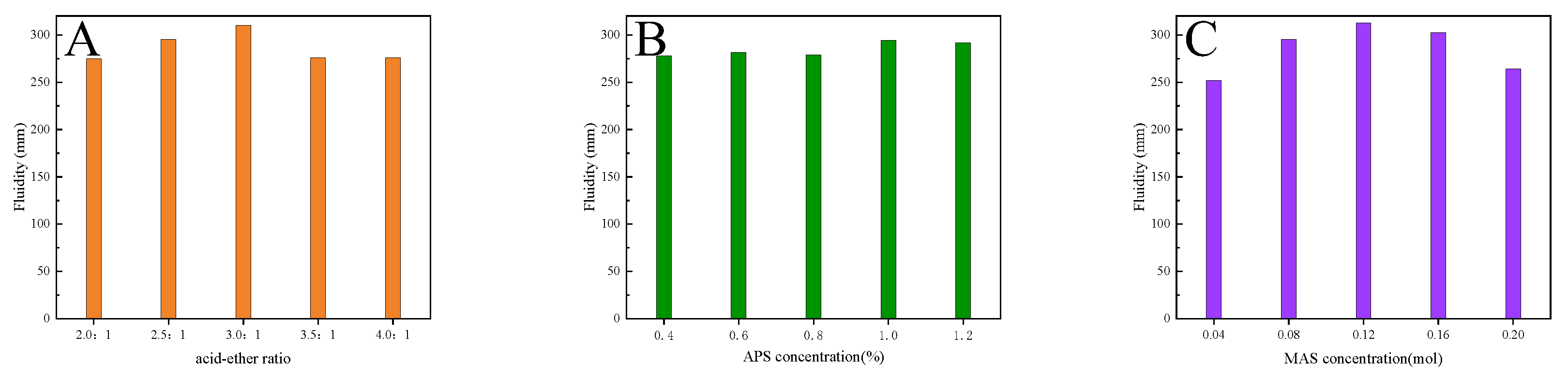
3.1. Orthogonal Test
3.2. Structural Characterization
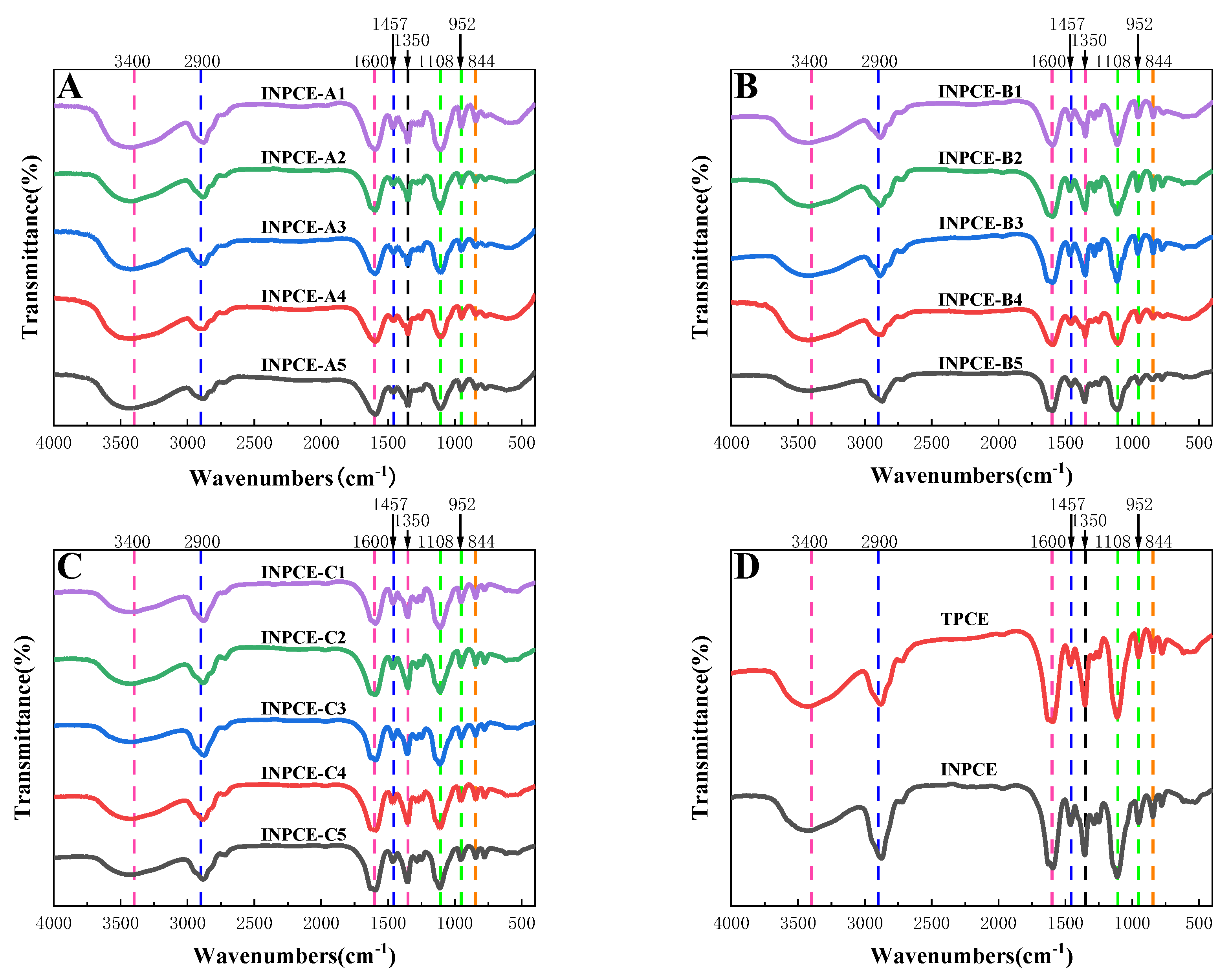
3.2.1. FT-IR
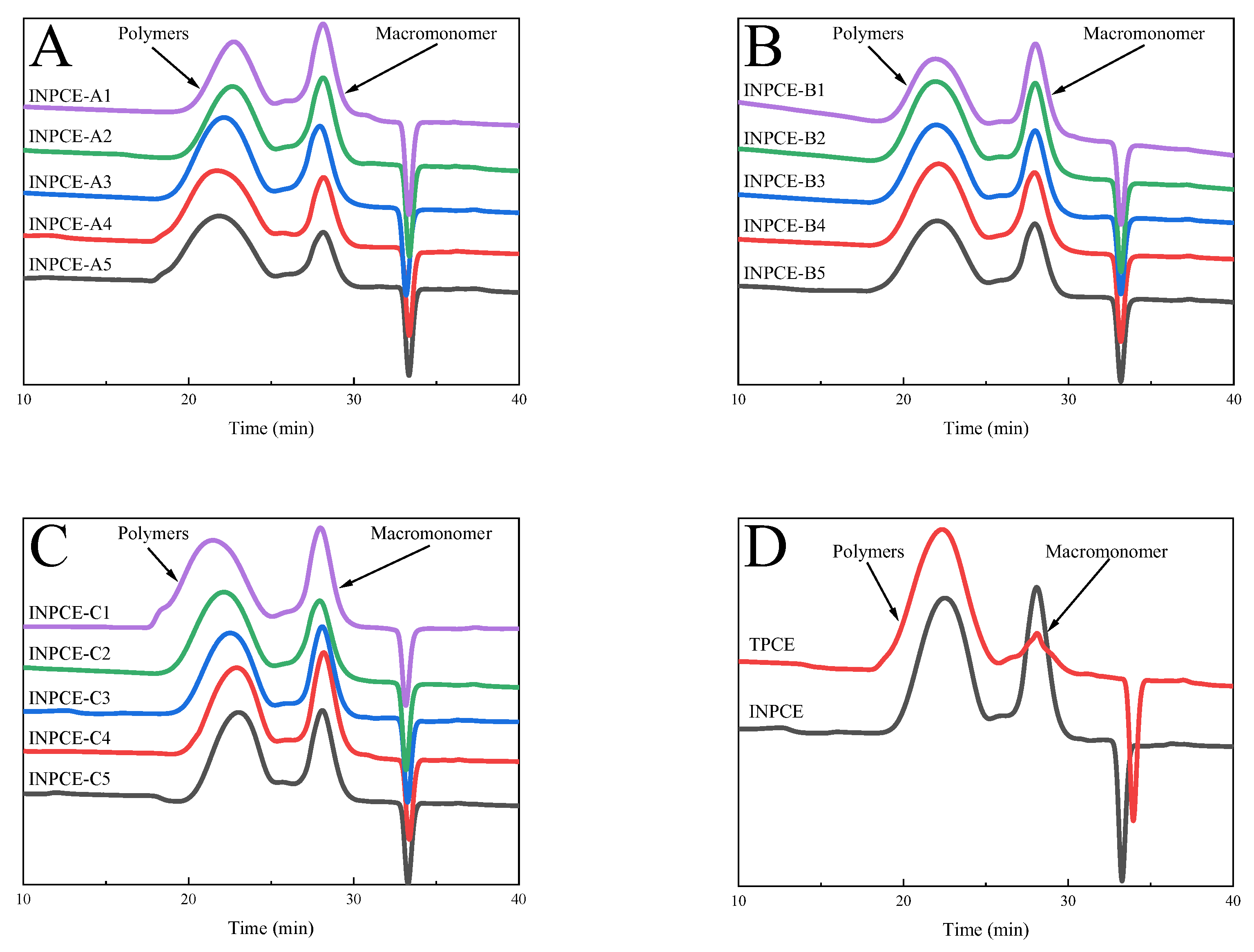
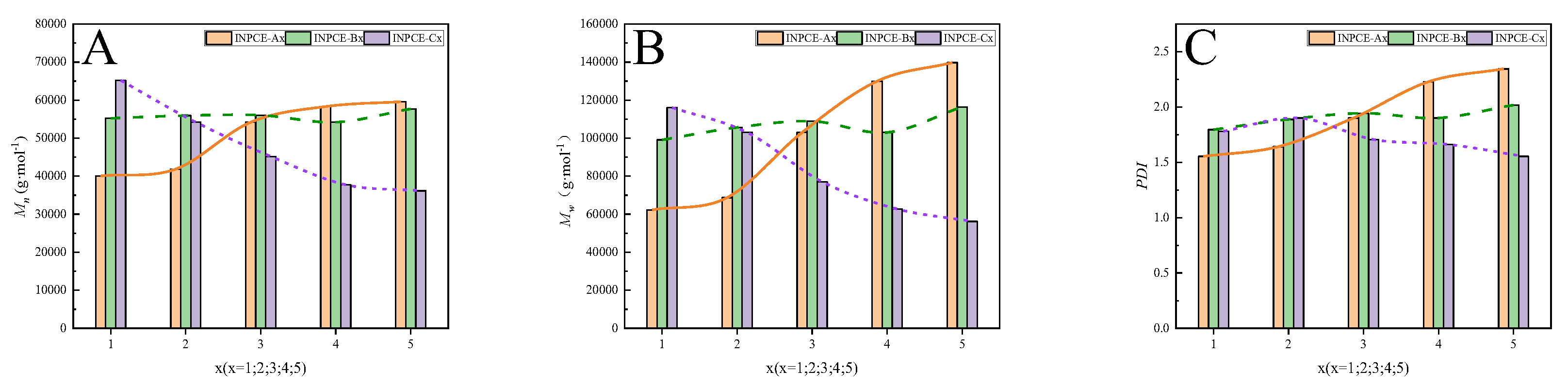
3.2.2. GPC
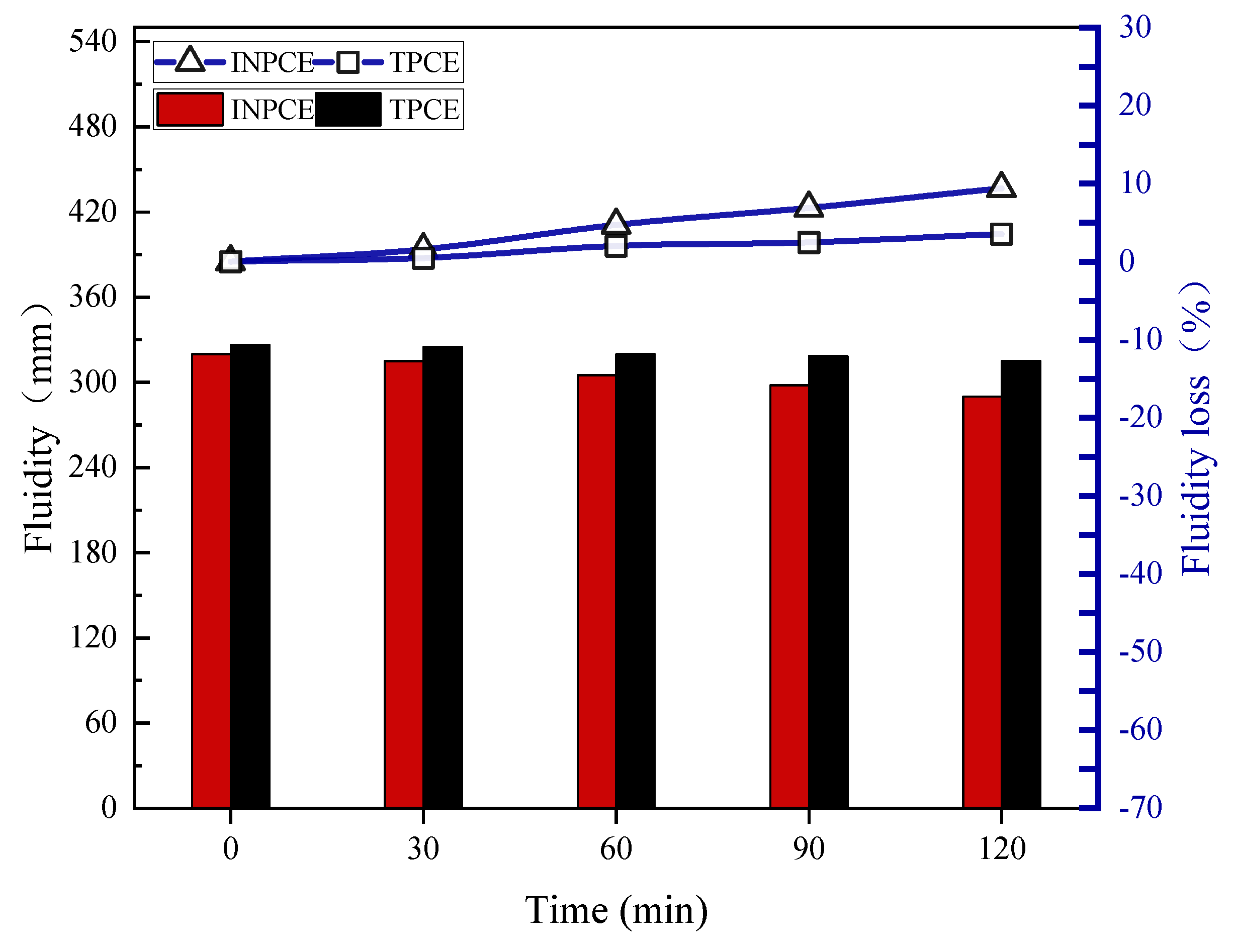
3.3. Fluidity of Cement Paste with INPCEs
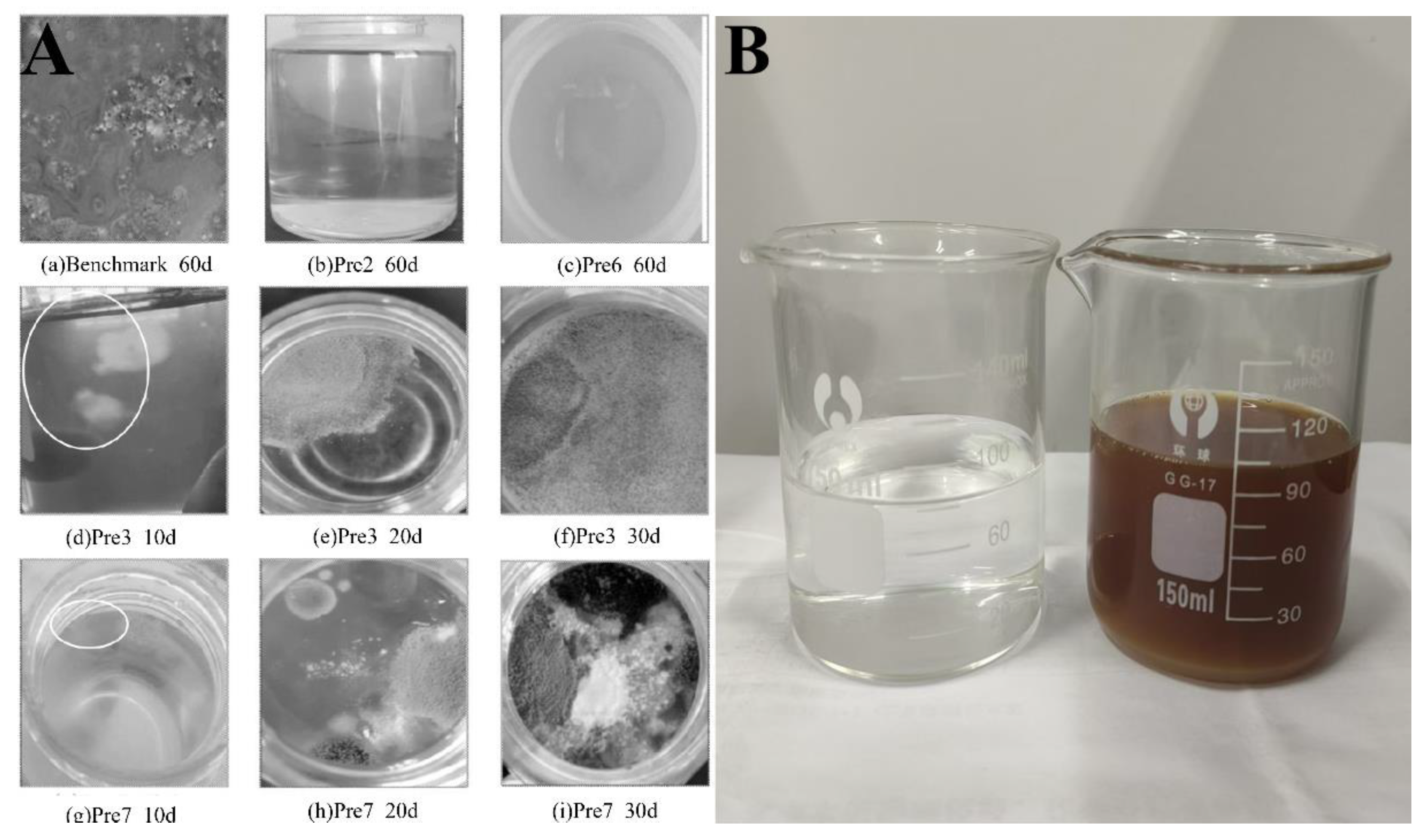
3.4. Effect of Different Synthesis Methods on the Performance of PCEs
3.4.1. Concentration
3.4.2. Fluidity Loss
3.4.3. Energy Consumption
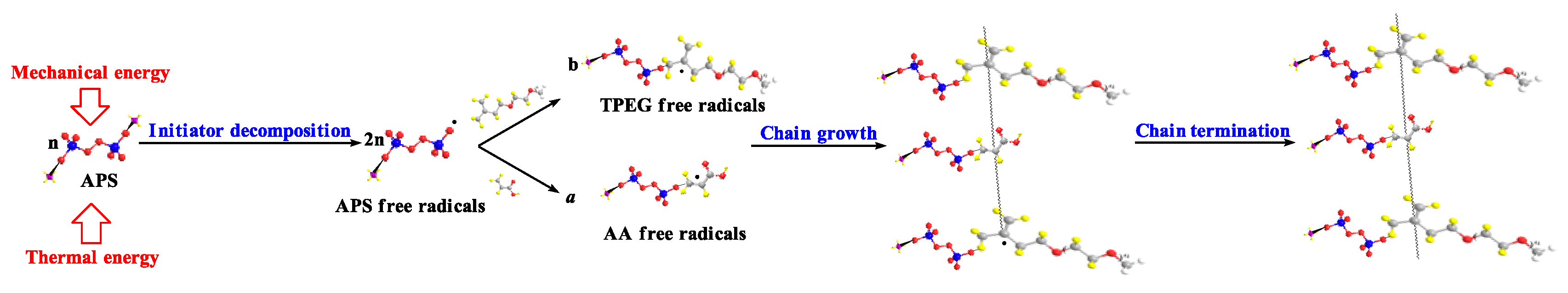
3.5. Reaction Mechanism and Prospects
4. Conclusions
- Varying the acid-ether ratio as well as the concentrations of MAS and APS affects the molecular weight and PDI of INPCEs. These properties are also dependent on the polymerization method used. However, the functional groups are the same regardless of the polymerization method.
- The optimal process parameters were found to be a reaction temperature of 60 ℃, reaction rotating speed of 70 R/min, reaction time of 60 min, nAA : nTPEG : nMAS = 3.5 : 1 : 0.12, and an APS concentration of 1 wt% relative to TPEG.
- The concentrations of all INPCEs were > 99.00 wt%, which is much higher than that of TPCE. Further, INPCE showed similar excellent dispersion and dispersion retention properties as TPCE while requiring less energy to synthesize in the laboratory.
Acknowledgments
References
- Chang, Q, Hu, M, Cheng, Y, Zeng, M, Liu, M, Pang, J, Xing, Y, Guo, J, Preparation of amphoteric polycarboxylate superplasticizer at low temperature and its application in cement-calcined kaolin blended system. J Clean Prod 2024, 435, 140542.
- Chuansheng, W, Xinxin, X, Minghui, X, Luyin, W, Huiguang, B, Zhifei, W, Influence of final-stage mixing temperature of mixer on properties of silica-filled compounds. China Rubber Industry 2023, 70(3), 220–224.
- Feng, H, Feng, Z, Mao, Y, Deng, Z, Zheng, B, Study on the polymerization process and monomer reactivity of epeg-type polycarboxylate superplasticizer. J Appl Polym Sci 2022, 139(30), 1–12.
- Feng, H, Feng, Z, Wang, W, Deng, Z, Zheng, B, Impact of polycarboxylate superplasticizers (pces) with novel molecular structures on fluidity, rheological behavior and adsorption properties of cement mortar. Constr Build Mater 2021, 292, 123285. [CrossRef]
- Hai, W, Xiangyin, M, Jun, L, Study on preparation and properties of compound functional polycarboxylic acid superplasticizer. Applied Chemical Industry 2024, 53(3), 511–515.
- Huizhou, S, Hui, W, Preparation of high slump-preserving polycarboxylate superplasticizers of its influence on concrete properties. Journal of Functional Materials 2022, 53(8), 8196–8201.
- Javadi, A, Jamil, T, Abouzari-Lotf, E, Soucek, MD, Heinz, H, Working mechanisms and design principles of comb-like polycarboxylate ether superplasticizers in cement hydration: quantitative insights for a series of well-defined copolymers. Acs Sustain Chem Eng 2021, 9(25), 8354–8371. [CrossRef]
- Jiankang, W, Aiying, S, Shijun, L, Xiaoming, C, Jing, L, Properties of solid polycarboxylate superplasticizer prepared by precipitation method. Bulletin of the Chinese Ceramic Society 2018, 37(6), 1856–1860,1867.
- Lai, H, Chen, H, Lin, Z, Study on mildew of polycarboxylic acid water reducing agent. Journal of Physics. Conference Series 2022, 2174(1), 12046.
- Li, X, Zheng, GJ, Bi, Y, Fu, CF, Yuan, MH, Wang, SF, Synthesis and characterization on a kind of polycarboxylate superplasticizer. Advanced Materials Research 2014, 989-994, 228–232.
- Lin, Z, Study on the influence of mildew on the structure and composition of polycarboxylic acid superplasticizer. Journal of Physics: Conference Series 2023, 2539(1), 012008.
- Lin, Z, Zhang, X, Chen, Z, Xiao, Y, Fang, Y, Study on adsorption, rheology and hydration behaviours of polycarboxylate(pce) superplasticizer synthesized by different acid to ether ratio. Journal of Physics. Conference Series 2021, 2133(1), 12007.
- Liu, X, Wang, Z, Zhu, J, Zheng, Y, Cui, S, Lan, M, Li, H, Synthesis, characterization and performance of a polycarboxylate superplasticizer with amide structure. Colloids and Surfaces. A, Physicochemical and Engineering Aspects 2014, 448, 119–129. [CrossRef]
- Lucas, E, Martín, AJ, Mitchell, S, Nabera, A, Santos, LF, Pérez-Ramírez, J, Guillén-Gosálbez, G, The need to integrate mass- and energy-based metrics with life cycle impacts for sustainable chemicals manufacture. Green Chem 2024, 26(17), 9300–9309. [CrossRef]
- Lufeng, P, Ligang, S, Zaibo, Z, Qingshan, C, Research and application of new superplasticized polycarboxylate powder. New Building Materials 2021, 48(7), 151–155.
- Plank, J, Li, H, Ilg, M, Pickelmann, J, Eisenreich, W, Yao, Y, Wang, Z, A microstructural analysis of isoprenol ether-based polycarboxylates and the impact of structural motifs on the dispersing effectiveness. Cement Concrete Res 2016, 84, 20–29. [CrossRef]
- Qiaoling, L, Qi, S, Peilin, S, Lufeng, P, Qingshan, C, Peng, F, Min, W, Huidong, L, Preparation of early-strength powder polycarboxylic acid superplasticizer by bulk polymerization. New Building Materials 2019, 46, 1–4,12.
- Ruijun, G, Xiao, L, Shanshan, Q, Bao, Z, Hongxia, W, Chunyong, G, Preparation methods of solid polycarboxylate superplasticizer:a review. China Powder Science and Technology 2023, 29(4), 61–70.
- Sanjayan, DJMF, Green house gas emissions due to concrete manufacture. Concrete Manufacture 2007, 5(12), 282–288.
- Schiefer, C, Plank, J, Co2 emission of polycarboxylate superplasticizers (pces) used in concrete. J Clean Prod 2023, 427, 138785. [CrossRef]
- Sha, S, Wang, M, Shi, C, Xiao, Y, Influence of the structures of polycarboxylate superplasticizer on its performance in cement-based materials-a review. Construction & Building Materials 2020, 233, 117257.
- Sun, W, Pan, L, Jiacheng, Xu, N, Guo, Z, Enhancing the application of mechanochemistry in the synthesis of high-concentration polycarboxylate superplasticizer: is aqueous copolymerization needed? J Disper Sci Technol 2023, 44(4), 660–668. [CrossRef]
- Tammer, M, G. Sokrates: infrared and raman characteristic group frequencies: tables and charts. Colloid Polym Sci 2004, 283(2).
- Wang, C, Kong, F, Wu, J, Pan, L, Adjusting concrete resistance to corrosive ions by varying carboxyl contents in chemical additives. Structures 2024, 62, 106168. [CrossRef]
- Wei, L, Chuansheng, W, Huiguang, B, Tianhao, C, Lin, Z, Luqi, Z, Study on compound properties and energy consumption of tandem mixing and conventional mixing. China Rubber Industry 2020, 67(5), 384–387.
- Weichao, W, Bo, W, Jianli, H, Analysis of the impact of refiners on energy consumption. Rubber and Plastic Technology and Equipment 2024, 50(08), 40–42.
- Wenting, S, Lisha, P, Zhifang, G, Jiacheng, L, Nai, X, Sujuan, P, Characterization and properties of polycarboxylate superplasticizer by mechanochemistry. Concrete 2019, (12), 88–91.
- Xia, Y, Shi, W, Xiang, S, Yang, X, Yuan, M, Zhou, H, Yu, H, Zheng, T, Zhang, J, Jiang, Z, Huang, L, Synthesis and modification of polycarboxylate superplasticizers-a review. Materials 2024, 17(5), 1092.
- Xiang, S, Shi, C, Gao, Y, Fan, G, Gu, F, Progresses in synthesis of polycarboxylate superplasticizer. Adv Civ Eng 2020, 2020(2020), 1–14.
- Xiao, L, Xiabing, B, Qian, X, Chunlei, X, Guanghong, L, Jianan, G, Synthesis and application performances of solid polycarboxylate superplasticizers using different initiators. Materials Science Forum 2020, 993, 1367–1372.
- Xiaojiang, L, Research status of solid polycarboxylate superplasticizer. China Building Materials Science & Technology 2019, 28(6), 51–52,54.
- Xingrong, L, Zhiqin, D, Jinbao, W, Song, W, Hai, J, Tao, Y, Research on antiseptic of compound polycarboxylate superplasticizer. New Building Materials 2021, 48(10), 104–107,129.
- Xitong, Z. Experimental study on the preparation of solid polycarboxylate superplasticizer by composite adsorbent. 硕士, Shenyang Architectural University, 2021.
- Xu, Y, Liu, X, Jiang, M, Lai, G, Li, S, Wang, Z, Cui, S, Effect of competitive hydrolysis of diester in polycarboxylate superplasticizer on the fluidity of cement paste. Colloids and Surfaces a: Physicochemical and Engineering Aspects 2023, 671, 131691. [CrossRef]
- Yang, M, Cai, G, Zhang, J, Liu, Y, Lu, T, Meng, F, Synthesis and properties of hyperbranched solid polycarboxylate superplasticizer. Journal of Physics. Conference Series 2022, 2393(1), 12028.
- Yanmei, L, Hua Zhen, L, Yuanqiang, G, Xiuxing, M, Preparation of solid polycarboxylate superlasticizer. Iop Conference Series: Materials Science and Engineering 2019, 631(2), 022045.
- Yuanzi, Z, Kangyuan, S, Lei, D, Jianrong, S, Review on development of polycarboxylate superplasticizer. China Building Materials Science & Technology 2024, 33(3), 94–98.
- Zhang, J, Ma, Y, Wang, J, Gao, N, Hu, Z, Liu, J, Wang, K, A novel shrinkage-reducing polycarboxylate superplasticizer for cement-based materials: synthesis, performance and mechanisms. Construction & Building Materials 2022, 321, 126342.
- Zhang, L, Du, W, Wang, D, Wang, F, Fang, K, Yu, J, Sheng, B, Syntheses of polycarboxylate superplasticizers: microwave induction versus conventional thermal induction. Composites Part B: Engineering 2021, 207, 108560. [CrossRef]
- Zhenpeng, D, Jiandong, Z, Hongbo, Y, Gang, W, Yunpeng, J, Preparation of polycarboxylic acid superplasticizer with high slump retention by bulk polymerization. Shandong Chemical Industry 2022, 51(7), 52–54,57.
- Zhenpeng, S, Erfei, G, Lelin, W, Liangliang, S, Yaling, X, Mingjie, L, Xi, L, Dangqiang, L, Current status of research on synthesis of polycarboxylic acid water reducing agents by propriety polymerization method. China Concrete 2016, (12), 37–40.
- Zuren, P, Polymer chemistry 5th edition. Chemical Industry Press: Bingjing, 2013; p 266.
- Zuyue, L, Zhenping, S, Yanliang, J, Haijing, Y, Min, P, Sheng, GH, Shaofeng, W, Xuebin, G, Lihua, Z, Yuanjie, H, Xiaofu, Z, Hazards, mechanisms and influencing factors of mold and mildew in liquid admixtures in summer. Concrete 2021, (06), 80-83,92.
| Chemical composition | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Composition | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | Na2Oeq | f−CaO | Loss | Cl− |
| Cement(wt%) | 20.94 | 4.31 | 3.28 | 63.46 | 2.76 | 2.23 | 0.56 | 0.80 | 2.31 | 0.036 |
| Levels | Factors | |||
|---|---|---|---|---|
| A-Reaction temperature (°C) |
B-Reaction rotating speed (R/min) |
C-Reaction time (min) |
D-Blank column* | |
| 1 | 60 | 65 | 60 | 1 |
| 2 | 65 | 70 | 90 | 2 |
| 3 | 70 | 75 | 120 | 3 |
| Samples |
nAA (mol) |
nTPEG (mol) |
nMAS (mol) | mAPS (g) |
VH2O (mL) |
Reaction temperature T(℃) |
Reaction rotating speed (R/min) (R/min) |
Reaction time (min) |
|
|---|---|---|---|---|---|---|---|---|---|
| INPCE-A | INPCE-A1 | 0.1750 | 0.0875 | 0.0070 | 2.10 | 1.5 | 60 | 70 | 60 |
| INPCE-A2 | 0.2188 | 0.0875 | 0.0070 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-A3 | 0.2625 | 0.0875 | 0.0070 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-A4 | 0.3063 | 0.0875 | 0.0070 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-A5 | 0.3500 | 0.0875 | 0.0070 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-B | INPCE-B1 | 0.2625 | 0.0875 | 0.0070 | 0.84 | 1.5 | 60 | 70 | 60 |
| INPCE-B2 | 0.2625 | 0.0875 | 0.0070 | 1.26 | 1.5 | 60 | 70 | 60 | |
| INPCE-B3 | 0.2625 | 0.0875 | 0.0070 | 1.68 | 1.5 | 60 | 70 | 60 | |
| INPCE-B4 | 0.2625 | 0.0875 | 0.0070 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-B5 | 0.2625 | 0.0875 | 0.0070 | 2.52 | 1.5 | 60 | 70 | 60 | |
| INPCE-C | INPCE-C1 | 0.2625 | 0.0875 | 0.0035 | 2.10 | 1.5 | 60 | 70 | 60 |
| INPCE-C2 | 0.2625 | 0.0875 | 0.0070 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-C3 | 0.2625 | 0.0875 | 0.0105 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-C4 | 0.2625 | 0.0875 | 0.0140 | 2.10 | 1.5 | 60 | 70 | 60 | |
| INPCE-C5 | 0.2625 | 0.0875 | 0.0175 | 2.10 | 1.5 | 60 | 70 | 60 | |
| Samples |
nAA (mol) |
nTPEG (mol) |
nMAS (mol) |
mAPS (g) |
VH2O (mL) |
Reaction parameters |
|---|---|---|---|---|---|---|
| INPCE | 0.2625 | 0.0875 | 0.0105 | 2.10 | 1.5 | The reaction temperature was 60 ℃, the reaction rotating speed was 70 R/min, and the reaction time was 60 min. |
| TPCE | 0.2625 | 0.0875 | 0.0105 | 2.10 | 357.0 | The reaction temperature was 60−75 ℃, the stirring speed was 1600 R/min, and the reaction time was 255 min. |
| Samples | Instrument number | Instruments names | Power rating (kW) | Usage time (h) |
|
|---|---|---|---|---|---|
| INPCE | 1 | XSS-300 torque rheometer from Shanghai Kechuang Rubber & Plastic Machinery Equipment Co., Ltd with a 300mL mixing chamber. | 4.00 | 1.25 | |
| TPCE | 1 | DF-101s Collector Type Constant Temperature Heating Magnetic Stirrer from Gongyi Yuhua Instrument Manufacturing Co., Ltd. | Motor power | 0.03 | 0.33 |
| 2 | Heating power | 0.50 | 4.83 | ||
| 3 | DX-204 Low-Temperature Circulator from Beijing Changliu Scientific Instrument Co., Ltd. | 0.30 | 4.83 | ||
| 4 | BT100-1L Peristaltic Pump Drive from Baoding Langer Constant Flow Pump Co., Ltd. | 0.05 | 2.25 | ||
| 5 | OPD-8 Spray Dryer of Shanghai Dachuan Yuan Drying Equipment Co., Ltd*. | 9.80 | 0.20 | ||
| Experimental number | A (Reaction temperature) | B (Reaction rotating speed) | C (Reaction time) | D (Blank column) | Fluidity (mm) |
|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | 237.5 |
| 2 | 1 | 2 | 2 | 2 | 237.5 |
| 3 | 1 | 3 | 3 | 3 | 232.5 |
| 4 | 2 | 1 | 2 | 3 | 210.0 |
| 5 | 2 | 2 | 3 | 1 | 212.0 |
| 6 | 2 | 3 | 1 | 2 | 230.0 |
| 7 | 3 | 1 | 3 | 2 | 228.0 |
| 8 | 3 | 2 | 1 | 3 | 247.0 |
| 9 | 3 | 3 | 2 | 1 | 208.0 |
| k1 | 235.8 | 225.1 | 238.2 | 219.2 |
|
| k2 | 217.3 | 232.1 | 218.5 | 231.8 | |
| k3 | 227.7 | 223.5 | 224.2 | 229.8 | |
| R | 18.50 | 8.667 | 19.67 | 12.67 | |
| Primary and secondary order | C>A>B | ||||
| Excellent level | A1 | B2 | C1 | ||
| Excellent combination | A1 B2 C1 | ||||
| Samples | Mn (g·mol−1) | Mw (g·mol−1) | PDI |
|---|---|---|---|
| INPCE | 45179 | 77074 | 1.71 |
| TPCE | 46513 | 99322 | 2.14 |
| Samples | Mn (g·mol−1) | Mw (g·mol−1) | PDI | |
| INPCE-A | INPCE-A1 | 40074 | 62257 | 1.55 |
| INPCE-A2 | 41845 | 68699 | 1.64 | |
| INPCE-A3 | 54188 | 102996 | 1.90 | |
| INPCE-A4 | 58314 | 129939 | 2.23 | |
| INPCE-A5 | 59575 | 139677 | 2.34 | |
| INPCE-B | INPCE-B1 | 55210 | 99104 | 1.80 |
| INPCE-B2 | 55925 | 105609 | 1.89 | |
| INPCE-B3 | 56015 | 108856 | 1.94 | |
| INPCE-B4 | 54188 | 102996 | 1.90 | |
| INPCE-B5 | 57681 | 116409 | 2.02 | |
| INPCE-C | INPCE-C1 | 65174 | 116062 | 1.78 |
| INPCE-C2 | 54188 | 102996 | 1.90 | |
| INPCE-C3 | 45179 | 77074 | 1.71 | |
| INPCE-C4 | 37741 | 62701 | 1.66 | |
| INPCE-C5 | 36151 | 56187 | 1.55 | |
| Samples | INPCE-A | INPCE-B | INPCE-C | TPEG | ||||||||||||
| A1 | A2 | A3 | A4 | A5 | B1 | B2 | B3 | B4 | B5 | C1 | C2 | C3 | C4 | C5 | ||
| concentration (wt%) | 99.34 | 99.29 | 99.03 | 99.19 | 99.15 | 99.20 | 99.16 | 99.33 | 99.03 | 99.05 | 99.24 | 99.03 | 99.08 | 99.00 | 99.06 | 39.73 |
| INPCE | TPCE | |||
| Energy(kW·h) | Ea, INPCE | E0, INPCE | Ea, TPCE | E0, TPCE |
| 5.00 | ≈E0, TPCE | 5.95 | ≈E0, INPCE | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




